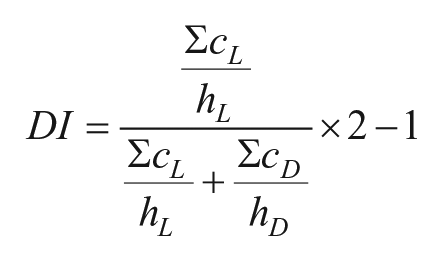Abstract
Several wild rodents, such as the subterranean tuco-tucos (Ctenomys famosus), switch their time of activity from diurnal to nocturnal when they are transferred from field to the laboratory. Nevertheless, in most studies, different methods to measure activity in each of these conditions were used, which raised the question of whether the detected change in activity timing could be an artifact. Because locomotor activity and body temperature (Tb) rhythms in rodents are tightly synchronized and because abdominal Tb loggers can provide continuous measurements across field and laboratory, we monitored Tb as a proxy of activity in tuco-tucos transferred from a semi-field enclosure to constant lab conditions. In the first stage of this study (“Tb-only group,” 2012-2016), we verified high incidence (55%, n = 20) of arrhythmicity, with no consistent diurnal Tb rhythms in tuco-tucos maintained under semi-field conditions. Because these results were discrepant from subsequent findings using miniature accelerometers (portable activity loggers), which showed diurnal activity patterns in natural conditions (n = 10, “Activity-only group,” 2016-2017), we also investigated, in the present study, whether the tight association between activity and Tb would be sustained outside the lab. To verify this, we measured activity and Tb simultaneously across laboratory and semi-field deploying both accelerometers and Tb loggers to each animal. These measurements (n = 11, “Tb + activity group,” 2019-2022) confirmed diurnality of locomotor activity and revealed an unexpected loosening of the temporal association between Tb and activity rhythms in the field enclosures, which is otherwise robustly tight in the laboratory.
Locomotor activity and body temperature (Tb) rhythms of rodents are tightly associated in time, with high Tb values occurring during the active phase. Locomotor activity also causes acute increases in Tb, further enhancing the high temporal correlation between the 2 rhythms through positive masking (Refinetti and Menaker, 1992; Weinert and Waterhouse, 1998). Due to this tight synchrony between daily high levels of Tb and activity, the phase of Tb rhythm has often been used as a proxy of the activity rhythm phase and, in extension, of the circadian oscillator (DeCoursey et al., 1998; Refinetti, 1999).
The comparison of activity rhythms in the field and laboratory has attracted particular interest of chronobiologists as an increasing number of studies have shown a switch of activity time, from diurnal to nocturnal, in several rodents when transferred from field to laboratory conditions (Hagenauer and Lee, 2008; Blanchong and Smale, 2000; Fritzsche et al., 2017; Kronfeld-Schor et al., 2013). For a long time, measurements of rodents’ activity rhythms in the field consisted of visual observations, trapping, or radio telemetry, while, in the laboratory, activity was measured by running wheel revolutions or as movement detected by infrared (IR) sensors (Silvério and Tachinardi, 2020). This raised questions on whether the diurnal to nocturnal switch might be an artifact of using different measurement methodologies in each condition. Before miniaturized activity loggers were available, measurement of Tb with implanted temperature loggers became the first choice for comparison of rhythmic patterns in the field and laboratory (Levy et al., 2007), using the daily Tb rhythm as a proxy of activity pattern. Another advantage was that implanted Tb sensors allow data to be recorded in otherwise inaccessible environments, such as the subterranean.
Subterranean tuco-tucos (Ctenomys famosus) are among the rodent species that display the abovementioned switch in activity time. In the laboratory light/dark (LD) conditions, tuco-tucos show robust nocturnal activity measured by wheel-running and IR sensors (Valentinuzzi et al., 2009; Yassumoto et al., 2019) and a tight temporal correlation between activity and Tb (Tachinardi et al., 2014). On the other hand, visual observations of tuco-tucos on the surface and automated recordings of light exposure in semi-field conditions strongly indicated that these animals would be diurnal in the wild (Tomotani et al., 2012; Flôres et al., 2016).
We report here a long-term study of 2 stages. The first stage was carried out between 2012 and 2016, in which abdominal Tb sensors were used as an indirect marker of activity (Tb-only group), to confirm if these animals are diurnal in the field. Our results revealed unexpected high prevalence of arrhythmic patterns of Tb. This contrasted with later studies carried out between 2016 and 2017 (activity-only group; Jannetti et al., 2019; Flôres et al., 2021), in which other individuals of tuco-tucos were released in semi-field using miniature movement biologgers (accelerometers). These results showed high prevalence of diurnality. The remarkable difference between these results motivated the second stage of this work, where we asked whether the tight correlation observed in the laboratory between Tb and activity was still held in the field. In this last stage (Tb + activity group), accelerometers and implanted Tb loggers were deployed in the same individuals, enabling our first simultaneous measurement of locomotor activity and Tb rhythms in both the field enclosures and the laboratory. These measurements revealed unexpected loosening of the temporal correlation between Tb and activity rhythms in semi-field conditions.
