Abstract
Historically thoracic MRI has been limited by the lower proton density of lung parenchyma, cardiac and respiratory motion artifacts and long acquisition times. Recent technological advancements in MR hardware systems and improvement in MR pulse sequences have helped overcome these limitations and expand clinical opportunities for non-vascular thoracic MRI. Non-vascular thoracic MRI has been established as a problem-solving imaging modality for characterization of thymic, mediastinal, pleural chest wall and superior sulcus tumors and for detection of endometriosis. It is increasingly recognized as a powerful imaging tool for detection and characterization of lung nodules and for assessment of lung cancer staging. The lack of ionizing radiation makes thoracic MRI an invaluable imaging modality for young patients, pregnancy and for frequent serial follow-up imaging. Lack of familiarity and exposure to non-vascular thoracic MRI and lack of consistency in existing MRI protocols have called for clinical practice guidance. The purpose of this guide, which was developed by the Canadian Society of Thoracic Radiology and endorsed by the Canadian Association of Radiologists, is to familiarize radiologists, other interested clinicians and MR technologists with common and less common clinical indications for non-vascular thoracic MRI, discuss the fundamental imaging findings and focus on basic and more advanced MRI sequences tailored to specific clinical questions.
Background and Context
Non-vascular thoracic magnetic resonance imaging (MRI) has increasingly been used over the last decade. However, its uptake in radiology departments has occurred at a slower pace compared to other MR clinical applications. 1 The unique challenges of cardiac and respiratory motion artifacts, the lower proton density of the lung parenchyma and the relatively long acquisition times are limiting factors that have impeded rapid growth of non-vascular thoracic MR applications in many radiology departments in Canada. However, recent technical advancements in MR hardware systems, improvement in MR pulse sequences (including parallel imaging techniques), diffusion weighted imaging, ultrashort echo sequences as well as postprocessing software have helped overcome many of these limitations. The excellent soft tissue contrast and lack of radiation make thoracic MR a unique problem-solving modality for characterization of thoracic diseases especially in young or pregnant patients, and patients who require long-term and frequent follow up. 2
Lack of familiarity with non-vascular thoracic MRI, paucity of dedicated literature addressing questions about clinical protocols and inhomogeneity in existing MRI protocols have prompted the creation of this guidance document. The purpose of this article is to serve as a practical reference for standardized and updated MRI protocols tailored to specific common and less common clinical indications. Where relevant, we provide the basic pathophysiologic mechanisms that justify specific sequences and facilitate thoracic MRI interpretation. We hope to clarify and demystify the use of non-vascular thoracic MRI thereby expanding its great potential in everyday clinical practice.
This practical guide, which was developed by the Canadian Society of Thoracic Radiology and endorsed by the Canadian Association of Radiologists, does not review imaging of the thorax that requires dedicated fellowship training such as breast and cardiac MRI. We discuss brachial plexus imaging only where relevant to superior sulcus tumor involvement, as it is part of the neuroradiology domain in most Canadian radiology departments. As per the title of this guidance document, we will not cover vascular applications including thoracic outlet syndrome.
Due to rapid continued technological advancements and improvement in MRI pulse sequences and hardware, this guidance document is a work in progress that will require future modifications to adapt to the changing technological environment and emerging new data arising from research studies.
Clinical Indications
Thymic Imaging
One of the most common indications for non-vascular thoracic MRI is characterization of masses in the anterior superior mediastinum that may be thymic in origin. 1
Thymic hyperplasia typically contains microscopic fat whereas neoplasms such as thymoma, thymic carcinoma and lymphoma do not. 3,4 In and out-of-phase T1 weighted gradient recalled echo (GRE) imaging makes use of this feature to identify the presence of microscopic fat. At certain echo times, intravoxel dephasing occurs due to the precession frequency differences between protons in fat and water. When there is microscopic fat, there is a loss of signal that occurs on the out-of-phase sequences (Figure 1). It is recommended that dual echo gradient sequences be used because they are acquired during the same breath hold, thereby avoiding misregistration from variable breath holds. A chemical shift ratio (CSR) is calculated by measuring a region of interest (ROI) signal intensity (SI) in the target lesion on the out-of-phase images and dividing this by the SI of an ROI placed in the paraspinal muscle. This ratio is then divided by the corresponding ratio of SI of target lesion to paraspinal muscle on the in-phase images. It is important to draw ROIs of the same cross-sectional area when calculating the CSR. Imaging at 1.5 T is preferred over 3.0 T for thymic protocols due to validation of the CSR at 1.5 T. 3,4
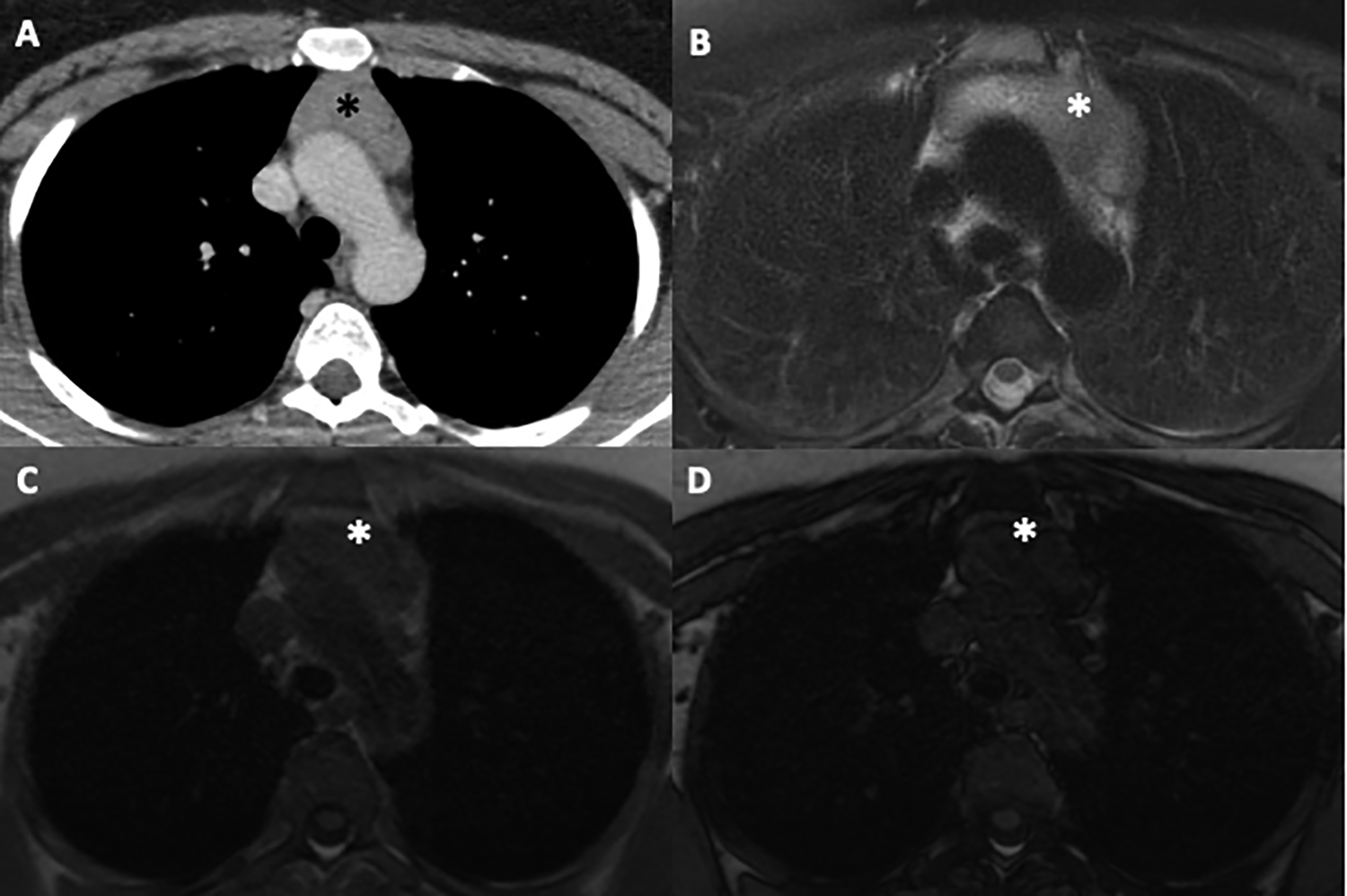
29-year-old woman with an anterior mediastinal mass detected on CT for staging of breast cancer. A, Contrast enhanced CT chest shows an anterior mediastinal mass (*) of soft tissue density (50-60 HU). B, Axial T2 fat saturated image demonstrates mild homogeneous T2 hyperintensity within the mass (*). C, Axial T1 GRE in-phase image and (D) Axial T1 GRE out-of-phase imaging shows loss of signal within the mass (*) typical of thymic hyperplasia. The CSR was calculated at 0.6, consistent with thymic hyperplasia. The mass decreased in size on follow up CT.
Chemical Shift Ratio (CSR) = (tSIop/mSIop)/(tSIin/mSIin)
A CSR value ≤0.7 is consistent with benign entities such as thymic hyperplasia. In contrast, neoplasms and cysts do not demonstrate signal loss on the out-of-phase imaging and therefore have CSR values around 1.0 (Figure 2). When using dual echo in and out-of-phase imaging acquired during the same breath hold, there is decreased need for normalization of the target lesion SI to paraspinal muscle. Furthermore, with increasing age, there may be fatty atrophy of paraspinal muscles that can lead to a drop in signal on out-of-phase imaging and spurious calculated CSR values. A SI index (SII) has been proposed as another parameter to evaluate thymic lesions with equal or better accuracy and specificity than the CSR. 5
SII = [(tSIin - tSIop)/ (tSIin)] × 100%.
