Abstract
Pancreatic cystic lesions (PCLs) are both common and often incidental. These encompass a range of pathologies with varying degrees of concern for malignancy. Although establishing a diagnosis is helpful for determining malignant potential, many PCLs are either too small to characterize or demonstrate nonspecific morphologic features. The most salient modalities involved in diagnosis and surveillance are magnetic resonance imaging, multidetector computerized tomography, and endoscopic ultrasound. Fine needle aspiration has a role in conjunction with molecular markers as a diagnostic tool, particularly for identifying malignant lesions. Although several major consensus guidelines exist internationally, there remains uncertainty in establishing the strength of the association between all PCLs and pancreatic adenocarcinoma, and in showing a benefit from extended periods of imaging surveillance. No consensus exists between the major guidelines, particularly regarding surveillance duration, frequency, or endpoints. This review paper discusses PCL subtypes, diagnosis, and compares the major consensus guidelines with considerations for local adaptability along with questions regarding current and future priorities for research.
Background
Pancreatic cystic lesions (PCLs) are present on up to 49% of abdominal magnetic resonance imaging (MRI) exams making these a common incidental finding, one that likely increases with age. 1 Definitions vary but a PCL is generally considered to be any closed sac-like structure with fluid or semi-fluid content within the pancreas on imaging, with or without communication with the main pancreatic duct (MPD). 2 In recent years, PCLs have been more widely detected than in the past, which could be due to increased usage of more sensitive imaging techniques such as MRI, MRI technological advances, an aging population, or due to a true increase in PCL prevalence.3,4 However, the overall mortality due to pancreatic malignancy has not changed to the same extent as the reported prevalence of PCLs.5,6 This challenges the notion that all PCLs have a strong association with pancreatic carcinoma as has been suggested in older publications.7-9
Pancreas cystic lesions can be neoplastic such as intraductal papillary mucinous neoplasms (IPMNs), mucinous cystic neoplasms (MCNs), or serous cystadenomas, or non-neoplastic such as pancreatitis-related collections or congenital lymphoepithelial cysts. Although establishing a diagnosis is helpful for determining malignant potential, many PCLs are either too small to characterize or demonstrate nonspecific morphologic features. Most incidental PCLs are likely branch-duct IPMNs, which can manifest as solitary lesions but are frequently multifocal.10,11 To risk-stratify these lesions, morphological features on imaging are routinely evaluated including PCL size, enhancing mural nodularity, wall thickening/enhancement, MPD dilatation, and others.11,12 Some PCLs may arise due to an underlying abnormality within the pancreas that also increases the risk of developing pancreatic adenocarcinoma elsewhere in the gland, known as the field defect theory, similar to hepatocellular carcinoma arising on a background of hepatic cirrhosis. 6 Multiple guidelines have been developed for the follow-up of incidental PCLs recommending many years to lifelong surveillance, including from the American College of Radiology (ACR), American College of Gastroenterology (AGA), the Canadian Association of Radiology (CAR), and others.5,13-16 Management is primarily based on PCL morphology and generally involves either continued imaging surveillance, further assessment using endoscopic ultrasound (EUS) or referral for surgical resection.
Considering the high prevalence of PCLs detected on abdominal MRI, the optimal management and follow-up strategy for incidental PCLs remains uncertain. Benefits of early detection of pancreatic adenocarcinoma must be weighed against the trade-offs of the costs, resource requirements, emotional impact, and other aspects of long-term surveillance of many patients. More low-bias data are needed establishing an association between PCLs and pancreatic adenocarcinoma, and showing a benefit from often extended periods of imaging surveillance. More optimal risk-stratification could focus surveillance strategies on those patients most likely to benefit.
In this article, we review the major consensus guidelines covering PCLs, the salient imaging modalities, the role for essential diagnostic tools including fine needle aspiration (FNA), association with malignancy, and current and future considerations for surveillance.
PCL Guidelines
Imaging and Biochemical Features of Intraductal Papillary Mucinous Neoplasms Requiring Further Workup Beyond Imaging as per the 2017 Revised Fukuoka Guidelines.

Adapted from Tanaka M, Fernandez-Del Castillo C, Kamisawa T, et al. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology 2017; 17:738–753.
Comparison of Major Pancreatic Cystic Lesion Guidelines.
