Abstract
Background
Given the multifaceted complexity in the nature of randomized controlled trials, identifying an appropriate and comparable control condition is an essential step to ensure methodological rigor, which allows for researchers to draw unambiguous conclusions concerning the efficacy of the intervention being studied.
Objectives
The objectives of this paper are to (a) review the current literature and analyze the control condition designs in exercise interventions targeted for cancer survivors; (b) provide an overview of the benefits and limitations of various types of control conditions used in exercise interventions; (c) discuss the considerations in the design of control conditions for exercise interventions; and (d) suggest recommendations for control condition design in future trials of behavioral interventions.
Results
The review of randomized controlled trials of exercise training interventions for cancer survivors revealed that the design of control conditions varied. The most commonly employed design could be classified into two major categories: (a) active controls including attention control, add-on controls, and
Conclusions
Careful planning for the control group design is as important as for the intervention group. Researchers can use the considerations presented in the paper to assist in planning for the most appropriate control condition for their study.
Background and Objectives
Background: Exercise Intervention for Cancer Survivors
Advances in cancer treatment and early detection efforts have yield encouraging results. The 5-year survival rate for all types of cancer has improved markedly in Canada since the early 90s (Canadian Cancer Society, 2020). The growing number of cancer survivors in the world has warranted the development of targeted health behavioral health interventions to mitigate the adverse effects of cancer diagnosis and the toxicities of treatments (Tollosa et al., 2019). While researchers have discovered a myriad of behavioral interventions that can benefit the health of cancer survivors, exercise training remains one of the options with the highest therapeutic value on both psychological and physical health (e.g., improved physical functioning and quality of life, and mitigation of anxiety) for this patient population (Liu et al., 2019; Segal et al., 2017).
The World Health Organization recommends that adults (aged 18- 64) should engage in a minimum of 150 min of moderate to vigorous-intensity aerobic physical activity (MVPA) per week (World Health Organization, 2020). While these guidelines were initially extended to cancer survivors, experts in the field have since reviewed the literature and recently published a series of evidence-based exercise prescriptions specifying the frequency, intensity, time, and type of exercise training for cancer survivors during and post-chemotherapy (Campbell et al., 2019). Besides the conventional aerobic or resistive exercise regimes, interventions using a variety of exercise modalities have also shown effectiveness in improving overall health status among cancer survivors (Stout et al., 2017). For the purpose of this study, exercise intervention is categorized as a sub-type of behavior interventions, of which the content comprises training that involves bodily movement aiming at increasing energy expenditure. Exercise training comprises structured, repetitive, and purposeful activities that gear towards enhancing or preserving one or more components of physical health (e.g., cardiorespiratory endurance, balance, flexibility, and musculoskeletal strength) (Campbell et al., 2019; Caspersen et al., 1985; Wolin et al., 2012).
Randomized Controlled Trials of Exercise Interventions
Exercise interventions are often considered complex interventions because they typically comprise multiple components. For instance, interventionists might be required to perform a variety of complex behaviors while delivering the treatment, the intervention design might consist of personalized and tailored activities, there might be multiple levels of study outcomes, and a large number of stakeholders can be involved at different stages of the process (Hecksteden et al., 2018; O’Cathain et al., 2019). For complex behavioral intervention studies, the phase III evaluation stage predominately relies on Randomized Controlled Trials (RCTs) as the gold standard experiment design in estimating the efficacy of the intervention, as well as the causal relationships in the study design (Hecksteden et al., 2018; Polit & Beck, 2021; Sidani, 2015). With increasing evidence supporting the relationship between exercise and physical functioning and psychological health among cancer survivors, clinical trials investigating the effectiveness of exercise intervention have been proliferated in the realm of cancer survivorship care in the past decades (Segal et al., 2017; Zhi et al., 2019). In fact, given that the methodological approach to conduct exercise training interventions is multifaceted and complex, an extension checklist of the CONsolidated Standards Of Reporting Trials (CONSORT) Statement titled the Consensus on Exercise Reporting Template (CERT) had been published in 2016 in order to provide supplementary information to report and document RCTs of exercise interventions (Slade et al., 2016).
