Abstract
Background
Canada is currently experiencing an opioid crisis.
Purpose
Nurses are the largest number of frontline healthcare professionals in Canada who administer narcotic pharmacotherapy, hence, they are ideally placed to improve narcotic stewardship in hospitals. Our study aims to understand the characteristics of narcotic incidents and hence recommend interventions for narcotic stewardship.
Methods
Our study was conducted within a 442-bed academic health sciences center in Ontario. We extracted anonymized narcotic incident reports which occurred over a 3-year period from the SAFER System. Descriptive statistics were utilized to analyze narcotic incidents and their contributory factors.
Results
272 narcotic incident reports were submitted to SAFER within the study period. Most incidents (51%) involved hydromorphone and morphine and were primarily categorized as Level I (n = 154) and Level II (n = 60). Incorrect narcotic dosing (44%), and narcotic count discrepancies (27%) were most commonly reported with active failures being the most commonly reported contributory factors such as failure to review medication orders prior to narcotic administration.
Conclusions
Nurses have an important role in narcotic safety as an intermediary between narcotic administration and incident reporting. Further research is needed to understand the enablers, barriers and opportunities for nurses and other healthcare professionals to improve narcotic stewardship.
Keywords
Introduction
Canada is currently experiencing an opioid epidemic and it is the second largest consumer of prescription opioids, internationally (Canadian Centre on Substance Abuse [CCSA], 2013; Expert Working Group on Narcotic Addiction, 2012; International Narcotics Control Board, 2015). Opioid pharmacotherapy is one of the most commonly utilized interventions for managing chronic non-cancer pain within healthcare settings, and this rate of utilization has been identified as a major contributor to the opioid epidemic (CCSA, 2017).
Prudent opioid prescribing is highly complex and can be defined as an iterative process involving steps of information gathering, clinical decision-making, communication and evaluation, which results in the initiation, continuation, adjustment or cessation of opioids (Nissen et al., 2010). Inappropriate prescribing of opioids, the inadequate or continued prescribing despite evidence of their ineffectiveness, continues to increase and can lead to prescription opioid-related harms such as opioid use disorders (OUDs) and deaths (Bohnert et al., 2011; Turner et al., 2015). For example, there has been a gradual increase in opioid-related deaths over the past 10 years and since January 2016, there has been approximately 15,393 apparent opioid-related deaths, and 19,377 hospitalizations due to opioid-related poisoning in Canada (Government of Canada, 2020).
Nurses are the largest number of frontline healthcare professionals (HCPs) in Canada (approx. 430,000) and are therefore ideally placed to improve opioid prescribing, administration and related interventions (Canadian Institute for Health Information, 2018). Opioid pharmacotherapy is often administered and monitored by registered nurses (RNs), however, these processes of care are not without its challenges (Moghabghab et al., 2016). Advanced practice nurses who treat patients with acute or chronic pain indicate that there are still significant challenges associated with utilizing opioid pharmacotherapy, which can have implications for narcotic safety (Moghabghab et al., 2016). Patient education, drug interactions, dosing, tapering, titration, therapeutic equivalency, and substitution were highlighted as some of the key challenges of utilizing opioids (Moghabghab et al., 2016).
Effective narcotic (opioid) stewardship is patient-centered and can be described as coordinated interventions designed to improve, monitor, and evaluate the use of prescribed narcotics, in order to support and protect human health (Institute of Safe Medication Practices Canada, 2020a). Essential components of narcotic stewardship are identifying, analyzing and reporting narcotic incidents and near-misses to ensure continuous quality improvement (CQI) within hospitals. Such incidents are predominately reported by nurses and can be defined as any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the HCP or patient (Cottell et al., 2020; Institute of Safe Medication Practices Canada, 2020b). Medication incidents including narcotic incidents may be related to professional practice, drug products, procedures, and systems which include prescribing, order communication, product labelling, compounding, dispensing, distribution, administration, education, monitoring, and usage (Institute of Safe Medication Practices Canada, 2020b). Near-misses also referred to ‘good catches’ and can be defined as an error that has the potential to cause harm (e.g., adverse events) but fails to do so because the error did not reach the patient by chance or because it is intercepted due to timely intervention by HCPs, patients or family members (Canadian Medical Protective Association, 2020; Canadian Patient Safety Institute, 2016; Institute of Safe Medication Practices Canada Safety Bulletin, 2007).