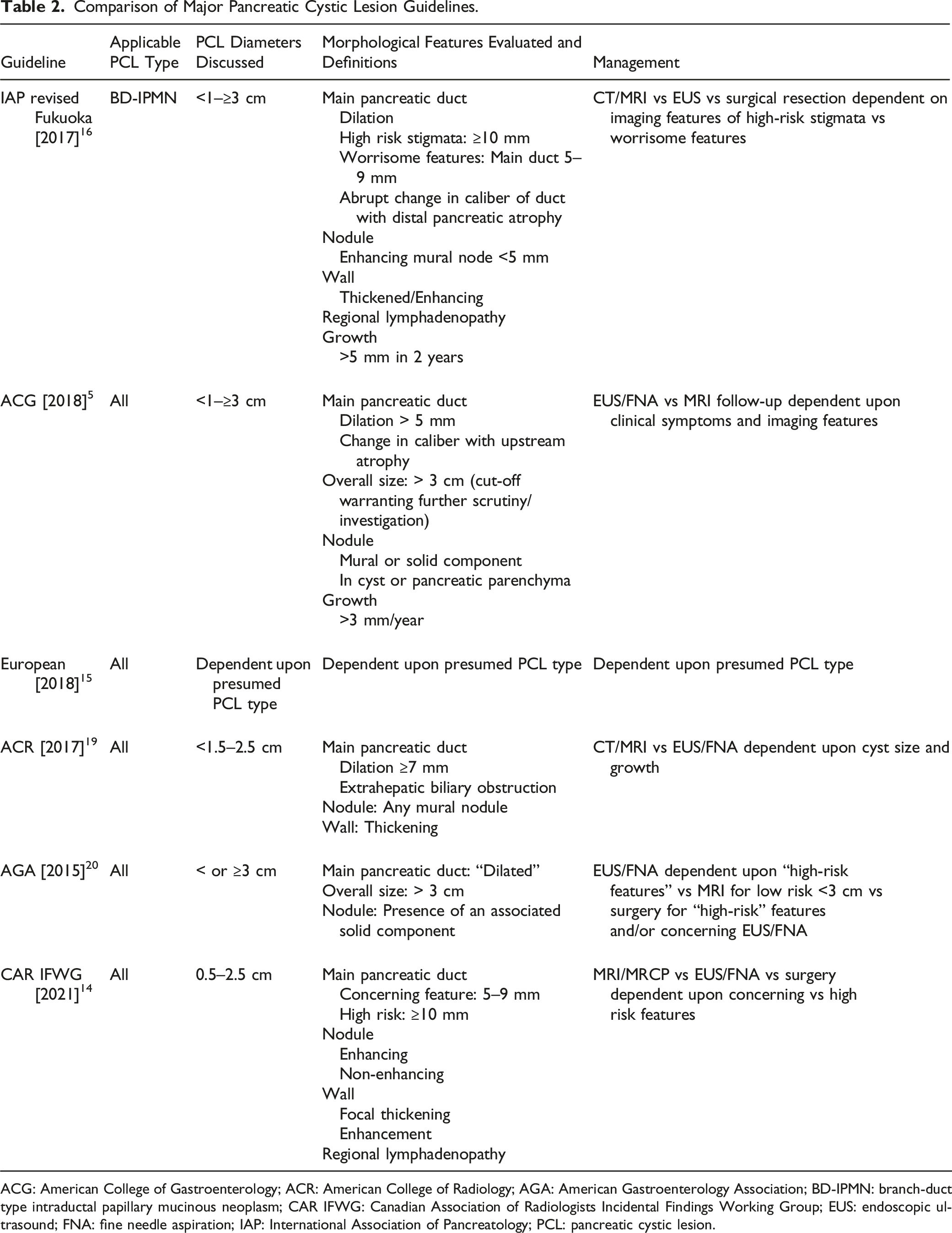
ACG: American College of Gastroenterology; ACR: American College of Radiology; AGA: American Gastroenterology Association; BD-IPMN: branch-duct type intraductal papillary mucinous neoplasm; CAR IFWG: Canadian Association of Radiologists Incidental Findings Working Group; EUS: endoscopic ultrasound; FNA: fine needle aspiration; IAP: International Association of Pancreatology; PCL: pancreatic cystic lesion.
Imaging Modalities (MRI vs MDCT vs EUS)
Pancreatic cystic lesions can be visualized on a variety of imaging modalities but owing to superior soft tissue contrast, magnetic resonance imaging/magnetic resonance cholangiopancreatography (MRI/MRCP) is often the preferred modality for non-invasive assessment. 1 Given the ubiquity of multidetector CT (MDCT) which is often used to image the abdomen prior to MRI, it is important to understand how it too performs for diagnosing and assessing PCLs. One study found that MDCT had an area under the curve of 0.76 and 0.82 for diagnosing malignancy within a PCL 23 while another found an area under the curve score of 0.639 and 0.806 in distinguishing malignant PCLs from benign lesions.24,25 In a study by Sahani et al, pancreatic protocol MDCT demonstrated sensitivities of 71–94% in detecting mural nodularity, MPD communication, and internal septations. 26 Udare et al 27 found that MDCT and MRI had sensitivities and specificities for differentiating benign and malignant PCLs that did not significantly differ in a meta-analysis that included only primary studies that had head-to-head direct comparisons of MDCT and MRI in the same cohort. 27 A benefit of MDCT is that it has superior depiction of calcification compared to MRI or EUS.
The superior soft tissue contrast of MRI/MRCP allows for improved depiction of ductal anatomy when compared to MDCT. 1 One study found that MRI/MRCP had an area under the curve from 0.85 and 0.91 for diagnosing malignant PCLs. 23 Particularly regarding PCLs measuring less than 3 cm, MRI/MRCP was more sensitive for detection of these lesions compared to MDCT. 23 The sensitivities of MRI/MRCP for assessing MPD communication and internal septations has been reported to be 91% and 100%, respectively.23,28 Also found in the meta-analysis by Udare et al, MRI had higher specificity than EUS for diagnosing malignant PCLs, and contrast-enhanced MRI was more sensitive than unenhanced MRI for diagnosing malignancy. 27
Endoscopic ultrasound can be used for both diagnostic and therapeutic purposes, for example, to drain pancreatitis-related collections. 29 The coupling of EUS with FNA provides an advantage relative to MDCT and MRI/MRCP, as both PCL fluid can be aspirated, and solid tissue can be biopsied. Endoscopic ultrasound can detect mural nodularity, other solid components, internal septations, vascular involvement, and lymph node metastases. 1 In a study by Kim et al, EUS was found to have a sensitivity of 91%, a specificity of 86%, and an accuracy of 88% for differentiating solid and cystic pancreatic lesions. 30 Analysis of EUS fluid aspirate can aid in diagnosis. Aspirate contents of interest include amylase, CEA, and mucin among others. 29
PCL FNA
Endoscopic ultrasound-guided PCL fluid aspiration is an essential diagnostic tool as PCLs can progress from adenomas to invasive ductal carcinomas via genetic mutations, gene silencing, and chromosomal deletions. 31 The molecular markers present in fluid samples include amylase, carcinoembryonic antigen (CEA), CA 19-9, and CA72-4. The presence of amylase is specific to the diagnosis of a pancreatitis-related collection; in high concentrations above 1000 ng/mL this marker is most suggestive of a pancreatitis-related collection, whereas below 250 ng/mL, this diagnosis is essentially excluded. The presence of CEA correlates highly with the diagnosis of mucinous cystic lesions (i.e., MCN and IPMN), especially at levels above 192 ng/mL. Furthermore, levels above 1000 ng/mL have been shown to be suggestive of malignancy, with the assumed risk proportionally increasing as the level increases. The additional presence of mucin within fluid aspirate containing CEA has been shown to further aid the diagnosis of a mucinous cystic lesion. Finally, in terms of further differentiating benign vs malignant mucinous lesions, the combined presence of multiple tumor markers in cyst fluid is most helpful for diagnosis. 31 For example, a study by Shin et al 32 revealed a specificity of 88% and a sensitivity of 89% when combining CEA and CA 19-9 levels. 32 Cytology of fluid aspirate can be very useful for diagnosing malignancy if an adequate volume of fluid and cells are obtained, however, this can be a challenge when sampling small PCLs.
