Abstract
Keywords
Introduction
Malignant ascites accounts for 10% of all ascites. 1 Approximately 60% of patients with malignant ascites present with some symptoms, which typically include abdominal distention, abdominal pain, nausea and vomiting, dyspnea, anorexia, and fatigue.2–4 In anticipation of symptom relief, systemic chemotherapy, paracentesis, and abdominal vein shunting 5 have been used; each approach has advantages and disadvantages. Japanese guidelines recommend reduced infusion in terminal cancer patients to achieve relief from malignant ascites symptoms. 6 Studies have also suggested the efficacy of diuretics for related abdominal symptoms.7,8 However, none of these studies provide definitive conclusions regarding the relationship between infusions and diuretics and the relief of symptoms of malignant ascites. Furthermore, the intraperitoneal administration of anticancer and molecular-targeted drugs has been attempted as a new treatment modality; however, the results have been inconsistent, and there is established no standard of care for malignant ascites.9–11
Paracentesis is an invasive procedure but provides rapid and reliable relief of symptoms.12–14 Ninety-eight percent of physicians perform paracentesis as a treatment for malignant ascites, and 89% report symptom palliation. 15 Furthermore, it is reported that an average of 94% of patients who receive paracentesis experience symptom relief. 10
However, due to the supportive nature of paracentesis, ascites often reaccumulates and related symptoms recur after drainage, necessitating frequent paracentesis. In addition, the effectiveness of each repeated paracentesis procedure tends to diminish, and complications (eg, pain associated with puncture, infection at the puncture site, bleeding, intestinal perforation, peritonitis, hypotension, and renal dysfunction) tend to increase. 16 Healthcare professionals are therefore concerned about the complications of paracentesis and its potential negative impact on survival and may hesitate to perform abdominocentesis even when a patient is symptomatic.
Although an observational study in palliative care units found that paracentesis did not affect survival in terminally ill cancer patients, 17 the impact of ascites drainage on survival in acute care hospital wards, where patients receive a variety of treatments (eg, chemotherapy and blood transfusions), is not clear. Unlike palliative care units, the impact of therapeutic interventions on survival is critically important for patients in acute care hospital wards. Frequent high-dose drainage reportedly increases the frequency of hemodynamic instability (eg, hypotension and renal dysfunction).10,16,18 Accordingly, the amount of ascites drainage may be reduced. This is also more likely to occur in small Japanese patients in comparison to Westerner patients, and the clinical guideline for gastrointestinal symptoms in cancer patients in Japan 19 suggests that 1000–3000 mL can be safely drained. Therefore, in clinical practice, frequent paracentesis—with drainage of a small volume of ascites in each procedure—is sometimes used in the hope of minimizing the impact on the patient's general condition and survival. However, it is not clear whether such a strategy is beneficial to the patient. We conducted a retrospective study of patients undergoing drainage of malignant ascites from digestive system cancer in acute care hospital wards to determine the association of different strategies for paracentesis with patient outcomes.
Methods
Study Design
This study is a retrospective cohort study conducted in an acute care hospital based on a chart review.
The study population included patients undergoing initial drainage for malignant ascites due to digestive system cancer (esophagus, stomach, small intestine, colon, rectum, liver, bile duct, gallbladder, and pancreas) between January 1, 2010 and April 30, 2022. Eligible patients had to be at least 18 years of age and have a clinical or pathologic diagnosis of malignant ascites. Patients who underwent only diagnostic (and not palliative) test puncture or who received prolonged (>6 months) continuous drainage of ascites were excluded from the study. Since the median prognosis for patients with malignant ascites is 5.7 months, 2 we excluded patients with longer survival because their prognosis was not affected by malignant ascites. Patients who underwent drainage for pleural and pericardial effusions were also excluded due to their significant impact on survival.
Procedures
Paracentesis was performed when the attending physician determined, based on the patient's symptoms, that paracentesis was appropriate for palliation of their symptoms. The method of paracentesis, amount of ascites drainage, fluid supplementation during or after puncture, and concomitant treatment were determined by the attending physician.
Extracted data included age, sex, performance status (Eastern Cooperative Oncology Group PS) at the time of the initial paracentesis procedure, site of the primary tumor, presence of liver metastasis, treatment for cancer (eg, surgery or chemotherapy), average infusion volume on the day of ascites drainage, use of diuretics or corticosteroids at the time of paracentesis, use of albumin/blood transfusion after paracentesis, ascites drainage volume per paracentesis procedure, total number of paracentesis procedures, total volume of drainage, Support Team Assessment Schedule Japanese (STAS-J) symptom version20,21 immediately before and during the 24 h after the first paracentesis procedure, survival outcome, and laboratory blood data (albumin, total bilirubin, creatinine, AST, and ALT) at the time of the initial paracentesis procedure. STAS-J is the Japanese version of STAS, a scale developed in the United Kingdom for healthcare providers to assess symptoms, and has been validated for its reliability.20,21 STAS-J symptom version is a scale on which physicians and nurses rate 22 symptoms. All symptoms can be rated on the same 5-point scale of 0–4. The unique feature of the scale is that it assesses symptoms based on the disturbance of activities of daily living. 21 Therefore, it is easily used as a proxy rating scale. Complications due to paracentesis were recorded according to the Common Terminology Criteria for Adverse Events version 5.0.
