Abstract
Parents make tradeoffs between care for offspring and themselves. Such a tradeoff should be reduced in biparental species, when both parents provide parental care. However, in some biparental species, the contribution of one sex varies greatly over time or between pairs. How this variation in parental care influences self-maintenance rhythms is often unclear. In this study, we used continuous video recording to investigate the daily rhythms of sleep and feather preening in incubating females of the Northern Lapwing (
Sleep and preening address basic animal life requirements (Van Iersel and Bol, 1957; Steinmeyer et al., 2010), and animals devote a lot of time to these self-maintaining activities (Connolly, 1968; Spruijt et al., 1992; Cotgreave and Clayton, 1994; Lesku et al., 2006). In most birds, a substantial part of the reproduction process involves time-consuming incubation. At the same time, bird parents need to forage, sleep, and take care of their bodies (Cotgreave and Clayton, 1994). As these activities may influence the risk of depredation, which is the most prevalent cause of nest failure (Ricklefs, 1969; Kubelka et al., 2018), incubating parents have to deal with a tradeoff between care for the offspring and the need for self-maintenance. The predation risk can be mitigated by subordinating the behavioral rhythms on the nest to the rhythm of predation pressure (Cervencl et al., 2011; Ekanayake et al., 2015), especially when both parents share the incubation duties (Komdeur and Kats, 1986; Weatherhead, 1990).
From a behavioral viewpoint, sleep is a temporary and rapidly reversible state of reduced susceptibility to surrounding stimuli with a restorative function (Siegel, 2003; Lima et al., 2005). Animals are more vulnerable to predation while they are sleeping (Lima et al., 2005; Lima and Rattenborg, 2007). In birds, unihemispheric slow-wave sleep may partially compensate for the reduced vigilance during sleep, because it allows birds to sleep with 1 eye open (Rattenborg et al., 1999; Rattenborg et al., 2000). At the same time, sleep makes individuals inconspicuous, and this may reduce detection by a predator (Lima et al., 2005; Lima and Rattenborg, 2007; Zimmer et al., 2011). Sleep on the nest can thus also be a defense tactic against a predator, particularly since the bird remains motionless and partially vigilant. Among birds, the length of sleep fluctuates widely from more than half of the day to a few hours per day, but also adaptive sleep loss during the reproduction period has been described (Roth et al., 2006; Lesku et al., 2012). Species differ in the extent to which they are flexible in the timing of their sleep (e.g., Hamilton et al., 2002; Chudzinska et al., 2013). Specifically, the timing of sleep during incubation may reflect not only the intensity of predation pressure but also the daily pattern of food availability (Meijer and Langer, 1995) and the ability or the willingness of the partner to guard the nest (Grønstøl, 2003).
Birds also need to spend a large proportion of their time using their bill to preen their feathers (Delius, 1988; Cotgreave and Clayton, 1994). This is necessary for feather maintenance, for distributing the preen wax, and for controlling ectoparasites (van Rhijn, 1977; Delius, 1988). Preening reduces the vigilance of an individual, and unlike sleep, it makes the individual much more visible to predators (Smith et al., 2012). The timing of preening is probably very flexible (Delius, 1988), unlike the timing of sleep (Randler, 2014), and this could enable individuals to schedule their preening during periods when it is less risky.
During biparental incubation, parents need to synchronize their activities to minimize the time for which the nest remains unattended (Bulla et al., 2016b; Sládeeˇek et al., 2019b). A large proportion of the off-nest time of each partner is necessarily spent foraging (Ashkenazie and Safriel, 1979; Grønstøl, 2003; Bulla et al., 2015); thus, the time for self-maintenance activities such as sleeping and preening may be limited. These activities might be carried out more on the nest during incubation. Consequently, when there is a substantial variation in the division of incubation duties between the parents, the contribution of the generally less care-giving partner can play an important role in the timing of self-maintenance of the incubating parent. In particular, a higher male contribution can enable the female to sleep and preen more while she is off the nest. She can therefore be more vigilant (sleep less) and less conspicuous (preen less) during incubation, at least during the peak activity of predators. However, studies on the rhythmicity of self-maintenance activities are scarce. To the best of our knowledge, no study has investigated the link between the self-maintenance rhythm of a care-giving parent and the daily rhythm of predation, or the link between the self-maintenance rhythm and the partner’s investment in parental care.
