Abstract

Introduction
Aside from trauma, multiple sclerosis (MS) is the most common cause of disability in young adults. 1 The greatest prevalence of MS has been reported at higher latitudes (>100/100,000), and the disease is 3 times more common in females .1,2 MS is the most common demyelinating disease, defined by demyelinating lesions in the central nervous system and their resultant neurological deficits. While the causative mechanisms are still actively being studied, certain risk factors have been observed, including genetic predisposition, vitamin D insufficiency, obesity, smoking, and Epstein–Barr virus infection.1,3 Progressive neurological deficits and demyelinating changes accumulate over time, although newer therapeutic agents have been developed which can achieve disease remission in some. 1
As part of the diagnostic work up, magnetic resonance imaging (MRI) helps exclude potential mimics of MS, such as cervical spondylosis with associated compressive myelopathy. It has been previously demonstrated, using MRI, that the prevalence of degenerative disc disease (DDD) dramatically increases with age. A classic paper by Modic et al. describes the range of DDD changes and shows that >85% of the population will show changes related to disc degeneration by the age of 50. 4 Associated myelopathy can potentially confound the interpretation of spinal cord lesions, erroneously fulfilling the diagnostic criterion of dissemination in space or signalling MS progression. As many as 90% of patients with MS may manifest with spinal cord signal abnormalities, most commonly in the cervical cord. 5 Concurrently, recent evidence suggests that cervical spine degenerative changes are more common in MS patients.6-8 While many factors have been implicated in cervical DDD, including disc dehydration due to natural aging and chronic flexion of the neck with increasing use of electronic devices, 9 the role of MS in this disease process is not well understood.
The purpose of the current study was to determine the presence and extent of degenerative disc disease of the cervical spine in young (<35) patients with MS, a cohort that is less well studied.
Methods
Patient Population
This retrospective study was approved by our institutional research ethics board (IRB) and the requirement for informed consent was waived. Medical record review was performed in accordance with IRB guidelines. We reviewed the MRI scans of consecutive patients aged <35 referred from The Ottawa Hospital MS clinic scanned between May 2005 and November 2014. 80 patients (51 female and 29 male) with MS of any type ranging between 16 and 32 years of age (average 26) were included. Patients with known history of cervical trauma, prior surgery, radiation, or spondylodiscitis were excluded.
Imaging Protocol
Each patient underwent an examination of the brain and cervical spine on a 3T MR scanner (Siemens Tim Trio, Siemens Medical Systems, Erlangen, Germany) using a 32-channel head coil. Protocols included the following sequences of the cervical spine: sagittal T2 FLAIR (Echo:1 TR:5000 ms TE:442 ms TI:1800 ms, Slice: 1.07 mm) with axial MPR reconstruction, sagittal T2 (Echo:1, TR:3300 ms, TE:108 ms, Slice: 3.00 mm), sagittal PD (Echo:1, TR:3050 ms, TE:13 ms, Slice:3.00 mm) and axial T2* MEDIC 2d (Echo:1, TR:755 ms, TE:12 ms, Slice: 3.00 mm). None of the studies included gadolinium enhanced sequences of the spine.
Image Assessment
MRI grading system for cervical spine degenerative disc disease (DDD).

Separately, the location of the cord signal abnormality was assessed in relation to the level of the degenerative changes, regardless of etiology (i.e., spondylotic myelopathy or MS plaque). Grade 0 was defined as no cord signal changes; grade 1 when cord signal change occurred only at the same level as DDD; grade 2 when cord signal changes occurred only at levels not affected by DDD; and grade 3 when cord signal changes occurred at both levels affected and unaffected by DDD.
Inter-rater agreement for DDD level, severity (5 point scale), and cord signal change was assessed using Kendall’s W coefficient of concordance. Inter-rater agreement for the binary classification of DDD severity (Miyazaki et al.) was assessed using Fleiss’ kappa. A Kendall’s W or Fleiss’ kappa of ≤.2 indicated poor agreement; .21-.40 fair agreement; .41-.60 moderate agreement; .61-.80 substantial agreement and .81-.99 indicated very good agreement. Differences in the proportions of grade 2 and 3 combined vs grade 1 cord signal change ratings were analyzed for each rater with Fisher exact tests.
