Abstract
Misalignment of behavior and circadian rhythms due to night work can impair sleep and waking function. While both simulated and field-based studies suggest that circadian adaptation to a nocturnal schedule is slow, the rates of adaptation in real-world shift-work conditions are still largely unknown. The aim of this study was to evaluate the extent of adaptation of 24-h rhythms with 6-sulfatoxymelatonin (aMT6s) and cortisol in police officers working rotating shifts, with a special attention to night shifts. A total of 76 police officers (20 women; aged 32 ± 5.4 years, mean ± SD) from the province of Quebec, Canada, participated in a field study during their 28- or 35-day work cycle. Urine samples were collected for ~32 h before a series of day, evening, and night shifts to assess circadian phase. Before day, evening, and night shifts, 60%-89% of officers were adapted to a day schedule based on aMT6 rhythms, and 71%-78% were adapted based on cortisol rhythms. To further quantify the rate of circadian adaptation to night shifts, initial and final phases were determined in a subset of 37 officers with suitable rhythms for both hormones before and after 3-8 consecutive shifts (median = 7). Data were analyzed with circular and linear mixed-effects models. After night shifts, 30% and 24% of officers were adapted to a night-oriented schedule for aMT6s and cortisol, respectively. Significantly larger phase-delay shifts (aMT6s: −7.3 ± 0.9 h; cortisol: −6.3 ± 0.8 h) were observed in police officers who adapted to night shifts than in non-adapted officers (aMT6s: 0.8 ± 0.9 h; cortisol: 0.2 ± 1.1 h). Consistent with prior research, our results from both urinary aMT6s and cortisol midpoints indicate that a large proportion of police officers remained in a state of circadian misalignment following a series of night shifts in dim-light working environments.
Approximately 16%-30% of the workforce in industrialized countries is regularly engaged in some form of shift work, which involves working outside standard daytime hours, including at night (Williams, 2008; Alterman et al., 2013; Australian Bureau of Statistics, 2013; Kubo, 2014; Eurofound, 2017). While this form of work is necessary to meet many of the expectations and requirements of an increasingly globalized society, it can place social and health burdens on the worker. Indeed, combined with its recognized effects on sleep loss and fatigue, growing evidence points to shift work as a health risk (Chellappa et al., 2019; Boivin et al., 2022). Shift work is associated with greater likelihoods of obesity (Sun et al., 2018), diabetes (Gan et al., 2015), cardiovascular disease (Kervezee et al., 2020), and cancer (Ward et al., 2019).
Although many behavioral and physiological factors contribute to these health outcomes in the broader population, their high propensity in shift work suggests disruption of the circadian system is an important contributing factor. Indeed, de-synchronization of behavioral cycles (e.g., sleep-wake) and endogenous circadian rhythms, common in shift work, interferes with healthy metabolic function (Banks et al., 2015; Kervezee et al., 2020; Sletten et al., 2020; Boivin et al., 2022). The circadian pacemaker is primarily entrained to the 24-h day by the light-dark cycle and is responsible for synchronizing the rhythms of physiological processes to fixed phase relationships with external zeitgebers (i.e., “time-givers”). Changing from a day-oriented to a night-oriented schedule when working at night induces a state of circadian misalignment. For most individuals, the circadian pacemaker is not able to quickly adapt to the new behavioral cycle, thus forcing rhythmic physiological functions to operate at suboptimal phases (Boivin and James, 2002a; Boudreau et al., 2013; Archer et al., 2014; Resuehr et al., 2019).
There have been numerous studies of simulated shift work (Crowley et al., 2004; Morris et al., 2016; Bescos et al., 2018; Kervezee et al., 2019a; Kervezee et al., 2019b; Sargent et al., 2022), but it is important to clarify the prevalence of circadian misalignment and the rate of circadian entrainment in real shift workers. The literature indicates that adaptation to a night-oriented schedule is a gradual process requiring extended, regular exposure to the new environmental conditions (Boivin et al., 2012b; Jensen et al., 2016). Indeed, sequences of fewer than three consecutive night shifts are unlikely to produce any signs of adaptation in rhythms driven by the central circadian clock, such as melatonin or cortisol (Grundy et al., 2009; Dumont et al., 2012; Jensen et al., 2016, 2020). Even with longer sequences comprising 4 to 7 consecutive night shifts, changes in the timing of physiological rhythms are usually not large enough to represent a complete realignment or adaptation for most individuals (Boivin and James, 2002a; Hansen et al., 2010; Harris et al., 2010; Boivin et al., 2012b; Ferguson et al., 2012; Stone et al., 2018).
Relatively few studies conducted in the field have assessed the circadian adaptation of night workers across more than 3 consecutive shifts (Boivin and James, 2002a; Boivin et al., 2012b; Ferguson et al., 2012; Jensen et al., 2016, 2020). Of those that have done so, the largest rates of adaptation to night work have consistently been observed with offshore oil rig workers following sequences of 7-14 consecutive shifts (Barnes et al., 1998; Gibbs et al., 2007; Hansen et al., 2010). However, they operate in isolated environments, and their regular schedules of daily activities starkly differ from those of other shift workers, such as police officers, nurses, and doctors who work a similar number of consecutive night shifts but also have to meet daytime social and domestic responsibilities (Boudreau et al., 2013; Stone et al., 2018). As circadian misalignment can affect sleep and alertness and have long-term unfavorable effects on individuals’ health, further studies are still needed to establish the rate of entrainment of regular night workers across longer sequences of shifts in the field, outside of a controlled environment.
Aim
In the present study, our aims were to investigate the extent to which shift-working police officers presented circadian adaptation to various types of shifts under real-world conditions and whether the centrally controlled circadian rhythms, urinary 6-sulfatoxymelatonin (aMT6s) and cortisol, differed in their rate of realignment to a series of nights. We also investigated the environmental lighting conditions and sleep-wake behavior that might contribute to circadian adaptation, as well as the impacts of circadian adaptation on sleep duration, self-reported measures of fatigue, and psychomotor performance at work.
Materials And Methods
Participants
With the support of management and union representatives, police officers aged 20-67 years, who worked full time on rotating shifts in the province of Quebec, Canada, were contacted via e-mail between September 2016 and December 2017. Police officers who expressed interest to participate were required to meet eligibility criteria during a phone-based screening questionnaire. Exclusion criteria included engagement in substantial commitments other than police patrol duties in excess of 10 h/week; medical conditions or prescribed medication that could disturb sleep or waking; and self-reported sleep disorders unrelated to shift work. Eligible participants underwent additional screening, including a physical assessment by a physician and assessment of blood and urine samples. During this process, anthropometric and psychometric data were also collected as previously reported (Kosmadopoulos et al., 2020; Kervezee et al., 2021).
Ethical approval for the study was granted by the Research Ethics Board of the Montreal West Island Integrated University Health and Social Services Centers (#2015-124). The study was conducted in accordance with the Declaration of Helsinki.
Study Procedures
Police officers participated in an observational field study between September 2016 and December 2017 in which they documented their sleep and work behaviors while working their habitual rosters. Participants provided their work schedule to the research team in advance of the study to facilitate planning. All but 4 police officers habitually worked a 35-day cycle with a roster comprising series of 9- and 12-h day shifts (0700-1600 h, 0700-1900 h), evening shifts (1500-2400 h), and night shifts (2230-0730 h, 2300-0800 h, or 1900-0700 h) separated by non-working days (Suppl. Fig. S1A and S1B). The remaining four officers worked a 28-day cycle with a roster comprising 12-h day shifts (0700-1900 h) and 12-h night shifts (1900-0700 h) separated by non-working days (Suppl. Fig. S1C).
Participants were required to collect, log, and freeze urine samples on multiple occasions throughout the protocol. Urine samples were scheduled to be collected for ~32 h prior to a series of day, evening, and night shifts and after a series of night shifts. The sampling window prior to shifts ended after waking from the main sleep period on the day of the shift and began with a void at bedtime of the prior sleep period (Suppl. Fig. S1D). The sampling window following the series of night shifts began with a void at bedtime on the day of the final shift and ended after waking from a main sleep period 2 calendar days later (Suppl. Fig. S1D).
Throughout their work cycles, police officers wore an actigraph (Actiwatch Spectrum; Respironics, Philips, OR) that comprised an accelerometer, light sensor, and event marker on the wrist of their non-dominant hand to monitor their sleep-wake and physical activity patterns (15-s epoch). They were asked to press the event marker at each bedtime and rise time. The device was required to be worn at all times except when showering, swimming, or engaging in contact sports to avoid equipment damage. Participants were provided a smartphone (Nexus 5; LG Electronics, Seoul, Korea) and instructed to log their bedtimes, rise times, and work times (i.e., work start, work end) using a smartphone application (movisensXS; movisens GmbH, Karlsruhe, Germany). In addition, ecological momentary assessment was used to record levels of mood, alertness, and fatigue measures at various time points throughout the day. Participants also performed several psychomotor performance tests with respect to each shift.
Participants were contacted weekly throughout the study to limit data loss and could contact a staff member at any time for study assistance. At the conclusion of the study, police officers returned their equipment with a copy of their final work schedules so the research team could verify their actual work hours. Self-reported work hours were compared against work schedules and the timestamps of questionnaires completed at the start and end of the shifts. Any conflicting work-start and work-end times were corrected via cross-verification.
Sleep-Wake Assessments, Light Exposure, and Physical Activity
As previously described by Kervezee et al. (2021), a standardized hierarchical algorithm adapted from the procedure described by Patel et al. (2015) was used to identify participants’ rest intervals (i.e., the period from bedtime to rise time) from sleep log information and timestamps, together with physical activity counts, event markers, and light exposure data retrieved from the actigraph in 15-s epochs. Data from the actigraphs were aggregated into 2-h bins by time of day per subject to assess sleep propensity, light exposure levels, and activity levels during the series of night shifts and in the 72-h periods before and after. To understand how sleep was affected across the series of shifts, the cumulative duration of sleep obtained from noon-to-noon each day was also calculated.