Methods
Trapping and Animal Maintenance
From 2012 to 2022, a total of 56 adult tuco-tucos (Ctenomys famosus) (158 ± 42 g; 31 females; 25 males) were captured inside a 10-km² area in Anillaco, La Rioja, Argentina (28°48′S; 66°56′W; 1350 m), using homemade polyvinyl chloride (PVC) traps (Tomotani et al., 2012) and taken to the laboratory in the research center CRILAR, located in the same village. This tuco-tuco species has previously been reported as Ctenomys cf. knighti or C. aff. knighti, but a recent species determination established that the Anillaco population is part of Ctenomys famosus, clade Velasco (Tammone et al., 2022). Captured tuco-tucos were weighed, sexed, chipped (Capri, Tecnologia Allflex, Brazil) and kept individually in acrylic cages (53 cm × 29 cm × 27 cm), with constant temperature at 24 ± 2 °C and natural illumination. Animals were fed ad libitum, with carrots, sweet potatoes, alfalfa, oatmeal, sunflower seed, rabbit chow, and greeneries. Food was replenished every 2 days during daytime. As other small desert mammals, tuco-tucos do not drink water (Buffenstein, 2000). All procedures of this work followed the guidelines of the American Society of Mammalogists for animal care and handling (Sikes, 2016), were approved by the Ethical Committee for the Use of Animals (CEUA) of the Institute of Biosciences—USP (protocols 273/2016; 339/2018), and authorized by Dirección General de Ambiente y Desarrollo Sustentable—Secretaría de Ambiente del Ministério de Producción y Desarrollo Local, La Rioja, Argentina (062/2008; 098/2018).
Semi-natural Enclosures
Three fenced outdoor enclosures were built (enclosure 1: 10 m × 5 m × 1 m and 1 m deep; enclosures 2 and 3: 12 m × 6 m × 1.5 m and 1 m deep) inside CRILAR’s facilities, in an area with native vegetation. Aboveground fences were made by wire mesh and concrete underground walls were built to prevent escaping. In addition, the top was covered by nylon net or wire mesh to prevent avian predation. Animals were kept individually in each enclosure and relied upon vegetation available (Larrea cuneifolia, Senna aphylla, Cercidium praecox, Opuntia sulphurea, Acacia aroma, among others), not being offered extra food. Environmental variables were collected with meteorological stations and external temperature loggers. Association of tuco-tucos rhythms with these factors will be treated in a separate study.
Animal Measurements
Measurements of tuco-tucos rhythms used in this work were divided into 3 groups described below with their corresponding sample size, considering only successfully retrieved data:
First, the group of tuco-tucos for which only Tb data were collected (n = 20), released in the enclosures from 2012 to 2016.
Second, tuco-tucos which had only activity monitored (n = 10), released from 2016 to 2017. Of those, 6 were already reported in the work by Flôres et al. (2021) and 1 in the work by Jannetti et al. (2019).
Finally, a third group had both Tb and activity rhythms collected (n = 11). These animals were released in semi-field from 2019 to 2022.
Implantation Procedure of Tb Logger
Daily rhythm of core Tb was measured with data loggers (diameter 17.35 mm, thickness 6.4 mm, weight 3.3 g; iButton; Maxim Integrated, USA; 5-60 min sampling rate, 0 to +50 °C range, 0.5-0.0625 °C precision) implanted in the abdominal cavity of tuco-tucos. Loggers were coated by a mixture of paraffin (Histosec; Merck, Germany) and ethylene vinyl acetate (Elvax; DuPont, USA), in a 4:1 proportion. Anesthetic consisted of isoflurane inhalation (5% in O₂ for induction and 2% for maintenance). Analgesic (flunixin; Banamine Schering-Plough, Rio de Janeiro, Brazil, 2.5 mg/kg) was administered through the abdominal incision for logger insertion. Incision was closed with absorbable surgical suture (Vicryl or polyglactin 910, Ethicon Inc., USA). Animals were kept under observation for 3 days. At the end of the experiment, tuco-tucos went through a second surgery to recover the logger. Extraction surgeries followed the same protocols.
Mounting Procedure of Activity Loggers
Two additional biologgers were used to detect body movement and tuco-tucos’ emergence to the surface. Accelerometers, for body movement (23 mm × 12 mm × 10 mm; 2 g weight; Axy-4; TechnoSmart, Italy), measured one acceleration value for each spatial axis (XYZ) at a 10-Hz sampling rate, range of −4 to +4 G force, with 8-bits resolution. Light loggers (0.65 g weight) were used for information on emergence of tuco-tucos to the surface. These data are being analyzed in a separate study. Both the light logger and the accelerometer were mounted on a collar made with cable ties inserted in a silicon tubing.
Experimental Protocol
Our enclosure/lab protocol followed the sequence used in the work by Tomotani et al. (2012). In that work, diurnal activity of tuco-tucos in the enclosures was first indicated by the high amount of times they were observed on surface during the day. To verify whether that diurnal activity was clock-controlled or due to masking (Kas and Edgar, 1999), tuco-tucos observed in the enclosures were transferred to lab constant darkness (DD) condition. If the free-running activity under DD occurred during subjective day, we would conclude that the observed diurnal activity in the enclosures was clock-controlled and that the clock had switched from nocturnal in the lab to diurnal in the field. On the other hand, if the free-running activity upon transference to lab DD immediately occurred in the subjective night, without transients, we would conclude that the phase of the clock had never changed between lab and field and the diurnal activity in the field was caused by masking or other downstream mechanisms (Tomotani et al., 2012). The results from Tomotani et al. (2012) suggested the second scenario and here we repeated the same protocol to verify the absence of transients, now using the same sensors across enclosure and laboratory.