Statistical Analysis
The primary endpoint was survival from the first paracentesis procedure. Secondary endpoints were changes in STAS-J symptoms before and after paracentesis and complications associated with paracentesis. Improvement in symptoms was defined as a ≥1 point decrease in the STAS-J score.
To examine the effect of the ascites drainage volume on the primary endpoint, patients were divided into 2 groups: Those with ≤1500 mL per paracentesis procedure (small volume drainage group) and those with >1500 mL per paracentesis procedure (standard volume drainage group). The rationale for group division was based on the median volume of ascites drainage per paracentesis procedure in the present data (1319 mL) and the lower limit of the single drainage volume (1500 mL) that was considered safe in a previous study. 22 Between-group patient background differences were examined using the χ2 test.
The Kaplan–Meier method was used to estimate survival, and the log-rank test was used to compare the small volume drainage group and the standard volume drainage group. In addition, the Cox proportional hazards model was used to examine the impact of variables on survival. We selected variables by referring to previous studies.2,17,22 Among the eligible cases in this study, there were 113 deaths, and we considered 11 variables were acceptable as independent variables to be applied to the Cox proportional hazards model. 23 Furthermore, we confirmed the Variance Inflation Factor of the variables and confirmed the absence of multicollinearity. The following variables: PS at the time of the first paracentesis procedure, primary site, presence of liver metastasis, blood albumin level at the time of the first paracentesis procedure, the mean ascites drainage volume per paracentesis procedure (total drainage volume/total number of drainage procedures), concurrent chemotherapy, the mean daily volume of fluid infusion, use of diuretics/corticosteroids during paracentesis, and the presence of albumin/blood transfusion after paracentesis were included as candidate explanatory variables.
Group differences in symptom improvement and adverse events associated with paracentesis were evaluated using the χ2 test. All statistical analyses were performed using JMP pro version 16.0 (SAS Institute Inc., Cary, NC, USA). P values of <.05 were considered to indicate statistical significance in all analyses.
Ethical considerations
In order to meet the ethical requirements for observational research regarding participation in the study, an opt-out method was used. All patients and their families were given the opportunity to view information about the study through documentation on the hospital website and to decline participation in the study. The study was conducted in accordance with the ethical standards of the Declaration of Helsinki and the ethical guidelines for epidemiological research of the Ministry of Health, Labor and Welfare of Japan. The study received approval from the Teikyo University Medical Research Ethics Committee (approval no. 21-037).
Results
Patient Characteristics
A flowchart of the study is shown in Figure 1. Among 365 patients of ≥18 years of age with digestive system cancer and ascites, 149 patients underwent drainage of ascites by paracentesis for the first time during the study period. Five of these 149 patients were excluded because they underwent pleural drainage at the same time as ascites drainage; thus, the data from 144 patients were finally analyzed.
