Abstract
Introduction:
Opioid overprescription after surgery results in a surplus of tablets that become available for diversion and abuse. This study aims to describe opioid consumption after common elective outpatient cosmetic surgeries to provide a reference for surgeons and deter preemptive overprescription. Secondary endpoints were risk factors for increased opioid use after cosmetic surgery and the influence of helium plasma technology (Renuvion; Apyx Medical Corporation, Clearwater, Florida) on postoperative opioid consumption.
Materials and Methods:
Patients completed a preoperative survey on the day of surgery and a postoperative survey 14 to 21 days after surgery. Data regarding procedure type, demographics, quantity of prescribed opioids, pain experience, amount of opioids consumed, and supplementation with over-the-counter pain medications or alternative therapies were collected.
Results:
Two hundred sixty-six patients were included. On average, 28 opioid pills were prescribed, and 13.8 pills were consumed across all cosmetic surgery types. Procedure type, body mass index, ethnicity, prior surgical history, and the incorporation of helium plasma technology did not influence opioid consumptions postoperatively. Patients with a surgical complication had a statistically significant higher opioid use than those without a surgical complication (P = .0397).
Conclusions:
Patients in our study required on average about half of the opioids prescribed during the immediate postoperative period. Means of opioids consumed per cosmetic surgery category are described and can serve as a reference for cosmetic surgeons.
Introduction
Short-term opioid use is a mainstay treatment for acute pain after surgery. 1 Undertreatment of pain postoperatively is associated with various adverse outcomes including increased risk for chronic pain, higher health care costs, and an overall increased morbidity and mortality.1,2 Despite their efficacy managing acute pain, opioids pose serious risks for patients, particularly relating to opioid use disorder and overdose. 3 From 1997 to 2017, nearly 218 000 deaths in the United States were related to overdoses from prescription opioids. 3 It is estimated that 1 in every 25 adults uses opioids regularly and about 2 million people suffer from opioid use disorder and addiction in the United States.3,4 In light of the opioid epidemic, there is increased awareness of physician opioid-prescribing patterns, specifically those of surgeons who account for 37% of opioid prescriptions. 5 About two-thirds of opioids prescribed after ambulatory surgery are unused and particularly in plastic surgery, only half of opioids prescribed are routinely consumed.5,6 This study aimed to describe opioid consumption after common elective outpatient cosmetic surgeries to provide a reference for cosmetic surgeons. Secondary endpoints were risk factors for increased opioid use in the immediate postoperative period and the influence of helium plasma technology (Renuvion; Apyx Medical Corporation, Clearwater, Florida) on postoperative opioid consumption.
Materials and Methods
Following Institutional Review Board (IRB) approval (Solutions IRB, #2021/02/38), a prospective, observational study was conducted investigating patients undergoing elective cosmetic procedures at an office surgery center in Miami, Florida, from April 2021 to June 2021. Patients undergoing the following procedures were eligible for inclusion: liposuction with or without autologous fat transfer, abdominoplasty with or without liposuction, breast augmentation, mastopexy with or without breast implants, gluteal augmentation, rhinoplasty, rhytidectomy, blepharoplasty, brachioplasty, cruroplasty, combined procedures involving 2 or more aforementioned surgeries, and cosmetic surgeries with radiofrequency-helium plasma technology. Exclusion criteria were participants aged less than 18 years, inability to read written English or Spanish, revision or emergency surgery, and intraoperative use of liposomal bupivacaine (Exparel). Patients operated by 2 cosmetic surgeons and 4 plastic surgeons were included in this study. No changes were made to the surgeons’ prescribing practices. Selection of opioid type, quantity, and use of adjunct pain medication were at the surgeon’s discretion.
Patients who elected to participate in this study completed a preoperative questionnaire assessing prior surgical history and ethnicity. Additional data collected from participants’ charts included age, sex, body mass index (BMI), opioid type, and quantity prescribed. A telephone questionnaire was then conducted between 2 and 3 weeks postoperatively. Patients self-reported use of prescribed opioid, supplementation with nonopioid pain medication, subjective pain experience, and surgical complications were recorded. Preoperative and postoperative telephone survey questions are listed in Table 1. All surveys were conducted by the cosmetic surgery fellow and research assistant in the participant’s preferred language. Data were deidentified for subsequent analyses.
