Abstract
Purpose:
Prior studies have described complications of radiofrequency ablation (RFA) of liver tumours. The aim of this study was to identify risk factors for hospitalization duration longer than 24 hours following RFA of liver tumours.
Methods:
This retrospective, single-centre study included patients with liver tumours undergoing RFA between October 2017 and July 2020. Medical records were reviewed to collect patient, tumours, and procedure characteristics for each RFA session. The association between potential risk factors and duration of hospitalization (less than or more than 24 hours) was analyzed using univariate and multivariate logistic regressions.
Results:
Our study included 291 patients (mean age: 65.2 ± 11.2 [standard deviation]; 201 men) undergoing 324 RFA sessions. Sixty-eight sessions (21.0%) resulted in hospitalization of more than 24 hours. Multivariate analysis identified each additional needle insertion per session (OR 1.4; 95% CI [1.1-1.9]; P = .02), RFA performed in segment V (OR 2.8; 95% CI [1.4-5.7]; P = .004), and use of artificial pneumothorax (OR 14.5; 95% CI [1.4-146.0]; P = .02) as potential risk factors. A history of hepatic encephalopathy (OR 2.6; 95% CI [1.1-6.0]; P = .03) was only significant in univariate analysis. Post-hoc, subgroup analysis of patients with hepatocellular carcinoma (69.8%) did not identify other risk factors.
Conclusion:
Risk factors for a hospitalization duration longer than 24 hours include a higher number of needle insertions per session, radiofrequency ablation in segment V, and use of an artificial pneumothorax.
Introduction
Liver resection is one of the main curative options in patients with liver tumours, yet surgical morbidity remains an important clinical concern. 1 More recently, image-guided percutaneous radiofrequency ablation (RFA) has become a common approach in the treatment of small primary or metastatic liver tumours especially in patients not fit for surgical resection or with poor functional hepatic reserve. 2 Other indications for liver RFA include bridge treatment of unresectable hepatocellular carcinoma (HCC) prior to liver transplantation and treatment of HCC recurrence. 3 It is also effective for treatment of liver metastases from different primary cancers. 3 Ablation procedures typically involve inserting an electrode (needle-like device) under image guidance into a target area, and delivering energy through a generator to induce coagulative necrosis. 2 It is considered a minimally invasive technique, with satisfactory local tumour control, technical ease, and safety in appropriately selected patients. 4
RFA requires shorter hospital stays and has lower complication rates compared to hepatic resection.5-10 Depending on the institution, patients are typically kept under observation for a few hours following the procedure or hospitalized overnight for surveillance of potential complications. The median duration of posttreatment hospital stay has been reported to vary between one to a few days, while the range is highly variable depending on the incidence of complications.8-11 A large multicentre study has reported major and minor complications rates of 2.2% and 4.7%, respectively, with the major periprocedural complications being intraperitoneal hemorrhage, intrahepatic abscess, and gastrointestinal tract perforation. 12 Another study reported liver failure as the most severe complication and associated with the highest mortality. 13
Few prior studies have identified risk factors for complications following RFA of liver tumours.1,12,14-22 However, some of them have only evaluated specific complications, such as hemorrhagic complications, 17 haemobilia, 18 liver abscess,15,20,21 and cholangitis. 20 Moreover, some studies had relatively small sample size14,18 and some have been published more than 10 years ago.12,14,15,17,20 Although complications influence length of stay, other factors may also have an impact. To our knowledge, no study has formally evaluated risks factors affecting length of stay as the primary outcome, which is an important information in order to optimize planning of hospital beds.
Therefore, the purpose of this study was to identify risk factors for hospitalization duration longer than 24 hours following percutaneous RFA of liver tumours.
Methods
Study Design
This was a retrospective, single-centre study of clinical and imaging data in adult patients undergoing RFA of liver tumours. This study was approved by the institutional review board of the Centre Hospitalier de l’Université de Montréal (CHUM), Montréal, Canada. Written informed consent was waived due to the retrospective study design.
Patient Selection
The study flowchart is shown in Figure 1. Consecutive subjects were included if they were an adult undergoing a percutaneous procedure for ablation of a liver tumour between October 2017 and July 2020. All patients were referred through multidisciplinary tumour boards at our institution. Subjects were excluded if they underwent: (1) ethanol injection, (2) microwave ablation, (3) cryoablation alone, or (4) transarterial chemoembolization before the RFA session during the same hospitalization. For subjects who underwent 2 or more RFA sessions at different dates, each session was considered an independent event since tumours and technique varied from one session to another for the same patient. Sample size was based on convenience sample of patients responding to eligibility criteria.
