Abstract
This is a visual representation of the abstract.
Introduction
Percutaneous vertebroplasty is a well-established minimally invasive procedure used for treatment of painful vertebral fractures through the injection of polymethyl methacrylate (PMMA) bone cement into fractured vertebrae. This technique was first described in the treatment of a haemangioma in the cervical spine and osteolytic metastatic lesions although it soon found more widespread use for treating osteoporotic vertebral compression fractures.1,2 Multiple studies have been published which demonstrate the efficacy of vertebroplasty in providing rapid and robust pain relief for patients with acute osteoporotic and pathological vertebral fractures.3-5 Vertebroplasty aims to decrease pain through the restoration of the load bearing capacity of fractured vertebra and reducing mechanical instability. The physical properties of PMMA allow it to be initially prepared in an injectable form which polymerizes to form a solid material with a high compressive strength that can resist axial loading forces. 6
There has been interest in the spatial distribution of PMMA following vertebroplasty and the correlation with outcomes including recompression and adjacent level fractures.7,8 Previous studies have attempted to classify PMMA spatial distribution or “fill” patterns using either qualitative descriptors based on visual assessment or geometric measurements.9,10 While these studies have shown a correlation with specific PMMA fill patterns and outcomes, these metrics can be subjective or limited in terms of quantification. It is therefore of interest to develop additional quantitative techniques for evaluating PMMA distribution following vertebroplasty used for the treatment of vertebral fractures. The pattern of PMMA distribution achieved following vertebroplasty is inherently related to the internal architecture of a vertebral body, which among other factors, may be modified by bone density, vertebral tumours, and fractures. Through performing vertebroplasty, we have consistently observed that PMMA assumes a different distribution in tumour related fractures compared to osteoporotic fractures, each showing a different level of morphological complexity (Figure 1). In osteoporosis, the reduction in bone density is caused by rarefaction of the trabecular network leading to increased porosity. 11 In osteolytic vertebral metastases there is both destruction of bone tissue combined with rarefaction of the trabecular network, while osteoblastic lesions may cause the opposite effect with a loss of bone marrow space due to internal remodelling. 12 The differences in these processes which modify the internal architecture presumably are related to the variable PMMA fill patterns observed in treated vertebrae.
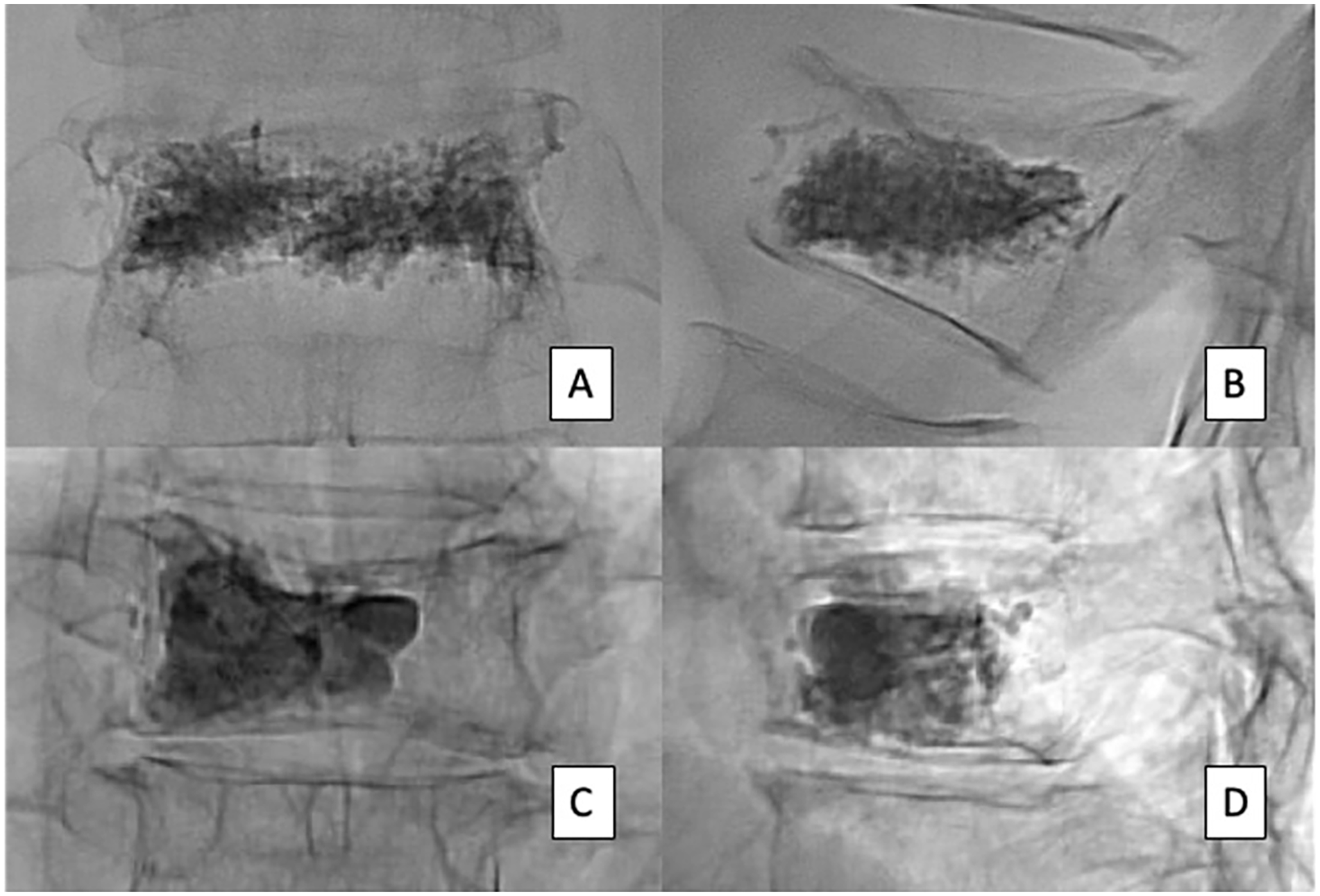
Comparison of PMMA distribution patterns in 2 patients with osteoporotic (A and C) and malignant vertebral compression fractures (B and D) on frontal and lateral radiographs. Frontal (A) and lateral (C) radiographs of the osteoporotic fractures at T12 demonstrates a diffuse or sponge-like filling pattern. The frontal (B) and lateral (D) radiographs of a T9 malignant compression fracture in a patient with multiple myeloma demonstrates a solid or globular pattern of PMMA distribution.
