Abstract
The existence of a microbiome-gut-brain axis has been established wherein gut microbiota significantly impacts host behavior and physiology, with increasing evidence suggesting a role for the gut microbiota in maintaining host homeostasis. Communication between the gut microbiota and the host is bidirectional, and shifts in the composition of the gut microbiota are dependent on both internal and external cues (host-derived signals, such as stress and immunity, and endocrine and environmental signals, such as photoperiod). Although there is host-driven seasonal variation in the composition of the microbiota, the mechanisms linking photoperiod, gut microbiota, and host behavior have not been characterized. The results of the present study suggest that seasonal changes in the gut microbiota drive seasonal changes in aggression. Implanting short-day Siberian hamsters (
The interrelationships between the gut microbiota and the host are an increasingly important area of research, with mounting evidence indicating that the microbiota plays significant roles in the development and maintenance of host physiology and behavior (Fava et al., 2019; Heiss and Olofsson, 2019; Icaza-Chavez, 2013; Rosshart et al., 2017). Dysbiosis, an imbalance of the microbiota from a “normal” state (Tamboli et al., 2004), is implicated in the etiology and/or persistence of gastrointestinal diseases (Carding et al., 2015; Chang and Lin, 2016; Saffouri et al., 2019), mood disorders (Dinan and Cryan, 2013; Foster and McVey Neufeld, 2013; Huang et al., 2019), and neurodegeneration (Hirschberg et al., 2019; Raval et al., 2020; Spielman et al., 2018), among other conditions. The gut microbiota also impacts host behavior, including exploratory (Bercik et al., 2011), motor (Diaz Heijtz et al., 2011; Sampson et al., 2016), and social (Münger et al., 2018; Needham et al., 2018; Sharon et al., 2019) behaviors. The mechanisms of communication between the host and the gut microbiota have not been resolved, but communication likely involves humoral and direct neural signaling, as well as signaling via other host or bacterial pathways (Bercik et al., 2012; Mayer et al., 2014; Medina-Rodriguez et al., 2020). Although the mechanisms underlying the host-gut relationship are complex and remain largely uncharacterized, it is evident that the brain and the gut microbiota communicate. The composition of the gut microbiota is photoperiod-dependent (Bailey et al., 2010; Ren et al., 2020) and pineal gland–dependent in Siberian hamsters (
Siberian hamsters are a classic model organism in the study of photoperiodism (Ebling, 2015), as they undergo robust seasonal changes in a number of phenotypic measures. For instance, their pelage color changes from agouti in the summer to white in the winter (Duncan and Goldman, 1984) and they lose up to one-third of their body mass (BM) in the winter (Bartness and Wade, 1985). The Siberian hamster reproductive tract regresses in the winter (rendering the animals incapable of reproducing during the short winter days), but then recrudesces so that reproductive function is restored by spring (Darrow and Goldman, 1985). Other rhythmic seasonal changes include body temperature, immune function, aggression, and social behavior (Prendergast et al., 2009).
Seasonal rhythms depend on the translation of photoperiod cues (i.e., day length) into physiological cues (e.g., hormone signals). Briefly, light information drives rhythmic synthesis and secretion of pineal melatonin (MEL; Goldman, 2001). MEL is primarily secreted during the dark phase; thus, the duration of the MEL signal is directly proportional to night length (Carter and Goldman, 1983). MEL signals communicate information about photoperiod to a variety of central and peripheral targets. Although the central pathway underlying photoperiodic time measurement has been investigated, there is still much to learn about the mechanisms underlying seasonal changes in both central and peripheral physiology, as well as behavior. In particular, the seasonal regulation of aggressive behavior has not been fully characterized. It is established that Siberian hamsters are significantly more aggressive during short photoperiods (Kramer et al., 2008), despite this being the season during which circulating gonadal steroid levels and reproductive activity are at their nadir (Jasnow et al., 2000). Previous studies of aggression in Siberian hamsters indicate a role for MEL (Demas et al., 2004), gonadal steroid secretion (or lack thereof; Jasnow et al., 2000; Scotti et al., 2007), seasonal alterations in gonadal steroid receptor patterns (Kramer et al., 2008), and adrenocortical steroids (Scotti et al., 2008) in mediating seasonal aggression. However, the precise signals and systems involved in the expression of aggression have not been fully characterized. Recently, the gut microbiota was implicated in seasonal aggression of Siberian hamsters (Ren et al., 2020), a promising new avenue to explore the physiology underlying aggression. Thus, our goal was to explore and clarify the relationships between gut microbiota and seasonal aggression.