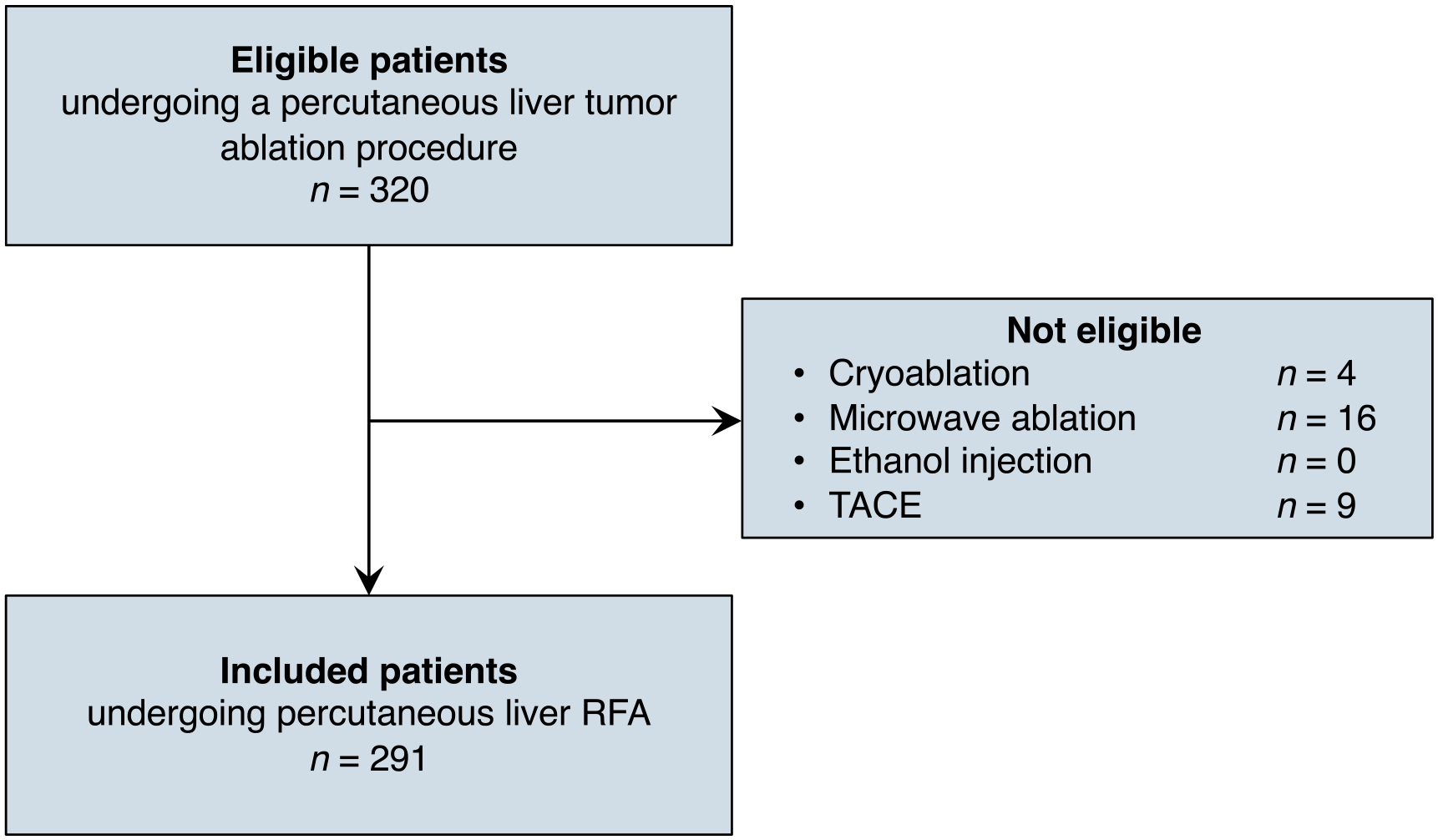
Study flowchart of patient selection.
Diagnosis of Liver Tumours
Confirmation of the diagnosis of liver tumours was made histopathologically via image-guided percutaneous biopsy of the tumour prior to the RFA procedure or on recent imaging by CT and/or MRI combined with clinical presentation if no biopsy was performed. Hepatocellular carcinomas (HCC) with a Liver Imaging Reporting and Data System (LI-RADS) 4 or 5 category were considered eligible for RFA treatment.23,24
RFA Procedure
RFA was performed using a percutaneous approach by fellowship-trained abdominal or interventional radiologists (J-S.B., 15 years of experience and D.O., 21 years of experience). Patients were under general anaesthesia for all the procedures. Various internally cooled applicators were used depending on tumour size (RFA Ablation Electrode kit E series or RF Ablation Cluster Electrode Kit E Series, Cool-tip™ Covidien, Medtronic, Dublin, Ireland; 2- or 3-cm active tip monopolar or 2.5-cm active tip cluster; 17 G gauge size; with different lengths depending on the depth of the tumour [from 15 to 25 cm] and various number of electrodes depending on the sizes and the number of the tumours). Some subdiaphragmatic tumours were accessed through a transthoracic extrapulmonary transdiaphragmatic approach with the creation of an artificial pneumothorax. Aero-dissection was performed with insertion of a Veress needle (15 cm, Conmed®, Largo, USA) in the pleural cavity anteriorly near the hepatic dome followed by controlled injection of CO2. All patients were given prophylactic antibiotics.
Energy was applied for 4 to 28 minutes using a 200-W power generator using a feedback algorithm that continuously monitored tissue impedance and automatically adjusted output to maximize energy delivery in order to obtain an ablative margin of 5 to 10 mm at the end of the session. Procedure was performed under ultrasound and/or CT guidance without intravenous contrast injection if the tumour was spontaneously visible and with intravenous contrast injection if not. Monitoring during the energy application was made by ultrasound and/or CT. To limit bleeding and tumour seeding, cauterization of the electrode tract was always performed during withdrawal of the applicator. Once the procedure was completed, axial non-enhanced CT was obtained for each patient to assess immediate treatment response and possible complications.
Data Collection
The duration of hospitalization post-RFA was collected for each session and dichotomized into 2 groups: less than 24 hours or more than 24 hours.
