Abstract
Most visual system functions, such as opsin gene expression, retinal neural transmission, light perception, and visual sensitivity, display robust day-night rhythms. The rhythms persist in constant lighting conditions, suggesting the involvement of endogenous circadian clocks. While the circadian pacemakers that control the rhythms of animal behaviors are mostly found in the forebrain and midbrain, self-sustained circadian oscillators are also present in the neural retina, where they play important roles in the regulation of circadian vision. This review highlights some of the correlative studies of the circadian control of visual system functions in zebrafish. Because zebrafish maintain a high evolutionary proximity to mammals, the findings from zebrafish research may provide insights for a better understanding of the mechanisms of circadian vision in other vertebrate species including humans.
Regulated by endogenous circadian clocks, visual system functions display robust day-night rhythms, which include opsin gene expression, neural transmission, light perception, and visual sensitivity (Foster et al., 1993; Argamaso et al., 1995; Okano and Fukada, 2001; Bellingham and Foster, 2002; Green and Besharse, 2004; Iuvone et al., 2005; Palczewski, 2012; McMahon et al., 2014). Cellular circadian rhythmicity is orchestrated by a set of core clock genes (e.g., Clock, Bmal1, Period, and Cryptochrome), the 24-h expression of which is regulated via interlocked transcription-translation feedback loops (Takahashi, 1995; Young and Kay, 2001; Reppert and Weaver, 2002; Cassone, 2014). In mammals, the master circadian clock is found in the suprachiasmatic nuclei of the hypothalamus, but additional clocks exist in other brain regions (Abe et al., 2002). In lower vertebrates (such as birds, salamanders, and fish), a preeminent clock is located in the pineal gland (Whitmore et al., 2000; Cahill, 2002; Pando and Sassone-Corsi, 2002; Cassone, 2014). The functional pathways of circadian clocks are highly conserved in vertebrate species (Cassone and Menaker, 1984; Schibler and Sassone-Corsi, 2002; Fukada and Okano, 2002; Cermakian and Sassone-Corsi, 2002; Panda, 2007).
While the circadian clocks that regulate the daily rhythms of animal behaviors reside in the forebrain and midbrain, in most vertebrate species, the retinas are also capable of generating self-sustained circadian oscillations (Reme et al., 1991; Terman et al., 1991; Berson, 2003; Besharse and McMahon, 2016). Retinal clocks directly regulate the circadian rhythms of visual system functions, such as photoreceptor outer segment disk shedding (Besharse et al., 1988; Bassi and Powers, 1990), retinomotor movement (Pierce and Besharse, 1985), retinal pigment epithelium granule migration (Bruenner and Burnside, 1986), and opsin gene expression (Korenbrot and Fernald, 1989; Pierce et al., 1993). In isolated frog retinas, for example, the expression of rhodopsin mRNA fluctuates between the day and night. In normal light-dark (LD) cycles, the expression is high in the late light phase (before lights-off) and low in the late dark phase (before lights-on). This pattern of fluctuation persists in animals kept in constant lighting conditions, suggesting that circadian clocks are involved in the regulation of rhodopsin gene expression (Korenbrot and Fernald, 1989). In isolated chick retinas, the expression of cone opsin genes also varies between the day and night (i.e., the expression is high in the late light phase and low during the dark phase). This rhythm persists in cultured retinal tissues in the absence of external timing cues, indicating that the circadian oscillators that regulate cone opsin expression are located in the retina (Pierce et al., 1993).
Zebrafish maintain a high evolutionary proximity to mammals (Itoh, 2007) and are suitable for research in developmental biology, physiology, neuroscience, and human disease (Appel, 2000; Grunwald and Eisen, 2002; Thisse and Zon, 2002; Fonseka et al., 2016). Zebrafish also offer a model for studying circadian biology. Zebrafish develop externally and rapidly, and the embryos are completely transparent. This provides a tool for easy molecular and cellular manipulation and time-lapse imaging, making it possible for real-time observation and quantification of the expression of circadian-related genes in live animals. In zebrafish, the circadian clock that regulates the rhythms of animal behaviors (e.g., motor activity) is found in the pineal gland (Whitmore et al., 2000; Cahill, 2002; Pando and Sassone-Corsi, 2002). Yet self-sustained circadian oscillations of gene expression and cellular function are also found in other tissues, such as the kidney, heart, spleen, and retina (Cahill, 1996; Whitmore et al., 1998; Kaneko et al., 2006; Yu et al., 2007; Huang et al., 2018; Nie et al., 2018). Zebrafish retinas share many similarities with human retinas, including opsin gene expression, photo transduction, synaptic formation, neurotransmission, and cellular organization (Klooster et al., 2009; Tarboush et al., 2012), which provide a tool for studying circadian vision in humans. This review will highlight some of the correlative studies on the circadian rhythms of visual system functions (e.g., rhythmic opsin expression, retinal neural activity, and behavioral visual sensitivity) that are mediated by retinal circadian clocks from research using zebrafish models. The findings from zebrafish research have been widely applied to other vertebrate species and have proven useful for research related to human vision (Whitmore et al., 2000; Li, 2001; Pando and Sassone-Corsi, 2002; Tamai et al., 2005; Idda et al., 2012; Blum et al., 2018).