The RCT is a robust study design most frequently used in pharmacological trials to establish safety, therapeutic efficacy, and tolerability of newly developed drugs or therapies. The gold standard design of an RCT consists of a double-blinding procedure, and placebo control, which is a pharmacologically inactive agent used to evaluate the effects of being given medications in the experimental group (U.S. Department of Health and Human Services, 2020). Choosing a proper control condition is one of the most fundamental yet critical aspects of the design in an RCT. The control is an essential element that allows the researcher to discriminate whether a treatment has an effect that is due to a hypothesized mechanism of action, and is not attributable to nonspecific effects or other confounders (e.g., sources of bias) (Sidani, 2015). In behavioral intervention studies, researchers can infer the improvements among participants receiving the intervention treatment to the salient features of the intervention itself based on the results of a well-controlled study.
Methodological Issues
Many nonspecific factors such as therapeutic environment, social interaction, and attention from research staff can impact the outcome of a RCT. A properly designed RCT of behavior intervention should allow for the identification of the “active ingredients” responsible for the desired behavior (Edmond et al., 2019; Sidani, 2015). To achieve this, the control condition should not have a significant effect on the hypothesized mechanism of action that explains the effectiveness of the intervention. Unfortunately, the active and inactive components in the intervention treatment are not as well defined as those of medications in pharmacological trials, given that behavioral interventions are often complex (O’Cathain et al., 2019). Further, since behavior intervention RCTs often rely on interpersonal interactions between participants and clinicians, blinding is challenging given everyone is usually aware of the true nature of their treatment allocation. Consequently, the gold standard design for RCTs is less clear in studies investigating the effectiveness of behavioral interventions (Hecksteden et al., 2018).
In exercise science, RCTs adopting robust methodologies are required to provide valid scientific evidence on the types of exercise, training routine, and dosage appropriate for different populations (Hecksteden et al., 2018). Yet, methodological consistency of RCTs of exercise intervention targeting cancer survivors has not been established, particularly in the choice of comparable control conditions (Boutron et al., 2017; Hecksteden et al., 2018; Pinto & Floyd, 2007). Since it is not always possible to develop a deceptive/mimic intervention (i.e., the equivalent to a placebo medication) that generates solely a placebo effect in RCTs of behavioral interventions, researchers have several options for their choice of comparison conditions including inactive control (i.e., wait-list control and usual care) and active/attention control (i.e., in which the participants engage in some activities or tasks during the intervention period.) (Segal et al., 2017; Zhi et al., 2019).
Inactive and Active/Attention Controls
Using an inactive control is a common practice in RCTs of behavioral interventions due to the challenge of identifying an appropriate behavioral “placebo”. Inactive conditions allow for the researchers to detect the outcome of the experimental intervention as compared to that of an un-intervened control group (Karlsson & Bergmark, 2015; Street & Luoma, 2002). The terminologies used to describe the subtypes of inactive controls have not yet been standardized, but they generally include usual care (standard care), no treatment, and wait-list control (Locher et al., 2018; Street & Luoma, 2002). In RCTs adopting usual care as the comparator, participants in the control arm receive the care they would normally get for their clinical conditions. Depending on the nature of the study, the research interventionists might also recommend that the participants maintain their usual habits or activity level, which applies to the context of an exercise training study (Lindquist et al., 2007). The terminology “no treatment” is less common practice in behavioral intervention RCTs; generally, the control arm not performing the intervention activities is described as usual care. Finally, in a wait-list controlled study, participants who are randomized to the control arm receive the intervention treatment upon the completion of the designated study time frame. Participants in the wait-list control arm typically receive usual care during the delay (Elliott & Brown, 2002; Lindquist et al., 2007; Street & Luoma, 2002).
Active controls are sometimes used in RCTs to account for the nonspecific effects of the intervention in a similar way that placebo medications are used to control for expectancy effects in a pharmaceutical trial (Aycock et al., 2018). Active controls are generally recognized as attention controls in behavioral intervention studies, given that participants receive an activity that is an inactive substitute of the intervention, with a similar amount of attention and contact (Aycock et al., 2018; Lindquist et al., 2007; Street & Luoma, 2002). Sub-types of attention controls include several types of component controls, where components of a pre-established intervention package are added on (i.e., additive control), or isolated from (i.e., dismantling control) the study arms in an attempt to identify the active ingredient hypothesized to contribute to the desired changes (Lindquist et al., 2007). In RCTs with multiple study arms, the experimental group is compared to two control groups, with one being a usual care/no-treatment group, and the other control group being an active attention control.