From 2012 to 2022, a total of 56 animals (158 ± 42 g; 31 females; 25 males) were released individually in outdoor enclosures for different deployment durations (5-28 days). Twenty-five of these animals (161 ± 47 g; 13 females; 12 males) had only implanted Tb loggers, while 17 (173 ± 35 g; 8 females; 9 males) had only accelerometers deployed and 15 (151 ± 27 g; 10 females; 5 males) had both Tb logger and accelerometer deployed (Table S1). One animal from summer of 2016 (male 128 g) was released with both Tb and activity loggers, but was considered in both Tb-only and accelerometer-only groups, since its Tb logger did not have sampling rate high enough to be suitable for our time association analysis described below, which requires 5-min-resolution Tb data to be compared with activity data. Deployment duration was longer for some animals due to their participation in parallel experiments. Four animals were recorded twice, and 1 was recorded 3 times, resulting in a total of 62 recording trials. Recapture of tuco-tucos had a rate of success of 64%, resulting in 40 recordings (average of 4 recordings per year; 24 recordings of females and 16 of males; 17 of summer, 19 of winter, and 4 of autumn/spring), being 12 with accelerometer and temperature logger (7 females and 5 males; 5 in summer and 7 in winter) (Table S2). After recapture, tuco-tucos were transferred to constant conditions in the laboratory to evaluate the features of their endogenous activity and Tb rhythms. In this DD condition, animals were placed in cages without access to running wheels, inside ventilated light-tight boxes, with constant environmental temperature (24 ± 2 °C). After 10 days, the collar with loggers was retrieved and the temperature logger was extracted. Laboratory data were successfully collected in only 30 of the 40 recordings (75%) due to death of animals after recapture from semi-field (n = 4), technical issues with loggers (n = 3), or participation of animals in parallel experiments (n = 3).
Analysis of the Collected Data
Tb was used in the following analyses as raw temperature data. Body movement was processed as the vectorial sum of movement in the 3-dimensional axes (VeDBA) and this resultant was averaged every 5 min. All analyses and data treatments were performed using R software (R Core Team, 2022). Unless indicated otherwise, statistical comparisons between semi-field and laboratory were performed using Student’s t test.
Activity and Tb recordings were visualized in actograms, plotted with El Temps software (Díez-Noguera, 2021). Minimum and maximum thresholds used for Tb and activity were standardized to include the 60% central values of the data distribution.
Two chronobiological parameters were calculated for Tb and activity rhythms to quantify the differences in daily rhythms observed between semi-field and laboratory conditions. Diurnality index (DI) was measured for activity and for Tb as the proportion of activity episodes or high Tb during daytime in relation to the total recording length in semi-field conditions (Tachinardi et al., 2015). R script used is available in Supplementary Material II. This index ranges from −1 (all activity bouts or high Tb concentrated in nighttime) to +1 (all activity bouts or high Tb concentrated in daytime) and was calculated using the following equation, extracted from Halle and Weinert (2000):
where
Tuco-tucos have shown long-lasting entrainment aftereffects under lab DD conditions (Tomotani et al., 2012; Flôres et al., 2021), which means their activity and Tb rhythms usually maintain a 24 h rhythm on the 10-20 first days of DD before expressing their free-running period. This allowed us to use lab DD data for rhythmic comparisons with semi-field conditions, aware that they were less regular than under lab LD. Finally, we performed a preliminary verification of the association of DI and RI with season, year, and sex using 1-way analysis of variance (ANOVA). A more thorough analysis of the influence of these factors will be presented in a separate study.
For tuco-tucos with both activity and Tb data, with exception of animal 202 mentioned above (“Animal Measurements” section), time association between activity and Tb rhythms was evaluated and compared between semi-field and laboratory. Time association was defined as the percentage of simultaneous occurrences of high Tb/activity or low Tb/rest in relation to the entire recording. Activity occurrences were VeDBA values above the arithmetic mean of each recording. For Tb data, raw values above the mean were considered “high-Tb” episodes.
Changes in time association between lab and enclosure conditions could be primarily due to a change in the phase relationship between activity and Tb rhythms or due to dampening of either activity or Tb rhythms in field enclosures. To verify these possibilities, phase relationship estimations and RIs were compared between semi-field and laboratory. While we can estimate phase relationship for lab data using onset and offset time references (Tachinardi et al., 2014), extracting these time points for semi-field data is more challenging, due to their highly irregular profiles. We described in Supplementary Material I a more suitable alternative method, adapted from cross-correlation analysis, that does not require phase references. Finally, DIs were also calculated for this group of data to verify its representativeness in relation to the previous groups (Tb-only group and activity-only group).
Results
Tb-Only Recordings
The preliminary exploratory analysis showed no association of the temporal Tb patterns with year (one-way ANOVA; for DI: F = 3.24, R 2 = 0.26, p = 0.05; for transformed RI: F = 0.11, R 2 = 0.16, p = 0.9), season (1-way ANOVA; for DI: F = 0.003, R 2 = 0.05, p = 0.95; for transformed RI: F = 3.53, R 2 = 0.12, p = 0.13), or sex (unpaired t test; for DI: t = −0.34, df = 13.4, p = 0.74; for transformed RI: t = −0.7, df = 14.7, p = 0.56), which led us to group all animals’ data when comparing semi-field against laboratory condition.
In the enclosures, Tb profiles showed high interindividual variation, without a consistent rhythmic pattern (Figure 1a). Three main outcomes were observed in all Tb data collected during semi-field conditions. 45% of individuals displayed rhythmic Tb patterns (i.e., RI above significance threshold) and 55% were arrhythmic (represented by #180 and #155 in Figure 1a). Of the rhythmic Tb patterns, 44% concentrated high Tb during daytime (i.e., DI > 0; represented by #111 and #127 in Figure 1a), while in 56% of rhythmic individuals, high Tb episodes remained concentrated during nighttime (i.e., DI < 0; represented by #159 and #154 in Figure 1a).
