Abstract
The Canadian Association of Radiologists (CAR) Cancer Expert Panel is made up of physicians from the disciplines of radiology, medical oncology, surgical oncology, radiation oncology, family medicine/general practitioner oncology, a patient advisor, and an epidemiologist/guideline methodologist. The Expert Panel developed a list of 29 clinical/diagnostic scenarios, of which 16 pointed to other CAR guidelines. A rapid scoping review was undertaken to identify systematically produced referral guidelines that provide recommendations for one or more of the remaining 13 scenarios. Recommendations from 21 guidelines and contextualization criteria in the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) for guidelines framework were used to develop the recommendation for these scenarios. During recommendation formulation, one additional scenario was mapped to an existing CAR guideline scenario, leaving 12 scenarios with new recommendations. The guideline focuses on cancer diagnosis and does not cover cancer staging, follow-up, and surveillance. This guideline presents the methods of development and the referral recommendations for suspected pancreatic cancer, suspected liver cancer, incidental liver mass, incidental colon mass or suspected colon cancer, suspected anal cancer, suspected penile cancer, suspected cervical cancer, suspected endometrial/uterine cancer, suspected vulvar cancer, suspected vaginal cancer, suspected haematologic malignancies, and suspected skin cancer. The guideline also points to other CAR guidelines for suspected neck, thyroid, brain, lung, intracardiac/pericardial, esophageal/gastric, renal, adrenal, bladder, testicular, prostate and ovarian cancers, suspected soft tissue mass or tumour, suspected bone tumour, suspected bone tumour --myeloma, suspected spine tumours, and incidental lung cancer.
Résumé
Le groupe d’experts en imagerie du cancer de l’Association canadienne des radiologistes (CAR) regroupe des médecins spécialistes des disciplines de la radiologie, l’oncologie médicale, l’oncologie chirurgicale, la radio-oncologie et l’oncologie dans le cadre d’une pratique générale, ainsi qu’une représentante des patients et une épidémiologiste spécialisée en méthodologie de l’élaboration de lignes directrices. Le groupe d’experts a élaboré une liste de 29 scénarios cliniques/diagnostiques, y compris 16 scénarios qui réfèrent à d’autres lignes directrices de la CAR. Le groupe a entrepris une revue rapide des articles publiés en vue de repérer les lignes directrices relatives aux demandes d’examen élaborées de façon systématique qui fournissent des recommandations pour les 13 autres scénarios. Les recommandations de 21 lignes directrices et critères de contextualisation du cadre GRADE (notation des recommandations, analyses, développements et évaluations) concernant la structure des lignes directrices ont été utilisées pour rédiger les énoncés de recommandations couvrant ces scénarios. Au cours de la rédaction des recommandations, une correspondance a été établie entre un autre des scénarios et des lignes directrices de la CAR; ce sont donc 12 scénarios qui ont fait l’objet de nouvelles recommandations. Les lignes directrices portent sur le diagnostic du cancer, et ne couvrent pas la stadification, le suivi, ni la surveillance de celui-ci. Ces lignes directrices présentent les étapes à suivre et les recommandations d’orientation dans les cas de cancer du pancréas présumé, de cancer du foie présumé, de masse hépatique de découverte fortuite, de masse du côlon de découverte fortuite ou de cancer du côlon présumé, de cancer de l’anus présumé, de cancer pénien présumé, de cancer du col de l’utérus présumé, de cancer de l’endomètre/de l’utérus présumé, de cancer de la vulve présumé, de cancer du vagin présumé, de malignités hématologiques présumées et de cancer de la peau présumé. Elles réfèrent également à d’autres lignes directrices de la CAR en cas de cancer présumé du cou, de la thyroïde, du cerveau, du poumon, du péricarde/de la région cardiaque, de l’œsophage/de l’estomac, du rein, de la glande surrénale, de la vessie, du testicule, de la prostate ou des ovaires, de masse ou tumeur présumée des tissus mous, de tumeur osseuse ou myélome présumé, de tumeur de la colonne vertébrale présumée et de cancer du poumon de découverte fortuite.
Introduction
Beginning in March 2024, an Expert Panel (EP) made up of physicians from the disciplines of radiology, medical oncology, surgical oncology, radiation oncology, family medicine/general practitioner oncology, a patient advisor, and an epidemiologist/guideline methodologist met to develop a new set of recommendations specific to referral pathways for suspected and incidental cancers. Through discussion (via a virtual meeting) followed by offline communication, the EP developed a list of 29 clinical/diagnostic scenarios to be covered by this guideline. These recommendations are intended primarily for referring clinicians (eg, family physicians, specialty physicians, nurse practitioners); however, they may also be used by radiologists, individuals/patients, and patient representatives.