Objectives
Given the multifaceted complexity in the nature of RCTs of exercise intervention and the specialized needs of cancer survivors, identifying an appropriate and comparable control condition is an essential step to ensure methodological rigor, which allows for researchers to draw unambiguous conclusions concerning the efficacy of the exercise training treatment being studied (Hecksteden et al., 2018; Karlsson & Bergmark, 2015). Because the design of control condition continues to be a challenge for researchers, the objectives of this paper were to (a) review the current literature and analyze the control condition designs in exercise interventions targeted for cancer survivors; (b) provide an overview of the benefits and limitations of various types of control conditions available for exercise intervention studies; (c) provide perspectives on considerations in the design of control conditions for exercise interventions; and (d) suggest recommendations for control condition design in future exercise intervention RCTs in cancer survivorship research. Given the authors’ specialization in oncology research, the literature reviewed in this paper are focused on the cancer survivorship studies. The motivation for writing this paper arose due to the challenges faced by the authors of this paper while undertaking an exercise intervention studies for cancer survivors. It is hoped that the cancer survivorship literature can elucidate some of the more salient issues in control condition design. While this paper primarily focuses on exercise training intervention adapted to cancer survivors, the issues and dilemmas explored here are applicable to other behavioral intervention research commonly conducted by nursing researchers such as diet modification, symptom self-management, and mindfulness intervention, etc.
Methods and Procedures
To examine the control conditions designs in RCTs evaluating the effects of exercise interventions for cancer survivors in current literature, a comprehensive literature search was conducted. Three databases, CINAHL, Medline_Ovid, and EMBASE, were searched using Boolean search strategies with truncated keywords. The search strategies for CINAHL are shown in Table 1. Criteria for inclusion were as follow: (1) RCTs of any exercise training interventions involving different training modes, such as aerobic, resistance, weight, and flexibility training were included; (2) the exercise training interventions could be conducted in different settings including home-based or community-based studies; and (3) participants involved in the RCTs had a confirmed diagnosis of any type of cancer, and had received and completed curative treatment. Studies included in the analysis were published in English since 2015.
Search Strategy for CINAHL.

Given the present review's focus on exercise training interventions, studies were excluded if the intervention involved mixed components combining exercise with other therapeutic approaches, such as psychotherapy, dietary modification, or cognitive-behavioral counseling. Finally, trials that compared exercise training with pharmacological and surgical treatments were excluded. When multiple publications from a single RCT were found, only the primary publication was included for this analysis in order to avoid double counting of studies using the same trial design.