We used fecal microbiota transplants (FMTs) to manipulate gut microbiota within treatment hamsters with microbial communities from donor hamsters. FMT is a proven method for transplanting gut bacteria (Borody and Khoruts, 2011; Pamer, 2014) and is increasingly used in health science research (Aroniadis and Brandt, 2013; Smits et al., 2013; Xu et al., 2015). FMT is now employed as a therapeutic for human gastrointestinal disorders (Choi and Cho, 2016), with further applications in neurology (Vendrik et al., 2020) and various other conditions—including metabolic, autoimmune, and allergy disorders (Xu et al., 2015)—currently in development. FMT is often implanted into a germ-free (GF) recipient animal: a GF animal represents a “blank slate” environment, which simplifies not only the engraftment of the transplant but also the linkage between specific bacteria to specific physiological and/or behavioral outcomes (Luczynski et al., 2016). However, development of the metabolic, gastrointestinal, neurological, and immune systems in GF animals is not normal (Grenham et al., 2011; Luczynski et al., 2016; Wrzosek et al., 2018), and furthermore, since GF animals do not occur in nature, these models are not as biologically relevant as a conventional animal model; for these reasons, we did not use GF animals in the present study. We believe that the Siberian hamster—with its robust seasonality and readily observable shifts in physiology and behavior—represents a highly relevant model for investigating the relationships among gut microbiota, behavior, and photoperiod.
While research into mammalian microbial communities often focuses on gut communities (generally through examination of fecal pellets), other body compartments house distinct communities, including the mouth (Wade, 2013), lungs (Beck et al., 2012), reproductive tract (Koedooder et al., 2019), and skin (Grice and Segre, 2011), among others. Often, however, these compartments are not fully independent (i.e., connections between the gut and other bodily compartments exist, allowing for microbial movement between interconnected systems), but this interconnectivity has been poorly studied. In addition to the gut microbiota, in the present study we were interested in exploring the potential roles for microbes outside of the gut in regulating seasonal behavior. Hamsters have large cheek pouches and a functional cecum; both tissues contain microbial communities, and they are connected to the gastrointestinal tract, providing food storage (Keyes and Dale, 1944) and digestive (Sakaguchi, 2003) functions (both cheek pouch and cecum are anaerobic environments). Thus, if a gut-brain connection exists, it cannot be precluded that hormonal or enzymatic production does not have its genesis in connecting bodily compartments, and products may get translocated (as seen in other animals, such as
The goal of this study was to clarify interactions among photoperiod, microbiota, and host behavior. If the composition of the gut microbiota drives seasonal changes in host behavior, then FMT from hamsters in a long photoperiod should alter the behavior of host hamsters housed in a short photoperiod, and vice versa.
Materials and Methods
Animals and Housing
Male Siberian hamsters (
Procedures
BM, Food Intake, and Testicular Measurements
BM (g ± 0.01 g) was measured once weekly, at the same time and day, for the duration of the study (weeks 0-5). Food was weighed weekly (g ± 1 g), 3 h prior to the dark phase, and food hoppers were refilled once per week, following food weighing.
Photoperiodic response was determined using estimated testes volume (mg ± 0.01 mg; testis length by width squared; Freeman et al., 2007). Testis measurements were obtained using analog calipers while animals were under light anesthesia (3% isoflurane and medical oxygen at a flow rate of 3 L/min). Estimated testes volume was measured on weeks 0, 2, and 4, on the same day and time. At the termination of the study (week 5), testes were surgically removed and weighed for final measurement of paired testes mass (mg ± 0.01 mg). All testes measurements (both estimated and paired testes masses) will be jointly referred to as paired testes mass. A reduction of ≥30% paired testes mass by week 5 was classified as gonadal regression (Freeman et al., 2007; Prendergast et al., 2001). Animals that failed to exhibit ≥30% decreases in testis size were classified as SD nonresponders and were excluded from all subsequent analyses.
Treatment Groups
Hamsters were randomly assigned into 1 of 4 treatment groups: LDfs, SDfl, LDfl, or SDfs (Figure 1).

Treatment group information. Abbreviations: LD = long day; FMT = fecal microbiota transplant; SD = short day; R-I = resident-intruder.
Oral Gavage: Polyethylene Glycol Cleansing and FMT
Oral-gastric gavage is a method used for infusion of materials directly into the gut of a recipient. Hamsters underwent FMT via oral-gastric gavage; methods followed those previously described (Wrzosek et al., 2018) with some modifications. Briefly, on week 1 (day 1) of the study, all treatment hamsters received 2 gavages (200 μL each) of polyethylene glycol (PEG 4000; Alfa Aesar, Ward Hill, MA) to cleanse the bowel contents and allow the donor’s transplanted microbiota to establish in its new host (Wrzosek et al., 2018). Four hours after polyethylene glycol administration, treatment animals underwent oral gavage with the corresponding FMT (either LD or SD slurry). Each hamster underwent oral gavage with 200 μL of FMT slurry on day 1, as well as once weekly on weeks 2, 3, and 4. Fecal samples (2-4 fresh pellets, deposited directly into a sterile container) from treatment animals were collected on weeks 0 and 5 and immediately frozen (−80 °C). Extra aliquots of FMT slurries were retained to be included in our sequencing libraries. Oral gavages and all fecal sample collections occurred on the same day of each week and within the first 2 h of the dark phase of the light:dark cycle. A full description of gavage methods and materials can be found in supplementary methods.
