Abstract
CRYPTOCHROMES (CRYs) are structurally related to ultraviolet (UV)/blue-sensitive DNA repair enzymes called photolyases but lack the ability to repair pyrimidine dimers generated by UV exposure. First identified in plants, CRYs have proven to be involved in light detection and various light-dependent processes in a broad range of organisms. In Drosophila, CRY’s best understood role is the cell-autonomous synchronization of circadian clocks. However, CRY also contributes to the amplitude of circadian oscillations in a light-independent manner, controls arousal and UV avoidance, influences visual photoreception, and plays a key role in magnetic field detection. Here, we review our current understanding of the mechanisms underlying CRY’s various circadian and noncircadian functions in fruit flies.
Genetic and Molecular Identification of Drosophila CRY as a Circadian Photoreceptor
CRYPTOCHROMES (CRYs) have evolved from photolyases, which are responsible for the repair of ultraviolet (UV)–generated pyrimidine dimers in a wide range of organisms (Sancar, 2008; Michael et al., 2017). CRYs and photolyases share a very similar domain that binds a flavin adenine dinucleotide (FAD) cofactor, the photolyase homology region. While photolyases bind a second chromophore such as a pterin that serves as a light-harvesting cofactor, CRYs do not appear to bear a second chromophore (Selby and Sancar, 2012). In photolyases, light activation of the FAD cofactor, either through direct photon absorption or Förster resonance energy transfer from the activated second chromophore, triggers a series of redox reactions that culminate with the repair of pyrimidine dimers (Sancar, 2008). The discovery of 2 CRYs as a novel class of proteins in Arabidopsis thaliana (Ahmad and Cashmore, 1996, 1993; Hoffman et al., 1996) implicated in blue-light responses but lacking DNA repair activity sparked the search for similar proteins in animals and other organisms. The identification of mammalian CRY1 and CRY2, expressed in the ganglionic cell layers of the retina, suggested that non–rhodopsin-based photoreception might occur in animals and could be involved in circadian entrainment (Hsu et al., 1996; Miyamoto and Sancar, 1998). Moreover, in fruit flies, it was known for decades that visual photoreception was dispensable for entrainment of circadian rhythms and that the nonvisual photoreceptor was sensitive to blue light (Frank and Zimmerman, 1969; Zimmerman and Goldsmith, 1971). In addition, the then-recently discovered light-dependent TIMELESS (TIM) degradation (Hunter-Ensor et al., 1996; Zeng et al., 1996; Myers et al., 1996), which appeared critical for circadian entrainment, was triggered by blue light specifically (Suri et al., 1998; Yang et al., 1998). The identification of Drosophila CRY as the blue-light circadian photoreceptor resulted from 2 converging approaches, reverse and forward genetics. First, the emerging Drosophila genome-sequencing project yielded a sequence with high homology to mammalian CRY1/2 (Emery et al., 1998). This cry gene proved to be under circadian control, its protein product was degraded after light exposure, and CRY overexpression caused flies to be hypersensitive to light pulses of low intensities. Second, a mutant was identified, crybaby (cryb), that showed severe circadian photoresponse defects: no light-dependent TIM degradation and no ability to shift the phase of circadian behavior in response to short light pulses (Stanewsky et al., 1998). The causative mutation was found to alter a key FAD binding residue. Another striking phenotype of cryb mutant flies was their circadian blindness to constant light: cryb flies remain rhythmic in constant light, while wild-type flies become rapidly arrhythmic because of constant TIM degradation (Emery et al., 2000a).
These results, and the many studies that followed, have clearly established that CRY is the primary circadian photoreceptor in Drosophila. However, visual photoreception contributes to entrainment of circadian behavior (Stanewsky et al., 1998; Helfrich-Förster et al., 2001). Recently, Rhodopsin 7 (Rh7) was found to be expressed in circadian neurons and to participate in circadian entrainment as well (Ni et al., 2017; Senthilan et al., 2019; Kistenpfennig et al., 2017). It is also important to mention that there are 2 CRY subfamilies in insects (Yuan et al., 2007). Type 1 CRYs, including Drosophila CRY, are light sensitive and likely important for circadian entrainment in all insects that carry them. Type 2 CRYs are more closely related to mammalian CRYs. The primary function of type 2 CRYs and mammalian CRYs is in the circadian molecular pacemaker that generates circadian rhythms, and they do not appear to have light-dependent circadian function, as was initially proposed based on the presence of mammalian CRYs in retinal ganglion cells (Kume et al., 1999; van der Horst et al., 1999; Reppert, 2007; Yuan et al., 2007; Merlin et al., 2013; Thresher et al., 1998; Miyamoto and Sancar, 1998). In fact, they are repressors of CLOCK/Bmal1 transcription, binding to PERIOD (PER) proteins. Drosophila do not have a type 2 CRY; it is TIM that binds to PER, and this dimer feedbacks on the per and tim genes by repressing the transactivator CLK/CYC, thus generating circadian oscillations in per, tim, and other clock-controlled gene expression (Zhang and Emery, 2012). Insects such as bees have only a type 2 CRY, whereas others such as butterflies have both (Yuan et al., 2007). However, mammalian/type 2 CRYs can absorb blue-light photons and undergo a flavin photocycle, but evidence that this ability influences their function in vivo remains limited (Hoang et al., 2008; Vieira et al., 2012; Foley et al., 2011; Gegear et al., 2010; Fogle et al., 2011). This review will focus on Drosophila CRY and its mechanism of action in the circadian clock, as well as its noncircadian functions.