Measurement of Urinary aMT6s and Cortisol
Concentrations of aMT6s in urine samples were determined in duplicate via enzyme-linked immunosorbent assay (ALPCO, Salem, NH). The average intra-assay and inter-assay coefficients of variation (CV) were 7.0% and 8.0%, respectively. Concentrations of urinary free cortisol were determined via radioimmunoassay (MP Biomedicals, Orangeburg, NY), and the inter-assay CV was 14.1%.
Circadian Rhythms and Adaptation Criteria
Once assayed, excretion rates of aMT6s and urinary cortisol concentrations per hour were derived at the mid-time between sample collections. Acrophases for each hormone were then calculated as the midpoint between the upward and downward crossing of the average excretion rate per hour (Boivin and James, 2002a; Boivin et al., 2012a). This method of analyzing the rhythms of individual workers was chosen due to irregular timing and frequency of samples in the field. The upward and downward crossings were determined with the consensus of two independent scorers (aMT6s: A.K. and L.K.; cortisol: A.K. and P.B.) who reviewed all the excretion profiles for each sampling period, blinded with respect to participant, time of day, and associated shift. Excretion profiles for which a midpoint could not be determined due to an ambiguous bimodal or pulsating rhythm were excluded, as were profiles for which the sampling period was <20 h.
A circadian rhythm was considered adapted to a given shift type if it demonstrated sufficient alignment with habitual sleep patterns in the corresponding sleep-wake schedule. For the purpose of evaluating circadian alignment, habitual sleep onset and waketimes on day-oriented schedules were calculated from officers’ main sleep episodes on non-working days that were neither preceded nor followed by a workday. For night-oriented schedules, habitual sleep times were calculated from officers’ main sleep episodes between consecutive night shifts.
Because melatonin secretion usually peaks during night-time sleep episodes, the interval between habitual sleep onset and waketime was used as the basis for aMT6s adaptation during day- or night-oriented schedules (Nowak et al., 1987; Shanahan and Czeisler, 1991; Benloucif et al., 2008). Thus, rhythms of aMT6s collected prior to day, evening, and night shifts were considered to be sufficiently aligned to a day-oriented schedule if acrophases occurred within participants’ average sleep times on non-working days. Similarly, rhythms of aMT6s after night shifts were considered to be sufficiently aligned to a night-oriented schedule if the acrophases occurred within participants’ average daytime sleep episodes between consecutive night shifts.
It was more difficult to establish criteria for the adaptation of the cortisol rhythm because the excretion of urinary cortisol typically increases throughout the nocturnal sleep period, peaking shortly after the habitual morning awakening (Gunn et al., 2016). As such, we used an iterative process to algorithmically identify an appropriate alignment window during which acrophases would be expected to occur to be considered adapted to a given sleep-wake schedule. Hourly offset increments (0-8 h) relative to habitual waketimes on non-working days and various window durations (4-8 h) were used to find the best fit. This iterative process was applied to days when participants were on a day-oriented schedule (i.e., days prior to day, evening, and night shifts) (See Suppl. Fig. S2). In selecting the optimal alignment window, the algorithm tried to achieve the best compromise between (1) the shorter window duration and (2) the higher percentage of captured acrophases. An increase in the window duration was considered only if it increased the percentage of acrophases captured by that window by ≥5%. After establishing the best parameter for detecting circadian adaptation on a day-oriented schedule, we transposed these criteria to night-oriented schedules.
Based on these criteria, the optimal window for establishing alignment was the 7-h window beginning 1 h prior to officers’ habitual waketime (74% of 153 cortisol acrophases prior to day, evening, night shifts; see Suppl. Table S1). Thus, acrophases were considered aligned to a day-oriented schedule if they occurred during these times of day. Conversely, acrophases were considered adapted to night shifts (aligned to a night-oriented schedule) if they occurred in the 7-h window starting 1 h prior to the average waketime of the main sleep episode between night shifts (Suppl. Figs. S1D and S2). While these criteria appear generous, they are reasonable considering the low and irregular sampling frequency in the field.
Self-report Questionnaires and Psychometric Performance
Timestamped self-reported and objective measures of neurobehavioral functioning associated with night work were obtained with the smartphone provided to each participant. For neurobehavioral functioning, officers were asked to perform a 5-min psychomotor vigilance test (PVT) at the start and end of each shift (Dorrian et al., 2005; Roach et al., 2006; Boivin et al., 2023). Measures extracted from the PVT were the median reaction time (msec) and the lapse count (reaction times > 500 ms). For self-reported measures, officers were asked to complete questionnaires at the start, middle, and end of the shifts. These questionnaires included visual analogue scales of alertness (0 = “low” to 100 = “high”) and mood (0 = “bad” to 100 = “good”), the Samn-Perelli fatigue checklist (7-point Likert-type scale, 1 = “fully alert” to 7 = “completely exhausted”) (Samn and Perelli, 1982), and the Karolinska Sleepiness Scale (9-point Likert-type scale, from 1 = “extremely alert” to 9 = “very sleepy”) (Åkerstedt and Gillberg, 1990). To facilitate the comparability of the self-reported ratings and PVT performance measures within and between participants across consecutive night shifts, data associated with atypical shifts, for example, night shifts that did not encompass the period between 0100 h and 0500 h, were shorter than 4.5 h (i.e., less than half a 9-h shift), or were longer than 13.5 h (i.e., overtime of more than half a 9-h shift), were excluded from analyses. Furthermore, performance and self-reported measures purported to have been collected at the start or end of shifts were excluded if timestamps were not within an hour of the verified shift times.
Statistical Analyses
Data analyses were performed with R Foundation for Statistical Computing, version 3.6.3 (R Core Team, 2020) using multiple statistical packages detailed in the following sections. Figures were created with the packages “ggplot2” and “cowplot” (Wickham, 2016; Wilke, 2019). Welch’s t-tests were used to compare the anthropometric and psychometric characteristics of participants who adapted to night work based on aMT6s phase markers with those of participants who did not.
Participants were divided into adaptation groups (“adapted” vs “non-adapted”) for each hormone based on the alignment of these rhythms to the type of schedule (day- or night-oriented). The statistical package “circular” (Agostinelli and Lund, 2017) was used for circular data (e.g., clock times) to describe the timing of adapted and non-adapted phase markers and the duration of phase shifts for all valid profiles, as well as for a subset of officers with a complete set of hormone data. Circular analyses of variance (ANOVA) were conducted for between-group differences (“adapted” vs “non-adapted”) for each hormone. Bayesian circular mixed-effects regression models (R package “bpnreg”; Cremers, 2020) were used to assess within-subject changes in acrophases before and after a series of night shifts.
All remaining analyses were conducted with linear mixed-effects models using the R packages “lme4” and “lmerTest” (Bates et al., 2015; Kuznetsova et al., 2017). Models included participant as a random effect and the fixed effects and interactions of the factors “adaptation group” (i.e., adapted vs non-adapted to night shifts), “time of day” (2-h bins), and “day” (i.e., pre- and post-night-shift days, or shift days 1-7) to assess changes in sleep, performance, light exposure, and physical activity. The adapted or non-adapted group effect on circadian parameters on hormonal rhythms was tested by adapting the cosinor.lm function from the “cosinor” package (Tong, 1976; Sachs, 2014) to linear mixed-effects models and setting the period to 24 h. Tukey’s post hoc pairwise comparisons were made using the package “emmeans” (Lenth, 2019). Data for urinary aMT6s and cortisol rhythms, light exposure, and activity levels underwent log-transformation prior to analyses but are presented in figures using raw values. For all analyses, significance was determined at p < 0.05.
Results
Participants
A total of 76 of the 81 police officers eligible to be included in the study were studied throughout a complete work cycle (Table 1). This sample was largely male (74%) and had a mean (SD) age of 32.0 (5.4) years and a body mass index of 24.8 (2.5) kg/m2. The other 5 officers had to be excluded due to dropout (n = 2), technical issues with equipment (n = 1), or withdrawal for medical reasons (n = 2) (Suppl. Fig. S3).
Characteristics of participants who completed a work cycle (n = 76) and the subset with valid rhythms for both aMT6s and cortisol before and after night shifts.