Resident-Intruder Paradigm
The resident-intruder (R-I) paradigm was conducted on week 5 to quantify the effects of FMT on aggressive behavior. The R-I procedure involves placing an “intruder” animal into the home cage of a “resident” animal and observing the subsequent displays of aggressive behaviors (Albers et al., 2002). Each R-I dyad lasted 10 min and was filmed to allow behavioral analyses at a later time (Kramer et al., 2008). To control for circadian variation in behavior and gut microbiota composition, each R-I dyad took place within the first 2 h of the dark phase of the light:dark cycle. This is a model method for studying Siberian hamster behavior, and procedures followed those previously described (Huhman et al., 2003; Jasnow et al., 2000; Kramer et al., 2008; see supplementary information for full R-I methods).
Following R-I testing, experimental hamsters were euthanized. The cecum and both cheek pouches were collected from each hamster. After extracting each cecum, the interior was scraped clean and only the internal contents were retained. The cheek pouches were collected whole. The cecal contents and cheeks were immediately frozen (−80 °C) following collection.
All experimental procedures and husbandry were approved by the University of Memphis Animal Care and Use Committee (Protocol #815) and comply with the criteria established by National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
DNA Extractions and Library Preparation
Methods for DNA extraction and library preparation followed those previously outlined (Brown et al., 2019; Shor et al., 2020). Bacterial amplicon libraries were generated by amplifying the 16S (V4) ribosomal RNA (rRNA) region. Primary (1°) and secondary (2°) polymerase chain reactions (PCRs) were conducted in 25 μL reactions. See supplementary methods for a complete description of methods.
Bioinformatics
Sequence data were processed using the program mothur (v.1.44.2; (97)), generally following Shor et al. (2020). See supplementary information for a full description of bioinformatics methods and for sequencing results.
Statistical Analyses
Weekly food intake was calculated as grams of food consumed per gram of BM. One-way analysis of variance (ANOVA) models were used to analyze paired testes mass, BM, and food intake against treatment (LDfl, LDfs, SDfl, SDfs). Treatment differences in aggression (latency to first attack, duration of aggression, number of aggressive bouts) were also analyzed using one-way ANOVA. Where applicable, post hoc planned comparisons including Tukey’s honestly significant difference (HSD) or Fisher’s Protected Least Significant Difference (PLSD) test were used to identify treatment differences.
To investigate community shifts among treatments, we utilized a Dirichlet multinomial mixture analysis (Ding and Schloss, 2014; Holmes et al., 2012) to classify operational taxonomic units (OTUs) into unique community types (CTs). The CTs created via Dirichlet multinomial mixture analysis represent metacommunities (enterotypes) whereby each CT represents a group of OTUs that behaves and responds similarly across experimentation. These data were analyzed using χ2 likelihood ratio tests to test whether the distributions of CTs within groups differed over time, as well as between groups on week 5 (terminal experimental date). Because of the relatively small number of samples, and that the expected number of occurrences within some combinations was less than 5, we verified the obtained results using Cochran-Armitage trend tests for week 0 versus week 5 comparisons.
In addition, to identify OTUs that differed in abundances between LD and SD donors (biomarkers), we used Linear Discriminant Analysis Effect Size (LEfSe) as implanted in mothur (Segata et al., 2011). LEfSe analysis was also used to identify biomarker OTUs disproportionally found within FMT treatment groups.
To examine the relationships between individual OTUs and aggressive behavior, we utilized zero-inflated negative binomial generalized regression analyses. Only OTUs with a total global relative abundance greater than 0.1% underwent regressions (95 most abundant OTUs). Due to the relatively small number of sample animals at week 5 and the large number of zero values that may impact robustness of regression results, we also simulated data based on obtained regression results to determine directionality of OTU correlations with aggression and to provide additional targets for future research on bacterial mediated aggression. Using JMP Pro v.15, we simulated OTU data using 10,000 iterations (randomized by model factors) for each OTU (within the top 95) to confirm and predict the direction and significance of all correlations in the context of a larger sample pool. Simulated OTU data were tested to see whether they were correlated with scored aggression values using the nonparametric Spearman’s ρ. Additional information regarding the statistics can be found in the supplementary information.
Results
Exclusions
Two hamsters from the SDfl group and 1 hamster from the SDfs group (see group descriptions in “Materials and Methods” section) failed to exhibit testicular regression; these hamsters were classified as nonresponders and excluded from analyses. One hamster from the LDfl group (Figure 1) died (reason unknown) on week 3 of the study. The final treatment group numbers after these exclusions were as follows: LDfl = 3, LDfs = 8, SDfl = 6, SDfs = 3, LD-control = 20.