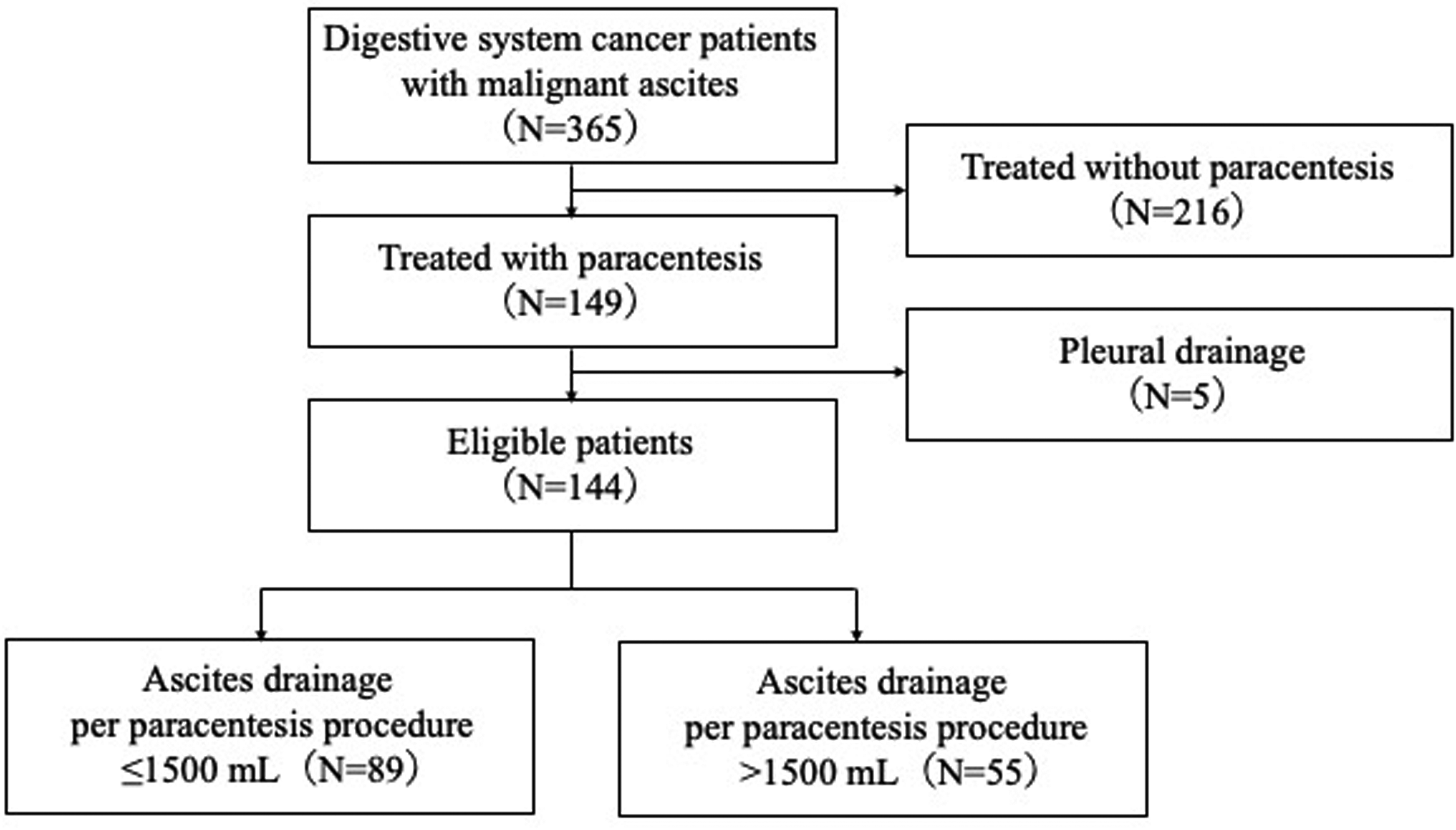
Patient selection flow chart.
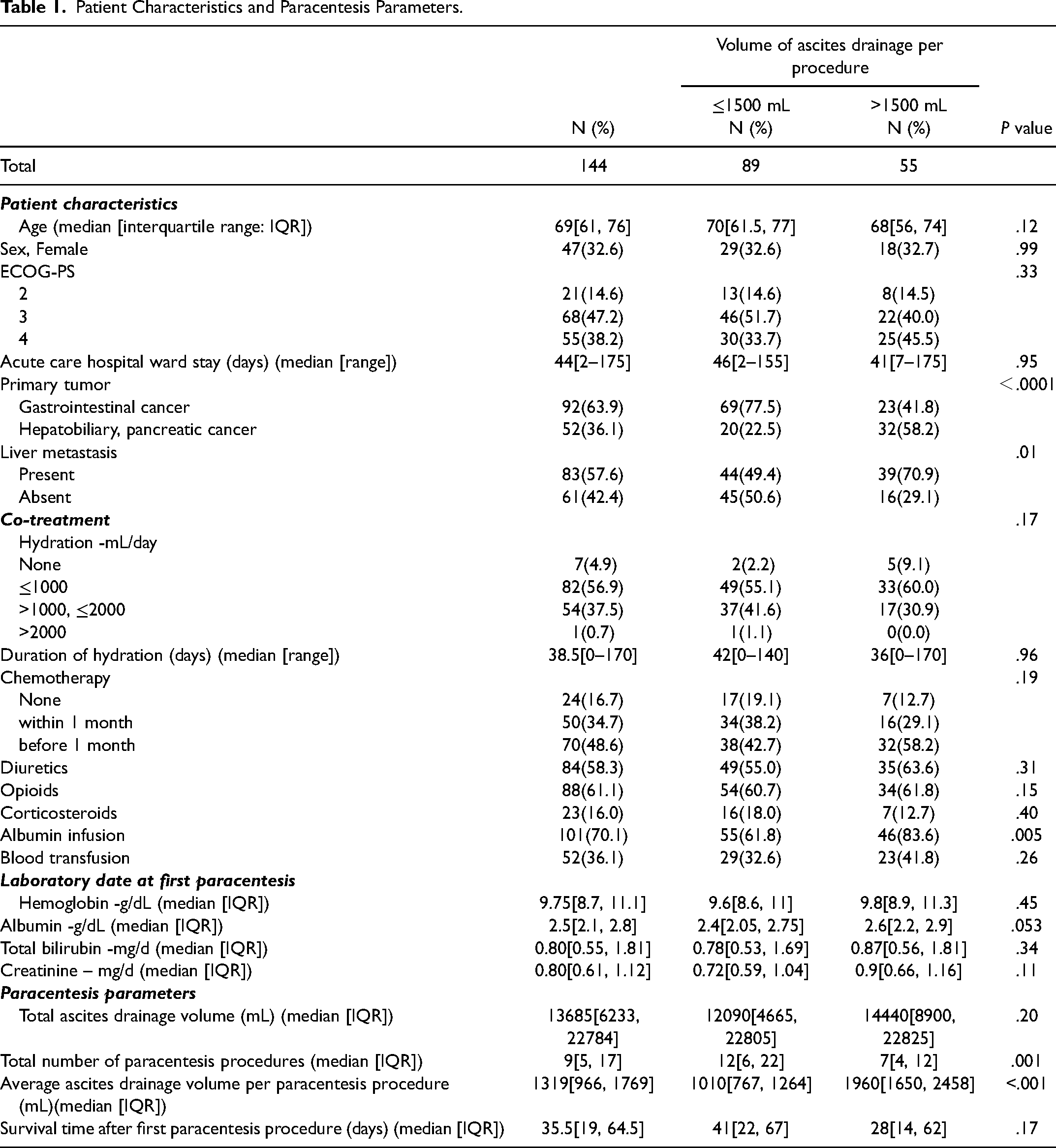
The patient characteristics are summarized in Table 1. The median age was 69 years, 33% of the patients were women, and patients with PS 3–4 accounted for 85.4% of the study population. The primary tumor was gastrointestinal cancer in 63.9% and hepatobiliary pancreatic cancer in 36.1%. Liver metastasis was present in 57.6% of patients.
Patient Characteristics and Paracentesis Parameters.
Treatments
The median total drainage volume from the initial paracentesis procedure to death was 13,685 (range: 750-86,270) mL, the median number of drainage procedures was 9 (range: 1-80), and the median volume per drainage was 1319 (range: 150-4500) mL. The infusion volume at the time of the initial paracentesis procedure was ≤1000 mL/day, in 56.9% of the patients. Albumin, opioids, diuretics, and corticosteroids were administered after the initial paracentesis procedure to 70.1%, 61.1%, 58.3%, and 16.0% of the patients, respectively.
Eighty-nine patients (61.8%) were classified into the small volume drainage group and 55 patients (38.2%) were classified into the standard volume drainage group according to the cutoff value (1500 mL). The total volume of ascites drained in the small and standard volume drainage groups was 12,090 mL and 14,440 mL, respectively (P=.20), and the number of paracentesis procedures was 12 and 7, respectively (P=.001). The standard volume drainage group had higher incidence rates of hepatobiliary pancreatic cancer (P<.0001) and liver metastasis (P=.0113), and a higher level of albumin products after ascites drainage (P=.0054).
Survival
During the follow-up period, death occurred in 74 patients in the low-drainage group and 39 patients in the standard-drainage group. In the 31 patients lost to follow-up because they were discharged home or transferred to other hospitals, the median follow-up time from the first paracentesis procedure was 35 days.
Kaplan–Meier curves for the small drainage and standard drainage groups are shown in Figure 2. The median survival in the small drainage and standard drainage groups was 50 (95%CI: 36-54) days and 44 (95%CI: 23-71) days, respectively (non-significant: P=.76).
