Abstract
Cancer is the second most common cause of death worldwide. Bowel emergencies in patients with cancer are becoming increasingly more prevalent due to advances in cancer therapy and longer overall patient survival. When these patients present acutely, they are often frail and may have pre-existing co-morbidities. This article discusses the imaging features of bowel emergencies commonly encountered in oncological patients in clinical practice. These include chemotherapy related colitis, neutropenia enterocolitis and typhlitis, toxic megacolon, bowel perforation, malignant bowel obstruction and gastrointestinal haemorrhage. The radiologist plays a key role in identifying these oncological emergencies and guiding further management.
This is a visual representation of the abstract.
Introduction
Cancer is the second most common cause of death worldwide. 1 With a rapidly increasing elderly population, it is likely to surpass heart disease as the leading cause of death in the coming years. 2 A total of 1.27 million people are predicted to die due to cancer in the European Union alone in 2024. 3 Colorectal cancer is the second most common cause of cancer-related mortality worldwide.
Not only is our ageing population living longer, but people of all ages with cancer are living longer too. In the past, the primary care goals for patients diagnosed with advanced or metastatic disease were predominantly symptom control, pain palliation and ultimately, end-of-life care. 4 However, there are now multiple tools in the armamentarium of physicians treating patients with cancer, which can help to prolong their survival, thereby increasing overall cancer survival rates. 4 Curative treatments may be considered for those diagnosed with early-stage cancer. Patients with metastatic cancer, however, may undergo life-prolonging treatments. For example, chemotherapy has been found to prolong median overall survival for some colorectal patients by more than 2 years compared to supportive care alone. 1 Subsequently, for some, their cancer may cycle through periods of quiescence and recurrence for a considerable time.
The development of new systems of cancer management has seen the majority of cancer care transition to the out-patient setting. 5 As a result, those living with cancer are at risk of encountering acute issues outside of the hospital setting, thereby necessitating multiple visits to the emergency department. 6 Each year, in the United States of America alone, approximately 4 million visits are made to the emergency department (ED) by patients with advanced cancer. 7 Approximately 29% of all new cancer patients are diagnosed due to an acute presentation to the emergency department. 7 It has been reported that patients diagnosed with cancer in the emergent setting have lower overall 1-year survival rates compared to those diagnosed via elective routes, regardless of tumour stage at time of diagnosis. 8 Up to 40% of patients with cancer who present to the emergency department do so secondary to an acute abdomen. 9 Due to the longer overall survival of patients living with cancer, coupled with the increasing complexity of therapeutic agents, such as immune checkpoint inhibitors in patients with melanoma, acute presentations are being seen in already frail individuals with multiple pre-existing co-morbidities.7,10 The causality of acute care visits is broad, ranging from first presentation due to symptoms of an undiagnosed malignancy, progression of known malignant disease, recurrence of disease previously in remission and possible complications or side-effects of cancer therapy and oncological surgical interventions. 7
This article will review the common emergencies of the small and large bowel in cancer patients that are most frequently encountered in the ED, as well as the role of the radiologist in the setting of these acute cancer emergencies (Table 1).
Summary of Common Imaging Presentations in Bowel Emergencies.
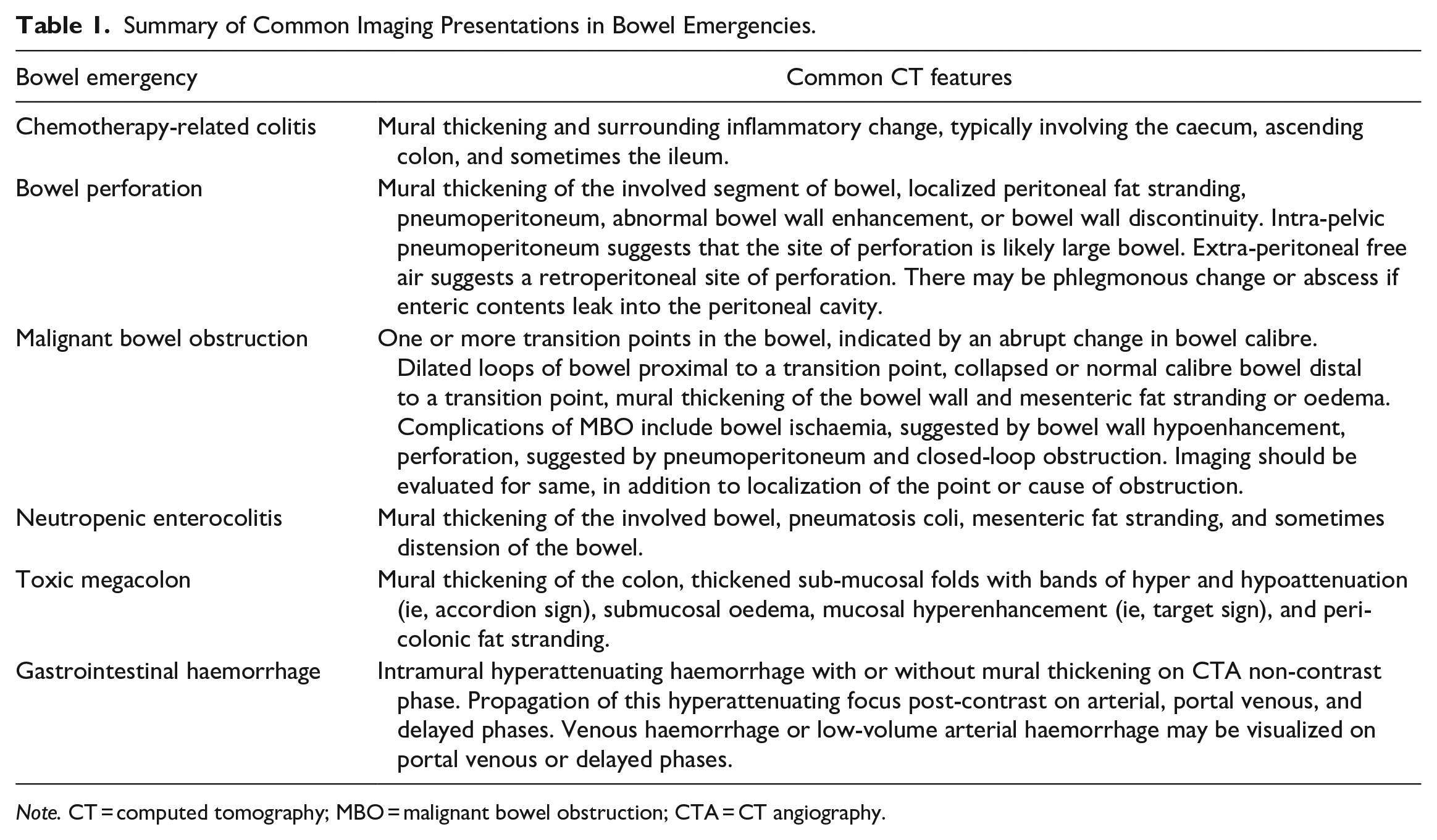
Note. CT = computed tomography; MBO = malignant bowel obstruction; CTA = CT angiography.