Endoscopic ultrasound can also be used to sample the PCL wall and any other solid components.33,34 This may improve diagnostic yield in addition to fluid aspiration, and is another advantage afforded by EUS relative to MRI and MDCT.
PCL Association With Malignancy
An important consideration driving concern and controversy with regards to PCLs is their strength of association with malignancy. This has implications for follow-up frequency and duration, choice of modality, considerations around intravenous contrast usage, healthcare costs, and patient counselling. 35 Earlier literature on branch-duct IPMNs reported a high percentage of these to be malignant, for example, a mean rate of malignancy including carcinoma in situ of 25% and invasive malignancy of 15%. 17 These studies typically consisted of small patient cohorts ranging from 17 to 60 in number.7-9,36-40 Many exclusively reported malignancy rates for PCLs that had undergone surgical resection which is likely a major source of selection bias.7-9 These limitations may have contributed to an overestimation of the association of PCLs with malignancy.
More recent works examining larger patient cohorts have found much lower incident rates of malignancy.41,42 A retrospective, high-powered cohort study of 2034 patients with PCLs and 6018 patients without PCLs imaged using mostly MDCT across 10 years found a three-fold increased risk of pancreatic ductal adenocarcinoma (PDAC) without increased all-cause mortality in patients over 65 years of age; for patients younger than 65 years, there was an association with increased all-cause mortality. 41 Incidence rate of pancreatic malignancy was 0.7 cases per 1000 patient-years of follow-up. 41 In another study, long-term follow-up of 267 patients with incidental PCLs smaller than 2 cm and greater than 0.5 cm for an average of 8.6 years (median of 9.2 years) found 0.9 cases of pancreatic malignancy per 1000 patient-years of follow-up. 42 This was similar to patients without PCLs.
The role of improving imaging technology may be a contributing factor to the increased incidence of PCLs. An analysis of 500 patients between 2005 and 2014 determined newer versions of MRI software and hardware led to higher numbers of PCLs being detected, in 30% of patients imaged with older hardware and 56% of patients imaged with newer hardware.
3
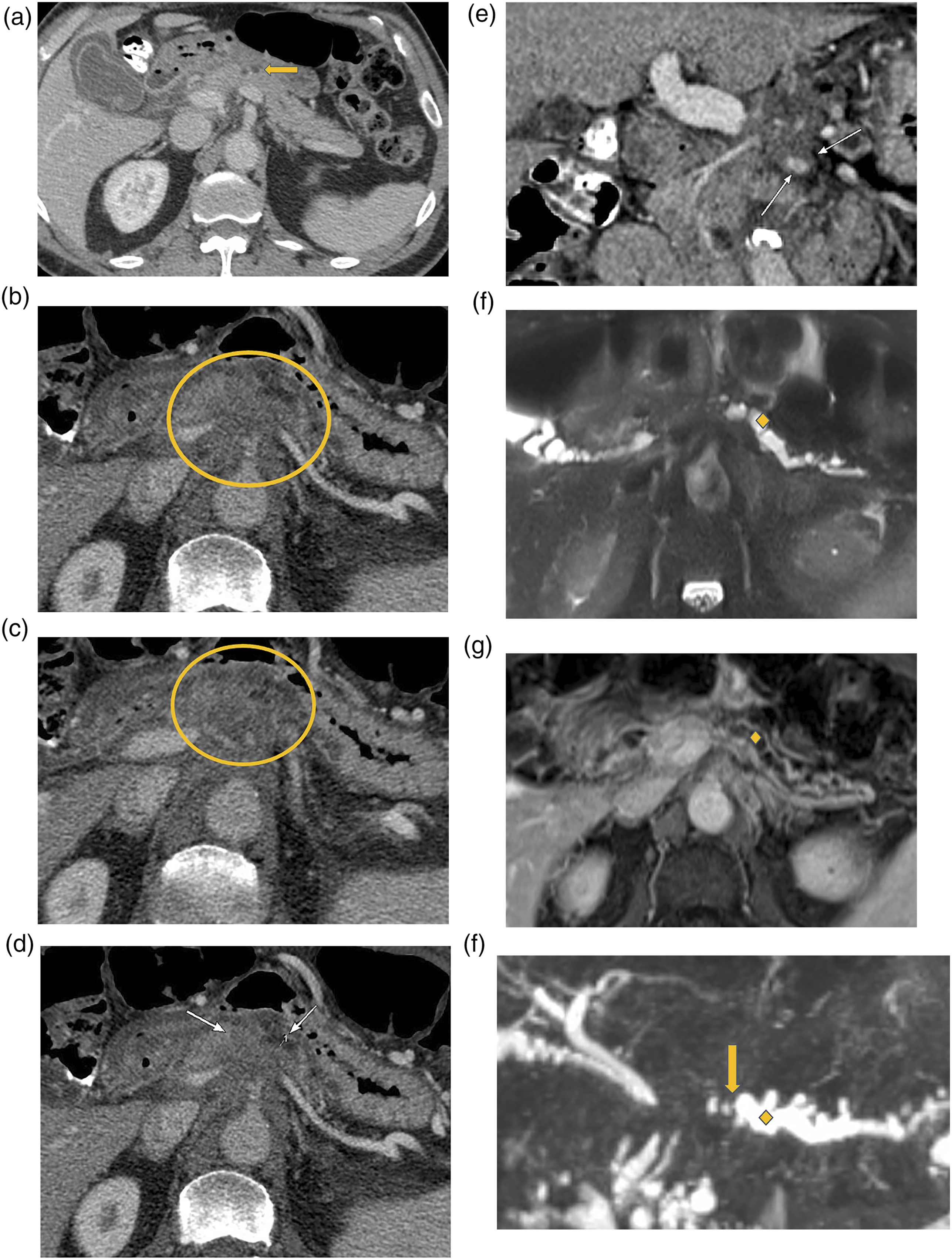