Results
There was substantial agreement between readers for the assessment of DDD level (Kendall’s W = .61, 95% CI 0.21-.74) and severity (Kendall’s W = .67, 95% CI 0.32-.79). Inter-rater agreement was very good for the assessment of cord signal change (Kendall’s W = .81, 95% CI 0.63-.86). When DDD severity was assessed using the binary Miyazaki classification scale, inter-rater agreement was considered moderate (Fleiss’ kappa = .53, 95% CI 0.40-.66).
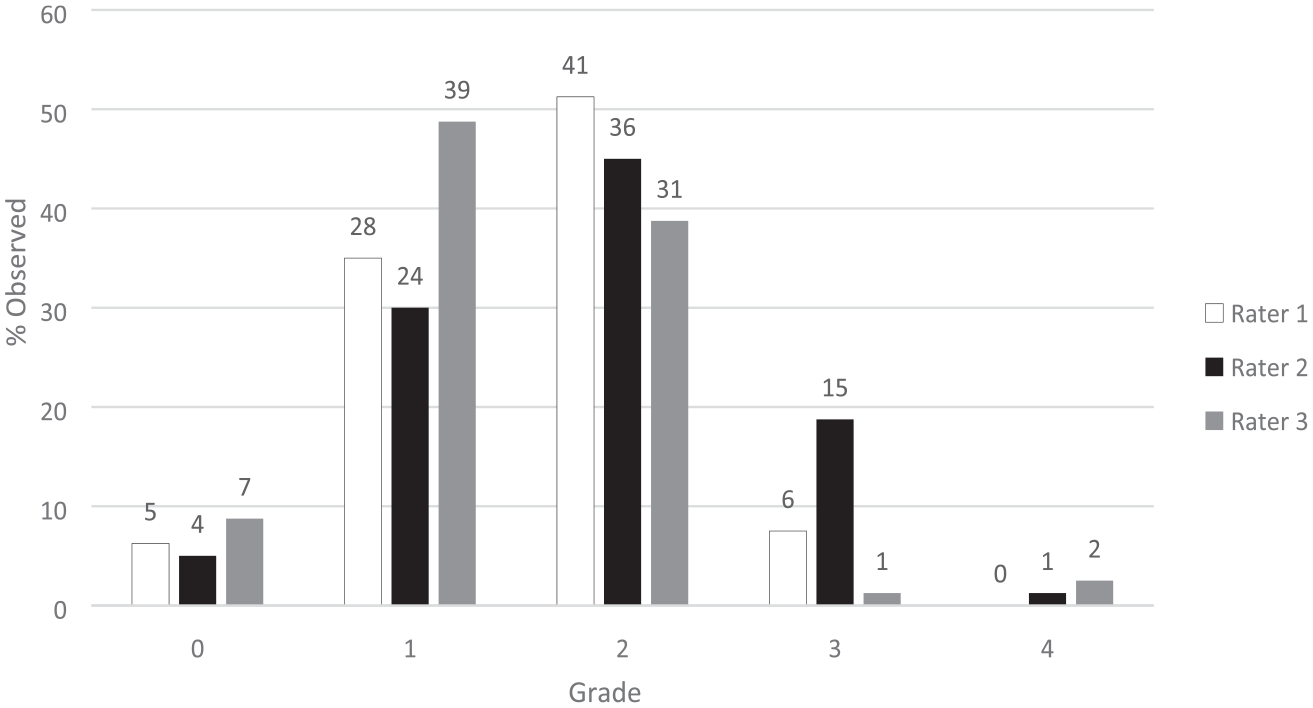
Figure 1 shows the DDD distribution ratings obtained from the 3 raters. No DDD (grade 0) was found in 5 (6%), 4 (5%), and 7 (9%) of patients by raters 1, 2, and 3 respectively. Single level DDD (grade 1) was found in 16 (20%),17 (21%), and 12 (15%) of patients by raters 1, 2, and 3 respectively. Multilevel DDD (grade 2) was found in 59 (74%) by both rater 1 and 2, and in 61 (76%) by rater 3. Figure 2 shows the DDD severity ratings from the raters. Grade 0 severity score matched the Grade 0 distribution score, by definition. Grade 1 severity was assigned in 28 (35%), 24 (30%), and 39 (49%) of cases, grade 2 in 41 (51%), 36 (45%), and 31 (39%), grade 3 in 6 (8%), 15 (19%), and 1 (1%), and grade 4 in 0 (0%), 1 (1%), and 2 (2.5%) patients (raters 1, 2, and 3 respectively). Converting the severity scores into the binary Miyazaki classification yielded 75 (94%), 76 (95%), and 73 (91%) patients with grade III-V DDD for raters 1, 2, and 3 respectively. Distribution of degenerative disc disease. N = Number above each bar. Severity of degenerative disc disease. N = Number above each bar.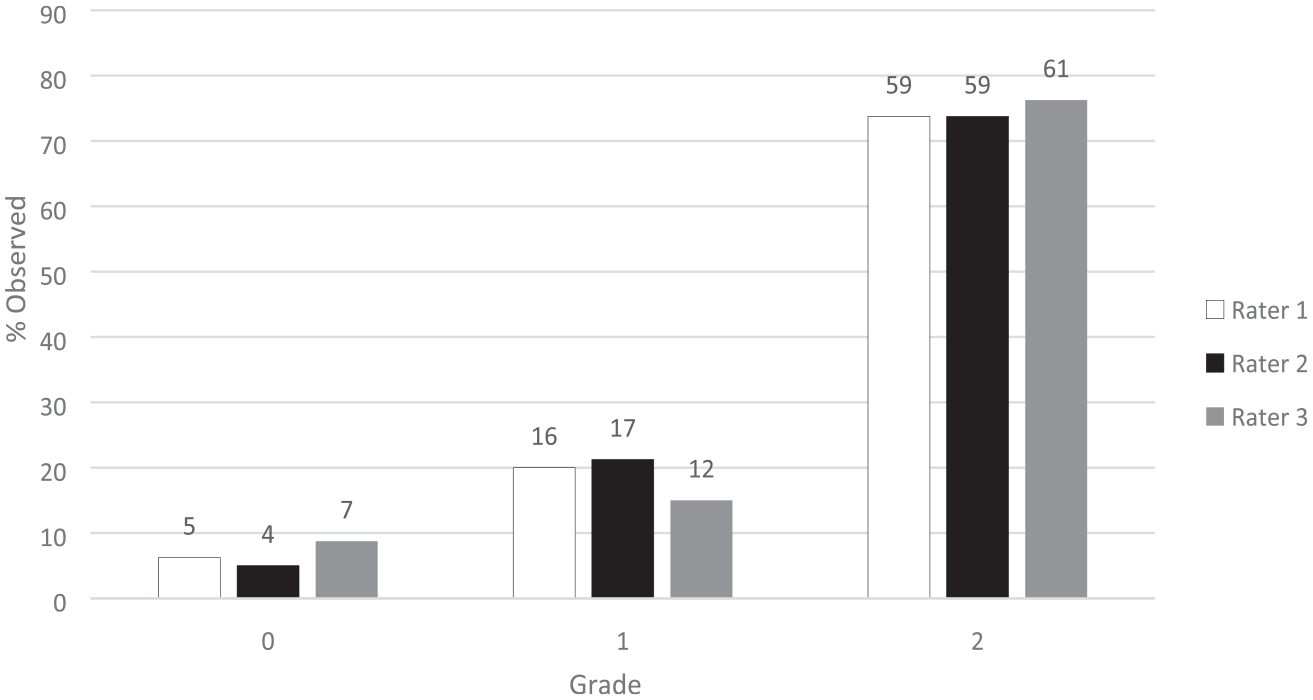