Cell-Autonomous Mechanisms of Circadian CRY-Dependent Photoreception
Circadian clocks are found throughout the body of Drosophila, albeit not necessarily in all cells of a given organ. This is particularly striking in the brain, in which only about 150 neurons of 100,000 express circadian clock genes (Plautz et al., 1997; Nitabach and Taghert, 2008). Interestingly, organs can be isolated in culture and are able to entrain autonomously to light (Plautz et al., 1997). This tissue-autonomous entrainment is mediated by CRY (Emery et al., 2000b; Levine et al., 2002a; Agrawal et al., 2017). CRY binds directly to TIM in a light-dependent manner to trigger TIM’s proteasomal degradation (Busza et al., 2004; Naidoo et al., 1999; Fig. 1). It does so by recruiting an E3 ubiquitin ligase complex that contains JETLAG (JET), an F-box protein that recognizes TIM (Koh et al., 2006; Peschel et al., 2006; Peschel et al., 2009). TIM degradation exposes PER to destabilizing phosphorylation and E3 ligase recognition and thus to its own proteasomal degradation, thereby resetting the circadian pacemaker (Lee et al., 1996; Ko et al., 2002; Grima et al., 2002). Since rhythmic PER and TIM protein levels peak approximately in the middle of the night under a 12 h:12 h light/dark cycle (Marrus et al., 1996), early-night light pulses slow down the accumulation of PER and TIM and delay the phase of circadian oscillations, while late-night light pulses accelerate the loss of PER and TIM and thus cause phase advances. The photic phase response curve (PRC) for circadian behavior, with its phase delays during the early night and advances with late-night light pulses (Levine et al., 1994; Suri et al., 1998), can thus be explained quite well through a CRY-dependent cell-autonomous mechanism. The PRC is also shaped by mRNA levels, since they drop rapidly after Zeitgeber Time (ZT)15 and contribute to the subsequent decline in protein levels (Ashmore and Sehgal, 2003). A recent study has shown that the PRC for molecular rhythms in peripheral tissues is shaped similarly to that of circadian behavior, reinforcing the notion that cell-autonomous CRY photoreception and TIM/PER degradation explain how delays and advances are generated (Lamba et al., 2018). This notion is also supported by the early finding that if CRY is overexpressed only in the circadian pacemaker neurons that drive circadian behavior (the ventral lateral neurons [LNvs]), then circadian behavior will be hypersensitive to short light pulses. Moreover, if functional CRY is present only in these pacemaker neurons, phase delays and phase advances will be partially restored in cryb mutant flies (Emery et al., 2000b). Finally, CRY expression in only the eyes restores light-dependent TIM oscillations specifically in this tissue (Emery et al., 2000b).