Circadian Rhythms of Opsin Gene Expression
The Expression of Opsin mRNA Fluctuates between the Day and Night
Because the perception of light is mediated by visual pigment in the photoreceptor cells, it is conceivable that differential expression of opsin genes at different times of day and night might be attributed to some extent to daily fluctuations of visual system functions. Li et al. (2005) examined the expression of long-wavelength cone (LC) opsin mRNA at different times of day and night in zebra-fish using quantitative reverse transcription polymerase chain reaction. They found that under normal LD conditions (14:10 LD, lights-on at 0800 h and lights-off at 2200 h), the expression of LC opsin mRNA was lowest at 0400 h (4 h before lights-on) and increased steadily from lights-on over the next 10 h to its 60-fold higher peak at 1800 h (4 h before lights-off). LC mRNA expression was maintained at high levels until it began to decrease at lights-off at 2200 h, reaching its nadir again at 0400 h (Fig. 1A).
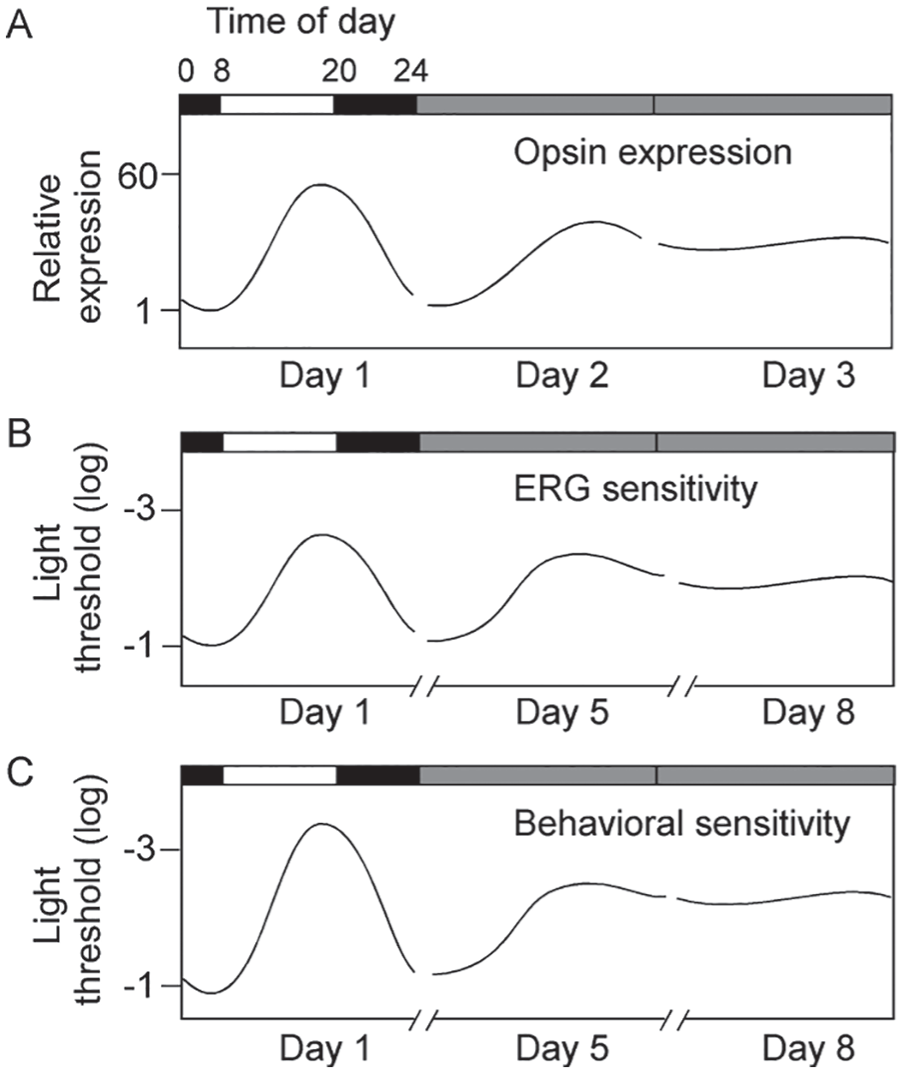
Diagrams showing the fluctuation of long-wavelength cone (LC) mRNA expression (A), retinal neural sensitivity (B), and behavioral visual sensitivity (C) in zebrafish kept in LD or DD. In LD, the expression of LC mRNA was lowest at 0400 h and highest at 1800 h. The rhythms persisted in DD for 1 day and then dampened out. The lowest expression at 0400 h was normalized to 1. Similarly, retinal neural sensitivity (determined by electrical physiological recordings from the cornea; see below) and behavioral visual sensitivity (determined by measuring the light threshold required to evoke visually-mediated escape responses; see below) fluctuated between the day and night. They were low at 0400 h and high at 1800 h. In both cases, the differences between the lowest and highest sensitivity levels were approximately 2 log units (100-fold).The lowest retinal neural sensitivity and behavioral sensitivity levels were normalized to log I = −1. The rhythms of retinal neural sensitivity and behavioral visual sensitivity persisted in DD for 5 to 7 days. Afterward, the rhythms dampened out and maintained at levels similar to those measured in the late light phase. Horizontal bars indicate lighting conditions: white, light phase; black, dark phase; gray, subjective day and night without light.
The fluctuation in LC opsin mRNA expression persisted in zebrafish that were kept in constant dark (DD), suggesting the involvement of circadian mechanisms in its regulation. However, the fluctuation in opsin gene expression was observed in DD for only 1 day, during which time the difference in gene expression between the subjective day and night was decreased. In the second day of DD, at all times of subjective day and night, the expression was maintained at a level approximately 40-fold higher than the previous expression at 0400 h (Fig. 1A).