Results
Findings
After screening procedures, 32 articles were selected for inclusion in this analysis (Figure 1). The review of RCTs of exercise training interventions for cancer survivors revealed that the design of control conditions varied. The most commonly employed design could be classified into two major categories: (a) inactive controls including no-treatment, usual care, and wait-list control; and (b) active controls including attention control, add-on controls, and dismantling controls, as shown in Table 2
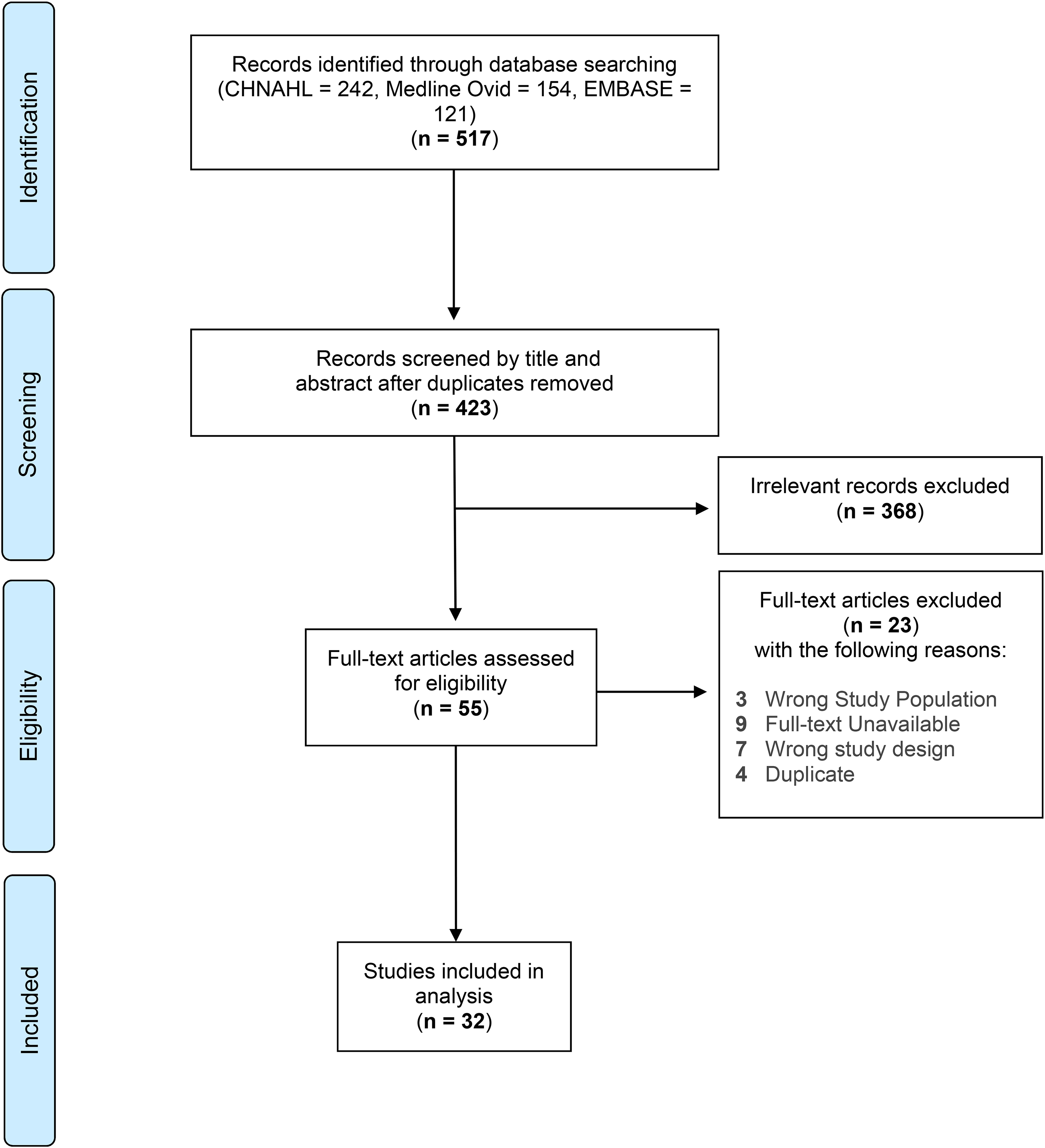
Study selection flow diagram.
Comparison of Control and Intervention Group Components in Exercise RCTs for Cancer Survivors.