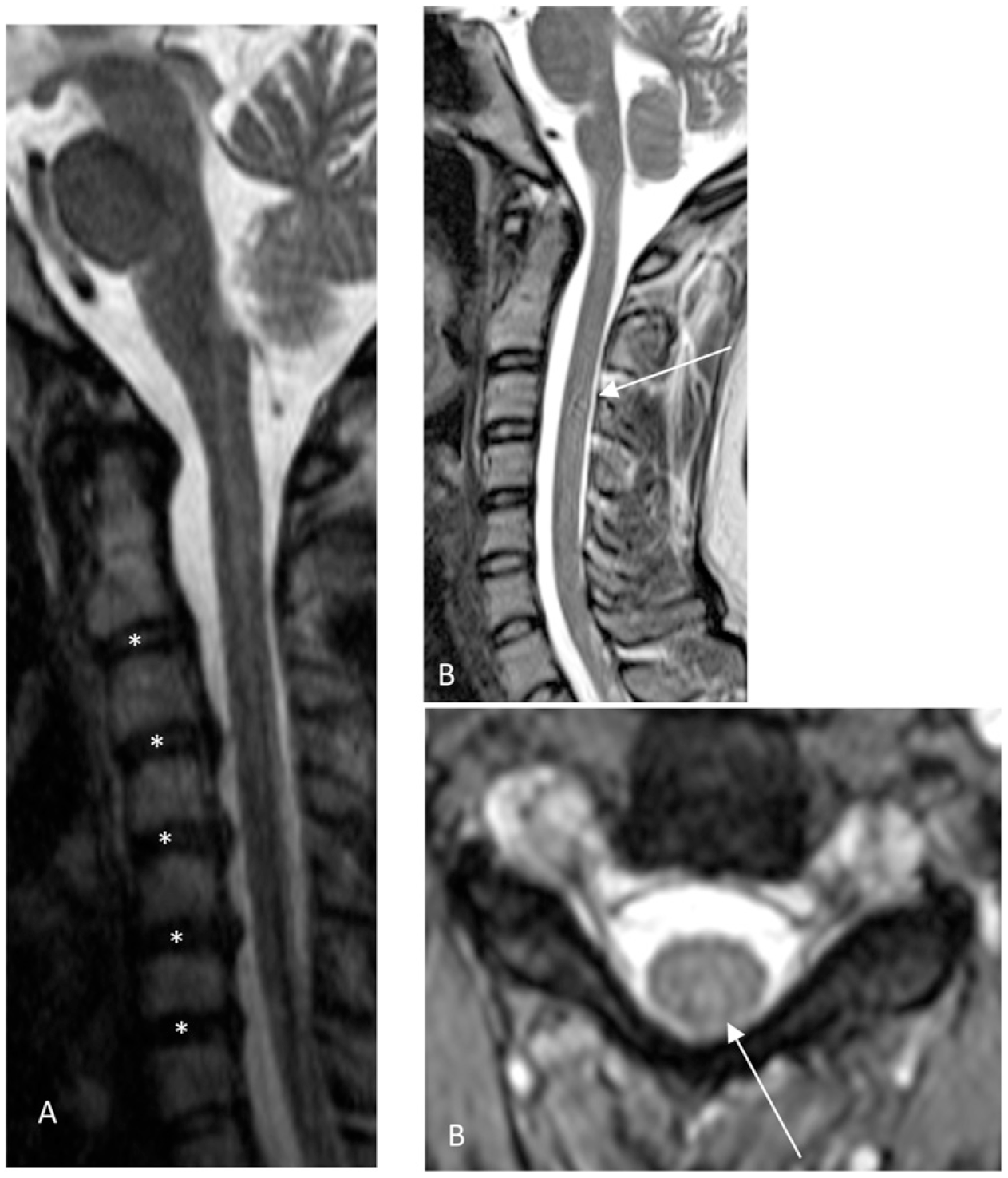
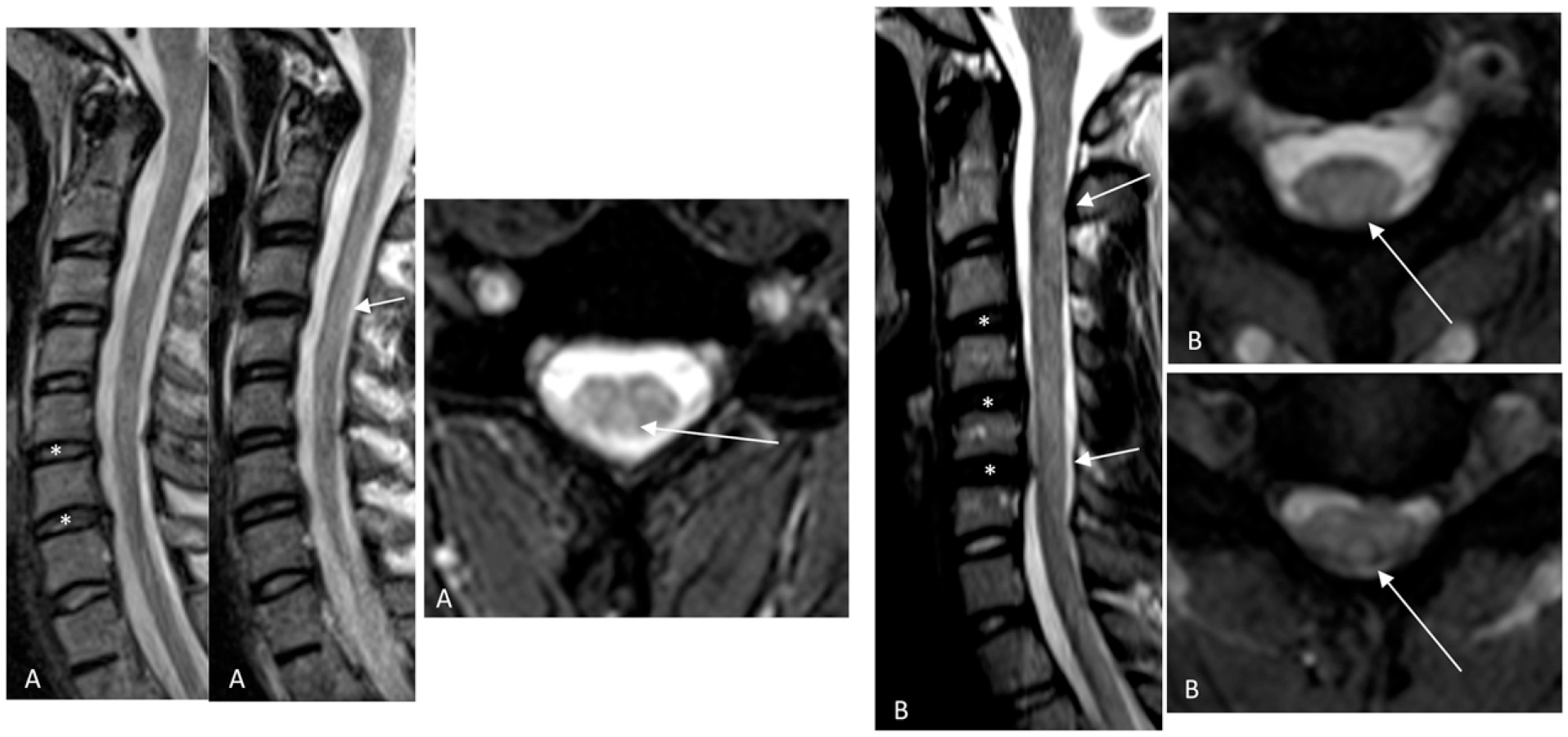
Cord signal change in relation to DDD level is shown in Figure 3. The raters did not find any cord signal abnormality (grade 0) in 35 (44% for raters 1 and 2), and 30 (38% for rater 3) patients. Grade 1 was found in 12 (15%), 8 (10%), and 9 (11%) patients, grade 2 in 24 (30%), 31 (39%), and 15 (19%) patients, and grade 3 in 9 (11%), 6 (8%), and 26 (33%) patients, by raters 1, 2, and 3 respectively. The proportion of patients graded 1, representing cases where cord signal occurred exclusively at degenerative disc levels, was significantly lower than other cord signal distributions in rater 1 (12/80 vs 33/80), rater 2 (8/80 vs 37/80), and rater 3 (9/80 vs 41/80), with P < .001 for all tests. Figures 4 and 5 highlight the spectrum of MRI findings and application of the scoring system. Cord signal and level of degenerative disc disease. N = Number above each bar. Opposite ends of the spondylosis – demyelination spectrum in the cervical spine of MS patients. A) Sagittal T2-weighted image of a 17-year-old female with multilevel degenerative disc disease (*) but no cord signal abnormality (distribution score 2, severity score 3, and cord signal score 0 by all raters, except for rater 3 who scored severity 2). B) Sagittal T2-weighted and axial T2*-MEDIC sequences of a 26-year-old female demonstrate no significant degenerative changes in the cervical spine but cord signal abnormality in