Fractal analysis (FA) is being increasingly used to examine the microstructure of biological tissues in medical research. 13 FA encompasses a range of mathematical tools which can define the geometrical complexity of structures which cannot be otherwise expressed within the familiar concept of three-dimension Euclidean geometry. Fractals are structures which exhibit self-similarity, describing that they can be broken down into smaller copies identical to the whole and can be expressed over a range of spatial scales. 14 Mandelbrot and Wheeler introduced the concept of fractal dimension for capturing geometric complexity of structures and is expressed as non-integer values between the dimensions of 1, 2, and 3. 15 The fractal dimension (FD) is a measure of how complex the surface is, with a higher FD value reflecting greater surface complexity and roughness. FD is predicated on the notion of the self-similarity of fractal objects within complex structures. The microstructure of biological tissues, however, do not routinely demonstrate high degrees of self-similarity or self-repeating textures. This is reflected on medical imaging studies where structures may demonstrate internal heterogeneity due variable size of the underlying structural unit or spatial dispersion of pixel tones within an image. To address this, the concept of lacunarity was introduced to provide additional textural information with regards to differences in how fractals fill space where structures containing larger “gaps” or heterogeneity exhibit greater lacunarity. 16 This can help differentiate between structures which have a similar range of FD. Assessment of the trabecular framework within a vertebra and the distribution of intervening bone marrow spaces is an example in which lacunarity is a useful feature. 17
Prior studies have utilized fractal analysis to examine the fractal dimension and lacunarity of untreated vertebra on conventional radiographs, computed tomography (CT), and magnetic resonance (MR) examinations. These studies have focused on establishing a relationship between fractal properties on imaging and bone mineral density in osteoporosis,17-20 predicting fracture risk in osteoporosis and myeloma,21,22 and differentiating between benign and malignant vertebral fractures. 23 The association between cement distribution and outcomes such as recompression and adjacent level fractures following vertebroplasty may suggest a role in this field for novel imaging tools such as fractal analysis. The aim of our study is to quantify the differences in PMMA spatial distribution following vertebroplasty using FA to examine osteoporotic and malignant vertebral compression fractures. We hypothesize that the PMMA distribution in osteoporotic and malignant vertebral fractures will show statistically different fractal dimensions and lacunarity values, reflecting the underlying pathological process with each entity.