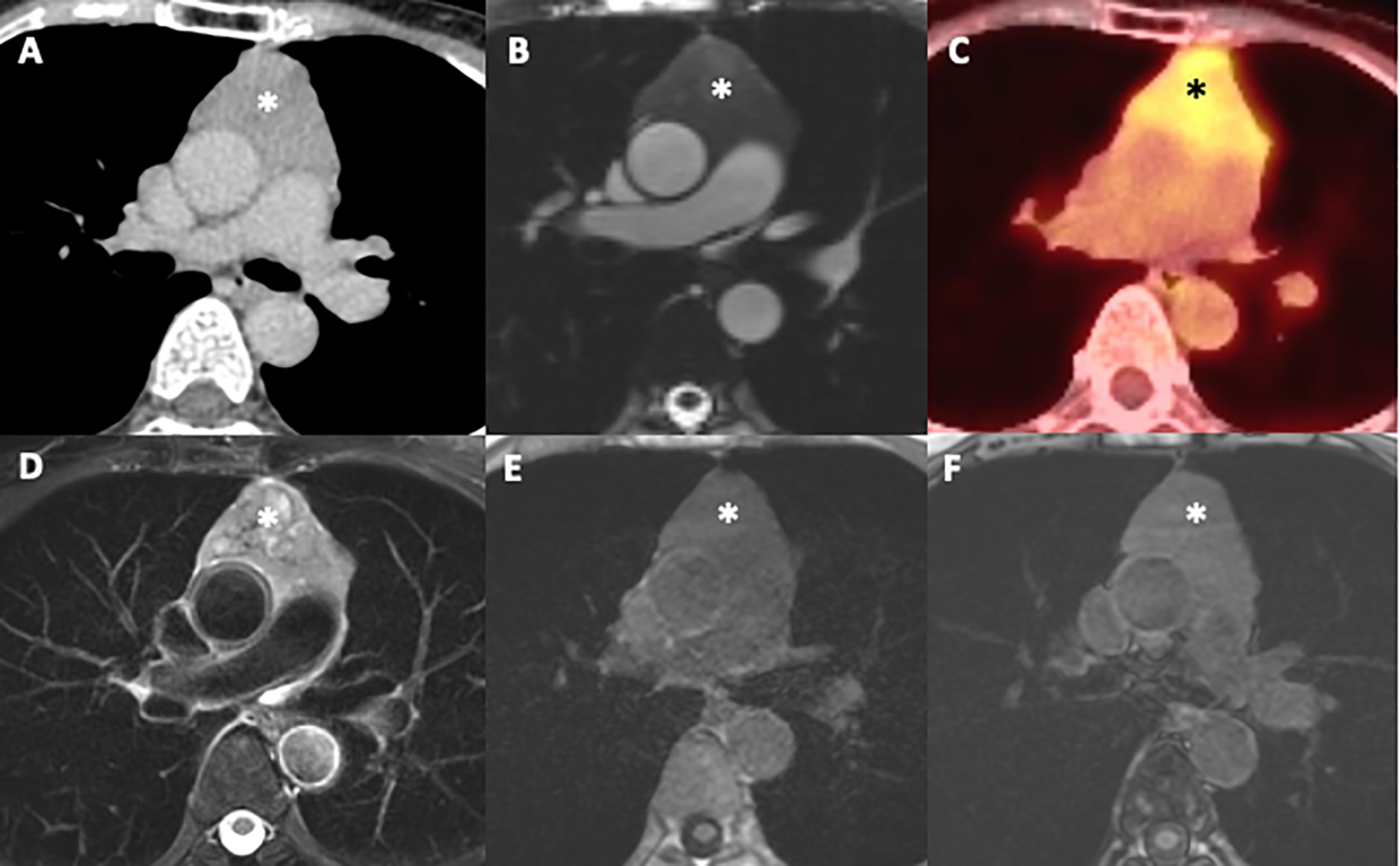
64-year-old woman with an anterior mediastinal mass (*) seen on axial contrast enhanced CT (A). B, Axial cine SSFP image shows isointensity of the mass (*) to skeletal muscle, and close proximity to the ascending aorta and main pulmonary artery but without vascular invasion. C, FDG PET-CT demonstrates standardized uptake value (SUV) of 4.1 within the mass (*) concerning for malignancy. D, Axial T2 fat saturated images demonstrate heterogeneous T2 signal within the mass (*). (E and F) In and out-of-phase T1 gradient dual echo images reveal no drop in signal on out-of-phase imaging within the mass (*). CSR was 1.1 consistent with a thymic neoplasm. Surgical resection confirmed thymoma.
For SII, a SI decrease of >8.92% has been used to differentiate thymic hyperplasia from neoplasms that do not demonstrate a drop in SI. The advantage of SII is that it is not influenced by age. In contrast, the CSR of thymic lesions may be age dependent, with overlapping CSR values between thymic hyperplasia and neoplasms especially in early adulthood when the normal thymus may lack fatty infiltration. 6,7 The normal thymus has a triangular, bipyramidal or quadrilateral shape, achieves maximum volume during puberty and undergoes a decrease in size over time due to progressive fatty involution (first microscopic then macroscopic fat is seen). The normal thymus has straight or concave peripheral margins in adults. Convex margins suggest thymic hyperplasia or neoplasm in adults but may be a finding associated with normal thymus in children. 7
Generally, spin echo (SE) T1 and T2 weighted sequences are used for basic tissue characterization but are often insufficient in isolation to make a definitive diagnosis. T1-hyperintensity in cystic lesions suggests the presence of blood products, proteinaceous fluid or fat within the lesion. Homogeneous T1-hypointensity often indicates serous fluid within a suspected cyst. T2-hypointensity within a lesion may indicate the presence of blood products, fibrous tissue, or high cellular density such as in lymphoma. Intravenous gadolinium contrast may be required to exclude vascular or mediastinal invasion or to help differentiate solid enhancing masses from cystic lesions in the anterior mediastinum. Diffusion weighted imaging (DWI) has been used to differentiate benign from malignant thymic lesions. Malignant lesions demonstrate more restricted diffusion as compared to benign lesions (lower mean apparent diffusion coefficient, ADC values). Higher grade neoplasms typically have further decrease in ADC values offering a means to differentiate thymoma from more aggressive thymic carcinomas. 8 However, optimal differentiating thresholds remain a topic of ongoing research. Routine incorporation of DWI into thymic MRI protocols is not widespread clinical practice across Canada. A suggested thymic imaging protocol is presented in the supplement section.
Mediastinal Masses
Due to its high soft tissue contrast resolution, 9 MRI is valuable in evaluating cystic mediastinal masses, such as thymic, foregut duplication or pericardial cysts with soft tissue attenuation; these may be misinterpreted as solid on CT, due to the presence of hemorrhage, proteinaceous components and occasionally calcium oxalate, or milk of calcium. 10 Unlike serous cysts, which demonstrate low T1 signal, proteinaceous or hemorrhagic cysts will show homogeneous high or intermediate T1 SI to muscle. 11 Simple cysts demonstrate homogeneous, T2 high SI-isointense to cerebral spinal fluid 9 with no central enhancement.
Axial pre- and post-gadolinium ultrafast-GRE fat saturated T1-weighted images should be performed with identical parameters, to enable subtraction of the pre- from the post-contrast image. Subtraction imaging facilitates evaluation of solid masses, especially those with high T1 SI on pre-contrast imaging (which may show only subtle enhancement). 12
MRI differentiates simple (Figure 3) from complex cystic lesions by detecting enhancing nodular wall thickening and septations, that may not be appreciated on CT. 11 This is significant, as complex cystic lesions require biopsy or surgical resection for definitive diagnosis (Figure 4).
