Abstract
This is a visual representation of the abstract.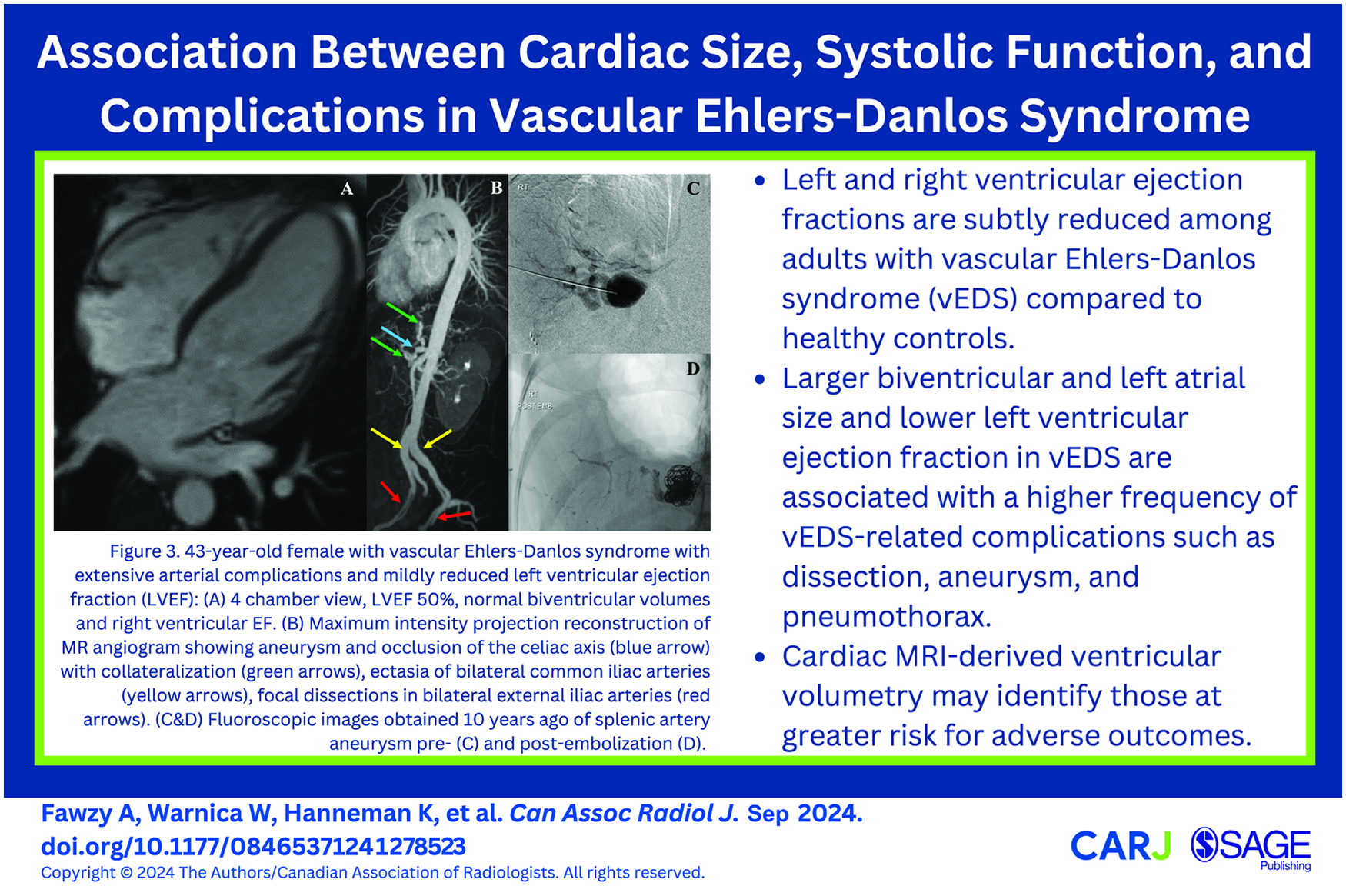
Introduction
Ehlers-Danlos syndrome (EDS) comprises a group of heritable connective tissue disorders, characterized by joint hypermobility, skin hyperextensibility, and tissue fragility. 1 The 2017 International Classification System for EDS specified 13 EDS subtypes, each with their unique genetic and clinical manifestations, and has since become the diagnostic standard. 1 The vascular EDS (vEDS) subtype, a heritable thoracic aortic disease (HTAD), is rare with an estimated frequency of 1/50 000 to 1/200 000. 2 vEDS is attributed to a pathogenic variant in the COL3A1 gene which encodes type III procollagen, a major component of the vessel walls and hollow organs.1,2 The resultant tissue fragility increases the risk of arterial dissection, rupture, and hollow organ rupture.1-5
Three α chains form the type III procollagen molecule and when the large globular domains are cleaved from both ends, type III collagen is formed. 6 Type III collagen is the second most predominant type of myocardial collagen, constituting 10% to 45%, and plays an important role in cardiac structure and function. 7 Therefore, abnormalities in type III collagen may potentially result in abnormal myocardial remodelling. However, cardiac involvement in vEDS has not been systematically investigated. An isolated case report described cardiomyopathy in an individual with vEDS, presumably attributable to the genotype. 8 Intrinsic cardiac impairment and a primary cardiomyopathy is known in other HTADs such as Marfan syndrome. 9 Therefore, the purpose of this study is to evaluate atrial and ventricular size and ventricular systolic function in individuals with vEDS and to explore their associations with vEDS-related complications. We hypothesize that individuals with vEDS may have subtle intrinsic cardiac impairment and the presence would be associated with vEDS-related complications.
Methods
Study Population
This single-centre retrospective cohort study was approved by the institutional research and ethics board. Waiver of informed consent was granted. Inclusion criteria were consecutive adults (≥18 years) with a genetically confirmed diagnosis of vEDS, referred for clinical cardiovascular MRI between February 2015 and March 2023 for routine surveillance of aorta and major branches or follow up of dissection and/or aneurysm. Exclusion criteria were lack of a contrast-enhanced MR angiography (MRA) and any artifacts degrading image quality. Demographics and clinical data, including medications, comorbidities including diabetes, hypertension, coronary artery disease (CAD) were retrieved from electronic medical records. vEDS-related complications such as aneurysms and dissection events, cardiovascular surgeries, extravascular complications including hollow organ rupture, pneumothorax, and pregnancy outcomes when applicable were also collected. For the control group, a convenience sample of adults ≥18 years referred for clinical cardiac MRI between January 2017 and October 2019 were identified from the institutional database. Subjects were included only if all investigations were normal, including electrophysiology, echocardiography, and cardiac MRI. Indications for screening cardiac MRI included family history of suspected cardiomyopathy, or workup of syncope or chest pain when the clinical suspicion of cardiac etiology was low. Since more than mild valvular regurgitation, prior type A aortic dissection or coronary dissection can be confounders for ventricular dysfunction, we conducted a subgroup analysis after applying these additional exclusion criteria among the vEDS group when comparing ventricular volumetry with controls.