The day-night fluctuation in LC opsin mRNA expression led to differential opsin protein expression. This was revealed by measuring the optical density of the outer segments of photoreceptor cells. Using micro spectrophotometry, Li et al. (2005) demonstrated that in red cone photoreceptor cells, the highest optical density of the outer segments was detected in the late light phase.
CLOCK Is Required for Circadian Expression of Opsin mRNA
Co-expression of core clock genes and opsin genes has been previously reported in retinal photoreceptor cells (Hayasaka et al., 2002; DeBruyne et al., 2007). Li et al. (2008) investigated the role of CLOCK in LC mRNA expression in zebrafish cone photoreceptor cells. They measured the pattern of LC mRNA expression in isolated red cone photoreceptor cells at different times of day and night while the expression of CLOCK was inhibited using clock sequence-specific morpholinos (see Thummel et al., 2011). In zebrafish retinas, the cone photoreceptor cells can be readily identified based on their cellular morphology and topographic arrangement (Raymond et al., 1995, 2014; Biehlmaier et al., 2003; Raymond and Barthel, 2004; Taylor et al., 2005). In morpholino-treated retinas, the expression of LC mRNA was detected in red cone cells at all times of day and night, but in DD, its expression was maintained at a constant level. This suggests that functional operation of the core circadian clock in photoreceptor cells is required for maintaining the circadian rhythm of opsin gene expression.
While the mechanisms underlying CLOCK-mediated circadian expression of LC opsin mRNA remain to be further studied, several lines of evidence suggest that CLOCK may exert its role on circadian opsin gene expression through cyclic adenosine monophosphate (cAMP)–mediated signaling transduction. First, cAMP is involved in opsin gene expression (Alfinito and Townes-Anderson, 2001; Yu et al., 2007). Second, the concentration of cAMP fluctuates between the day and night, and this pattern of fluctuation can be modified by shifting the LD cycles (Li et al., 2008). Third, when the expression of CLOCK is inhibited, the day-night fluctuation of cAMP concentration and LC mRNA expression is diminished (Li et al., 2008). Fourth, in CLOCK knock-down animals, the application of 8-Bromo-cAMP (a membrane-permeable cAMP analog) restores the circadian fluctuation of LC opsin mRNA expression (Li et al., 2008).