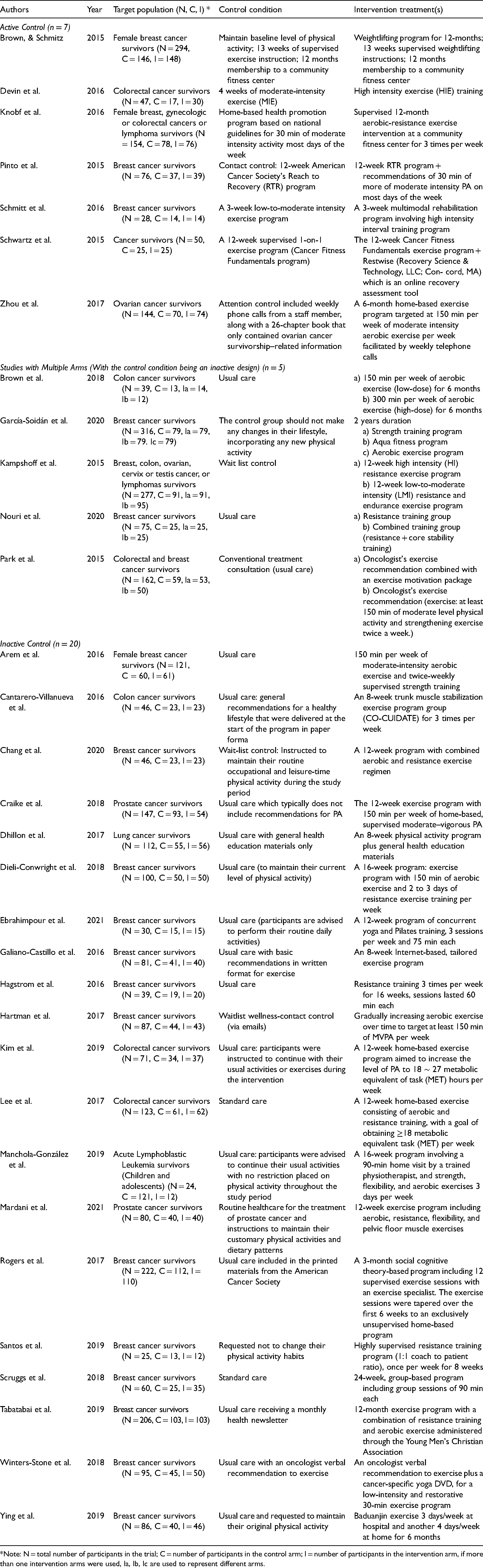
*Note: N = total number of participants in the trial; C = number of participants in the control arm; I = number of participants in the intervention arm, if more than one intervention arms were used, Ia, Ib, Ic are used to represent different arms.
Inactive Controls: Limitations and Benefits
Exercise intervention RCTs included in the analysis most frequently adopted “usual care” as the control condition design (n = 25, 78.1%). Nevertheless, the contents of the usual care activities can vary among studies (Edmond et al., 2019; Lindquist et al., 2007; Thompson & Schoenfeld, 2007). For instance, in a study of a 12-week exercise intervention for prostate cancer survivors, participants in the control arm were not referred to the exercise program. Instead, they received usual care which did not involve any physical activity recommendations (Craike et al., 2018). In another study testing the effects of a 16-week home-based exercise program among acute lymphoblastic leukemia survivors, participants in the usual care control arm received a “physical activity and usual care brochure” identical to that of the intervention group, and were instructed to continue their normal activity levels without restrictions placed on exercising throughout the study period (Manchola-Gonzalez et al., 2020). Lastly, in a study of an eight-week exercise program for lung cancer survivors, the researchers described that the usual care control arm receives general health education material, without specifying the contents of the material (Dhillon et al., 2017). This lack of standardization to quantify the contents of activities in the control condition is considered a key limitation of inactive control designs (Kinser & Robins, 2013; Street & Luoma, 2002; Thompson & Schoenfeld, 2007). As a result, the frequency of participant contacts and the level of attention received among the study arms may vary tremendously (Street & Luoma, 2002).
Using a design that assigns participants to usual care or wait-list control will also render the blinding of participants and research interventionists impossible. Moreover, it is also observed that there is an inadequate reporting, documentation, or oversight of what activities comprise usual care conditions across study settings (Karlsson & Bergmark, 2015). For example, authors may provide limited details regarding the duration of usual care activities or the implementation of standardization of usual care activities. This non-transparency in study design reporting might further conceal the effects of certain biases and non-specific actors that impact the study outcome (Kinser & Robins, 2013; Street & Luoma, 2002; Thompson & Schoenfeld, 2007). The inability to differentiate the treatment effects and nonspecific treatment effects can eventually lead to the inability for researchers to ascertain the intervention effectiveness.