In this study, we used continuous video recordings to investigate the behavioral rhythms of incubating Northern Lapwing (
Specifically, we (1) investigated the timing of nest predation events within our population. Then we (2) tested whether sleeping and preening followed any daily rhythm and, if so, whether such rhythm was similar to the daily rhythm of predation pressure. Finally, we (3) tested whether females that were paired with more caregiving males slept and preened less during incubation, thereby changing the possible self-maintenance rhythm.
Methods
The study was conducted between March and June 2015 and 2016, in the Cˇeské Budeˇjovice basin, Czech Republic (49° 15′N, 14° 05′E). To assess the daily pattern of nest predation pressure, we used 33 cases in which Northern Lapwing nests were depredated within our study area and where the time of depredation was known. We further assumed that other ground-nesting waders in the study area experienced the same predation pressure (Macdonald and Bolton, 2008; Mason et al., 2018), and we therefore also included 17 cases of Little Ringed Plover (
We monitored the incubation on 55 nests of Northern Lapwing. Using small cameras placed approximately 1.5 m from the nest, we obtained continuous video recordings of 3 days (median, range: 1-5; Sládeček et al., 2019c). Since it takes an immense amount of time to extract the detailed behavior, we randomly chose a 1-day complete record for each nest (i.e., 24 h of uninterrupted recording).
We extracted behaviors from the recordings using Boris software version 6.3 (Friard and Gamba, 2016), with precision to within 1 s. First, we determined the beginnings and the ends of all incubation bouts, taken as the time when the bird stands on both legs in the nest. Within the pair, we identified the sex of the incubating bird, using a set of sex-specific plumage traits, for example, the crest length and the extent of melanin-based ornaments on the face and breast, which are well identifiable features (Meissner et al., 2013; Schonert et al., 2014).
Second, we extracted the beginning and the end of each sleeping bout. Of the 2 sleeping postures described elsewhere (Amlaner and Nigel, 1983; Dominguez, 2003; Gauthier-Clerc and Tamisier, 2012), the birds slept predominantly with the head turned backward and partly tucked between the shoulder coverts. In rare cases (i.e., ~2% of the sleeping bouts), the birds slept with their head forward (as during regular incubation) but drooping (see videos in the Supplementary Material). Note that while remaining in the sleeping position, the bird often opened 1 eye for a short time and scanned the surroundings. We interpret these periods as sleep, and we include them in the sleeping bouts, as this behavior seems to be connected with unihemispheric slow-wave sleep, which has frequently been reported in birds (Rattenborg et al., 1999; Rattenborg et al., 2000; Roth et al., 2006).
Third, we extracted preening, defined as rapid bill movements between the feathers and the preen gland (van Rhijn, 1977). Since preening is often performed in clusters of preening bouts lasting from 1 s to several seconds, interrupted by breaks of similar length, we extracted preening as an occurrence (“yes”/”no”) of this behavior during each 30-s interval of incubation by the female.
Statistical Analysis
All procedures were performed in R version 3.5.0 (R Core Team, 2017). General linear models were fitted using the “lm” function, and general linear mixed-effects models were fitted using the “lmer” function from the “lme4” R library (Bates et al., 2015). For all model-based parameter estimates, we report the effect sizes as the median and the Bayesian 95% credible interval (95%CrI), based on the posterior distribution of 5000 values simulated by the “sim” function from the “arm” R library (Gelman et al., 2016).