Hospital incident reporting systems are often utilized to capture and report narcotic-related incidents and near-misses, which can be indicative of system failures or provider performance issues (CMPA, 2020; Institute of Safe Medication Practices Canada Safety Bulletin, 2007). Subsequently, these incidents should be analyzed and communicated to all clinical staff involved in narcotic pharmacotherapy. Based on incident analyses, organizations can devise tailored preventive interventions such as educational programs for HCPs about narcotics and subsequently, these interventions could be routinely monitored and evaluated for their effectiveness. Also, communication of narcotic incidents and near-misses to senior management is pertinent to facilitate prudent decision-making about hospital-based narcotic stewardship at an organization-level. Therefore, our study aims to obtain a comprehensive understanding of the nature and type of narcotic incidents and their associated contributory factors (where reported) and hence recommend interventions to improve narcotic stewardship.
Methods
Our study was conducted within a 442-bed acute care academic health sciences center in Ontario, Canada. The Safety and Feedback Event Reporting (SAFER) system is the center’s incident e-reporting tool, which has been based on the National Coordinating Council for Medication Error Reporting and Prevention (NCCMERP) framework which provides a standardized taxonomy for medication errors (NCCMERP, 2001). The SAFER dataset consists of 32 mandatory and optional data fields for medication incident reporting including patient demographics (e.g., age and sex), most responsible physician, incident description/details including medication(s) involved (i.e., ordered vs. administered), incident categories (n = 25) e.g., wrong dose, severity level (see Table 1), immediate actions undertaken (e.g., medication discontinued or dose adjusted), reporter designation and contributory factors. We abstracted anonymized narcotic incident reports which occurred between April 2016 and March 2019, from the SAFER e-reporting tool. Next, we analyzed narcotic incident reports using incident categories and other incident characteristics such as severity levels e.g., mild and moderate severity. Within SAFER, incident reporters can use a free-text option to provide a factual account of medication incidents (i.e., incident narratives), these narratives were analyzed to identify proximal and distal contributory factors, using the Yorkshire Contributory Factors Framework (YCFF), where provided (Lawton et al., 2012). This framework uses 20 domains that describe active failures, and other factors e.g., latent factors that can affect patient safety, and has been designed specifically for healthcare settings and is based on systems theory (Lawton et al., 2012). Descriptive statistics were utilized to identify the type and frequency of reported narcotic incidents using IBM SPSS Statistics v25.0. Free-text incident descriptions were coded and analyzed using NVivo v12.0.
Level of harm categories and their descriptions used in SAFER.
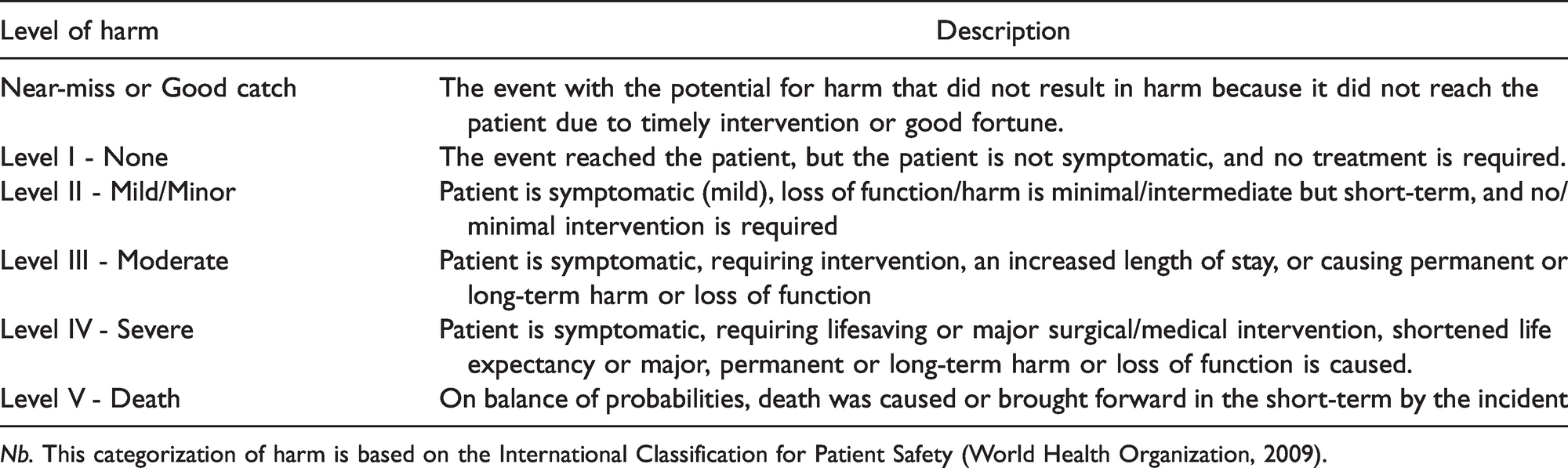
Nb. This categorization of harm is based on the International Classification for Patient Safety (World Health Organization, 2009).