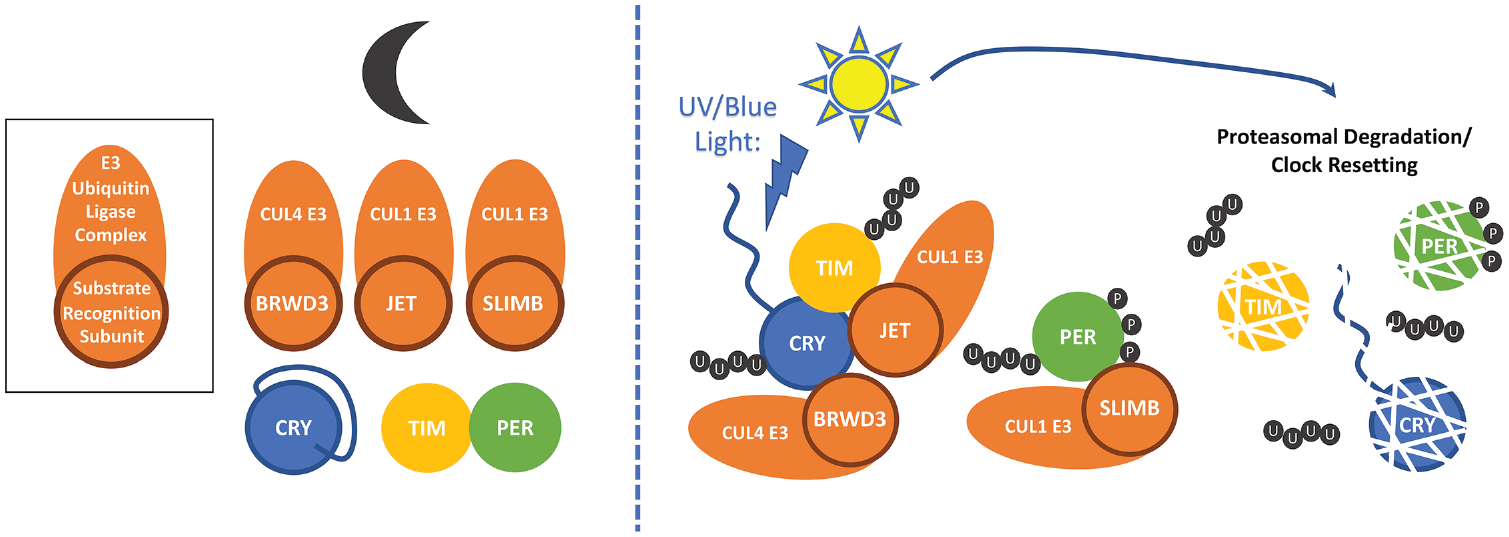
Light activation of CRY resets the circadian clock. In the dark (left), CRY’s C-terminal tail (CTT) occludes key binding sites for TIM and the E3 ligase subunits JET and BRWD3. TIM and PER are bound in a repressive complex. Light activation of CRY (right) causes a conformational change that involves release of the CTT and exposure of interaction surfaces. TIM associates with CRY and JET and is ubiquitinated (U) by the JET/CUL1 E3 ligase. CRY associates with the BRWD3/CUL4 E3 ligase and is ubiquitinated. PER is unbound from TIM and progressively phosphorylated (P). This promotes binding and ubiquitination by the SLIMB/CUL1 E3 ligase. Ubiquitination of TIM, CRY, and PER targets them for proteasomal degradation, resetting the clock.
What does light do to CRY to allow it to bind to TIM? The study of CRY mutant proteins (CRYM and CRY∆) that are missing the short C-terminal tail (truncation at amino acid 521 and 524, respectively) revealed that this domain is critical to modulate CRY function (Busza et al., 2004; Dissel et al., 2004). Indeed, in its absence, CRY binds to TIM whether the protein is exposed to light or not, while wild-type CRY binds to TIM only in the presence of light. Interestingly, CRY itself is degraded after light exposure through proteasomal degradation (Lin et al., 2001; Busza et al., 2004), which at least in Drosophila S2 cells is mediated by the DDB1 and CUL4 associated factor (DCAF) protein RAMSHACKLE/BRWD3 (Oztürk et al., 2013; Fig. 1). Confirming that this protein is indeed required for CRY degradation in vivo has proved challenging, as loss of BRWD3 is cell lethal. As is the case for TIM degradation, CRY degradation is also modulated by the C-terminal tail (Busza et al., 2004; Dissel et al., 2004). The most parsimonious explanation would thus be that CRY’s C-terminal tail masks a key interaction domain for TIM and BRWD3 (Fig. 1). Alternatively, it could bind a protein that masks this domain. Based on crystallographic studies followed by mutagenesis, the simplest explanation appears to be correct (Levy et al., 2013; Zoltowski et al., 2011; Czarna et al., 2013). Indeed, in CRY’s dark conformation, the C-terminal tail resides inside a groove that functions in the structurally closely related 6-4 photolyases as the active site for 6-4 photoproduct binding and repair. Moreover, limited tryptic proteolysis studies demonstrate that a light-induced conformational change in full-length CRY exposes residues that are also exposed in mutants missing the C-terminal tail, as well as an arginine residue within the C-terminal domain (Oztürk et al., 2011; Vaidya et al., 2013). Of note, these conformational changes have a long half-life (15 min), which could explain how CRY can integrate very low intensity of light over long period of time and thus trigger TIM degradation to phase shift circadian behavior (Oztürk et al., 2011; Vinayak et al., 2013). It is also interesting that CRY harbors both a negatively charged and positively charged surface. Since JET is basic while TIM is acidic, these 2 surfaces might be used to bring together JET and TIM in a light-dependent manner (Czarna et al., 2013). Thus, structural and in vitro biochemical studies fit strikingly well with previous molecular and genetic studies.