To test the daily rhythmicity of the predation, we calculated the general linear model with the number of known predation events for each hour of the day as response variable. We used 2 mixed-effect models to explain the variation in female sleeping behavior. In the first model, the dependent (response) variable was the ratio of female sleep to the overall time for which she attended the nest within a particular hour (“sleep”). That is, the hours when a female did not incubate were excluded from this analysis. We weighted the model by the square root of the female incubation time during a particular hour. Note that an alternative approach, with the absolute time of sleep within an hour (i.e., regardless of female nest attendance per hour), yields similar results (Suppl. Table S1). In the second model, we used the length of the sleeping bouts as a response variable. To describe the variations in preening behavior, we used the number of preening records, divided by the overall time for which the female attended the nest within a particular hour (“preening”) as a response variable. For this analysis, we used only hours with more than 10 min of female incubation (to exclude possible extreme proportions of preening events during short video recordings), and we weighted the model by the square root of the female incubation time during a particular hour.
We used a similar set of predictors in all models. To test for the daily rhythmicity in a response, we transformed the time to radians (2 × time × π/period of supposed rhythmicity) and fitted the sine and the cosine of the radians (Bulla et al., 2016a). As the period of rhythmicity, we used either a 24-h cycle or a 12-h cycle, based on a general pattern visualized from the raw data (Suppl. Figs. S1, S2; supplementary actograms in Sládeček et al., 2019a). Moreover, because birds (and also predators) probably react to actual changes in the light, we included in each model a binomial predictor indicating whether it was “day” or “night” during a particular hour. An hour was assigned as “night” when the sun was more than 6° below the horizon for more than one half of the hour, and vice versa. As a measure of the male contribution to incubation, we used the proportion of male nest attendance in a given day (i.e., 24 h). In all models, we also included the interaction between the male incubation effort and the time of day.