The combination of increased PCL detection, and recent evidence supporting relatively lower incidence of PCL-associated malignancy, prompt the need to re-evaluate surveillance strategies. Figures 1 and 2 are examples of PCLs that remained stable for the duration of follow-up, with the lesion in Figure 2 having undergone FNA which was negative for malignancy. Figure 3 highlights malignant transformation of incidentally detected PCLs in patients who may have benefited from surveillance imaging and early detection. Fifty-year-old female with a 2 cm uncinate process cystic lesion (yellow circle), present since CT in 2007 and followed until 2019. (A-C) Axial T2, axial T2 fat-saturated, and axial T1 post-gadolinium images from index MRI from August of 2015. (D-F) Axial T2, axial T2 fat-saturated, and axial T1 post-gadolinium images from follow-up MRI May 2018. (G) Axial PV phase CT in 2019. The lesion demonstrated septations with minimal enhancement and no nodules or worrisome features. No intralesional calcifications on CT. The differential considerations were for a branch duct IPMN vs serous cystic neoplasm. No FNA was performed. MRI: magnetic resonance imaging; CT: Computerized tomography; IPMN: intraductal papillary mucinous neoplasms; FNA: Fine needle aspiration. Sixty-seven-year-old female with a PCL at the time of initial MRI in 2015 (yellow circle). (A-B) Axial T2 fat-saturated and MRCP images from MRI 2015. Interim FNA [prompted by increase in lesion size] performed in January 2017 demonstrated mildly elevated amylase, normal CEA, and no malignancy. (C-E) Axial T2, MRCP, and axial T1 post-gadolinium images on MRI preceding FNA. (F-G) Axial T2 and MRCP images on final follow-up MRI. As per the last clinical note, optimal follow up had been achieved as per CAR guidelines with absence of worrisome features and due to patient age on the last follow-up (73 years). Recommended follow up with primary care provider. PCL: Pancreatic cystic lesions; MRI: magnetic resonance imaging; FNA: Fine needle aspiration. Seventy-two-year-old male. Initial PV phase CT in October 2016 (A) was performed for abdominal pain and elevated liver enzymes demonstrated circumferential gallbladder wall edema, no cirrhosis, and a hypodense pancreatic lesion (yellow arrow) without evidence of pancreatitis, for which imaging follow-up was recommended. Patient presented with pancreatitis and weight loss in May 2018 and found to have pancreatic cancer. (B-E) Axial and coronal PV phase images demonstrate a hypoenhancing pancreatic neck/body mass (yellow circle) with vascular encasement involving the splenic vein, portosplenic confluence, and superior mesenteric vein (SMV) (white arrows). (F-H) Axial T2, axial T1 post-gadolinium, and MRCP radial images demonstrate distal pancreatic ductal dilatation (yellow diamond) with upstream abrupt cut-off (yellow arrow) at the level of the mass.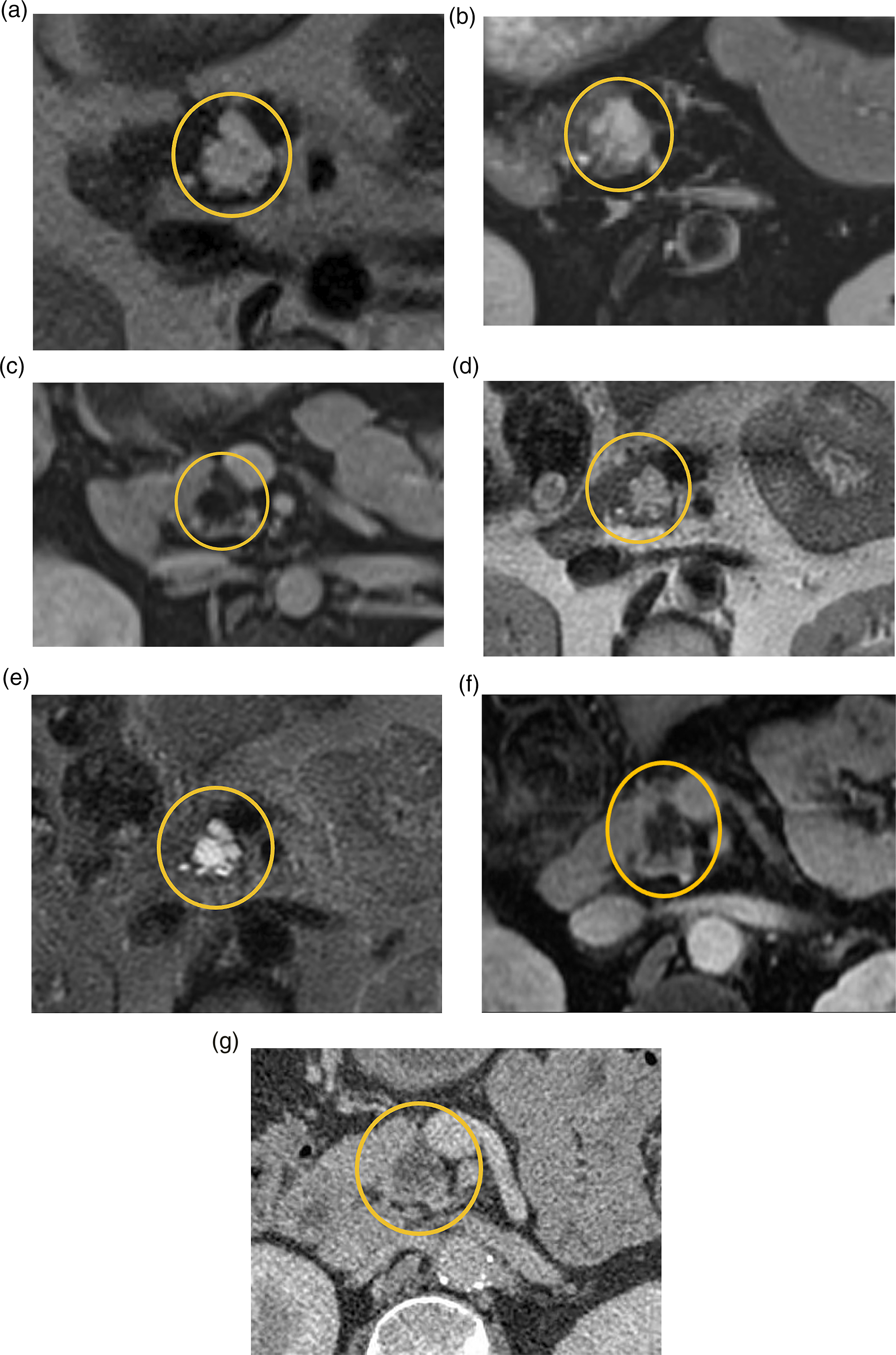
The concept of a field defect refers to PCLs as background parenchymal abnormalities that may predispose to future alternate-site pancreatic malignancy.