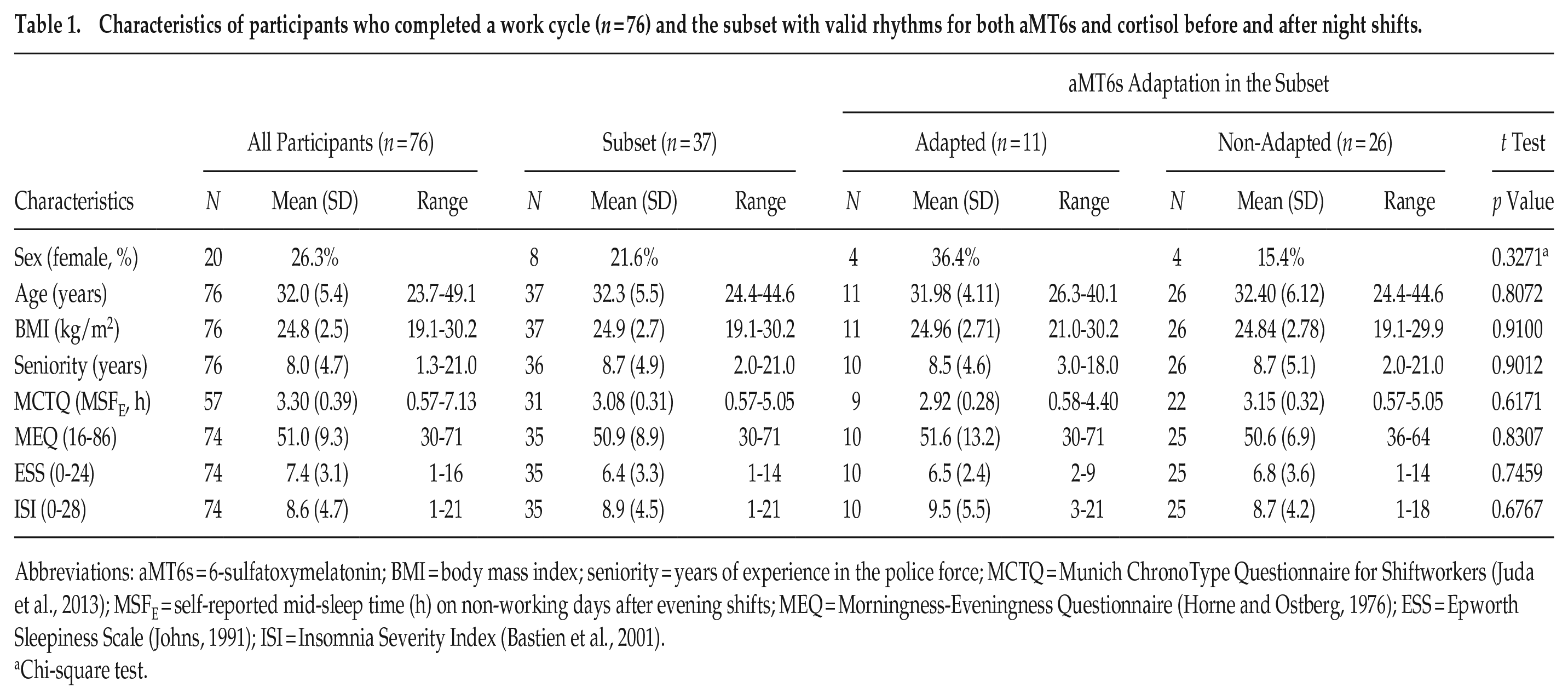
Abbreviations: aMT6s = 6-sulfatoxymelatonin; BMI = body mass index; seniority = years of experience in the police force; MCTQ = Munich ChronoType Questionnaire for Shiftworkers (Juda et al., 2013); MSFE = self-reported mid-sleep time (h) on non-working days after evening shifts; MEQ = Morningness-Eveningness Questionnaire (Horne and Ostberg, 1976); ESS = Epworth Sleepiness Scale (Johns, 1991); ISI = Insomnia Severity Index (Bastien et al., 2001).
Chi-square test.
Data Retention
Hormone rhythms were assessed for 75 officers who collected urine samples for ≥20 h before or after at least one shift series (Suppl. Fig. S3A). One officer collected urine samples for less than 20 h during all collection windows, so these were discarded. The median duration of the remaining sample collection windows was 31.1 h (interquartile range [IQR] = 5.9 h) from initial void before bedtime until the final awakening sample. The median duration between successive samples within each window was 3.5 h (IQR = 1.9 h), and the median number of samples for each window was 7 (IQR = 3). Refer to Figure 1A and 1B for a representative depiction.
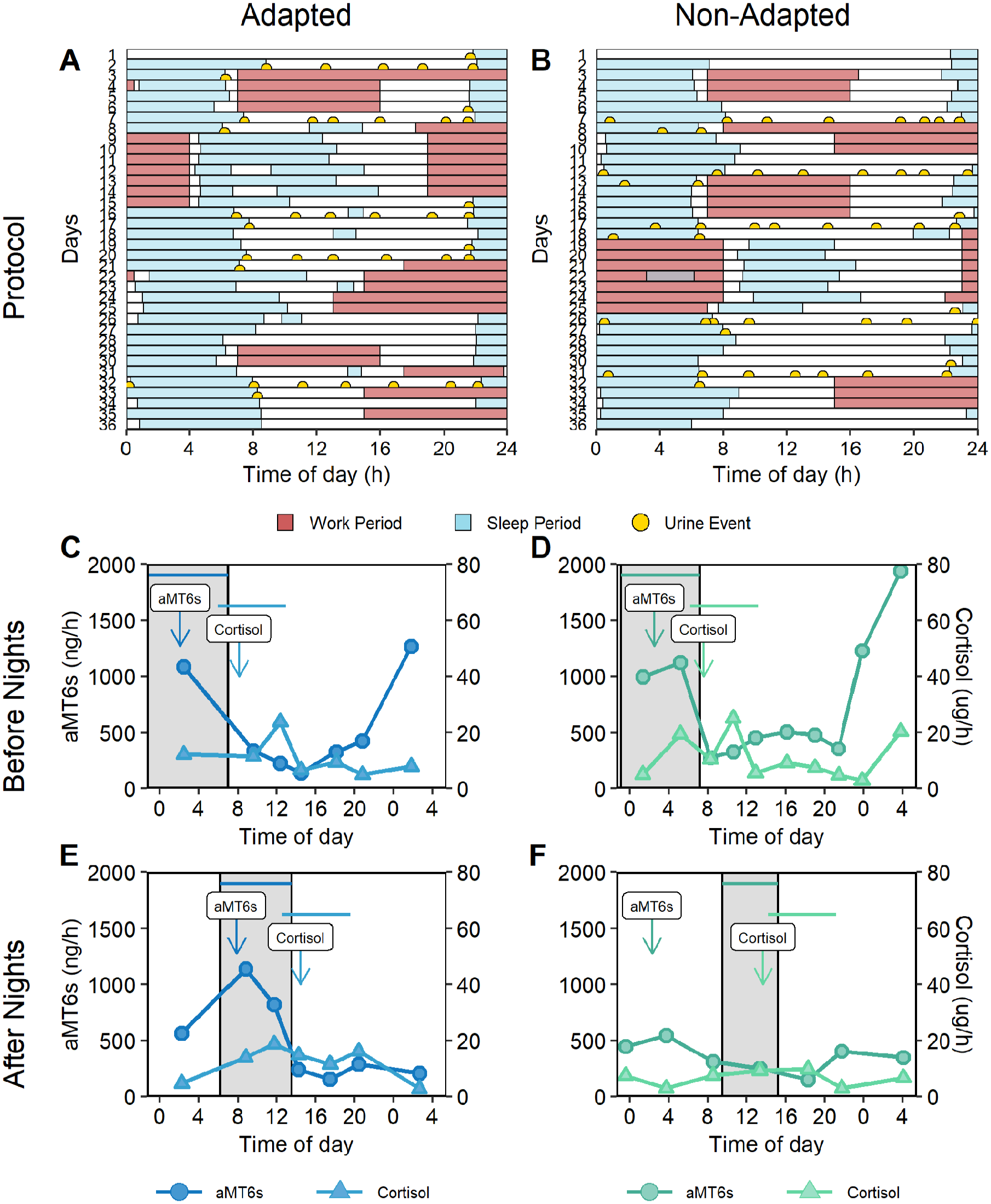
Representative depictions of sleep, work, and urine sampling schedules during the study protocol (A, B) and excretion profiles of urinary 6-sulfatoxymeltatonin (aMT6s) and cortisol before (C, D) and after (E, F) a series of night shifts (C-F). The left panels (A, C, E) represent an individual whose hormone rhythms were aligned to a night-oriented schedule following the series of night shifts, and the right panels (B, D, F) represent an individual whose rhythms did not. In the protocol diagrams (A and B), the x-axis represents time of day, and the y-axis represents consecutive days in the work cycle. Urine samples are depicted with semi-circles. Pale blue rectangles represent sleep periods, and darker red rectangles represent shifts at work. For the hormone profiles (C-F), the x-axis indicates time of day, and the y-axis indicates the excretion rate per hour. The gray rectangles illustrate the participants’ habitual main sleep period on non-working days (C, D) and between consecutive night shifts (E, F). Each data point illustrates the excretion rate at the mid-time between consecutive urine samples; circles identify aMT6s rhythms, and triangles identify cortisol rhythms. The downward arrows indicate the acrophases of the aMT6s and cortisol rhythms, before and after night shifts, for the adapted (C, E) and non-adapted (D, F) participants. Horizontal lines near the top of the panels (C-F) illustrate the time ranges during which acrophases (aMT6s and cortisol) needed to occur to be considered adapted based on the sleep behavior before or during night shifts.
Initial agreement between scorers for the hormone profiles was 89% (aMT6s) and 74.5% (cortisol), and discrepancies were resolved via discussion. Profiles that were still deemed ambiguous by both scorers were excluded (aMT6s: n = 6, 3%; cortisol: n = 87, 24.7%). Unambiguous phase markers were obtained for 94%-100% of the aMT6s rhythms and 72%-85% of the cortisol rhythms that were assayed for the various series of shifts (Suppl. Fig. S3C). These were used to ascertain the proportion of rhythms properly aligned to the shift type.
Adaptation to night work was further analyzed in a subset of 37 officers with circadian phase assessments available for both aMT6s and cortisol before and after the series of night shifts (Table 1). This allowed us to compare adaptation of these circadian rhythms in the same individuals and to quantify phase shifts that occurred during the series of night shifts. These 37 police officers all worked the 35-day work cycles comprising a series of 7 consecutive night shifts (Suppl. Fig. S1A and S1B). The median number of night shifts completed by these officers was 7 (IQR = 1; range = 3-8), with 92% working at least 6 night shifts in this sequence (Suppl. Fig. S4D). During their participation in the study, officers had a median of 15 non-working days (IQR = 4; range = 8-20). Figure 1 illustrates representative examples of the timing of sleep, work schedules, and urine collections of a participant who did not adapt to his series of night shifts and one participant who did.
Adaptation of aMT6s and Cortisol Rhythms According to Shift Type
The alignment of aMT6s and cortisol rhythms to a type of day was only assessed for unambiguous phase estimates and for officers with satisfactory actigraphic sleep data on non-working days or between consecutive shifts. For the assessment of aMT6s prior to day (n = 15), evening (n = 18), and night shifts (n = 65), 60%-89% of officers were considered aligned to a day-oriented schedule based on the timing of aMT6s acrophase between officers’ habitual night-time sleep onsets (n = 73; circular mean ± SD = 2345 h ± 0017 h) and waketimes (n = 73; circular mean ± SD = 0748 h ± 0016 h) (Figure 2A). It was found that 16 (24%) of all officers (n = 68) were substantially aligned to a night-oriented schedule following the night shifts, with aMT6s phase markers occurring between their habitual daytime sleep onsets (n = 71; circular mean ± SD = 0910 ± 0017 h) and waketimes (n = 71; circular mean ± SD = 1525 h ± 0023 h).