Methods
Patient Selection
This retrospective study was performed following institutional research and ethics board approval; the requirement for informed patient consent was waived. A retrospective database of 29 consecutively registered patients who underwent percutaneous vertebroplasty procedures between November 2019 and December 2020 was created. Patients were recruited to the study if they fulfilled the following inclusion criteria: age older than 18 years, availability of radiographs taken both in the frontal and lateral projections after completion of the vertebroplasty procedure, recent diagnosis of an osteoporotic or pathological vertebral compression fracture. The exclusion criteria included patients with missing clinical records, vertebral levels with the presence of metallic hardware or significant cement extravasation on post-procedural radiographs, and vertebral levels that received balloon kyphoplasty.
Baseline patient metadata recorded included age, sex, fracture etiology, pathology of malignant cases, and total number of treated vertebral compression fractures. The patient demographic data are summarized in Table 1. The study included 12 males and 17 females with average age of 70.1 years. Patients were categorized into osteoporotic or malignant fracture groups based on their fracture etiology, which were diagnosed on MR and/or CT imaging by a neuroradiologist with more than 25 years of experience. The pathology of malignant fractures was based on a combination of unequivocal radiological findings in combination with an established history of a metastatic solid or haematological malignancy. Characteristic imaging findings on standard of care MRI included a paravertebral soft-tissue mass, abnormal signal intensity and thickening of the pedicle and posterior element, development of a convex posterior border with the vertebral body, and evidence of other spinal metastases. The 12 patients with malignant compression fractures had the following underlying primary malignancies: multiple myeloma (n = 8), lung cancer (n = 2), prostate cancer (n = 1), and leiomyosarcoma (n = 1). Six of these patients, including all the solid cancer diagnoses and 2 of the multiple myeloma patients, had a history of radiation in vertebra that were subsequently treated with vertebroplasty (n = 17, 34.2%). The median interval between radiation and vertebroplasty in these 6 patients was 41.5 days (range 13-433). The severity of vertebral height loss was calculated as a percentage of the maximal height loss compared to the mean height of the adjacent superior and inferior vertebral bodies, or the historical vertebral body height if prior imaging was available.