Preoperative and Postoperative Surveys.

Statistical analyses were performed using SAS software 9.4 (SAS Institute Inc., Cary, North Carolina). The R package ggplot2 was used to create bar plots and boxplots showing the opiate pills distribution across surgical subgroups. Summary statistics were used to describe the patient cohort. Associations between categorical covariates and continuous variables were assessed with χ2 tests and independent-sample t tests, respectively. Differences across means in surgical subgroups were tested using the analysis of variance (ANOVA). All tests were 2-tailed, and P value of <.05 was statistically significant. Patients who did not fill opioid prescriptions were excluded from statistical analysis.
Results
A total of 302 patients consented for participation, of which 266 patients completed the study. Three patients requested removal from the study when contacted to complete the postoperative telephone survey and 33 patients were lost to follow-up. Baseline demographics and distribution of survey answers are listed in Table 2.
Demographics.
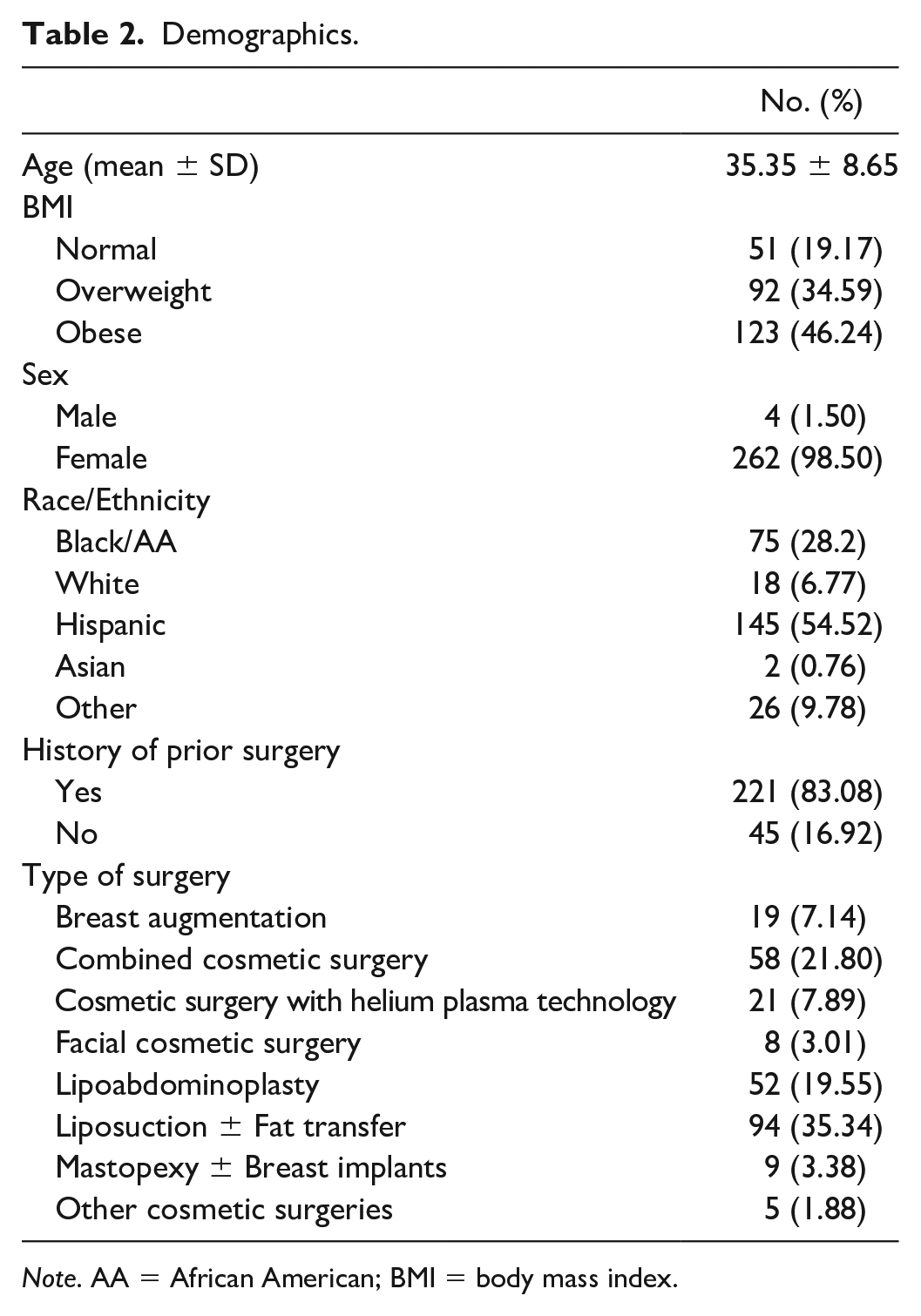
Note. AA = African American; BMI = body mass index.