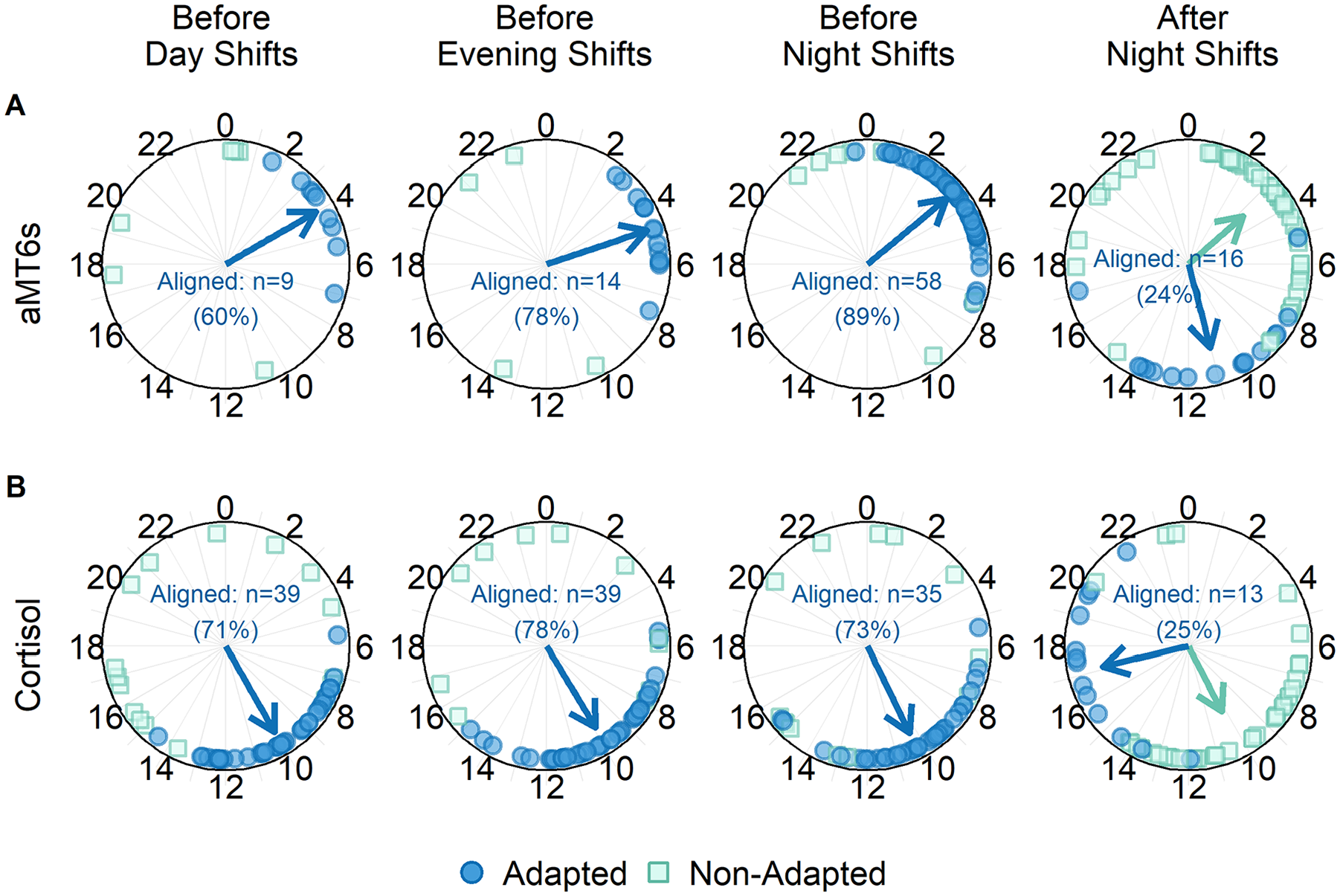
Phase markers of 6-sulfatoxymelatonin (aMT6s) and cortisol rhythms before and after a series of shifts. Phase markers are depicted by time of day (hours) and grouped based on alignment to a day-oriented schedule (prior to day, evening, and night shifts) or a night-oriented schedule (following night shifts). Circles represent rhythms that are adapted, and squares represent those that are not adapted with the sleep-wake schedule. Arrows indicate the circular mean time of phase markers for aligned or misaligned rhythms. No arrows are shown for the misaligned rhythms prior to day, evening, and night shifts because the phase markers were not non-uniformly distributed (all p > 0.05, Rayleigh test). Percentages indicate the proportion of officers for whom urinary aMT6s (A) or cortisol (B) phase markers were aligned. Day-oriented alignment before day, evening, and night shifts could not be determined for the phase markers of 2 participants who had inadequate or faulty sleep data on non-working days.
Of the officers with valid cortisol rhythms prior to day (n = 55), evening (n = 50), and night (n = 48) shifts, 71%-78% were deemed to be aligned to a day-oriented schedule and had their acrophase occurring within a 7-h window beginning 1 h prior to habitual waketime on non-working days (Figure 2B). After the consecutive night shifts, 25% of cortisol rhythms (n = 52) were found to be aligned to a night-oriented schedule and had their acrophase occurring within a 7-h window beginning 1 h prior to habitual waketime on days between night shifts.
Subgroup Analyses on the Effects of Night-Shift Work
To clarify the relative adaptation of each hormone rhythm and quantify phase shifts that occurred during the series of consecutive night shifts, we restricted all subsequent analyses to the subset of 37 officers with unambiguous phase markers of both hormones before and after the series of night shifts. The habitual sleep schedules of this subset were comparable to the entire group both for day-oriented non-working days (n = 36; sleep onset: 2348 h ± 0018 h, waketime: 0744 h ± 0016 h) and for days between night shifts (n = 37; sleep onset: 0926 ± 0017 h, waketime: 1543 h ± 0020 h). Adequate sleep data was missing for one officer in the subset on non-working days, so it was not possible to ascertain the day-oriented alignment prior to night shifts for that individual.
Adaptation of Circadian Markers
Before night shifts, the circadian rhythms of aMT6s and cortisol were aligned to a day-oriented schedule for 33 (92%) and 28 (78%) officers (n = 36), respectively. After the series of night shifts, the circadian rhythms of aMT6s and cortisol were aligned to a night-oriented schedule for 11 (30%) and 9 (24%) officers (n = 37), respectively. Following the night shifts, 14% of the officers were aligned to a night-oriented schedule for both their aMT6s and cortisol rhythms. The group of officers whose aMT6s rhythms adapted to a night-oriented schedule did not differ from the non-adapted group with regard to any demographic characteristics (Table 1).
Circadian Phase Shifts
Circular ANOVA demonstrated that, prior to night shifts, aMT6s acrophases did not differ between the adapted (n = 11; circular mean ± SD; 3.8 ± 0.6 h) and non-adapted (n = 26; 3.1 ± 0.5 h) groups (Figure 3A). Circular mixed-effects models showed that the acrophases of the adapted aMT6s group after night shifts (11.4 ± 0.7 h) were significantly delayed compared to those before night shifts (difference = −7.6 h, 95% CI: −9.8 to −5.6 h). In contrast, the acrophases of the non-adapted aMT6s group after night shifts (2.1 ± 0.8 h) did not differ from those before night shifts (difference = 0.8 h, 95% CI: −0.5 to 2.0 h) (Figure 3A). For between-group comparison, circular ANOVA showed that the phase shift for officers whose aMT6s rhythms adapted (−7.3 ± 0.9 h) was significantly different from that for those whose aMT6s did not adapt (0.8 ± 0.9 h; p < 0.0001) (Figure 3B).