BM, Testicular Measurements, and Food Intake
BM was dependent on photoperiod, but not treatment, at both the start and end of the study. On week 0, there were significant differences in BM between LD and SD (one-way ANOVA:
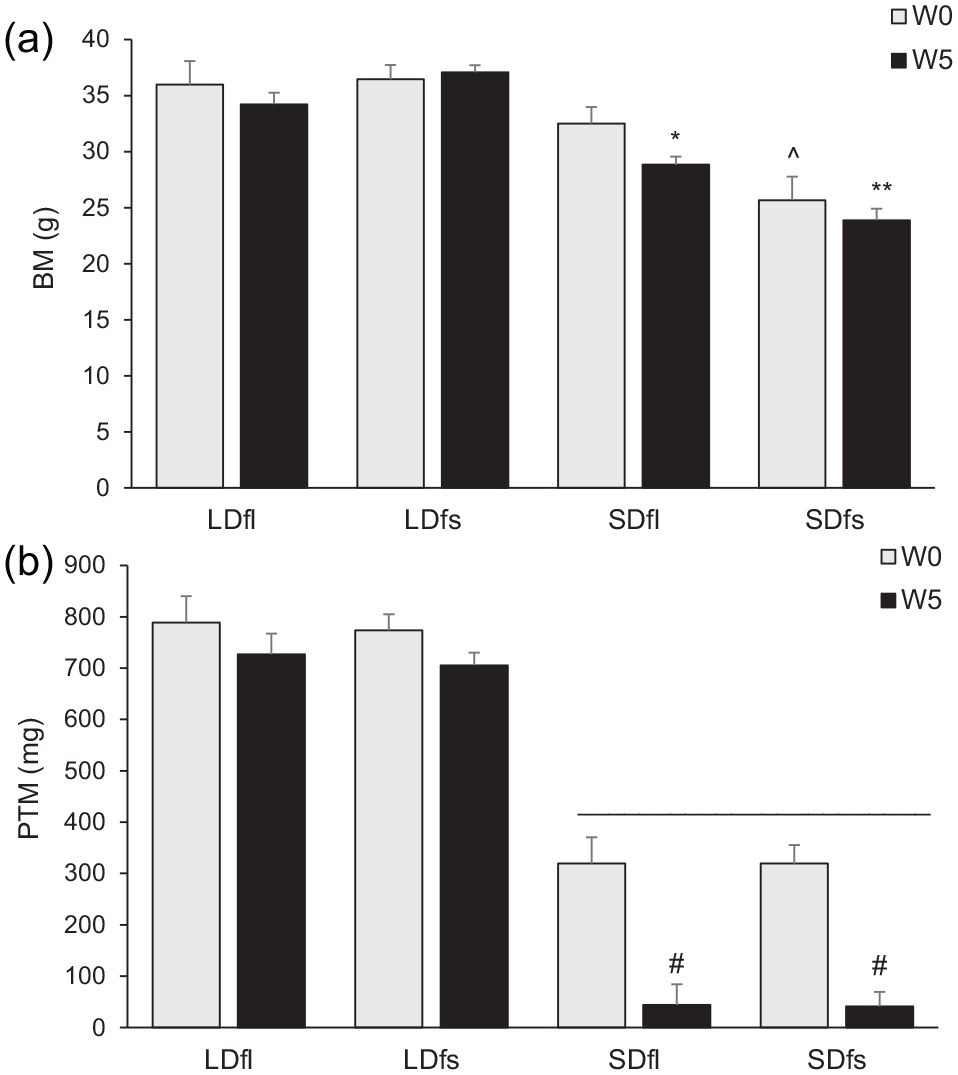
Somatic responses over time. Body mass (a) and paired testes mass (b), W0 versus W5. (A) * SDfl W5 < SDfl W0 (
Testes were larger in LD compared with SD hamsters at both weeks 0 and 5, regardless of the treatment (one-way ANOVA: week 0:
Food intake was independent of the photoperiod throughout the study (one-way ANOVA: week 0:
R-I Paradigm: Aggressive Behavior
The number of aggressive bouts was significantly greater in the SD-housed hamsters that were transplanted with SD microbiota (SDfs) compared with all the other groups; none of the other groups differed from each other (one-way ANOVA:
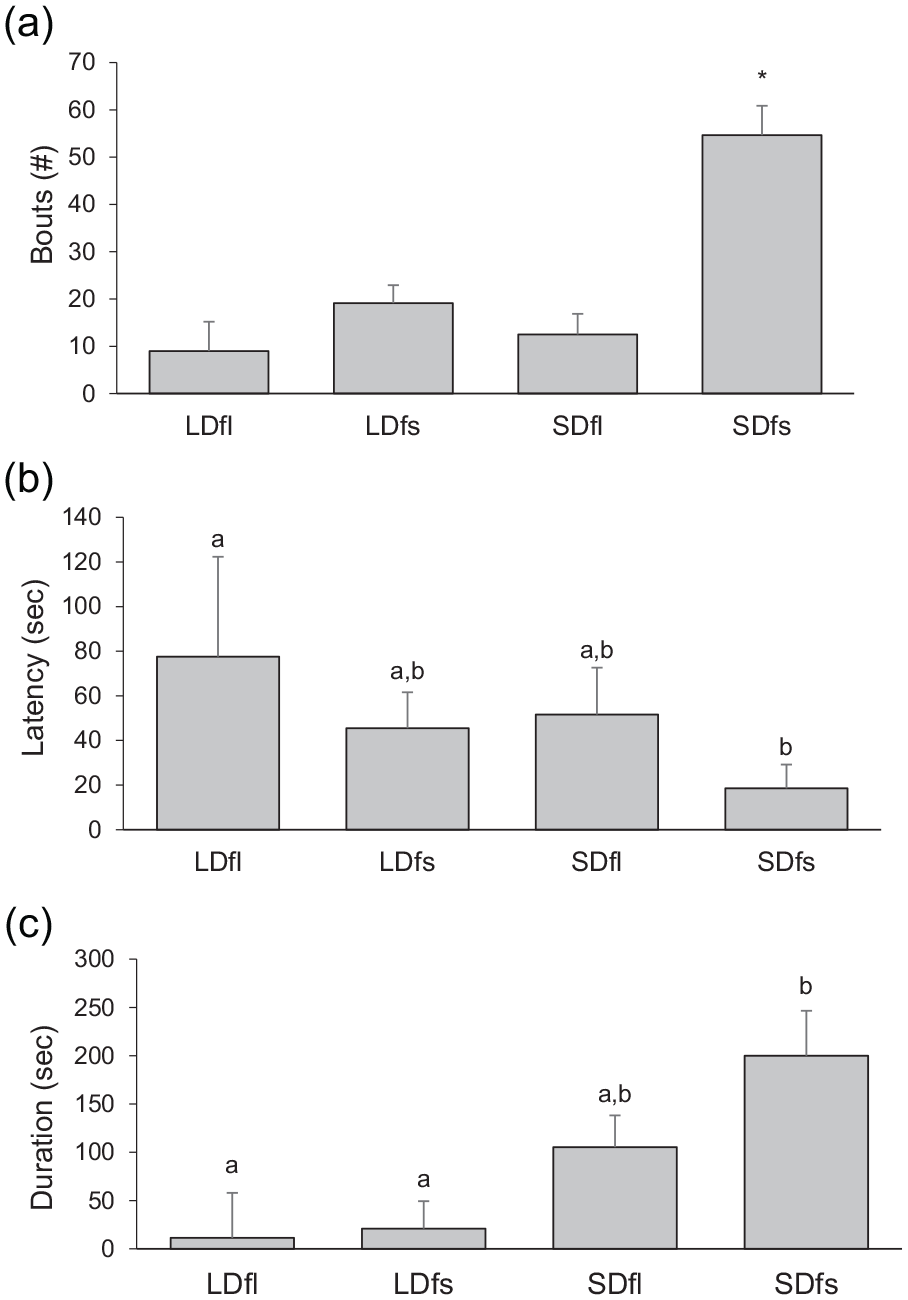
Treatment differences in aggressive behavior. Number of aggressive bouts (a), latency to first attack (b), and total duration of aggression (c). (a) SDfs > all other groups (*