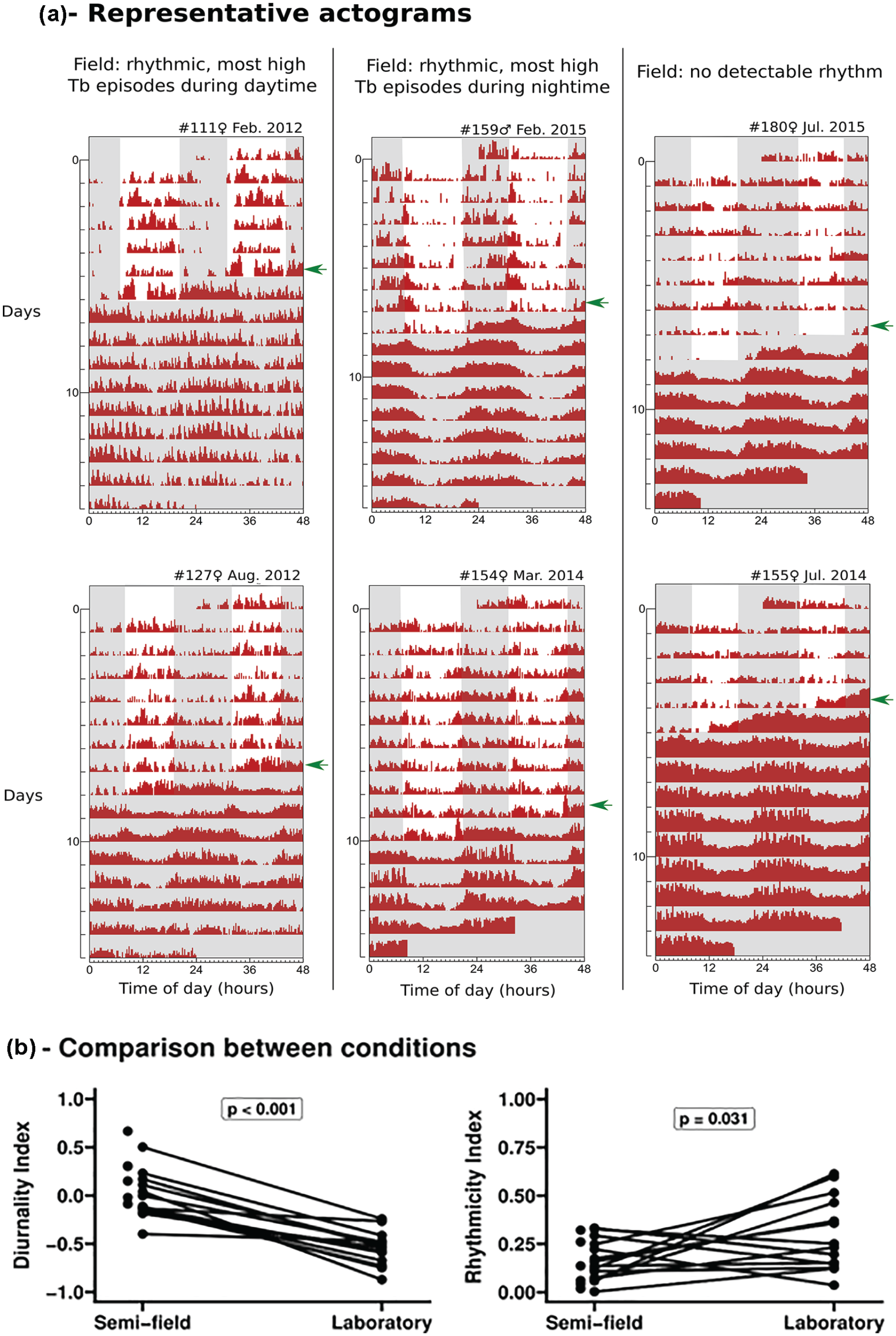
Chronobiological parameters and representative data of tuco-tucos from Tb-only group, compared between semi-field and laboratory conditions. (a) Double-plotted actograms of Tb values above a given threshold for 6 representative individuals for different Tb rhythmic patterns under semi-field conditions: animals from the left column are rhythmic (RI>0) and display high Tb predominantly during daytime (DI>0); animals from the middle column are rhythmic (RI>0) and display high Tb predominantly during nighttime (DI<0); and animals from the right column show no detectable rhythms (RI<0). Abbreviations: Tb = body temperature; DI = Diurnality index; RI = Rhythmicity index. Arrows on the right of each actogram indicate the moment the animal was recaptured and released into laboratory DD. Shaded gray areas indicate the dark phase of the day-night cycle when the animal was in the field enclosure or under laboratory DD. Labels above each actogram indicate animal number, gender, as well as month/year during which monitoring occurred. (b) Comparison of individual diurnality indexes (left) and rhythmicity indexes (right) of Tb rhythms. Lines connect values under each condition for a given individual. Statistics used Student’s t test with DI values and transformed RI values (see “Analysis of the collected data”). Data in the laboratory were lost for some animals, thus only semi-field parameters were plotted for these individuals (points without lines).
When transferred to lab DD, most animals displayed Tb rhythms with high levels concentrated during their subjective night, without transients, reproducing the same aftereffect patterns shown for wheel activity (Tomotani et al., 2012). When transformed RI and DI for Tb were compared pairwise between semi-field and laboratory (Figure 1b), there was a nonsignificant tendency of decrease of rhythmicity and significant increase of diurnality in the enclosures (paired t test; DI: p < 0.001; 0.52 mean difference; transformed RI: p = 0.03; −0.13 mean difference).