To identify risk factors for hospitalization longer than 24 hours, the following patient, tumour, and procedure characteristics were collected for each RFA session: age, sex, histology (eg, HCC, metastasis, adenoma, hemangioma, and cholangiocarcinoma), history of cirrhosis, ascites, hepatic encephalopathy, portal hypertension, high blood pressure, diabetes, chronic obstructive pulmonary disease, cardiac valvular disease, heart failure, aortic stenosis, atrial fibrillation, coronary artery disease, etiology of chronic liver disease (hepatitis B, hepatitis C, hepatitis D, alcohol-related, and nonalcoholic steatohepatitis), Child-Pugh class, number of ablated tumours per session, pre-RFA laboratory results (serum alpha-fetoprotein, platelet count, serum total bilirubin, serum albumin, and international normalized ratio), size of the largest tumour per session, tumour location (intraparenchymal, subcapsular, parapedicular, suprapedicular, and gallbladder proximity), duration of the procedure, duration of thermoablation, number of insertions per session, electrode type (Cool-tip™ Covidien 2 cm, Cool-tip™ Covidien 3 cm, Cool-tip™ Covidien Cluster 2.5 cm), hepatic segment location where RFA was performed, creation of an artificial pneumothorax during the procedure, and additional procedures performed during the same treatment session such as temporary venous occlusion.
Subcapsular was defined as a tumour location within 5 mm of the liver capsule, parapedicular as a tumour within 5 mm of the liver pedicle, and suprapedicular as a tumour within 5 mm superior to the pedicle of the liver. Proximity with gallbladder was defined as tumours that were less than 5 mm from the gallbladder.
The size of tumours was determined by the largest measured diameter of the tumour by either CT or ultrasound performed before the procedure.
All data were collected retrospectively by 2 trainees (O.R. and L.X.Z.) from the hospital electronic medical records and radiological information system.
Statistical Analysis
Statistical analyses were performed by J.Z. using SAS® software (version 9.4; SAS Institute Inc., Cary, NC, USA) and SPSS Statistics® software (version 26; IBM Corp., Armonk, NY, USA) with a significance level of 5%. Categorical variables were expressed as numbers and percentages. Continuous variables were expressed as means ± standard deviations, with number of patients in parentheses when applicable.
For characteristics of patients, tumours, and procedures, odds ratios (ORs) with corresponding 95% confidence intervals (CIs) were obtained using logistic regressions to compare the 2 groups (hospitalization duration less than vs more than 24 hours). Variables with a P-value ≤ .2 were included in the stepwise logistic regression using .05 and .1 as significance levels for entering and for staying in the model, respectively. It is conceivable that some patient characteristics like the Child Pugh-score are more specific to treatment of HCC. Post-hoc subgroup analyses were therefore conducted for RFA sessions for HCC.
Results
Patient Characteristics
The study included 291 patients (201 [69.1%] men and 90 [30.9%] women) with a mean age of 65.2 ± 11.2 years undergoing a total of 324 RFA sessions for liver tumours at the Centre hospitalier de l’Université de Montréal (CHUM), Montréal, Canada. Main characteristics of the patients, tumours, and procedures are summarized in Table 1, with additional details provided in Supplemental Table 1. Of the 324 sessions, 256 (79.0%) resulted in a hospitalization duration shorter than 24 hours and 68 (21.0%) resulted in a hospitalization longer than 24 hours. In the latter group, the median duration of stay was 2 days and ranged from 2 to 15 days. Two hundred and twenty-six (69.8%) sessions were performed for HCCs, 92 (28.4%) for metastases, 3 (0.9%) for adenomas, and 3 (0.9%) for other histological types of tumours. The mean number of tumours per session was 1.2 ± 0.5 (range, 1-4). The mean diameter of the largest tumour per session was 2.0 ± 0.8 cm (range, 0.4-5.5 cm), and the mean sum diameter of all tumours treated per session was 2.2 ± 1.0 cm (range, 0.4-5.6).
Characteristics of Patients, Tumours, and Procedures for the Percutaneous Liver RFA Sessions Included in the Study.