Despite their limitations, inactive control designs are still preferable in many circumstances. Studies adopting inactive control designs such as wait-list and usual care are ideal to control for threats to internal validities including regression to the mean and spontaneous improvement due to the course of illness (Locher et al., 2018). Such designs will be more likely to produce larger effect sizes than a study comparing two active interventions (Kinser & Robins, 2013; Locher et al., 2018). Furthermore, studies using an inactive control group also require smaller sample sizes and fewer resources overall (Williams et al., 2016). Three studies that adopted the wait-list control design in the literature review reported that the use of wait-list control had enhanced study participants recruitment and retention (Chang et al., 2020; Hartman et al., 2018). Wait-list control is often considered an ethical design, given that all participants will eventually receive the treatment intervention. Because the beneficial effects of exercising are well-established and well-known among the general population, this design is particularly appealing and credible to the study participants because they are guaranteed the benefits of the exercise intervention (Hecksteden et al., 2018).
Active Controls: Limitations and Benefits
In exercise interventions, a variety of attention control designs have been implemented. Among the studies included for analysis in this paper, seven RCTs had an active control condition, and five RCTs employed a multiple-arms design. The component control design was implemented in the study aiming to test the efficacy of weightlifting training to preserve muscle mass among a group of breast cancer survivors. In the study, both the control and intervention groups received a 13-week supervised exercise program. The hypothesized active ingredient, weight training, was added-on to the program only in the intervention group (Brown & Schmitz, 2015).
Furthermore, a different dosage of exercise (i.e., different levels of intensity) could be assigned in the intervention and control arms, respectively. For instance, in a study examining the effectiveness of a 4-week high-intensity exercise training program on cardiovascular health among colorectal cancer survivors, the participants in the attention control arm received a moderate-intensity training program of equal length to that of the experimental group (Devin et al., 2016). Likewise, the attention control arm might be assigned to receive a different modality of activity other than exercise training. For instance, Zhou and colleagues designed an RCT where ovarian cancer survivors in the intervention arm were assigned a 6-month home-based aerobic exercise program, and the attention control arm received weekly phone calls from a research team member, along with a book containing ovarian cancer survivorship–related information (Zhou et al., 2017). In multi-arm exercise intervention RCTs, multiple experimental treatment groups of different exercise dosage (Brown et al., 2018; Kampshoff et al., 2015) or different types of exercise training (Garcia-Soidan et al., 2020; Nouri et al., 2020) are compared against an inactive control condition.
Attention control is an ideal comparison condition because it omits the unique ingredients of the intervention treatment while sharing the common factors across conditions to allow for equal measure and comparison (Aycock et al., 2018). In the aforementioned attention control design, researchers aimed to control for nonspecific factors including the amount of attention, participant expectancy, treatment contact, and social support given to both study arms (Aycock et al., 2018). Attention control, therefore, allows researchers to confer an unambiguous conclusion of the hypothesized unique component of the behavioral intervention (Whitehead, 2004). Furthermore, participants in the attention control arm might benefit from the activities assigned regardless (Aycock et al., 2018; Street & Luoma, 2002). Attention control designs present many advantages over the usual care or no-treatment inactive control designs; nevertheless, they are not without limitations. Unfortunately, little guidance concerning the design standards or the required components in an attention control arm for an exercise intervention has been established in the literature (Hecksteden et al., 2018).
Holding the important active study variables constant across study arms (e.g. intensity and timing of intervention) could be challenging (Pagoto et al., 2013). The amount of attention time participants in the control arm receive might not be equivalent to that received by the participants in the intervention arm. Since RCTs often require considerable human resources, time, infrastructure, and financial support, the resources allowance of a given study might constrain the extent to which the activities in the attention control are set up in parallel to the intervention arm (Lindquist et al., 2007). Researchers must also carefully consider the effects that the activities in the control arm might have on the study outcomes (Aycock et al., 2018; Lindquist et al., 2007). Finally, when the attention control consists of activities with completely different structure, modality, or type as compared with the intervention arm, it is also possible that the attention control activities alter the participant's behavior and through a different mechanism of action, thus becoming an alternative intervention itself. If the control arm shares too many common therapeutic qualities with the intervention arm, it renders the control condition limited in comparability with the intervention treatment (Aycock et al., 2018; Gross, 2005). As a result, it becomes difficult to infer an unambiguous cause-and-effect relationship between the active ingredient and desired outcome specified in the study hypothesis (Kinser & Robins, 2013).
