Abstract
Colon cancer is the third most common malignancy in Canada. Computed tomography colonography (CTC) provides a creditable and validated option for colon screening and assessment of known pathology in patients for whom conventional colonoscopy is contraindicated or where patients self-select to use imaging as their primary modality for initial colonic assessment. This updated guideline aims to provide a toolkit for both experienced imagers (and technologists) and for those considering launching this examination in their practice. There is guidance for reporting, optimal exam preparation, tips for problem solving to attain high quality examinations in challenging scenarios as well as suggestions for ongoing maintenance of competence. We also provide insight into the role of artificial intelligence and the utility of CTC in tumour staging of colorectal cancer. The appendices provide more detailed guidance into bowel preparation and reporting templates as well as useful information on polyp stratification and management strategies. Reading this guideline should equip the reader with the knowledge base to perform colonography but also provide an unbiased overview of its role in colon screening compared with other screening options.

Introduction
Colorectal cancer (CRC) is the third most common malignancy in Canada with over 26,000 cases per annum in 2020. Since 2017, all 10 provinces and Yukon have implemented organized screening programs. Nevertheless, almost half of CRC is diagnosed at stage III or IV, this is paralleled by suboptimal participation, with no program meeting the minimum target of 60% uptake. 1 COVID-19 had a detrimental impact on screening programs, and CRC was no exception. Although CT colonography (CTC) has not been used for mass screening in any province, it remains an integral adjunct to conventional colonoscopy and offers choice to participants who are reluctant or unable to proceed with conventional screening methods.
This paper describes the current state of CTC in the adult population based on the evolving scientific evidence in the field. The Canadian Association of Radiologists last provided guidance on CTC in 2010. 2 The following practice update offers recommendations and minimum standards for the acquisition, interpretation, reporting, and maintenance of competence in CTC, along with a tool kit for radiologists and technologists who are performing or embarking on CTC in their practice. A sample reporting template is provided in Appendix 1.
Role in Screening
Detection Rates of Screening Tests for Colorectal Cancer.

In general, CTC should be offered for screening every 5 years after the age of 50 if optical colonoscopy is medically contraindicated, technically challenging, or incomplete.10-14 Patient preference may also be a reason to choose CTC. Some data suggests that individuals at low and moderate risk of colon cancer can be screened with CTC, while high-risk patients should be screened with colonoscopy where possible. 11
There is extensive published evidence in support of the reliability of CTC for screening for CRC. For example, a study showed that CTC identified 90% of adenomas or cancers measuring 10 mm or more in diameter. 15 Validation trials in screening cohorts and subsequent meta-analyses have demonstrated high sensitivity,11,16 often exceeding OC, with no missed cancers. 3 Randomized controlled trials in Europe evaluated CTC as the primary test for population screening of CRC, compared to fecal immunochemical test (FIT), sigmoidoscopy and OC. The detection rate of CTC for CRC and advanced adenomas was 5.1-6.1%. 14
The Value of CTC in a Screening Paradigm
A Canadian study showed it is feasible to run a high-volume CTC service, reducing the requirement for optical colonoscopy in over 70% of patients, confirming that CTC is a valuable triaging tool where long OC waitlists exist. 17 The participation rates for screening CTC are higher than for optical colonoscopy.14,18 Therefore, despite a slightly lower detection rate of polyps with CTC than with colonoscopy, the detection rates per patient of CTC and colonoscopy are comparable. The higher participation rates also render CTC more cost-effective, justifying the inclusion of CTC in organized screening programs for CRC.
Indications for CTC
Indications for CTC.
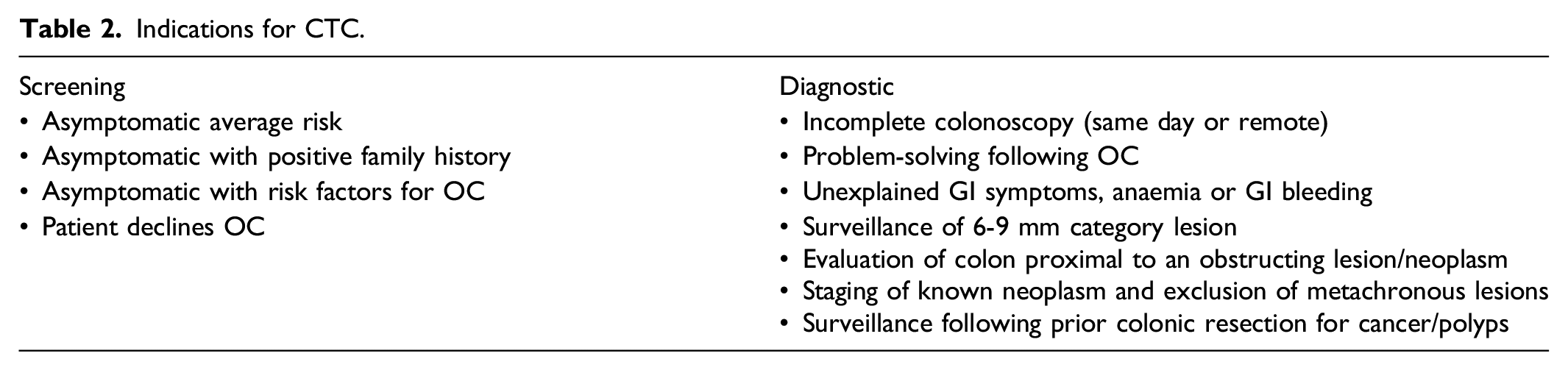
Screening
Contraindications for CTC.