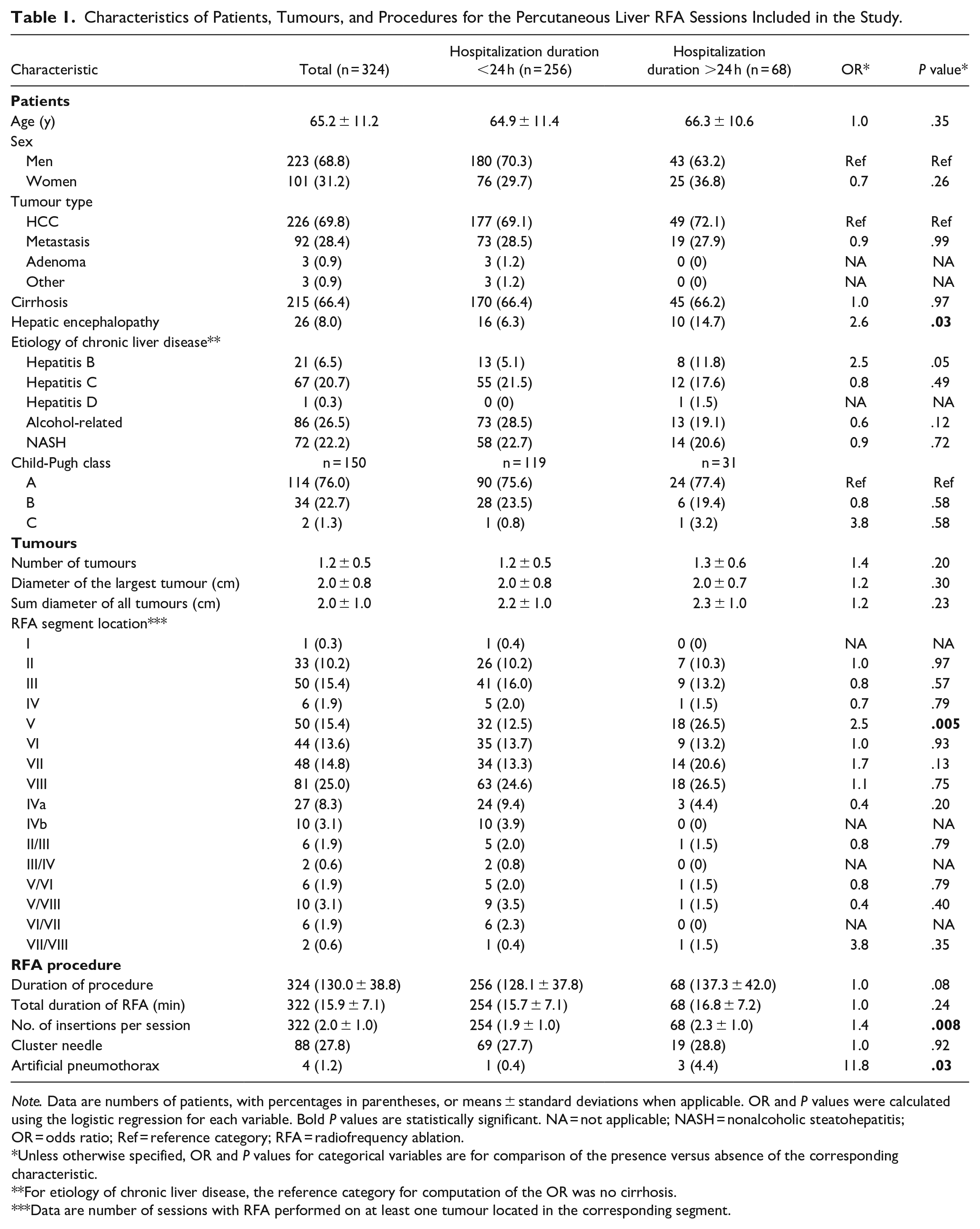
Note. Data are numbers of patients, with percentages in parentheses, or means ± standard deviations when applicable. OR and P values were calculated using the logistic regression for each variable. Bold P values are statistically significant. NA = not applicable; NASH = nonalcoholic steatohepatitis; OR = odds ratio; Ref = reference category; RFA = radiofrequency ablation.
Unless otherwise specified, OR and P values for categorical variables are for comparison of the presence versus absence of the corresponding characteristic.
For etiology of chronic liver disease, the reference category for computation of the OR was no cirrhosis.
Data are number of sessions with RFA performed on at least one tumour located in the corresponding segment.
Analysis of Risk Factors
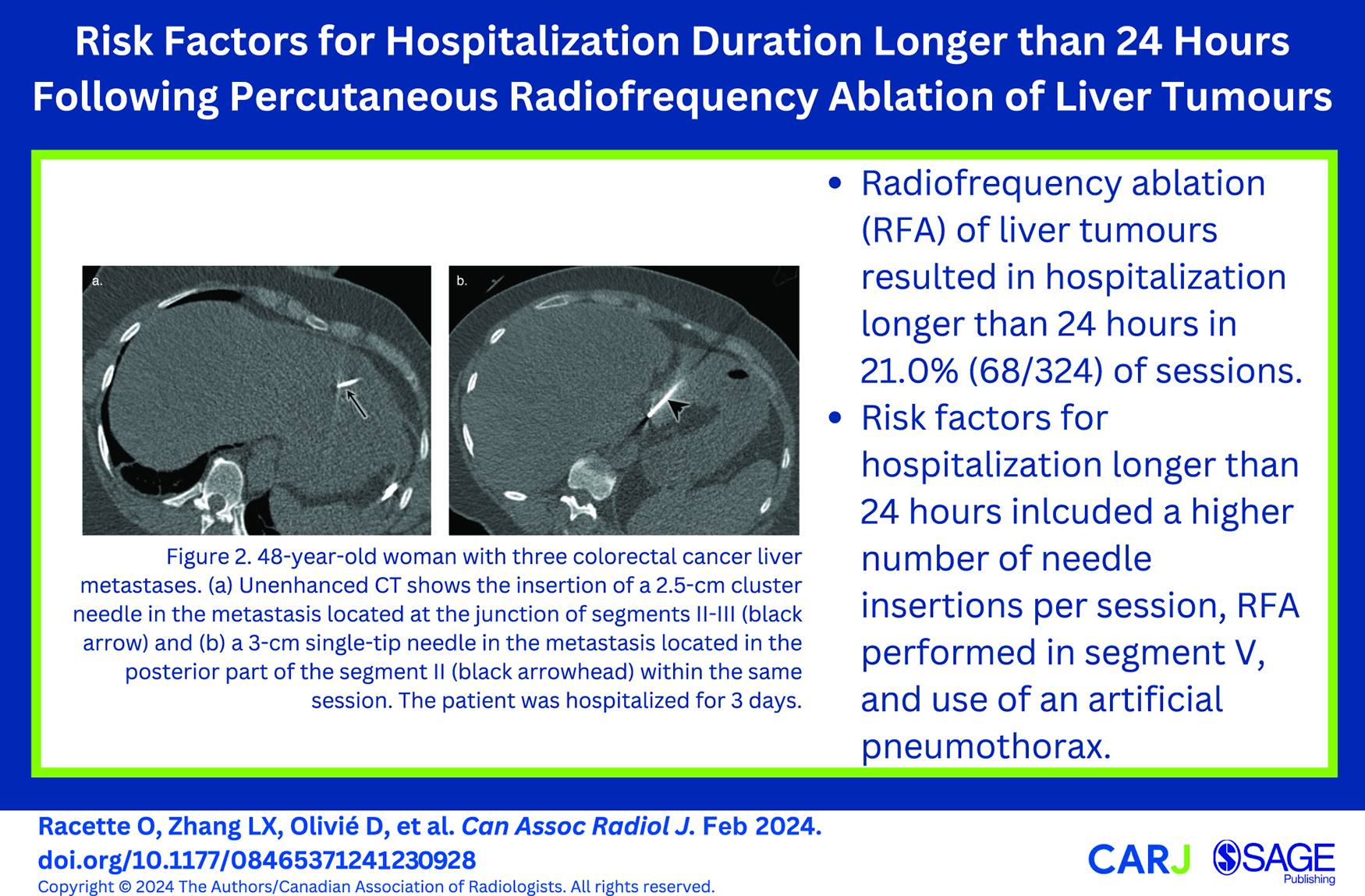
Table 2 summarizes the risk factors determined to be significant in predicting a hospitalization duration longer than 24 hours with univariable and