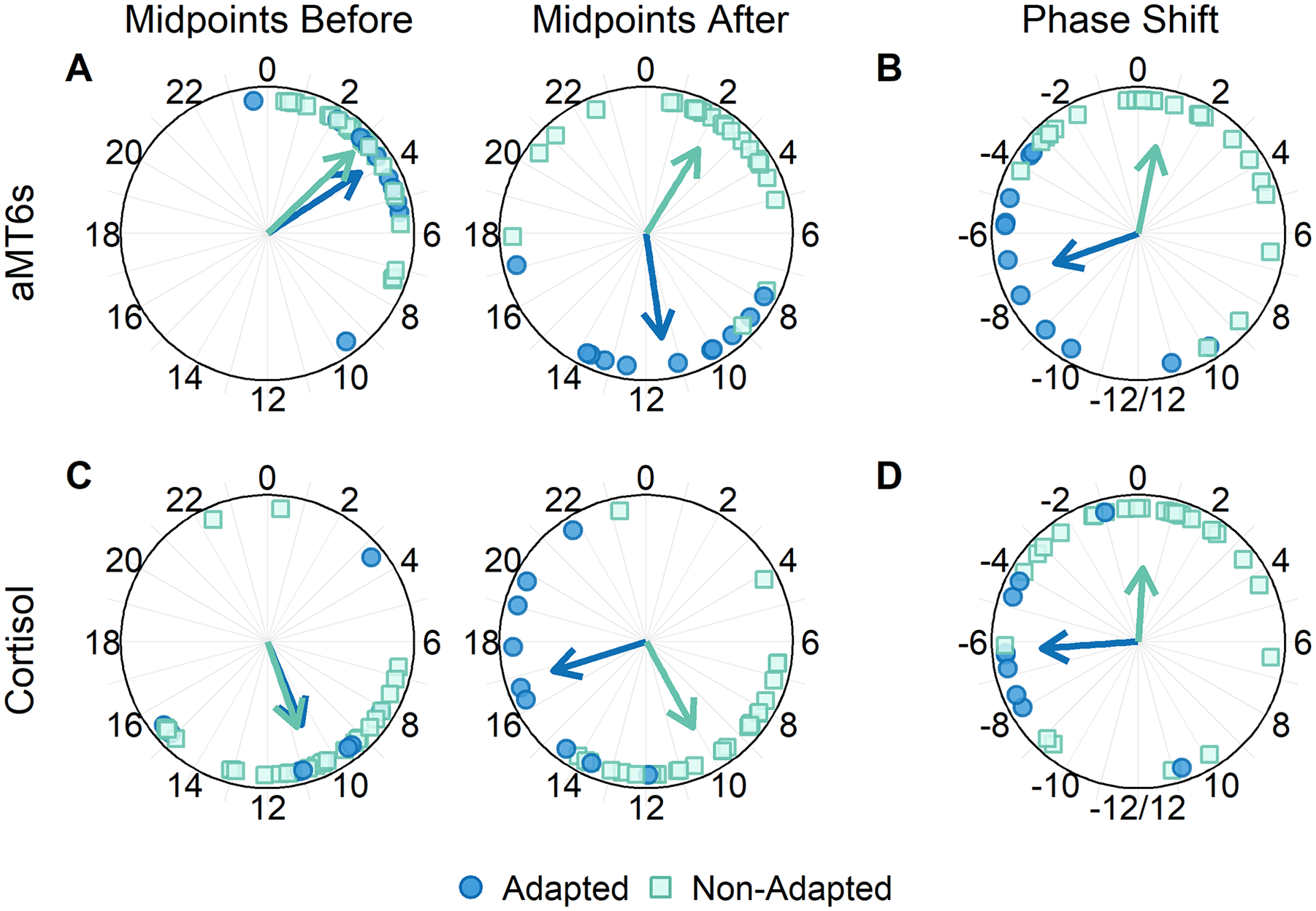
Phase markers and phase shifts of officers with both unambiguous 6-sulfatoxymelatonin (aMT6s) and cortisol rhythms before and after a series of night shifts (n = 37), grouped by adaptation to a night-oriented schedule following a series of night shifts. Acrophases of urinary aMT6s (A) and cortisol (C) rhythms are depicted as a function of time of day in hours. Phase shifts for aMT6s (B) and cortisol (D) are depicted such that negative values represent delays and positive values represent advances in the rhythms following night shifts. Circles represent the acrophase or phase shifts of officers whose rhythms adapted to a night-oriented schedule (aMT6s: n = 11, 30%; cortisol: n = 9, 24%), and squares represent those of officers whose rhythms did not adapt (aMT6s: n = 26, 70%; cortisol: n = 28, 76%). Arrows indicate the average acrophase time or phase shift for each group of officers.
Circular ANOVA showed that, prior to night shifts, acrophases of the cortisol adapted group (n = 9; 10.5 ± 0.9 h) did not differ from those of the non-adapted group (n = 28; 10.7 ± 0.9 h; p = 0.876) (Figure 3C). Circular mixed-effects models showed that acrophases of the adapted group after night shifts (16.8 ± 0.8 h) were significantly later than those before night shifts (difference = −6.3 h, 95% CI: −9.6 to −3.1 h). In contrast, acrophases of the non-adapted cortisol group after night shifts (10.1 ± 0.8 h) did not differ from those before shifts (difference = 0.5 h, 95% CI: −1.1 to 2.0 h) (Figure 3C). Phase shifts of urinary cortisol were significantly different for officers whose rhythms adapted (−6.3 ± 0.8 h) compared with those for officers whose rhythms did not adapt (0.2 ± 1.1 h; p < 0.0001) (Figure 3D).
Additional Exploration of Circadian Rhythmicity
After establishing the average acrophases and phase shifts for individual officers, additional analyses were conducted on group data using linear mixed-effects models. These analyses served to confirm our prior analyses and to investigate whether circadian adaptation to night work affected other rhythmic parameters (i.e., mesor, amplitude).
When compared to a null model, inclusion of a “time of day” factor with one harmonic revealed significant rhythmic variations of aMT6s for officers in the adapted group (χ2[4] = 11.4, p = 0.022) and non-adapted group (χ2[4] = 157.8, p < 0.001) (Figure 4A). Inclusion of “day” (before vs after nights) × “time of day” interaction term only improved the model for the adapted group (χ2[2] = 34.6, p < 0.001), not for the non-adapted group (χ2[2] = 1.6, p = 0.451). Cosinor-fitted regressions demonstrated that the group mesors of the aMT6s rhythms before and after night shifts did not differ for either the adapted officers (583 ± 51 ng/h vs 592 ± 53 ng/h; p = 0.873) or the non-adapted officers (774 ± 74 ng/h vs 771 ± 67 ng/h; p = 0.970). Similarly, there were no significant changes in amplitude for the adapted (302 ± 69 ng/h vs 302 ± 75 ng/h; p = 0.996) and non-adapted (566 ± 115 ng/h vs 490 ± 104 ng/h; p = 0.419) groups before and after night shifts. These rhythmic analyses confirmed a significant phase delay for officers whose aMT6s rhythms adapted following night shifts (phase shift: −7.1 h, 95% CI: −4.6 to −9.6 h) but not for officers who did not adapt (phase shift: 1.0 h, 95% CI: −0.4 to 2.5 h).
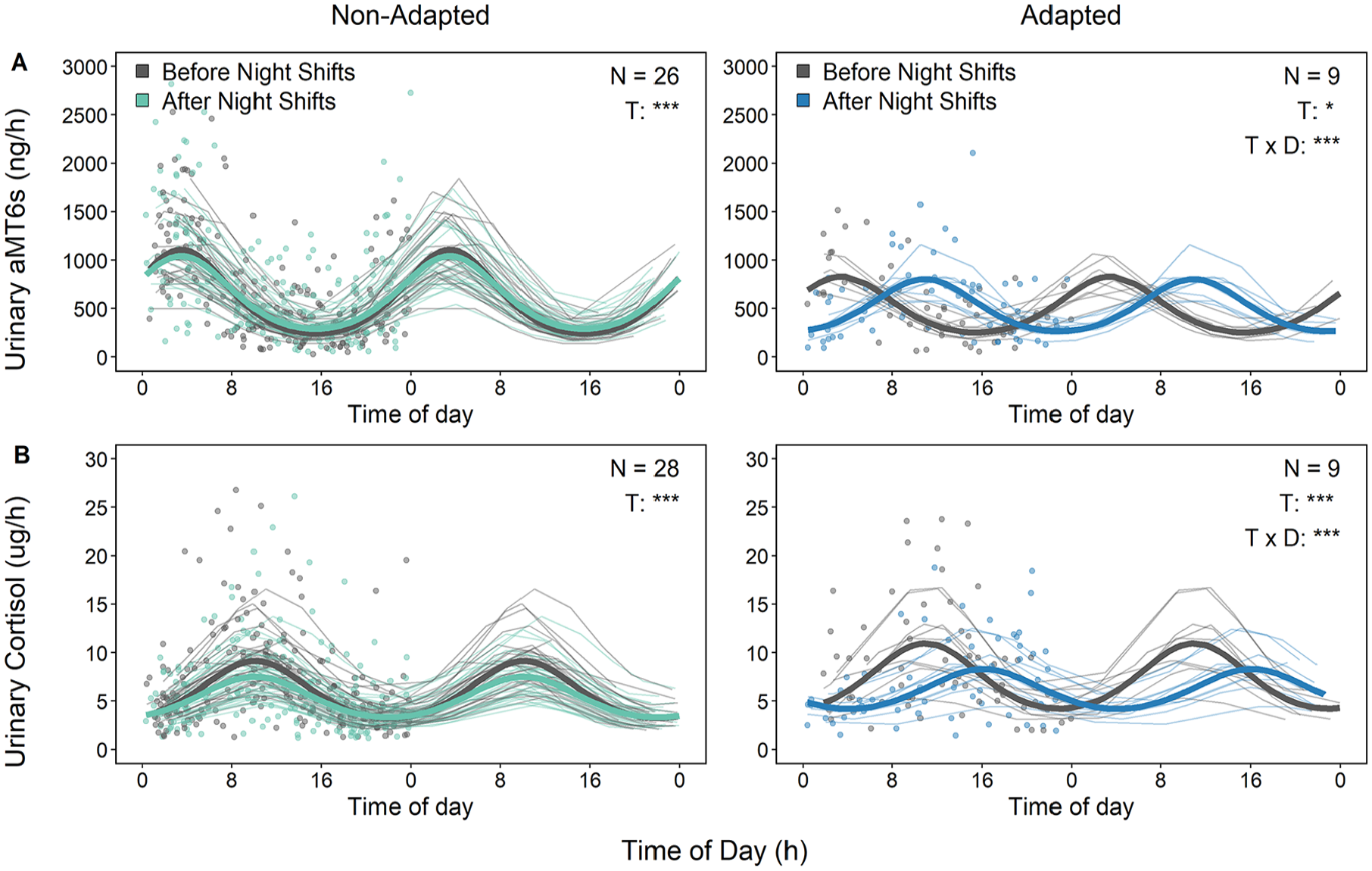
Profiles of urinary (A) 6-sulfatoxymelatonin (aMT6s) and (B) cortisol excreted before and after night shifts, grouped by adaptation to night shifts (double-plotted). The aMT6s data are from 35 participants (26 non-adapted, 9 adapted). Two of 11 participants with adapted aMT6s rhythms were excluded due to extreme outliers for some samples that affected the group mesor and amplitude. Cortisol data are from 37 participants (9 adapted, 28 non-adapted). Data points represent the average excretion rate per hour at the mid-time between samples. The thick lines depict the model-fitted rhythms of each group on the day before and day after night shifts. The thin lines depict the model-fitted rhythm for each participant. Significant effects of time (T), day (D), and their interaction (T × D) are depicted in each panel.