Activity-Only Recordings
Exploratory analysis for temporal activity patterns indicated only weak association of DI with year (year: 1-way ANOVA; for DI: F = 10.8, R 2 = 0.5, p = 0.01; for transformed RI: F = 2.64, R 2 = 0.15, p = 0.15; season: 1-way ANOVA; for DI: F = 0.36, R 2 = 0.2, p = 0.78; for transformed RI: F = 0.5, R 2 = 0.2, p = 0.7; sex: unpaired t test; for DI: t = −0.57, df = 7.3, p = 0.58; for transformed RI: t = −0.2, df = 7.64, p = 0.79). The only 2 individuals collected in 2016 showed the lowest RI indexes for activity which diminished their DI indexes and created the association between DI and year. None of the 10 recordings in the field enclosures were arrhythmic and all of them showed activity concentrated during daytime (DI > 0).
When DI and transformed RI for activity were compared pairwise between semi-field and laboratory (Figure 2), the only significant difference was the increase of diurnality in the enclosures (paired t test; DI: p < 0.001; 0.8 mean difference; transformed RI: p = 0.41; −0.02 mean difference).
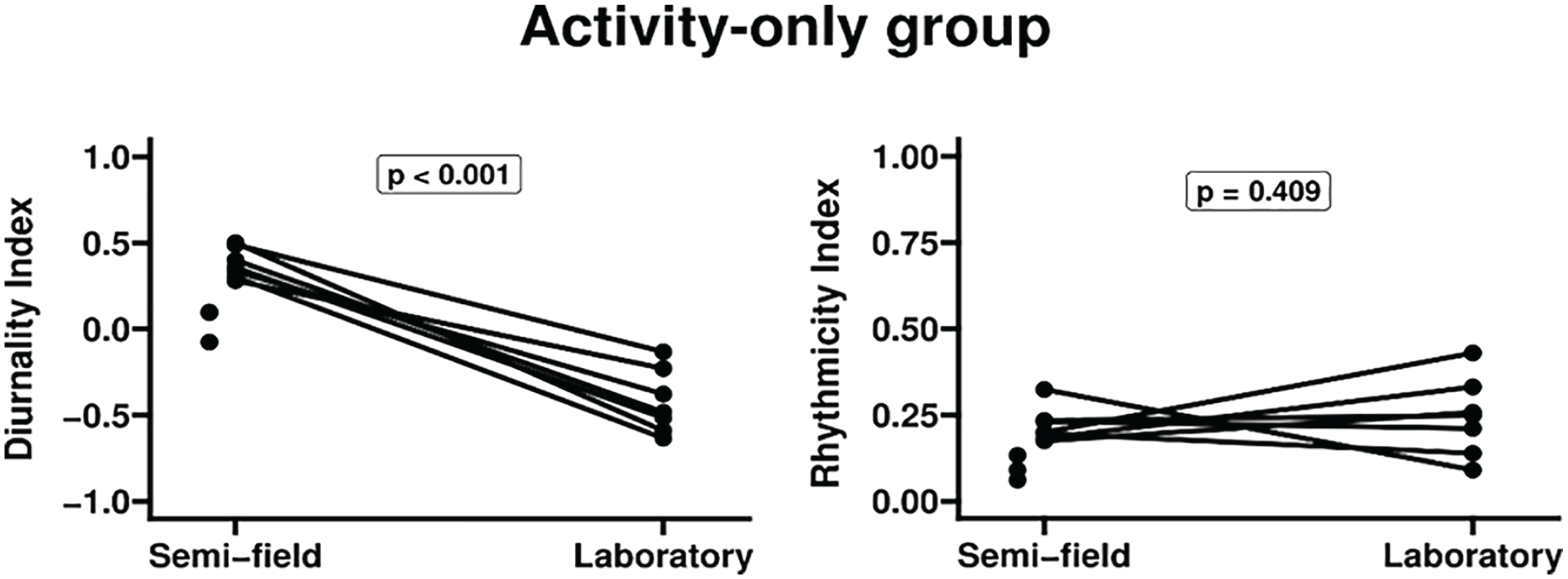
Chronobiological parameters of tuco-tucos’ data from activity-only group, compared between semi-field and laboratory conditions. Comparison of the individual diurnality indexes is shown on the left and comparison of rhythmicity indexes, on the right. Lines connect values under each condition for a given individual. Statistics used Student’s t test with DI values and transformed RI values (see “Analysis of the collected data”). Points without lines represent animals that had no measurements under laboratory conditions. Actograms for this group of data (activity-only) were already published in the works by Jannetti et al. (2019) and Flôres et al. (2021). Abbreviations: DI = diurnality index; RI = rhythmicity index.
Tb + Activity Recordings
The simultaneous measurement of activity and Tb revealed the same profiles described above, with activity rhythms showing more consistent patterns than Tb rhythms (Figure 3a). DIs (Figure 3b and 3c) confirm that these more recent data are representative and consistent with the previous recordings where Tb and activity were measured separately (Figures 1 and 2). Both activity and Tb are more diurnal in semi-field, with Tb showing lower DI than activity (paired t test for the difference between DI of activity and Tb; t = 4.4364; df = 10; p = 0.001). Transformed RI of Tb decreases in semi-field, while transformed RI of activity remains in a low level for both conditions (Figure 3b and 3c). Analyses performed with simultaneous measurement of activity and Tb revealed that there is a trend of lower temporal association between both rhythms when animals are in the enclosures (Figure 4a). When testing the causes of the decrease in time association between high Tb/activity and low Tb/rest, we found, first, that estimated phase relationship between activity and Tb is significantly different between semi-field and laboratory (Figure 4b). In semi-field, activity rhythm is estimated to be more advanced in relation to Tb rhythm than in laboratory, which is consistent with the qualitative evaluation of actograms in Figure 3a. Second, the lower rhythmicity of Tb in semi-field, independently of its phase relationship with activity, is another factor that contributes to the lower overall time association between both rhythms.