multivariable analyses. Figure 2 shows an example of multiple needle insertions within the same RFA session, Figure 3 an example of RFA performed in segment V, and Figure 4 an artificial pneumothorax induced prior to RFA.
Risk Factors for Hospitalization Longer Than 24 Hours After Percutaneous Liver Radiofrequency Ablation in Univariable and Multivariable Analyses.

Note. OR, 95% CI, and P values were calculated using the logistic regression for each variable. Bold P values are statistically significant. CI = confidence interval; OR = odds ratio; RFA = radiofrequency ablation.
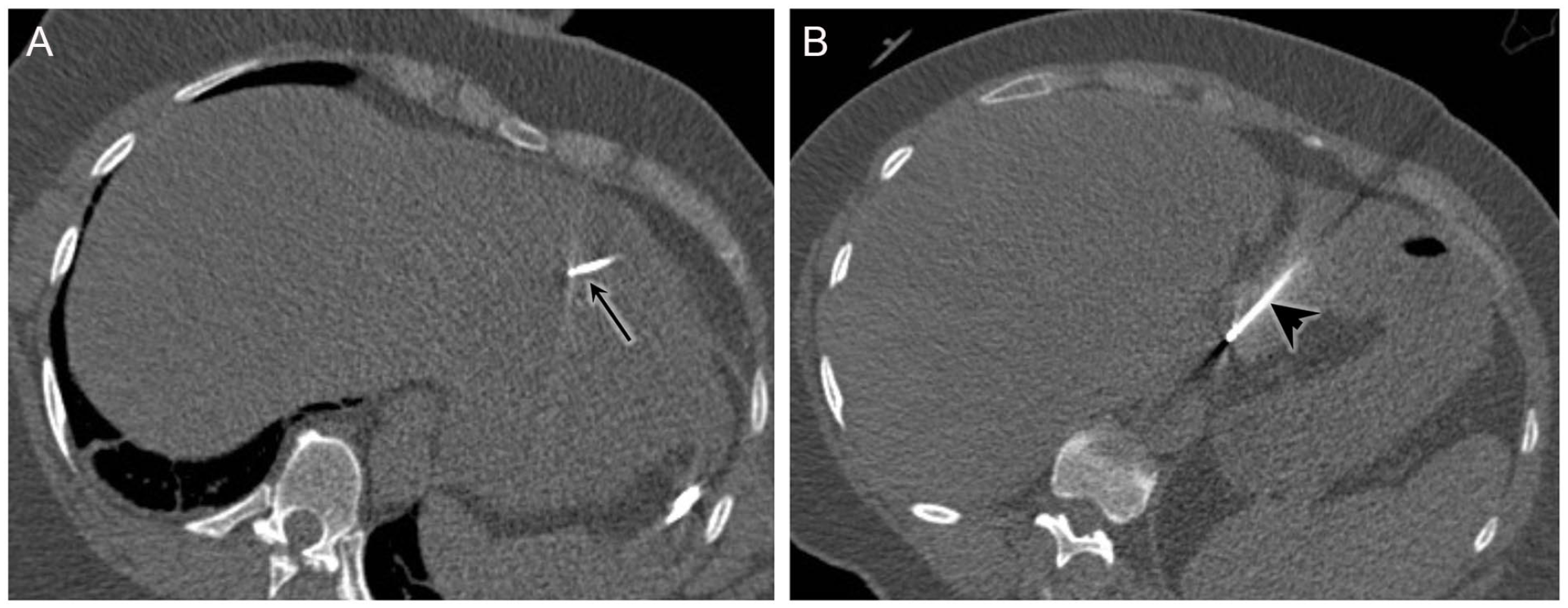
48-year-old woman with 3 colorectal cancer liver metastases. (A) Unenhanced CT shows the insertion of a 2.5-cm cluster needle in the metastasis located at the junction of segments II-III (black arrow) and (B) a 3-cm single-tip needle in the metastasis located in the posterior part of the segment II (black arrowhead) within the same session. The patient was hospitalized for 3 days.