There is one additional interesting set of observations that was made while studying crym mutant flies (Busza et al., 2004). The CRYM protein is expressed at a very low level, because it is constantly degraded. Nevertheless, crym mutant flies still respond weakly to constant light and show reduced, but clearly detectable, responses to short light pulses. CRYM, however, binds to TIM whether in light or in darkness, and it would thus be predicted that it constantly triggers a low level of TIM degradation, irrespective of the presence of light. Supporting this idea, overexpression of CRY∆ indeed reduces TIM levels and lengthens circadian period under constant darkness, as if the flies were exposed to low constant light (Dissel et al., 2004). Thus, if CRYM is constantly in an active state, there should be no CRY-dependent circadian photoresponses left in crym flies, but this is clearly not the case. The implication is that light activation of CRY is needed for efficient TIM degradation even if CRY can bind to TIM irrespective of the presence of light. The mechanisms by which light promotes TIM degradation after CRY binding remain unclear. However, it is clear that CRY conformational changes are not limited to the C-terminal domain (Oztürk et al., 2011; Vaidya et al., 2013), and these additional conformational changes might be necessary to bring JET and TIM into the correct position for TIM to be efficiently ubiquitinated.
As mentioned above, photolyases repair DNA through redox reactions involving the FAD catalytic cofactor. FAD needs to be fully reduced for DNA repair activity, and this is achieved through light-induced electron transfer involving 3 tryptophans (the TRP triad; Byrdin et al., 2003). For Drosophila CRY, the ground state is indeed a fully oxidized FAD, while the active state is a semiquinone (Hoang et al., 2008; Berndt et al., 2007). The TRP triad of dCRY also appears to be important for light-dependent reduction of FAD (Oztürk et al., 2008; Lin et al., 2018; Zoltowski et al., 2011; Czarna et al., 2013). Unexpectedly, whether the TRP triad (or perhaps a tetrad with the addition of W394) is actually required for CRY and TIM degradation has been controversial (Lin et al., 2018; Froy et al., 2002; Oztürk et al., 2014). This might be because mutations of TRP residues are insufficient to completely eliminate FAD reduction or CRY function and that the exact conditions that were used in different studies (light pulse intensities or duration, cellular concentrations of CRY, TIM, and JET) might explain the different results that were obtained (Lin et al., 2018). Clearly, it would be important to generate mutants in vivo to address the function of these residues. CRISPR/Cas9-guided mutagenesis should provide a clean and efficient way to test the impact of specific CRY mutations on CRY and TIM photic degradation and circadian entrainment. An interesting recent set of observations indicated that the mechanisms of CRY and TIM degradation might be distinguished by specific mutations of a histidine residue located between the FAD and C-terminal tail (Ganguly et al., 2016). Indeed, the effect of such mutations is much stronger on TIM than on CRY degradation. Again, it would be interesting to assess the role of such mutations directly in vivo.
Non–Cell-Autonomous Mechanisms in CRY-Dependent Circadian Light Responses
As discussed above, the shape of the Drosophila behavioral PRC with phase delays before the middle of the night (ZT18) and phase advances afterward can be explained by the properties of tim and per mRNA and protein cycles and the cell-autonomous degradation of TIM by the photoreceptor CRY (Ashmore and Sehgal, 2003). This autonomous mechanism explains why most tissues in Drosophila are directly light sensitive (Emery et al., 2000b; Levine et al., 2002a; Agrawal et al., 2017); although, at least in Malpighian tubules (the fly’s kidneys), an additional and as yet unidentified photoreceptor might be present as well (Ivanchenko et al., 2001). In the brain, however, things are not as simple. As mentioned earlier, there are 150 neurons in the fly brain that express circadian genes (Nitabach and Taghert, 2008). They are named after their anatomical locations and their sizes and then subdivided based on neurotransmitter and neuropeptide content. The small ventral lateral neurons (sLNvs) control circadian behavior in constant darkness, imposing their pace on the other circadian neurons through secretion of the neuropeptide PDF (Renn et al., 1999; Stoleru et al., 2005; Yao and Shafer, 2014). These cells also control morning anticipatory behavior and are thus frequently referred to as M-oscillators (Stoleru et al., 2004; Grima et al., 2004). A subset of dorsal lateral neurons (LNds) along with a PDF-negative sLNv control evening anticipatory behavior and are referred to as E-oscillators (Stoleru et al., 2004; Grima et al., 2004; Rieger et al., 2006). Interestingly, rhythms in calcium levels and thus neuronal activity in M- and E-oscillators precedes (by about 4 h) the peak of morning and evening activity, respectively (Liang et al., 2016). There are also the large LNvs (l-LNvs), 3 groups of dorsal neurons (DN1, 2, 3), as well as 3 lateral posterior neurons. Among the approximately 150 circadian neurons, only about one-third of them expresses CRY (Yoshii