Results
272 narcotic incident reports were submitted to the SAFER system between April 2016 to March 2019 (see Figure 1), representing approximately 20% of all medication incidents reported over the same time period. Reported narcotic incidents predominantly involved hydromorphone (51%) and morphine (20%) and occurred within the departments of Surgery and Oncology (49%), and Urgent and Critical Care (41%). Some reports indicated that incidents occurred within other departments i.e., Women’s and Infant Health (9%) and Pharmacy (<1%). We found that the majority of narcotic incidents were reported by nursing staff (73%), pharmacy staff (5%) however, there was no reporter designation recorded in 22% of submitted reports. The majority of incidents occurred between 6 pm and 12midnight (34%) and between 6am and 12noon (28%). Incidents were categorized as near-miss or ‘good catch’ (n = 13), Level I (n = 154), Level II (n = 60), Level III (n = 31), Level IV (n = 1), and unknown (n = 13) (see Figure 2). There were no incident reports categorized as level V. Some incidents/errors were recategorized to a higher severity level (n = 7) or lower severity level (n = 10) after internal review. We found that the quality of incident narratives was inconsistent across submitted reports. Although, contributory factors were recorded within most incident narratives (78%), their descriptions varied from limited descriptions such as ‘administration error’ to comprehensive descriptions of incidents for analysis within the YCFF and examples are provided in Table 2. An overview of the incident categories utilized for narcotic incident reporting are presented below with illustrative examples.
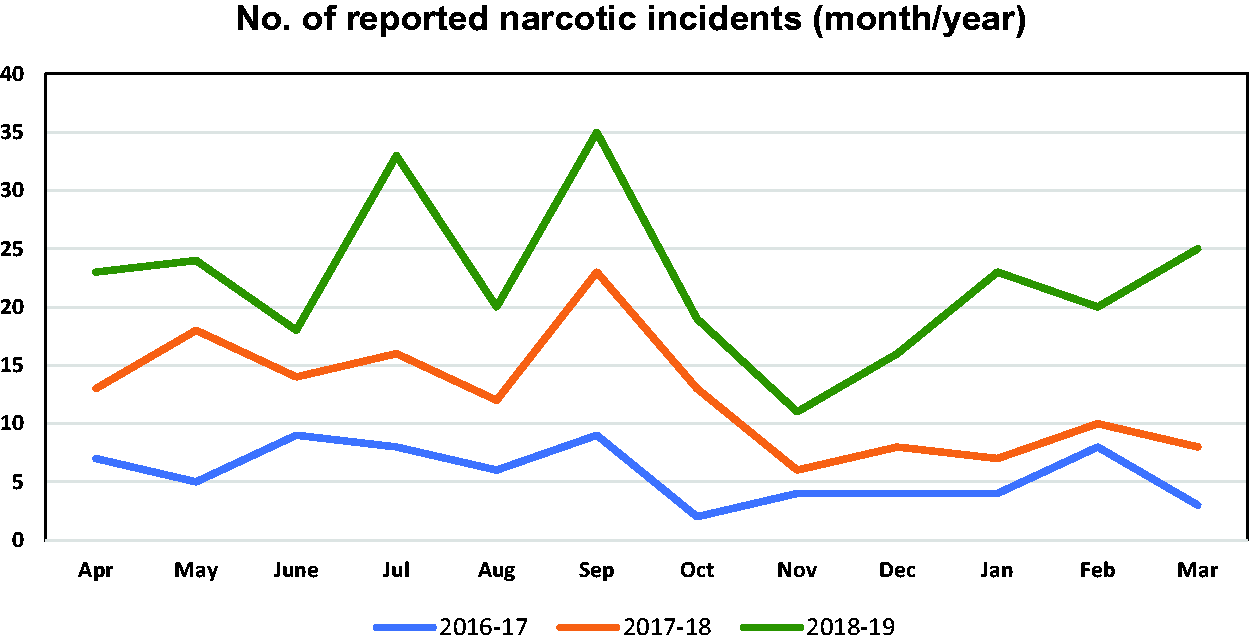
Number of reported narcotic incidents between April 2016 and March 2019—SAFER System (month/year).