The latency to first attack was significantly greater in the LD-housed hamsters receiving LD microbiota (LDfl) compared with hamsters housed in SD lengths that received SD microbiota (SDfs). Latency to attack in the groups that received the opposite microbiota (LDfs and SDfl groups) was intermediate and did not differ from any other group (one-way overall ANOVA:
The total duration of attacks differed among the treatment groups (one-way ANOVA:
Determination of Bacterial CTs
Communities were demarcated into 4 CTs based on Dirichlet multinomial mixture analysis (Supplemental Figure S2). CT demarcation was optimized into Dirichlet components using minimum Laplace approximations. Each partition had similar community variability except CT3, which was much more heterogeneous (CT1 θ = 2371.45, CT2 θ = 1713.12, CT3 θ = 125.08, C4 θ = 1321.02). These CT data were used in likelihood-based contingency analyses for all sample types.
CTs Across Samples
Neither photoperiod nor treatment affected the distribution of CTs in cheek and cecum samples. Overall, fecal sample communities were significantly different than the other sample types (χ2 = 100.93, df = 6,
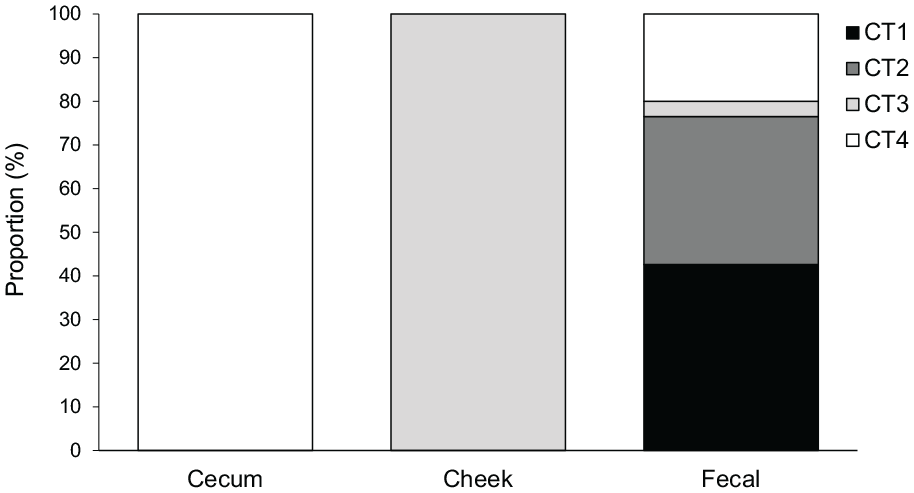
Community types (CTs) by tissue at week 5. Cecum and cheek pouches were 100% demarcated into CT4 and CT3, respectively. Fecal samples were classified as a mixture of CT1 (42.6%), CT2 (33,9%), CT3 (3.5%), and CT4 (20.0%).