Cardiac MRI Acquisition
Cardiac MRI was performed using either a 1.5T scanner (Magnetom Avanto fit; Siemens Healthcare, Erlangen, Germany) or a 3T scanner (Magnetom Skyra fit; Siemens Healthcare, Erlangen, Germany) equipped with commercially available cardiac surface coils. Retrospectively gated balanced steady-state free precession (bSSFP) cine images were obtained in long axes and a stack of short-axis sections with coverage from cardiac base to the apex (repetition time ms/echo time ms, 40/1.19; field of view, 360 mm; section thickness, 8 mm; intersection gap, 2 mm). A stack of bSSFP images were obtained through the aortic root in double-oblique cross-section (slice thickness 6-8 mm, 0 mm interslice gap, and temporal resolution 30-40 ms). In the vEDS group, non-electrocardiogram gated contrast-enhanced MRA was performed in the sagittal plane after administering 10 to 15 cc gadobutrol (Gadovist, Bayer Healthcare, Berlin, Germany) intravenously. The acquisition parameters were optimized to achieve maximal aortic enhancement during the arterial-phase acquisition.
Cardiac MRI Analysis
Ventricular volumetry was performed using the stack of short-axis cine SSFP images. To calculate ventricular volumes, ejection fraction, and mass, a semi-automated contour detection method was employed, with manual adjustment when necessary. Papillary muscles and trabeculations were included as part of the left ventricular (LV) volumes. Cardia MRI post-processing was performed using commercially available software (CVI42, Circle Cardiovascular Imaging Inc., Calgary, Alberta, Canada). Qualitative assessment of the valves was carried out using long-axes cine SSFP images to determine the presence and severity of regurgitation, stenosis, and prolapse. The largest dimensions of the aortic root, measured from sinus to sinus, were obtained from the aortic root cross-section cine SSFP image at end-diastole. The maximum dimensions of the thoracoabdominal aorta were measured from double oblique measurements derived from MRA using commercially available software (Aquarius iNtuition Viewer, TeraRecon, Durham NC, USA). The vasculature was also evaluated for the presence of dissections. We used the pre-existing clinical radiology reports to extract the cardiac MRI metrics.
Statistical Analysis
Categorical variables were reported as frequencies (percentage) and continuous variables as means ± standard deviation or median (interquartile range), as appropriate. For continuous variables, the distribution of data was tested using Kolmogorov–Smirnov test to check for normality. To compare continuous variables among groups, we employed either a two-tailed independent samples t-test or a non-parametric Mann-Whitney test. For the comparison of categorical variables, we utilized the Chi-square test or Fisher’s exact test as appropriate. To assess associations between continuous variables, we used Pearson correlation. To assess associations between dichotomous and continuous variables, we used multivariable logistic regression analysis. For all statistical tests, a P-value <.05 was considered statistically significant. All statistics were performed using Statistical Package for the Social Sciences (SPSS Statistics 28.0, Chicago, IL).
Results
Demographic and Clinical Characteristics
We included a total of 26 individuals with vEDS and 26 controls, who were similar in age (38.6 ± 15.6 vs 38.0 ± 13.8 years, P = .88) and sex distribution (both 50.0% female, P > .99). Demographic and clinical characteristics are detailed in Table 1. Hypertension was the most common comorbidity in vEDS (19.2%) and beta-blockers were the most commonly used antihypertensive medication (53.8%). Median follow up time was 2.4 (1.1-3.6) years.
Demographics and Clinical Characteristics in vEDS and Controls. .
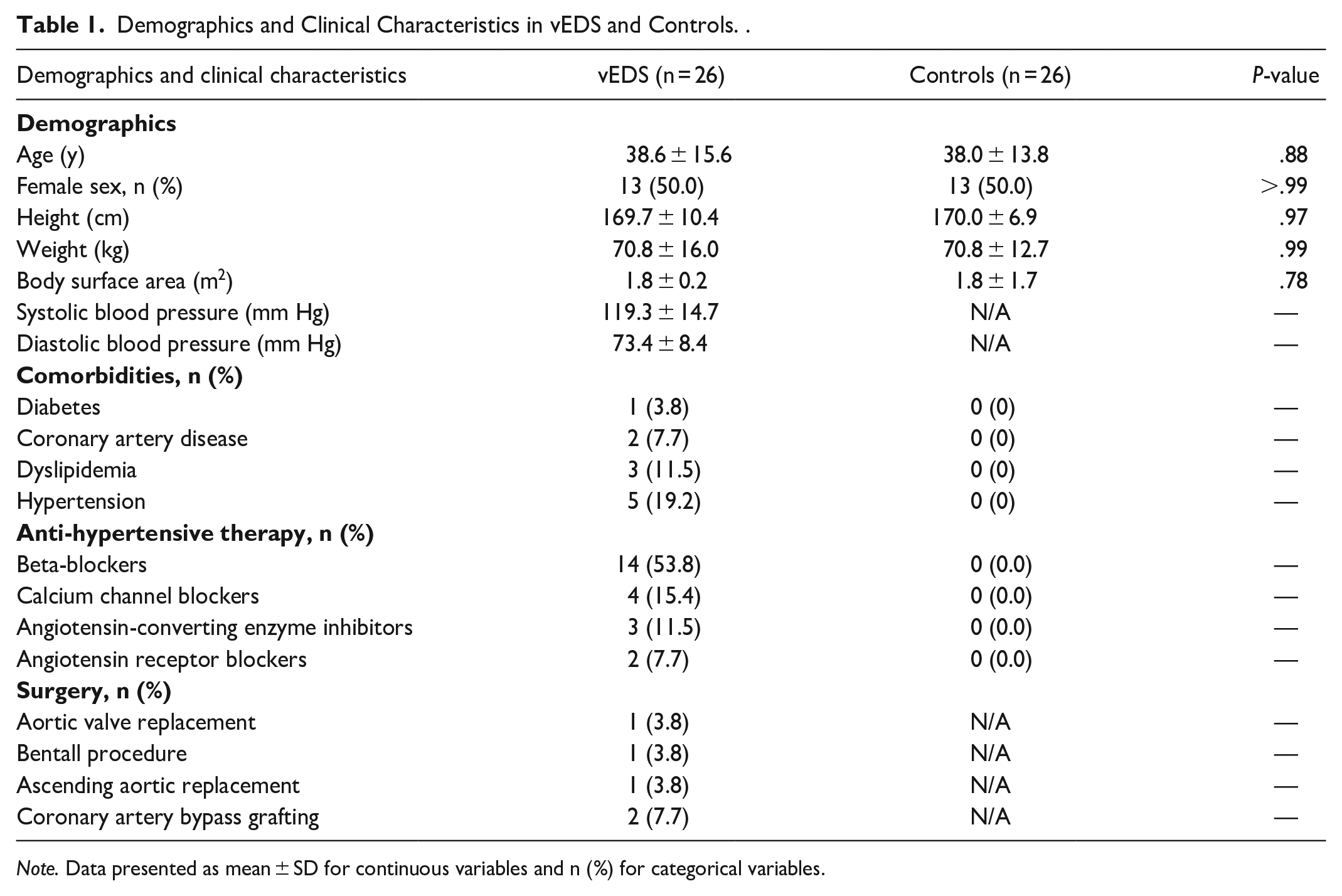
Note. Data presented as mean ± SD for continuous variables and n (%) for categorical variables.