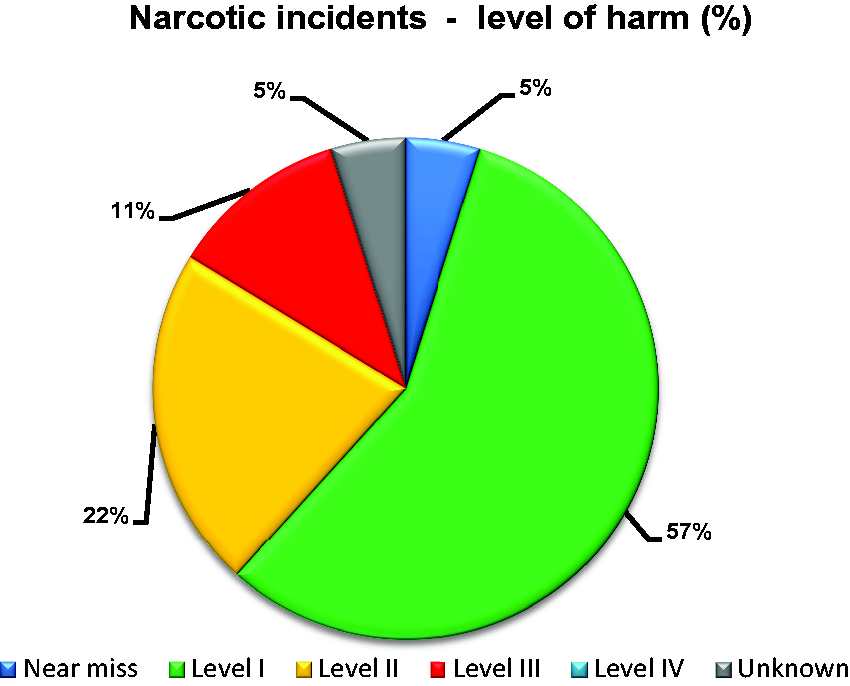
Percentage of narcotic incidents by level of harm—SAFER System. Nb. Level IV incidents were <1%.
Narcotic incidents with examples of contributory factors categorized using the YCFF.

Dose-related incidents (wrong dose)
One hundred and fifteen incidents were reported and occurred when narcotic doses were not administered to patients as indicated on medication orders. Incidents were categorized as Level I (61%), Level II (22%) and Level III (8%) and near-misses (4%). Six errors were not assigned a severity category. Most incidents resulted in patients receiving supratherapeutic or subtherapeutic doses of narcotics and numerous contributory factors were provided within reports for incorrect narcotic doses being administered to patients. For example, incorrect patient charts were reviewed for medication orders prior to narcotic administration by newly hired staff (n = 2), failure to review or confirm medication orders and/or medication administration records (MARs) (n = 25), misinterpretation of medication orders and/or MARs (n = 7), and failure to sign MARs to indicate that medication administration occurred or stopped (n = 2). Dispensing errors (n = 26) were reported, for example, a Level III incident involved 125 epidural cassettes which were prepared in pharmacy containing approximately fentanyl 1 mcg/ml instead of 2 mcg/ml, of which, 50 cassettes were dispensed to a clinical unit. Once this incident was identified, patient charts were reviewed which determined that no patients were harmed as a result of this incident. Dispensing errors also led to the administration of immediate release (IR) instead of continuous release (CR) formulations (n = 5) and vice versa (n = 8). For example, a patient was administered oxycodone 40 mg IR instead of oxycodone 30 mg CR as ordered and another patient was given oxycodone 10 mg CR and not oxycodone 10 mg IR as ordered. Pump programming errors (n = 12) also led to the administration of incorrect narcotic doses, such as 5 mcg/h instead of 5 mcg/ml and 250 mcg/h instead of 100 mcg/h being programmed into pump drug delivery systems. Communication errors (n = 4) were reported such as lack of communication about hydromorphone administration during shift change-overs, failure to confirm a verbal order for hydromorphone and also poor communication about medication order changes between departments i.e., internal medicine, pharmacy and palliative care via the electronic medical record which led to double dosing of hydromorphone 24 mg IR.
Dose calculation errors (n = 2) were reported, one of which occurred on a neonatal unit and involved morphine 35 mg being prescribed instead of morphine 0.35 mg every 8 hours PRN. This calculation error was identified by a pharmacist and the medication order was rectified by the prescribing physician. This incident was categorized as a ‘good catch’ because it was identified before an incorrect dose was administered to the patient. Documentation errors such as incorrect doses recorded on MARs and narcotic sheets (n = 9), lack of knowledge of about narcotic formulations e.g., IR vs. CR formulations (n = 1), patient refusal due to family interference (n = 1), failure to waste narcotics appropriately (n = 2) e.g., 24 mg of unused hydromorphone contin was left at a patient’s bedside (n = 2), workload constraints (n = 2), human error due to distraction or fatigue (n = 2), narcotic storage issues (n = 2), and busy/high utilization of small rooms to prepare narcotics for patient administration (n = 3) were also provided as reasons for incorrect dosing. There were 10 incident reports which did not include descriptions related contributory factors.