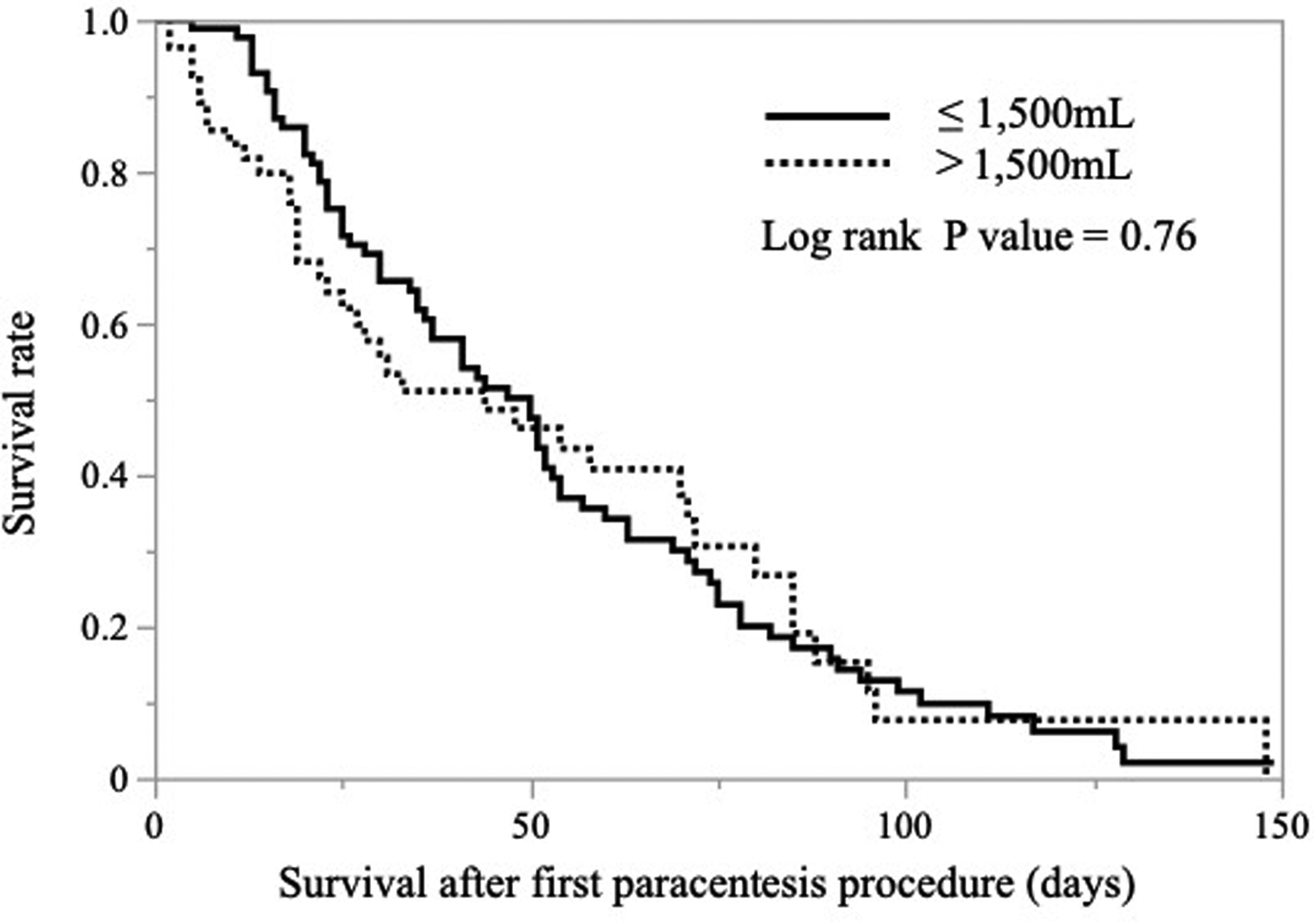
Kaplan–Meier survival curves for the small-drainage and standard-drainage groups.
Table 2 shows the results of a multivariate Cox proportional hazards analysis. No liver metastasis, good PS before paracentesis, blood transfusion after paracentesis, and chemotherapy within 1 month of paracentesis were significantly associated with longer survival, but not with the amount of ascites drainage per paracentesis procedure.
Multivariate Cox Proportional Hazards Analysis of Factors Associated With Survival.