6
Multiple studies support the field defect theory, including most recently a study by Ikegawa et al
43
that found a relationship with multifocal PCLs and the incidence of PDAC synchronous with IPMNs.
43
Instances of malignancy that subsequently developed were at a separate location from the PCL that was being followed.
42
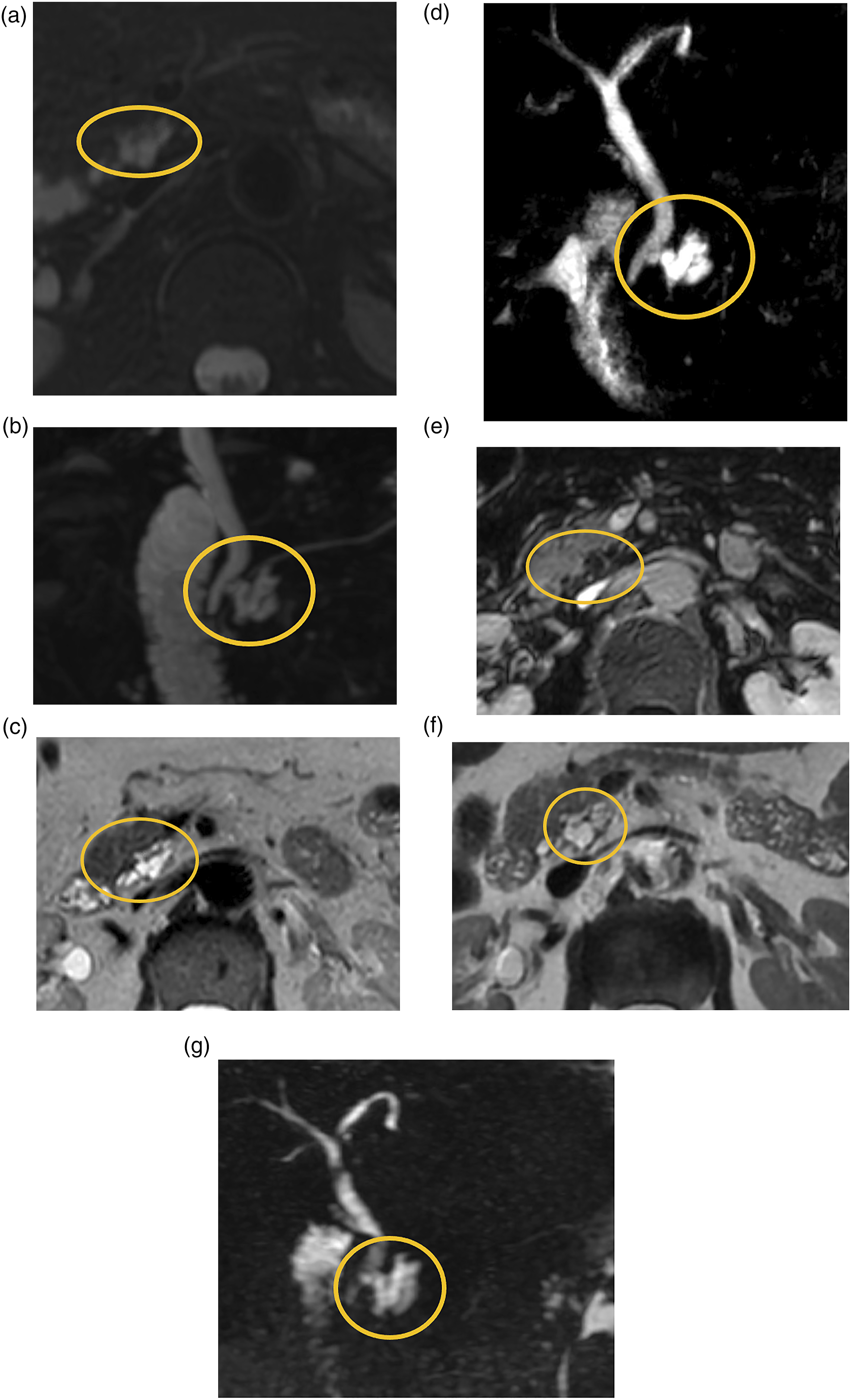
A consideration is whether decreased frequency of follow-up for PCLs below 2 cm and increased follow-up frequency if PCLs exceed 2 cm in size may help maximize utility of follow-up examinations. Figure 4 provides an example of a PCL exceeding 3 cm in size accompanied by MPD dilatation >5 mm, thereby meeting more than one revised Fukuoka guideline worrisome feature. This was a case of surgically resected IPMN found to contain invasive carcinoma. Fifty-seven-year-old male with initial MRI in October 2016 demonstrating a 3.2 cm pancreatic cyst (yellow circle) with distal ductal dilation (yellow diamond), meeting both worrisome features and high-risk stigmata (A-B, Coronal T2 and axial T2 images). EUS-guided FNA showed atypical ductal epithelium/IPMN. Subsequently, the patient presented 12 months later with pancreatitis and jaundice. (C-D) Coronal PV phase CT images demonstrate increased lesion size (yellow arrow), pancreatic and common bile ductal dilation (yellow diamond) with