Abstract
As Canadian policy-makers recognize the urgency for concerted actions to reduce plastics (e.g., Canada’s involvement in the international plastics treaty negotiations, zero plastic waste strategy, and single-use plastics regulations), the healthcare sector must also consider a more sustainable plastics system. In this context, the potential for novel bioplastics to mitigate healthcare’s substantial plastic waste problem must be carefully interrogated. Our analysis examines the complexities of bioplastics, highlighting the technical challenges of identifying legitimate sustainable alternatives, and the practical barriers for implementing bioplastics as substitutes for consumable plastics in healthcare. We focus on the Canadian healthcare sector and regulatory landscape with the insights gained being applicable to other sectors and countries. Given the limitations identified, the focus on reducing consumption should remain the priority.
Introduction
The healthcare industry relies heavily on plastics due to their low-cost and functional properties of versatility, durability, sterility, and biocompatibility. Plastics are used extensively in all aspects of clinical practice, including in diagnostics, devices, and associated packaging1,2; they also meet many non-clinical needs, such as for furniture and building materials. This dependency has consequences, including emissions of Greenhouse Gases (GHGs), microplastics, and chemical contaminants across the plastic lifecycle (resource extraction, manufacturing, transportation, and use and disposal). 3 Canada’s healthcare system is one of the world’s most carbon-intensive, 4 generating almost 5% of Canada’s GHG emissions. 3 It is also estimated that Canada has one of the highest healthcare waste generation rates in the world, at 8 kg/bed/day. 5 Plastics contribute approximately 25%-30% of all general healthcare waste, although absolute quantities of plastics waste are challenging to determine and likely vary across settings and regions.2,6
The substantial scale of plastics use and waste in healthcare is of global concern, threatening both environmental and human health. 6 As healthcare stakeholders consider ways to reduce these harms, attention is turning toward bioplastics marketed as “eco-friendly.” 7 In this article, we clarify the confusing constellation of concepts associated with the term “bioplastics” and identify impediments to their role as solutions to environmental sustainability challenges in healthcare, drawing on the waste hierarchy framework to inform the management of plastic pollution. 8 Further, we examine whether the existing regulatory context in Canada is adequate to support the evidence-informed adoption of current or future “bioplastic” solutions. While we address specific issues for the Canadian healthcare sector, the issues raised are broadly applicable to other sectors and countries since the substantial scale of plastics use and waste in healthcare is of global concern.6,7
How is healthcare responding to the imperative to reduce plastics?
As the world moves toward an international legally binding instrument on plastic pollution, 9 the healthcare industry needs to pursue concerted actions to reduce plastics use and waste. In Canada, the Single-use Plastics Prohibition Regulations 10 provide clear impetus for change. The regulations ban specific single-use plastics (e.g., plastic straws, checkout bags, and cutlery) and grant healthcare only a partial exemption: for single-use plastic straws for people who require them for medical or accessibility reasons. 10
From an environmental management perspective, plastic waste is best addressed by reducing the use of plastics, as recommended by the waste hierarchy 8 and the concept of the circular economy, 11 both of which have clear application in healthcare.12,13 Yet, given the difficulties of reduction and achieving circularity through recycling, there is growing interest in “bioplastics” as a hoped-for environmentally preferable alternative, alongside reduction of overuse (e.g., of non-sterile gloves),14,15 and increased use of reusables, where feasible.7,16–18
Healthcare already has experience with some “bioplastics,” specifically those made of biodegradable polymers.1,19 Such products have been successfully used in biomedical applications for the last 50 years, for example as delivery vehicles for controlled drug release, suture materials, and therapeutic device implants such as adhesion barriers and stents.1,19
Examples of plastic applications in healthcare and available bioplastics alternatives promoted for their environmental sustainability potential.

aBioplastics alternatives available.
What is the problem with bioplastics?