The mean age was 35.35 ± 8.65 and the mean BMI was 29.09 ± 4.24. Most patients (83.1%) reported a history of prior surgery, with 42% having had a prior cosmetic procedure.
Opioids most frequently prescribed in this study were oxycodone-acetaminophen 5/325 mg (80%) and hydrocodone-acetaminophen 7.5/300 mg (15%). Other opioids prescribed (5%) were hydrocodone-acetaminophen 10/325 mg, codeine-acetaminophen 30/300 mg, and tramadol 50 mg. Across all cosmetic surgeries, an average of 28 opioid pills was prescribed per patient.
The postoperative telephone survey was conducted at 15.78 ± 2.12 days after surgery. When asked about pain experience, 48.5% of patients reported that their pain was better than expected. Most patients (87.6%) reported being satisfied with their surgery. Nine patients (3.4%) expressed being unsatisfied with their surgery and 24 patients (9%) declined to answer the question at the time the survey was conducted. Fourteen patients (5%) did not fill the opioid prescription.
Among patients who filled the opioid prescription, an average of 13.8 pills were consumed per patient across all cosmetic surgeries. Nine patients (3.6%) who filled the prescription did not consume any opioid pills, whereas 61 patients (24.2%) finished the opioid prescription. Most patients (93.3%) reported no longer using opioids at the time of the survey. Only 17 patients (6.7%) continued to use the prescribed opioids. More than half of the patients (79.4%; 200/252) supplemented their postoperative pain regiment with acetaminophen and/or nonsteroidal anti-inflammatory drugs (NSAIDs). A total of 103 study patients (38.7%) used herbal or additional supplements for pain relief. Among these patients, topical formulations and supplements containing arnica were most commonly reported (73.8%, 76/103). The reported mean opioids consumed by study patients who did not use acetaminophen, NSAIDs, herbal, or additional supplements was 13.7 pills per patient. A total of 2045 tablets were reported unused by all patients.
Cosmetic surgeries were grouped into surgical categories for statistical analysis as outlined in Table 2. Most combined cosmetic surgeries included lipoabdominoplasty with a breast procedure (82.6%). Facial cosmetic surgeries in this study included 5 rhinoplasties, 1 otoplasty, 1 rhytidectomy with submental liposuction, and 1 rhytidectomy with platysmaplasty. Five procedures were grouped under Other Cosmetic Surgeries: 2 brachioplasties, 2 breast implant exchanges, and 1 gluteal implant augmentation with liposuction. There was no statistically significant difference in the mean opioid consumption between the surgical categories (P = .3922, Figure 1). Box-plot figure detailing opioid amounts prescribed, consumed, and unused by surgical category is shown in Figure 2.