Diagnostic Imaging Modalities
Contrast enhanced computed tomography (CT) is the imaging modality of choice in the assessment of patients with cancer who present acutely to the ED with a suspected bowel emergency. 11 Traditionally, the use of oral contrast was deemed to be of benefit for CT examinations of the abdomen and pelvis, in particular if older-generation CT scanners were used to perform the study. 12 More recent studies have shown that intravenous ( IV) contrast alone can produce satisfactory diagnostic results.13,14 In addition, it must be noted that extraluminal leakage of oral contrast has not been found to be a particularly sensitive sign of bowel injury.15,16 Omitting oral contrast has numerous benefits. Firstly, it can expedite the pathway for the diagnosis and treatment of an acutely ill patient in the ED. 17 Oral contrast can require a delay of up to 90 minutes to achieve opacification of the distal bowel. 18 Secondly, for the evaluation of mucosal enhancement in cases where bowel viability, bowel inflammation, or hyperenhancing tumours are in question, the use of intraluminal contrast would limit the diagnostic ability of the study.19,20
In addition, the use of oral contrast can pose an aspiration risk in patients who are vulnerable to same. 17 Normally, IV contrast enhanced CT is performed in the portal venous phase, approximately 60 to 80 seconds post contrast material injection. 21 This is usually sufficient for the imaging of hypovascular tumours and hypovascular metastatic disease. For the detection of hypervascular heaptic tumours or hypervascular hepatic metastatic lesions, multi-phase imaging (arterial and portal venous phases) is required to demonstrate arterial phase enhancement and subsequent washout on portal venous phase images. 21 Unenhanced CT can be performed in patients with acute kidney injury, chronic kidney disease, or iodinated contrast allergy. While generally considered a sub-optimal investigation, nonetheless, unenhanced can be of diagnostic value and particular attention should be paid to its interpretation. 22
Chemotherapy-Related Complications
Chemotherapy has long been a treatment option for select patients with malignancy. The imaging appearances of chemotherapy-related toxicities, however, are extremely broad and can vary depending on the stage of malignancy and the timing or recency of chemotherapy administration. 23 Toxicity is also one of the main factors that limits chemotherapy use. 24 Thus, it is important for radiologists to be aware of how they may present. Classically, cytotoxic chemotherapeutic agents act on rapidly proliferating cells in order to induce cell death. As a result, not only are malignant cells affected, but healthy cells with a short cell-cycle undergoing rapid cellular division, such as those in the gastrointestinal (GI) tract, can also be affected. 24 In recent decades, therapies that target specific phases of tumour growth, such as apoptosis evasion, angiogenesis, and insensitivity to growth-inhibition signals, as well as monoclonal antibodies, have been developed. 24 Similarly, however, healthy cells that express the molecular target of interest can also be injured, leading to adverse effects.
Toxic complications of chemotherapeutic agents involving the GI tract occur relatively frequently. 23 Common causative agents include taxanes, methotrexate, alkylating agents, vascular endothelial growth factor receptors tyrosine kinase inhibitors (TKIs), and anti-angiogenic TKIs. 23 The most common type of colitis secondary to taxane therapy is ischaemic colitis, however, haemorrhagic colitis and neutropenic enterocolitis (or typhlitis) in immunocompromised patients have also been reported.25,26 Patients with melanoma may be subjected to acute colitis when they receive immune checkpoints inhibitors. In this regard, colitis is observed in 1.4% of patients with metastatic melanoma receiving nivolumab and in 19% of those receiving ipilimumab. 10 CT appearances of colitis often demonstrate mural thickening and surrounding inflammatory change, typically involving the caecum, ascending colon, and sometimes the ileum (Figures 1 and 2). 23
Seventy-year-old woman with stage IIIC cutaneous melanoma with complete response. The patient presented with acute abdominal pain and severe diarrhoea while receiving a fourth session of nivolumab. (A) CT image in the axial plane shows mild thickening and mucosal hyperaemia of sigmoid colon (arrows). (B) CT image in the coronal plane shows mucosal hyperaemia involving sigmoid colon, descending colon, and also ascending colon (arrows). (C) 18F-FDG positron emission tomography-CT image in the axial plane shows avid uptake of 18F-FDG by sigmoid and descending colon (arrows). Colonoscopy and endoscopic biopsy confirmed colonic inflammation and mucosal injury consistent with nivolumab-associated colitis.
Sixty-one-year female on Sunitinib for RCC presents with diarrhoea and acute pain. CT exam shows marked thickening of the large bowel (arrowheads, A, B, and C) extending from the distal transverse colon to the proximal sigmoid colon, highly suspicious for ischaemic colitis.