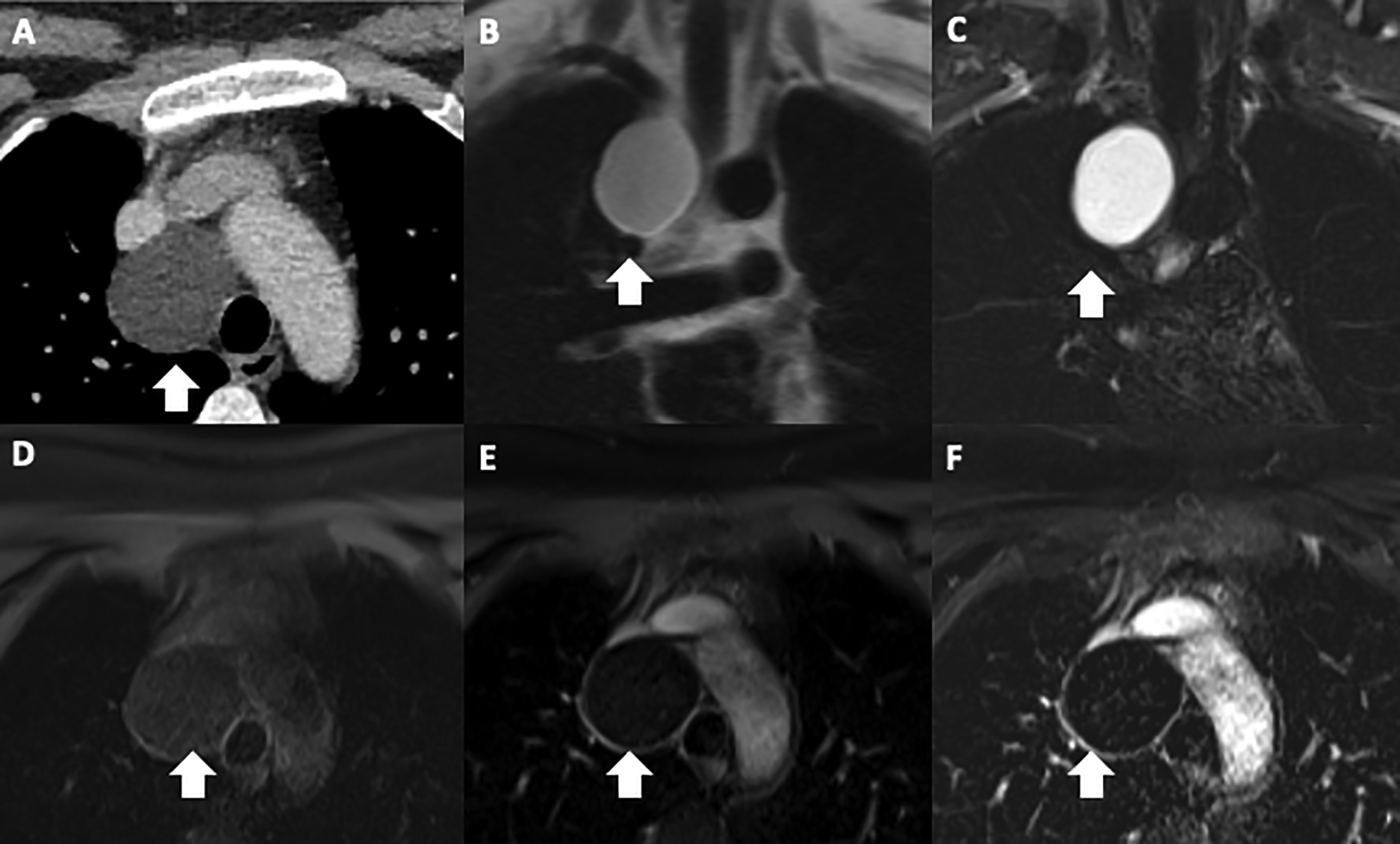
25-year-old woman with chronic cough. Contrast enhanced CT chest (A) shows a right paratracheal mass (arrow) of fluid density (5 HU). Coronal BH ultrafast SE T2-weighted image (B) and coronal fat-suppressed BH STIR image (C) demonstrate homogenous T2 hyperintensity. Axial T1-weighted 3D GRE fat saturated pre-gadolinium (D), post-gadolinium (E) and subtraction images (F) show the mass is homogeneously T1 hypointense (D), with no evidence of central enhancement. There is mild enhancement (arrow) of the thin wall of the cyst (E, F). Appearances are typical of a bronchogenic cyst.
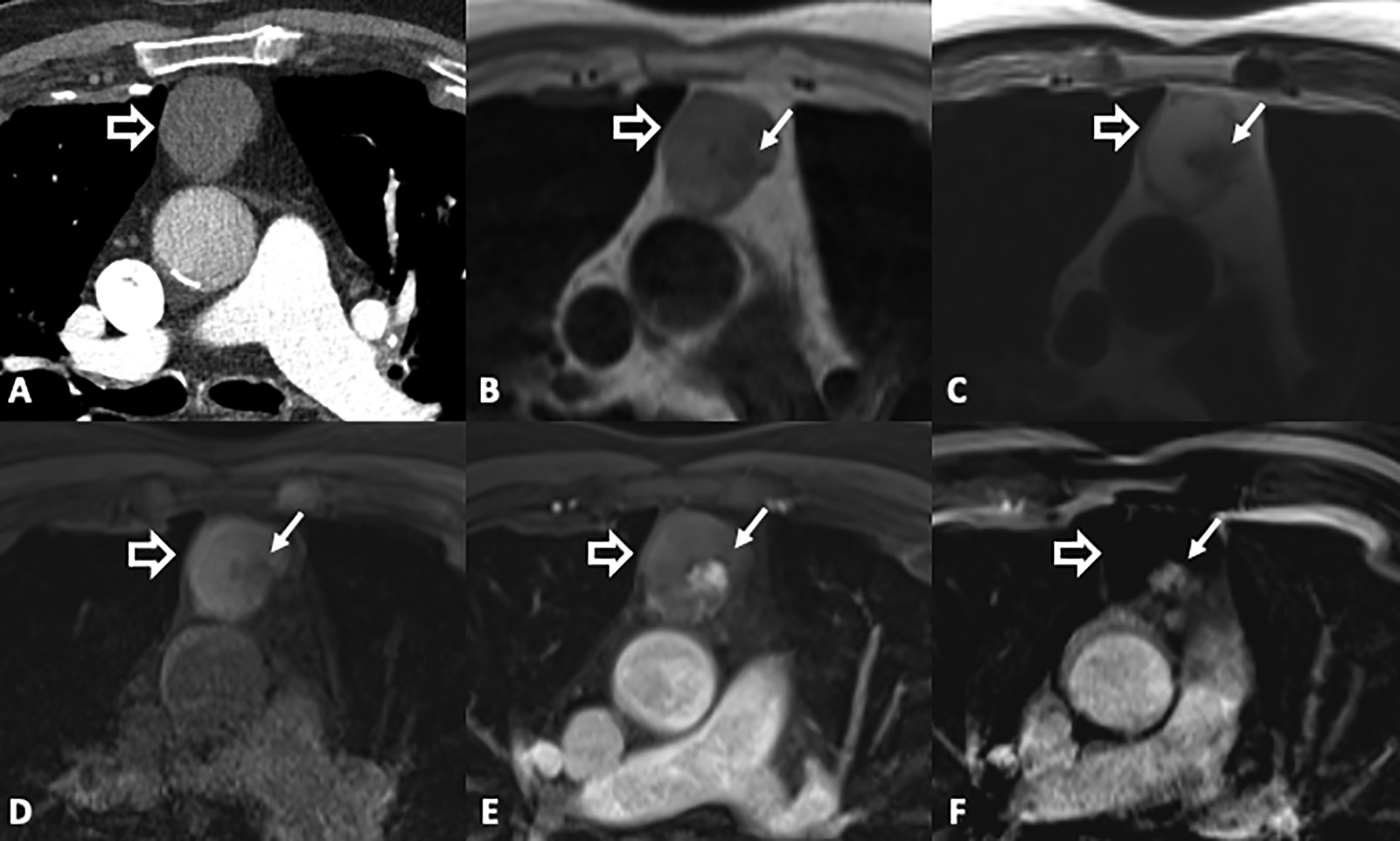
79-year-old man with chest pain. CT pulmonary angiogram (A) shows a homogeneous anterior mediastinal mass (open arrow) of soft tissue density (30 HU). Axial T1-weighted 3D GRE image (B) shows the mass has intermediate T1 signal intensity with a small low T1 nodular focus (solid arrow). Axial BH ultrafast SE T2-weighted image (C) shows T2 hyperintensity except for the small low T2 nodular focus. Axial T1-weighted 3D GRE fat saturated pre-gadolinium (D), post-gadolinium (E) and subtraction images (F) show enhancement of the nodular component (E, F). Findings are in keeping with a complex cystic mass. Surgical resection revealed a cystic thymoma.
MRI plays a vital role to evaluate lymphatic malformations, which contain fluid, but may appear solid on CT. Lymphatic malformations demonstrate variable T1 SI, due to different amounts of proteinaceous fluid present 10,13 and high T2 SI, often showing multiple tubular channels or septa. Some may demonstrate no enhancement or slow enhancement due to third spacing of gadolinium, especially in cases of lymphatic duct obstruction, or diffuse non-mass-like enhancement if they have a venous component such as in venolymphatic lesions. However, nodular enhancement should be viewed with suspicion and either closely followed or biopsied. 12
The high soft tissue contrast of MRI helps in guiding the site for biopsy in areas with enhancing nodular components or restricted diffusion on DWI. On DWI, tissues with high cellularity (i.e. tumors) demonstrate restricted water diffusion and produce a higher SI on higher b-value imaging, and low ADC values. 14
MRI is valuable to identify microscopic and macroscopic fat. Chemical shift T1 GRE imaging utilizing in- and out-of-phase imaging enables identification of microscopic (intravoxel) fat by demonstrating significant signal decrease on the out-of-phase compared to in-phase sequences. This is useful fto detect not only thymic hyperplasia but also dermoid cysts containing intravoxel fat. 15 Additionally, macroscopic fat in mature teratomas, thymolipomas, lipomas and liposarcomas (Figure 5) can be identified on fat saturation imaging. Macroscopic fat will show high T1 and T2 SI and demonstrate signal decrease on fat saturated sequences. 16
42-year-old woman with an incidental mediastinal mass discovered on chest radiograph (not shown) performed to screen for tuberculosis. Contrast enhanced CT chest (A) demonstrates a large anterior mediastinal mass occupying a large portion of the left hemithorax (open arrows). The mass contains fat (stars) and soft tissue density components including large round soft tissue mass (solid arrow). Axial T1-weighted 3D GRE image without (B) and with fat saturation (C) pre-gadolinium, and axial BH ultrafast SE T2-weighted (D) images demonstrate areas within the mass of high T1 signal intensity (B, stars) and high T2 signal intensity (D, stars) and SI drop on T1-weighted fat saturation image (C, stars), consistent with macroscopic fat. Axial T1-weighted 3D GRE fat post-gadolinium (E) and subtraction (F) images show partial enhancement of the rounded mass (curved arrow). Surgical resection confirmed liposarcoma. The rounded mass component showing heterogeneous signal intensity on all pulse sequences corresponded to myxofibrosarcomatous pattern on pathology. The enhancing solid component which demonstrated low T2 signal intensity corresponds to the fibrous component, while the subtle T1 hypointense (B, triangle) and subtle T2 hyperintense (D, triangle) non-enhancing component (E, F triangle) correspond to the myxoid component.