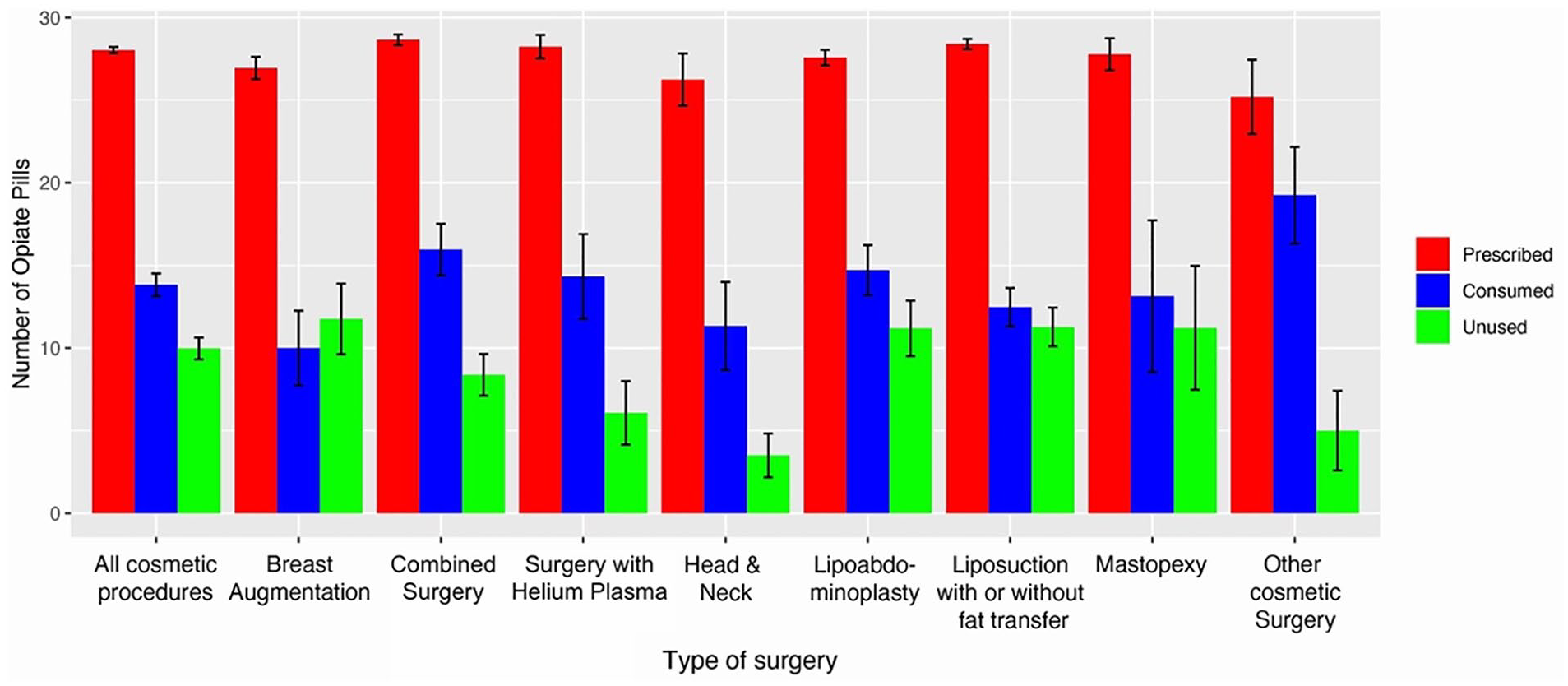
Prescribed, consumed, and unused opioid tablets after outpatient cosmetic surgery.
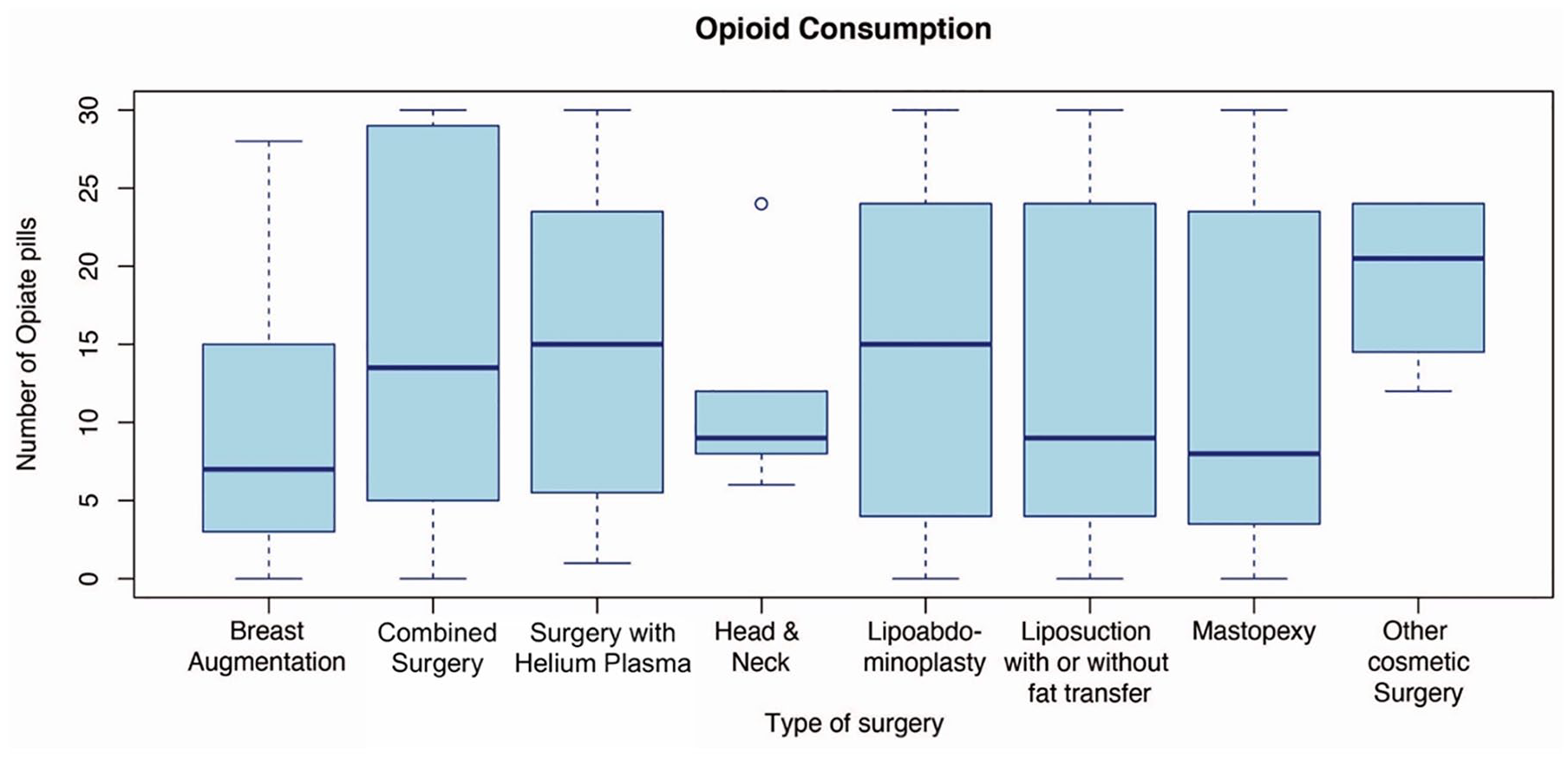
Consumed opioids after outpatient cosmetic surgery.
Across all cosmetic surgeries, the reported mean of opioids consumed by patients with a BMI >30 was 14.2 pills per patient compared with 13.5 pills per patient with a BMI <30 (P = .6465). Patients with a prior surgical history reported an average opioid consumption of 13.8 pills per patient compared with 13.97 pills per patient without any prior surgeries (P = .9286). The average opioid consumption by reported ethnicity was as follows: 13.0 pills per patient who identified was black/African American, 13.3 pills per patients who identified as Hispanic/Latino, 17.6 pills per patient who identified as white/Caucasian, and 17.09 pills per patient as other race/ethnicity (P = .1749).
Thirty-seven study patients had complications related to their surgery. The most common complications were wound dehiscence (43.2%) and seroma (19.4%). Four patients (1.5%) were hospitalized following their surgery: 1 patient for anemia requiring blood transfusion, 2 patients for treatment of wound infections with intravenous antibiotics, and 1 patient for a complicated wound dehiscence. One hospitalized patient reported not filling the opioid prescription. The mean reported number of opioid pills consumed per patient with a surgical complication was 17.2 pills compared with 13.2 pills per patient without a complication (P = .0397). In addition, 33.3% (12/36) of patients who had a surgical complication finished the opioid prescription compared with 22.7% (49/216) of patients without a surgical complication (P = .1673).
Cosmetic Surgeries With Helium Plasma Technology
A total of 21 cosmetic surgeries incorporated helium plasma technology. Most were liposuction procedures with or without autologous fat transfer (76.2%). The reported average of opioid pills consumed across all cosmetic surgeries with helium plasma technology was 14.3 pills per patient compared with 13.8 pills across all cosmetic surgeries without helium plasma technology (P = .8422). There was no statistically significant difference in the mean opioid pills consumed between liposuction procedures with autologous gluteal fat transfer with and without helium plasma technology (Table 3). A total of 2 complications were reported after cosmetic surgeries with helium plasma technology—a wound infection and a seroma, both treated outpatient.