Neutropenic Enterocolitis
Neutropenic enterocolitis, or typhlitis, is a potentially life-threatening complication of chemotherapy. Mucosal injury secondary to a chemotherapeutic agent, combined with neutropaenia and associated immunosuppression can leave an individual vulnerable to microorganism invasion, typically bacterial or fungal. 27 The presence of bacteraemia, fungaemia, or hypotension is associated with poorer prognosis in these patients. 27 Neutropenic enterocolitis classically presents with a clinical “triad” of fever, abdominal pain, and diarrhoea. 27 Neutropenic enterocolitis commonly involves the caecum and in such cases is referred to as typhlitis. 27 CT features can include mural thickening of the involved bowel, pneumatosis coli, mesenteric fat stranding, and sometimes bowel distension. 28 Treatment of neutropenic enterocolitis involves broad-spectrum antibiotic therapy, nasogastric tube decompression, and nutritional supportive therapy.27,28 Surgery in neutropenic patients is associated with 30-day mortality rates of up to 30% and should, therefore, be considered only in cases of perforation with peritonitis or haemodynamic instability, or if deemed necessary, until neutrophil counts improve. 27
Bowel Perforation
One of the most severe and potentially life-threatening complications of enterocolitis is perforation. 24 GI tract perforation may also be secondary to a small or large bowel tumour. In general, the main factors leading to GI tract perforation are ischaemia and necrosis. 29 Necrotic degeneration of neoplastic tissue, vascular invasion by tumour cells and luminal distension secondary to obstruction can all contribute to bowel ischaemia and subsequent perforation. 29 Bowel perforation can present with a constellation of symptoms, including abdominal pain, rebound tenderness, guarding secondary to peritonitis, nausea, vomiting, and diarrhoea, which may be bloody. Signs and symptoms of sepsis are often present and are most commonly due to gram-negative enteric bacteria in the peritoneal space. 24