Further, to avoid misinterpretations of the results, we defined and tested the effect of potentially confounding variables, that is, the date within the season on which the nesting started, the time within the incubation period at which the video recordings were made, and 2 weather variables, temperature (daily means) and precipitation (daily sums). Measurements from Cˇeské Budeˇjovice (H. Zajícˇková, Czech Hydrometeorological Institute, Cˇeské Budeˇjovice) were used for both weather variables. Since neither of these predictors has a substantial effect on sleep (Suppl. Table S2a, b) or preening (Suppl. Table S2c), we did not include these predictors in the models presented in the main text. The full models including the effects of these predictors are presented in Supplementary Table S2a-c.
All continuous predictors, except for the time of day, were included in all models z-transformed (mean centered and divided by the standard deviation; Schielzeth, 2010). In all models, we fitted nest identity as a random intercept; time predictors were included as random slopes (Schielzeth and Forstmeier, 2009).
Results
Daily Variation in Predation
Nests were depredated almost exclusively at night; that is, 42 of 50 depredation events occurred when the sun was >6° below the horizon (Fig. 1a; Table 1a). In addition, the video-recorded predators were mammals only: 15 Red Foxes (
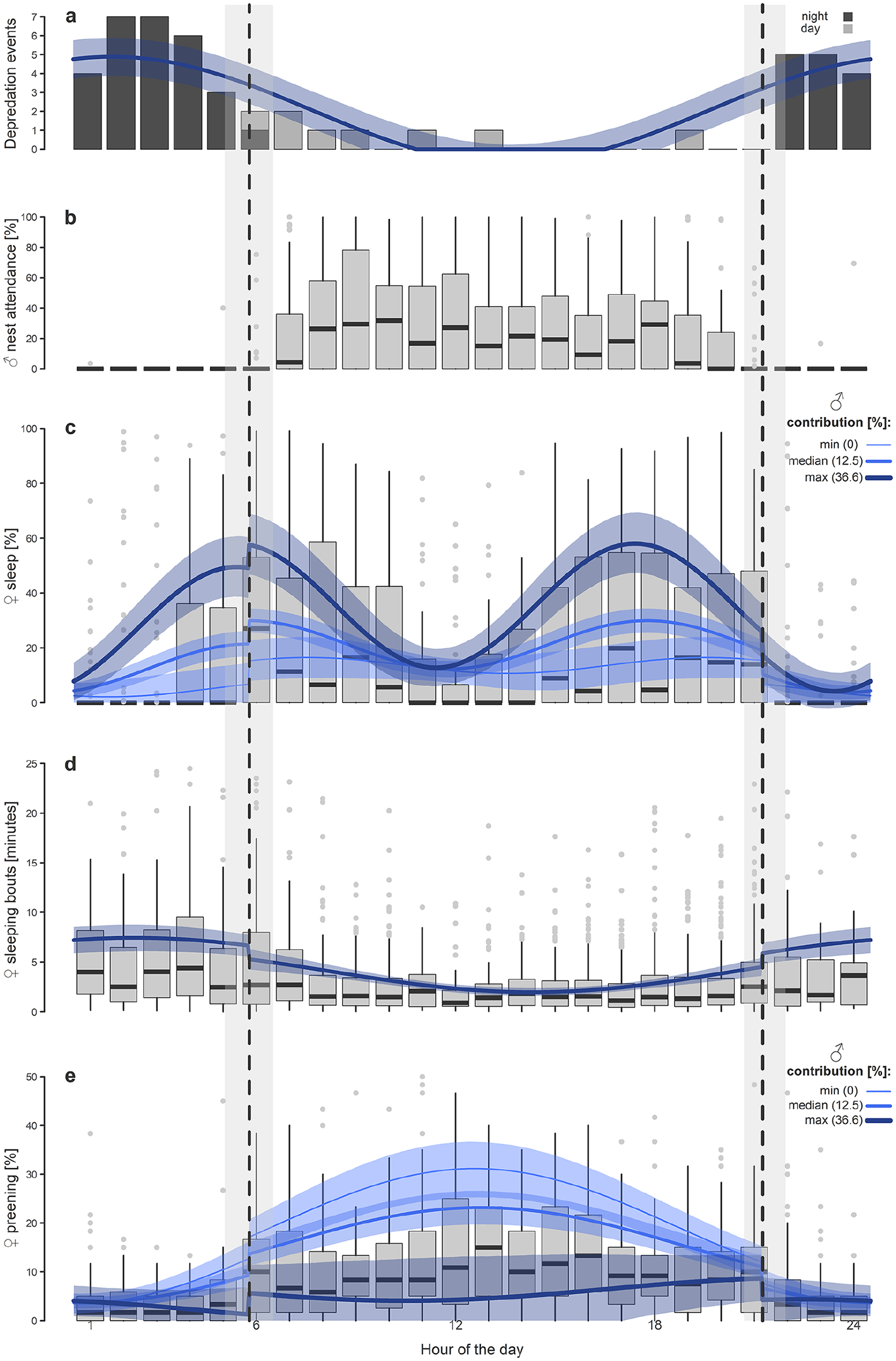
(a) Daily variation in nest predation. Bars depict the number of predation events in particular hours in the study area. (b) Daily pattern of male contribution to incubation. (c) Daily variation in female sleep. (d) Daily changes in the length of female sleeping bouts. (e) Daily variation in female preening behavior. Percentages in (b), (c), and (e) refer to the proportions within the given hour. Boxplots (b-e) depict the median (horizontal line inside the box), 25th to 75th percentiles (box), 25th and 75th percentiles minus or plus the 1.5× interquartile range, respectively, or the minimum and maximum value, whichever is smaller (whiskers), and outliers (circles). Curves (a, c, d, e) with shaded areas indicate the model prediction, with 95%CrIs based on the joint posterior distribution of 5000 simulated values from the model outputs (Table 1) and generated by the “sim” function in R (Gelman and Hill, 2007). Note that fits in (c-e) also include the effect of the night (i.e., whether sun was >6° below the horizon or not), which causes skips in the fit. Male contribution to incubation in (d) has been set to the mean value. Vertical dashed lines indicate the median and gray polygons indicate the range for the beginning and end of the dark part of the day (i.e., when the sun was >6° below the horizon).
Predation pressure in relation to time of day.