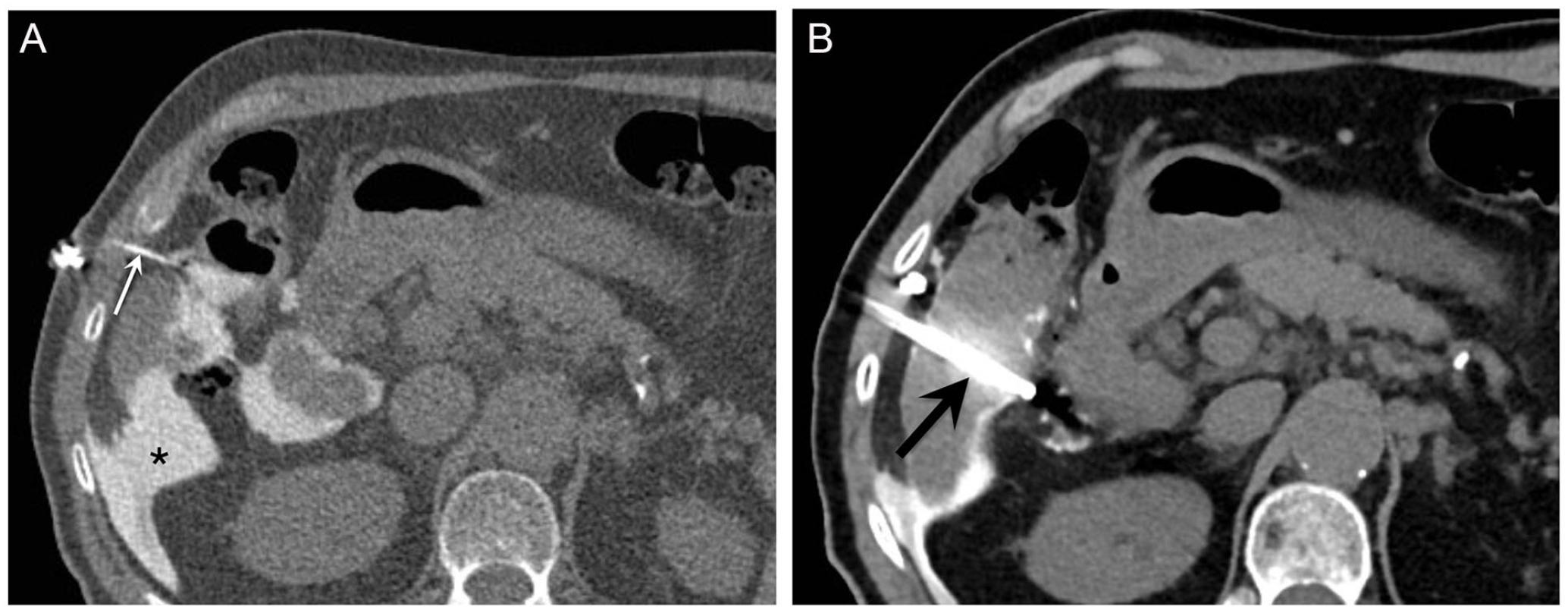
74-year-old man with hepatitis B virus-related liver cirrhosis undergoing radiofrequency ablation of a 17-mm HCC in segment V. (A) Hydrodissection was performed with a 5-Fr catheter (thin arrow) by injection of diluted contrast (asterisk) to displace the colon from the liver capsule. (B) A radiofrequency ablation was then performed with a cluster needle positioned in the tumour (black arrow). The patient presented fever after the percutaneous treatment and stayed 4 days at the hospital.