When combined with chemotherapy, the anti-angiogenic TKI bevacizumab has been found to significantly improve progression-free and overall survival in patients with metastatic colorectal cancer.30,31 However, this combination has also been associated with GI tract perforation in approximately 2% of patients. 32 The majority of perforations were found to occur within the first 3 months after therapy initiation, with the highest rates of perforation seen in those with an intact primary tumour, recent intervention such as colonoscopy or sigmoidoscopy or prior pelvic radiotherapy.24,32
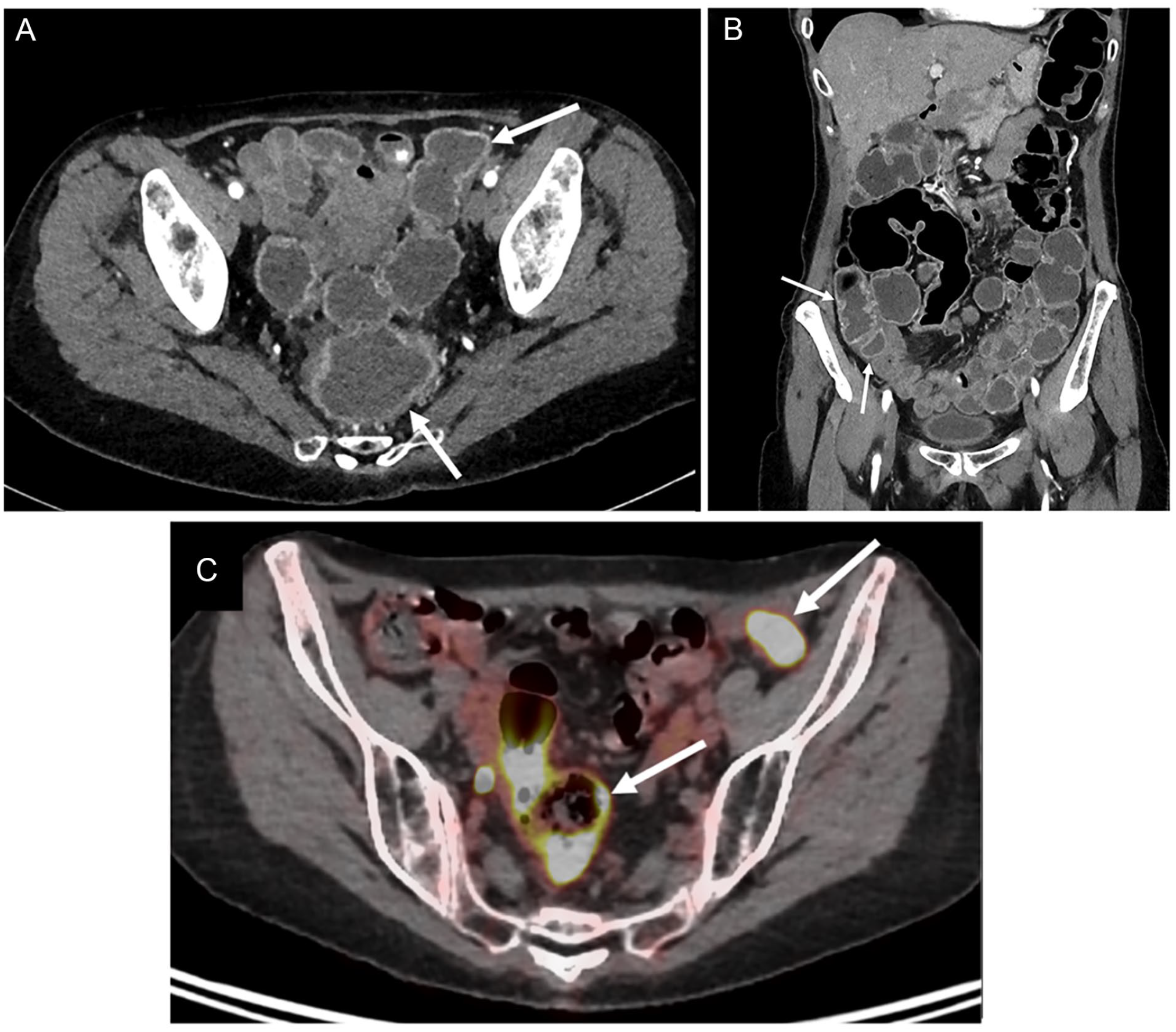
CT of the abdomen with intravenous (IV) contrast is the imaging modality of choice for the investigation of suspected GI tract perforation, which shows 85% accuracy for the identification of the perforation site.33,34 CT features of bowel perforation may include mural thickening of the involved bowel segment, localized peritoneal fat stranding, pneumoperitoneum, abnormal bowel wall enhancement, or discontinuity of the bowel wall (Figure 3). 34 If the pneumoperitoneum is located in the pelvis, the site of perforation is likely to be large bowel, whereas extra-peritoneal free air is suggestive of a retroperitoneal site of perforation. 34 If there is extra-luminal leakage of enteric contents, there may be associated phlegmonous change or abscess (Figures 4 and 5). 34
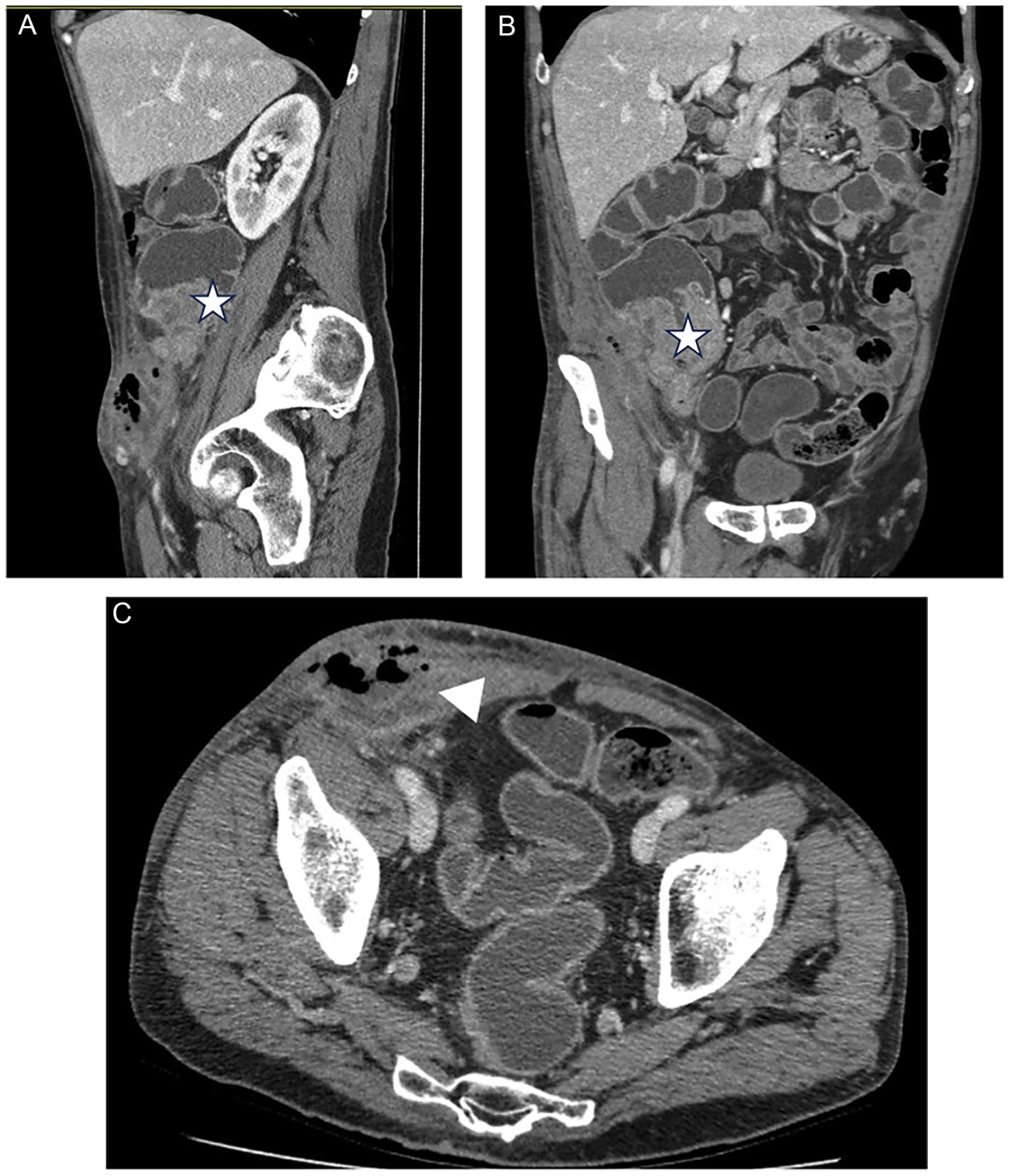
Sixty-one-year male with primary cecal malignant mass (star, A and B), causing frank perforation of the bowel wall. Inflammatory changes involve primarily the abdominal wall, with secondary myositis and abscess formation in the right lower abdominal musculature (arrowhead, C).