The Circadian Oscillators That Regulate Rhythmic Opsin Expression Are Located in the Retina
Using transgenic zebrafish Tg(rhodopsin::GFP), Yu et al. (2007) examined the oscillation of retinal circadian clocks. In the transgenic fish, the rod photoreceptor cells expressed green fluorescent protein (GFP) under the transcriptional control of the rhodopsin promoter. The GFP was modified with a short half-life time of approximately 30 min. Thus, rhodopsin promotor activities could be reflected by differential expression of GFP (i.e., when the promoter activity was high, brighter GFP would be observed, and when the promoter activity was low, dimmer GFP would be observed). By monitoring GFP expression, Yu et al. (2007) reported that in DD, the activity of the rhodopsin promoter fluctuated between day and night. The pattern of fluctuation, however, was different among individual photoreceptor cells. In some cells, the highest promoter activity was seen in the late subjective day, whereas in other cells, the peak activity was observed in the early subjective day (Fig. 2). The data suggest that in the retina, each photoreceptor cell may function as an independent circadian clock, but the oscillation of individual clocks is not synchronized. Yu et al. (2007) further demonstrated that via dopamine-mediated Ca2+ signaling transduction pathways, light transiently decreased rhodopsin promoter activity and reset the expression of the rhodopsin promoter to its baseline levels. Thus, the multiphasic circadian oscillations among individual photoreceptors could be synchronized.
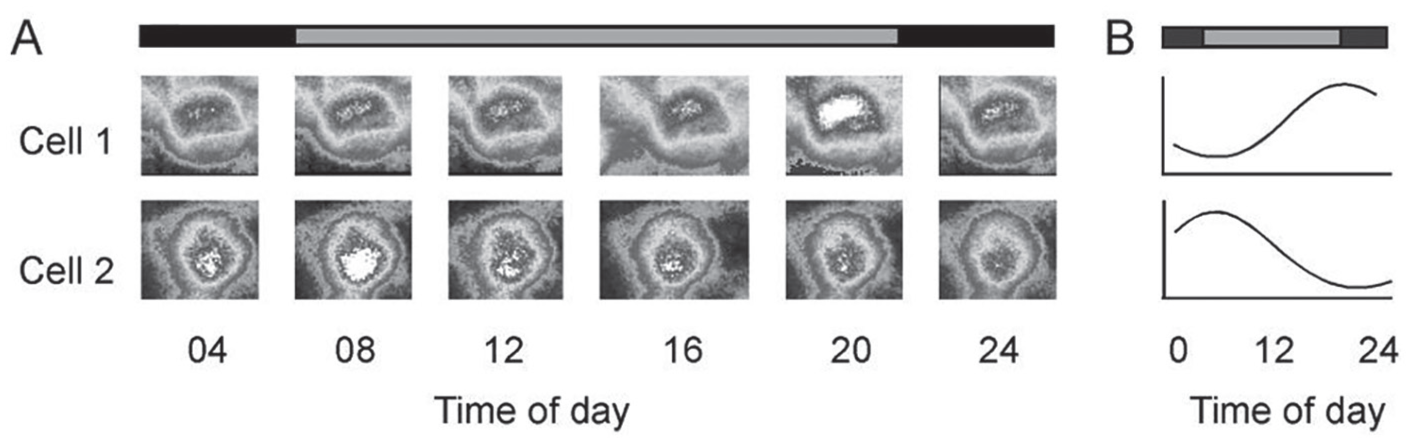
Circadian expression of the rhodopsin promoter in rod photoreceptor cells in DD. (A) Time-lapse images of rhodopsin promoter-driven green fluorescent protein expression at different times of day and night in 2 rod photoreceptor cells from the same retina. In cell 1, the highest expression was seen in the late subjective day, whereas in cell 2, the highest expression was observed in the early subjective day. (B) Diagrams of circadian oscillation of rhodopsin promoter activity in these 2 photoreceptor cells over 24 h in DD. The oscillations in these 2 cells were out of phase. Horizontal bars indicate lighting conditions: gray, subjective day; black, subjective night.