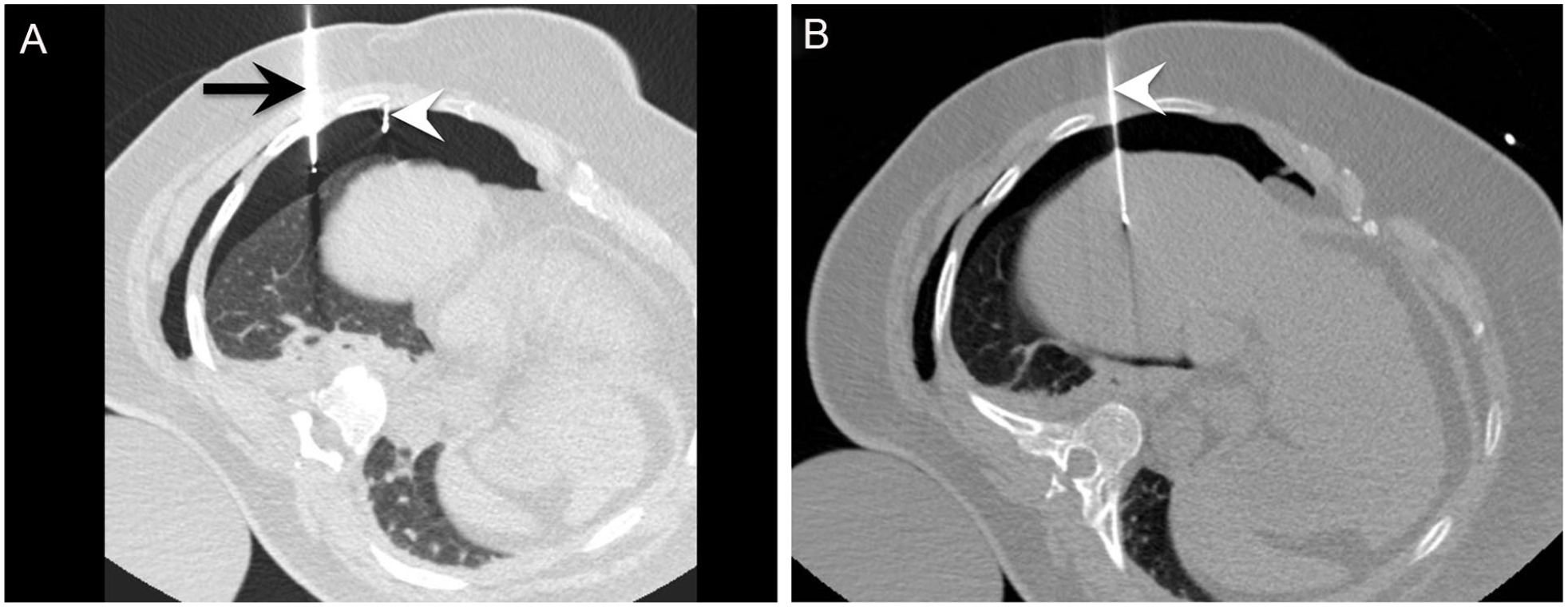
59-year-old woman with nonalcoholic steatohepatitis liver cirrhosis undergoing artificial pneumothorax for radiofrequency ablation of a 13-mm HCC in segment VIII. (A) Because this HCC was located too high in the liver dome, an artificial pneumothorax was created with a Veress® needle (black arrow) without puncturing the lung to separate the visceral from the parietal pleura through injection of CO2, which created a safe trajectory for placement of a RFA needle (white arrowhead). (B) A Cool-tip™ Covidien 15-cm/3-cm RFA needle was positioned within the HCC for tumour ablation. A chest X-ray performed 24 hours after the procedure (not shown) revealed the persistence of a pneumothorax. The patient was hospitalized for 5 days.
Univariable Analysis
A higher number of needle insertions per session, RFA performed in segment V, induction of an artificial pneumothorax, and a history of hepatic encephalopathy were identified to be significant risk factors. The OR of a hospitalization duration longer than 24 hours was 1.4 (95% CI [1.1-1.9]; P = .008) for each additional insertion per session, 2.5 (95% CI [1.3-4.8]; P = .006) for RFA performed in segment V compared to RFA not performed in this segment, 11.8 (95% CI [1.2-115.0]; P = .03) for sessions using an artificial pneumothorax compared to sessions not requiring this procedure, and 2.6 (95% CI [1.1-6.0]; P = .03) for a patient with a history of hepatic encephalopathy compared to a patient with no such history.
Multivariable Analysis
A higher number of needle insertions per session, RFA performed in segment V, and usage of an artificial pneumothorax were found to be significant independent risk factors. The OR for hospitalization duration longer than 24 hours was 1.4 (95% CI [1.1-1.9]; P = .02) for each additional insertion per session, 2.8 (95% CI [1.4-5.7]; P = .004) for RFA performed in segment V compared to RFA not performed in this segment, and 14.5 (95% CI [1.4-146.0]; P = .02) for sessions using an artificial pneumothorax compared to sessions not requiring this procedure.
In our cohort, RFA sessions were performed for HCCs in 69.8% of the RFA sessions. Post-hoc subgroup analyses conducted on RFA sessions for HCCs did not yield additional risk factors.
Overall, for the 68 sessions with a post-procedure stay of more than 24 hours, the main types of events recorded that could have potentially contributed to the prolonged stay include (more than one for some patients): surveillance for suspected or confirmed traumatic complications such as hematoma (6), hemoperitoneum (1), and persistent artificial pneumothorax (3); pain (20); worsened liver function tests (6); hepatic encephalopathy (4); fever and/or confirmed source of infection (7); other medical comorbidities requiring surveillance or management such as urinary retention, acute renal failure, electrolyte imbalances, decompensated diabetes, pulmonary edema, cardiac events, uncontrolled blood pressure, delirium (29); and administrative reasons such as waiting for other consultants or for transfer in another institution (3).
Discussion
This retrospective analysis evaluated the potential risk factors for hospitalization duration longer than 24 hours following radiofrequency ablation of liver tumours. A higher number of insertions per session, radiofrequency ablation performed in segment V, and artificial pneumothorax were found to be independent risk factors. A history of hepatic encephalopathy was a significant risk factor at univariable, but not at multivariable analysis.
Our results showed that each additional needle insertion per session was an independent risk factor for a hospitalization longer than 24 hours, with an OR of 1.4. A higher number of needle insertions per session usually reflects a higher overall complexity of the procedure, and thus is associated with a higher risk injury and longer recovery time. Our results are consistent with those of 2 previous studies, one of which found that 2 or more punctures represented a significant risk factor for minor hemorrhage, 22 and the other suggesting that the number of coaxial needles was an independent predictive factor for major complications. 1 On the other hand, the use of cluster electrodes was not a significant risk factor in our study, whereas it was identified as a risk factor for minor hemorrhage in the former study above. 22 However, it constituted a minor immediate complication without specification of the impact on the length of stay.