Cardiac MRI Characteristics
Cardiac MRI characteristics are summarized in Table 2. The vEDS group had normal mean thoracic aortic dimensions as measured on MRA. Mild tricuspid regurgitation was the most common valvular abnormality, occurring in 34.6% of individuals with vEDS. While the LV and right ventricular end-diastolic and end-systolic volumes indexed to body surface area (LVEDVi, RVEDVi; LVESVi, RVESVi) were similar between the vEDS group and controls, the LV and RV ejection fractions (LVEF, RVEF) were significantly lower in the vEDS group compared to controls (58 ± 6% vs 61 ± 4%, P = .03; 54 ± 5% vs 58 ± 4%, P = .03). In the subgroup analysis, a total of 16 individuals with vEDS were included after excluding confounders for ventricular dilatation and/or dysfunction (flow-limiting CAD/coronary dissection or myocardial infarction [n = 3], prior type A aortic dissection [n = 2], and moderate or severe valve regurgitation [n = 2]). The reduction in LVEF and RVEF in vEDS compared to controls persisted (58 ± 5% vs 61 ± 4%, P = .04; 53 ± 4% vs 58 ± 4%, P = .003), detailed in Supplemental Table 1.
Cardiac MRI Characteristics in vEDS and Controls. .
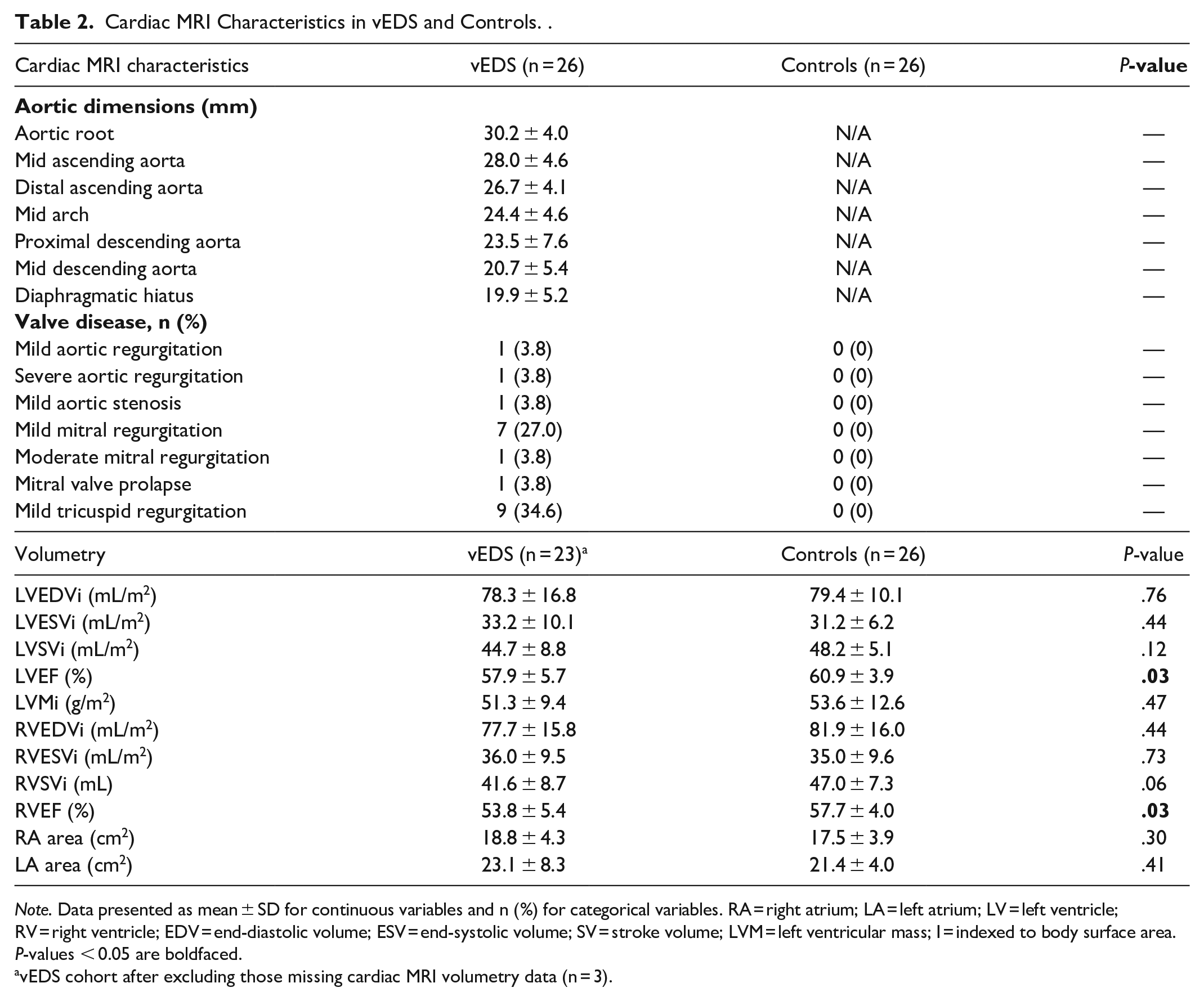
Note. Data presented as mean ± SD for continuous variables and n (%) for categorical variables. RA = right atrium; LA = left atrium; LV = left ventricle; RV = right ventricle; EDV = end-diastolic volume; ESV = end-systolic volume; SV = stroke volume; LVM = left ventricular mass; I = indexed to body surface area.
P-values < 0.05 are boldfaced.
vEDS cohort after excluding those missing cardiac MRI volumetry data (n = 3).