Dynamic MRI utilizing cinematic balanced SSFP can assess adherence or invasion of a mediastinal mass to adjacent chest wall 17,18 or cardiovascular structures. The absence of a sliding motion between a lesion and an adjacent structure is highly suspicious for involvement or invasion.
Finally, MRI is indicated to differentiate the types of neurogenic tumors (schwannomas, neurofibromas and ganglioneuromas) 19 -21 (Figure 6) that may have similar CT features, and in the evaluation of intraspinal and neural extension of the tumor. 22
56-year-old woman with a mass discovered on chest radiograph. Sagittal T1-weighted (A), sagittal T2-weighted fast SE (B) and axial T2-weighted FSE (D) images demonstrate a large left lower paraspinal mass (solid arrow) at the T10 vertebral body, isointense to muscle on T1-weighted and low T2 signal intensity. The mass contains one large (star) and multiple small (open arrows) low T1 and high T2 signal intensity foci that do not enhance on the post-gadolinium fat saturated T1-weighted sagittal (C) and axial (E) images, in keeping with cystic components. The mass otherwise shows homogeneous enhancement on the post-gadolinium fat saturated T1-weighted GRE images of the solid components. The lesion does not involve the neural foramen. Surgical resection confirmed a cystic schwannoma.
Image quality is optimized by choosing breath-hold imaging over respiratory gating to eliminate respiratory motion artifact, and is best achieved with sequences which last < 20 seconds. 23 Multiple stacks (ie. multiple breath holds) should be used to cover the necessary volume when imaging cannot be obtained in 20 seconds.
Chest Wall and Pleural Masses
The diagnostic performance of imaging the chest wall and pleura with CT and MRI is similar. 12 However, the main advantage of MRI is in its superior contrast resolution for delineation of anatomy, evaluation of local invasion and the real time imaging capabilities.
Imaging the chest wall is unique due to the rounded asymmetric contour and obliquely orientated structures (i.e ribs) that render evaluation using conventional imaging planes (axial, coronal, sagittal) challenging. In these situations, oblique or parallel planes to anatomical structures (ribs, brachial plexus, etc.) are required. Malignant chest wall invasion is evident on CT and MRI with the presence of bone destruction. Superior contrast resolution of MR 12,24 is used to advantage to help determine invasion and provide additional tumor staging information that helps to guide patient management. 25 Although a maintained fat-soft tissue plane is reassuring, obliteration of the fat tissue plane should raise suspicion for tumor invasion (Figure 7). 12 Due to magnetic field inhomogeneities, for large masses, GRE fat suppression techniques may be suboptimal and adequate fat suppression is often better achieved using STIR. 26
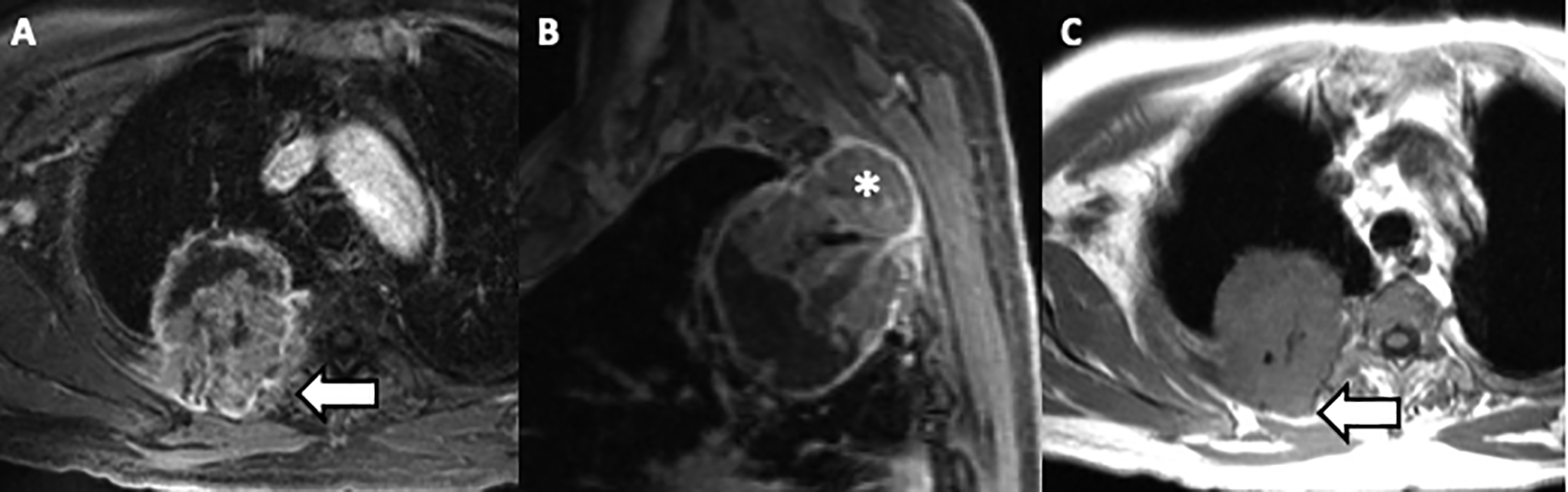
71-year-old man with a right upper lobe non-small cell lung cancer demonstrating posterior chest wall invasion. Axial (A), sagittal (B) T1 VIBE post contrast and axial T1 SE (C) show a right upper lobe lung mass that is T1 isointense compared to muscle and has solid areas of enhancement and non-enhancing cystic spaces. The lung mass invades through the subpleural fat and chest wall (arrow) and into the 3d-4th intercostal muscles and ribs (*). However, the fat plane with the right rhomboid muscle is maintained (arrow).
While CT is often the initial cross-sectional modality for chest wall and pleural based masses, determining the exact location of origin on CT can be challenging. For these masses protruding into the chest cavity, real time MRI cine imaging helps determine if there is chest wall or lung parenchymal invasion. 12,25 A cine SSFP sequence in normal individuals shows the pleural surface and lung parenchyma gliding smoothly during the respiratory cycle and with diaphragmatic motion. Tumors with chest wall or lung parenchyma invasion are adherent and move in unison. This is most pronounced in the mid and lower lobes where lung motion is greatest. In addition, real time cine MRI can be useful to determine if a mass arises from the lungs or from the pleura. Real-time cine MRI can also be used to evaluate diaphragmatic paralysis by demonstrating paradoxical diaphragm motion during inspiration. 12 Access to real-time MRI may be a limiting factor but it has an advantage over the fluoroscopy sniff test by avoiding radiation.
Pancoast Tumors, Recurrent Cancers, and Brachial Plexus Involvement
Pancoast tumors are non-small cell carcinomas that originate in the lung apex and cause the Pancoast syndrome which is characterized by ipsilateral shoulder pain and Horner’s syndrome. Superior sulcus tumors encompass all non-small cell carcinomas that arise from the lung apex and invade the chest wall or the soft tissues of the thoracic inlet. The role of MRI in Pancoast tumors is to assess the spine and extrathoracic soft tissues, as well as subclavian vessels and brachial plexus involvement to help determine resectability.
Superior sulcus tumors are defined as T3 because they invade extrathoracic soft tissues. Invasion of the trunk or root of C7 or above, subclavian vessels, vertebral bodies, spinal cord, trachea, or esophagus is considered T4 disease. 27,28 Tumors with hilar (N1) and ipsilateral supraclavicular (N3) nodal metastases can be resected. However, mediastinal (N2) and contralateral supraclavicular (N3) nodal metastases, brachial plexus invasion at a level above the T1 nerve, more than 50% vertebral body invasion, invasion of the esophagus or trachea and distant metastasis are contraindications to resection of the dominant lung mass. 29 Familiarity with the anatomy of the superior sulcus and brachial plexus is required to accurately describe the tumor extent that will guide appropriate management. 30 -32
The standard chest MRI with addition of axial oblique and sagittal oblique T1-weighted fast spin echo sequences typically without fat saturation provides adequate information. Axial T1-weighted FSE sequences from the upper cervical to upper thoracic spine is best for localization of the primary tumor and for visualization of the spinal canal and neural foramina. Sagittal T1-weighted SE sequence provides the best anatomic information to depict the scalene muscles, the brachial plexus, the subclavian vessels, the apical pleura, and posterior arcs of the ribs. Images should include from contralateral paraspinal vertebral bodies to the retropectoral region at the level of coracoid process (Figure 8A and B). Coronal T2-weighted sequences are useful to detect surrounding inflammation but do not often contribute additional important information regarding resectability. Post-contrast 3D GRE images facilitate assessment of vascular or neural foraminal extension (Figure 8C-F).