Drug selection incidents (wrong drug)
Forty-four incidents were reported which were predominantly categorized as Level I (46%) and Level II (36%) incidents. Most incidents were reported from surgical wards and involved incorrect administrations of morphine instead of hydromorphone and vice versa e.g., morphine 10 mg was administered to a patient instead of hydromorphone 10 mg. Other incidents were reported and involved oxycocet, oxycodone, fentanyl, and methadone being administered instead of the drug(s) prescribed. For example, one incident involved administration of morphine instead of diazepam, the reporter indicated that both drugs were located within the same container and this contributed to the incorrect drug being selected. Another incident involved fentanyl 25 mcg being administered of instead of methadone 2.5 mg, the reporter indicated that the major contributory factor was that both these medications ‘looked-alike’ i.e., both were clear fluids contained within syringes with purple labels. One Level III incident involved a patient being treated for alcohol withdrawal who was given hydromorphone 2 mg on four occasions instead of lorazepam 2 mg as prescribed, which resulted in moderate harm to the patient. Dispensing errors, transcription errors and failure to review medication orders were the most commonly reported causes of these incidents. Other reasons provided included failure to verify patient identity, incorrect interpretation of the medication order and incorrect documentation within nursing records.
Route of administration incidents (wrong route)
Nine incidents were reported, of which (78%) were categorized as Level I incidents.
For example, although an order was double-checked by two nurses, a patient continued to receive an IV administration of hydromorphone 3 mg/ml via a continuous ambulatory delivery device (CADD) infusion pump instead via subcutaneous administration. This report stated, ‘the nurse and I who double-checked the order together and totally missed the route’. Another example involved fentanyl being administered subcutaneously for nine days although IV administration was originally ordered. Misinterpretation and/or failure to review medication orders and MARs were the reported causes of these incidents.
Time/frequency of administration incidents (wrong time/frequency)
Ten incidents were reported which were categorized as Level I (n = 5), Level II (n = 3), near-miss (n = 1) and unknown (n = 1). There was one incident which was recategorized from Level III to Level II after internal review. For example, multiple hydromorphone orders were administered at the incorrect time intervals e.g., an order stated that hydromorphone should be administered every two hours or four hours PRN but it was administered every three hours. Other examples involved a patient who refused to take two doses of hydromorphone 12 mg CR as prescribed (i.e., at 0500 and 0800 BID), however the patient self-administered the prescribed doses together later in the shift. Another incident occurred because the bolus administration frequency for hydromorphone was incorrect because the CADD pump settings were not changed from 20-minute to 30-minute intervals as indicated by the medication order. Communication errors between the primary and covering nurses at handover, and failure to review medication orders were the main reasons for these medication incidents.
Patient identification incidents (wrong patient)
Failure to verify patients’ identification using a two-patient identifier process resulted was the primary reason provided for the three Level I incidents reported. For example, a nurse reported that they administered hydromorphone 1 mg to a patient with a serious hydromorphone allergy before verifying their identity by checking the patient’s wristband. This incident was recategorized to level II after internal review.
Documentation-related incidents (wrong documentation)
Two Level I documentation errors were reported such as the correct medication was given to a patient but documented within an incorrect patient’s MAR. Another incident occurred because an RN utilized a MAR from a transferring health facility to verify which drugs should be administered to a patient. Other documentation errors were reported within the study sample however reporters identified them as contributory factors and not as the main incident category i.e., wrong documentation.
Narcotic count discrepancies
Narcotic counts are conducted as part of the organization’s narcotic reconciliation and security procedures. Narcotic count discrepancies occurred if there were differences between the number of narcotics on the Narcotic and Controlled Drug Inventory Record and the amount on clinical units. Organizational policies specify that if discrepancies are identified, the reason(s) should be documented that includes an explanation of count variance within the SAFER e-reporting tool. Seventy narcotic count discrepancies were reported and were categorized as Level I (n = 44), Level II (n = 6), Level III (n = 7), Level IV (n = 6), near-miss (n = 5) and unknown (n = 2). Reported contributory factors included documentation or transcription errors on narcotic count sheets (n = 7), failure to adhere to the narcotic count and wastage policies (n = 6), and also dispensing and/or administration errors (n = 3). Some discrepancies occurred although there were no patients scheduled for narcotic administration on clinical units. Also, there was one of clinical unit did not conduct narcotic counts for approximately one year. Most reporters indicated that they were unable to locate missing drugs and/or provide a rationale for narcotic count discrepancies (n = 54), for example, reporters stated, ‘unable to locate missing dose(s)’ or ‘unable to determine where discrepancy occurred’. Therefore, it was recommended that narcotic counts should be routinely conducted at the end of each shift and also audits of narcotic cabinet keys on wards.