Community Difference in Donor Slurries
PERMANOVA results indicated that microbiota communities were significantly different in LD versus SD donor slurries (
Fecal Samples: Treatment Differences of Communities
CT Over Time (Week 0 vs Week 5)
FMT treatment did not significantly change the composition of CT from week 0 to week 5 in LDfl, LDfs, or SDfs (Table 1; Figure 5a, b, and d). There was, however, a significant change in SDfl. On week 0 versus week 5, CTs in SDfl were marginally different with likelihood ratio analysis (χ2 = 5.545,
Community types across treatments, week 0 vs week 5.

Abbreviations: CT = community types. Cochran-Armitage trend testing (
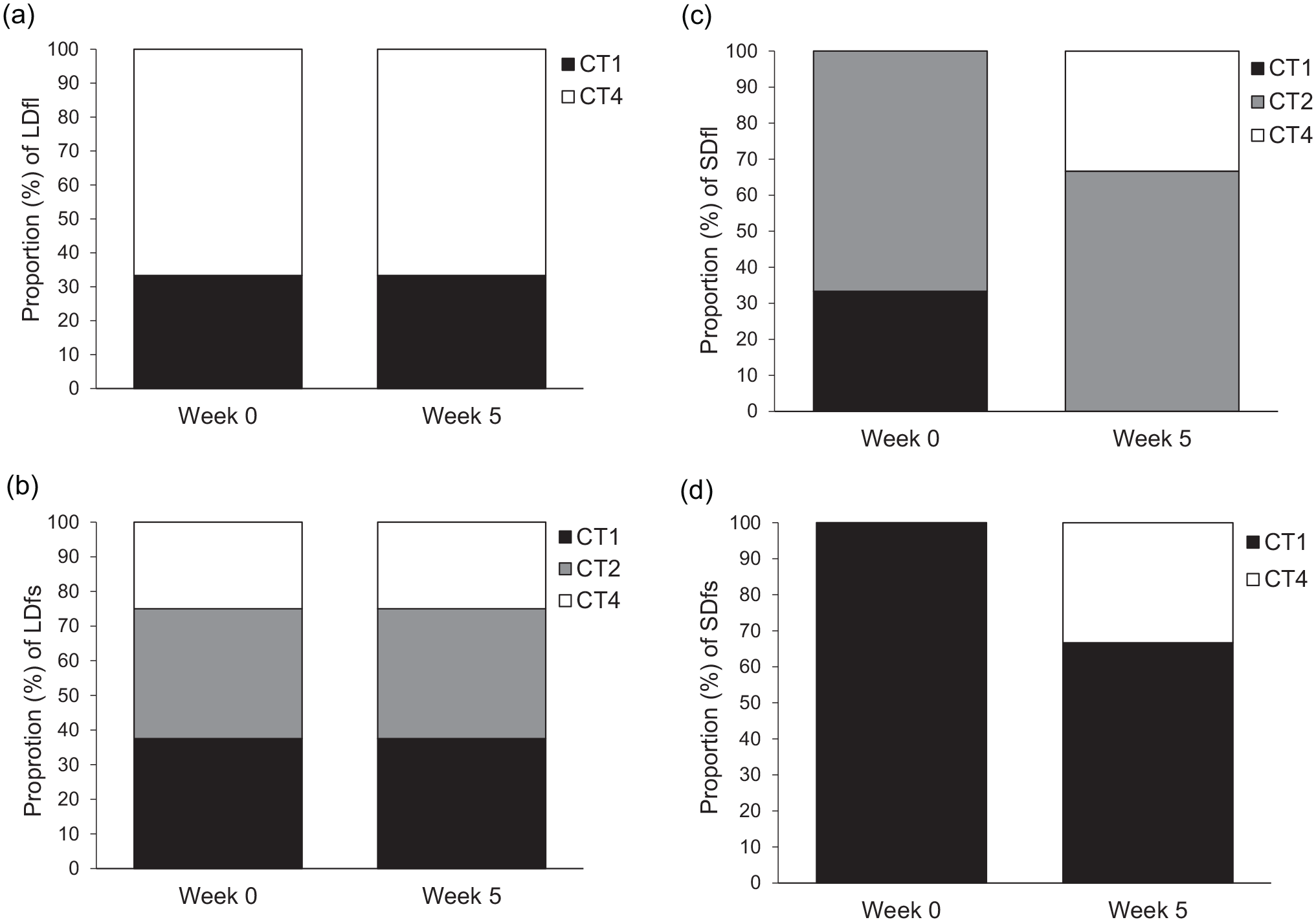
Community type (CT) distributions by treatment group, week 0 vs week 5. SDfl (c) communities changed significantly in ratio and identity over time (
CT Within Groups (LD and SD, Week 5)
Treatment did not affect CT composition within LD groups: LDfl was not significantly different from LDfs on week 5 (χ2 = 2.847,
Taken together, the CT results suggest that LD hamsters are more resistant to microbiota perturbation than SD hamsters. Furthermore, SDfl hamsters were the only group to show changes in CT along with changes in aggressive behavior.