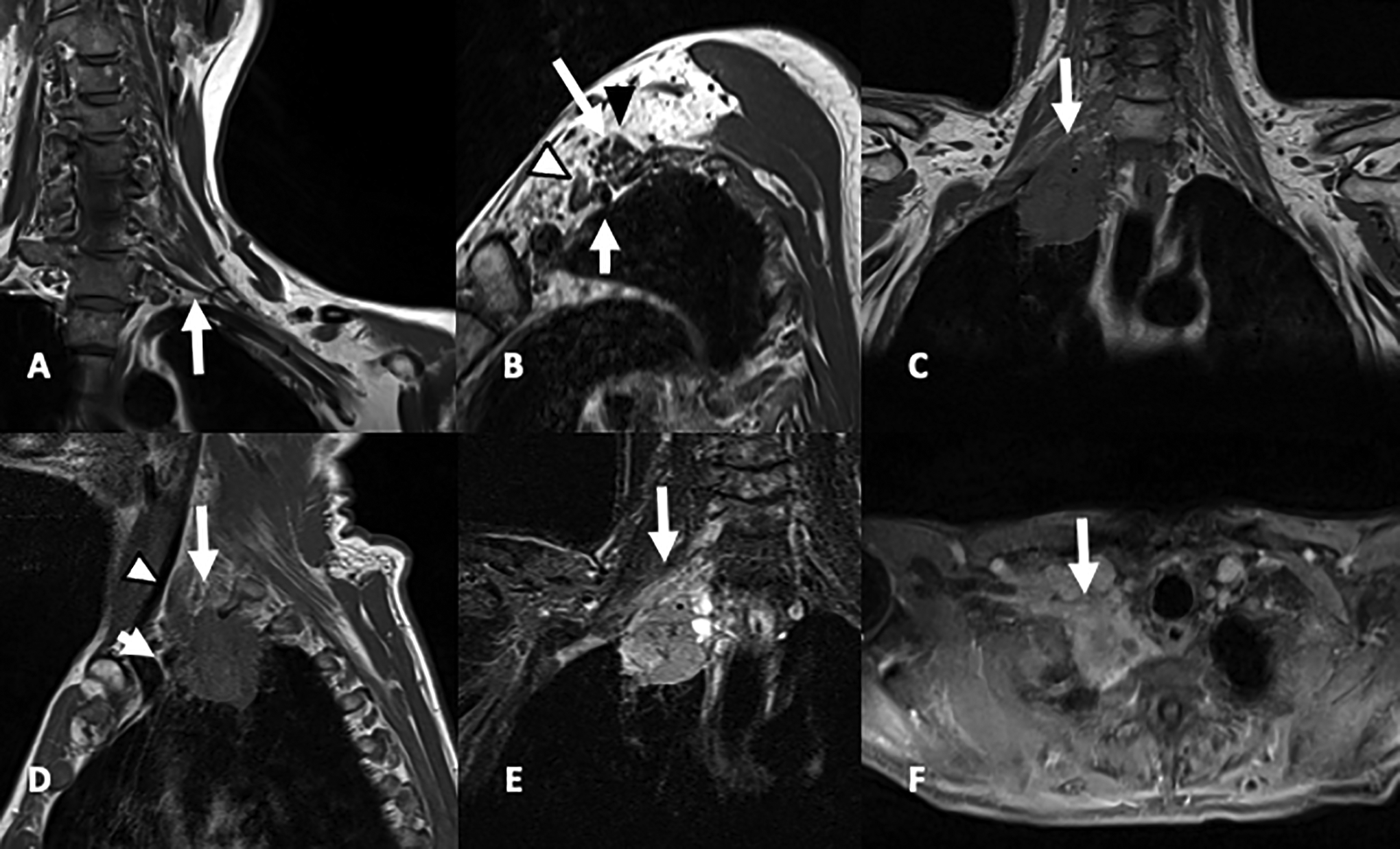
24 year-old woman with normal anatomy: (A) coronal oblique T1-w FSE in plane of brachial plexus, arrow is pointing to C8 nerve root, (B) sagittal oblique T1-W FSE: anterior scalene muscle (white arrowhead), middle scalene muscle (black arrowhead), brachial plexus in interscalene triangle (long arrow), subclavian artery (short arrow). 54-year-old man with superior sulcus tumor: (C) coronal T1-weighted FSE shows extrapulmonary extension of the lung adenocarcinoma into the superior sulcus, (D) Sagittal T1-weighted FSE shows extension in to lower interscalene triangle (long arrow), anterior scalene muscle (arrowhead), right subclavian artery (short arrow), (E) T2-weighted with fat saturation shows limited surrounding edema (arrow), (F) Axial T1-weighted post gadolinium shows encasement of the right subclavian artery (arrow).
In patients who have undergone neoadjuvant therapy, there is often blurring of fat planes and abnormal enhancement that can be seen in both residual and recurrent tumors as well as post treatment changes including fibrotic tissue. However, new nodular enhancement or new destructive bony lesions are findings suggestive of disease recurrence. PET-CT scanning can help differentiate, but is not always reliable to differentiate residual or recurrent tumor from post-treatment changes. DWI imaging has been used to help differentiate recurrent tumors from benign lesions with promising preliminary results 33 as well as to predict and monitor treatment response. 34,35
Less Common Indications
Pulmonary nodules and lung cancer staging
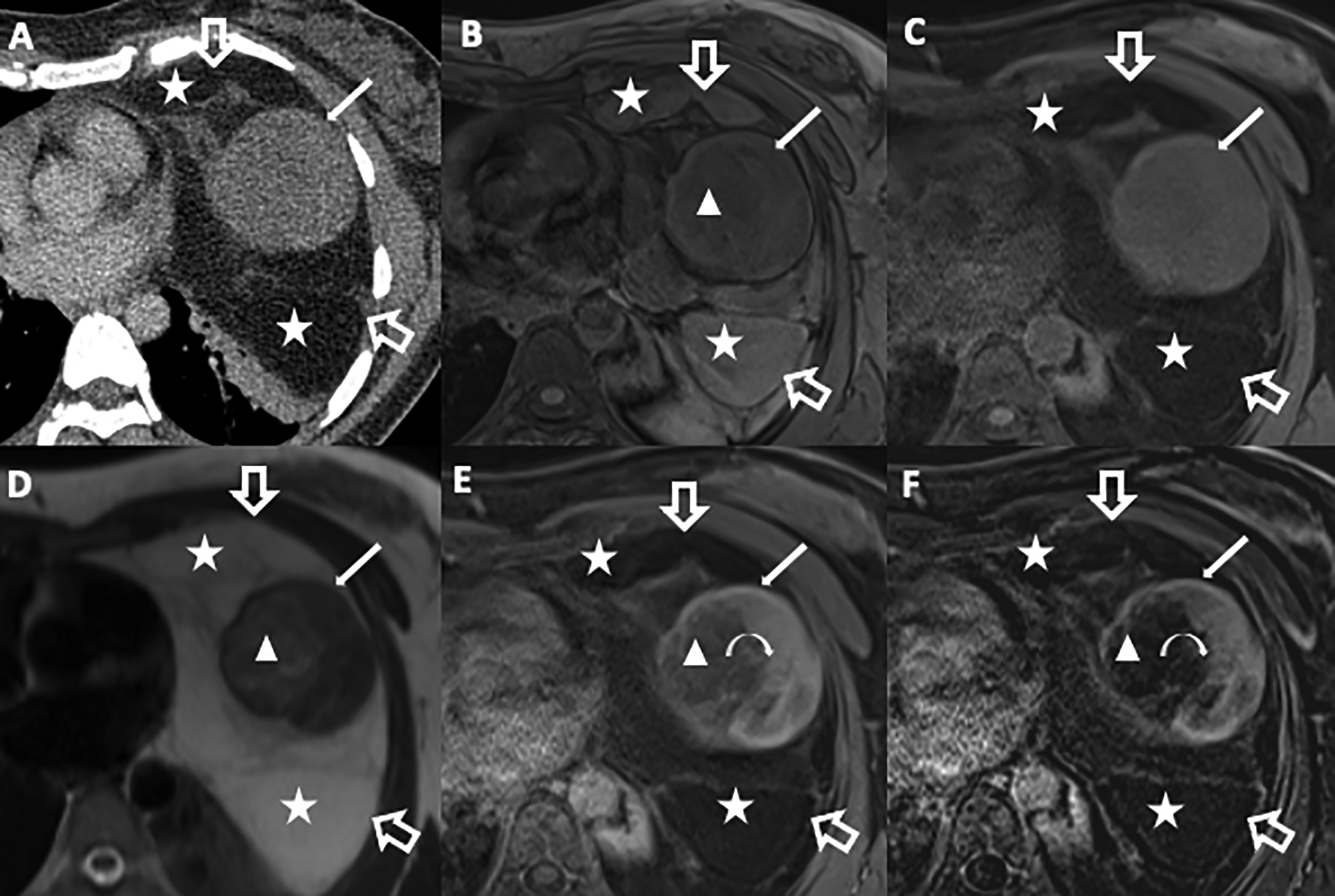
Pulmonary nodule detection and characterization is often performed on routine chest radiography and CT as well as in lung cancer screening programs using low dose CT. 36 The Fleischner Society suggests that nodule type and size are effective biomarkers for nodule management to reduce lung cancer specific mortality. 37 Moreover, lung cancer is the most common cancer worldwide and is the leading cause of cancer-related mortality. 