Additionally, the number of tumours treated per session, and the maximum diameter or the sum diameter of the tumour treated were not significant risk factors in our cohort. The relationship between the number of tumours per session and their size, and complications after RFA remain controversial in previous studies.12,14,15,17-22 Interestingly also, the duration of the procedure was not found to be a significant risk factor, and neither was the total duration of RFA, in accordance with a previous study. 17
We found that a RFA performed in segment V was an independent risk factor with an OR of 2.8. A possible reason for this relationship is the proximity to the colon, gallbladder, and bile ducts.25,26 Also, some of these patients had hydrodissection or carbodissection, procedures that could require monitoring until resolution and thus lengthen the hospital stay out of precaution. Previously, Goto et al 17 reported that tumour location in segment I or in segment VII were risk factors for hemobilia or hemothorax, respectively. Schullian et al 1 found that tumours located in segment IVa or VIII were independent prognostic factors for major complications presumably because of the proximity to the diaphragm, and segment VII as another negative prognostic factor but only on simple logistic regression. Hence, tumour location thus appears to be an inconsistent predictor of complications.
Artificial pneumothorax was a significant risk factor for prolonged stay in our cohort. Three out of the 4 patients with artificial pneumothorax for a sub-diaphragmatic tumour in segment VIII or IVa had persistent pneumothorax in addition to other events (ileus and hemoperitoneum, thrombopenia refractory to transfusion, and pain). They respectively stayed 4, 4, and 5 days. Artificial pneumothorax was initially described in 6 patients as a useful and safe technique for CT-guided RFA of liver dome tumours. 27 Two other studies then reported similar results, but were retrospective in nature and with limited sample sizes (16 and 26 respectively).28,29 The former did not mention the length of post-procedure stay, and in the latter all procedures were performed by the same interventional radiologist. It is plausible that the presence of a persistent pneumothorax after RFA incites a cautious approach and prolonged hospitalization.
In our cohort, the Child-Pugh score was not a risk factor for hospitalization duration longer than 24 hours. The severity of chronic liver disease, as assessed by the Child-Pugh class, has been studied as a risk factor for complications after liver RFA, with conflicting results. While some studies have identified Child-Pugh class as a risk factor for complications,14,21 others have not concluded to a significant difference.15,18,20,22 Unlike prior studies, which examined Child-Pugh score in aggregate, we have included each individual component of the Child-Pugh score and have identified a history of hepatic encephalopathy as a risk factor in univariate analyses with an OR of 2.6. This is likely explained by the observation that patients with a history of hepatic encephalopathy are more prone to recurrent episodes, and are at increased risk of decompensation post-RFA. 30 In our cohort, 4 patients developed hepatic encephalopathy and stayed more than 24 hours post-procedure, all of which have a prior history of hepatic encephalopathy. Incidentally, 2 patients in our cohort were Child-Pugh C, one was discharged within 24 hours and the other stayed an additional day for increased bilirubin and fever with negative workup.
Our study has a number of limitations. First, we included more than one session for the same patient as independent events in a small number of cases. However, each session targeted different tumours with different procedure characteristics and was of interest in this study. Moreover, this is representative of a real-world clinical setting, where a patient may require multiple RFA sessions. Second, we did not have access to all the laboratory results or clinical information required to compute the Child-Pugh score for some patients. Instead, we relied on the scores provided by clinicians when available. Third, our study included patients with different types of tumours with potentially distinct risk factor profiles. For instance, the Child-Pugh score is relevant to patients treated for HCCs, but less for patients treated for metastases. To account for this, we performed sub-group analyses for the group treated for HCCs.
Conclusion
Our study identified risk factors for hospitalization duration longer than 24 hours following RFA of liver tumours, including a higher number of needle insertions, RFA performed in segment V and the use of an artificial pneumothorax. The findings could potentially help stratify high-risk patients and inform resource planning for post-procedure stay. Larger, multi-centre studies are required, and other clinical endpoints such as hospital readmission could be explored in the future.
Supplemental Material
sj-pdf-1-caj-10.1177_08465371241230928 – Supplemental material for Risk Factors for Hospitalization Duration Longer Than 24 Hours Following Percutaneous Radiofrequency Ablation of Liver Tumours
Supplemental material, sj-pdf-1-caj-10.1177_08465371241230928 for Risk Factors for Hospitalization Duration Longer Than 24 Hours Following Percutaneous Radiofrequency Ablation of Liver Tumours by Olivier Racette, Li Xin Zhang, Damien Olivié, Kim-Nhien Vu, Jeanne-Marie Giard, Franck Vandenbroucke-Menu, Gilles Soulez, Justine Zehr, An Tang and Jean-Sébastien Billiard in Canadian Association of Radiologists Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fonds de Recherche du Québec–Santé (FRQ-S) and Fondation de l’Association des Radiologistes du Québec (FARQ) Clinical Research Scholarship–Senior Salary Award (FRQS-ARQ no. 298509) to A.T.
Supplemental Material
Supplemental material for this article is available online.
Authors’ Note
Institution from which the work originated: Centre Hospitalier de l’Université de Montréal (CHUM), 1000, rue Saint-Denis, Montréal, Québec, Canada, H2X 0C1.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