Ambiguous terminology
The term “bioplastics” is an umbrella concept that has been used to refer to a range of plastic types, including those that are bio-based, biodegradable, or both (Figure 1). The many different meanings of “bioplastics” create confusion, and the misconception that all are safer for human and ecosystem health or more sustainable.21,22 Bioplastics can refer to bio-based, biodegradable, and/or compostable plastics.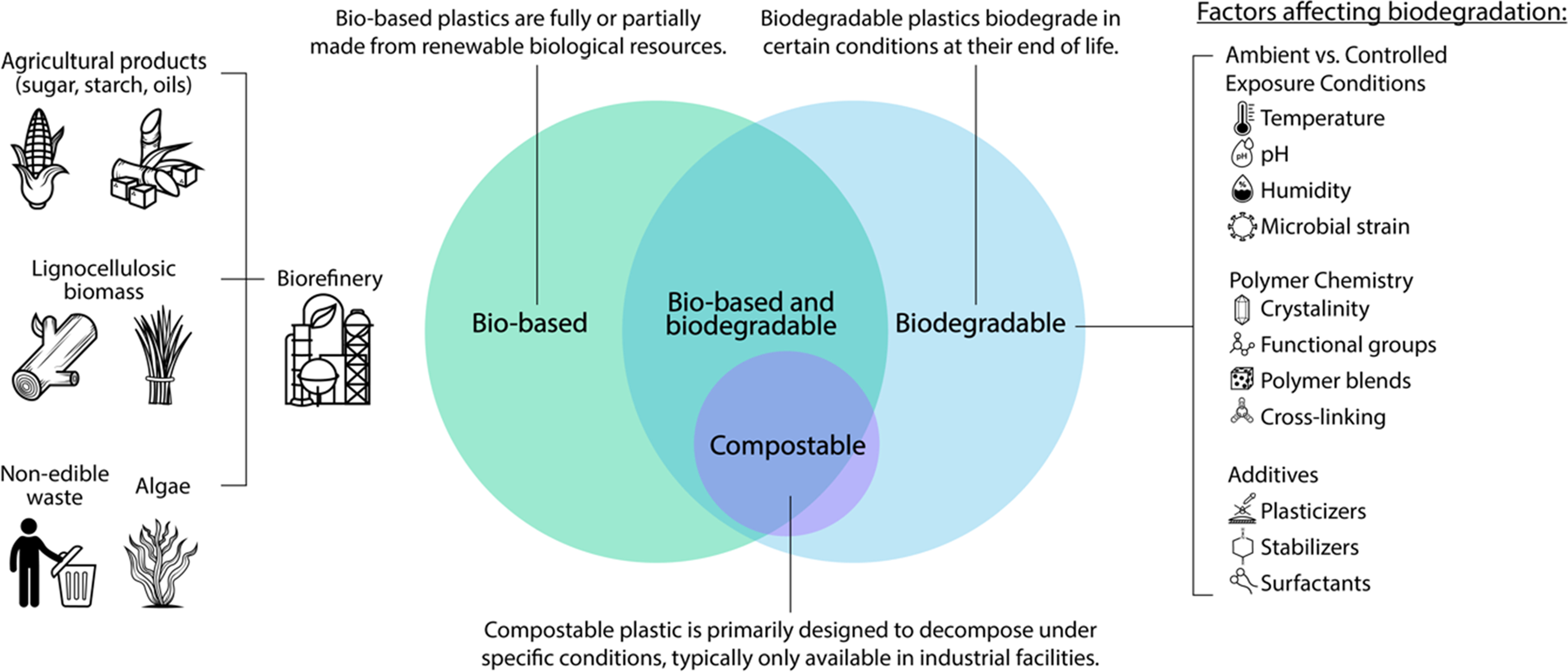
The term “bio-based” refers only to the use of renewable resources for polymer production. 23 That is, it refers to bio-based polymers or bio-polymers, as distinct from fossil-fuel-based polymers, and does not imply end-of-life outcomes such as degradability.21,23,24 Bio-based plastics are not necessarily biodegradable. 21
Biodegradation refers to the process of degradation by microorganisms 23 and does not specify the timeframe for which these processes occur. “Biodegradable” plastics are not necessarily made of bio-based feedstocks; they may be derived from fossil-based materials. 21 Indeed, the term “bioplastics” has been used to describe fossil-fuel-based plastics that are engineered to biodegrade in specific environmental conditions (e.g., polybutylene adipate terephthalate).21,24
Compostability refers to complete decomposition or mineralization 23 by microorganisms to produce carbon dioxide, water, minerals, and biomass, without visually distinguishable or toxic residues. 25 Thus, compostable plastics represent only a subset of biodegradable plastics (Figure 1).
Biodegradable is not necessarily compostable, and not all “compostables” are the same
Plastics’ biodegradability depends on the chemistry of the polymer and conditions for biodegradation21,26 (Figure 1). Whether subject to ambient environmental conditions (e.g., as occurs in soil, freshwater, and marine water) or controlled industrial facilities (e.g., as occurs in some composting facilities), the biodegradation rate (i.e., how quickly) and performance (i.e., how complete) vary widely as they are a function of the material itself, as well as exposure conditions such as presence and types of microbial communities, temperature, moisture, pH, and the time spent in that particular environment. 26
Most commercially available bio-polymers or bio-based materials that are marketed as compostable require conditions only available in specific industrial composting facilities,21,22 emphasizing the importance of end-of-life considerations for each product and their composites. Indeed, the compostability performance of new bio-based plastics requires testing in municipal or industrial waste management facilities. 26 Meanwhile, biodegradable plastics that do not claim to be compostable, or even compostables that end up in non-compositing conditions after use, degrade through partial physical breakdown, which generates microplastics and nanoplastics, with associated risks to human and ecosystem health.27,28 An additional practical challenge posed by biodegradable or compostable plastics is that they are considered contaminants in both conventional recycling and organics waste streams when collected together. 29 Thus, if adopted by healthcare organizations, bioplastic waste threatens existing recycling and composting programs.