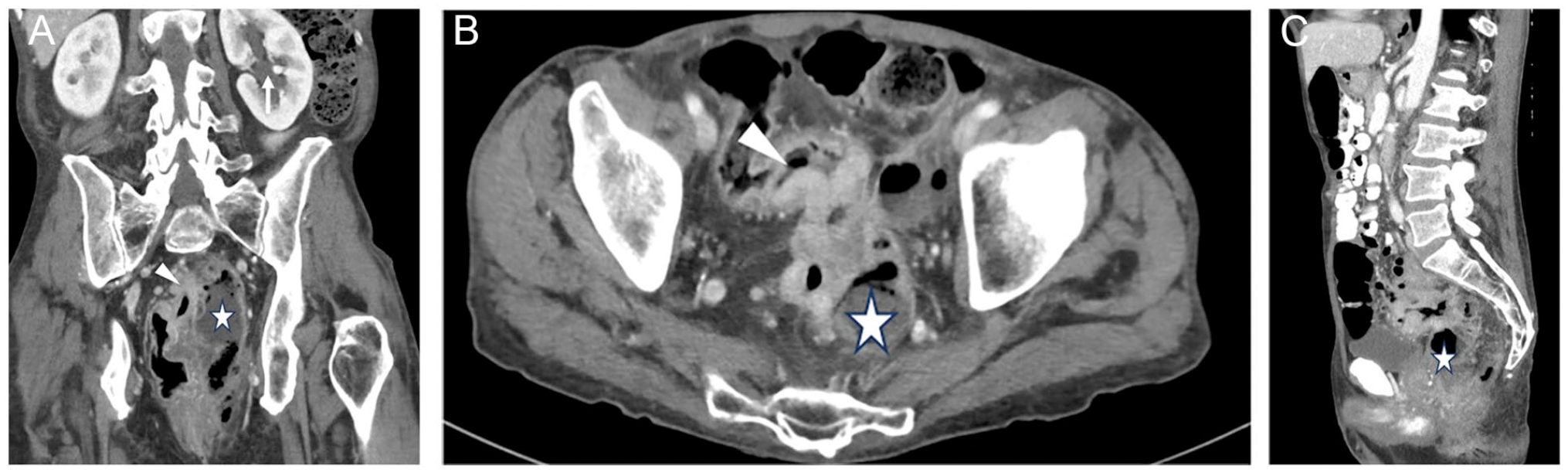
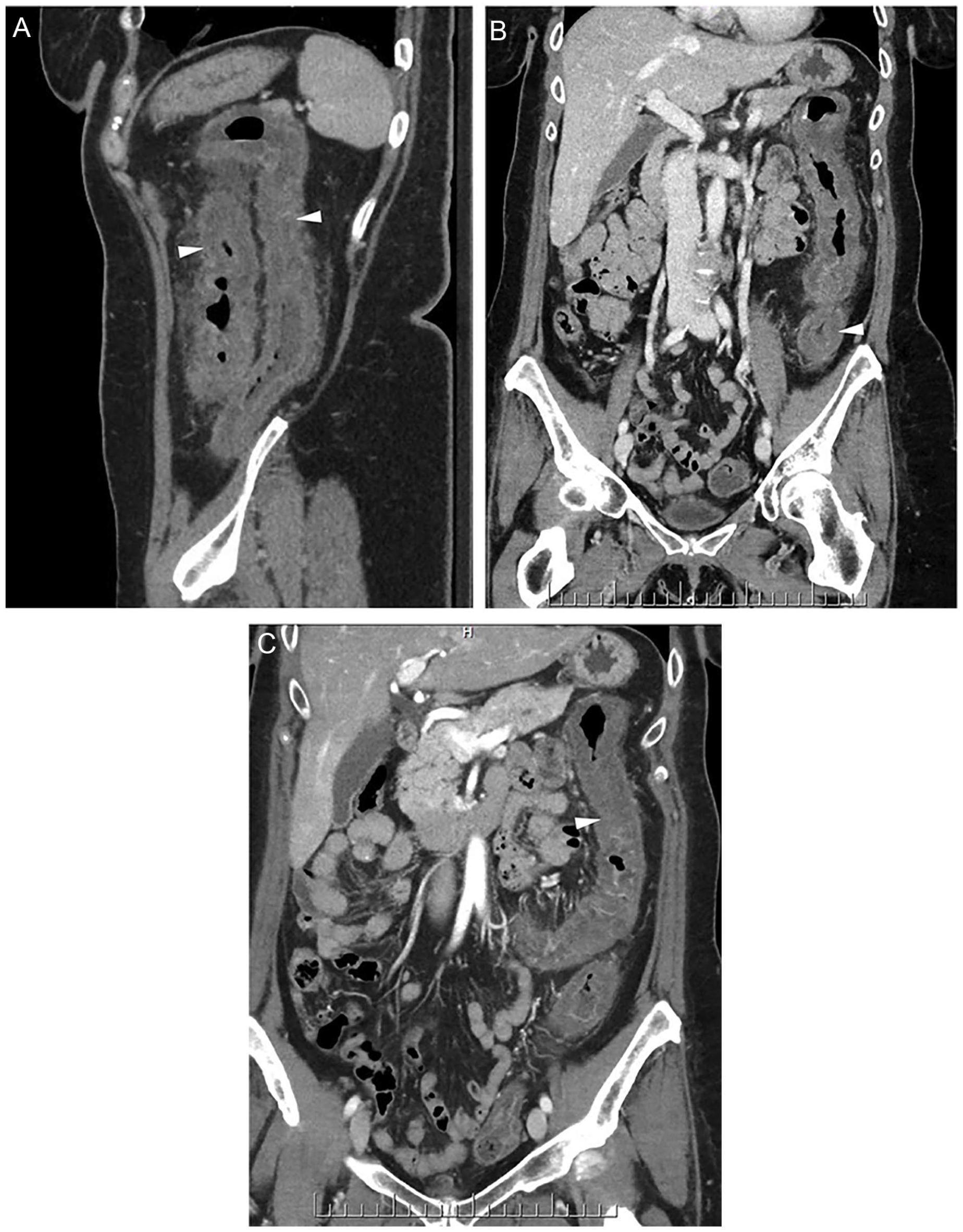
Sixty-six-year male with a perforated cancer (arrowhead, A and B) of the rectosigmoid junction with multiple pelvic abscesses (star, A, B, and C), one of them causing mild left hydroureteronephrosis (arrow, A).