the upper cervical levels as seen on the axial plane (arrow). The lesion is not well seen on the sagittal plane (arrow points to corresponding axial image level). All raters graded distribution/severity score 0, and cord signal score 2. Intermediate findings of both degenerative changes and demyelination in the cervical cord of MS patients. A) Sagittal T2-weighted and axial T2*-MEDIC sequences of a 30-year-old male with degenerative disc disease (*) at different levels from the cord signal abnormalities (arrows, best seen on axial). Severity was graded 3 by rater 2 and grade 2 by raters 1 and 3; they all rated distribution and cord signal as 2. B) Sagittal T2-weighted and axial T2*-MEDIC sequences of a 25-year-old female with diffuse cervical cord signal abnormalities (arrows) both at degenerative (*) and nondegenerative levels. Cord signal abnormality extended from C2-T2 (not shown). All raters graded distribution as 2, severity as 3, and cord signal as 3, except rater 3 graded severity as 4.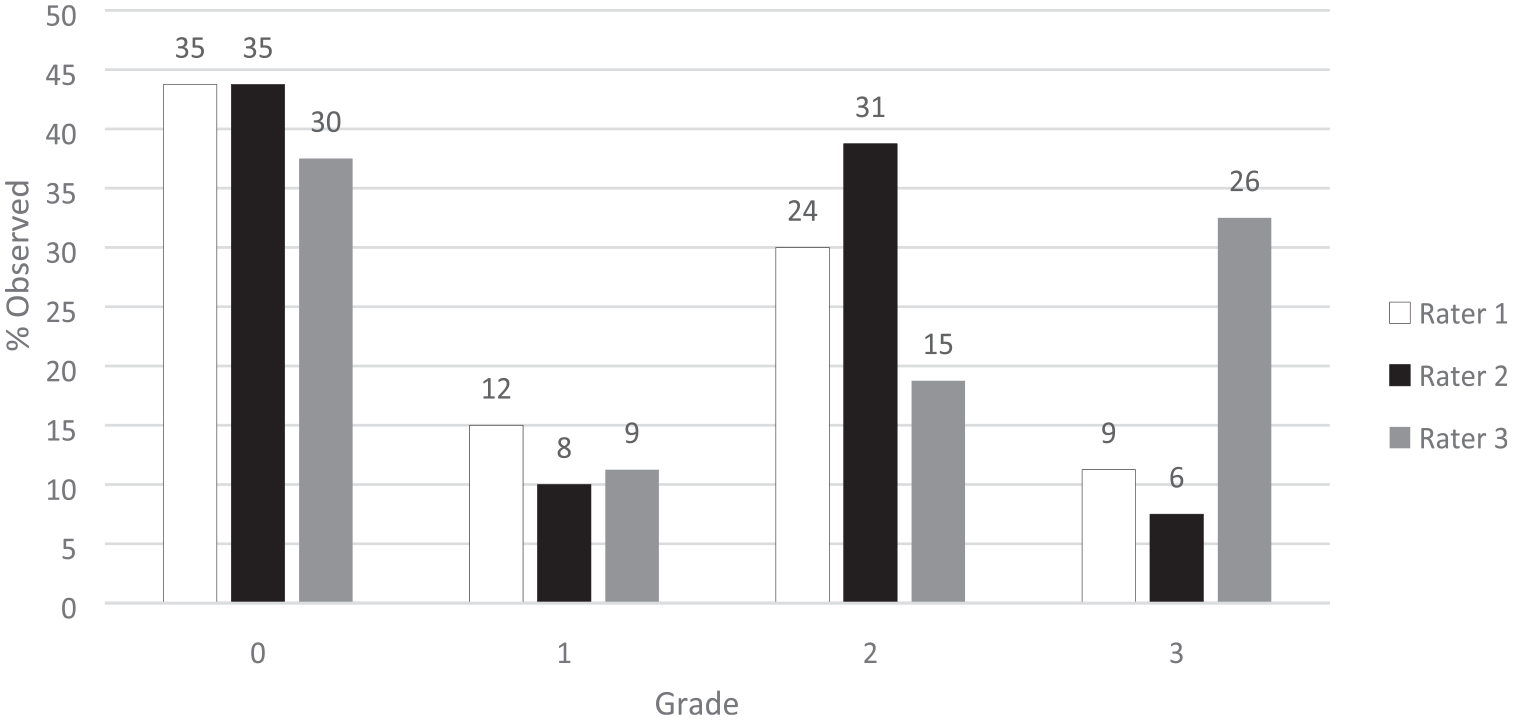
Discussion
Spinal cord demyelinating plaques are a hallmark feature of patients with multiple sclerosis, yet relatively few studies have examined the presence and prevalence of associated degenerative disc changes in the cervical spine. In the general non-MS population, cervical DDD becomes common typically after the age of 40, and if present at a younger age, it begins at higher spine levels with progressive disc signal loss. 11 Limited data exists on the prevalence of DDD in young (<30) asymptomatic