et al., 2008; Agrawal et al., 2017). Yet even CRY-negative neurons can acutely degrade TIM after a 1-h light exposure (ZT1), which could be indicative of non–cell-autonomous CRY function (Yoshii et al., 2008). Furthermore, in CRY-negative LNds, a brief 5-min light pulse is sufficient to trigger TIM degradation, and this in a JET-dependent manner (Lamba et al., 2014). It should be noted, however, that at least in larvae, TIM degradation can also be triggered within an hour through visual inputs, and in adults, a recent study shows that all circadian neurons receive such input (Mazzoni et al., 2005; Li et al., 2018; Keene et al., 2011). Thus, visual input could contribute to TIM degradation in CRY-negative neurons. Further supporting the idea that non-cell-autonomous mechanisms might contribute to CRY photoreception was the observation that phase delays appeared to correlate better with the degree of TIM degradation in DN1s than in sLNvs after a short light pulse (Tang et al., 2010). Additionally, elimination of the l-LNvs specifically compromises phase advances (Shang et al., 2008). Rescue and RNAi experiments targeting JET, which is the protein directly responsible for TIM proteasomal degradation, show that light-dependent TIM degradation needs to occur both in M- and E-oscillators for proper phase delays and advances when the circadian neural circuit is intact (Lamba et al., 2014). However, apoptotic elimination or electrical silencing of either the M- or the E-oscillators does not compromise phase delays or advances, implying that either of them are sufficient if the others are eliminated or inhibited (Lamba et al., 2018). The most likely explanation for this apparent paradox is that the M- and E-oscillators are mutually coupled and exchange rhythmic signals. If either the M- or the E-oscillators fail to undergo acute TIM degradation and thus proper rapid resetting of their local clock, these rhythmic signals are desynchronized, and this compromises the ability of the M-E–oscillator subcircuit to resynchronize. This is supported by the fact that if the M-oscillator neuropeptide PDF is eliminated in flies that are defective for JET specifically in M-oscillators, normal phase shifts are observed, while when PDF is present, these phase shifts are severely disrupted (Lamba et al., 2018). Resetting of the whole circadian network follows resetting of the M/E subcircuit, since the M-oscillators control the circadian phase and pace of the whole circadian neuronal network in constant darkness (Stoleru et al., 2005). Interestingly, live imaging performed on dissected brains exposed to a short light pulse shows that M-oscillators and most circadian neuronal subgroups are acutely desynchronized, but the LNds (which include most E-oscillators) are the fastest group to reset to the phase that will ultimately be adopted by the whole circadian neural network (Roberts et al., 2015). This further supports the importance of non-cell-autonomous mechanisms underlying photic resetting of the circadian neural network. What remains unclear is the role played by non–cell-autonomous acute TIM degradation and its mechanisms. At least for the LNds (3 of 6 lack CRY), the evidence indicates that CRY-negative neurons receive input in a JET-dependent manner from CRY-positive neurons to trigger TIM degradation (Lamba et al., 2014). It is unclear at this point how CRY and JET trigger non-cell-autonomous TIM degradation. However, it should be noted that an acute increase in neuronal firing activity of PDF-positive LNvs, using the thermosensitive TRPA1 channel (Pulver et al., 2009; Hamada et al., 2008), promotes phase delays and advances in a PDF-dependent manner and causes Cullin-3 (CUL3)–dependent TIM degradation in E-oscillators (Grima et al., 2012; Guo et al., 2014; Eck et al., 2016). Perhaps a similar CUL3-dependent mechanism mediates non-cell-autonomous TIM degradation in response to light pulses.
In summary, the cell-autonomous properties of CRY-dependent TIM degradation explain how the circadian neural network can advance or delay its phase. Supporting this notion, the PRC observed with nonneuronal peripheral oscillators is virtually identical to that of circadian behavior (Lamba et al., 2018). However, the intricate connections between circadian neurons requires coordinated resetting of the local circadian clocks of the M- and E-oscillators to reset the whole circadian neural network and thus circadian behavior. More broadly, communication within the clock network might be needed for M-oscillators to integrate different modalities. Indeed, specific circadian neurons—in particular CRY-negative neurons—provide temperature input to the M-oscillators (Busza et al., 2007; Yoshii et al., 2010; Yadlapalli et al., 2018), and the M-cells also receive (directly or indirectly) visual and olfactory cues (Li et al., 2018; Helfrich-Förster et al., 2001; Levine et al., 2002b). It remains unclear how and why loss of large LNvs specifically affects phase advances (Shang et al., 2008). In addition, some DNs can play a role in phase advances when the E-oscillators are missing (Lamba et al., 2018). It is therefore possible that under specific conditions (internal or external), neurons beyond the M- and E-oscillators can modulate circadian behavioral photoresponses in a time-dependent manner.