Adapted from ACR-SAR-SCBT practice parameter for the performance of CTC in adults. 26
Incomplete OC
CTC is a critical tool in the evaluation of patients with signs and symptoms suggestive of colorectal cancer for whom OC is incomplete or contraindicated due to an occlusive tumour or patient factors (incomplete bowel preparation, anatomical factors, patient discomfort, sedation risk, severe co-morbidities). Almost all incomplete studies performed with lumen-occluding lesions can be interrogated on CTC. As many as 35.1% of synchronous lesions occur in 1 or more segments away from the offending lesion and the depiction of these significantly impacts patient management.
Synchronous CRC prevalence is reported at 5.7% (95% CI 4.7%–7.1%), 20 highlighting the key role CTC plays in preoperative evaluation of the entire colon. Identification of synchronous lesions can alter patient management in up to 21.5% of patients with an occlusive CRC and incomplete colonoscopy. 21
Contraindicated OC
CTC is a useful and appropriate tool for patients who have a contraindication (absolute or relative) to OC. In the event of a significant finding, the patient should be counselled that subsequent OC may be required.
Signs or Symptoms of Colorectal Cancer
CTC plays a role in evaluation of patients with signs and symptoms suggestive of colorectal cancer for whom OC is incomplete or contraindicated. CTC20,21 and OC have similar sensitivity (93% vs 92% respectively) for detecting CRC in patients over 65 years, 16 and large polyps in symptomatic patients. 22
Post-Operative
CTC is indicated in patients after hemicolectomy for whom OC is contraindicated or unfeasible. Patients should be informed that CTC is inferior to OC in detection of polyps in this setting, with a sensitivity of 44.0% for polyps ≥6 mm and 79.6% for polyps ≥10 mm. 23
Other Clinical Scenarios
Patients with a history of prior colon cancer or neoplasia can undergo CTC if there is a contraindication to OC. In this setting, it offers a one-stop shop for restaging and colonic assessment.
Safety
CTC and colonoscopy are both associated with low risk of perforation, although lower with CTC. Meta-analyses published in 2014 and 2016 report rates of perforation of .04% in CTC 24 and .5% in colonoscopy. 25 Further, the overall risk of perforation is higher in symptomatic/diagnostic individuals compared to those undergoing screening. 24 Most perforations are treated conservatively.24,26
Risk factors for perforation include manual insufflation with room air or an inexperienced operator, recent colonoscopy with polypectomy, older patient age, inguinal hernias containing colon, diverticulosis, or obstructing neoplasm.27,28 CTC can be performed after a colonoscopy with polypectomy, although many authors advocate waiting 2-4 weeks.27,29 Risk can be further mitigated post-colonoscopy with a low-dose non-contrast CT before insufflation to exclude a clinically-occult perforation. 12 Minor adverse events such as vasovagal reactions are also rare and self-limited.27,30,31 Radiation exposure with CTC is small, with the estimated number of CRCs prevented with a 5-year screening interval outweighing the number of radiation-induced cancers (benefit-risk ratio of 24:1). 32 While there is variation in effective dose between institutions, CTC doses are now lower than reported in this study. 33
With advances in CT technology, such as iterative reconstruction and lower tube voltage, CTC effective doses have been reported as low as sub-millisievert level. 34 Iodinated IV contrast is not typically administrated for CTC. However, if administered, there is a very low risk of iodinated contrast media reactions, (for example anaphylaxis), contrast-associated acute kidney injury, 35 and hypersensitivity. 36
Benign colonic pneumatosis is seen in approximately .11% of patients and has a predilection for the right colon. 37 The patients are usually asymptomatic. Radiologically, there is intra-mural gas but no free air or extrinsic to the colonic wall. This phenomenon has only been described with CO2 insufflation.
Extracolonic Findings
One of the common criticisms levelled at CTC is the detection of extracolonic findings (ECF). To reduce undue patient anxiety, cost and unnecessary further investigations, ECF should be stratified from clinically unimportant (E2) to potentially important findings (E4) as per CT Colonography Imaging Reporting and Data System (C-RADS). 38 (Appendix 2). Although up to 70% of CTCs may produce extracolonic findings, the majority of these (84-88%) are insignificant/normal variants (E1) or unimportant/benign, not requiring further work up (E2). 39 Potentially significant ECF (E4) are more frequently identified in older and symptomatic patient populations. 40
Apart from CRC and advanced adenoma detection, CTC offers an opportunity for limited screening of the lower thorax, abdomen and pelvis. Up to 4.0% of potentially important ECF (E4) represent malignancy 39 (most commonly RCC, lung, and lymphoma). The findings that may warrant further investigation (E3) or are potentially clinically significant (E4) include extracolonic malignancies, abdominal aortic aneurysms, or undiagnosed pathologies such as urolithiasis and chronic liver disease. Artificial Intelligence offers an opportunity to improve the accuracy of incidental extracolonic lesion characterization as well as opportunistic screening for extracolonic pathology.