Symptom Palliation
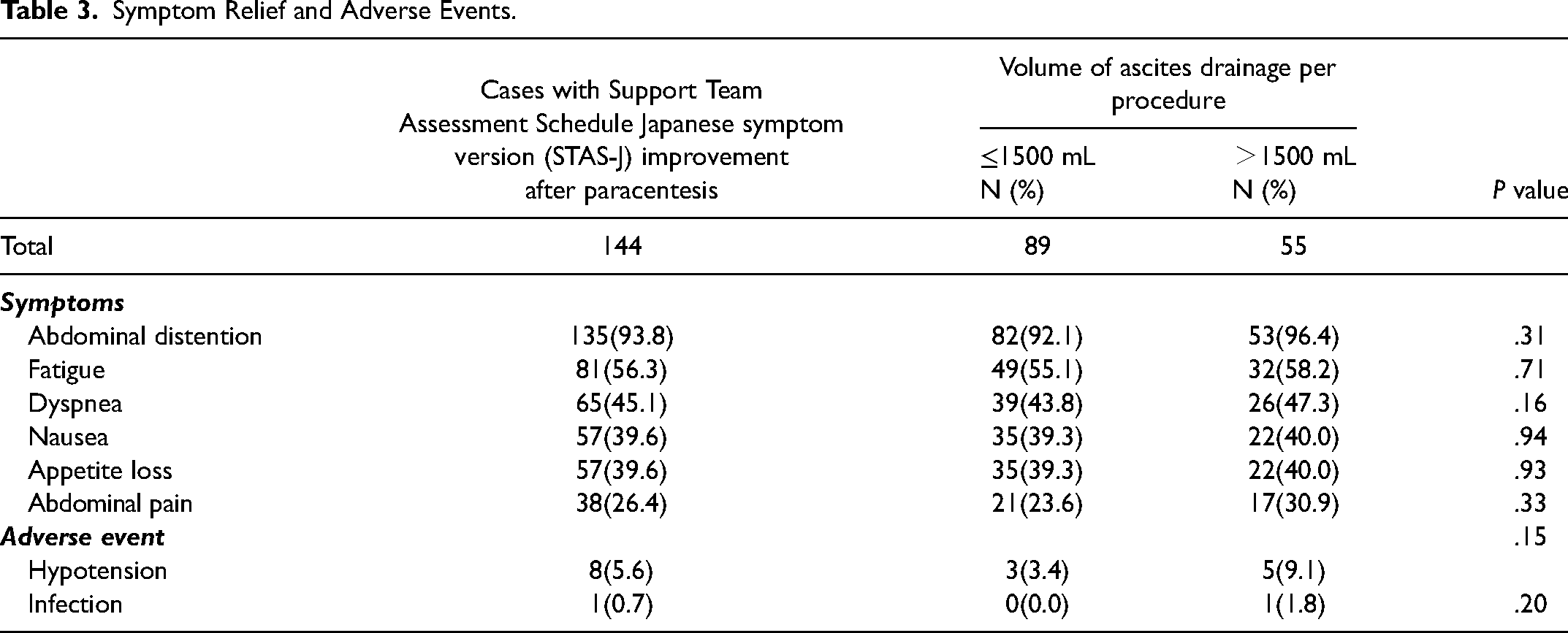
All patients had some symptoms that were considered to have been caused by malignant ascites. The percentages of cases in which symptoms improved (according to the STAS-J score) from before to after ascites drainage were as follows: Abdominal distention, 93.8%; fatigue, 56.3%; dyspnea, 45.1%; nausea, 39.6%; decreased appetite, 39.6%; and abdominal pain, 26.4% (Table 3). None of these improvements were significantly associated with the amount of ascites drainage.
Symptom Relief and Adverse Events.
Adverse events
Adverse events included Grade 1–2 hypotension in 8 patients (5.6%). There was no association between the volume of ascites drainage and the occurrence of hypotension (P=.15). Only 1 patient in the standard drainage group developed a local infection at the puncture site, which improved after 5 days of antibacterial treatment. No serious adverse events (grade ≥3) were observed
Discussion
This study demonstrated that a strategy of limiting the amount of drainage and increasing the number of drainage procedures is not associated with longer survival and that paracentesis is effective and safe for patients with malignant ascites from digestive system cancers who were admitted to wards in acute care hospital. Complications associated with paracentesis were infrequent and not severe, and there was no association between the volume of per paracentesis procedure and the occurrence of adverse events.
We think the strength of this study is that it is the first to examine whether limiting the amount of ascites drainage is associated with survival in acute care hospital wards.
A previous study in palliative care units suggested that ascites drainage of approximately 2000 mL in a single procedure is not associated with survival in terminally ill cancer patients. 17 However, the previous study did not take place in acute care hospital wards, where the concurrent treatments are more varied and where the impact on survival is more likely to be emphasized. In the present study, the small-drainage group had the same total drainage volume as the standard-drainage group, despite having a less than approximately 2000 mL of ascites drained per paracentesis procedure, which a previous study 17 suggested was not associated with survival; instead, puncture was performed more frequently. The results of the present study showed that limiting the amount of drainage per drainage was not associated with prolonged survival or the incidence of adverse events.
On the other hand, it was also shown that various symptoms caused by malignant ascites could be improved by the drainage of a small amount of ascites. This may support the results of a previous study 24 that reported symptom relief after the first few liters of drainage of 5000 mL of ascites by paracentesis. In this study, abdominal distention, in particular, was markedly improved by drainage of ascites, possibly because it is a symptom of increased intra-abdominal pressure due to fluid retention. Even if the volume of ascites suggested by imaging is not large if a physical examination reveals strong abdominal distension, drainage of ascites with the aim of improving abdominal distention, may be considered.