Baseline Demographic and Clinical Characteristics.
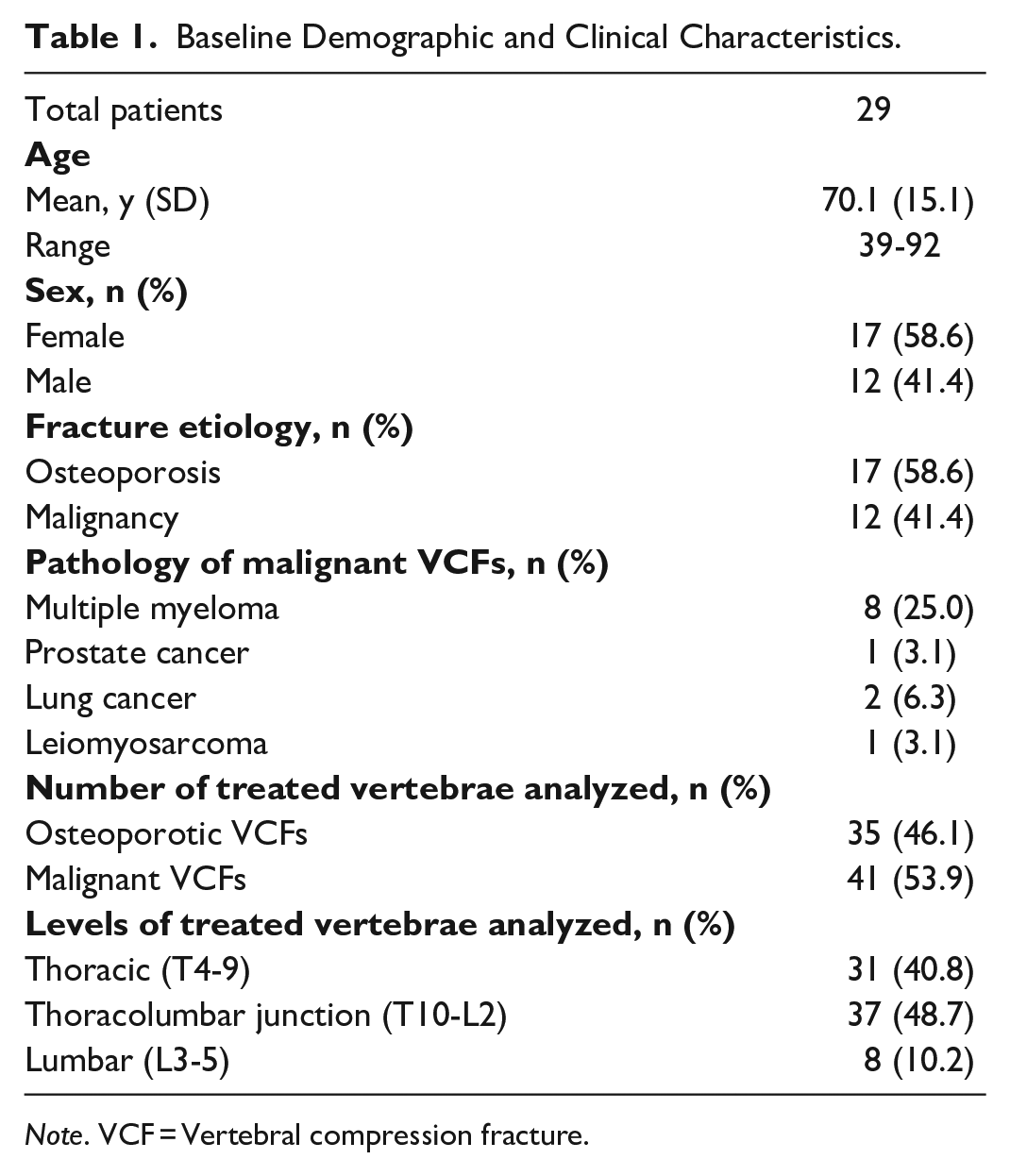
Note. VCF = Vertebral compression fracture.
Vertebroplasty Technique
Vertebroplasty procedures were carried out by 2 board-certified neuroradiologists with more than 25 years of experience. All procedures were performed using biplane fluoroscopy machine (Allura Xper FD20/20, Philips Healthcare, Best, the Netherlands). Standard technique was employed with unilateral or bilateral transpedicular injection through 11- or 13-gauge vertebroplasty needles. A unipedicular approach was used at 60 levels (78.9%), and a bi-pedicular approach at 16 levels (21.1%). Eleven-gauge needles were used at 62 levels (81.6%) and 13G needles were used at 14 levels (18.4%). High-viscosity PMMA bone cement (Kyphon HV-R bone cement, Medtronic, Minneapolis, USA) was utilized for all vertebroplasty procedures. Radiofrequency or cryoablation ablation were not performed at any of the vertebroplasty levels. Cement was prepared using the proprietary mixer and injected after 5 minutes from initial mixing to ensure a standardized viscosity. Injection was performed until satisfactory vertebral cement distribution was achieved. A set of completion radiographs were obtained in the frontal and lateral views after cement injection and vertebroplasty needle removal.
Image Analysis
Post-procedural frontal and lateral radiographs of individual treated vertebral levels were obtained in patients that underwent vertebroplasty for osteoporotic and malignant Vertebral compression fracture (VCFs) (n = 84). Images of postprocedural vertebroplasty levels degraded by motion artefacts or with superimposition from significant cement extravasation or metallic implants were excluded (n = 16). A total of 76 individual vertebral levels were taken forward for analysis, which included 35 osteoporotic and 41 malignant treated VCF levels respectively. The frontal and lateral images were segmented and stored using stacked TIFF files. The images were pre-processed in python by applying a Contrast Limited Adaptive Histogram Equalization (CLAHE). Images were then manually segmented and following which, thresholding and binarization was performed to ensure that the treatment was standardized using the IsoData protocol (Figure 2). The fractal dimension and lacunarity values were calculated using the box counting and gliding box approach, respectively, using ImageJ (version 1.50c, National Institutes of Health, Bethesda, MD, USA), a widely used tool in image analysis, and the FracLac plugin.