Circadian Rhythms of Retinal Function
Evidence of Circadian Rhythms in Retinal Sensitivity
In zebrafish, the functions of retinal cells can be evaluated in live animals by recording full-field electrical potentials from the cornea, known as the electroretinogram (ERG; Li and Dowling, 1997, 1998; Saszik et al., 1999, 2002; Saszik and Bilotta, 2001). The ERG consists of 3 major components: a negative a-wave followed by a positive b-wave in response to lights-on and a positive d-wave in response to lights-off. The a-wave is mainly derived from the photoreceptor cells, and the b- and d-waves are derived from secondary order cells, such as bipolar cells and glial cells (Kondo et al., 1998).
Li and Dowling (1998, 2000b) evaluated the circadian rhythms of retinal neural sensitivity in zebrafish by recording the minimum light intensities required to evoke threshold ERG responses (e.g., 20 µV b-wave amplitude) at different times of day and night. They recorded ERGs in dark-adapted zebrafish using either narrow-band red light (to measure cone-system sensitivity) or wide-spectrum white light (to measure rod-system sensitivity). In LD, when tested using either red or white light, the minimum light intensities required to evoke threshold ERG responses were high in the late dark phase (e.g., 0400 h, 4 h before lights-on) and low in the late light phase (e.g., 1800 h, 4 h before lights-off; Fig. 1B). This pattern of fluctuation persisted in DD, suggesting the involvement of circadian clocks. The fluctuation in cone- and rod-mediated threshold ERG responses persisted for 5 to 7 days in DD. Afterward, they dampened out and maintained constant levels at all times during the subjective day and night, similar to thresholds measured in the late light phase (Fig. 1B).
Evidence of Circadian Rhythms in Cone- and Rod-dominant Transition
In most vertebrate retinas, the photoreceptor cells are coupled through gap junction channels (Cook and Becker, 1995; Abd-El-Barr et al., 2009; Kihara et al., 2009; Asteriti et al., 2017). In zebrafish, the coupling between the cone and rod photoreceptor cells is regulated by circadian clocks (i.e., the junction is strong at night but weak in the day; Li et al., 2009). The cone- and rod-mediated visual pathways function differently at different times of day and night. In the daytime, retinal functions are largely regulated by the cone system; at night, they are mainly driven by the rod system (Nagy et al., 2014). The cone- and rod-dominant visual system functions may be shifted in response to alterations in lighting conditions, and the transitions between cone- and rod-dominant vision are regulated by gap junction channels (Anderson et al., 1986; Weiss et al., 1998; Xu and Yang, 2002; Burkhardt, 2011).
Ren and Li (2004a, 2004b) examined whether the transitions between cone- and rod-dominant vision in zebrafish are regulated by circadian clocks. This was done by monitoring the dynamic changes of ERG waveforms at different times during dark adaptation after bright-light adaptation. In early dark adaptation, when visual system functions are cone dominant, both the ERG b- and d-waves could be readily recorded. In late dark adaption, when rod-mediated retinal functions become dominant, the b-wave amplitude increased and the d-wave amplitude decreased. To quantitatively analyze the transitions between cone- and rod-dominant vision, Ren and Li (2004a, 2004b) recorded the time of dark adaptation at which the b-wave amplitude (microvolt) became 10 times greater than the d-wave amplitude (microvolt), referred to as Rb/d = 10. In LD, the transitions between cone- and rod-dominant vision took longer in the late dark phase (e.g., 0400 h) than in the late light phase (e.g., 1800 h). For example, when tested in normal LD, it took approximately 55 min to reach Rb/d = 10 at 0400 h but only 35 min at 1800 h. This pattern of timing was also observed in zebrafish kept in constant light (LL), and the pattern of fluctuation could be completely reversed when the animals were kept in reversed LD cycles. Together, the data suggest that circadian clocks are involved in the transition of cone- and rod-dominant vision.
Evidence of Circadian Rhythms in Behavioral Visual Sensitivity
Li and Dowling (1997) developed a behavioral assay, based on visually mediated escape responses, to measure the behavioral visual sensitivity in zebrafish. The test apparatus consisted of a transparent container surrounded by a rotating drum. The drum was illuminated by a light source from above, and the light intensity could be adjusted by changing neutral density filters. The drum could rotate in either clockwise or counterclockwise directions. The inside of the drum was covered by white paper marked with a black segment. The fish was allowed to freely swim in the container, and a post was placed in the middle of the container to prevent the fish from swimming through the center of the container. The swimming behavior was viewed via a TV monitor connected to an infrared video camera. Normally, the fish swam slowly along the wall of the container in either clockwise or counterclockwise directions. However, when challenged by the black segment rotating outside the container, the fish displayed robust escape responses (i.e., as soon as the black segment came into view, the fish immediately turned and rapidly swam away; Fig. 3).