Though the shift from conventional to low-dose CTC has reduced sensitivity in detecting potential ECF, recent meta-analysis demonstrates that potentially significant ECF (E4) are identified in up to 5.2% of symptomatic and 2.8% of asymptomatic patients, with recommendations for further investigations in up to 8.2% of all patients. 41 Referring clinicians should be aware of the current paradigm for low-dose CTC and be cognizant of the downstream impact on the detection of extracolonic findings. Reports should acknowledge diminished sensitivity.
Ultimately, opportunistic screening of asymptomatic populations for ECF (outside the spectrum of malignancy) has a dual advantage as it maximises the cost-effectiveness of this examination. Potential areas include osteoporosis, abdominal aneurysm screening and fat quantification.
Flat/Serrated Lesions
Serrated polyps represent an alternate pathway to the development of colorectal cancer and likely account for 15-30% of sporadic non-familial cases.42,43 The WHO has subclassified serrated lesions into hyperplastic polyps, sessile serrated lesions (SSL), SSL with dysplasia, traditional serrated adenomas (TSA) and unclassified serrated adenomas.
SSL with and without dysplasia are superficially elevated lesions >3 mm in height relative to the normal mucosa with a plaque-like morphology and have a risk of malignant transformation. These lesions can be subtle with minimal protrusion into the lumen. They have a propensity for the proximal colon and are usually larger (>10 mm). Prevalence of these lesions in asymptomatic Western populations is estimated at around 3.1% for the larger lesions. 44 There is a wide variation in reported prevalence due to lack of consensus on the definition, detection strategies and histopathologic assessment. 45 A small proportion of these lesions (Type IIC = central depression) are more aggressive than their counterparts; this risk has been incorrectly assigned to all flat lesions.
Awareness of their typical morphology and location can help improve diagnostic accuracy in the detection and reporting of these lesions. Concurrent improvements in technique have resulted in improved detection rates at CTC. Emerging data shows many of these lesions have an indolent natural history with many showing little or no progression at follow-up CTC studies. Older studies have shown inferiority in detection of flat lesions relative to conventional polyps. Unlike traditional polyps, sessile lesions are risk-stratified by morphology, 46 and not by size alone (Appendix 3). 47
Colonic distension is an important prerequisite for polyp detection, however, readers need to be aware of the impact of distension on the minimal profile of sessile lesions. 48 The so-called “disappearing phenomenon” can further diminish lesional conspicuity. The presence of adherent contrast, as well as concurrent confirmation of a fixed location on both 2D and 3D are invaluable methods of detection. Lack of tagging may contribute to their inferior detection on CTC relative to OC. 46 A recent study (N = 7940) demonstrated that CTC with a 6- and 10-mm threshold had a higher yield and positive predictive value for the high-risk SSLs as compared with non-invasive testing such as mt-Sdna. 45 PPV and overall screening yield for high-risk SSLs were 5.5% vs 14.4% and .7% vs 1.7% for mt-sDNA vs CTC at a 6 mm threshold, respectively (both P < .001).
Polyp Management Strategy
Risk Stratification According to Polyp Size.
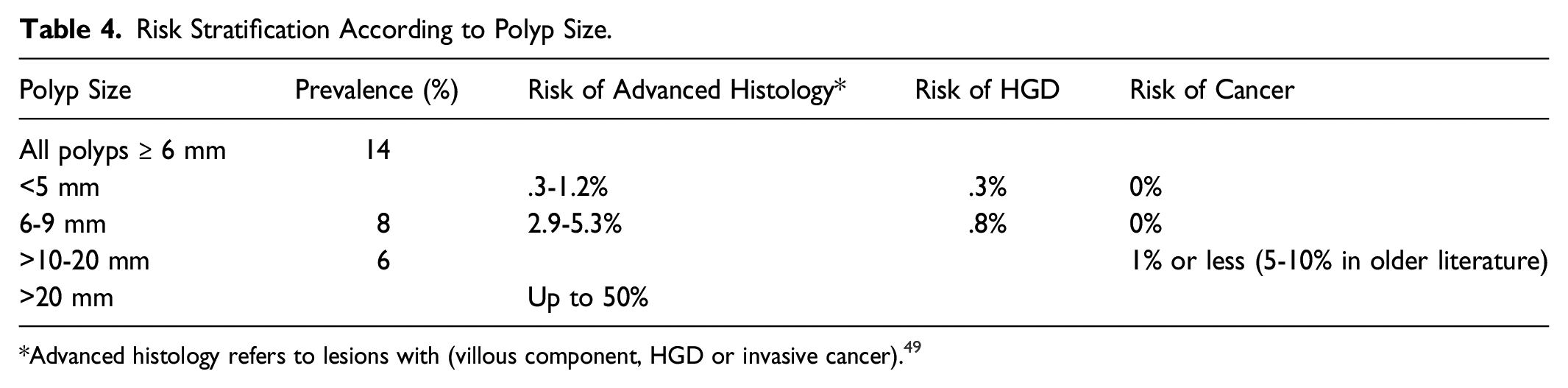
*Advanced histology refers to lesions with (villous component, HGD or invasive cancer). 49
30-40% of the population will develop an adenoma by age 60, 50 and risk of developing colorectal cancer increases with age, particularly between 60-70 years of age.51-54 The lifetime cumulative risk of developing cancer is 5.5%. 50 However, the natural history of polyps is not always predictable nor stepwise, and not all colonic adenomas progress to malignancy. 15-20% of cancers arise via an alternate pathway where size is not the determinant of risk.