Discussion: Considerations and Recommendations
Researchers investigating exercise interventions for cancer survivors face numerous challenges in optimal study design, which are predominantly attributed to the multi-component and multifaced nature of such interventions (Hecksteden et al., 2018; Pinto & Floyd, 2007). Based upon the brief review of current literature, it is apparent that there are numerous pitfalls surrounding control condition designs in RCTs of exercise interventions. A flawless RCT design does not exist, and the researcher must consider each option and make careful decisions by weighing the pros and cons when choosing the most appropriate approach in their studies. Researchers should focus on several considerations while designing a control condition in RCTs of exercise interventions targeting cancer survivors. Four principal considerations concerning control condition design, are discussed in this last section of the paper: appropriateness, credibility, appeal, and comparability. Recommendations on how to avoid some major threats to validity and potential biases are also provided along with the discussion.
Appropriateness
One fundamental principle in control condition design in RCTs is appropriateness. A control condition should be aligned with the overall purpose and objectives of the study, thus enabling the study outcomes to answer the research questions unequivocally (Kinser & Robins, 2013). For instance, when the primary research question is to detect the efficacy of a specific type of exercise training to lessen anxiety among cancer survivors, the intervention arm should be compared to a control condition adopting a different activity while holding the same level of attention and social contact constant (Street & Luoma, 2002). Inactive control such as usual care or waitlist control could also be used to establish the efficacy of the intervention treatment, but the researchers have to take into consideration if the inactive control allows them to adequately isolate the intervention effects from other non-specific effects such as participant expectation. In other words cautious conclusions about the intervention efficacy should be made (Karlsson & Bergmark, 2015).
Alternatively, if the research question concerns the identification of appropriate dosing (e.g., intensity, strength, length, or frequency) of a specific exercise training, the control should be an active control of the same type of exercise training, prescribed at a different dosage. In this case, inactive control conditions would not be appropriate. Finally, an exercise intervention study could aim to examine the efficacy of a specific mechanism of action (e.g., tailored regime vs. traditional exercise regime) or a theory-based variable (e.g., peer support, self-efficacy) on increasing cancer survivors’ level of physical activities (Street & Luoma, 2002; Wolin et al., 2012). For instance, it is common that the exercise intervention consists of training regimes that are personalized to address individual survivors’ physical abilities and specialized needs at the post-treatment transitioning period (Hecksteden et al., 2018; Slade et al., 2016). In this context, active control or wait-list control is the appropriate choice. For ethical reasons, active control or wait-list control are also superior to usual care in cancer survivorship research, considering that the benefits of exercising are well recognized. A no-treatment or usual care control denies the opportunities for the participants to receive a potentially beneficial intervention based on random chances, which might be regarded as ethically problematic (Elliott & Brown, 2002; Street & Luoma, 2002).
Credibility
The extent to which the study participants perceive the intervention as credible can affect their response to the intervention treatments (Street & Luoma, 2002). Ideally, the control condition and the intervention treatment in an RCT should be equally
Appeals and Potential for Social Threats
Relatedly, another essential consideration when designing the control condition in behavioral intervention RCTs is the participant's perceived overall appeal of the study. Both
Participant retention is also associated with the perceived appeal of the study. Participants who regard the control condition as unappealing could withdraw from the study, resulting in attrition, and their data not being available for the final analyses procedure (Davidson & Kaszniak, 2015; Pagoto et al., 2013; Street & Luoma, 2002). Researchers should adopt strategies to enhance the appeal of the study. Matching the control condition with the potential participants’ needs and interests should be considered (Kinser & Robins, 2013; Lindquist et al., 2007). For instance, in a study investigating the effect of a weight training exercise intervention on cancer survivors’ muscle strength, offering the control arm participants a comparable alternative such as aerobic exercise could be an ideal design, since the ultimate desired outcome of the study is health promotion in the post-treatment period in both cases. Seeking a comparable yet acceptable control condition might be more challenging in the case of a novel intervention study (Street & Luoma, 2002). Given that the effect of the intervention treatment has not been previously established, an alternative control condition design such as a delay-start or wait-list control group would be ideal.