Complications and Associations With Cardiac Size and Systolic Function
In terms of vascular complications, a total of 24 dissections were observed in 50.0% of the vEDS group (13/26), with the common iliac artery being the most affected site (20.8%, 5/24; Figure 1). A total of 25 aneurysms were observed in 30.7% of the vEDS group (8/26), with the common iliac artery being the most affected site (32.0%, 8/25; Figure 2). Overall, 53.8% (14/26) had arterial complications (either a dissection or an aneurysm or both; Figure 3).
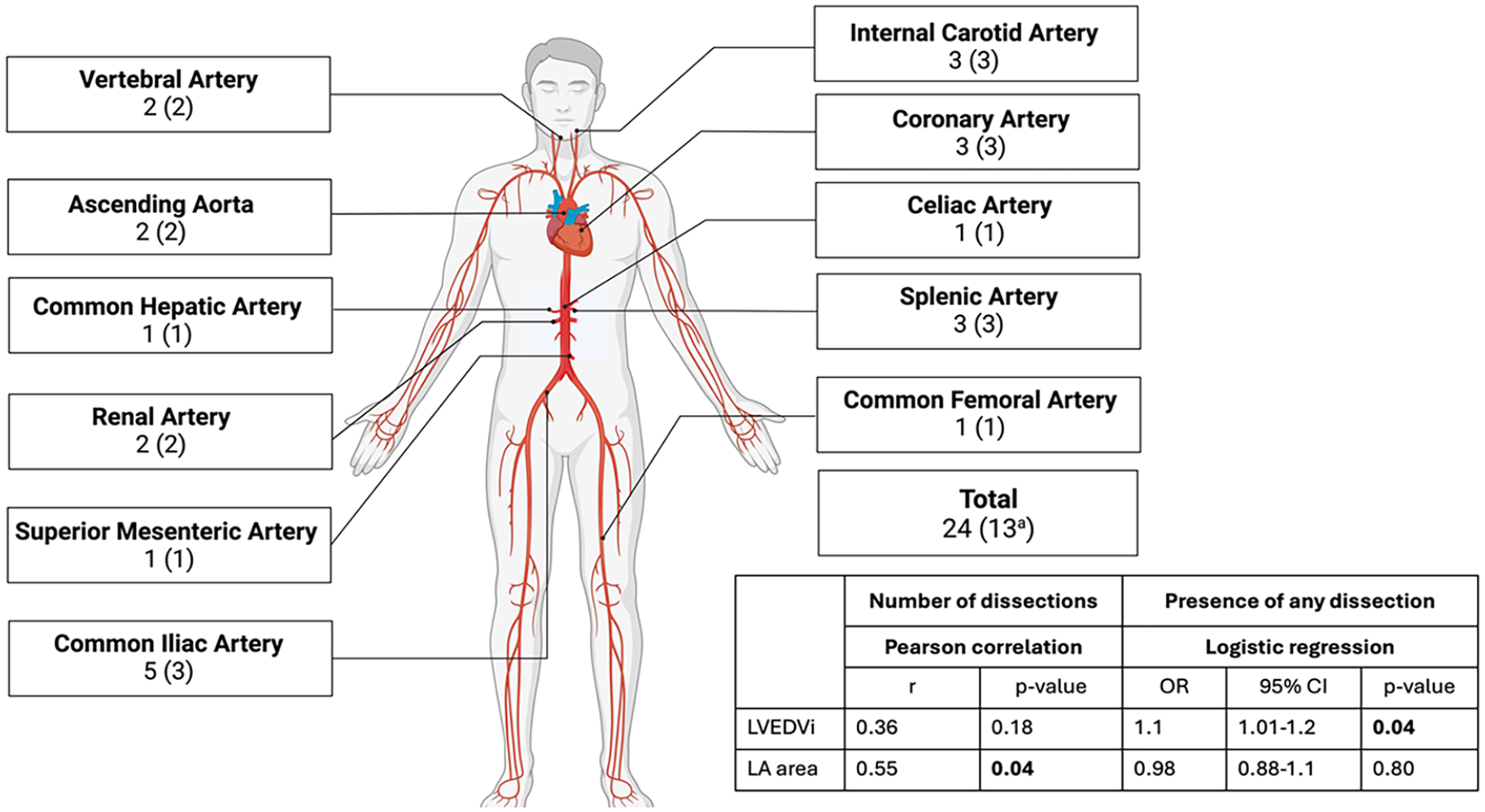
Total arterial dissections in vEDS, presented as number of dissections (number of patients). Multivariable logistic regression controlling for age, sex, and antihypertensive medication.
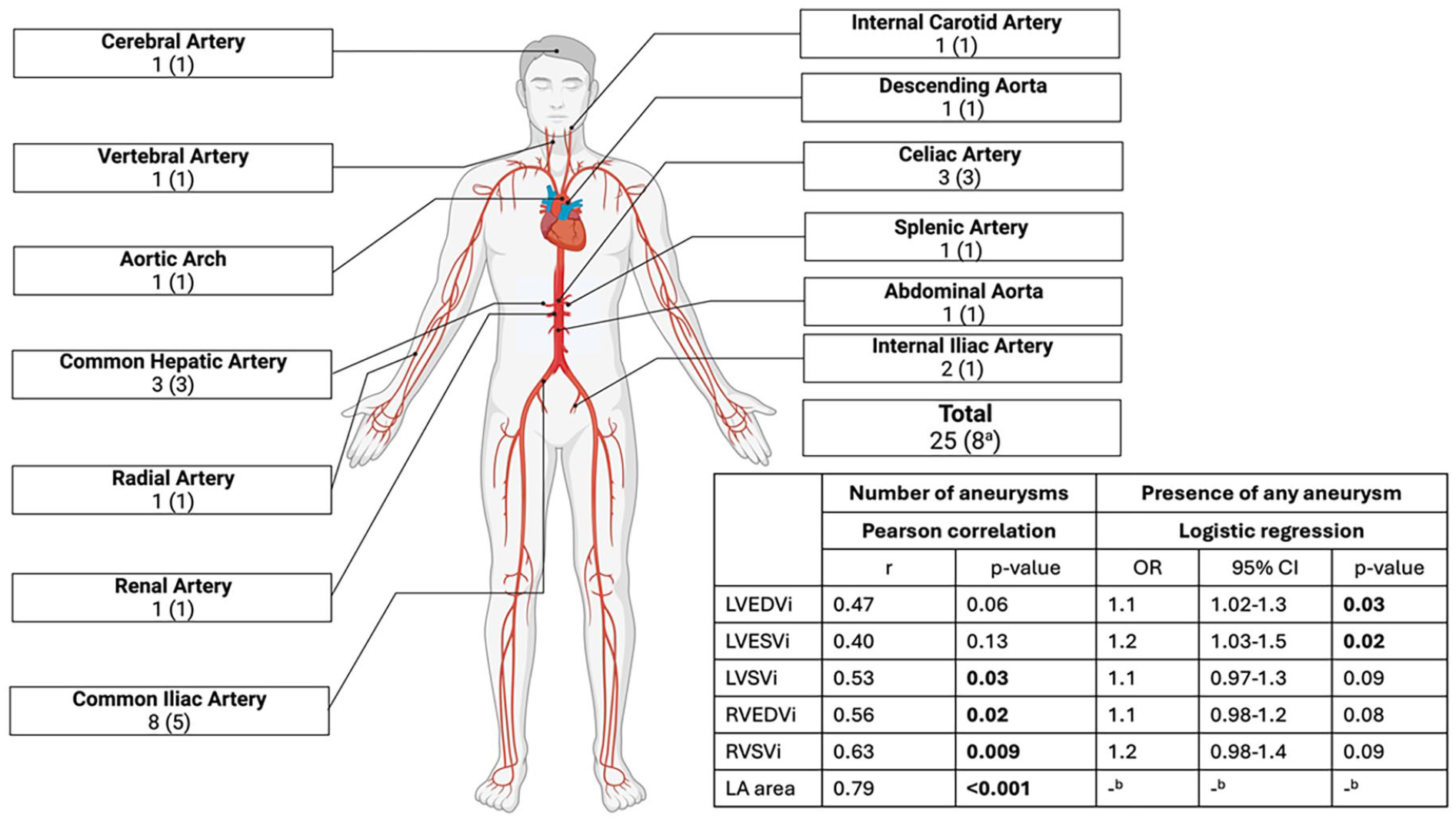
Arterial aneurysms in vEDS, presented as number of aneurysms (number of patients). Multivariable logistic regression controlling for age, sex, and antihypertensive medication.