When compared to a null model, linear mixed-effects regressions revealed a significant effect of time of day on the rhythmicity of cortisol regardless of whether adaptation was observed (χ2[4] = 24.0, p < 0.0001) or not (χ2[4] = 123.0, p < 0.0001) following night shifts (Figure 4B). Inclusion of the group factor in the model indicated no overall effect on cortisol levels before (χ2[1] = 3.6, p = 0.058) or after the series of night shifts (χ2[1] = 1.2, p = 0.27) for officers whose cortisol rhythms adapted and for those whose rhythms did not adapt to a night-oriented schedule. The inclusion of neither “day” nor “day” × “time of day” interaction terms improved the model for the non-adapted cortisol group (χ2[2] = 2.12, p = 0.347), but the interaction was present for the adapted group (χ2[2] = 16.4, p < 0.001). Cosinor-fitted regressions demonstrated that the group mesors of the urinary cortisol rhythms before and after night shifts did not differ for either the adapted officers (8.33 ± 0.91 μg/h vs 7.13 ± 0.75 μg/h; p = 0.873) or the non-adapted officers (6.87 ± 0.51 μg/h vs 6.39 ± 67 μg/h; p = 0.241). Similarly, there were no significant changes in amplitude for the adapted (4.01 ± 0.96 μg/h vs 2.43 ± 0.79 μg/h; p = 0.151) and non-adapted (3.47 ± 0.48 μg/h vs 2.47 ± 0.50 μg/h; p = 0.091) groups. These analyses confirmed there was a significant overall phase delay in cortisol rhythms for the adapted group (phase shift: −5.37 h, 95% CI: −1.97 to −8.76 h) but not for the non-adapted group (phase shift: −1.00 h, 95% CI: −2.53 to 0.52 h).
Sleep, Activity, and Light Exposure Behavior
During the series of night shifts, there were significant effects of time of day in the distribution of sleep propensity, activity levels, and light exposure (all p < 0.001) (Figure 5). During the series of night shifts, the greatest propensity for sleep occurred between 0800 h and 1600 h, coincidental with the lowest activity levels. In comparison, prior to night shifts, sleep propensity was greatest at night. In the 72 h prior to night shifts, group differences in sleep propensity were observed, and police officers whose aMT6s rhythms adapted spent a greater percentage of this period asleep than those in the non-adapted group (estimated marginal mean: 34% vs 29%; p = 0.010). There were no other significant main effects of adaptation group before night shifts. Similarly, there were no group differences for sleep propensity, light exposure, or activity during and after night-shift days.
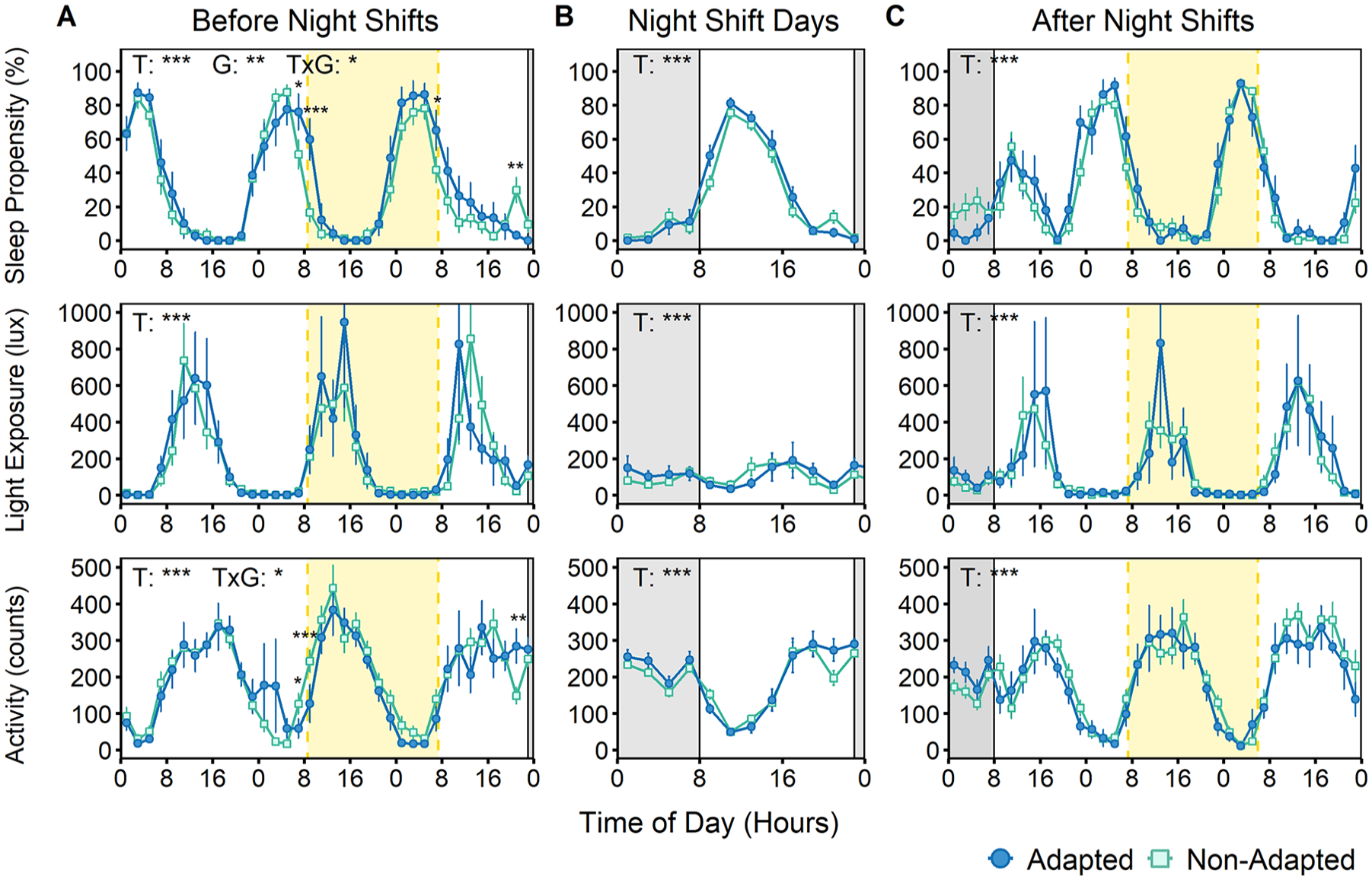
Time of day distribution of sleep propensity, light exposure, and activity levels before, during, and after night shifts for police officers with urinary aMT6s rhythm adapted (n = 11) or not adapted (n = 26) to a night-oriented schedule. (A) The left panel depicts data obtained during a 72-h period before midnight on the day of the first night shift. (B) The center panel depicts data obtained between the start of the first night shift until the end of the final night shift, folded by time of day. (C) The right panel depicts data obtained during a 72-h period following midnight on the day of the final night shift. Dark gray rectangles in all panels represent a theoretical night shift from 2300 h to 0800 h. The pale yellow rectangles bordered by vertical dashed lines (panels A and C) indicate the average urine collection windows before and after night shifts. Before, during, and after the series of night shifts, there were significant effects of time of day for sleep propensity (all p < 0.001), light exposure (all p < 0.001), and activity levels (all p < 0.001). Prior to night shifts, police officers who adapted to night shifts had a higher sleep propensity than those who did not (p = 0.010). There were no other significant effects of group. Interactions of time of day and adaptation group were found for sleep propensity and activity prior to night shifts. Significant main effects of time (T) and group (G), interactions (T x G), and pairwise comparisons are depicted in each panel with asterisks.
Behavioral Impact of Circadian Alignment
When evaluating the total sleep duration obtained noon-to-noon across consecutive night-shift days, linear mixed-effects models revealed a significant main effect of day number (p < 0.001). Post hoc tests revealed that sleep was shortest in the 24 h comprising the first night shift and then stabilized (Figure 6A). There was a significant group effect (p = 0.02) with more sleep on average each day in the group with adapted aMT6s rhythms. Analysis of total sleep cumulated over successive night shifts revealed a significant interaction between shift day and adaptation group (p = 0.026). This indicated that participants with aMT6s rhythms that adapted to night work obtained cumulatively more sleep per day throughout the series of shifts than those with rhythms that did not adapt (average 6.4 h per day vs 5.8 h per day). It took until the fourth day for officers who adapted to night shifts to obtain significantly more sleep than officers who did not (Figure 6B).