Comparability
Comparability of the intervention treatment and control condition must be taken into consideration in the study design. Researchers need to warrant that the study outcomes are not attributable to the activities assigned in the control arm through either the same or a different mechanism as the intervention treatment (Aycock et al., 2018; Lindquist et al., 2007; Locher et al., 2018). A fundamental function of using a comparison group in an RCTs is that it provides a comparable condition (i.e., a condition able to control for the nonspecific features such as the natural course of the disease) to that of the intervention, thus allowing researchers to draw a conclusion regarding the efficacy of the hypothesized active ingredient (Aycock et al., 2018; Karlsson & Bergmark, 2015; Whitehead, 2004). Preferably, similar to the premises given to ensuring the credibility of the study, a comparable control condition should have a parallel design to that of the intervention group, with the sole difference being the active feature in the study hypothesis. Nonetheless, given the level of complexity in behavioral intervention studies, the researcher must ensure that the activity in the control condition does not unintentionally skew the study outcome. This issue can be addressed by limiting the possibility of
Tackling the issue of comparability is challenging in behavioral intervention RCTs, particularly in exercise intervention (Hecksteden et al., 2018). Elements in exercise intervention studies such as format and the nature of engagement could be difficult to mimic. To illustrate, if the intervention involves the use of an individualized, tailored exercise prescription, the alternative activities offered in the control arm probably would not generate the same level of participants’ engagement, social contact, and expectancy. Likewise, it is unrealistic to ask participants in the control arm of an exercise intervention study to stay completely inactive (Hecksteden et al., 2018).
Recommendation for Future Research
Piloting of an RCT is necessary in order to assess for barriers to participant recruitment and to prevent a high rate of attrition (Feeley & Cossette, 2015; Feeley et al., 2009), which can provide insights concerning uncertainties of control condition design of a study. Any issues attributed to control condition designs detected in the pilot trial should be adequately addressed before proceeding to the phase III efficacy trial. In addition, scientific concerns about methodological rigor in RCTs can be overcome by transparent study reporting. It is recommended that researchers provide detailed reports of study design, recruitment, randomization, as well as rationales for choosing a specific design using the guidelines from the CONSORT Statement and the CERT extension (Karlsson & Bergmark, 2015).
Conclusion
In conclusion, the state of science in exercise training is flourishing in the field of cancer survivorship research. Yet, many systematic reviews have recognized the need for more sophisticated methodological approaches and more appropriate controlled studies (Segal et al., 2017; Stout et al., 2017; Zhi et al., 2019). The gold standard RCT design involves the implementation of placebo controls, which are unfortunately not applicable in most exercise intervention studies. Researchers also need to pay special attention to the design of RCTs, especially concerning the effects of the various types of control conditions on study and participant outcomes. To further examine the challenges in developing optimal control conditions, current literature reporting RCTs of an exercise intervention targeting the cancer survivor population were reviewed in this present paper. Furthermore, to address the methodological challenges of exercise intervention studies, four key considerations were reviewed, and recommendations to address each consideration were provided. A well-designed RCT could deliver valid conclusions about the efficacy of an intervention and can smooth the transition of evidence into clinical practice. More meticulous control condition designs are a crucial step towards making exercise intervention more readily available to cancer survivors.
Footnotes
Acknowledgments
Wing Lam Tock is a doctoral nursing student at the Ingram School of Nursing, McGill University. She would like to thank her course instructor, Dr. Nancy Feeley for their support as the manuscript was being developed. She would also like to acknowledge the support from the McGill Nursing Collaborative for Education and Innovation in Patient and Family Centered Care (Newton Foundation/McGill Faculty of Medicine) and Graphos/McGill Writing Centre (Ms. Mariève Isabel) through the Writing for Publication initiative.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author Wing Lam Tock has received financial support from The Faculty of Medicine and Health Sciences, McGill University: Graduate Excellence Award in Nursing; Eileen Peters Award; Réseau de recherche en interventions en sciences infirmières du Québec (RRISIQ): Doctoral Fellowship; and the Cole Foundation Doctoral Fellowship.