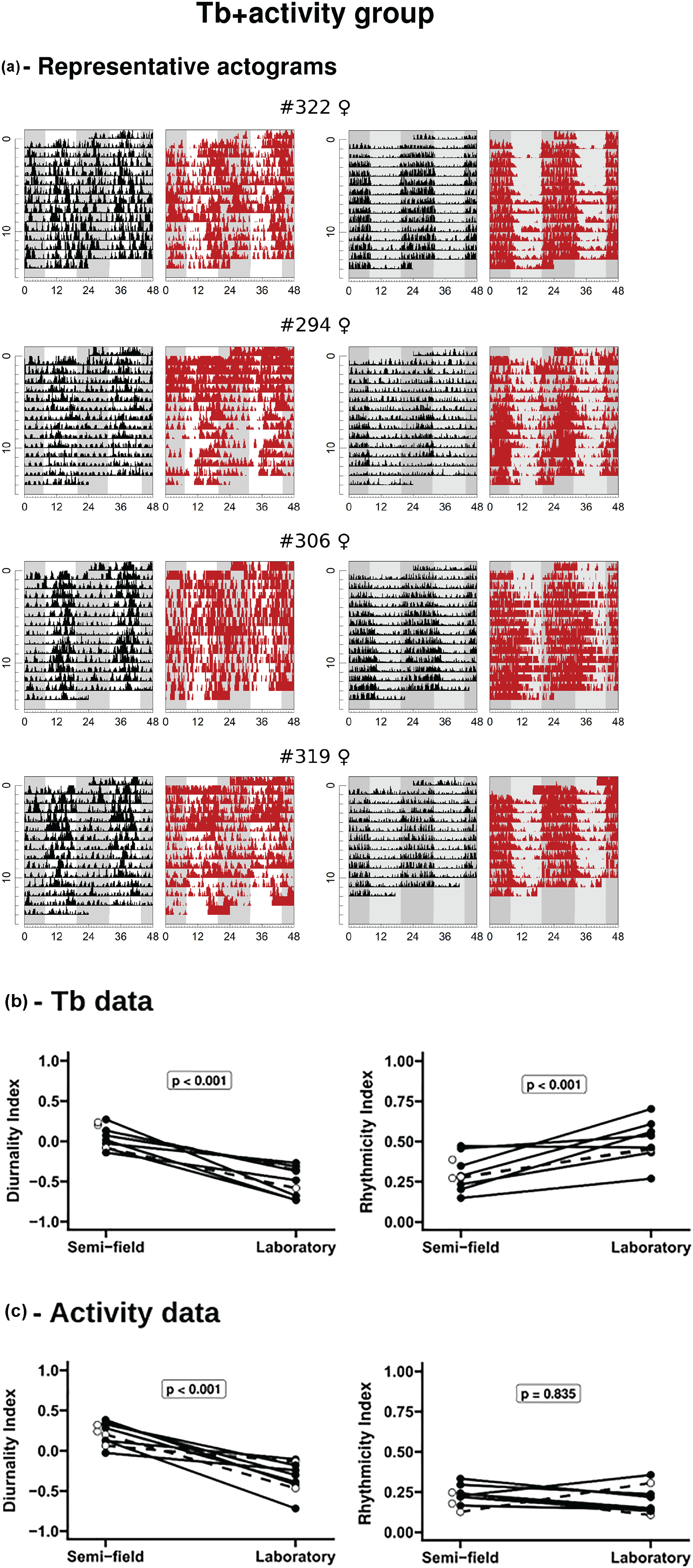
Chronobiological parameters and representative data of tuco-tucos from Tb + activity group, compared between semi-field and laboratory conditions. (a) Double-plotted actograms of 4 representative individuals for activity and Tb for both conditions. Left: semi-field condition; white and gray background represent natural photoperiod to which animals were exposed. Right: laboratory constant DD condition; background with different gray shades is a reference for external day and night. Activity is shown as 5-min summarized VeDBA (black marks) and Tb as raw data (gray marks). Abbreviations: Tb = body temperature; DI = diurnality index; RI = rhythmicity index; VeDBA = vectorial sum of movement in the 3-dimensional axes. (b) Comparison of individual diurnality indexes (left) and rhythmicity indexes (right) of Tb rhythms. (c) Comparison of individual diurnality indexes (left) and rhythmicity indexes (right) of activity rhythms. Statistics used Student’s t test with DI values and transformed RI values (see “Analysis of the collected data”). Open circles and dotted lines: recordings obtained in summer. Points without lines represent animals that had no complete measurements under laboratory conditions.