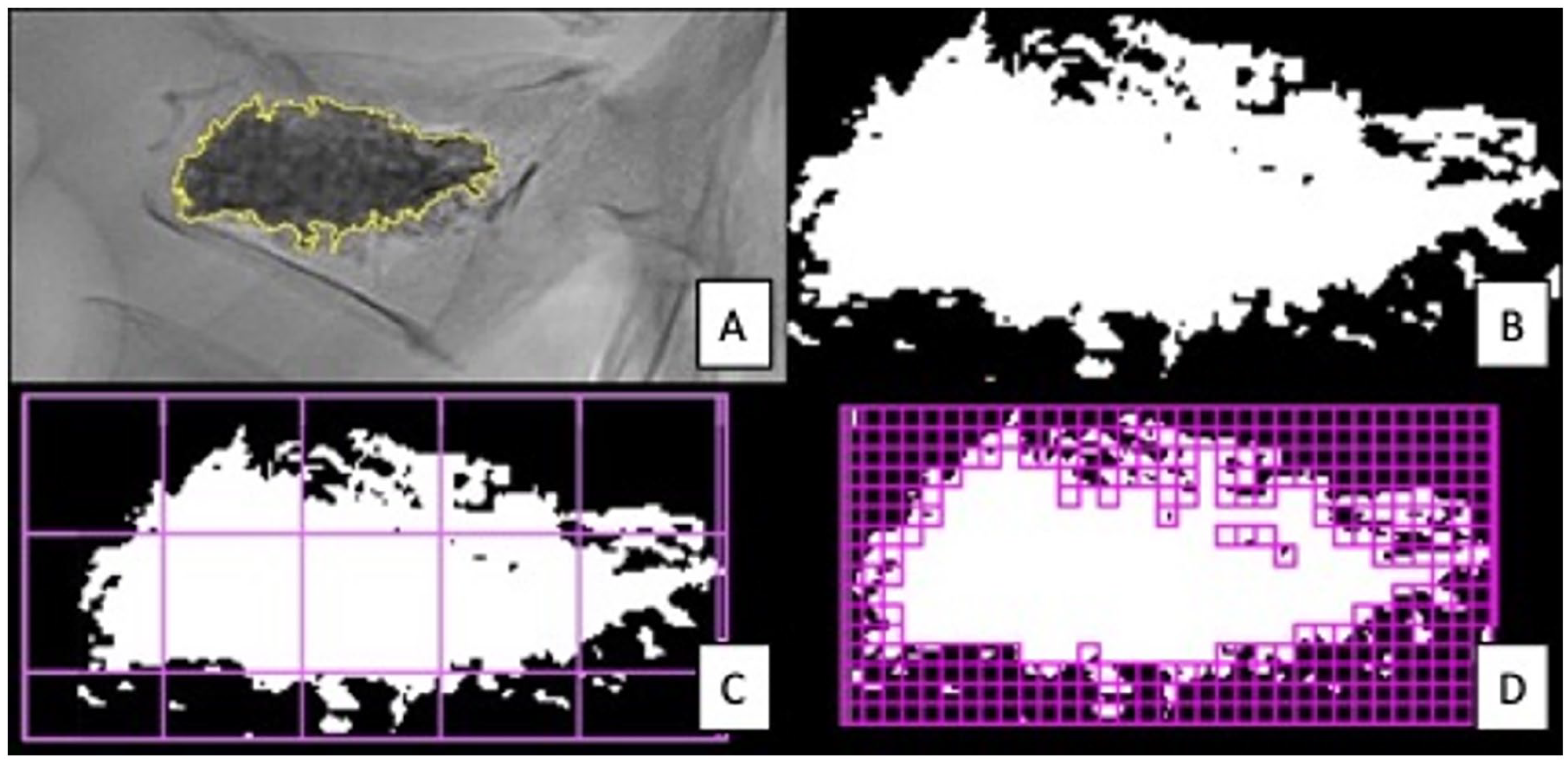
Image series demonstrating the fractal image analysis process of a vertebroplasty level. Lateral radiograph demonstrating segmentation of the region of interest (ROI) containing the cement distribution (A). Panel B demonstrates binarization of the segmented ROI. The binarized image is subjected to the box-counting method (C) which is subsequently processed with a smaller box width length “r” (D), increasing the number of boxes required to cover the image.