Despite these 2 pathways, it is widely acknowledged that the dwell time for this progression is likely a decade or more. Awareness that selective polypectomy is more cost-effective and results in wiser utilization of limited resources with a lower incidence of complications is an important paradigm shift; it is a divergence from the gastrointestinal approach of universal polypectomy (Figure 1). Correlation between C-RADS categories and risk stratification based on endoscopic findings, with guidance for follow-up intervals.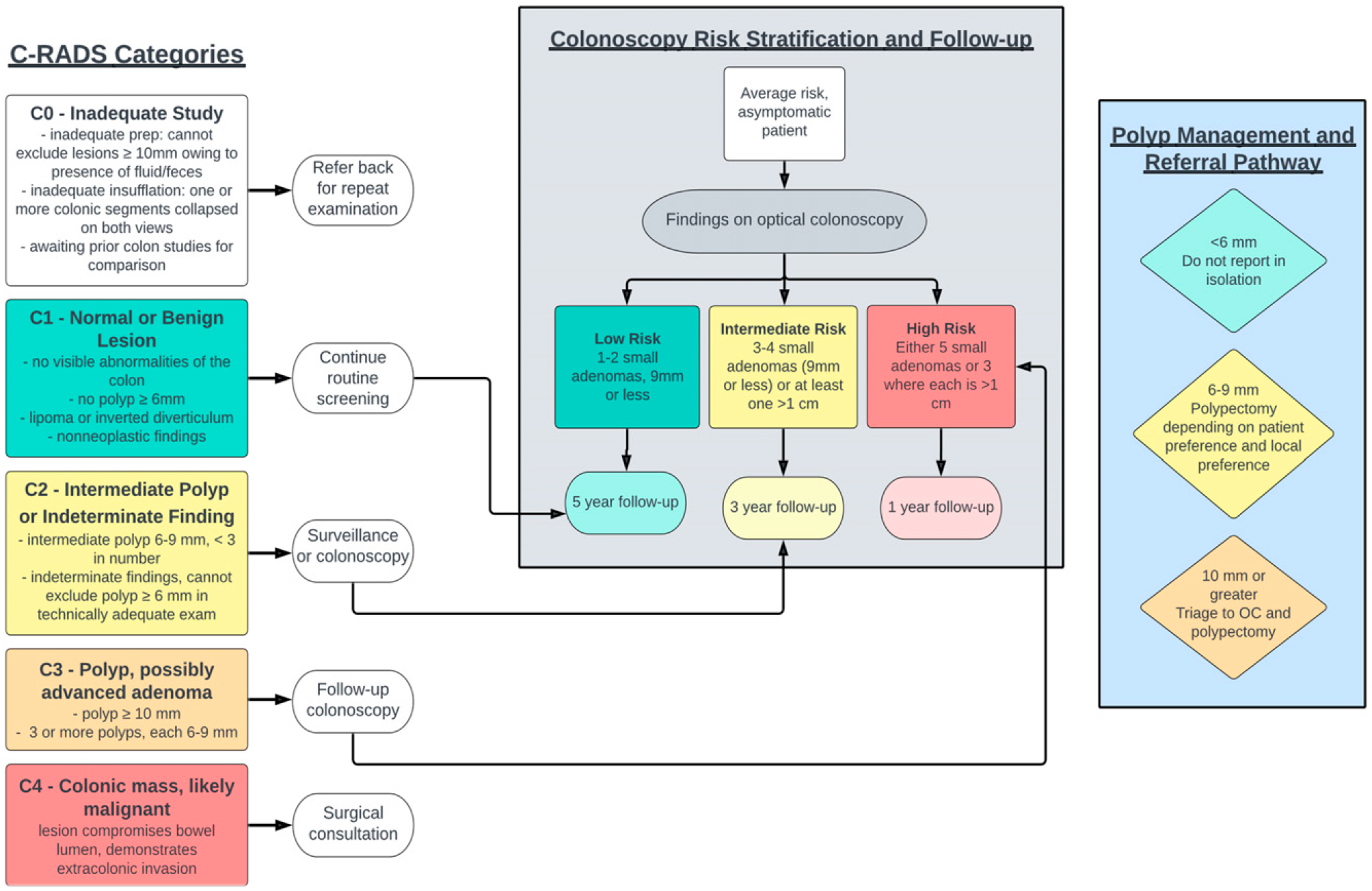
Traditionally, polyps >1 cm and cancers are triaged to immediate polypectomy. The management of lesions in the 6-9 mm category is variable and will be informed by local policy (as discussed with GI), patient preference and individual risk factors. For example, medium and even large polyps are frequently not referred for polypectomy if the risk for conventional scope appears to outweigh the potential benefit.