38 Pulmonary imaging with MR was historically limited by low proton density, high signal decay, long acquisition time and respiratory and cardiac motion artifacts. Recent technical advancements achieved stronger gradients, multichannel coils, increased field homogeneity, parallel imaging and shorter echo times. Advancements in postprocessing software have made pulmonary MRI a promising radiation-free alternative to CT for pulmonary nodule detection/assessment and lung cancer staging. 2,39 Non-contrast sequences have been used for nodule detection with single shot T2-weighted turbo SE (HASTE) and STIR turbo SE with detection rates ranging from 46% to 100%. 40,41 A novel free-breathing 3-dimensional GRE ultrashort echo time (UTE) sequence (echo time <200 µsec) has become a “game changer” in the assessment of pulmonary disease and specifically in the detection of pulmonary nodules, given that shorter echo times reduce magnetic susceptibility artifacts in the soft tissue-air interfaces and provide a higher signal-to-noise ratio from the lung’s relative short T2*. 2,42,43 The use of UTE achieved a detection rate of more than 90% for ground glass, part-solid and solid nodules ranging from 4 to 29 mm37 and had comparable results to low dose CT in lung cancer screening with MRI (Figure 9). 44 Clinical and experimental studies have shown a threshold size of 3-4 mm for solid nodules and detection rates ranging from 60 to 90% for 5-8 mm lesions and near 100% for nodules over 8 mm (Figure 10). 41 Given that the probability of cancer in pulmonary nodules < 5 mm has been reported to be <1%, MR threshold would suffice for the detection and follow-up of clinically significant lesions. 45
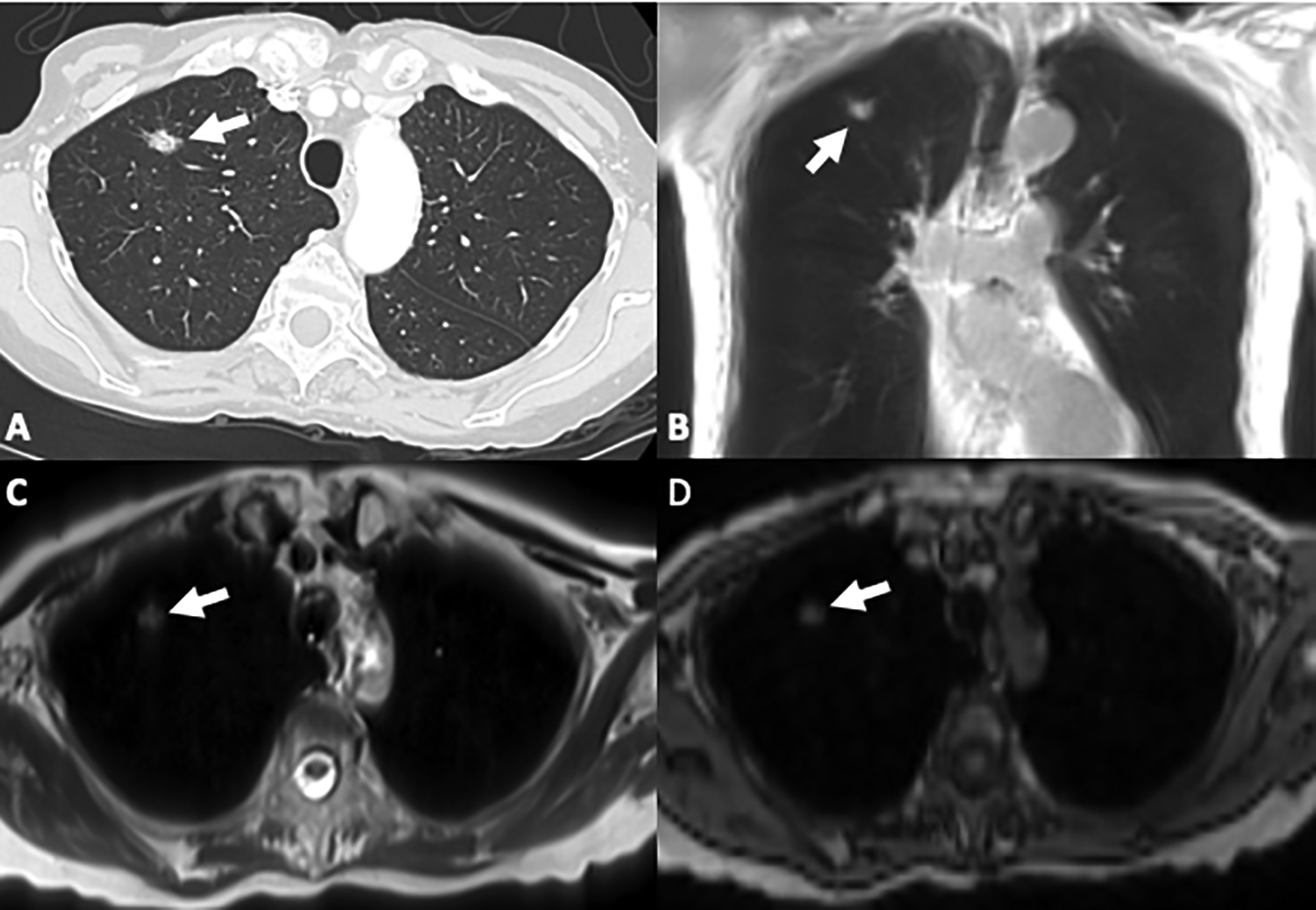
78-year-old male with biopsy proven lung adenocarcinoma. Chest CT shows (A) a right upper lobe spiculated nodule in keeping with the known primary malignancy (arrow). Coronal UTE (B) and axial HASTE (C) and VIBE (D) images clearly depict the lesion (arrows).
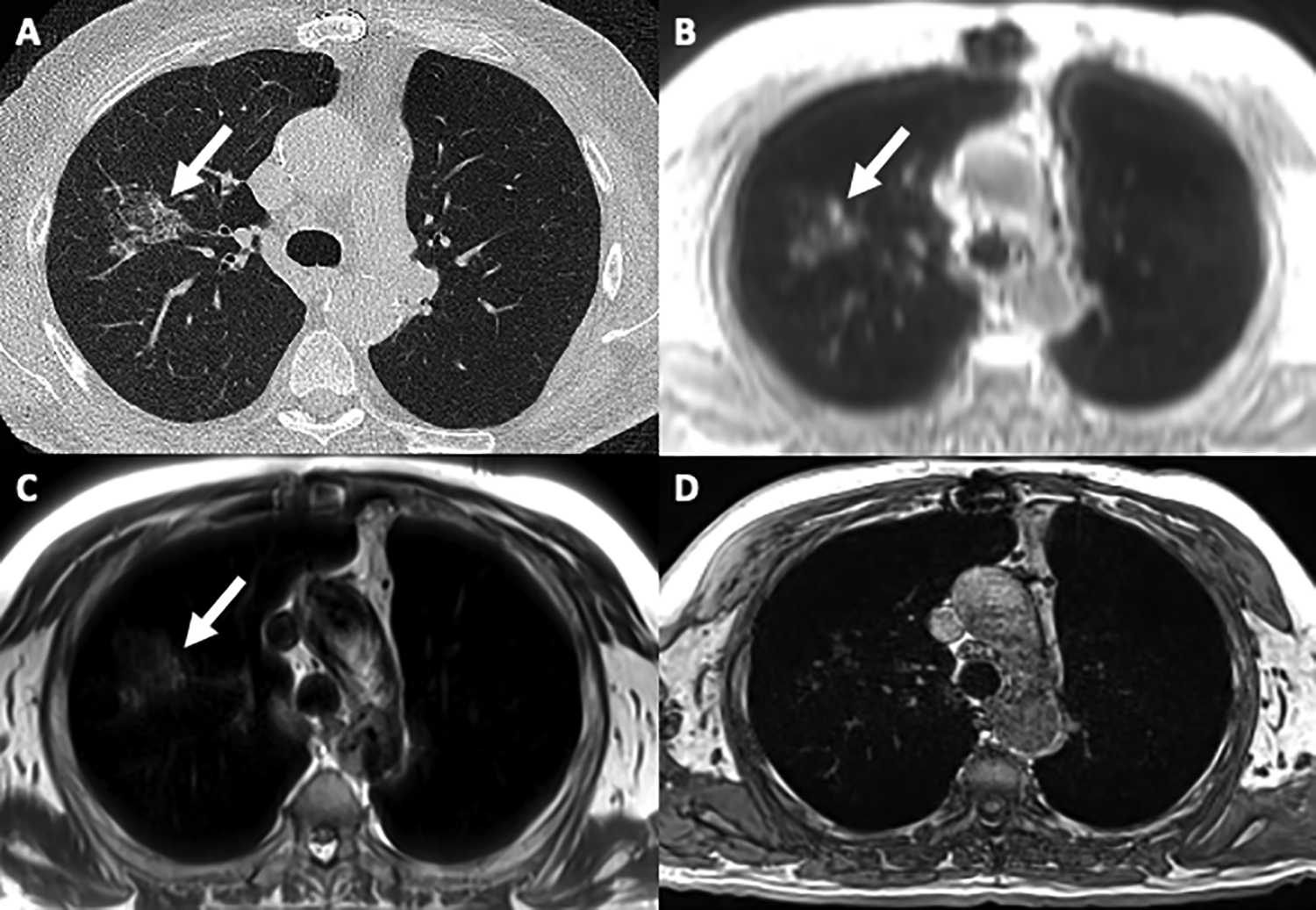
77-year-old man being followed for a slow-growing right upper lobe ground glass lesion. Axial chest CT (A) shows a centrally-located right upper lobe subsolid nodule (arrow). The lesion is clearly depicted in UTE (B) and T2 HASTE (C) (arrows), but not well demonstrated in T1 GRE (D).