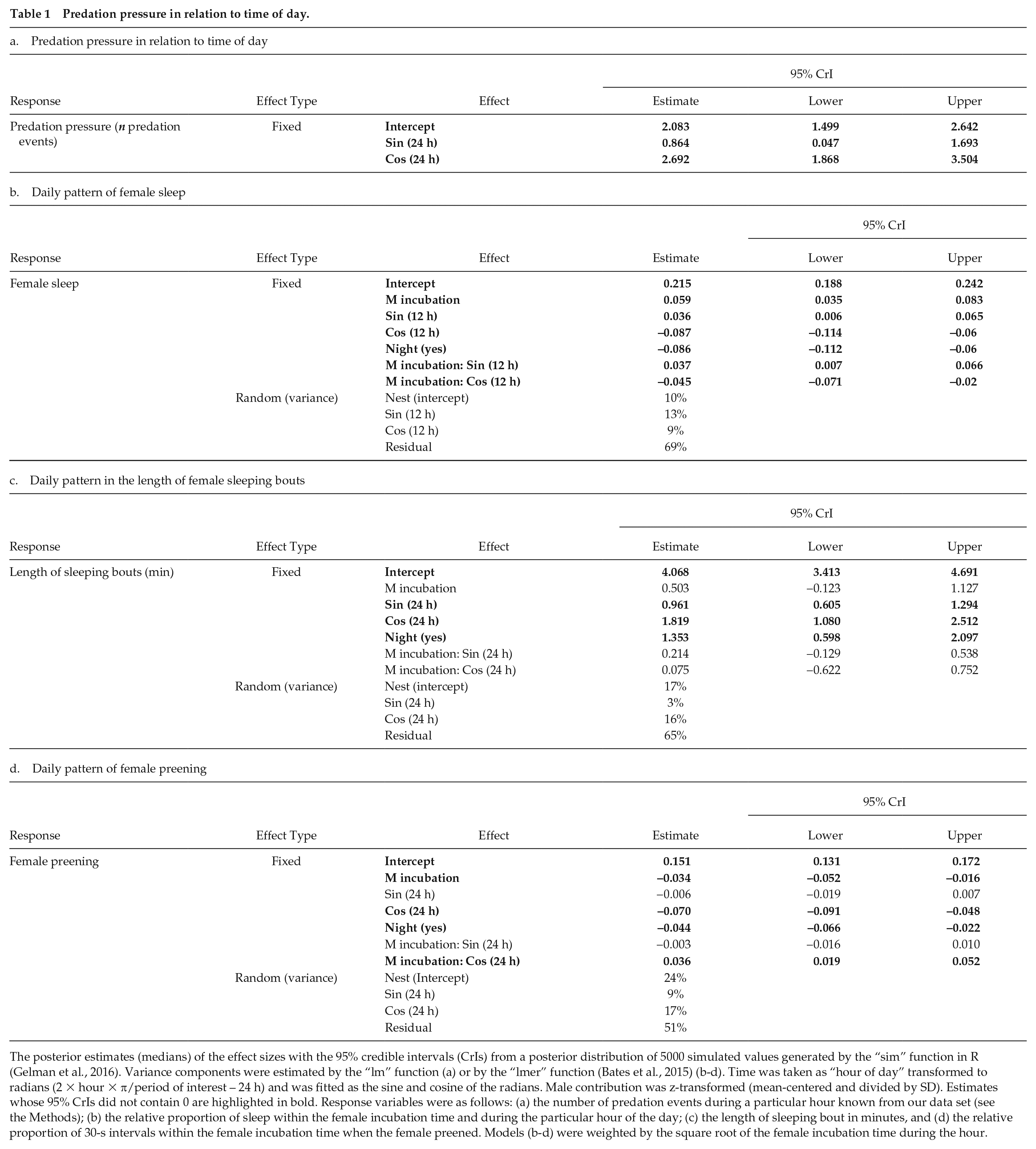
The posterior estimates (medians) of the effect sizes with the 95% credible intervals (CrIs) from a posterior distribution of 5000 simulated values generated by the “sim” function in R (Gelman et al., 2016). Variance components were estimated by the “lm” function (a) or by the “lmer” function (Bates et al., 2015) (b-d). Time was taken as “hour of day” transformed to radians (2 × hour × π/period of interest – 24 h) and was fitted as the sine and cosine of the radians. Male contribution was z-transformed (mean-centered and divided by SD). Estimates whose 95% CrIs did not contain 0 are highlighted in bold. Response variables were as follows: (a) the number of predation events during a particular hour known from our data set (see the Methods); (b) the relative proportion of sleep within the female incubation time and during the particular hour of the day; (c) the length of sleeping bout in minutes, and (d) the relative proportion of 30-s intervals within the female incubation time when the female preened. Models (b-d) were weighted by the square root of the female incubation time during the hour.