Furthermore, we were able to examine the efficacy and safety of ascites drainage in patients undergoing chemotherapy and blood transfusion, as this study involved patients in an acute care ward. In the multivariate analysis performed in this study, a good PS (reflecting the general condition) and liver metastasis (reflecting the stage of cancer), chemotherapy, and blood transfusion after abdominal drainage were selected as factors that could prolong survival. It is reasonable to assume that patients undergoing chemotherapy will have good survival, since their PS and organ function are preserved to some extent, and the tumor itself is expected to be controlled by chemotherapy.
The eligible patients in this study were a population with a poor PS; approximately 80% had PS3 or PS4. Since it is possible that drainage of a larger volume of ascites in a single procedure may have a greater effect on patient survival in patients with a poorer PS, we performed a sensitivity analysis (Figure 3) to determine if the effect of a single drainage volume on survival varied according to PS; however, we found no change in the effect with PS. Considering the results of this sensitivity analysis, we believe that the findings of this study are consistent, at least in patients with PS3 and PS4.
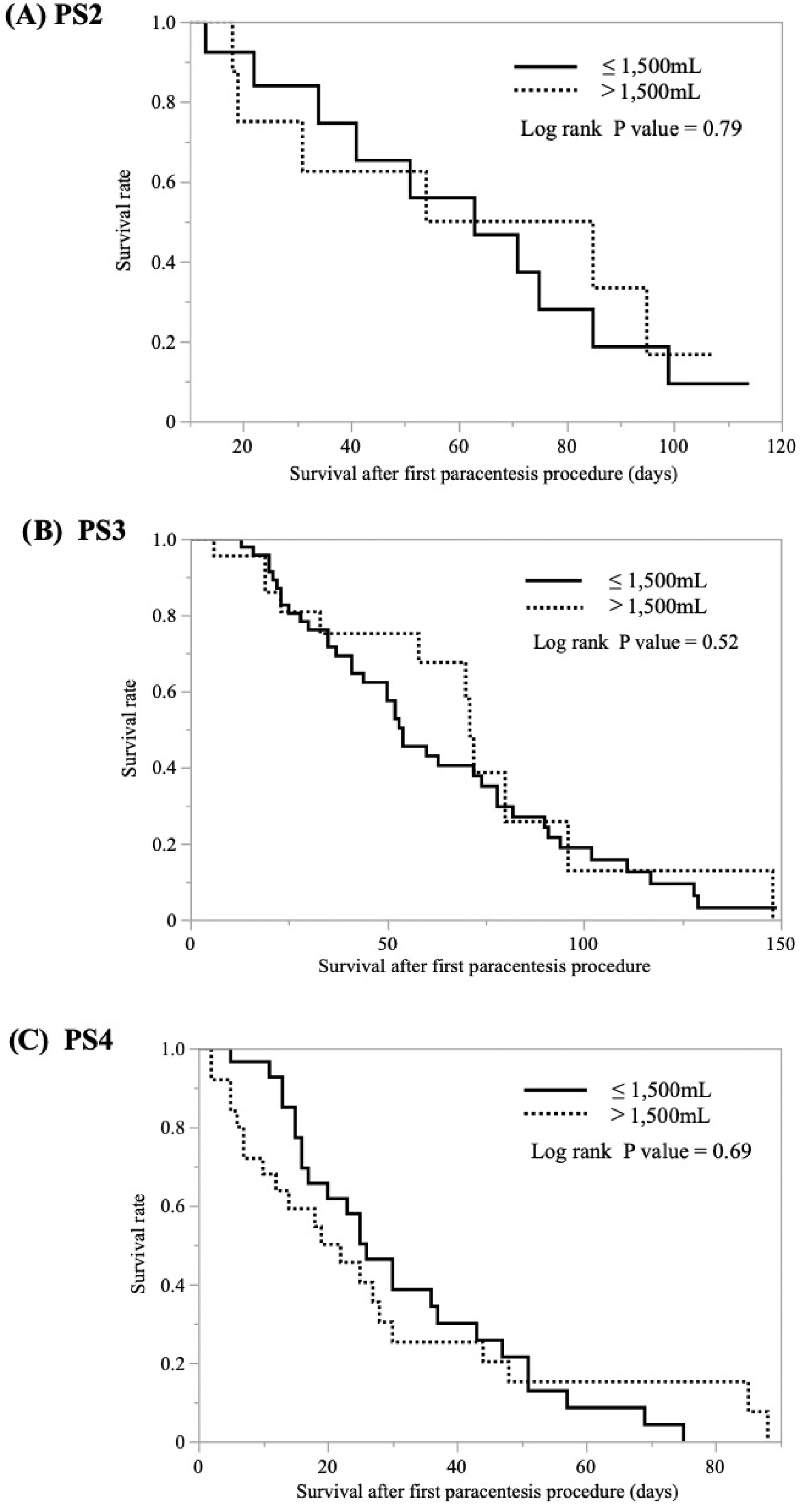
Kaplan–Meier survival curves for the small-drainage and standard-drainage groups for each Performance Status (PS 2-4).