CTC with a selective polypectomy strategy is the most cost-effective method of performing CTC and compares favorably with conventional scopes when this strategy is employed.55,56 Additionally, natural history data shows mid-size polyps can be safely observed, with a least 20% of lesions in this category showing net regression. 57 Moreover, CTC confers the benefit of observing volumetric (and not linear) threshold for growth as well as proving an accurate localization of location and morphology. 58
Artificial Intelligence
Vendor specific recommendations are beyond the scope of this document but we urge readers to try out colon software prior to purchase to personally assess performance, characteristics and range of plug in options (e.g., CAD) prior to committing to a product. Commercially available computer-aided diagnosis (CAD) tools are based on second-era machine learning which was developed prior to wider access to deep learning tools. Deep learning has enabled the learning and linking of patterns in datasets and allows for the creation of more robust solutions. These are more commonly referred to as Artificial Intelligence (AI). 59 CAD tools/plugins are offered by most vendors on their 3D visualization platforms; however, their role in a routine workflow is less well established. CAD tools as a second reader have been shown to improve reader sensitivity, especially for less experienced readers, 60 but also led to reduced specificity and an increase in false positives in those studies. Studies have shown that comprehensive training can result in performance akin to that of CAD as a second reader. 60
However, current research shows that AI/ML tools have the potential to improve detection of clinically significant polyps and overcome some of the shortcomings of CAD tools. 61 A proof of concept study showed deep learning could discriminate premalignant from benign polyps on CT and therefore identify the subset of patients who would benefit from endoscopic polypectomy. AI tools allow for improved image reconstruction of low-dose CT acquisitions. 62 An exciting area for AI/ML applications is opportunistic screening for osteoporosis, vascular aneurysms, coronary artery disease, visceral fat and metabolic syndrome, and sarcopenia.63-65 Future AI/ML research may also improve the characterization of incidental extracolonic findings; however, there are no current active projects in this space.
Role in T Staging
Accurate T staging is an important prognosticator in the pretreatment setting of colorectal cancer. Preoperative staging impacts surgical approach and the extent of nodal dissection. Clinical trials such as the FOxTROT 66 and PRODIGE-22 67 have shown the merits of pre-operative chemotherapy in high-risk patients (T3 and T4). Overtreatment remains a concern and accuracy of T staging is paramount in stratification. Accuracy of conventional CT remains suboptimal. Despite the advent of multi-detector CT, accuracy of T staging for colorectal cancer is 73% if axial images were assessed alone and improved to 83% when interpreted in the setting of multiplanar reconstruction (MPR). Pre-operative T staging by CTC with MPR shows improved diagnostic confidence in discriminating between T2 and T3 disease as neoadjuvant therapy is indicated in the context of the higher-risk lesions. A prospective study of patients with colon cancer who underwent CTC pre-operatively showed an accuracy of 87.1% in discriminating T1/T2 vs T3/T4 tumours, with high sensitivity (88.5%) and specificity (84.1%). For high-risk tumors (T3 ≥ 5 mm and T4), CTC showed an accuracy, sensitivity, and specificity of 82.7%, 86% and 80%, respectively (kappa = .65). 68
Imaging criteria based on the spatial relationships of tumours and the bordering vessels (both marginal and subserosal) are useful in distinguishing T3 from T2 and T4 disease. 58
One of the strengths of CTC is being able to screen the entire colon even in the setting of stricturing or obstructive lesions. In this scenario, a diagnostic enhanced study in the portal venous phase, with optimized MA and kVp is performed as the second acquisition (supine series by convention). The parameters are lower for the non-contrast series. Even at low dose, unenhanced CT can evaluate the colon wall deformity and fatty infiltration demonstrating serosal involvement.
Utilization in Times of Stress in Healthcare
To date, CTC has not been used in population-based screening due to availability, cost, expertise, and concern with regards to widespread implementation. The recent COVID-19 pandemic has reinforced that exposure-prone procedures including upper and lower GI endoscopy can be heavily impacted at times of stress on the healthcare system. There is modelling data showing that even transient interruptions to screening can impact the numbers of individuals whose CRC diagnosis is delayed or worse not made with impact on overall patient outcome.69,70 Prioritization tools have been suggested to triage the small subset of patients who do need endoscopic intervention. CTC is 1 such method that can allow noninvasive, risk-free screening and help minimize wait times.
Bowel Preparation
Bowel preparation 71 is crucial for a successful CTC examination, affecting sensitivity and performance for the detection of pathology (Appendices 4 and 5). A low residue diet may be required, especially for patients with poor preparation or refractory constipation. Preparation involves a cathartic agent (laxative) and a tagging kit, with a low-volume, iso-osmolar cathartic being preferred. The agent should be safe for patients with fluid restriction, cardiac issues, hepatic or renal insufficiency. Dry regimens with hyper-osmolar agents have higher patient acceptance and leave less residual fluid. Factors such as patient preference, medical comorbidities, and prior reactions influence the choice of agent. The preparation should leave the lumen clean and dry, and a 1-day prep is well tolerated and effective for most patients. 72 Although residual fluid in the lumen can be easily managed on OC by suction, this is not an option on imaging; moreover, patient tolerance is low. 73
The tagging kit comprises a 2% w/v barium solution and an iodinated contrast agent gastrografin (diatrizoate) or iohexol are 2 options. 74 Tagging helps distinguish polyps or cancers from soft tissue density and improves reader performance, with less reliance on dual positioning for diagnosis. The tagging incorporates contrast into any residual fluid (iodine) or fecal material (barium) and has a secondary cathartic impact. 71 The material can be electronically subtracted from the lumen using a thresholding algorithm. 75
The by-product of tagging is the coating of the ingested contrast to the surface of polypoid lesions (both sessile and convex lesions). This phenomenon is related to the presence of altered mucoproteins resulting in adherence of contrast material on the surface of polyps.76,77 This also improves lesional conspicuity and diagnostic performance. An awareness of the impact of tagging in terms of artifact on 2D imaging (beam hardening at low kVp) as well as subtraction artifacts on 3D images is important, but beyond the scope of this guideline.