patients, with sample sizes too small 12 to enable meaningful comparisons with our study. In a larger prospective cohort study of the general population in Japan, 26.3% of men and 27.9% of women aged 21-49 demonstrated cervical DDD, 13 significantly less than what we found in our study with younger subjects ranging between 16 and 32 years of age in whom over 90% had some DDD (i.e. scored 1 or higher on our severity scale). However, comparison is limited given differences in DDD grading, as only the Pfirrmann classification was used in that Japanese study, ignoring morphologic disc changes. 13 While our data suggests a greater than expected prevalence of DDD for age among MS patients, further studies are needed with age-matched healthy controls for confirmation. Recent case-control studies however have supported our results.6,7
The mechanism by which DDD may be accelerated in MS patients also requires further study. One theory suggests that inadequate posture of the cervical spine in MS patients may alter biomechanics leading to early degenerative changes. 7 Worsening posture is associated with increased disability, 7 which may be in part due to cord or nerve root compression. Another proposed mechanism is that the increased fibronectin seen within the microvasculature and macrophages of MS plaques 14 may also occur in the intervertebral discs, as fibronectin has been shown to accelerate disc degeneration. 15 In our study, a common clinical feature among patients with the highest severity scores (3/4) was hyperreflexia and spasticity. Given that spinal loading is predominantly determined by muscular forces, greater muscle tone and dysregulated contractions may overload the discs and accelerate DDD. 16 Conversely, studies show that reduced mobility can also accelerate DDD. 16 Patients with MS are known to be less physically active, likely due to fatigue, weakness, and poor coordination. 17 A combination of these factors likely contributes to DDD, but dedicated studies are needed.
The etiology of cord lesions in MS has been questioned by some.18,19 In 1988, Al-Mefty et al. described compressive myelopathic MRI changes in the cervical cord related to degenerative disease in non-MS patients, 20 which have since been firmly established, and similarly seen in traumatic cases. Interestingly, the pathophysiology of MS has been theorized to include traumatic in addition to autoimmune mechanisms. 18 Ocak et al. reported that the presence of cervical disc herniations and MS plaques were correlated and occurred at the same levels, suggesting that the plaques were caused by disc herniation and micro-trauma. 19 Studies of cervical spine biomechanics in football players have demonstrated lesions in the spinal cord even in the absence of bone or joint injuries, 21 in theory supporting an “occult trauma” mechanism in MS. However, most spinal cord lesions in our study were located at different levels from the sites of DDD. Our findings support the idea that the degenerative disc changes in the cervical spine are independent of MS plaques, and that compressive myelopathy represents a separate disease process.
This single institution retrospective cross-sectional study has several limitations. First, our lack of a control group limits our conclusions as DDD is naturally prevalent in the general population. However, we have uniquely selected a very young population ranging between 16 and 32 years of age, which would be expected to have little to no degenerative disease. 9 Second, our sample size is relatively small. Third, the retrospective design prevents assessment of DDD progression or response to MS treatment. Finally, the classification of severity used in our study is not a standardized measure, limiting our comparison to other studies. However, we benchmarked our severity scale against a validated scale and internal validity was demonstrated by substantial to very good interrater agreement.
Conclusion
Our results demonstrate that young patients with MS show various degrees of DDD in the cervical spine, greater than expected for age. Furthermore, we found no clear association between demyelinating plaques in the spinal cord and the degree of degenerative changes and therefore our study does not support a micro-traumatic etiology for MS. While not the objective of the current study, we postulate that early degenerative changes may be related to altered biomechanics. Future studies with clinical correlates such as cervical posture, muscle tone, and physical activity levels are needed to better understand the pathophysiology of DDD in the MS population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