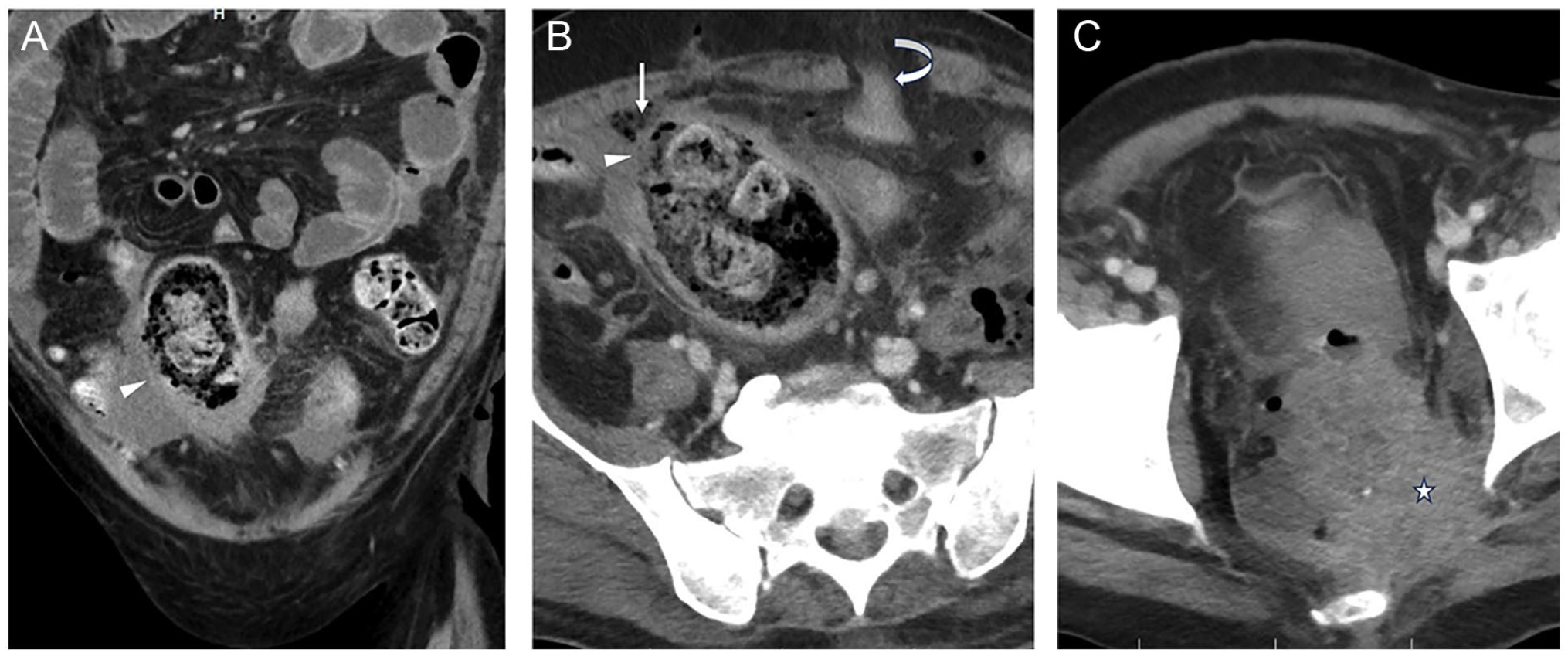
Sixty-one-year male with known rectal malignancy presents with acute abdomen. CT exam shows a large infiltrative lower ano-rectal cancer (star, C) with upstream bowel obstruction and segment of diminished enhancement along the anterior wall of the distal sigmoid colon with a defect (arrowhead, A and B) within the anterior wall close to the rectosigmoid junction. Adjacent pockets of gas and fluid are seen with loculated contents within the peritoneal cavity (straight arrow, B). Defunctioning loop colostomy in the left lower quadrant (curved arrow, B).
Treatment of a suspected bowel perforation in patients with cancer involves supportive care, including IV access, nasogastric tube decompression, nil-per-mouth (cessation of oral intake), haemodynamic support if signs of haemodynamic instability or sepsis are present, and broad-spectrum IV antibiotics. 35 In the haemodynamically stable patient with a locally contained perforation and no signs of peritonitis, non-operative management with observation and IV antibiotics may be continued in conjunction with surgical consultation. 36 If the locally contained perforation is amenable to percutaneous drainage, then consultation with interventional radiology may be considered.35,37 However, should symptoms of sepsis or peritonitis develop despite conservative or interventional radiology management, surgical intervention becomes a vital consideration. Surgical intervention in the acutely ill patient with advanced cancer is an increasingly common challenge faced by surgeons due to higher prevalence of advanced cancer and longer survival rates in these patients. 38 Ideally, multidisciplinary discussion and consultation with both the patient and their family is important, however, the urgency of the situation may not lend itself to same. 38 In the emergent setting of bowel perforation, the primary aim of surgical intervention is to achieve source control in order to manage sepsis. In the case of perforation secondary to GI tract tumours, source control commonly takes precedence over achieving complete tumour and lymph node resection. 38