The limits of bio-based feedstock sourcing
In addition to challenges with the degradation potential of bioplastics, challenges arise where plastics are sourced from renewable resources, as is the case for bio-based plastics.22,24,30 In theory, the use of renewable rather than fossil-fuel feedstocks (e.g., edible agricultural products, food wastes, and algae) can reduce the GHGs associated with the polymer source itself (Figure 1). Yet these benefits may be counterbalanced by higher energy and water use and GHG emissions over the life cycle of the product.30,31 Evidence shows that using agricultural products for plastics create competition for land and the resources for food production, in addition to other socio-economic impacts associated with industrial scale plastic feedstock production.24,32 Of note, the harms of bio-based polymer feedstocks are experienced more acutely in rural settings and developing countries through impacts on food and water security, and detrimental land use change.32,33
The challenge of chemical additives
Even if the challenges of bio-based feedstocks and biodegradation could be readily solved, bioplastics create environmental challenges because of the mix of chemicals used to create plastic products that meet functional requirements such as durability and flexibility. 21 Most plastics have intentionally added chemical additives (e.g., UV stabilizers, plasticizers, and dyes) or consist of blended polymers and composites, with the aim of improving the desired functionality of the plastic or expanding the range of applications.34,35 However, such composition complicates claims of bio-based sourcing and reduces biodegradability performance, which is inherently at odds with durability. 21 Other additives enter plastics unintentionally, for example, during manufacturing. 34 The presence of both intentionally and Non-Intentionally Added Substances (NIAS) reduces their recyclability and is of concern, particularly upon release while processing plastic waste, as these chemicals may be of toxicological concern.34,36 Simply, claims that bioplastics are safer alternatives lack sufficient evidence.
Canada needs a regulatory framework
A minimum requirement for generating confidence in the environmental sustainability potential of marketed bioplastic alternatives is a consistent set of standards and regulations to guide their application. This minimum requirement has not been met for bioplastics globally, with added challenges in the Canadian context.
Standards aim to add certainty regarding terminology and material characteristics (e.g., compostable and biodegradable), and to minimize inconsistencies in the labelling of products. Unfortunately, several standards exist for each of bio-based content, biodegradability, and compostability, including those from the International Organization for Standardization (ISO) and the American Society for Testing and Materials (ASTM). Therefore, harmonized international standards do not yet exist. 37 Moreover, third-party organizations are relied on to verify performance according to these various standards and provide a label or conformity mark. 37 However, such verifications do not mean that these materials will perform as expected under “real world” conditions—for example, the full-scale industrial facilities that exist in practice—since the tests are often predicated on the use of optimal conditions in controlled laboratory settings. 21
Even if consistent and robust standards exist, they may not necessarily be used by companies marketing the relevant products. Jurisdictions such as California and Washington State have enacted laws 38 requiring that products labelled as compostable meet established ASTM composting standards. 25 In Canada, by contrast, adherence to standards is voluntary. Thus, manufactures and suppliers in Canada are at liberty to label or market their products as they see fit, without third-party verification. Canada’s proposed regulatory framework for compostability labelling of plastics may address these challenges. 29 The regulations would require accredited third-party certification following ASTM (D6400 and D6868) and/or ISO (17088) standards for an item to be labelled as “compostable.” In addition, third-party certification would pertain to real world performance, as the regulations require in-field testing at a composting facility in Canada.
Of course, the challenges posed by bioplastics extend beyond transparency about end-of-life outcomes such as degradability. Information about feedstock (e.g., fossil or bio-based) and chemical additives is also needed to inform practice, but such transparency is rare, with the relevant information routinely deemed proprietary.
Conclusion
Bioplastics have long addressed some biomedical needs, but their ability to address growing demands for environmentally sustainable healthcare is doubtful. Our analysis of the use of bioplastics in healthcare highlights the challenge of vetting legitimate alternatives and ensuring their proper management at end-of-life, suggesting that novel bioplastic alternatives such as biodegradable PPE should not be viewed as a lower impact substitute for single-use consumable plastics. Even if terminology was clarified and product labelling regulations were implemented, bioplastics could only serve as partial solutions to the plastics challenge in healthcare. Fundamental solutions rely on drastically reducing the consumption of plastics, regardless of their feedstock (i.e., bio-based or fossil-fuel-based) or degradation potential, in alignment with the waste hierarchy framework. Health leaders should seek to align with the best practice aim of reduction, a solution that has been reinforced by numerous scientific investigations, including the recent international summary report by Wagner et al. 35
Footnotes
Acknowledgements
The authors thank Luz Paczka Giorgi for figure visualizations.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was supported by an investigator-initiated grant from the Canadian Institutes of Health Research (Funding Reference # 178146).
Ethical approval
Institutional Review Board approval was not required.