CRY’s Light-Independent Circadian Function
As discussed above, Drosophila are missing a type 2 CRY, which, when present in other organisms, is critical for circadian pacemaker function. However, there are indications that Drosophila CRY can play a light-independent role in the control of circadian oscillations, at least in specific tissues. Indeed, molecular and physiological rhythms observed in the antennae (olfaction) are disrupted in CRY mutant flies (Krishnan et al., 2001). Furthermore, CRY is required to maintain robust molecular rhythms in Malpighian tubules in constant darkness (Ivanchenko et al., 2001). It should also be noted that behavioral locomotor rhythms tend to be of lower amplitude in cry0 flies at low temperature, but they are normal at 25 °C or 29 °C (Dolezelova et al., 2007). Thus, CRY can contribute to the maintenance of circadian oscillations in constant conditions. The mechanism for this is not entirely clear, but results in cell culture and in fly eyes indicate that CRY can contribute to PER repression of CLK/CYC activity, at least when overexpressed (Collins et al., 2006). However, it is clear that CRY is dispensable for rhythmicity in DD when PER and TIM levels are measured in whole head protein extracts (in which the eyes contribute to most of the PER and TIM signal (Zeng et al., 1994)) after temperature entrainment (Stanewsky et al., 1998). CRY is also dispensable for circadian cuticle deposition rhythms (Ito et al., 2008). CRY’s light-independent role in the circadian pacemaker might thus be limited to specific tissues or specific environmental conditions.
CRY’s Role in Regulating Neuronal Activity
CRY also plays important roles independent of the circadian molecular pacemaker. Mechanistically, the best understood of these noncircadian functions might be the regulation of arousal and UV avoidance. This work stemmed from a very surprising electrophysiological observation: the electrical activity of large LNvs is stimulated by light in a CRY-dependent manner (Sheeba et al., 2008). Subsequent studies indeed revealed that CRY’s photoreceptive properties are critical for this increased activity, although the C-terminal tail is not required to modulate CRY activity of neuronal firing, further supporting the notion that the photolyase homology region itself undergoes functionally relevant conformational changes. In addition, TIM was not involved in this process (Fogle et al., 2011). Actually, CRY regulates neuronal activity through hyperkinetics (HK), which is a potassium channel regulatory subunit with a putative redox sensing domain (aldo-keto reductase domain or AKR; Fogle et al., 2015). Strikingly, altering the redox status of the LNvs genetically or pharmacologically compromises CRY-dependent changes in l-LNv activity, and the HK redox sensing domain is required for its function in the photic modulation of l-LNv activity. HK binds to Ether-A-Gogo (EAG) potassium channel family members. In the case of LNv activation, the relevant members are EAG and EAG-related-gene (ERG). Thus, a model would be that upon blue-light activation, CRY activates HK through redox reactions and thus modulates HK/EAG and HK/ERG channels to depolarize the large LNvs’ membrane potential and increase firing activity (Fig. 2). This might be achieved through decreased kinetics of channel inactivation and increased K+ current based on studies with the Shaker channel (Pan et al., 2008; Weng et al., 2006). This would hasten repolarization and subsequent firing. Whether CRY and HK directly interact is not known, but potential biophysical mechanisms for their coupling have been proposed (Hong et al., 2018).
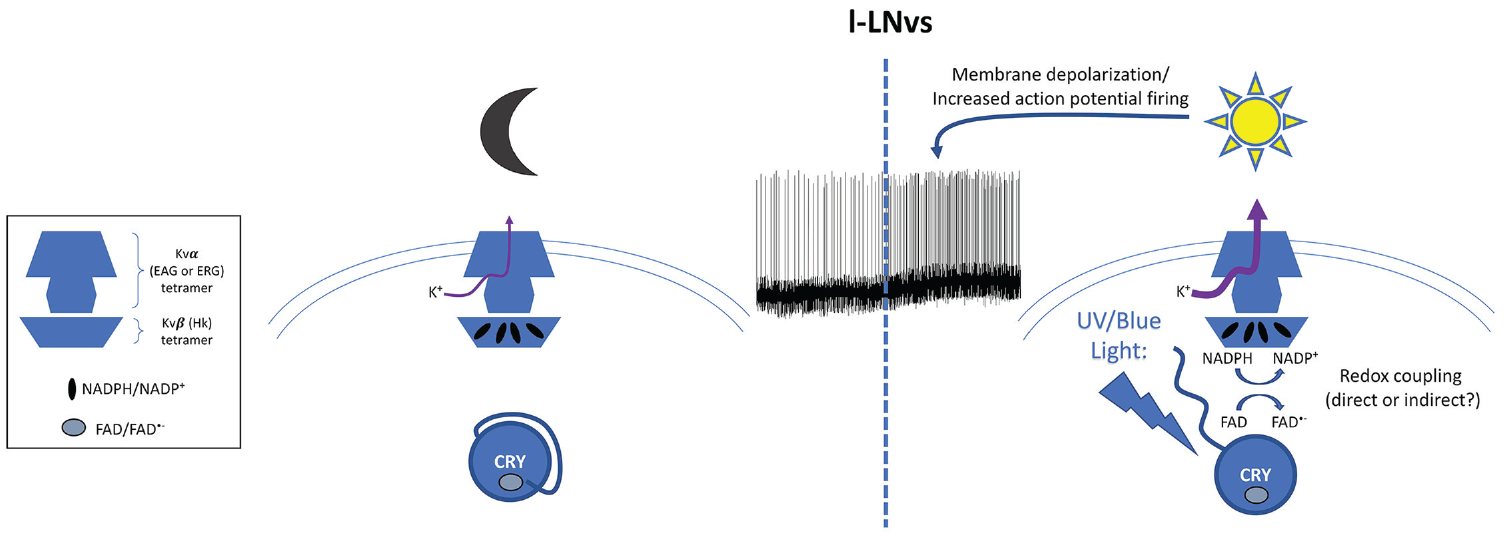