OTUs and Aggression
LEfSe analysis of fecal samples revealed that 3 OTUs (from the top 95 most abundant) were enriched in a subset of SDfl hamsters (Table 2). These OTUs were enriched in 2 SDfl hamsters that displayed pronounced aggressive behavior typical of the SD photoperiod, suggesting that elevated levels of these taxa may be related to aggressive behavior (or, conversely, that diminished levels of these taxa may be related to lack of aggression).
Enriched OTUs in aggressive SDfl hamsters.

Abbreviations: OTU = operational taxonomic units; LDA = linear discriminant analysis; LEfSe = linear discriminant analysis effect size. OTU is significantly enriched in aggressive phenotype SDfl hamsters. LDA was calculated using LEfSe analysis.
OTU could not be identified at the genus level.
Several OTUs were also correlated with aggression (zero-inflated negative binomial regression, top 95 OTUs): 10 OTUs were correlated with aggression at α ≤ 0.05, and an additional 8 OTUs were correlated at α ≤ 0.1 (Supplemental Table S4). These OTUs represent 9 genera (and/or families, where genus could not be identified). After these regressions, we followed with simulations using our predictive model (10,000 iterations) and found that 88 OTUs correlate significantly with aggression (13 positive, 75 negative;
Discussion
The relationships among the brain, gut microbiota, physiology, and behavior are complex, and considering photoperiod within this network creates an additional layer of complexity. In this study, we characterize this network by investigating the relationship between seasonal gut microbiota and seasonal behavior. Previous results established that the gut microbiota changes seasonally (Bailey et al., 2010) and that many of these changes are dependent on the pineal gland (Shor et al., 2020). This left unanswered the question of whether changes in gut microbiota are merely another photoperiod-dependent measure, or, alternatively, whether seasonal changes in the gut microbiota drive other seasonal rhythms in physiology and behavior. The present results suggest that seasonal changes in the gut microbiota drive seasonal changes in aggressive behavior. Multiple measures of aggressive behavior were impacted by transplanting microbiota from the opposite photoperiod. For example, SD hamsters implanted with LD gut microbiota displayed a reversal in seasonal aggression with respect to both the number and duration of attacks: Thus, these hamsters expressed LD-like low levels of aggression rather than the typical SD-like increase in aggression. The results also indicate intermediate aggressive behavior with regard to the latency to attack in LD-housed hamsters that received SD microbiota. We also found numerous correlations between individual OTUs and aggressive behavior. These results support our hypothesis that photoperiod-dependent changes in the gut microbiota are a driver of seasonal changes in aggression. Furthermore, although the microbiota in cheek pouches, cecum, and feces all differed from one another, the composition of microbiota in cheek pouches and ceca were independent of the photoperiod and FMT. We thus rejected our alternative hypothesis that seasonal behavior is driven by photoperiod-dependent changes in cheek and/or cecum microbiota.
Despite the lack of photoperiodic or treatment differences, the cheeks and ceca still provide important insights into the distribution of microbial communities throughout the body. It is interesting to note differences in the composition of cheek versus cecum versus fecal microbiotas. The lack of significant overlap in CTs between cheek and fecal samples is noteworthy. Hamsters are coprophagous: they consume feces and often store fecal pellets within their cheek pouches. In addition, there was a possibility of unintentionally depositing some of the donor fecal microbiota in the recipient mouth during the oral-gastric gavage procedure. Given these factors, we could have expected more similarity in fecal and cheek microbiotas. Furthermore, we could have expected more overlap between cecum and fecal microbiotas, due to both their close physical proximity and the integration of the cecum within broader gastrointestinal physiology (Grant, 2014). The interesting lack of overlap in our results is an indicator of the diversity of microbial environments, even within a single host system. Overall, significant differences in CTs throughout the hamster body demonstrate that there is a microbial gradient between tip and tail.