Fractal Dimension
The most common method of calculating FD is box-counting. 15 A complex geometry can be broken down into smaller parts, or boxes, and analyzed on a smaller scale (Figure 2). This technique views the binarized image by changing the box width of length “r,” which represents the size of the individual box used to cover the segmented area and is typically quantified in pixels, to inspect the image. The FD quantifies the complexity of a set across varying scales using the given formula:
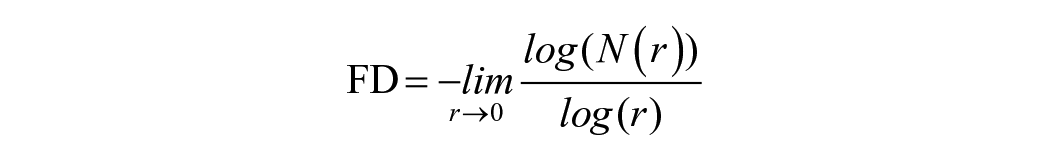
The size of the box is represented as “r” (r was set from 2:7), whereas “N(r)” represents the number of boxes needed to encompass the entirety of the image.
Lacunarity
Lacunarity is a counterpart to the FD which adds further textural information through analyzing the gap distribution at different scales. In the case of binary images, the gaps are defined as a specific foreground pixel value. The Gliding-Box technique developed by Allain and Cloitre is a common method to compute the lacunarity of an image by gliding boxes at different scales over the image. 24 The lacunarity at measure “r” can be defined as:
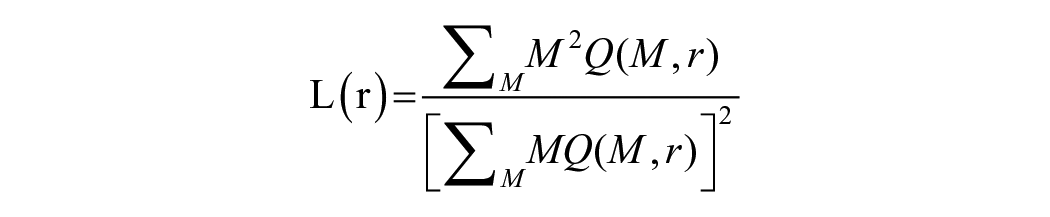
The number of gliding boxes is defined as n (M,r) with radius (r) and a box-mass (M) which is the number of occupied sites within each box. A mass distribution probability function, Q(M,r), is calculated as the boxes glide across the image.
Statistical Analysis
Statistical analysis was performed using SPSS, version 29.0 for MacOS (IBM SPSS Statistics, Armonk, NY: IBM Corp). Continuous variables were recorded as means (± standard deviation) or median (interquartile range, IQR) and subjected to two-sample t-tests and Mann-Whitney U tests respectively. Categorical data were reported as proportions (%) and subjected to a two-sided χ2 statistic. The confidence interval was set at 95% for all statistical analyses.
Results
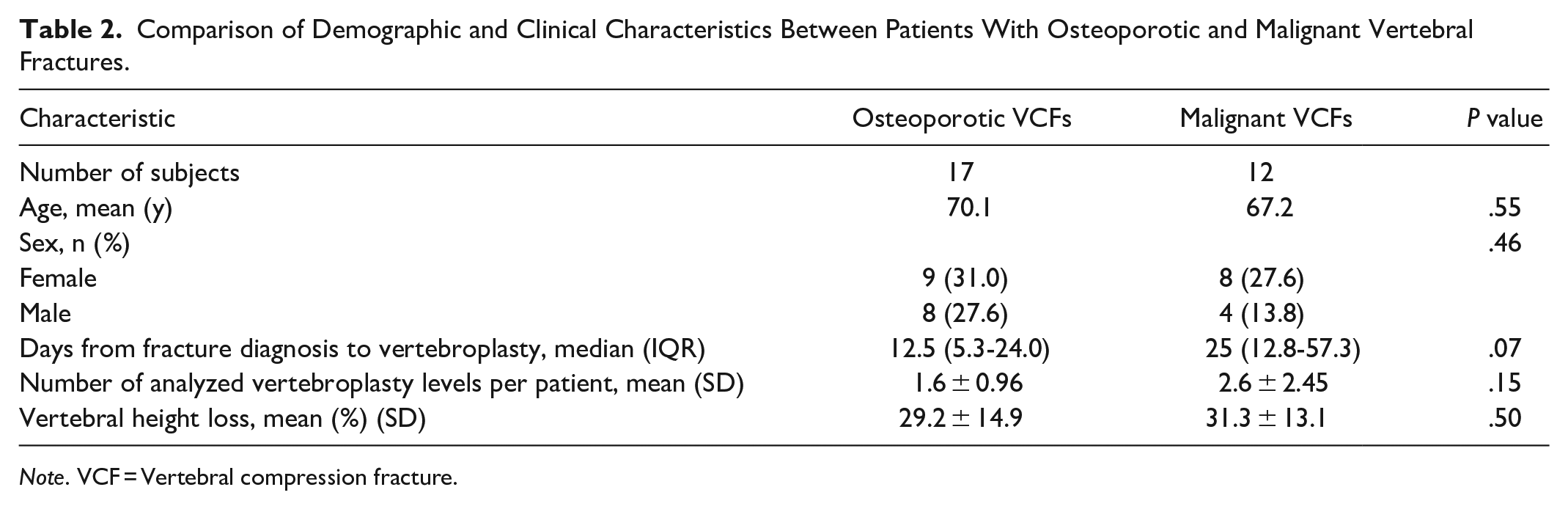
The baseline characteristics observed were similar in both the osteoporotic and malignant fracture vertebroplasty groups, with no significant difference in the age or sex between groups (Table 2). All the tumoural lesions were lytic in nature (n = 41, 100%). Most procedures were performed with a unipedicular technique (n = 60, 78.9%), in both osteoporotic (n = 21) and malignant groups (n = 39), which predominantly used 