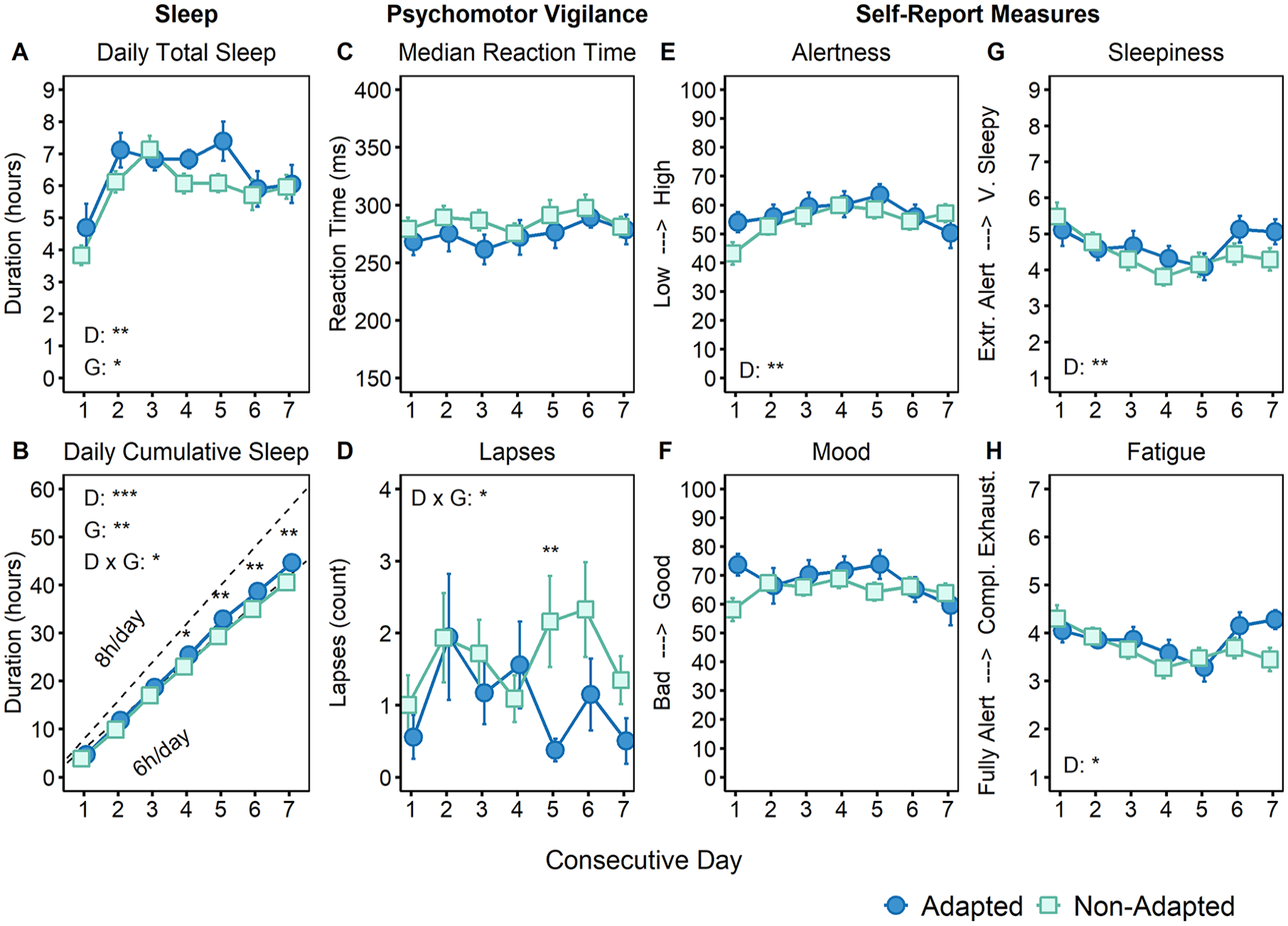
Impact of circadian adaptation to night work on sleep duration, self-reported measures, and psychomotor performance. Total daily (A) and cumulative sleep (B) obtained during consecutive noon-to-noon periods (referred to as “days”) throughout the week. The dotted line (B) represents the cumulative sleep of a participant who would have slept 8 h per day. Psychomotor vigilance performance as median reaction time (ms; C) and lapse count (reaction times > 500 ms; D). Visual analogue scales of alertness (low to high; E) and mood (bad to good; F). Ratings of sleepiness (extremely alert to very sleepy; G) and fatigue (fully alert to completely exhausted; H). Data for each consecutive shift are grouped based on adaptation of 6-sulfatoxymelatonin (aMT6s) rhythms to a night-oriented schedule following a series of night shifts. Circles represent the group of officers who adapted to night shifts, and squares represent the group that did not. Values are expressed as mean ± SEM. Significant effects of day (D), group (G), and interaction (D × G) are depicted in each panel.
Self-report Ratings and Psychomotor Performance Across Consecutive Night Shifts
When self-report ratings for each participant were averaged per shift, linear mixed-effects models indicated that there were significant main effects of shift day for alertness (p = 0.005), fatigue (p = 0.013), and sleepiness (p = 0.002) (Figure 6E, G, and H). These all showed a tendency to improve across the first five night shifts before plateauing or worsening for the final two. It should be noted that 48% of the officers contributing self-report data worked 12 h or longer for at least one of the final two shifts, which may explain the U-shaped curve for these self-report measures. For the psychomotor vigilance task, there was an interaction between shift day and adaptation group for lapse count (p = 0.031; Figure 6D). This interaction suggested a reduction in the average number of lapses in the adapted group beginning on the fifth night shift. On the final night shift, alertness significantly declined (p = 0.048), whereas fatigue (p = 0.033) and sleepiness (p = 0.046) increased with time on shift. However, there were no main effects in the aMT6s adaptation group for the self-report ratings or performance measures.
Discussion
The present study investigated the prevalence of circadian adaptation of police officers on a rotating shift schedule comprising multiple day, evening, and night shifts. This was achieved by evaluating changes in diurnal rhythms of urinary aMT6s and cortisol produced throughout the work cycle. Overall, 75 police officers contributed data before or after at least one shift, and most of these officers were adjusted to a day-oriented schedule when they began a given series of shifts. In contrast, only ~25% of them presented aMT6s and cortisol urinary rhythms adapted to night work after completing a series of consecutive shifts.
When focusing on our subsample of 37 officers with valid acrophase markers for both hormones before and after consecutive night shifts, 30% had aMT6s rhythms that adapted, and 24% had cortisol rhythms that adapted. As only 14% of the officers were deemed to have both rhythms of aMT6s and cortisol adapted to night shifts, this low number may be suggestive of internal de-synchrony between the two rhythms or differing sensitivity of the hormones to masking factors in the environment. Officers who adapted to night shifts demonstrated significantly longer phase delays than officers who did not for either rhythm. Group-level analyses of rhythmic parameters confirmed these results but revealed no between-group differences in mesor or amplitude. Interestingly, when officers were compared on the basis of the adaptation of their aMT6s rhythms, the officers who adapted obtained cumulatively more sleep per day throughout the series of night shifts than those who did not. These groups did not differ on any self-report measures of sleepiness or alertness, but the adapted group had fewer lapses in psychomotor performance toward the end of the night-shift sequence. There is evidence that personal characteristics such as age, sex, and chronotype can modulate adaptation to shift work (Blok and de Looze, 2011; Gamble et al., 2011; Kervezee et al., 2021; Boivin et al., 2023), although circadian markers are not reported in these publications. In the present study, officers did not differ in terms of the personal attributes that were measured whether they were circadian-adjusted to night shifts or not (Table 1).
Adaptation of Melatonin Rhythms
The prevalence of aMT6s adaptation to night work in the current study, based on the timing of the acrophase within the daytime sleep period, is similar to the 25% of permanent night workers estimated by Folkard (2008) to exhibit “substantial” adaptation, defined as a minimum of either (1) suppressed melatonin levels throughout the entirety of a night shift or (2) a peak in melatonin 2-3 h after sleep onset. Eastman and Martin (1999) proposed that the nadir of core body temperature, which typically occurs about 2 h after the acrophase of aMT6s (Nowak et al., 1987; Shanahan and Czeisler, 1991), should at least coincide with the first half of the sleep episode for partial adaptation and the second half for complete adaptation. When these criteria are applied to the subset of 37 aMT6s acrophases after night shifts, 8 police officers would be partially adapted, and 3 completely adapted, representing 22% and 8% of them, respectively. However, it should be acknowledged that due to the limitation in the temporal resolution of urine samples, a precise quantification of complete or partial circadian adaptation remains difficult.
The average delay observed for the aMT6s adapted group (−7.6 h) was longer than that of the non-adapted group (0.8 h). This shift for the adapted group is similar to the average phase delay observed for a group of 11 police officers on patrol in a separate study by Koshy et al. (2019). In that study, the rhythm of aMT6s profiles was also compared before and after 7 consecutive night shifts. Notably, however, phase assessments were derived from urine samples collected at more frequent intervals in a laboratory setting (Koshy et al., 2019). This may have facilitated a greater level of precision than was possible for samples collected in the field.
Field studies comprising sequences of 2-4 consecutive night shifts, shorter than those in our study, primarily described differences in the diurnal excretion profiles of aMT6s between day shifts and night shifts, rather than changes in circadian phase (Vangelova and Dalbokova, 1998; Hansen et al., 2006; Grundy et al., 2011). In contrast, work cycles comprising longer sequences of shifts, comparable to those of our study, which included the collection of circadian markers, were more likely to show signs of melatonin adaptation. Ferguson et al. (2012) conducted a study of 24 participants at a live-in mining site, wherein saliva samples were collected hourly between 1900 h and 2300 h each day for 7 night shifts (1745-0600 h). A significant delay in the timing of salivary melatonin was observed by the end of the shift sequence, but the effect was small (~30 min) and there was no indication of the prevalence of adaptation. Although light exposure was not monitored, sunrise occurred at ~0530 h, before the end of their shift, thus presumably exposing miners to light during the advance portion of the phase response curve to light and counteracting adaptative circadian delays.
Most of the studies that found at least substantial adaptation of melatonin rhythms, and were conducted entirely in the field, comprised offshore oil rig workers who collected urine samples every 2-3 h while awake on 14-day work cycles (i.e., either alternating 7 nights and 7 days, or fixed nights or days) (for a review, see Boivin et al., 2022). Barnes et al. (1998) found that the aMT6s acrophase shifted between 1.3 and 1.8 h per day, on average, across 2 weeks of night shifts. Workers had a high rate of complete adaptation (96%), with 22 of 23 workers having their final acrophase within the second half of their daytime sleep period by the end of the work cycle. Work cycles comprising a single week of night shifts also found high rates of adaptation, with Gibbs et al. (2002) and Hansen et al. (2010) observing average phase delays of 5.4 h and 4.1 h, respectively. Similarly, Gibbs et al. (2007) considered 82% of oil rig workers to be adapted based on an aMT6s phase delay of at least 3 h. Oil rig workers were exposed to light between 3 and 243 lux inside the ship on the night shift (Hansen et al., 2010), with an average of 80 lux/min during these work hours (Gibbs et al., 2007).