Non-cathartic or prepless examinations are not the preferred method when polyp detection is the desired outcome. However, there is a sufficient body of evidence to indicate this technique has a role to play in the exclusion of cancer/large polyps in older/frail patients 31 for whom a full bowel preparation may be contraindicated or technically unfeasible. A study utilizing only gastrografin prep showed a very high NPV of .99 for exclusion of CRC. 78 DECT may potentially impact the quality of non-cathartic CTC as artifacts typically associated with electronic cleansing are not seen in this technique. A dual energy index can aid discrimination of air, fecal tagged material from soft tissue density lesions.81When reading an examination where preparation is suboptimal a 2D approach may be necessary for segments where assessment is limited by the presence of stool/residual material. A comment in the report that sensitivity is limited and confined to detection of larger polyps or masses may also be appropriate.
For same-day CTC following incomplete colonoscopy we recommend 30 mls of ionic water-soluble iodinated contrast. 79 This is administered orally once the patient is fully recovered from sedation. A minimum of 2-3 hours wait is preferred to allow adequate opacification of any untagged fluid. In a study with a 2 hour wait time, there was failure to reach the left colon in 26% of patients. 79 This may not be an issue where the left colon has been adequately seen on OC. There are studies showing longer wait times improve homogeneity, performance of flat lesion detection and degree of tagging 80 but for scheduling and patient-related factors, this may not be a viable option. 81
Technique
Colonic Insufflation
Before the examination, it is prudent to ask questions about compliance with the bowel preparation and tagging regime. Success is usually indicated by passage of clear or yellow stained fluid. If the patient is still passing solid stool additional preparation may be needed (Appendix 5e-f).
Troubleshooting the CTC exam.
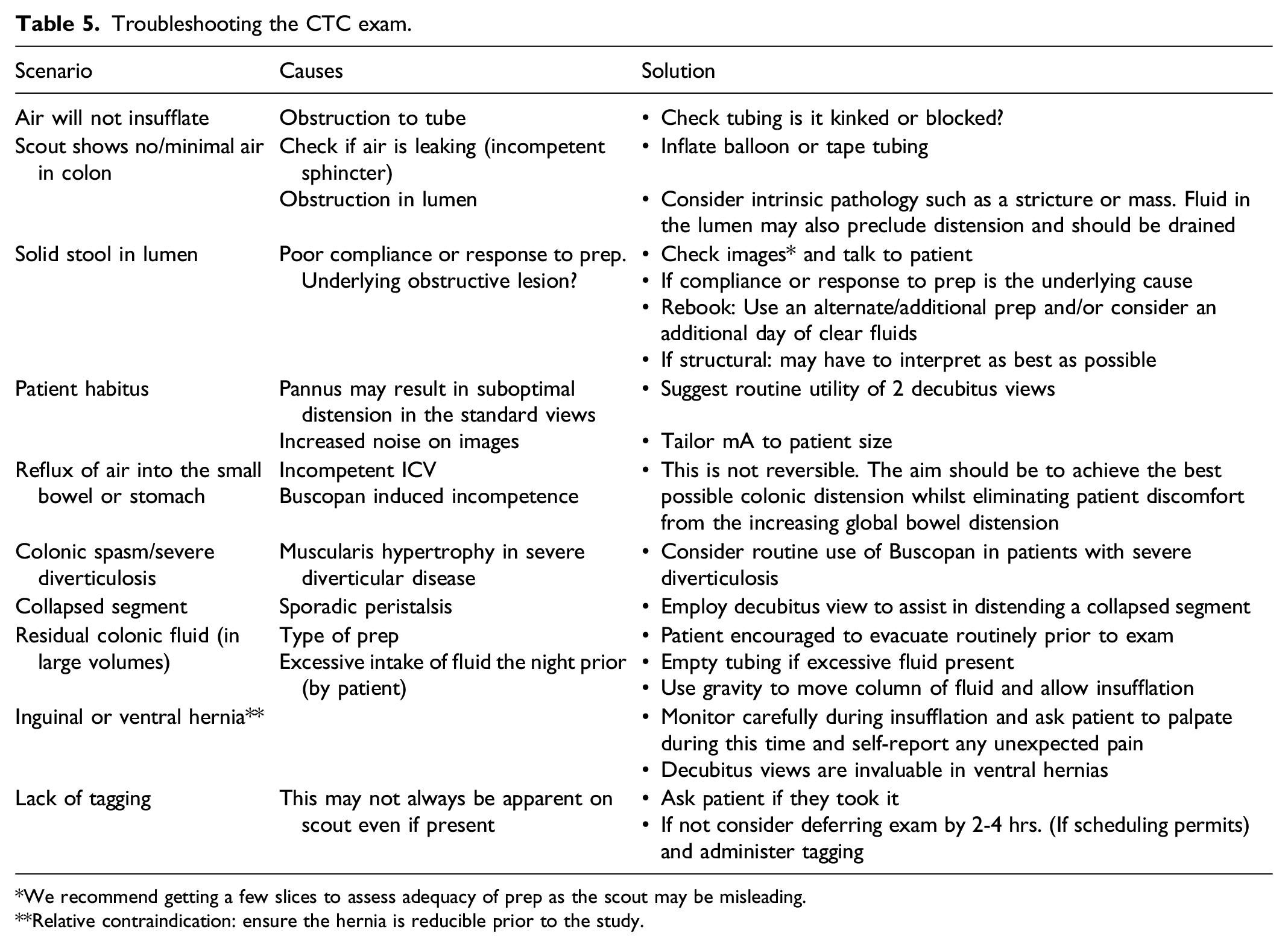
*We recommend getting a few slices to assess adequacy of prep as the scout may be misleading.