T-Test Comparing Means of Pills Consumed in Patients Undergoing Liposuction With Fat Transfer With and Without Helium Plasma Technology.
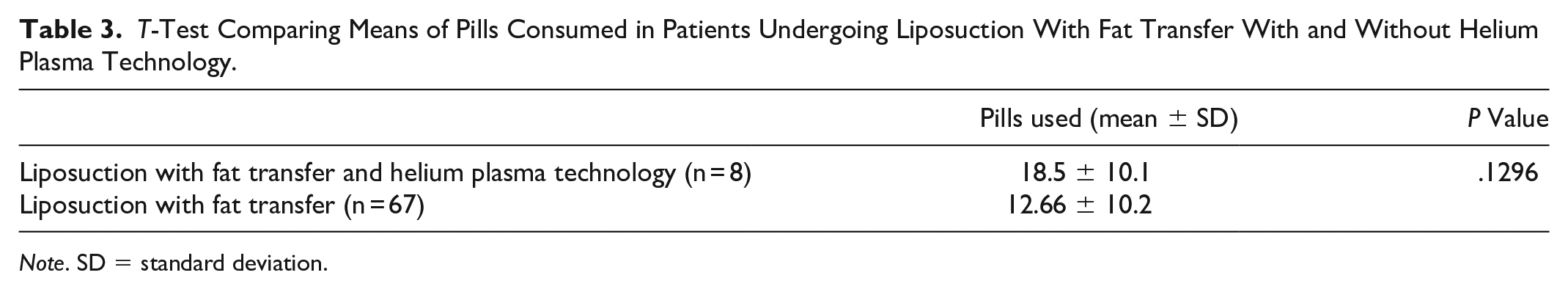
Note. SD = standard deviation.
Discussion
Opioid misuse, addiction, and overdose have been a growing public health concern for the past 30 years and are now amplified amidst the COVID-19 pandemic.7,8 Surgeons must balance patients’ access to opioids postoperatively while optimizing their pain control. Uncontrolled pain after surgery is a well-known risk factor for the development of chronic pain requiring long-term opioid use, 1 whereas overprescription of opioids is associated with chronic use and misuse. It is estimated that 6% of patients develop new persistent opioid use after both minor and major surgical procedures. 9
In our study, prescription practices exceeded patients’ postoperative opioid requirements across all cosmetic surgery categories. We show that patients use, on average, half of the opioids routinely prescribed after an elective cosmetic surgery. A mean of 28 opioid tablets was prescribed across all surgical categories, whereas patients consumed an average of 13.8 pills postoperatively. Our study findings are similar to those of prior studies.4,10,11 A survey of 177 patients after outpatient plastic surgery procedures by Rose et al 4 showed that patients were prescribed on average 26 tablets and 13 tablets were consumed.
Overprescription of opioids has been noted across various surgical specialties including general surgery, orthopedic, gynecologic, urologic, and dental surgery. 6 Common cited reasons for overprescription after breast augmentation are inability to prescribe controlled substances over the phone and concerns for patient refill requests. 12 This preemptive overprescription can lead to a surplus of tablets for patients to later misuse and abuse. A previous study showed that 61.3% of patients reported saving unused narcotics for future use. 13 A total of 2045 tablets in our study were reported unused that were available for diversion and possible misuse.
Identification of risk factors for increased opioid requirement or development of persistent opioid use after surgery would aid surgeons preoperatively recognize patients who may need co-management with a pain specialist or an aggressive nonopioid regiment. In a cohort of 29 068 patients by Brummett et al, 9 tobacco use, alcohol abuse, anxiety, and mood disorders were independent risk factors for persistent opioid use after a surgical procedure in opioid naive patients. 8 Previous studies have identified surgery type and elevated BMI as risk factors for the development of persistent pain after breast cancer surgery requiring long-term pain control. 14 In our study, cosmetic surgery procedure, BMI, ethnicity, and prior surgical history were not associated with an increased opioid consumption in the immediate postoperative period. Patients with any surgical complication on average had a statistically significant higher opioid use than those without a surgical complication. A subset analysis showed that incorporation of helium plasma technology did not increase opioid consumption.