Disposal-related incidents (wrong disposal)
Inappropriate narcotic disposal procedures resulted in Level I incidents (n = 7). Examples included syringes containing unused narcotics left unattended in a patient’s room on a neonatal unit, a syringe containing fentanyl 1 ml left on an anesthesia cart and another left unlabeled in an operating room, all of which were subsequently wasted as per the narcotic policy. As a result of these incidents, narcotic safety was discussed with both nursing and anesthesia staff during safety huddles.
Other narcotic incidents
Other reported incidents involved incorrect pumps used for narcotic administration which have different drug libraries and drug concentration limits (n = 4), medication not ordered (n = 3), adverse drug reactions or allergy (n = 2) which included a Level IV incident, inappropriate narcotic storage (n = 2) and suboptimal patient monitoring during transfer to another ward (n = 1). However, there was limited description of the causes of these incidents within incident narratives.
Discussion
Our primary objective was to analyze narcotic incident reports submitted to the SAFER system over a three-year period and to understand their characteristics and associated contributory factors and hence to recommend interventions to improve narcotic stewardship. Based on submitted reports, incorrect narcotic dosing (44%), narcotic count discrepancies (27%) and wrong drug (17%) were most commonly reported incidents. Similar to other studies, active failures were the most commonly reported contributory factors such as dispensing errors, failure to review medication orders and MARs and also failure to use two-patient identifiers to verify patients’ identity prior to medication administration (Carson et al., 2009; Dean et al., 2000; Desai et al., 2013; Dy et al., 2007; Heneka et al., 2018; Lawton et al., 2012; Parry et al., 2015; Simon et al., 2005). Active failures can be considered as any failure in performance or behaviour (e.g., slips, lapses, mistakes, and violations) of the person at the ‘sharp end’ within the prescribing pathway and are considered as proximal contributory factors (see Table 2). For example, slips and lapses are categorized as skill-based errors and mistakes are categorized as knowledge-based errors (Reason, 1990, 1995).
Communication systems defined as the effectiveness of processes and systems for the exchange and sharing of information between staff, patients and departments, including written and verbal communication systems was another commonly reported factor (see Table 2). Similar to other studies, hybrid models of clinical documentation and communication (i.e., verbal, written and electronic) were often utilized within the prescribing pathway and these were often cited as underlying causes of narcotic and other medication incidents (Dingley et al., 2008; Frydenberg & Brekke, 2012). We found instances of suboptimal electronic and written clinical documentation which have been shown to be associated with increased rates of medication incidents, particularly prior to medication administration (Parry et al., 2015). Also, we found that incidents due to failure to effectively communicate narcotic administrations tend to occur during shift changes and patient handovers. Staff workload which refers to the level of activity and pressures during shifts, was another contributory factor (see Table 2). For example, some reports highlighted that workload pressures due to nurse-to-patient ratios contributed to active failures resulting in the administration of incorrect doses and/or drugs. Individual nurse factors which included lack of knowledge and experience with narcotic pharmacotherapy of nursing students and agency staff was highlighted as a cause of dosing incidents (see Table 2). Patient-related factors were only reported in two incidents where patients refused to take prescribed medication as stipulated on medication orders (see Table 2). Latent contributory factors describe the organizational conditions in which active failures occur such as physical environments and implementation of narcotic policies and procedures (Reason, 1990, 1995). For example, factors related to the physical environment were reported such as the high utilization of small medication preparation rooms by nurses and other HCPs (see Table 2).
Remedial interventions (error mitigation)
Some incident reports described actions undertaken by clinical units to rectify and mitigate future narcotic incidents and improve narcotic stewardship. Team debriefs of narcotic incidents during safety huddles was the most commonly reported intervention. These incident reviews could be led by advance practice nurses such as nurse practitioners due to their expertise in organizational processes related to medication administration. Additional interventions included reviews of narcotic prescribing processes for methadone, dispensing procedures within pharmacy and on clinical units including methadone labelling, and clinical documentation processes such as the utilization of paper-based and electronic MARs (eMARs). Reviews of medication administration processes including utilizing the rights of medication administration, two-patient identifiers, and also reviews of narcotic policies including the controlled drug withdrawal policy were also conducted. Education and retraining/reinstruction of nursing staff were conducted including narcotic safety, narcotic administration with infusion pumps e.g., CADD Solis pumps, and narcotic wasting procedures. It was recommended within reports that narcotic safety education was particularly important for new agency staff and student nurses since they were responsible for some reported narcotic incidents. However, a recent systematic review has shown that such interventions can have little to moderate effects on medication safety and both didactic and experiential educational strategies may be more effective depending on the environment of care (Härkänen et al., 2016). Improved communication about narcotic administration at breaks and shift handovers was recommended because some communication errors at these times led to patients receiving supratherapeutic narcotic doses. However, there was limited description about whether these interventions were intradisciplinary (i.e., involving nurses only) or interdisciplinary. Patient-oriented interventions included patient reinstruction about patient-controlled analgesia as well as education and counselling about narcotics. We recommend that the center should evaluate the effectiveness of these strategies in improving narcotic stewardship by using frameworks such as the Hierarchy of Intervention Effectiveness framework which provides an approach for rating high leverage interventions e.g., forcing functions to low-leverage interventions e.g., educational programs (Cafazzo & St-Cyr, 2012).