**Relative contraindication: ensure the hernia is reducible prior to the study.
In the post-operative scenario CTC is technically feasible in the setting of a colostomy (e.g., following APR). We recommend inserting the Foley catheter several centimeters beyond the cutaneous surface to avoid the risk of balloon expulsion while inflating with CO2. Modifications to image acquisition include substitution of a prone series with a decubitus for obvious reasons. 85 In the setting of an ileostomy the examination may be more technically challenging. In both scenarios a modification to bowel prep is needed. Patients with ileostomies are susceptible to dehydration and electrolyte disturbance and require a clear fluid preparation only (No cathartic), with a colostomy a split preparation is often needed. Both scenarios offer the opportunity for a “1 stop shop” in the surveillance of colorectal cancer.
Antispasmodics
Hyoscine Butylbromide (Buscopan), can be used to improve colonic distention and increase patient comfort during the examination. One study found that Buscopan improved colonic distention significantly for CTC and administering Buscopan had an odds ratio of 6.49 for adequate colonic distention throughout all colonic segments compared to patients who did not receive it. 71
When administered, patients should be pre-screened for any potential contraindications, including acute coronary syndrome, uncontrolled cardiac failure, and arrhythmias, and should be withheld in these situations. Screening for glaucoma is a misnomer and technologists or nursing staff must receive adequate training to recognize contraindications and potential side effects of Buscopan as well as awareness to counsel patients for potential precipitation of acute glaucoma. 86 Side effects include urinary retention, dry mouth and blurred vision, Patients are counselled about these and advised not to drive following the procedure if they receive Buscopan. 71 Glucagon is a viable alternate to Buscopan in the setting of contraindications. Published data shows inferiority to Buscopan, 87 and its higher cost and less favourable side effects profile ensure it less commonly utilized than Buscopan.
Image Acquisition and Post Processing
• The images are acquired as a single breath hold with coverage of the entire colon and rectum as guided by the scout images. • Use a low-dose, non-enhanced CT with dose adaptation based on the patient’s body habitus. • Target a CT Dose Index Volume (CTDIvol) of 5 mGy or less per acquisition, or a Dose Length Product (DLP) of 600 mGy.cm or less for the whole exam.88,89 ○ This can be achieved by setting the tube voltage to 120 kVp and using automatic dose modulation with a reference tube current of 35-70 mA, or a minimum/maximum tube current 40 mA and 450 mA respectively. ○ Titrate parameters based on the patient’s body habitus to ensure diagnostic image quality. • Employ dose reduction techniques such as optimum positioning, automatic exposure control, and deep-learning image reconstruction. • Acquire thin section images (.6 – 1.5 mm) using a soft-tissue reconstruction algorithm. • View images on a workstation or as 2D multiplanar reconstructions, 3D intraluminal views, or both. • Optional: use digitally cleansed images.
Patient Positioning
The entire colon should be imaged in a minimum of 2 positions, ensuring adequate distension and utilizing any combination of supine, prone and decubitus techniques. Patient preference and anatomy can influence choice; in larger patients, in the setting of hernia(s) or a stoma a decubitus is invaluable. Similarly, the right lateral decubitus position90-96 is helpful for problem solving in patients with diverticulosis of moderate or greater severity.
IV Contrast
IV contrast is not used for routine screening CTC, only for staging CTC (in the setting of a known mass/neoplasm) or problem solving. The low-dose, alternate position should be acquired first to reduce artifacts from contrast being excreted into the ureters and urinary bladder.
It should be administered in the supine position thereafter and a standard technique (full dose) CT of the abdomen and pelvis should be acquired.
Dual Energy CT
Dual Energy CT (DECT) can improve subtraction of contrast material on virtual non-contrast images compared to electronic cleansing. Improved detection of polyps on virtual monoenergetic images has also been reported. DECT may also offer improved image quality due to the benefits of greater contrast from the low-energy images and reduced noise from the high-energy images.
Contrast-enhanced DECT may alleviate the need for bowel cleansing and fecal tagging while still offering high sensitivity and specificity for the detection of polyps, which would potentially improve patient acceptance of CTC as a screening modality.90-96
Quality and Safety Check
Following image acquisition, CT technologists should be adequately trained to recognize under distension and acquire additional images prior to discharging the patient. A safety check should include assessment for any potential perforation or extra-luminal air prior to discharge from the department.