Pulmonary MRI has been used for the characterization and management of a pulmonary nodule or mass. In this setting, STIR, TSE and DWI have reported to be at least as or even more effective as PET-CT for differentiation of malignant from benign nodules. 36,41,46 Moreover dynamic first-pass contrast-enhanced MRI with 3D GRE sequence and UTE is a fast technique which has superior performance compared to FDG PET-CT. 47
MR also has a potential role in the staging of known lung cancer. Given its superb contrast resolution, MR can differentiate tumoral tissue from adjacent atelectasis or consolidation and can accurately depict invasion of adjacent structures, such as the chest wall, pericardium, or diaphragm. However, although dynamic CE MR in a 3 T MR system was found to have higher performance compared to multidetector CT in detecting pleural and mediastinal invasion, currently MDCT remains the modality of choice for the assessment of T descriptor. 2,48 The significant role of pulmonary MRI in assessing the N descriptor has been reported in many studies and STIR turbo SE and DWI are promising techniques. 49 Furthermore, a recent study showed that staging of NSCLC with whole-body MR was comparable to the standard of care staging pathway and reduced staging times and costs. 50
Endometriosis
Endometriosis is defined as endometrial tissue outside the uterus and is associated with inflammation and fibrosis. The pelvic cavity is the most common location. The thorax is the most common site of extra-pelvic endometriosis that may result in the thoracic endometriosis syndrome (TES) with catamenial pneumothorax, hemothorax, hemoptysis, lung nodules, isolated catamenial chest pain and/or shortness of breath. Thoracic endometriosis is often easier to identify at the dome of the right hemidiaphragm. MR imaging is fundamental for evaluation and management. 51
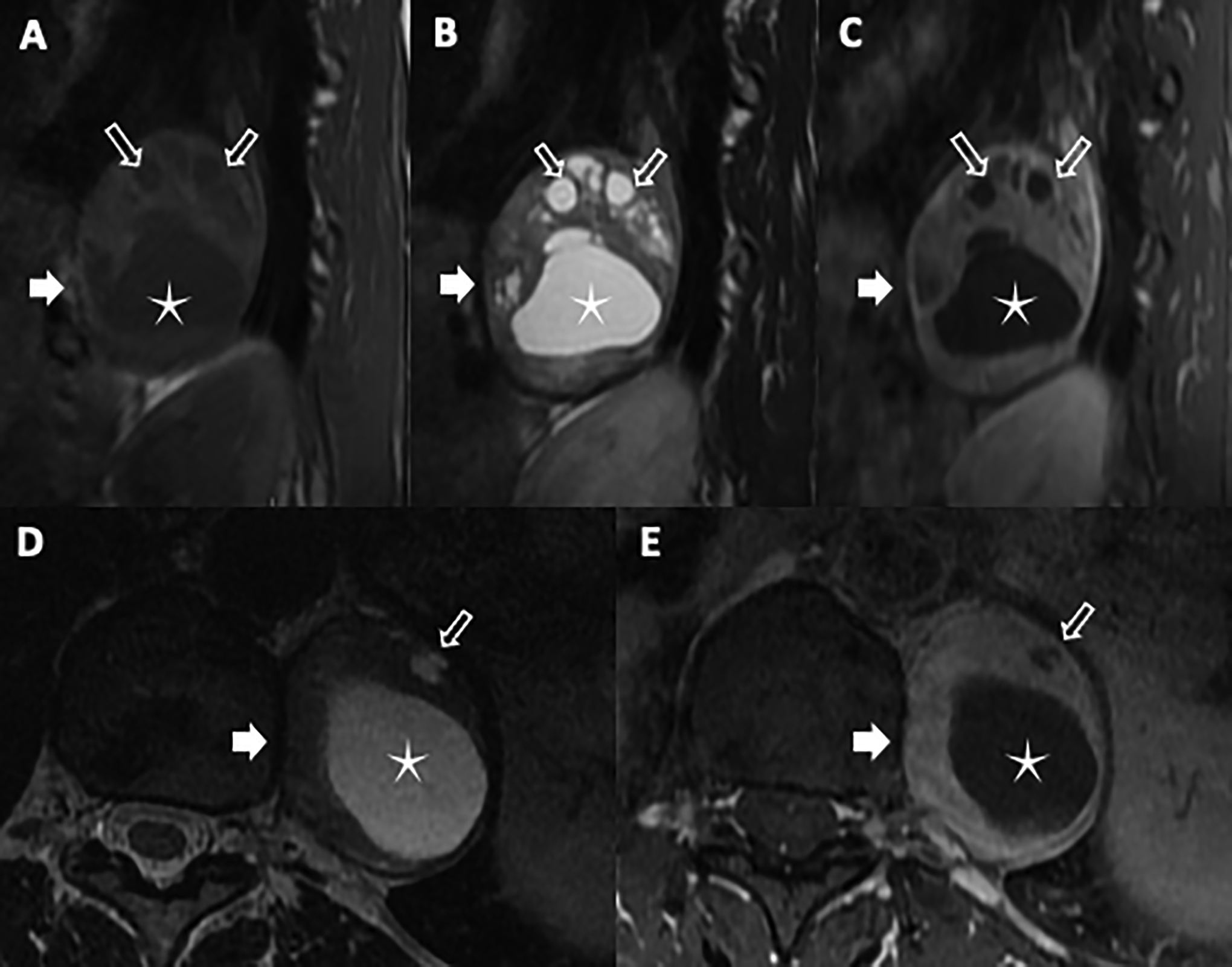
In the appropriate clinical setting, a nodule with high signal intensity on T1-weighted images, high signal intensity on T2-weighted fat-saturated images, enhancement post gadolinium and restriction on DWI is suggestive of endometriosis. 52 -55 In patients with high clinical suspicion of thoracic endometriosis, T1-weighted imaging is the most important. However, post gadolinium sequences and DWI may add additional supportive evidence to increase diagnostic certainty (Figure 11).
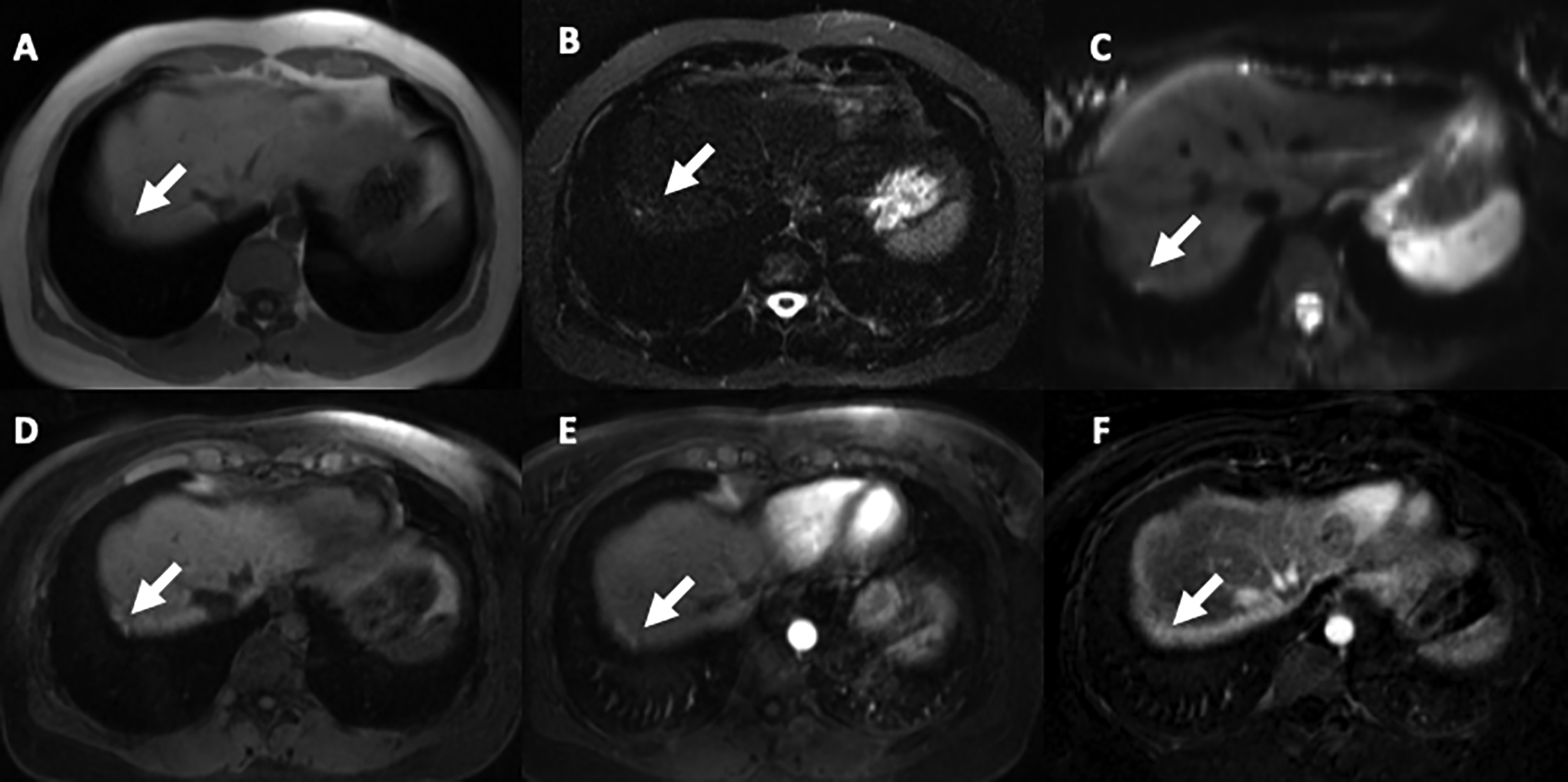
36-year-old woman with known pelvic endometriosis and symptoms suggestive of thoracic endometriosis: Axial thoracic MR images show 2 adjacent foci of high signal intensity at the dome of the right hemidiaphragm with features in keeping with endometrial deposits (arrow). Diagnosis was confirmed pathologically after surgical resection (A) T1-w FLASH in-phase, (B) T2 STIR (only one of the foci is high signal intensity), (C) DWI b50, (D) T1-W GRE fat saturated pre-contrast, (E) T1-W GRE Fat saturated postcontrast, (F) subtraction images confirm enhancement.
Artifacts and Possible Solutions
Thoracic MRI is particularly susceptible to patient-related artifacts due to respiratory and cardiac motion. This can cause diffuse image blurring or ghost images in the phase encoding direction that can obscure pathology and present challenges to accurate interpretation. 56 Therefore, it is important to minimize or remove motion artifacts as much as possible.