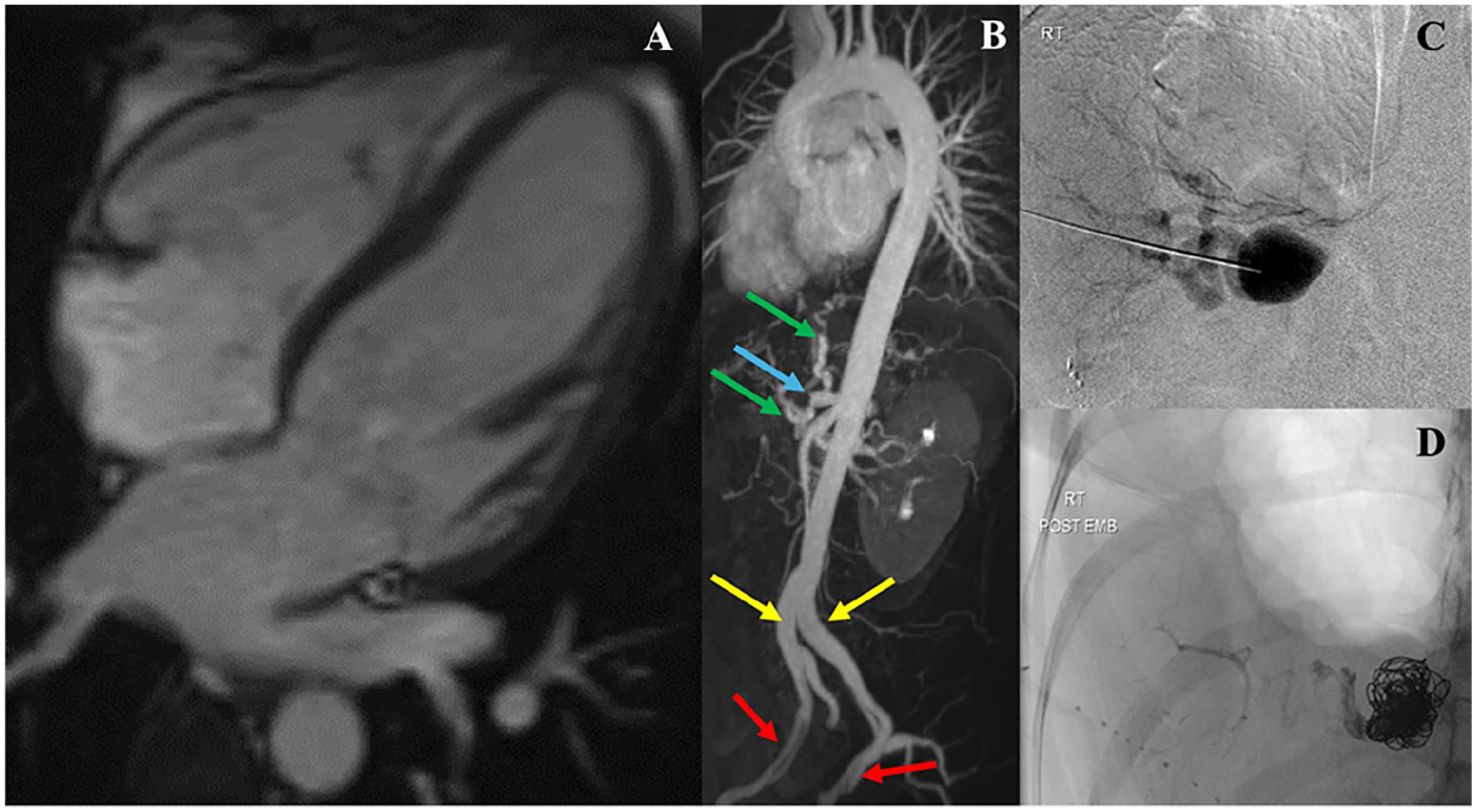
Forty-three-year-old female with vascular Ehlers-Danlos syndrome with extensive arterial complications and mildly reduced left ventricular ejection fraction (LVEF): (A) 4 chamber view, LVEF 50%, normal biventricular volumes and right ventricular EF. (B) Maximum intensity projection reconstruction of MR angiogram showing aneurysm and occlusion of the coeliac axis (blue arrow) with collateralization (green arrows), ectasia of bilateral common iliac arteries (yellow arrows), focal dissections in bilateral external iliac arteries (red arrows). (C and D) Fluoroscopic images obtained 10 years ago of splenic artery aneurysm pre- (C) and post-embolization (D).
Other vascular (myocardial infarction, stroke, thromboembolism, haemorrhage), non-vascular (organ rupture, pneumothorax), and pregnancy-related complications are listed in Table 3. Pneumothorax was the most common non-vascular complication, occurring 18 times in 5 individuals. Two out of three coronary artery dissections occurred during pregnancy.