Malignant Bowel Obstruction
Malignant bowel obstruction (MBO) is a common complication encountered by patients living with advanced cancer. It is estimated to occur in up to 28% of patients with cancer of the GI tract and up to 51% of patients with advanced ovarian malignancy. 39 In 2007, an international clinical protocol committee defined MBO as (i) clinical or radiological evidence of bowel obstruction, (ii) bowel obstruction distal to the ligament of Trietz, (iii) non-curable intra-abdominal cancer, or (iv) extra-abdominal cancer with peritoneal disease. 40 MBO is typically classified as mechanical or functional. Mechanical causes include extrinsic compression of the bowel secondary to intra-abdominal masses, adhesions, peritoneal or omental disease, and intra-mural or intra-luminal obstruction due to cancer growth. 39 Functional MBO results from dysmotility secondary to tumour infiltration of the enteric neural plexi or the mesentery. 39 Additional factors may worsen MBO, including medications (opioids, anticholinergics, intraperitonal chemotherapy), post-surgical adhesions, and post-radiotherapy fibrosis. 39 Another cause of malignant bowel obstruction is intussusception. This typically happens in patients with small bowel metastases from melanoma, in whom polypoid metastases may act as leading points for intussusception (Figure 6). 41
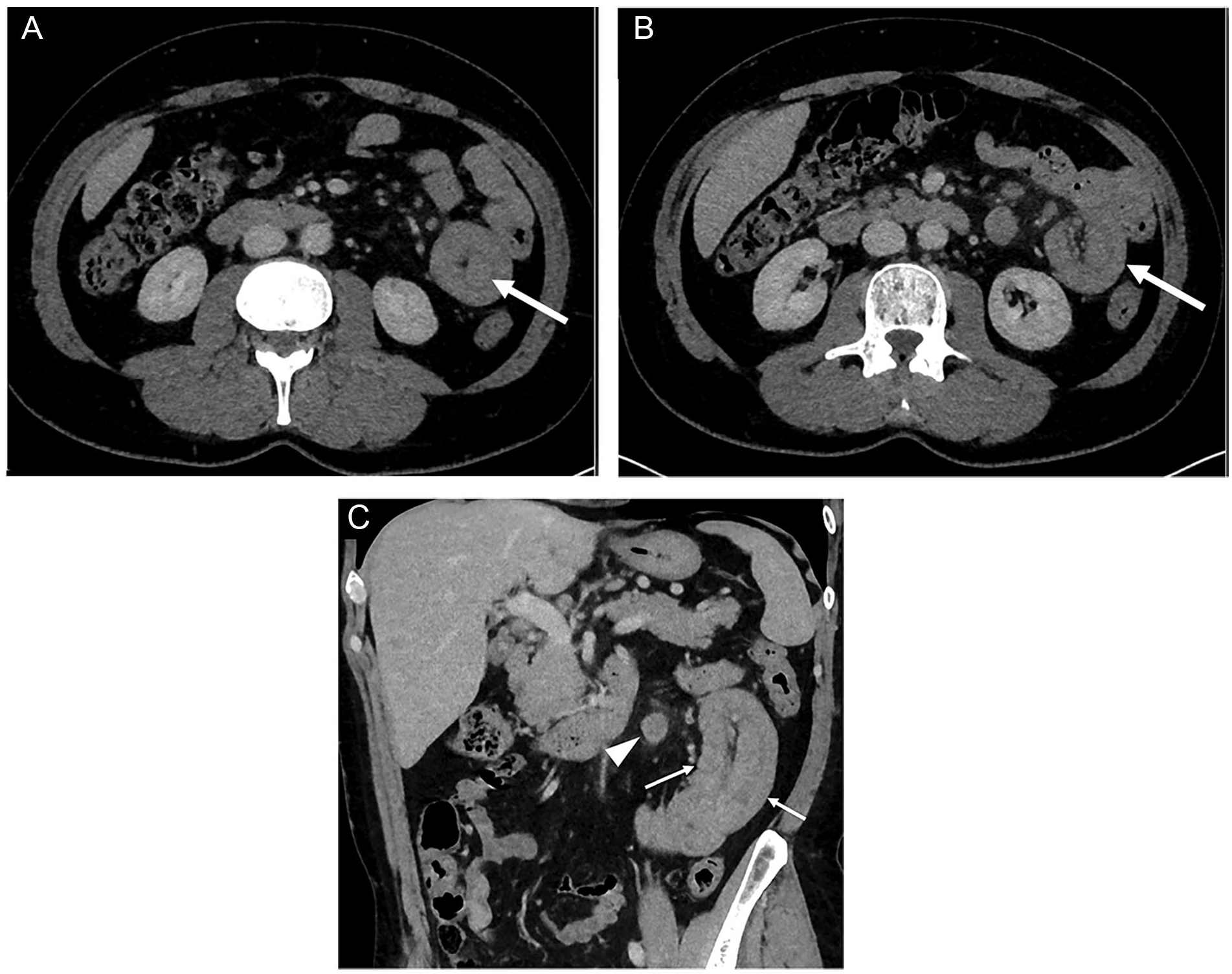
Sixty-two-year-old man with stage IV cutaneous melanoma presenting with abdominal pain and vomiting. The patient has no ongoing treatment. (A) CT image in the axial plane reveals jejunal mass (arrow) displaying aneurysmal pattern. (B) At a different level of slice, CT image in the axial plane shows marked circumferential thickening of a jeunal loop (arrow). (C) CT image in the coronal plane reveals typical pattern of jejunal intussusception (arrow) and adjacent metastatic lymph node (arrowhead). Surgery confirmed small bowel metastasis from melanoma.