Respiratory motion can be minimized by respiratory gating, respiratory compensation, or breath-hold imaging. 57 Breath-hold imaging is preferred because it is faster than other alternatives and can completely eliminate respiratory motion in compliant patients. Usually, the entire chest can be scanned axially in two or three 20-second breath-holds. Given this, our suggested protocols include primarily fast breath-hold sequences such as ultrafast GRE and single-shot fast spin echo (SSFSE) techniques. Coaching the patient before the examination about the breath-hold technique with clear breathing instructions during the examination increases the chances of a successful breath-hold (Figure 12). Also, administration of oxygen via nasal canula (2-4 L) and hyperventilation can improve a patient’s breath-hold capability and subsequent image quality. 58
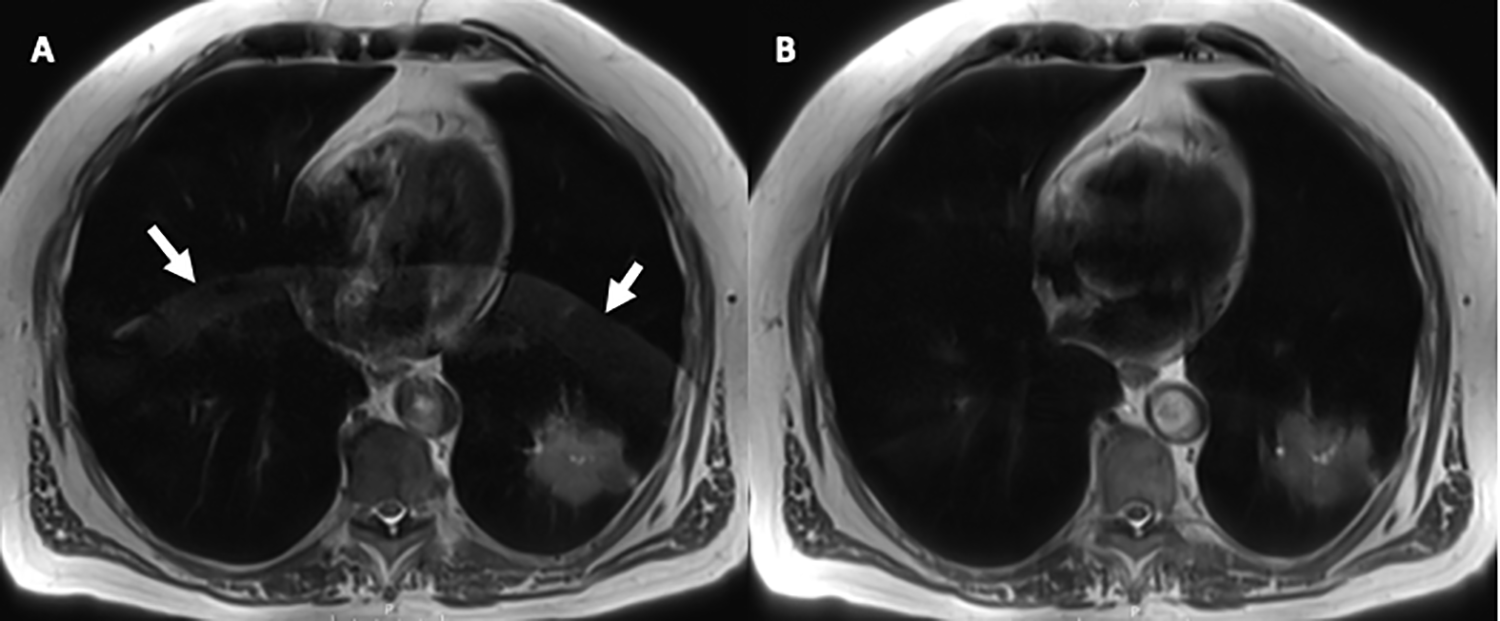
63-year-old man with left lower lobe lung adenocarcinoma. Single shot T2-weighted turbo SE (T2HASTE) resulted in respiratory motion artifact (yellow arrows) despite breath-holding (A). The sequence was repeated after recoaching the patient with breath-holding instructions and repeat T2HASTE immediately after shows significant improvement and almost complete resolution of the artifact (B).
Respiratory triggering is a reasonable alternative for patients unable to breath hold. However, it increases scan times given that images are only acquired during a specific phase of the respiratory cycle. It can also reduce image contrast because the repetition time (TR) is longer than in breath-hold images. 57,59
Cardiac motion can be reduced by ECG gating. However, this technique should be used selectively as it significantly increases scan times given that images will be acquired only during a specific phase of the cardiac cycle and particularly adds to scan time in bradycardic patients. Obtaining high-quality black blood images of the whole chest with double or triple inversion recovery techniques (DIR/TIR) can take several minutes instead of the 40-60 seconds needed for a SSFSE technique. 57,59
Ghosting artifact caused by the thoracic aorta is easily identifiable as a rounded (axial) or longitudinal (sagittal/coronal) structure that repeats vertically or horizontally in the phase-encoding direction in the same plane of the aorta. When it interferes with the evaluation of another anatomical structure, a practical solution is to swap the phase and frequency encoding directions to clear the area of interest or apply a signal saturation band.
Magnetic susceptibility artifact can also be present in chest MR, especially in patients with a history of previous sternotomy and cardiac surgery. In these patients, the sternotomy wires or cardiac devices can cause severe blooming that may limit the evaluation of adjacent structures. Shortening the echo time (TE), widening the bandwidth, or using FSE instead of GRE sequences may help to reduce the amount of artifact. 56
Aliasing or wrap-around artifact occurs when the chest wall outside the field of view produces a signal that reaches the coil. The body part outside the FOV appears “wrapped” or moved to the opposite side of the image. This can be solved by using specific filters (“no-wrap”) or increasing the scan FOV. 56,57 (Table 1).
Frequent Thoracic MR Artifacts and Possible Solutions.

Contraindications and Drawbacks
Non-vascular thoracic MRI can provide vital information and be a problem-solving tool. The non-vascular diseases often evaluated via MRI are infrequently seen, because the malignancies and pathologies that MRI is helpful to assess are not common. As a result, gaining familiarity with protocols, imaging and anatomy can be a challenge for both the interpreting radiologist and the technologist trying to optimize the scans. A technologist who is unfamiliar with anatomy may take longer to plan the sequences and imaging planes. Time is often wasted while a radiologist reviews and adds additional imaging sequences to the scan. This can lead to degradation of imaging as the patient struggles with compliance during longer scan times.
Image interpretation can also be challenging for the radiologist. Familiarity with subtle nuances of the disease and challenges that each of the scans may present is a limiting factor, especially when infrequently encountered. In 2014, Ackman et al. surveyed practicing radiologists and fellows, finding that 77% had reported or interpreted <10 Thoracic MRI studies during residency. Moreover, 64% of respondents had reported fewer than 10 thoracic MRIs in the previous year. 1 This low number of thoracic MR studies completed and interpreted severely limits comfort with the modality.
Finally, imaging protocols for non-vascular thoracic MRI are not fully standardized. Approximately 40% of those surveyed stated that the lack of standardized protocols within and between institutions because of different MRI vendors and/or software was a perceived obstacle to performing non-vascular thoracic MRI. 1 Vendor basic MRI protocols may not result in the highest quality images. Thoracic MRI protocols should be built and modified with combined input from the radiologist, technologist and possibly the MR physicist to ensure image optimization, while balancing overall scan times and patient tolerance. As a result, there may be variations across institutions depending on the most common clinical indications, scanner availability and scheduled time slots. We provide suggested MRI protocols in the supplement section that will work in most clinical practice environments.
Conclusion
Non-vascular thoracic MRI, with the help of recent technological advancements, is becoming increasingly utilized. It is emerging as a powerful problem-solving imaging tool for characterization of thymic, mediastinal, pleural, chest wall and superior sulcus tumors as well as for the assessment of endometriosis, lung nodules and lung cancer staging. Non-vascular thoracic MRI is a recognized imaging modality without ionizing radiation that is particularly valuable for young patients, women of childbearing age, pregnant women and patients requiring frequent serial follow-up imaging. Familiarity with up-to-date standardized MRI protocols that address specific clinical indications and are easily applied across different MR vendors, is the most critical step toward expanding the great potential of non-vascular thoracic MRI.
Supplemental Material
Supplemental Material, sj-pdf-1-caj-10.1177_0846537121998961 - Canadian Society of Thoracic Radiology/Canadian Association of Radiologists Clinical Practice Guidance for Non-Vascular Thoracic MRI
Supplemental Material, sj-pdf-1-caj-10.1177_0846537121998961 for Canadian Society of Thoracic Radiology/Canadian Association of Radiologists Clinical Practice Guidance for Non-Vascular Thoracic MRI by Elsie T. Nguyen, Hamid Bayanati, Ana-Maria Bilawich, Felipe Sanchez Tijmes, Robert Lim, Scott Harris, Carole Dennie and Anastasia Oikonomou in Canadian Association of Radiologists Journal
Footnotes
List of Acronyms
Acknowledgments
We would like to thank Casey Hurrell for her valuable contributions in coordinating our work and preparing this guidance document.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Anastasia Oikonomou: (1) Funding Institution: AFP Innovation Fund—Academic physicians, Ontario Medical Association (OMA) and the Ontario Ministry of Health and long-term care (MOHLTC): “Combination of radiomics signature of lung cancer on pre-treatment PET and MRI and its role in prognostication of patients treated with stereotactic body radiotherapy.” (2) Funding Institution: University of Toronto, Medical Imaging, Heart Lung Vessel Group: “Accuracy of MRI ultrashort TE sequence in detecting solid and subsolid pulmonary nodules and correlation of MRI-based radiomic features with metabolic activity on PET.”
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