Other Vascular and Extravascular Complications in the vEDS Group (n = 26).
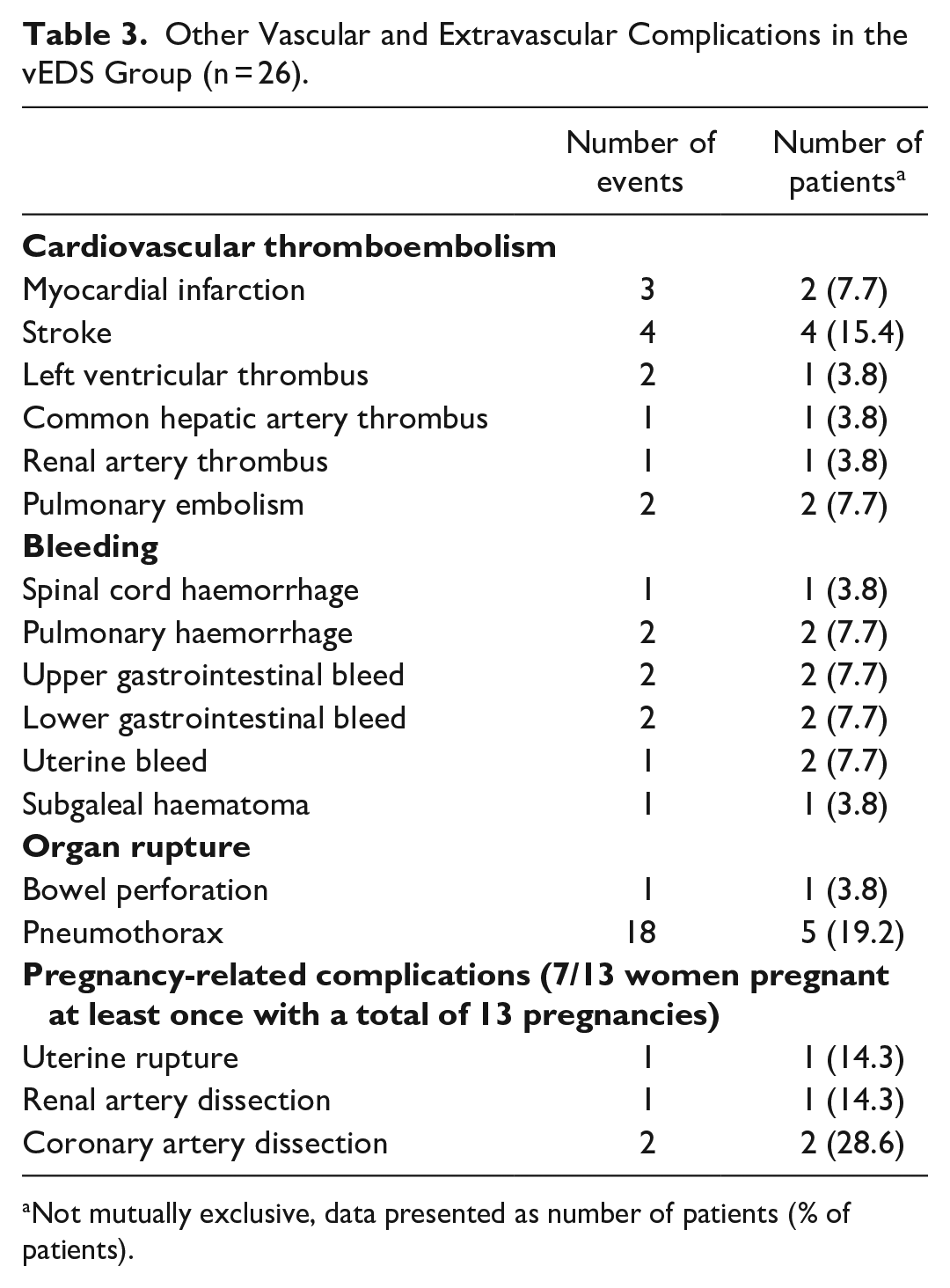
Not mutually exclusive, data presented as number of patients (% of patients).
The multivariable logistic regression model revealed that LVEF was predictive of any vEDS-related complication, even after controlling for age, sex, and antihypertensive therapy (Supplemental Table 2), with every 1% reduction in LVEF, the odds of presence of any vEDS-related complication were 1.41 times higher (OR 1.41, 95% CI 1.01-1.92, P = .04). LVEDVi was predictive of presence of dissection (OR 1.1, 95% CI 1.01-1.2, P = .04; Figure 1). LVEDVi and LVESVi were predictive of presence of any aneurysm (OR 1.1, 95% CI 1.01-1.3, P = .03 and OR 1.2, 95% CI 1.03-1.5, P = .02, respectively; Figure 2). Other ventricular volumes or RVEF were not predictive of complications (Supplemental Table 2).