Our methods describing the guideline development process, including the rapid scoping review to identify the evidence base, has been published in CMAJ Open 1 and an editorial to this series of guideline publications is available in CARJ. 2 The application of well-established scoping review and rapid review guidance (JBI, 3 Cochrane Handbook, 4 Cochrane Rapid Review Methods Group 5 ) and guideline methodology (ie, Grading of Recommendations Assessment, Development, and Evaluation or GRADE6,7) were used to identify the evidence-base and to guide the Expert Panel in determining the strength and direction of the recommendations for each clinical scenario (Table 1). The quality of conduct and reporting of the included guidelines identified in the scoping review were evaluated with the AGREE-II checklist, 8 using a modified scoring system. In instances where guidelines were lacking, expert consensus was used to develop the recommendation. Contextualization to the Canadian health care system was considered for each recommendation, with discussion around the factors found in the Evidence to Decision framework in GRADE for guidelines (eg, balance of desirable and undesirable outcomes, values and preferences, resources implications). 7
Recommendation Text, Symbol, and Interpretation.
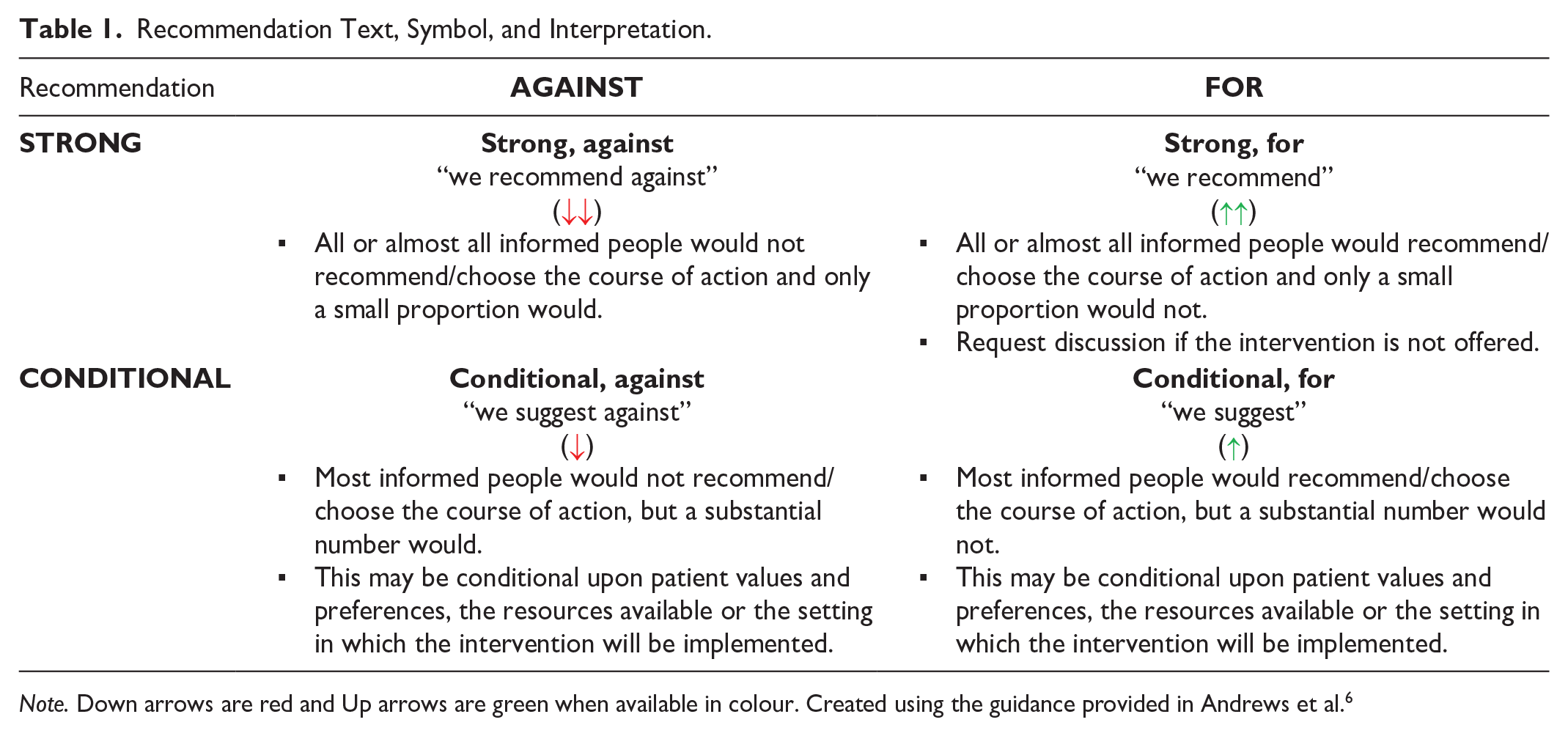
Note. Down arrows are red and Up arrows are green when available in colour. Created using the guidance provided in Andrews et al. 6
A systematic search for guidelines (with an a priori defined inclusion criteria) was run in Medline and Embase on May 25, 2024. The search was limited to publications from 2019 onward (Supplemental Appendix 1). Supplemental searching included the following national radiology and/or guideline groups: the American College of Radiology and the National Institute for Health and Care Excellence. Recommendations for each clinical scenario were formulated over one virtual meeting on October 19th, 2024. External review and feedback were obtained from radiologists, an emergency physician, a family physician, a spine surgeon, and a nurse practitioner. The full guideline can be found on the CAR website (www.car.ca).