Adaptation of Cortisol Rhythms
At the end of the series of night shifts, only 24% of the police officers’ cortisol midpoints occurred during the 7-h daytime window used to classify circadian adaptation. Of the few other studies that investigated the adaptation of cortisol rhythms over the course of several night shifts, most have been conducted for work cycles comprising 2 consecutive night shifts (Costa et al., 1994; Jensen et al., 2016). Costa et al. (1994) measured plasma cortisol levels in 15 nurses at the beginning, middle, and end of 10-h night shifts and did not observe any deviations from a day-oriented schedule in the pattern of cortisol on the second shift. In a separate study comprising both permanent and rotating night workers, Kudielka et al. (2007) found similar daily variations in salivary cortisol values between the day shift and the second night shift, indicating no adaptation. A study by Harris et al. (2010) conducted with offshore workers compared the cortisol awakening response (CAR) in 14-day permanent day-shift and night-shift schedules with 7-night/7-day (swing) shift schedules. On the first night-shift day, the CAR was dampened compared to that in the day-oriented schedule. However, it showed signs of adaptation for both the permanent night and swing shifts by the end of the first week, with peak cortisol levels occurring near awakening. Despite the lack of circadian phase assessment in that study, it suggests that the offshore workers had a higher rate of adaptation than the police officers in our study (Harris et al., 2010). As described for melatonin rhythms, this high rate of adaptation for cortisol is likely a product of the environments unique to offshore oil rigs that do not apply to workers in other shift-work settings.
Sleep Duration and Environmental and Behavioral Exposures
Across the night-shift days, total sleep duration was shortest in the 24 h comprising the first night shift and then stabilized at longer durations for subsequent shifts. This pattern is consistent with the acute sleep deprivation often associated with the transition from a diurnal to nocturnal schedule (Santhi et al., 2007; Kosmadopoulos et al., 2016). Indeed, as many as 50% of workers do not nap immediately prior to their first night shift, thereby accumulating as much as 24 h of wakefulness by its end the following day (Knauth et al., 1980). The average total amount of sleep obtained each day (~6 h) is consistent with what has previously been reported for night-shift workers, across different chronotypes (Roach et al., 2003; Kervezee et al., 2021). While there was no difference in the overall 24-h sleep propensity rhythm between night shifts, the adapted aMT6s group began to obtain cumulatively more sleep than the non-adapted group by the fourth night shift, which is consistent with what would be expected of an adapting circadian system.
It has been estimated that the relative timing of light exposure might explain as much as 47% of the variability in the melatonin phase response to night work (Stone et al., 2018). Indeed, we previously demonstrated that a carefully-timed combination of bright light exposure at work and tinted goggles during the morning commute to home could delay melatonin rhythms by as much as 11.3 h (compared to 5.1 h in the control group) after ~12 night shifts (Boivin and James, 2002a). In addition, in a prior study from our laboratory, we observed that police officers who adapted to night work were exposed to higher light levels during their shift than police officers who did not (Boudreau et al., 2013). In the current study, average light exposure monitored by wrist-worn actigraphs was <200 lux, and the 24-h variation in light exposure on night-shift days was considerably dampened compared to that before and after the series of night shifts. This is likely due to the combined effect of dim working environments while on night-time patrol and also sleeping during the day. In contrast, in one of our prior studies (Boivin et al., 2012b; Boudreau et al., 2013), 8 out of 17 officers were exposed to bright light while working at night. While it is possible that wrist actigraphy did not accurately capture corneal light exposure, the light exposure measured for both aMT6s adaptation and non-adaptation groups was similar. It is also possible that individual factors other than exposure to light affected the rate of circadian adaptation to night work. The only between-group difference we observed was related to the diurnal rhythm of sleep propensity in the 72 h prior to the series of night shifts, although its clinical significance is unclear, and further field studies of shift workers will be necessary to investigate the individual factors that affect circadian adaptation to night work.
Alertness, Fatigue, and Psychomotor Performance
Based on other shift-work studies (Lamond et al., 2003; Crowley et al., 2004; Lamond et al., 2004; Harris et al., 2010), it was expected that sleep loss associated with a night-oriented schedule would result in considerably impaired psychomotor performance on the first night shift, with gradual improvement over subsequent days. In our subgroup of 37 police officers, there were no differences in median reaction time across days or between adaptation groups, but there was an interaction effect of aMT6s adaptation and shift day on the number of lapses (reaction times > 500 ms). These results showed no differences between groups for the first four night shifts, but a significantly improved performance was observed in the adapted group during the fifth night shift. There were no differences between groups for the self-report measures, but we did observe significant differences in the day-to-day variation of sleepiness, fatigue, and alertness, which were consistent with expectations from other studies (Crowley et al., 2004; Lamond et al., 2004). That is, these measures tended to be worst on the first night shift and gradually improved until the fourth night shift, after which subjective assessments plateaued. Ratings of fatigue and alertness worsened on days 6 and 7. However, this is most likely a consequence of the work schedule. Of the 76 police officers who were eligible to participate in the study, 26 worked rosters with 12-h night shifts scheduled on the final two nights. It was calculated that 48% of the officers who contributed self-reported measures worked a minimum of 12 h for at least one of the final two shifts. Importantly, we did not observe any group effects on performance or self-reported measures, by time of day, during the final shift.
Limitations
There are several limitations that should be considered when interpreting these results. The first is that a large number of urine-collection periods were excluded from analyses because the excretion profiles of aMT6s or cortisol were too ambiguous to reliably assign a midpoint. This meant that the subset of police officers who had available profiles for both hormones before and after night shifts was considerably reduced compared to the overall number of officers who completed the work cycle. While pruning the participant sample in this way increases confidence in the reliability of the phase markers that are included in the analyses, it is plausible that the physiological rhythms of these included officers may not reflect the range of possible effects of circadian misalignment and adaptation. A related issue is that all urine samples were collected outside of controlled laboratory conditions. While this is not unusual for field-based shift-work studies, it does mean that the rhythms of melatonin and cortisol may have been masked by environmental and behavioral confounders, such as light exposure, activity levels, and work-related stressors (Gander et al., 1986; Boivin and James, 2002b; Duffy and Dijk, 2002; van Eekelen et al., 2003). However, since both the adapted and non-adapted groups, based on their aMT6s profiles, had similar exposures to light, activity levels, and work schedules, it is reasonable to assume comparable masking effects between the groups. It is possible that irregularity in the timing and frequency of samples collected in the field may have also affected the precision of acrophases. However, given the windows used to ascertain circadian alignment were based on the known phase-relationships of aMT6s and cortisol acrophases to sleep (i.e., during the sleep period for aMT6s and around waketime for cortisol), we do not believe this would substantially affect classifications of adaptation to night work. Finally, it should be noted that the participant sample was predominantly male and sex-differences might exist (Boivin et al., 2023). While the proportion of female police officers in the study is comparable to the proportion in their workplace, the small number of female participants limited the investigation of sex-related differences in circadian adaptation to night work.
Conclusions
Circadian adaptation to a night-oriented schedule is important for mitigating the negative effects of working at night for both general health and workplace functioning. In this study, we investigated the extent to which police officers on rotating shift schedules were able to adapt to a series of night shifts. Two different hormonal circadian rhythms were used as markers of the circadian system—urinary aMT6s and cortisol. We found that only 30% and 24% of police officers substantially adapted to a night-oriented schedule based on their aMT6s and cortisol rhythms, respectively. The acrophases of aMT6s and cortisol rhythms occurred significantly later for the adapted group than for the non-adapted group. This supports findings from other field studies, involving health care workers and miners, that a minority of workers will show circadian adaptation to night work. Police officers whose aMT6s rhythms adapted to night work cumulatively obtained more sleep during the day between shifts. This may have been reflected in the improvement of psychomotor performance for adapted officers across days. Overall, the absence of meaningful circadian adaptation in most of the police officers suggests a need to either (1) reconsider the benefit of long sequences of night shifts in the work roster or (2) implement targeted interventions to facilitate faster adaptation.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231196280 – Supplemental material for Circadian Adaptation of Melatonin and Cortisol in Police Officers Working Rotating Shifts
Supplemental material, sj-docx-1-jbr-10.1177_07487304231196280 for Circadian Adaptation of Melatonin and Cortisol in Police Officers Working Rotating Shifts by Anastasi Kosmadopoulos, Philippe Boudreau, Laura Kervezee and Diane B. Boivin in Journal of Biological Rhythms
Footnotes
Acknowledgements
We wish to thank the research participants, staff, and students of the Centre for Study and Treatment of Circadian Rhythms for their contributions to this investigation. We thank Dr Dominique Walker for assistance with assaying the urine samples, Fernando Gonzales-Aste and Alexandre Gervais for helping with recruitment and data collection, Dr. Sylvie Rhéaume for clinical advice, and Johanne Gauthier and Manon Gagnon for administrative support. This study was supported by grant 2013-0046 to D.B.B. from the Institute de recherche Robert-Sauvé en santé et en sécurité du travail. A.K. and L.K. were funded by postdoctoral fellowships from the Fonds de Recherche du Québec–Santé.
Author Contributions
Conceptualization, D.B.B.; Data curation, A.K., L.K., and P.B.; Formal analysis, A.K., L.K., and P.B.; Funding acquisition, D.B.B.; Investigation, P.B.; Methodology, A.K., L.K., P.B., and D.B.B.; Project administration, D.B.B.; Resources, D.B.B.; Supervision, P.B. and D.B.B.; Visualization, A.K. and L.K.; Writing—original draft, A.K.; Writing—review & editing, A.K., L.K., P.B., and D.B.B. All authors qualify for authorship and have read and agreed to the published version of the manuscript.
Conflict Of Interest Statement
D.B.B. provides consultation services and legal expert advice on shift-work and sleep-related topics. Other authors have no conflicts of interest to declare.
Data Availability Statement
The data underlying this article cannot be shared publicly because participants and their organizations have not agreed that their data be placed in a publicly accessible database. Thus, for ethical and confidentiality reasons, the authors cannot provide public access to these data. Nevertheless, materials and data may be shared for investigation of scientific integrity if necessary.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