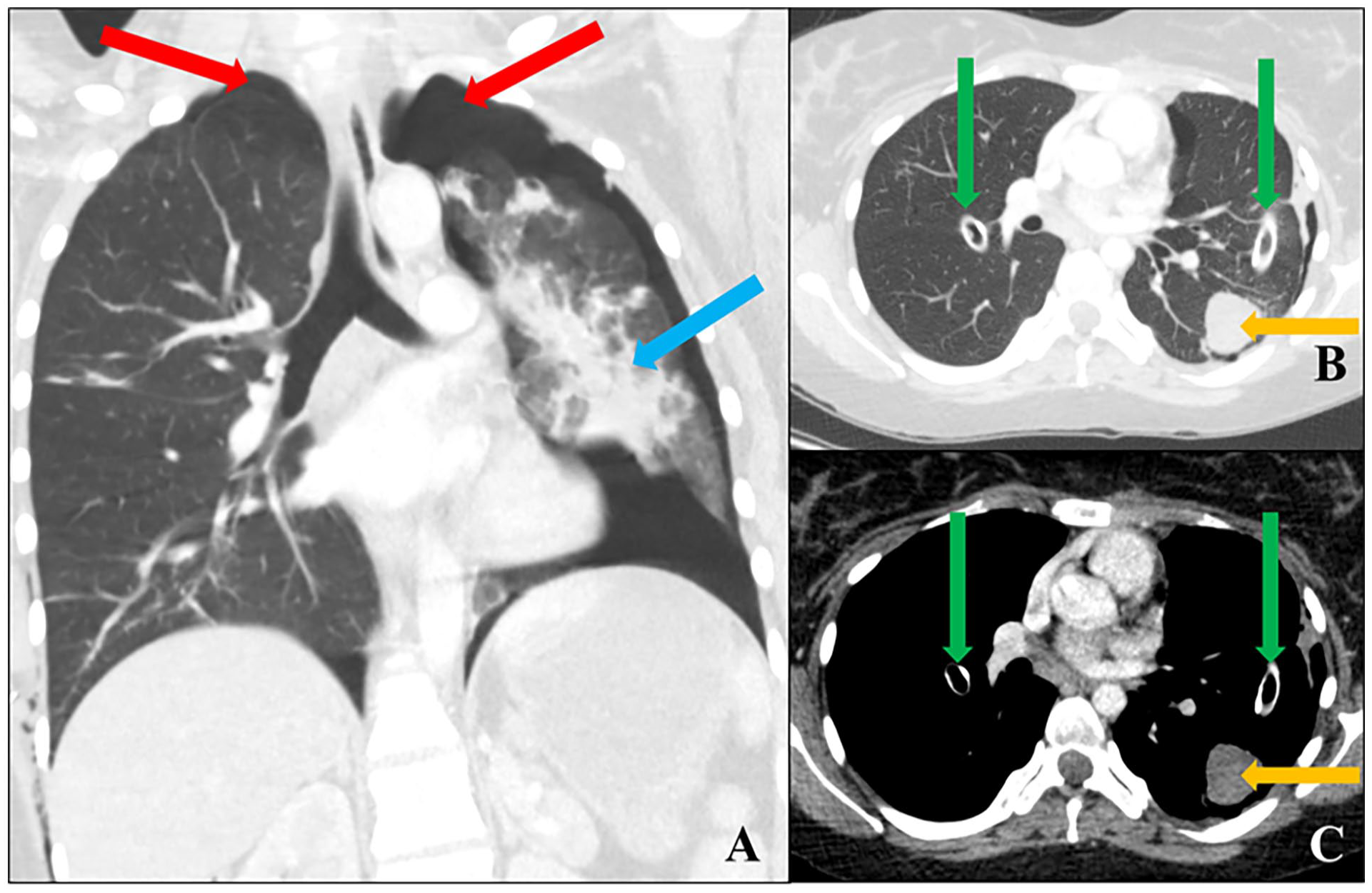
Nineteen-year-old female with vascular Ehlers-Danlos syndrome with a history of recurrent pneumothoraces: (A) Coronal CT chest in the lung window demonstrates bilateral pneumothoraces (red arrows) and pulmonary haemorrhage in the left lung (blue arrow). Eleven months later, chest tubes were inserted (green arrows), seen on (B) axial CT chest in the lung window and (C) mediastinal window. A haemorrhagic cavity (45 Hounsfield Units) evolved in the left lower lobe (orange arrow). The left ventricular ejection fraction was minimally reduced at 54%. There were no arterial complications.
LA area correlated with number of dissections (r = .55, P = .04; Figure 1). RVEDVi, LV, and RV stroke volume indexed to body surface area (LVSVi, RVSVi), and left atrial (LA) area correlated positively with the number of aneurysms (r = .56, P = .02; r = .53, P = .03; r = .63, P = .009; r = .79, P < .001 respectively; Figure 2). Ventricular EFs or other ventricular volumes did not correlate significantly with complications (Supplemental Table 3).
Within the vEDS cohort included for subgroup analysis, pneumothorax occurred exclusively among those with LVEF below the mean (LVEF <58%, 8/16) compared to those with LVEF equal to or above the mean (LVEF ≥58%, 8/16; 50.0% (4/8) vs 0.0% (0/8); P = .02), Figure 4. The fifth individual with pneumothorax did not have volumetry data. Individuals with vEDS with LVEF <58% also had more frequent dissection and/or aneurysm as compared to individuals with vEDS with LVEF ≥58% (75.0% [6/8] vs 12.5% [1/8]; P = .04). Additionally, individuals with vEDS with LVEF <58% had more frequent complications (any vEDS-related) as compared to those with LVEF ≥58% (87.5% [7/8] vs 37.5% [3/8]; P = .03).
Discussion
The novel findings to arise from our study include: (1) Biventricular EFs are slightly lower in vEDS as compared with controls (2) Larger LV, RV, LA volumes and lower LVEF are associated with a higher frequency of complications in vEDS (3) Spontaneous pneumothoraces occur exclusively in those with lower LVEF. To our knowledge, this is the first study to have systematically evaluated for primary functional cardiac impairment in vEDS and the associations between cardiac size and systolic function, and vEDS-related complications.
Primary Cardiomyopathy in HTADs
A pathogenic variant in the COL3A1 gene is responsible for the manifestations of vEDS. 10 COL3A1 gene encodes the pro-α1 chains of type III collagen. 6 We know that type III collagen is a major constituent of the extracellular matrix of the arteries and hollow organs.6,7 Furthermore, it constitutes up to 50% of the myocardial collagen. 7 Therefore, it is plausible that the dysregulation of the extracellular matrix could result in abnormal myocardial remodelling, along with causing the known vEDS-related arterial and hollow organ ruptures. This may manifest in the form of a subtle cardiomyopathy characterized by slight reduction in ventricular function, with or without dilatation of the ventricles and increase in LV mass.
This understanding builds on prior studies demonstrating a genotype-related cardiomyopathy in other HTADs such as Marfan syndrome. 11 In Marfan syndrome, fibrillin-1 (FBN1) mutations are thought to alter myocardial structure and function.12,13 Marfan cardiomyopathy is recognized as a primary cardiomyopathy, replicated in numerous studies in the absence of significant valve disease, aortic dilatation, and other confounders for cardiac impairment, thereby a presumed consequence of the FBN1 gene mutations.14-20 Clinically, this manifests as a dilated cardiomyopathy phenotype with biventricular dilatation and systolic and diastolic dysfunction.15,16 A dilated cardiomyopathy has also been described in individuals with pathogenic SMAD3 variants, which give rise to another form of HTAD. 21 A primary cardiomyopathy has also been described in individuals with Loeys-Dietz syndrome, where the genetic mutation results in dysregulation in the transforming growth factor beta pathway, similar to Marfan syndrome.22,23
There are isolated descriptions of concomitant cardiomyopathy with EDS. Hypertrophic obstructive cardiomyopathy was reported in an individual with EDS (type not specified). 24 Left ventricular non-compaction cardiomyopathy was also described in an individual with vEDS. 8 In both reports, the authors contemplate the role of the genetics in the development of cardiomyopathy. However, among our vEDS group, we did not encounter any morphologic abnormalities pertaining to elevated LV mass focally or globally or in the form of hypertrabeculation. The observed lower LVEF and RVEF in the vEDS cohort compared to controls in our study persisted even after the exclusion of individuals who had confounders for ventricular dilatation or dysfunction. Blood pressure lowering medications such as beta blockers and angiotensin receptor blockers are routinely prescribed in HTAD to reduce the stress on the weakened arterial wall. 25 These are known to halt disease progression and are also cardioprotective. In keeping with the current guidelines, majority of individuals with vEDS were on anti-hypertensive medications. Even in the presence of this cardio-protective therapy for individuals with vEDS, the subtly lower EFs were seen in vEDS compared to controls. These findings support the hypothesis of COL3A1 mutation-induced abnormality in the myocardial phenotype.