Results
Systematic Scoping Review
A total of 6217 records were identified through the electronic database and 2 additional records were added from the supplemental search. Twenty-one guidelines were included (Figure 1). Potentially relevant guidelines published in languages other than English can be found in Supplemental Appendix 2. A list of excluded records with justifications for exclusion is available upon request. Most guidelines were rated as moderate or high quality, using the modified AGREE-II checklist 8 (Supplemental Appendix 3). The number of guidelines included per clinical/diagnostic scenario ranged from 0 to 7, with a median of 2 guidelines per clinical scenario.
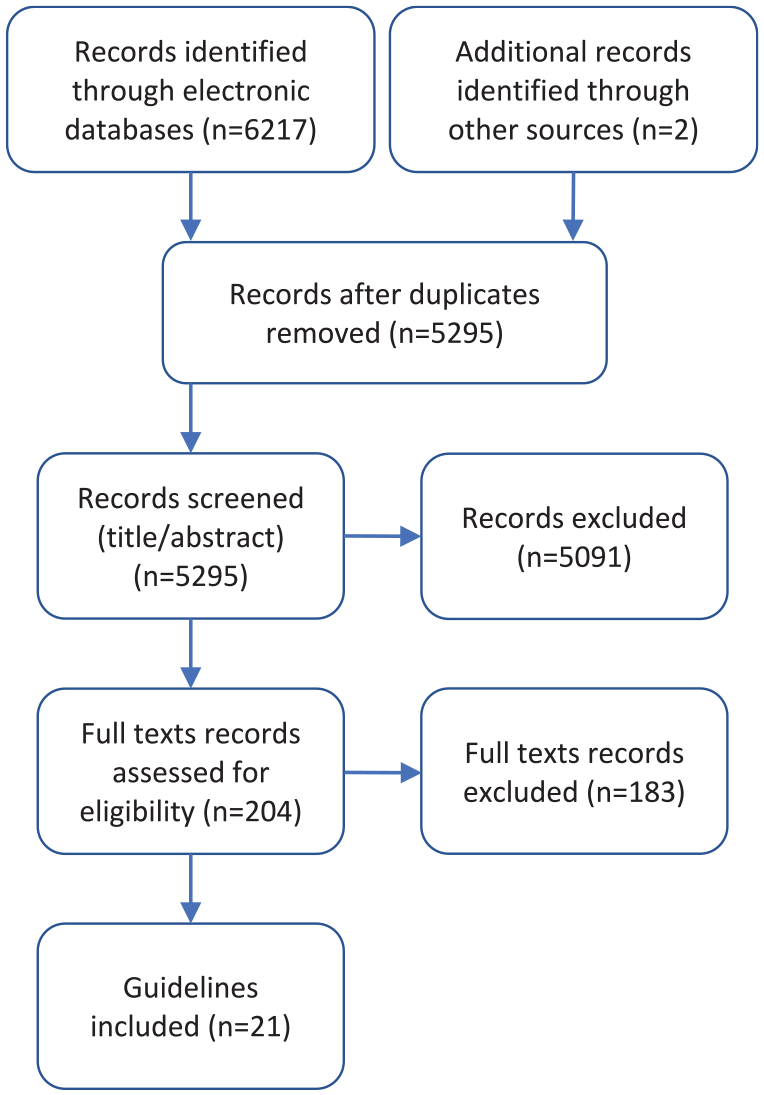
PRISMA flow diagram.