Patient Preference
CTC leads to higher patient satisfaction97,98 and better screening compliance than colonoscopy. 99 Of CTC patients who had a prior colonoscopy, 77.1% preferred CTC and 13.8% preferred colonoscopy. 98 Preference for CTC is attributed to its minimally-invasive nature, limited bowel preparation, non-sedation and low risk of complications compared with colonoscopy.98,100 CTC is convenient, with 37% of patients saying they wouldn't have screened otherwise. 100 In post-operative patients, however, preference for colonoscopy was higher (42.6%) with no preference for 35.4%. In 1 study, only 3% of CTC patients experienced significant pain compared to 34% during colonoscopy; polyp detection shifted preference to colonoscopy. 101 In a comparative trial, participants expected colonoscopy to be more burdensome, but found CTC more burdensome after the procedure. 102 Use of carbon dioxide for colonic insufflation results in less pain and bloating compared to room air insufflation. CO2 usage in colonoscopy leads to less post-procedure discomfort and less retained colonic gas. 103
Cost-Effectiveness
The cost of a screening test such as CTC is often compared with the gold standard of OC. The average cost of a colonoscopy varies widely across international jurisdictions. In the Canadian setting, CTC compares favorably against OC when a rationalized referral approach is adopted (≥6 mm).
The cost-effectiveness of a screening test is also dependent on rate of participation as well as the numbers of screening rounds. The COCOS trial showed CTC was the most cost-effective strategy in participants who underwent >2 lifetime screens and was the preferred test by participants. 18 A recent Canadian study suggests that increasing the screening interval to 10 years would increase the cost-effectiveness of CTC. 104 However, the study failed to address several important considerations including patient compliance (higher in CTC) and the time commitment (both by the patient and their accompanying friend/family) when sedation is given. This loss of time also contributes to the overall economic burden imposed by OC and the authors acknowledge this likely undermined the true cost effectiveness of CTC.
Training and Certification
Recommended Requirements
• Physician with specialist training in diagnostic radiology and abdominal CT, equivalent to Royal College curriculum, is required. • Basic training in virtual colonoscopy, including didactic training and buttonology, should be completed with review of 50 cases with endoscopic correlation. • Cases should showcase a range of colonic pathology and complexity and include a mix of contrast and non-contrast techniques. • During training, the review process should use dedicated CTC software with instruction and supervision, including reporting and interpretation. • Double-reading and self-audit with endoscopy feedback is suggested. Awareness of 7-12% positive findings in average risk population helps benchmark referral rates for follow-up endoscopy.
Rationale
Various national and international bodies have published recommendations and benchmarks for radiologist training prior to independent reporting of CTC, though the minimum standards and suggested training methods vary. 105 (Appendix 7). The minimum standard is 50 pathology-proven cases with a spectrum of pathology, but there is limited detail about requirements for the spectrum of complexity assessed in these cases, nor are there stated requirements for PPV or polyp identification rates.
There is evidence that training in CTC interpretation incrementally improves reader sensitivity and performance with the number of cases read, 106 but reading a pre-determined number of cases is not a guarantor of performance. 107 Training and feedback are important components of diagnostic performance in addition to clinical experience. Studies which have focused on delivering feedback to readers, with or without tailored individualized training, have demonstrated significant gains in reader sensitivity.106,108,109
Maintenance of Competence
Maintaining the quality of CTC reporting is imperative to the credibility of the technique and will minimize perceptual errors when interpreting these exams. This in turn influences polyp or cancer detection and directly impacts patient outcome. 108
As of 2019, the ACR recommends a 50-case review with endoscopic correlation every 2 years. 88 Ideally, a periodic audit of all exams including endoscopic feedback and internal peer review process will help facilitate this ongoing learning and maintain reader sensitivity and performance. The CAR recommends that the latter is a more feasible and realistic approach in the Canadian context. This audit should be performed every 3 years as a minimum.
We recognize that this may be a challenging number to achieve in some centres. Possible solutions to increase reader volumes include: 1. Restricting CTC reading to those with expertise/interest in the area. 2. Continuing to double read to gain mutual experience and feedback.
Conclusion
CT colonoscopy is a mature technology with proven value add to the paradigm of colon cancer screening and problem solving in colonic pathology. Understanding its strengths and limitations in this regard equips the performing radiologist with a powerful tool to facilitate patient choice and provide a desirable outcome.
Supplemental Material
Supplemental Material - Canadian Association of Radiologists Practice Guidelines for Computed Tomography Colonography
Supplemental Material for Canadian Association of Radiologists Practice Guidelines for Computed Tomography Colonography by Tanya Chawla, Casey Hurrell, Valerie Keough, Chris M. Lindquist, Mohammed F. Mohammed, Caroline Samson, Gavin Sugrue and Cynthia Walsh in Canadian Association of Radiologists Journal
Footnotes
Acknowledgments
The authors would like to thank the members of the Canadian Association of Radiologists who took the time to provide their feedback and peer review during the drafting of these guidelines. The working group would like to extend a special thank you to Tracy Wakeford for her work at Mount Sinai to compile the patient-facing prep materials that were the basis for the ![]() , and to the other sites and institutions who shared their materials to contribute to the version appended here.
, and to the other sites and institutions who shared their materials to contribute to the version appended here.
Disclaimer
CAR Clinical Practice Guidelines are based on evidence review and consensus recommendations derived from the available evidence at the time of preparation. The guidance provided herein should not be utilized to establish a legal standard of care and should be interpreted in the setting of specific clinical situations and resource availability. Adherence to the guidance cannot assure an accurate diagnosis or a successful outcome. Deviation does not, in and of itself, indicate or imply that such medical practice is below an acceptable level of care. The ultimate judgment regarding the propriety of any specific procedure or course of conduct must be made by the physician and imaging care team considering all circumstances presented by the individual situation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