Limitations of SAFER system
We identified a number of limitations of the SAFER e-reporting tool which had negative implications on the data quality of incident reports and should be addressed to improve narcotic stewardship at the study site. Clinical indication was not recorded within SAFER so therefore we were unable to verify the reason for narcotic administration. The center should consider including clinical indication as a mandatory data field because it is necessary to be able to examine adherence to narcotic prescribing guidelines within incident reports. Also, the center should continually incorporate nurse and pharmacist review of narcotic orders and the inclusion of clinical indication during the narcotic ordering processes. We found potentially redundant incident categories which were not mutually exclusive such as ‘wrong time’ vs. ‘wrong frequency’ and also ‘dose extra’, ‘dose omitted’ vs. ‘wrong strength/concentration’ which could be interpreted as ‘wrong dose’. The center should consider collapsing these redundant incident categories into one incident category e.g., wrong dose which would improve the sensitivity of incident categorizations. Other incident categories could be included such as ‘wrong equipment’ to indicate when an incorrect medical device has been utilized e.g., incorrect infusion pump. Within SAFER, only one incident category can be utilized for reporting narcotic incidents, however, we found that there were 29 incidents where multiple incidents categories could be utilized. For example, morphine 10 mg was administered intravenously, instead of morphine 2 mg subcutaneously, which could be categorized as both ‘wrong dose’ and ‘wrong route’. The inability to report or capture multiple categories and causative factors has been highlighted in other studies as a limitation of incident reporting (Carson et al., 2009; Frydenberg & Brekke, 2012). However, the quality of reports improved after updates to SAFER e-reporting tool as well as the introduction of mandatory online educational modules on incident reporting in January 2018.
Although, incident reporting systems can provide pertinent insights into the incident characteristics, several other limitations associated with these systems have been identified. One key limitation is the time lag between incident occurrence and incident reporting which can negatively affect the recall accuracy of incidents. To compensate, incident reporters may use episodic estimation as opposed to episodic enumeration strategies due to the lack of inbuilt recall prompts within reporting systems (Jobe et al., 1993). Reporters tend to report active failures as opposed to distal or latent factors (system factors), potentially due to a lack of understanding of these factors, their interrelationships and the implications on the occurrence of narcotic incidents (Bicket et al., 2017; Califf et al., 2016; Dy et al., 2007; Heneka et al., 2018). Healthcare organizations have a responsibility for improving narcotic incident reporting which is an essential component of quality improvement models targeted at medication safety. Due to its limitations, we recommend that narcotic incident reporting should not be a standalone strategy for improving narcotic stewardship within hospitals. Supplementary strategies including patient chart reviews using the IHI Global Trigger Tool for identifying adverse events, auditing concordance between dispensing and administration records, process mapping, ‘real-time’ incident monitoring and direct observations should be utilized as well as incident reporting (Carson et al., 2009; Classen et al., 2008; Frydenberg & Brekke, 2012; World Health Organization, 2014). Continual reporting and analysis of narcotic incidents and their characteristics such as type, severity, frequency and contributory factors are pertinent for effective risk management and mitigation strategies and seek to limit recurrent narcotic incidents and improve narcotic management (Frydenberg, & Brekke, 2012). Further, inherent system failures (latent failures) should be identified as part of an effective incident reduction strategy, utilizing the principles of systems thinking and clinical operations management (i.e., the examination of inputs and transforming processes that result in outputs related to care provision and delivery) (Carson et al., 2009; Vissers & Beech, 2005). Also, healthcare organizations have roles and responsibilities in ensuring effective narcotic stewardship by addressing latent factors such as ensuring appropriate staffing levels and skill mix of new and experienced staff during shifts and supportive practice environments including medication preparation rooms. Such factors need to be explored further to understand their implications on narcotic safety. Confidential, anonymous and non-punitive narcotic incident reporting could be part of an organization’s safety culture and should be supplemented by effective feedback mechanisms to key stakeholders to promote and implement narcotic safety strategies (Frydenberg & Brekke, 2012). This is important because substandard feedback loops (or communication strategies) for medication incidents to key stakeholders has resulted in recurring and preventable incidents and has been highlighted in other studies (Elden & Ismail, 2015; Shojania, 2008; Vincent, 2007). To promote narcotic stewardship, this center has adopted a ‘Just Culture’ approach for managing medication incidents. This approach fosters a balanced culture between blamelessness and punishment while enabling an environment of openness and fairness to promote honest error reporting (Rogers et al., 2017).