biliary drain in situ, and vascular involvement (white arrows). (E-F) Endoscopic retrograde cholangiopancreatography (ERCP) was performed for brushings and stenting, at which time atypical cells raised concern for malignancy. Following chemotherapy and Whipple’s procedure, surgical pathology demonstrated IPMN with invasive carcinoma. MRI: magnetic resonance imaging; CT: Computerized tomography; IPMN: intraductal papillary mucinous neoplasms; FNA: Fine needle aspiration; EUS: endoscopic ultrasound.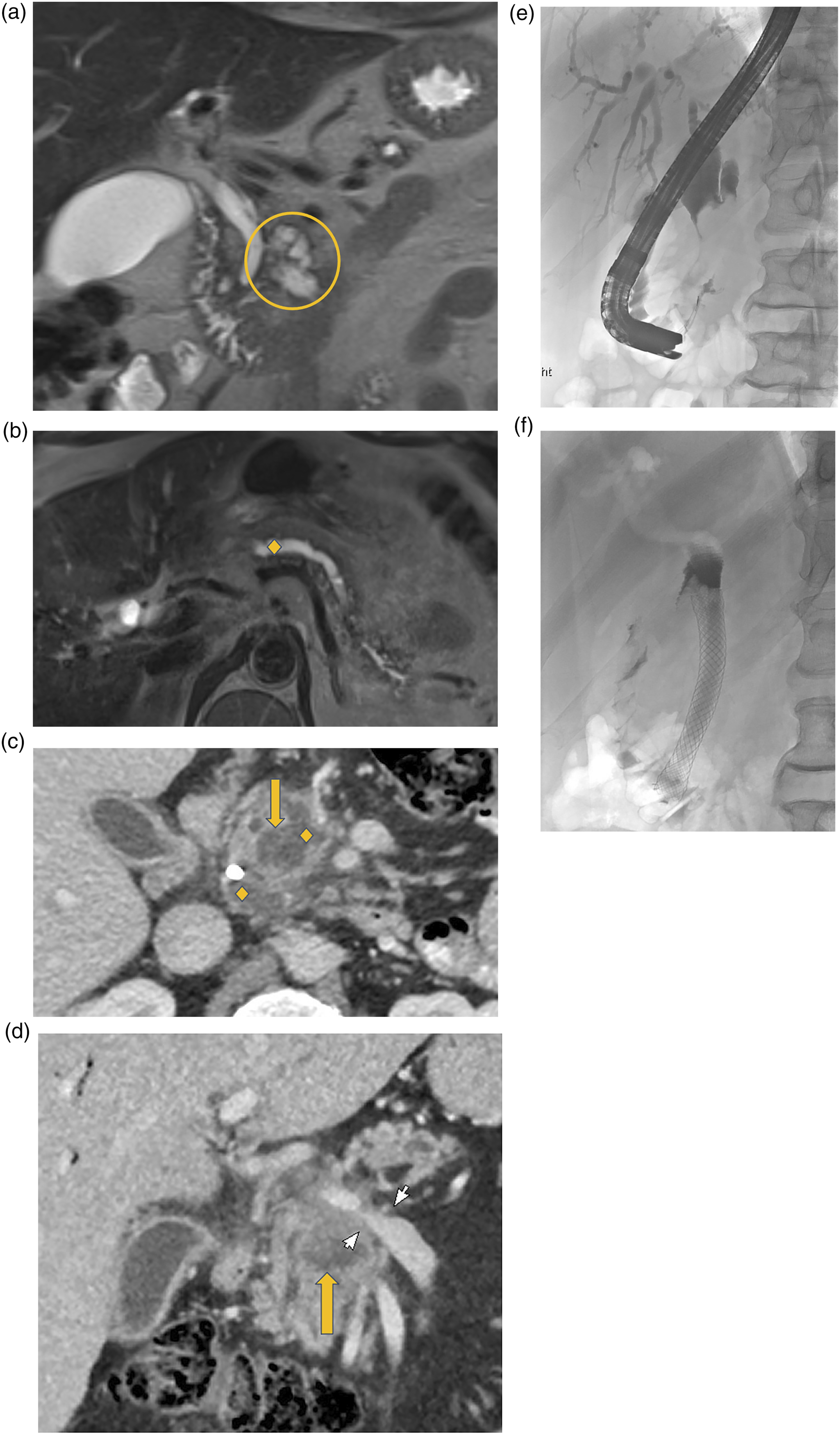
PCL Guidelines and Surveillance Strategies—Current and Future Considerations
There exists no international nor interdisciplinary consensus regarding the follow-up duration or criteria for cessation of follow-up for small PCLs. This is emphasized in all major published guidelines. There is considerable variability in the most recent major guidelines, most notably with regards to long-term surveillance.
6
Set endpoints are only in a minority of guidelines, for example, at 5 years and 10 years in the AGA and ACR guidelines, respectively, despite some evidence for long-term risk in developing pancreatic malignancy (Figure 5).
6
A commonality of importance amongst guidelines is the emphasis for ongoing follow-up in younger patients guided by clinical factors as well as the incorporation of surgical candidacy.