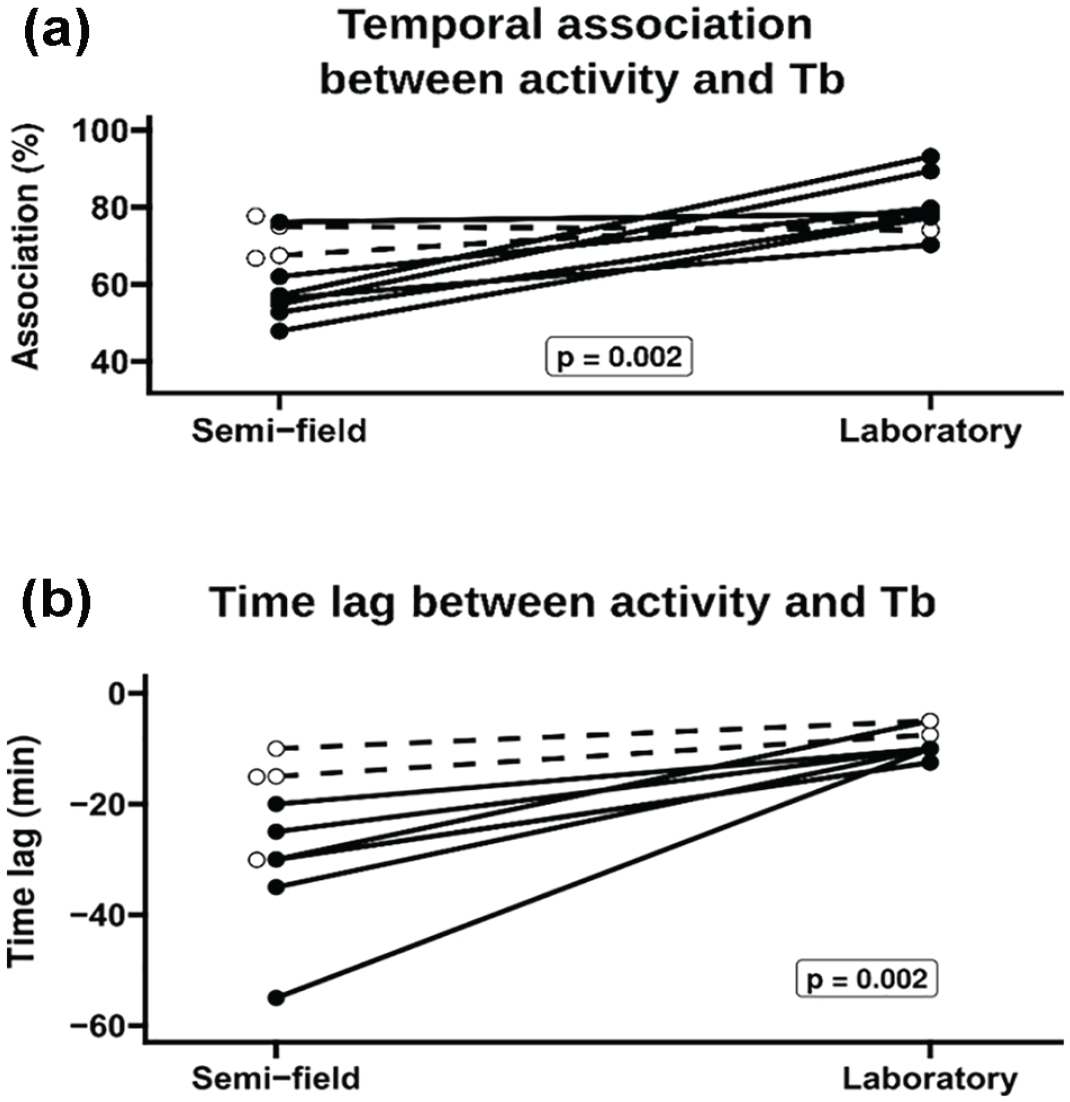
Analyses of temporal association and estimated phase relationship between Tb and activity when tuco-tucos were in semi-field and laboratory. (a) Percentage of time association (see “Analysis” section) between Tb and activity rhythms in both conditions. (b) Estimated phase relationship (time lag) between Tb and activity rhythms, extracted from an adapted cross-correlation analysis (Supplementary Material I). Abbreviations: Tb = body temperature. Open circles and dotted lines: recordings obtained in summer. Points without lines represent animals that had no complete measurements under laboratory conditions.
Discussion
Tuco-tucos display tight nocturnal synchrony between activity and Tb rhythms in the lab (Tachinardi et al., 2014; Yassumoto et al., 2019). This tight synchrony has been resistant to several experimental manipulations, such as under a constant light regimen, when both activity and Tb rhythms split into 2 components of different periods (splitting) (Flôres et al., 2021) or when both activity and Tb switch from nocturnal to diurnal in respirometry chambers (Tachinardi et al., 2015).
In the present work, we measured the Tb rhythms of a large number of tuco-tucos released in semi-field conditions in different years and seasons. Implanted Tb loggers did not preclude tuco-tucos’ normal behavior and recorded data for the entire experimental protocol. Individuals released with only temperature logger showed great variability in degrees of diurnality among them, with 56% of the total showing DI lower than 0 (Figures 1 and 2a). A significant amount (55% of the total) also showed arrhythmicity. This result made us ask whether tuco-tucos were rhythmic at all in natural conditions, where they may face more unpredictable challenges such as predation risk.
On the other hand, more recent activity recordings of tuco-tucos carrying only accelerometers (without Tb loggers) (2016-2017) showed that none of the individuals had DI < 0 and none were arrhythmic (Figure 2b). The great discrepancy between RI and DI of activity and those previously obtained for Tb (Figure 2a) led us to ask whether the tight synchronization between Tb and activity would hold in natural conditions, in the same way it has long been established for laboratory (Tachinardi et al., 2014).
The simultaneous measurement of Tb and activity allowed us to directly test the synchronization between these variables through the percentage of time association. Our analysis showed indeed a tendency of decrease of time association between activity and Tb (Figure 3). Animals measured in summer (n = 4) did not show a decrease of the same magnitude of those measured in winter. Although this sample size is too small to justify a separate analysis by season, we speculate that these animals contributed in part to the lack of significance of the difference between synchronization in the field and in the lab. This is further sustained by the fact that summer animals also expressed less divergent phase relationships in semi-field, suggesting that the change in the synchrony of these rhythms is less dramatic in summer than winter. This is also consistent with actograms (Supplementary Figure S3) and requires further investigation.