Nest Attendance
In total, we monitored 55 nests and extracted the incubation record of a random complete day (24 h) for each nest. Within this time, the females spent 17.8 ± 2.7 h on incubating (mean ± SD, range: 6.5-21.8 h), while the males spent 2.0 ± 1.6 h on incubating (mean ± SD, range: 0-6.4 h), with clear preference for daylight incubation (Fig. 1b). Thus, the nests were not attended by either of the parents for 4.1 ± 2.3 h (mean ± SD, range: 0.8-14.7 h). For a detailed description of the Lapwing incubation pattern, see Sládeček et al. (2019c).
Sleep
Incubating females slept on their nests for 3.0 ± 1.8 h of 24-h days (mean ± SD, range: 13 min–7.0 h), which corresponds to 17.5% of their incubation time (mean, range: 1.2%-45%). The sleep was divided mostly into very short sleeping bouts, with a median length of only 1.7 min (range: 3 s–1 h; Fig. 1d, see also Suppl. Fig. S3 and supplementary actograms in Sládeček et al., 2019a). The between-female variation in the overall sleep length has been associated with the number of sleeping bouts, rather than with the length of the sleeping bouts (Suppl. Fig. S4). The daily sleeping rhythm was strongly bimodal, with maxima in the morning and in the late afternoon and minima in the middle of the day and especially at night (Table 1b; Fig. 1c; Suppl. Fig. S5). In addition, female sleep was associated with male contribution to incubation. Contrary to our hypothesis, females with more help from their mate slept more, and the bimodal ~12-h sleep rhythm was stronger than in females with little or no male help (Table 1b; Fig. 1c; Suppl. Figs. S1, S5).
The length of the sleeping bouts followed a daily rhythm, with the longest sleeping bouts during the night (median around midnight: 4 min) and the shortest sleeping bouts in the middle of the day (median around noon: 1 min; Table 1c; Fig. 1d). This pattern was not associated with the male contribution to incubation.
Preening
Preening of females on their nests occurred 122 ± 83 times per 24 h (median ± SD, range: 7-398), which corresponds to a 6.7% median probability that preening occurs within a 30-s interval of female incubation. However, the median probability of preening during a 30-s interval ranged from 0% to more than 27% between females.
The preening behavior followed a daily rhythm, with the maximum in the middle of the day (median after midday: 21.7%) and the minimum at night (median after midnight 1.7%; Table 1d; Fig. 1e). In contrast to sleep, the more the male helped the female with incubation, the less the incubating female preened (Table 1d; Fig. 1e; Suppl. Fig. S6). Also, the females receiving more help had a weaker daily rhythm of preening (Table 1d; Fig. 1e; Suppl. Fig. S2, S6).
Discussion
With the use of continuous video recordings of breeding Northern Lapwings, we revealed a strong daily rhythmicity in the self-maintenance behavior of incubating females. Female sleep showed a bimodal ~12-h rhythm, while female preening showed a unimodal 24-h rhythm. Self-maintenance was suppressed particularly during the night, when the predation risk for our population is the highest. We also revealed that the male contribution to incubation correlated with the intensity of female self-maintenance. Specifically, an increased male contribution to incubation was associated with a stronger sleep rhythm and a weaker preening rhythm of incubating females.
Dynamics of Behavioral Rhythms
The 3 self-maintenance traits—sleep, length of sleeping bouts, and preening—followed 3 different rhythms. Whereas the sleep peaked at dawn and before sunset, the sleeping bouts were longest during the night. In contrast, preening showed a unimodal pattern, with the maximum around noon. In other words, both sleeping and preening were suppressed during the night hours, when the predation pressure was also highest. In fact, predation risk has been suggested as an important driver of the timing of sleep and preening in various bird and mammal species (Randler, 2005; Amo et al., 2011; Javůrková et al., 2011).