6
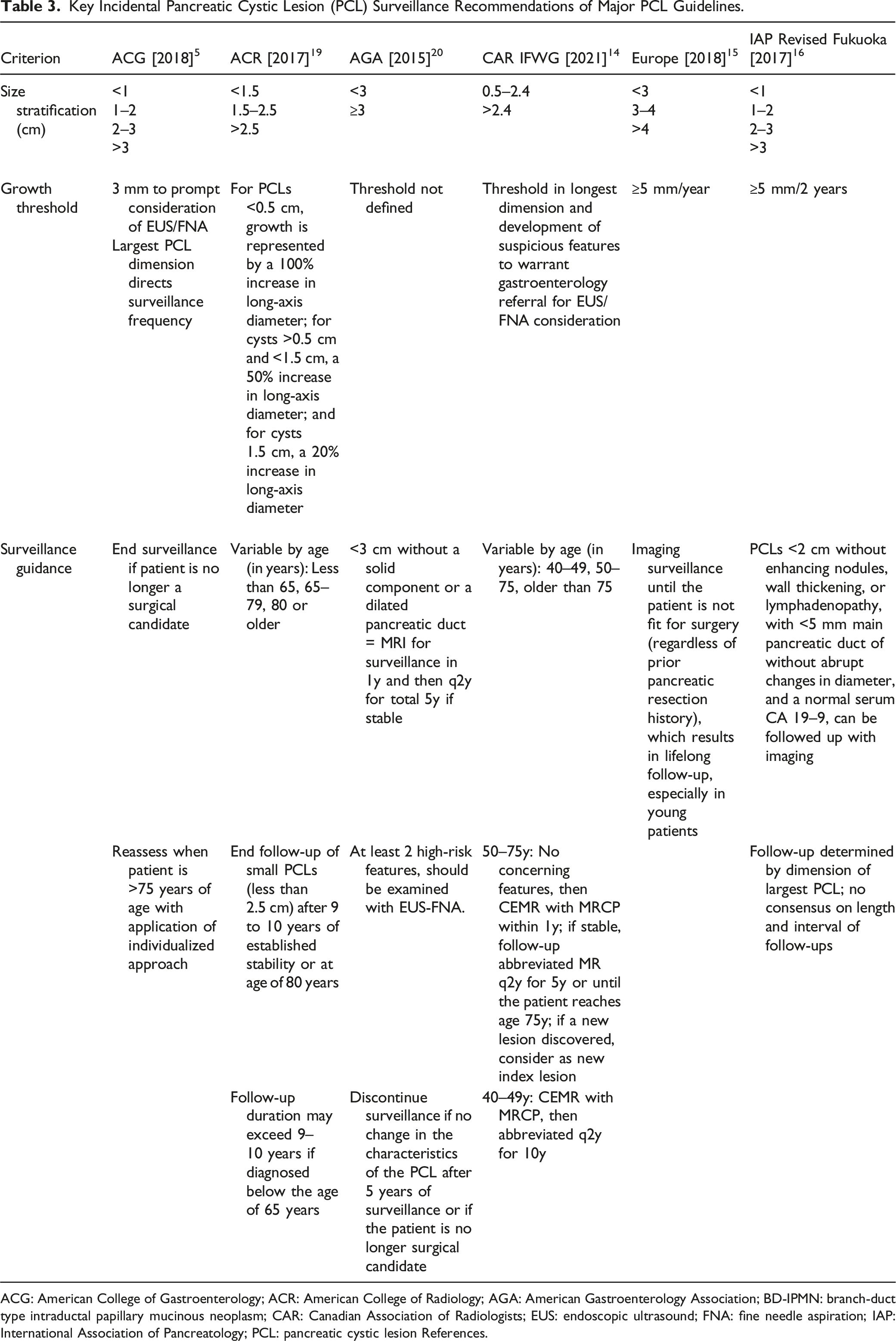
Table 3 summarizes the key features of surveillance proposed by each major guideline. Sixty-three-year-old female with history of idiopathic pancreatitis. Incidentally detected 8 mm PCL at the head (yellow circle) without worrisome features or high-risk stigmata on MRI 2013, considered a BD-IPMN vs pancreatitis-related pseudocyst (A, MRCP radial). Stable on MRI 2016 (B, MRCP radial). Lesion enlarged up to 2.0 cm by 2020 with worsened MPD dilatation up to 13 mm at the uncinate process (yellow diamonds), most concerning for main duct IPMN (C-D, Coronal PV phase CT at uncinate process and body). Surgical pathology confirmed IPMN with high grade dysplasia. PCL: Pancreatic cystic lesions; CT: Computerized tomography; IPMN: intraductal papillary mucinous neoplasms; MPD: main pancreatic duct. Key Incidental Pancreatic Cystic Lesion (PCL) Surveillance Recommendations of Major PCL Guidelines. ACG: American College of Gastroenterology; ACR: American College of Radiology; AGA: American Gastroenterology Association; BD-IPMN: branch-duct type intraductal papillary mucinous neoplasm; CAR: Canadian Association of Radiologists; EUS: endoscopic ultrasound; FNA: fine needle aspiration; IAP: International Association of Pancreatology; PCL: pancreatic cystic lesion References.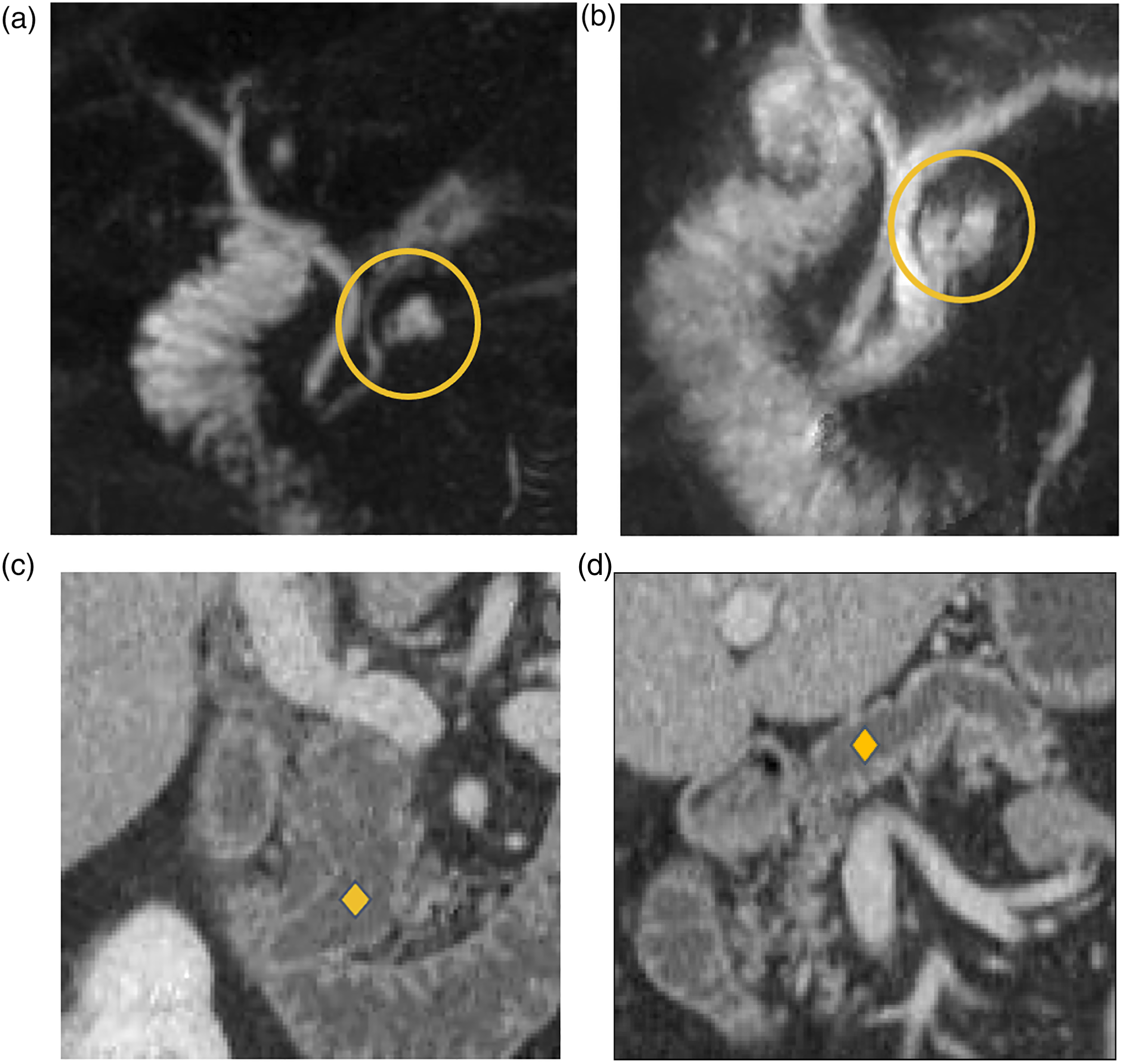
Adaptation of the variability in guidelines on an institutional or nation-wide basis would help locally navigate the lack of consensus. The Canadian Association of Radiology Incidental Findings Working Group recommendations for the management of incidental pancreatic findings in adults provides the most recent applicable guidelines stemming from collaboration between academia and community radiologists, accounting for Canadian practice patterns, and factoring for cost-effectiveness in the approach while respecting the goals of highest patient care. 14 Further exploration needs to be done for standardization of age vs surgical candidacy-based endpoints for surveillance, which can impact allocation of limited healthcare resources. Patient perspectives should be incorporated into local and national decision-making around surveillance guidelines and revisions. For example, responses from 109 international survey participants before and during surveillance for PCLs demonstrated low psychological burden in association with surveillance. 44 Incorporation of age, surgical candidacy, and lesion size in aggregate may help patients and caregivers make joint decisions regarding length of imaging surveillance.