Complications in vEDS
Arterial complications are the hallmark of vEDS, with arterial dissection and aneurysm observed in 40% to 77% of individuals with vEDS.2,10,26 In symptomatic individuals with vEDS, the first major event is predominantly an arterial complication.4,27 These complications can occur in major arteries such as the aorta and iliac arteries, leading to acute, life-threatening events.2,26-29 In our study, 50.0% of individuals with vEDS experienced at least one dissection, and 30.7% had at least one aneurysm, with an overall arterial complication rate of 54.0%, aligning with what is reported in the literature.
Gastrointestinal (GI) complications are also common, found in up to 40% of vEDS and include bowel perforations and gastrointestinal bleeding.2,4,27,30 These conditions often necessitate urgent surgical intervention. A recent large retrospective Dutch study of 142 individuals with vEDS reported a GI complication rate of only 6.0%, underscoring the variability in reported rates. 26 Our finding of GI complications in 19.2% of individuals with vEDS is within this broad range. Type III collagen is also essential for the integrity of the lung interstitial extracellular matrix.6,31 Mutations in the COL3A1 gene have been associated with spontaneous pneumothoraces,32-35 occurring in approximately 6% to 16% of individuals with vEDS.2,26,27,29 The incidence of spontaneous pneumothorax in our study was in line with the literature, at 19.2%.
Pregnancy is particularly high-risk for women with vEDS, with complications reported in up to 31% of pregnancies. 29 These complications include uterine rupture, severe perineal tears, and both antepartum and postpartum haemorrhage.2,4,27-29 Pregnancy is associated with a mortality rate of 5% among women with vEDS, with 50% of those occurring during the first pregnancy.2,32 While a small number of these deaths are due to pregnancy-specific complications, such as uterine rupture, the majority result from arterial complications similar to those seen in non-pregnant women, which are exacerbated due to the physiologic stresses of pregnancy.4,27 Pregnancy-related complications in our vEDS group were markedly higher at 57.1%. We had a small sample size further reduced when limiting to women who experienced at least one pregnancy. Additionally, our institution is a tertiary care referral centre, both for HTAD and high-risk obstetric patients. Particularly these individuals with a higher risk profile undergo more close imaging surveillance which could induce a selection bias. These factors could explain the higher pregnancy-related complications in our study.
Neurological and/or intracranial complications in vEDS are less common, occurring in 3% to 7% of individuals, with stroke (ischaemic and haemorrhagic) occurring in 5% and intracranial aneurysm in 2%.26,28 Although rare, these complications can lead to significant neurological deficits, and require careful monitoring and management. The prevalence of stroke in our vEDS group was higher at 15.4%. The same factors described above could contribute to this discrepancy.
We were intrigued to find that certain larger cardiac MRI-derived volumetric indices and lower LVEF were associated with multiple vEDS-related complications. Notably, lower LVEF was associated with the presence of any vEDS-related complication. According to our data, for every 1% increase in LVEF, the odds of having any complication decreases by 29%, highlighting the importance of LVEF as a predictive marker for complications in vEDS patients. These associations persisted after adjusting for age, sex, and antihypertensive medication, and perhaps, further support the hypothesis that the COL3A1 mutations probably affect the myocardium simultaneously and result in remodelling. When the mutation’s penetrance is severe enough to result in pronounced vEDS manifestations characterized by vessel wall weakening, atrial ruptures, and organ ruptures, changes in myocardial phenotype may also become overt.
Cardiac-valvular EDS (cvEDS) is a subtype of EDS with known cardiac involvement; and is associated with mutations in the COL1A2 gene which encodes type I collagen. 1 Both type I and III collagen contribute to myocardial tensile strength. 7 Despite the unique genetic mutations, owing to the resultant collagen abnormalities, vEDS and cvEDS may have overlapping myocardial changes. Further research is required to explore the possible similarities between the 2 subtypes.
Limitations and Future Directions
This study has some limitations, mainly due to its retrospective nature and a small sample size, owing to the rarity of the disease. Due to the small sample size, we could not control for important confounders in our analysis such as the use of antihypertensive therapy or comorbidities in the vEDS group which could potentially contribute to changes in ventricular function and/or volumes. There was also selection bias since clinicians refer those with a high-risk profile for closer cardiac MRI surveillance. We did not have data on cardiac MRI techniques such as parametric mapping to examine for interstitial fibrosis or 4D flow to explore any altered haemodynamics. Furthermore, since we relied on clinical radiology reports to extract cardiac MRI metrics, small inter-observer differences are possible. Finally, our study does not touch upon the molecular and cellular mechanisms linking COL3A1 mutations to any myocardial phenotype alternations.
A multidisciplinary approach involving comprehensive genetic, cardiologic, and imaging assessment would be beneficial for clinical management. Prospective multicentre longitudinal studies with larger cohorts are necessary to confirm the findings and understand the underlying pathophysiologic mechanisms. Additionally, understanding the impact of the pathogenic variants of COL3A1 mutations on the myocardium and their clinical manifestations would also be important.
Conclusion
Our study sheds light on the subtle myocardial abnormalities in vEDS. Larger cardiac size and lower ventricular function is associated with vEDS-related complications. Cardiac MRI-derived ventricular volumetry may aid in the prognostication in vEDS.
Supplemental Material
sj-docx-1-caj-10.1177_08465371241278523 – Supplemental material for Association Between Cardiac Size, Systolic Function, and Complications in Vascular Ehlers-Danlos Syndrome
Supplemental material, sj-docx-1-caj-10.1177_08465371241278523 for Association Between Cardiac Size, Systolic Function, and Complications in Vascular Ehlers-Danlos Syndrome by Aly Fawzy, William Warnica, Kate Hanneman, Rachel M. Wald, Erwin Oechslin, Paaladinesh Thavendiranathan and Gauri R. Karur in Canadian Association of Radiologists Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: GRK is supported by the Genetic Aortic Disorders Association Canada (GADA), Loeys-Dietz Syndrome Foundation Canada (LDSFC), and University Medical Imaging Toronto (UMIT).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