MBO can occur in the large and small bowel, however, MBO of the small bowel is most common. 39 Symptoms generally include abdominal distension, abdominal discomfort, nausea, vomiting, obstipation, lack of flatus passage and, sometimes, overflow diarrhoea. 39 Malnutrition, raised C-reactive protein, leukocytosis, and hypoalbuminaemia are associated with poorer prognosis in patients with MBO. 39
Common findings on contrast enhanced abdominal CT include one or more transition points where there is an abrupt change in bowel calibre, dilated loops of bowel proximal to the transition point, collapsed or normal calibre bowel distal to the transition point, mural thickening of the bowel wall and mesenteric fat stranding or oedema.42,43 It is of vital importance to assess the viability of the involved bowel and to interrogate the images for the presence of any possible complications of MBO, such as bowel wall hypo-enhancement, suggestive of ischaemia, pneumoperitoneum, indicative of perforation, or closed-loop obstruction.42,43
Treatment of MBO consists of supportive care including IV hydration and nutritional therapy, often parenteral. 39 Temporary decompression for symptom relief may be achieved with nasogastric tube placement and anti-secretory medications may also be used. 44 Long-term decompression of gastric contents has been achieved with placement of an endoscopic or percutaneous gastrostomy tube. 39 Endoscopic stenting may also be a consideration. 45 Palliative surgery for MBO may be of benefit, however, surgical intervention should be evaluated on an individual patient basis due to high risk of post-operative morbidity and mortality.44,45 Surgical intervention for MBO can include tumour resection if feasible, bowel resection or proximal diversion and decompression with stoma formation.45,46
Pseudomembranous Colitis and Toxic Megacolon
Pseudomembranous colitis is a form of infectious colitis and is a common cause of nosocomial diarrhoea, secondary to antibiotic therapy. 26 Patients with cancer are at an increased risk of developing this condition and are found to have higher rates compared to the non-oncologic hospital in-patient population. 47 Broad-spectrum antibiotics are the most common causative agents. 26 Antibiotic therapy disturbs the equilibrium of the native intestinal flora, allowing Clostridium Difficile, a gram-negative, spore-forming bacteria, to colonize the GI tract. Cytotoxins and enterotoxins are released by Clostridium Difficile, which damage the integrity of the colonic mucosa and eventually lead to intestinal ulceration and necrosis. 26 This gives rise to the “pseudomembranes” seen on the colonic mucosa during endoscopic evaluation.26,48
Toxic megacolon is a potentially life-threatening complication of non-infectious and infectious colitis, including pseudomembranous colitis. 48 Rarely, cases of toxic megacolon have been reported with the use the antimetabolite methotrexate.24,49 Folate supplementation has been found to be effective in alleviating GI tract symptoms related to methotrexate.24,49 When the colitis inflammatory process extends into the colonic smooth muscle, ganglion cells in the colonic mucosa are injured when the inflammatory process extends into the colonic smooth muscle layer, which results in impaired peristalsis and progressive colonic dilatation. 48 Often, clinical signs of sepsis are also present. 48
CT features of pseudomembranous colitis include pericolonic fat stranding, mural thickening of the colon with hyperenhancement of the mucosa, and hypoenhancement of the submucosa secondary to oedema (ie, target sign). 26 The appearance of the hyperaemic, hyperenhancing mucosa, and the thickened, oedematous submucosal folds can resemble the folds of an accordion (ie, accordion sign). 26 Toxic megacolon is characterized by segmental or total non-obstructive dilatation of the large bowel equal to or greater than 6 cm.26,48 Colonoscopy is not used as an investigation of choice in the acute setting due to a high risk of colonic perforation. 48
Gastrointestinal Haemorrhage
Acute gastrointestinal haemorrhage (GIH), classified as haemorrhage into the lumen of the GI tract, can often be the first presentation of a GI tract cancer. 50 Acute haemorrhage can be caused by a number of factors, including tumour-bleeding secondary to tumour hypervascularity and abnormal tumour vasculature, tumour invasion of intra-abdominal vasculature and also as a complication of surgery or endoscopic intervention. 51 Acute GIH can also be induced by anti-tumour chemotherapy and radiotherapy. 51 For example, alkylating agents such as cyclophosphamide have been associated with incidences of haemorrhagic colitis, which terminated post discontinuation of therapy. 24 Exacerbating factors may include adjunct therapies, such as non-steroidal anti-inflammatory use, anti-coagulants, and thrombocytopaenia induced by malignancy or anti-cancer therapy. 51
In the setting of acute upper GIH (proximal to the ligament of Treitz) in the haemodynamically unstable patient, endoscopic therapy is the intervention of choice. 52 For acute lower GIH (distal to the ligament of Treitz), in haemodynamically unstable patients presenting with either haematochezia or melaena, colonoscopy is the diagnostic and therapeutic intervention of choice. 53
In the emergency setting, multiphase CT angiography is commonly the imaging modality of choice for suspected GIH. 52 CT features of acute GIH include intraluminal or intramural hyperattenuating haemorrhage on the non-contrast phase, which may also be associated with mural thickening.52,54 Subsequent enlargement or change in shape of this hyperattenuating focus on arterial or portal-venous imaging can help to confirm the presence of active GIH. 54 Portal venous phase imaging should also be scrutinized to assess for venous haemorrhage or low-volume arterial haemorrhage not immediately apparent on arterial phase imaging. 54 In the absence of active contrast extravasation, identification of abnormalities of the bowel wall, intra-abdominal vasculature, or progression of malignant disease can aid in the localization of potential sources of GIH. 54 CT angiography is reported to have a sensitivity of up to 90% in identifying the location of an active GIH. 55 However, in the setting of intermittent bleeding, sensitivity of CT angiography falls to approximately 45%. 55 Catheter angiography via interventional radiology can be used as both a diagnostic and therapeutic modality, especially in refractory cases of GIH, however, it is not commonly used as a first line investigation. 55
Conclusion
Bowel emergencies in patients with cancer are becoming increasingly more prevalent due to advances in cancer therapy and longer overall patient survival. When these patients present acutely, they are often frail and may have many pre-existing co-morbidities. Furthermore, surgical intervention in patients with advanced cancer is an increasingly common challenge faced by surgeons and warrants multidisciplinary discussion where possible, as well as patient and family consultation, to determine the appropriateness of same. The role of the radiologist in the timely diagnosis of bowel emergencies in cancer patients is crucial to ensure prompt and appropriate management in the acute setting.
Footnotes
List of Abbreviations
CT computed tomography
GI gastrointestinal
GIH gastrointestinal haemorrhage
IV intravenous
MBO malignant bowel obstruction
TKI tyrosine kinase inhibitor
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.H. is the Editor-in-Training of Canadian Association of Radiologists Journal. M.N.P. is the Editor-in-Chief of Canadian Association of Radiologists Journal.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