11G vertebroplasty needles (n = 53). A greater proportion of the treated osteoporotic VCFs (n = 14, 40.0%) were treated with a bi-pedicular technique compared to the malignant group (n = 2, 4.9%). There was no significant difference in the degree of vertebral height loss between fracture groups (P = .5). There was no significant difference in the mean PMMA volumes injected in the osteoporotic group (3.22 ml ± 1.56 ml) and the malignant group (3.22 ml ± 1.24 ml; P = .9) although cement volumes were recorded for 65.8% of the total number of treated vertebrae. Specifically, cement volumes were reported for approximately a quarter of osteoporotic levels (n = 9, 25.7%) but reported for all malignant levels (n = 41, 100%). The median number of days between fracture diagnosis and vertebroplasty was greater in the malignant vertebral fracture group (25 days; IQR 12.8-57.3) compared to the osteoporotic patients (12.5 days; IQR 5.3-24.0), although this did not reach statistical significance (P = .07).
Comparison of Demographic and Clinical Characteristics Between Patients With Osteoporotic and Malignant Vertebral Fractures.
Note. VCF = Vertebral compression fracture.
The results of fractal analysis of both the osteoporotic and malignant vertebroplasty groups are shown in Table 3. The mean FD of PMMA distribution following vertebroplasty was significantly higher in malignant VCF group (1.53 ± 0.08) compared to treated osteoporotic VCFs group (1.34 ± 0.17; P < .001). Similarly, the mean lacunarity of PMMA distribution was significantly greater in the malignant VCF group (0.50 ± 0.09) compared to the osteoporotic VCF group (0.37 ± 0.10; P < .001).
Comparison of Fractal Dimension and Lacunarity Values Between Osteoporotic and Malignant Vertebral Fractures.

Note. VCF = Vertebral compression fracture.
Discussion
Our study aimed to investigate and quantify the differences in PMMA cement filling patterns following vertebroplasty in patients with benign osteoporotic and malignant vertebral compression fractures using fractal analysis. We found that fractal dimension and lacunarity values of PMMA distribution post-vertebroplasty were significantly greater in patients with malignant compression fractures compared to osteoporotic fractures. To our knowledge, this study represents the first attempt to quantify cement distribution patterns using fractal analysis techniques.