Model for CRY’s light-dependent regulation of firing rate of the large ventral lateral neurons. In the dark (left), CRY is inactive, and its cofactor flavin adenosine dinucleotide (FAD) is in the oxidized state. The potassium channel
What is the impact of this CRY-dependent regulation of neuronal activity? The l-LNvs promote arousal, and indeed, cry mutant flies show defective arousal upon exposure to blue light (Baik et al., 2017). Moreover, both HK and CRY defective flies show compromised arousal to UV light pulses and UV avoidance (Baik et al., 2019; Baik et al., 2018; Baik et al., 2017). Thus, a possible role for modulation of l-LNvs activity is the avoidance of noxious light exposure. Surprisingly, a similar CRY-dependent mechanism is involved in maintaining membrane physiology in larval salivary glands, which do not express other circadian genes (Agrawal et al., 2017). Curiously, even though EAG and HK are involved (as well as Shaker [SH], which HK can bind to; Wang and Wu, 1996), CRY’s role in controlling electrical input resistance is light independent. In addition, CRY levels are not light sensitive in larval salivary glands. CRY activity and levels are thus regulated very differently there than in other tissues, and it will be critical to understand how salivary gland CRY functions in a light-independent manner.
CRY’s Impact on Visual Photoreception
It was recently recognized that besides playing a role in the entrainment of the local circadian clocks in visual photoreceptors, CRY can also interact with proteins critical for visual photoreception. Indeed, CRY binds in a light-dependent manner to Inactivation No Afterpotential D (INAD), a scaffolding protein for complexes involved in visual phototransduction (Mazzotta et al., 2013). This interaction is mediated by CRY’s C-terminal domain, which contains PDZ interacting motifs (Hemsley et al., 2007) and a region of INAD that contains 2 PDZ domains. CRY also binds to F-actin and localizes to the rhabdomeres (the site of light detection in visual photoreceptors) in a light-independent manner (Schlichting et al., 2018). Surprisingly, CRY levels do not change in rhabdomeres in the presence of light, but they do so in the cell body of visual photoreceptors. This means that either the E3 ubiquitin ligase complex ubiquitinating CRY in a light-dependent manner (and containing BRWD3 probably, see above) is excluded from the rhabdomeres or it is unable to bind CRY there, perhaps because CRY is bound to INAD.
Flies defective for CRY show reduced motion vision and phototaxism, decreased ability to adjust the amount of locomotor activity as a function of light intensity during the day, and weaker behavioral entrainment under red light/dark cycles (Schlichting et al., 2018; Mazzotta et al., 2013). Since CRY does not detect red light, its role in the eyes for entrainment is independent of CRY activation. Thus, CRY function in the rhabdomeres might be structural, perhaps helping to properly align proteins in the phototransduction cascade through its interaction with F-actin. However, the role of the light-dependent INAD-CRY interaction is not yet clear. In addition, CRY in the photoreceptors’ cell bodies is required for entrainment of the local circadian clocks, which then affect rhythms in visual sensitivity and motion detection (Mazzotta et al., 2013; Emery et al., 2000b). Finally, in visual photoreceptors, CRY can also interact in a light-dependent manner with Bruchpilot (BRP), a key presynaptic active zone protein, and regulate its level (Damulewicz et al., 2017). CRY might thus also affect synaptic connectivity or synaptic transmission in the visual system.