Amidst the lack of consensus and underlying potential for confusion with guideline variability, there is also a question of whether PCL surveillance clinically impacts patients in a meaningful manner. Application of a Monte Carlo method evaluating outcomes for 10 000 patients comparing surveillance by two different guidelines (2017 International Consensus Guidelines and 2015 AGA Guidelines) demonstrated that the primary outcome of mortality related to PCLs (125 deaths) were vastly outnumbered by unrelated causes (1422 deaths). 45 Meanwhile, application of the AGA guidelines resulted in more missed malignancies while the 2017 Consensus guidelines resulted higher cumulative costs as well as more surgeries and imaging exams. 45 A current randomized clinical trial underway (ECOG-ACRIN Cancer Research Group 2185) aims to compare the clinical impact of PCL surveillance programs, namely the Fukuoka (high intensity surveillance) and AGA (low intensity surveillance) guidelines, amongst 4606 asymptomatic patients with newly identified PCLs ≥1 cm in diameter followed prospectively for 5 years. 46 The results of this trial may be a key component of future locally or nationally adapted guidelines.
Pancreatic cystic lesion surveillance guidelines would benefit from an improved understanding of the added risk for PDAC from a PCL relative to other independent risk factors for PDAC. It is possible that some patients may benefit from imaging follow-up of PCLs whereas others with the same imaging characteristics may not benefit based on their overall risk profile. More research is needed to improve patient risk-stratification and optimize surveillance strategies.
Conclusion
The last two decades have marked large strides on consensus around the diagnosis, management, and surveillance of PCLs. Major international consensus guidelines, however, remain variable, particularly with regards to length of surveillance follow-up and when to stop follow-up of incidentally detected PCLs. Pancreatic cystic lesion association with malignancy needs further study to clarify the true degree of association and alternate-site predisposition to pancreatic adenocarcinoma. Ongoing research is warranted targeting surveillance efforts primarily at patients with greatest risk, thereby minimizing excess follow-up of lesions that are highly likely benign.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