In light of the opioid epidemic and physician overprescription, there has been a paradigm shift toward creating opioid prescription guidelines and promoting the use of opioid alternatives. It is well established that multimodal pain management decrease the need for opioids as the cornerstone of pain control postoperatively with a proven safety profile.15-17 Nguyen et al 18 showed that a nonopioid protocol combining celecoxib and gabapentin controlled postoperative pain as effectively as traditional opioid protocols after outpatient breast surgery. Preoperative patient education also effectively reduces opioid consumption and increases safe and proper opioid disposal.19,20 Eagan et al 21 noted a 33% decrease in opioid consumption in breast surgery patients who received a single-page handout discussing pain expectations and multimodal pain management during their preoperative visit. This handout was formatted to be read in 5 minutes or less and implementation of this brief intervention did not increase the time of the preoperative visit. 21
Most surgeons are in favor of establishing societal evidence-based opioid prescription guidelines. 12 The Centers for Disease Control and Prevention (CDC) recommends limiting opioids to a 3- to 7-day supply for acute pain and advocates for use of the lowest effective dose. 22 The mean opioid amount prescribed in our study was 28 tablets, which is well within this limit. However, despite being in accordance with this guideline, patients on average used only half of the prescription. Opioid-restrictive protocols, such as the Plastic Surgery Initiative to provide Controlled Analgesia and Safe Surgical Outcomes (PICASSO), counteract this preemptive overprescription. 23 This protocol emphasizes multimodal analgesia and argues for an aggressive limit of opioids prescribed. 23 Opioid-restrictive protocols have been successfully implemented without impacting patient satisfaction and refill requests. 24 In a survey by Long et al, 25 less than one-third of patients expected to receive opioids after plastic surgery and half of patients preferred a nonopioid pain regiment, suggesting that surgeons can limit opioid prescription without affecting patient satisfaction.
Still, we recognize that our study is not without its limitations, such as recall bias, which is inherent to survey studies. Opioid consumption was self-reported during the telephone postoperative survey and patients may have incorrectly recalled the amount of opioid pills used and not used. Exact pill counts by an unbiased third party during postoperative visits can more accurately identify opioid consumption. Another limitation of this study is that some patients partially answered the survey, and information for either opioids used or leftover was missing. Patients with preexisting chronic opioid use were not identified and could have been included in this study, resulting in higher reported opioid use. Long-term opioid requirements and development of new persistent pain were not assessed in this study. Further research is needed to evaluate if cosmetic procedure type and presence of any surgical complication predisposes the development of new persistent pain requiring prolonged opioid use. This is a single-institution study and findings may not be generalizable to the general population. Finally, this is a small sample of patients and was likely underpowered to detect a statistical difference between cosmetic surgical procedures.
However, despite its limitations, this study adds to the growing body of pain management literature in the field of cosmetic surgery. This study describes average opioid pills consumed after common elective cosmetic surgeries, which can serve as a reference for physicians and help deter preemptive overprescription. To our knowledge, this is the first study to evaluate opioid consumption after surgeries incorporating the helium plasma technology. Our study included an ethnically diverse cohort of patients that is often underrepresented in the medical literature. In addition, most studies focusing on enhanced recovery pathways with opioid-sparing protocols are within the inpatient hospital setting. This study provides data on opioid consumption after ambulatory cosmetic surgery and underscores physician overprescription in this setting.
Conclusion
Overprescription of opioids after outpatient cosmetic surgeries is common. Patients in our study required about half of the opioids prescribed during the immediate postoperative period. Preemptive overprescription allows for a surplus of opioid pills that can be misused and abused. Safely limiting the amount of opioids prescribed postoperatively is a simple practice change that can help curb the opioid epidemic. Development of evidence-based prescription guidelines after cosmetic surgery is needed to achieve this goal.
Footnotes
Acknowledgements
The authors thank Dr Victoria Karlinsky, MD, FACS, Dr Alexander Zuriarrain, MD, FACS, Dr Benjamin Liliav, MD, FACS, Dr Stephanie Stover, MD, and Dr Gary Vela, MD, FACS for their participation and contribution to this research study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Astrid Marina Zorrilla and Dr Michael Rodriguez received a research grant from Apyx Medical to cover the costs of the IRB fee and publication of the study in an open access journal. The data collected from this study was analyzed by a third party (independent statistician) to prevent any bias. The other authors have no conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Apyx Medical (grant number IIS-2021-01).