Recommendations
Additional details of the included guidelines, including which imaging modalities (eg, computed tomography [CT], magnetic resonance imaging [MRI], radiograph [XR], ultrasound [US]) that were discussed can be found in Supplemental Appendix 4.
A guideline is intended to guide and not be an absolute rule. Medical care is complex and should be based on evidence, a clinician’s expert judgment, the patient’s circumstances, values, preferences, and resource availability. Not all imaging modalities are available in all clinical environments, particularly in rural or remote areas of Canada. Decisions about patient transfer, use of alternative imaging or serial clinical examination and observation can be complex and difficult. Therefore, the expected benefits of recommended imaging, risks of travel, patient preference, and other factors must be considered. The guideline recommendations are designed to assist the choice of imaging modality in situations where it is deemed clinically necessary to obtain imaging.
Unless the panel agreed a specific protocol is required to optimize patient care/diagnosis, the recommendations do not specify when contrast should or should not be used, as this decision may vary based on clinical presentation, regional practice preferences, preference of the referring clinician, radiologist, and/or the patient, and resource availability.
We reviewed relevant recommendations related to the 13 clinical/diagnostic scenarios previously published by radiology and specialty societies, including: the Canadian Association of Radiologists, 9 the American College of Radiology, 10 American Association for the Study of Liver Diseases, 11 the American Society of Clinical Oncology, 12 the Argentinian clinical practice guideline, 13 the British Gynaecological Cancer Society, 14 the British Society for Haematology, 15 the European Association of Urology-American Society of Clinical Oncology, 16 the European Society of Gynaecological Oncology, the International Society of Ultrasound in Obstetrics and Gynecology, the International Ovarian Tumour Analysis group and the European Society for Gynaecological Endoscopy, 17 the European Society of Gynaecological Oncology jointly with the European Society for Radiotherapy & Oncology and the European Society of Pediatric Oncology, 18 the German Clinical Practice guideline, 19 the German Society for Digestive and Metabolic Disorders, 20 the German Society for Gynecology and Obstetrics, the Gynecology Oncology Working Group of the German Cancer Society and the German Cancer Aid, 21 the Italian Association of Medical Oncology, 22 the Italian Society for Ultrasound in Obstetrics and Gynecology, 23 the Japan Pancreas Society, 24 the Joint French clinical practice guideline, 25 the Korean Liver Cancer Association, 26 the Korean National Cancer Center, 27 the Liver Cancer Study Group of Japan, 28 and the Philippine guideline. 29
Seventeen scenarios (see note below) in this guideline refer to the CAR Head and Neck guideline, 30 the CAR Central Nervous System guideline (accepted in CARJ December 2024), the CAR Thoracic guideline, 31 the CAR Cardiovascular guideline, 32 the CAR Musculoskeletal guideline, 33 the CAR Spine guideline, 34 the CAR Gastrointestinal guideline, 35 the CAR Genitourinary guideline, 36 and the CAR Obstetrics and Gynecology guideline. 37
Note: During the formulation of the recommendations for CA23. Suspected ovarian cancer, the Cancer EP noted that the recommendations from the CAR Obstetrics and Gynecology guideline aligned with the recommendations being discussed. Therefore, we direct the reader to that guideline. 37
Recommendations are presented in Table 2.
Cancer Recommendations.

Supplemental Material
sj-pdf-1-caj-10.1177_08465371241312087 – Supplemental material for Canadian Association of Radiologists Cancer Diagnostic Imaging Referral Guideline
Supplemental material, sj-pdf-1-caj-10.1177_08465371241312087 for Canadian Association of Radiologists Cancer Diagnostic Imaging Referral Guideline by Candyce Hamel, Barb Avard, Howard Lim, Alex Mathieson, Alan Michaud, Kristoff Nelson, Devang Odedra, Jason Pantarotto, Anna Wilkinson and Karim Samji in Canadian Association of Radiologists Journal
Footnotes
Acknowledgements
We would like to thank: Becky Skidmore for creating the search strategies for the systematic scoping review, and the following individuals on the Diagnostic Imaging Referral Guidelines Working Group and external reviewers for providing feedback on the guideline (listed alphabetically): Alanna Coleman, Nicolas Dea, Cathy MacLean, Ryan Margau (WG co-chair), Paul Pageau (WG co-chair), Charlotte Yong-Hing, Kaitlin Zaki-Metias.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Medical Association.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