The composition of the gut microbiota affects host behavior (Desbonnet et al., 2014; Dinan and Cryan, 2013; Foster et al., 2016), although the mechanisms by which this occurs have not been fully characterized (Capuco et al., 2020; Cryan and Dinan, 2012; Mayer et al., 2014). Our results suggest that the microbiota plays a significant role in seasonal changes in aggression. We found various relationships between individual OTUs and aggression. The 10 OTUs that were significantly correlated (
Although the overall model for duration of aggression indicated an intermediate difference between the SDfl and SDfs groups, it is interesting to note that whereas 4 hamsters displayed LD-like non-aggression, 2 hamsters displayed higher duration more typical of SD. Interestingly, bacteria from 3 orders were significantly enriched in the two aggressive SDfl hamsters but were not enriched in the four SDfl hamsters that exhibited the reversed (non-aggressive) phenotype. Although we cannot firmly establish the relationships between these bacteria and aggression from the relatively small sample sizes in the present study, particularly due to the correlative nature of these results, these results indicate a need to further explore the roles of these bacteria in behavior. Using observed and simulated OTU data, we identify bacterial taxa that are likely to be associated with aggression, but additional work is needed to verify. One of these elevated bacteria was the genus
It is interesting to note that
Although we did not specifically investigate the effects of
Disturbance of the gut microbiota and its metabolome also elicits changes in host physiology and behavior (Carding et al., 2015; Clarke et al., 2014; de Palma et al., 2014). Perturbation of the microbiota, or dysbiosis (Tamboli et al., 2004), is characterized by an imbalance in the enteric gut flora (Hughes et al., 2018). Dysbiosis has been increasingly linked to pathologies, including neurodegenerative (Hirschberg et al., 2019; Raval et al., 2020; Spielman et al., 2018), mood (Dinan and Cryan, 2013; Foster and McVey Neufeld, 2013; Huang et al., 2019), gastrointestinal (Carding et al., 2015; Chang and Lin, 2016; Saffouri et al., 2019), and immune system (Clemente et al., 2018; Levy et al., 2017; Petersen and Round, 2014) disorders. The relationship between dysbiosis and disease indicates that the microbiota may play a significant role in the establishment and maintenance of host health. Furthermore, the microbiota shifts with age (Odamaki et al., 2016; O’Toole and Jeffery, 2015; Rinninella et al., 2019), photoperiod (Bailey et al., 2010; Ren et al., 2020; Shor et al., 2020), and time of day (Liang and FitzGerald, 2017; Paulose et al., 2016; Teichman et al., 2020). These temporal shifts suggest that changes in host physiology, health, and behavior across developmental stages and life-history events may be at least partly regulated by cyclical changes in the gut microbiota. Our current results support the hypothesis that seasonal fluctuations of behavior are related to temporal fluctuation of the microbiota.
In this study, we transplanted hamsters with gut communities that were either similar (LDfl and SDfs groups) or significantly different (LDfs and SDfl groups) from their own. The results indicate a more robust response in aggressive behavior to microbiota transplant in SD-housed hamsters compared with those in LD. One explanation for this difference may be related to differences in the susceptibility of the different phenotypes to dysbiosis. It is possible that there is a greater resistance to dysbiosis in LD-housed hamsters because maintaining gut homeostasis may provide selective advantages relating to socialization, reproduction, and/or health during spring and summer. As with immunity and reproduction, resistance to dysbiosis may thus be regulated as a seasonal trade-off.
Despite increasing evidence that the gut microbiota significantly influences host behavior and physiology, it is important to note that communication between microbiota and host is bidirectional (Carabotti et al., 2015). This suggests that signals deriving from the host can influence gut community. Furthermore, the bidirectional nature of the microbiota-host relationship suggests that the actions of the microbiota on the host may be modulated by host-related factors, including genetics (Bonder et al., 2016), endocrine signaling (Neuman et al., 2015), and immune function (Zhang et al., 2017). The groups of hamsters in this study were inherently different: half expressed the summer-like LD phenotype, and half expressed the winter-like SD phenotype. As such, the microbiota transplants were introduced into 2 different phenotypic backgrounds. In addition, the FMTs were transplanted into 2 different gut environments—we did not use a germ-free rodent model, so the transplanted bacteria would also have initially interacted with seasonally different communities of bacteria. We did not investigate the photoperiod-dependent intermicrobial interactions, nor did we investigate how the seasonal physiologies differed in terms of host-gut communication mechanisms.
Although our results are consistent with a multitude of studies showing that fecal microbiota transplant alters the gut microbiota, and that the gut microbiota does impact the host, there is a need for further investigation of the host’s role in regulating the microbiota. However, the current results do indicate that manipulation of the gut microbiota induces shifts in behavior, as well as opening a new avenue of investigation regarding seasonal regulation of aggression. In sum, this study represents a new tool to explore photoperiodism and behavior, and it contributes toward a better understanding of the relationships between microbiota, host, and environment.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304221092105 – Supplemental material for Bacteria and Bellicosity: Photoperiodic Shifts in Gut Microbiota Drive Seasonal Aggressive Behavior in Male Siberian Hamsters
Supplemental material, sj-docx-1-jbr-10.1177_07487304221092105 for Bacteria and Bellicosity: Photoperiodic Shifts in Gut Microbiota Drive Seasonal Aggressive Behavior in Male Siberian Hamsters by Elyan K. Shor, Shawn P. Brown and David A. Freeman in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors are grateful for the support provided by members of the Department of Biological Sciences, Center for Biodiversity research, and Animal Care facility at the University of Memphis. Funding for this research was provided by the Department of Biological Sciences at the University of Memphis.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