Our findings suggest that incubating Northern Lapwing females strived to be vigilant at night. In contradiction with our results, previous studies on the Mallard (
If lack of female sleep during the night may protect females against predation, what stands behind the sleeping peaks after sunrise and before sunset and the lack of sleep around noon? We provide 2 explanations that are not mutually exclusive. First, the females may need to sleep after and before the long sleepless nights. Second, the morning and afternoon sleep peaks may be another form of antipredator tactic, this time against daytime predators. Most predators that are active during the daylight (e.g., corvids and hawks) have peak activity similar to the peak sleeping activity of incubating female lapwings (Fig. 1a; Rutz, 2006; Roth and Lima, 2007) and usually use sight to detect their prey at a long distance. Notably, a male partner in lapwings is often on the watch. Thus, for an approaching visual daytime predator, a vigilant nonincubating partner will always be easier to detect than a sleeping (immobile) bird incubating the eggs. Moreover, the nonincubating vigilant parent emits a loud warning sound whenever he (or she) detects or actively deters approaching predators during the daylight hours (Elliot, 1985a; Kis et al., 2000), giving enough time for the sleeping bird on the nest to react. In addition, daytime predators can be detected at a great distance, so incubating birds can change their behavior in time, if necessary. Thus, sleeping on the nest during daylight may be an appropriate combination of antipredator tactics and self-maintenance. However, why females prefer preening to sleep during noon remains unclear. One explanation might be that predators are scarce around noon, and females can thus perform other activities associated with conspicuous movement on the nest, such as preening, egg turning, improving the nest lining, and feeding, which it is useful to perform at the time of lower predator activity. Noon is also the time when incubation attendance drops (Sládeček et al., 2019c).
Although night sleeping bouts were generally rarer than daytime sleeping bouts, the night sleeping bouts were paradoxically somewhat longer (median ~4 min) than the daylight sleeping bouts (median ~2 min). As Dukas and Clark (1995) suggest, the continuous vigilance of birds during the night may not be sustainable for the whole night, thus perhaps explaining why sleep-deprived birds occasionally fall into longer sleep bouts.
We found that the proportion of night predations (in the dark) was 84% (42 of 50 depredation events). Although the dynamics of the behavioral rhythms provide a good reflection of the pattern of real predation events, we can consider that a principal driver for changing the sleep behavior during incubation may be a switch in the light conditions, which can play the role of a proxy for the perception of predation risk. It is if the female cannot see the predator in the dark, she will resist the sleep. Note that the binomial predictor day/night was an important factor in all of our models. However, females started to sleep even before the end of the night, that is, before the approach of daylight (sunrise), while their sleep sharply ended after twilight (dusk; Table 1b; Fig. 1c). Interestingly, the Red Fox has a very similar pattern to the start and end of female sleep from March to May (i.e., a sharp drop in the dark before sunrise as well as peak activity shortly after dusk; Kämmerle et al., 2020). Thus, it is possible that the direct experience with dominant predators at the local level can adjust the general effect of light conditions on the sleep rhythm during incubation.
This high predation risk during the night multiplied by the inability to detect a predator over a long distance might be a crucial cause of using different antipredator behavior on the nest between night (vigilance) and daylight (sleep). Further research should therefore investigate whether reduced night vigilance or reduced sleep of incubating parents after sunrise and before sunset may increase nest and/or adult depredation.
Effect of Male Incubation Effort
Our study provides correlative evidence that the contribution of males to incubation is linked to the self-maintenance behavior of their female incubating partners. In contradiction to our hypothesis, females that were paired with more caregiving males slept more during incubation and also during the night. The male contribution to incubation is perhaps a proxy for general male investment; that is, males that incubate more are also more vigilant and active in defending the nest, patrolling around the nest, giving warning calls, and chasing away predators (Cramp and Simmons, 1983; Elliot, 1985a, 1985b; Kis et al., 2000). Indeed, Northern Lapwing females with a slightly greater off-nest sleeping time paired with males that incubated more (Grønstøl, 2003). If a male’s defensive activity correlates with his willingness to incubate, females paired with more territory-defending males may be better protected and could afford to be less attentive. Such division of parental roles may be an example of social synchronization, which has only recently been described in wild animal populations (Bulla et al., 2016b; Leniowski and Wȩgrzyn, 2018).