Study limitations
We analyzed 272 narcotic safety reports which were submitted to the SAFER system on a voluntary basis. However, the number of reports submitted to SAFER may not be representative of the actual number of narcotic incidents which may have occurred within the study period, since underreporting of medication incidents is prevalent in healthcare (Armitage & Knapman, 2003). It may be difficult to identify each narcotic incident that occur within the prescribing pathway which includes the preparation, dispensing, administration and monitoring of narcotics. Voluntary reporting may be subjected to reporting bias or selective reporting (e.g., some reporters may consider some incidents to be insignificant to narcotic safety so therefore these incidents may not be submitted). Others may not report narcotic incidents due to actual or perceived punitive implications. We were unable to authenticate the content of narcotic incident reports by reviewing patient charts and other data sources, to determine and/or verify other contributory factors and associated patient outcomes so therefore we were only reliant on the incident narratives provided by reporters. Some reports provided limited details therefore it was difficult to ascertain whether distal contributory factors such as organizational safety culture, design of equipment and supplies, scheduling and bed management contributed to narcotic incidents. Further, our analysis provided insights into the type and nature of narcotic incidents and related contributory factors (where reported) which could be context-specific so these results may not be generalizable to other care settings and should be interpreted with caution.
Recommendations for further research
Interventions are needed to improve the quality of narcotic incident reporting in order to understand the current status of narcotic safety at the study site. Hence, we need to understand the enablers, barriers and opportunities for narcotic incident reporting at the HCP-, unit-and hospital-level including human and other contributory factors. Similar to other studies, this study highlights the important role of nurses in identifying, mitigating and reporting narcotic incidents within current models of care for managing pain, since nurses tend to report the majority of narcotic incidents (Heneka et al., 2018). Therefore, the factors that enable and prevent nurses from conducting these important roles warrant further investigation (Heneka et al., 2018).
A comprehensive understanding of the interrelationships between contributory factors and the conditions that facilitate their prevalence will assist in the development and implementation of robust narcotic safety initiatives at the study site. For example, most narcotic incidents (51%) involved hydromorphone, therefore, we need to understand why there was a high prevalence of incidents involving this drug. Most reports were submitted from surgical, oncological and critical care units however further research is required to understand if there is underreporting from other units. Further understanding of nurse workflows, nurse workloads, the accessibility and usability (e.g., utility of data elements and response options) within SAFER, and also the prioritization of narcotic incident reporting within the current models of care for managing pain, is required. Supplementary examination of the patient educational requirements undergoing narcotic therapy should be conducted, particularly those utilizing patient-controlled analgesia. Ongoing nurse education about narcotic therapy should be continually prioritized particularly for newly qualified nursing staff such as independent double checks before administration for problem-prone drugs e.g., hydromorphone. Further, an essential component of narcotic safety is examining the effectiveness of narcotic incident mitigation strategies within diverse clinical areas.
Conclusion
Current narcotic incident identification and reporting frameworks can be improved. Determining incident causation can be complex and multiple strategies are required to understand the type, and frequency of narcotic incidents and associated their proximal and distal contributory factors such as higher-order organizational factors. The National System for Incident Reporting (NSIR) criteria and accident causation models such as the YCFF could be used as a foundation for designing incident reporting systems supplemented by education about the role and identification of contributory factors in incidents, to facilitate improved reporting by nurses and other HCPs (Canadian Institute for Health Information, 2017; Lawton et al., 2012). Concepts such as stewardship, which is a patient-centred, interdisciplinary approach for improving prescribed drug therapy, could be used as a basis for optimizing hospital-based narcotic risk management strategies and safety cultures. Since nurses are pivotal in administering and monitoring narcotic therapy and reporting narcotic incidents, one approach could be to devise nurse-led narcotic stewardship interventions within current models for managing pain. With the move towards learning health systems, it is essential to optimize data quality for performance monitoring and also knowledge translation processes, to advance narcotic stewardship.
Footnotes
Acknowledgments
We would like to kindly thank Sinai Health System for their time and participation in this study.
Ethical Approval
Ethics approval was granted by the Research Ethics Board (REB) at the Sinai Health System and the University of Toronto (MSH 10–0204-E).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by a Health System Impact Fellowship which was jointly funded by the Canadian Institute for Health Research (Grant no. 503625), MITACS Elevate (IT10235) and the Canadian Nurses Association (CNA).