In the adopted semi-field setup, tuco-tucos were exposed to natural environmental conditions, which must also play an important role in Tb modulation. Since these mechanisms behind Tb modulation are independent of locomotor activity, they might dissociate Tb and activity rhythms, reducing the temporal association between them in nature. For instance, mammals’ Tb may suffer acute changes according to variations in the animal’s thermal environment (Geiser and Drury, 2003; Warnecke et al., 2008). Specially in the subterranean, thermodynamic factors can also modify Tb rhythms more chronically. Inside their tunnels, subterranean rodents dissipate heat mainly by conduction and have anatomic characteristics that increase the efficiency of this venue for heat transfer (Okrouhlík et al., 2015). The increased heat transfer by conduction, in comparison with the laboratory setup, may also affect the dynamics of heat dissipation when animals perform energetically demanding activity. While an activity in the laboratory results in a determined Tb increase, the same activity performed in the field may result in a lower or quicker Tb increase, as for 2 species of mole-rats (Okrouhlík et al., 2015) and Tb-activity association in the field would be reduced.
Switching of activity time, from nocturnal to diurnal, has been observed in different groups of animals and in different environmental contexts, being generally attributed to ecological causes, such as predation and competition pressures, or physiological causes, such as changes in thermoregulation strategies due to altered conditions of light, temperature, humidity, and food availability (Hut et al., 2012). Among rodents, there is a growing number of studied species that are diurnal in the field, but nocturnal when brought to the laboratory. For the tuco-tucos Ctenomys famosus studied here, the lack of transients in both Tb and activity rhythms when they were transferred from semi-field to laboratory favors the hypothesis that diurnal activity in natural conditions is determined downstream from the circadian oscillator (Tomotani et al., 2012). Also, the nocturnal Tb and activity rhythms under laboratory DD confirmed that, in this condition, both rhythms are tightly synchronized with the circadian clock, which was equally entrained in lab and field.
A downstream mechanism to explain diurnality in the field is in agreement with previous studies that argue that this activity switch is triggered by energetic challenges. For an animal exposed to energetically challenging environmental conditions, such as low ambient temperature and shortage of food, it can be an advantageous strategy to allocate demanding activities to the warmer daylight conditions, allowing energy savings with thermoregulation (Hut et al., 2011; Kronfeld-Schor et al., 2013). In this context, the fact that Tb and activity of Anillaco tuco-tucos did not always coincide in the increase of diurnality in semi-field suggests that the pathway that coordinates activity switches must be separated from the coordination of Tb rhythms.
Switching the timing of daily activity rhythms is only part of a wide repertoire of physiological and behavioral adjustments that animals express to cope with energy challenges. Investigating tuco-tucos’ daily rhythms alongside other strategies they use to survive in their habitat can help us understand why there is high interindividual variation in their rhythmic patterns, as well as why the relationship between activity and Tb is looser in the field than in the lab. Our future work will integrate the data shown in this study with other environmental, physiological, and behavioral variables to further investigate the effect of environmental factors on Tb of tuco-tucos and better interpret Tb rhythms in their complexity. For example, we have indications that tuco-tucos perform basking, detected from episodes of low body movement while animals were aboveground (Jannetti et al., 2019). This could be a strategy to maintain high Tb with minimal energy expenditure. Whether this is a relevant behavioral thermoregulation strategy in the field could be answered by integrating body movement, Tb, light exposure, and environmental temperature data. Understanding the intricate relationship among timing of activity, Tb, and environmental variables is a crucial step to unveil how subterranean rodents cope with energy challenges in their natural habitat.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231154715 – Supplemental material for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures
Supplemental material, sj-docx-1-jbr-10.1177_07487304231154715 for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures by Milene G. Jannetti, Patricia Tachinardi, Veronica S. Valentinuzzi and Gisele A. Oda in Journal of Biological Rhythms
Supplemental Material
sj-docx-2-jbr-10.1177_07487304231154715 – Supplemental material for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures
Supplemental material, sj-docx-2-jbr-10.1177_07487304231154715 for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures by Milene G. Jannetti, Patricia Tachinardi, Veronica S. Valentinuzzi and Gisele A. Oda in Journal of Biological Rhythms
Supplemental Material
sj-docx-3-jbr-10.1177_07487304231154715 – Supplemental material for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures
Supplemental material, sj-docx-3-jbr-10.1177_07487304231154715 for Temporal Dissociation Between Activity and Body Temperature Rhythms of a Subterranean Rodent (Ctenomys famosus) in Field Enclosures by Milene G. Jannetti, Patricia Tachinardi, Veronica S. Valentinuzzi and Gisele A. Oda in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors thank Johana Barros for excellent technical assistance; Jose D. Paliza, Eugenio Sanchez (Charly), Carlos Herrera (Carlitos), and Juan Mulet for the construction and maintenance of the enclosures; Dr. Loren Buck for introducing us the accelerometers and Carlo Catoni, from Migrate Company, for his kind assistance; André Yamachi, Danilo Flôres, Jefferson Silva, Giovane Improta, Tamíris Yassumoto for data collection; Lucas A. Zena for kind suggestions on handling the temperature sensors.
This work was supported by the Agencia Nacional de Promoción Científica y Tecnológica (grant number PICT 2013/2753); Consejo Nacional de Investigaciones Científicas y Técnicas (grant number PIP-11220120100415CO); Consejo Nacional de Investigaciones Científicas y Técnicas (grant number PUE-201622920160100125); Fundação de Amparo à Pesquisa do Estado de São Paulo (grant numbers 2012/23393-5, 2014/09324-6, 2014/20671-0, 2016/25058-0, 2017/19680-2, 2017/22973-1, 2019/26752-5); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brazil (CAPES) (grant numbers 001, 142215/2012-0, 88882.377169/2019-01)
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.