Since our analysis covered a single randomly selected day from the incubation period of each nest, we cannot exclude the possibility that females with an extremely low amount of sleep slept more during the previous days or would sleep more in the subsequent days. However, incubation patterns, such as incubation attendance and male contribution, remain almost consistent in the course of the incubation period and are highly repeatable in the Northern Lapwing (Sládeček et al., 2019c). In addition, the sleeping effort changed little over the incubation stage and season (Suppl. Table S2a). Notably, biological rhythms such as sleep also remain repeatable in other species (Steinmeyer et al., 2010; Stuber et al., 2015; Stuber et al., 2016). Whether the sleep-deprived females compensate for the sleep deficit and thus sleep more during subsequent days deserves further investigation.
Sleep deprivation can have a negative influence on attention, motivation, memory (Rolls et al., 2011; Vyazovskiy et al., 2011), and reproductive output (Potdar et al., 2018). Note that females with little or no help from their partner can hardly compensate for the lack of sleep during their off-nest time, because they are off-nest for only 10% to 15% of the time, during which they need to forage. In addition, the Northern Lapwing seems to spend a negligible proportion of the off-nest time sleeping (Grønstøl, 2003). Note that lack of sleep over extended periods of time (even weeks) during reproduction has also been reported in the territory-defending Pectoral Sandpiper (
In accordance with our hypothesis, females paired with more caregiving males spent less time preening on the nest. This suggests that these females can preen when they are off the nest. The reduced preening on the nest likely reduces movement on the nests and may increase vigilance, which can reduce nest detectability by predators. However, whether the time spent preening on the nest increases nest predation risk awaits future testing.
Conclusion
We have revealed different daily rhythms in the sleep and preening of incubating Northern Lapwing females. Their sleep followed ~12-h periodicity associated with sunrise and sunset, which corresponds with predator activity patterns. In contrast, preening followed a 24-h rhythm, with a peak in the middle of the day, when predation pressure was lowest. Interestingly, the intensity of the rhythms was modulated by the male contribution to incubation. Females paired with more contributing males had a stronger sleep rhythm and, conversely, a weaker preening rhythm. How the modulation of daily self-maintenance rhythms in species with biparental care affects reproductive success and individual fitness awaits further investigation.
Supplemental Material
Supporting_information – Supplemental material for Daily Rhythms of Female Self-maintenance Correlate with Predation Risk and Male Nest Attendance in a Biparental Wader
Supplemental material, Supporting_information for Daily Rhythms of Female Self-maintenance Correlate with Predation Risk and Male Nest Attendance in a Biparental Wader by Kateřina Brynychová, Miroslav E. Šálek, Eva Vozabulová and Martin Sládeček in Journal of Biological Rhythms
Footnotes
Acknowledgements
We would like to thank Hana Vitnerová, Vojteˇch Kubelka, Jana Hronková, Tereza Kejzlarová, and many others for their help in the field. Bart Kempenaers and Martin Bulla from the Max Planck Institute for Ornithology lent us some of the field equipment. We are grateful to Kristýna Nohejlová for her help with the incubation extraction. We thank Martin Bulla for his enormous assistance in revising the manuscript and an anonymous reviewer for his valuable comments. We thank Robin Healey for his kind language assistance. This work was funded by IGA FZP (20184221) to K.B. and M.E.Š. and by CIGA (20184210) and CIGA (20184208) to M.S., M.E.Š., and E.V.
Author Contributions
M.S., K.B., and M.E.Š. conceived the study, M.S. and E.V. collected the incubation data and extracted the incubation behavior from the recordings, K.B. extracted other behavioral data from the recordings, M.S. analyzed the data, M.S., K.B., and M.E.Š. wrote the manuscript.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the current laws of the Czech Republic.
Availability of Data and Materials
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