CRY as a Putative Magnetoreceptor
How animals detect the Earth’s magnetic field is very poorly understood. Two main hypotheses have been proposed. The first is the use of a magnetic iron oxide crystal called magnetite, which is found in many species from magnetic field sensing bacteria to vertebrates (Ritz et al., 2010). The second is based on the sensitivity of electron spins of radical pairs to the magnetic field, which would affect the activity or conformation of a protein (Ritz et al., 2010). Cryptochromes, interestingly, have the basic properties to function as radical pair magnetic field sensors (Ritz et al., 2010; Müller and Ahmad, 2011). Using a learning paradigm in which flies are trained to associate a strong magnetic field with food, it was found that cry mutant flies show poor learning performance (Gegear et al., 2008). It was also shown with this assay that blue/UV light, to which CRY is sensitive, is required for magnetic field learning. This indicates that CRY either is able to sense the magnetic field or at least is part of a magnetosensing signaling cascade (perhaps also containing a putative magnetosensor called MagR; Qin et al., 2016). Interestingly, rescue experiments with human CRY2 indicate that this protein can substitute for Drosophila CRY and suggest that the magnetosensing function of cryptochromes could be conserved in mammals (Foley et al., 2011). Further supporting the idea that CRY can sense the magnetic field, it was shown that a CRY-dependent circadian light response (circadian locomotor behavior of long period under low-intensity constant light) is affected by magnetic fields and by the level and activity of CRY (Yoshii et al., 2009; Fedele et al., 2014a). Magnetic fields can also alter negative geotaxis and locomotor activity level, and Drosophila larvae avoid a weak pulsed electromagnetic field in a CRY-dependent manner (Fedele et al., 2014a; Fedele et al., 2014b; Sherrard et al., 2018). Moreover, magnetic field– and CRY-dependent seizures have been observed in Drosophila larvae (Marley et al., 2014), and the ability of CRY to promote neuronal activity of motoneurons (when ectopically expressed) is sensitive to the magnetic field (Giachello et al., 2016).
Thus, that CRY is implicated in magnetic field sensing is supported by multiple experimental approaches. However, conceptually, how CRY would provide directionality sensing and thus be helpful for magnetic field orientation remains unclear. It would have to be somehow organized in a geometric manner to provide directionality information. One possibility is that its expression in the rhabdomeres of the compound eye provide the needed geometry. One potential mechanism could involve the aforementioned light-dependent interaction between CRY and INAD in rhabdomeres (Mazzotta et al., 2013), which would be modulated by the magnetic field in a direction-dependent manner, affect the efficiency of phototransduction in specific photoreceptor cells, and thus result in a patterning “filter effect” of the field of vision, as proposed in the visual system of birds (Solov’yov et al., 2010). Interestingly, CRY expression in different visual photoreceptor cells can restore magnetic field suppression of negative geotactic responses, and flies without eyes are defective in this assay (Fedele et al., 2014b). However, antennae also appeared to be both necessary tissues and sufficient with CRY rescue. Finally, CRY expression in M- and E-cell circadian neurons together was sufficient as well. It seems that sensory input is required from both the antennae and the eyes for a normal behavioral response in this assay, but CRY expression in these tissues is not necessary; rather, CRY may mediate magnetic field–sensitive negative geotaxis independently from multiple sites. There are other geometric structures in the insect brains, such as the central complex. Interestingly, in butterflies, CRY2 is present in neuronal projections of these structures, which are known to play a critical role in flight orientation (Zhu et al., 2008). CRY is also detected in the central complex ellipsoid bodies of fruit flies (Yoshii et al., 2008). Clearly, much more work is needed to resolve the locus of CRY-mediated magnetoreception. Its molecular mechanism also needs to be revisited with improved approaches such as CRISPR/Cas9 mutagenesis, as rescue experiments with CRY mutant proteins have suggested that CRY-dependent magnetoreception might be (at least partially) independent of the classical TRP triad (Gegear et al., 2010; Fedele et al., 2014a).
Conclusion
Drosophila CRY’s functions are remarkably diverse. After being discovered for its crucial role as a circadian photoreceptor, CRY has now been implicated in the modulation of visual function, magnetoreception, and neuronal firing and can function both in a light-dependent or light-independent manner. Drosophila has proven to be a unique model organism to understand cryptochrome function, with its genetic arsenal and wide range of behavioral and physiological assays. The amazing progress made with genome editing now opens the way to rigorously test putative roles for cryptochrome in various animal species and to uncover novel functions. For example, establishing genetically that cryptochromes are involved in magnetoreception in vertebrates as has been proposed would be particularly important to understand seasonal migration as well as year-round orientation and navigation abilities. It would also be fascinating to determine the role of cryptochromes in marine organisms such as annelids or cnidarians, which use moonlight to coordinate reproduction (Levy et al., 2007; Raible et al., 2017). As mentioned earlier, genome editing is also a very potent approach to understand mechanistically the function of cryptochrome in vivo. There is thus little doubt that the coming years of research will transform our understanding of cryptochromes.
Footnotes
Acknowledgements
Patrick Emery’s laboratory is supported by a MIRA award from the National Institute of General Medicine Sciences (1R35GM118087).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