The study findings are consistent with our understanding of the underlying pathological processes involved in both osteoporosis and malignant vertebral fractures, with fractal parameters of cement distribution appearing to be inherently related to the underlying architectural changes in both processes. The significantly greater fractal dimension values of PMMA distribution with malignant vertebral compression fractures indicates a greater complexity and roughness to the filling patterns when compared to osteoporotic vertebrae. In addition, significantly greater lacunarity values in the malignant fracture vertebroplasty group reflects both a larger and more heterogeneous arrangement of cavities with the vertebral body. These findings reflect the distinct microarchitectural changes of heterogeneous and irregular bone destruction in osteolytic bone tumours as compared to trabecular bone rarefaction in osteoporosis. 12 Further support of this comes from previous studies which quantified increased fractal dimension and lacunarity in myeloma and predicted fracture risk. 22 Overall, these findings may also help to explain and provide quantitative validation for the commonly observed patterns of PMMA distribution by interventionists with PMMA filling cavities in an irregular and heterogeneous patterns related to destructive malignant processes as opposed to a more evening dispersed or sponge-like filling pattern in osteoporosis.
This imaging tool while experimental may have potential clinical applications, such as the refinement of technical components of vertebral augmentation procedures. Specific intravertebral PMMA distribution patterns have been associated with specific adverse outcomes following vertebroplasty in osteoporotic patients including adjacent level fractures 10 and recompression fractures.7,8,25 Solid and compact cement distribution patterns have been associated with an increased rate of adjacent level fractures compared with more diffuse or sponge-like fill patterns in osteoporotic patients. 10 Similarly, another demonstrated that solid-filling or discontinuous-filling patterns were associated with an increase recompression fracture risk and height loss within treated vertebral bodies. 7 Although the insights from these studies are certainly useful, the cement filling patterns in these types of studies were classified based on visual scoring systems rather than quantitative metrics. 25 Furthermore, other studies have shown that greater filling in the vertical dimension is protective against recompression. 26 A combined metric utilizing fractal analysis and geometric-derived measurements such as vertebral volume fill fraction 9 may provide a more robust assessment of fill patterns, their association with potential adverse events such as recompression or adjacent level fractures, and determining a satisfactory procedural endpoint. This could be a potential subject for future clinical research. Further applications may also include optimization of operator-dependent factors during cement preparation and administration, or in the evaluation of novel polymers.27,28
While this study offers some insights into the applications of fractal analysis in PMMA distribution patterns, there are a few limitations which require acknowledgement when considering these results. The retrospective study design may have introduced unintended selection bias and limits the control over confounding variables. Exclusions of patients with vertebral levels with overlying metallic hardware may have contributed bias in this regard. Additionally, our study mostly focused on PMMA distribution and did not consider all possible factors which may influence study outcomes, such as bone mineral density which was not available for all our patients. While our results demonstrated a significant difference in fractal dimension and lacunarity values, a larger sample size would also improve the generalizability of this type of study. Specifically, the relatively small range of primary cancer diagnoses in the malignant VCF group, two thirds of which were related to multiple myeloma, may limit the generalizability of our results. A subgroup analysis by tumour type in the malignant group (solid vs haematological malignancies) was not performed as any conclusions drawn would likely be limited for the same reasons. The results may have been influenced by the injected cement volumes. The mean volumes were similar between both groups; however, cement volumes were unmeasured for approximately three quarters of the osteoporotic VCF group which may confound outcomes in this group. Prior radiotherapy in around a third of the malignant vertebroplasty levels may have conceivably affected cement distribution due to local effects on tumour viability and bone quality, although a portion of these irradiated vertebra (29.4%) underwent augmentation within 30 days post-radiation. Finally, not all treated malignant VCFs were pathologically proven with biopsy in our study, although the underlying pathology of the fractures were almost certainly secondary to either osteoporosis or malignancy based on unequivocal imaging findings and a known history of malignancy in relevant patients.
Conclusion
In conclusion, our study demonstrates that fractal analysis can differentiate cement spatial distribution patterns between osteoporotic and malignant vertebral compression fractures, with malignant vertebra demonstrating increased fractal dimension and lacunarity values. These findings are in keeping with our understanding of the underlying processes and changes in bone microarchitecture in both conditions. Furthermore, this study illustrates the technical implementation of fractal analysis and serves as a proof of concept of this novel imaging tool for quantifying cement distribution patterns following spine augmentation procedures. While this remains a novel tool for extracting textural features, the results warrant further investigation with testing of the presented methods on larger and more diverse datasets to verify our conclusions and further develop this technique.
Footnotes
Abbreviations
FA fractal analysis
FD fractal dimension
PMMA polymethylmethacrylate
VCF vertebral compression fracture
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