The results of this study may reduce the concern among healthcare providers and patients in acute care hospital wards that the drainage volume of malignant ascites should be limited to avoid shortening patient survival. In addition, it may help in the decision to choose ascites drainage for symptom palliation.
The present study was associated with several limitations. First, this was a single-center, retrospective study, which may have led to a selection bias. There may be unobservable confounding factors, such as the preference for paracentesis by the physician in charge and the frequency of patient requests for peritoneal puncture, which should be validated on a larger scale in a multicenter setting. Second, the study was limited to patients with digestive system cancers. In particular, patients with malignant ascites due to ovarian cancer, which is estimated to account for 25%–40% of all malignant ascites, are known to have a better long-term prognosis than those with malignant ascites due to digestive system cancer, because ovarian cancer causes malignant ascites from a relatively early stage in the disease course and the response rate to chemotherapy is relatively high.1,25 Whether the findings obtained by this study can be applied to malignant ascites due to gynecological and other cancers needs to be examined in another study. Third, there may be confounding factors other than the data we collected. For example, the cardiac function and oral fluid intake may affect the rate of ascites retention, and patient body size may play a role in the effect of ascites drainage on the general condition. However, given the sample size of this study, it was not appropriate to include all potential explanatory variables in the statistical analysis; thus, appropriate variables were selected a priori through a literature search. Fourth, we were not able to track the changes in albumin levels after paracentesis. Hypoalbuminemia, which is frequently observed in patients with malignant ascites, has been shown to be a prognostic factor in patients with malignant ascites. 2 In the present study, many cases lacked data on albumin levels, despite the fact that 70% of the patients were treated with albumin transfusion after paracentesis. However, the multivariate analysis showed no significant association between albumin supplementation and survival, suggesting that albumin levels have a limited effect on survival in patients with malignant ascites. Fifth, this study covers more than 12 years, and chemotherapy and other anticancer therapies have changed during the study period. Therefore, it is possible that patients at the beginning and end of the study period received different anticancer therapies, which may result in a different speed of ascites accumulation.
Conclusion
This study suggests that paracentesis is effective and safe for acutely ill digestive system cancer patients with malignant ascites and that there is no association between limiting the volume of ascites drainage to ≤1500 mL and survival in such patients. However, because this was a retrospective observational study in a single center, further prospective studies are needed to establish more effective and safer methods of ascites control.
Supplemental Material
sj-jpg-1-pal-10.1177_08258597231170049 - Supplemental material for Is Low Volume Drainage of Ascites Associated With Improved Survival in Digestive System Cancer Patients With Malignant Ascites?—A Retrospective Cohort Study
Supplemental material, sj-jpg-1-pal-10.1177_08258597231170049 for Is Low Volume Drainage of Ascites Associated With Improved Survival in Digestive System Cancer Patients With Malignant Ascites?—A Retrospective Cohort Study by Shunya Hoshino, Yusuke Takagi, Takeo Fukagawa, Keiji Sano, Nobuhiko Seki, Yojiro Hashiguchi and Etsuko Aruga in Journal of Palliative Care
Footnotes
Acknowledgement
The authors would like to thank Miyoshi Sakai, MPH (Department of Hygiene and Public Health, Teikyo University School of Medicine) and Shuji Ando (Department of Information Sciences, Tokyo University of Science) for advice on the statistical analyses and Kiyoko Uno, MD, PhD (Teikyo Academic Research Center) for her advice on study design. We would like to thank Japan Medical Communication (![]() ) for editing the English language of this manuscript.
) for editing the English language of this manuscript.
Author Contribution
Shunya Hoshino made contributions to the conception and design of the study, acquisition of data, analysis of data, data interpretation, and revision of the manuscript. Shunya Hoshino and Yusuke Takagi drafted the manuscript. All authors approved the submitted version. Yusuke Takagi and Etsuko Aruga made contributions to the study design, data interpretation, and revision of the manuscript. Takeo Fukagawa, Keiji Sano, Nobuhiko Seki, and Yojiro Hashiguchi contributed to the data collection and assembly, data interpretation, and revision of the manuscript. All authors have approved the submitted version of the manuscript and agreed to be accountable for any part of the work.
Data Availability
The datasets collected and analyzed in this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
Ethical Approval
The study received approval from the Teikyo University Medical Research Ethics Committee (approval no. 21-037).
Informed Consent
In order to meet the ethical requirements for observational research regarding participation in the study, an opt-out method was used. All patients and their families were given the opportunity to view information about the study through documentation on the hospital website and to decline participation in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