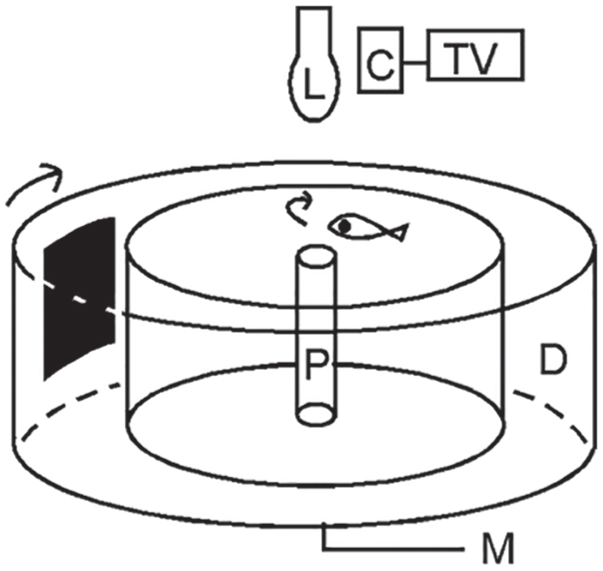
The apparatus for measuring behavioral visual sensitivity in zebrafish. Arrows indicate the direction of drum rotation (i.e., clockwise) and fish swimming (i.e., initially counterclockwise). The fish displayed escape responses to the approach of the black segment. Upon the black segment coming into view, the fish immediately turned and swam away (i.e., in the clockwise direction). Abbreviations: C, camera; D, rotating drum; L, light source; M, motor; P, post; TV, television monitor.
By measuring the minimum light intensities required to evoke the escape responses, Li and Dowling (1998, 2000b) evaluated the circadian rhythm of behavioral visual sensitivity in zebrafish. The experiments were performed under either background illumination conditions (to measure cone sensitivity) or in complete darkness (to measure rod sensitivity). In LD, under both test conditions, the minimum light intensities required to evoke the escape responses were high in the late dark phase (e.g., 0400 h) and low in the late light phase (e.g., 1800 h; Fig. 1C). This pattern of fluctuation persisted in DD, suggesting that circadian clocks are involved in the regulation of zebrafish behavioral visual sensitivity. After 5 to 7 days of DD, the fluctuations in behavioral cone and rod sensitivities were no longer evident. The merged sensitivity levels were maintained at constant levels, similar to those measured in the late light phase (Fig. 1C).
Modulation of Retinal Circadian Clocks
The Pineal Gland and Melatonin
In zebrafish, the pineal gland functions as a circadian pacemaker that regulates behavior (Kazimi and Cahill, 1999; Vuilleumier et al., 2006). The gland consists of photoreceptor cells that rhythmically synthesize and release melatonin (Cahill, 1996; Kazimi and Cahill, 1999). The function of the pineal gland is regulated by endogenous circadian mechanisms as well as by ambient LD conditions (Whitmore et al., 2000; Vatine et al., 2001; Idda et al., 2012; Laura et al., 2012; Lima-Cabello et al., 2014).
Li et al. (2012) tested the role of the pineal gland in the circadian rhythm of visual sensitivity in zebrafish. The research was conducted in transgenic zebrafish Tg(Gnat2:gal4-VP16/UAS::nfsB-mCherry), in which nitroreductase was expressed under the transcriptional control of an opsin promoter. In developing embryos and young adults, the transgene was expressed in both retinal and pineal photoreceptor cells. During aging, however, the expression of the transgene in retinal photoreceptor cells gradually diminished, whereas its expression in pineal photoreceptor cells persisted. By 8 months of age, nitroreductase was no longer expressed in retinal photoreceptor cells. This provided a tool for ablation of pineal photoreceptor cells; that is, after treatment with the antiprotozoal compound metronidazole, nitroreductases become active and destroy DNA in the nuclei of pineal photoreceptor cells (see also Pisharath et al., 2007; Curado et al., 2007; Montgomery et al., 2010). The treatment, however, does not affect the survival of retinal photoreceptor cells because the transgene is no longer expressed in the retina. Li et al. (2012) reported that in the absence of pineal photoreceptor cells, behavioral visual sensitivity levels measured using either narrow-band color light (to measure cone sensitivity) or wide-spectrum white light (to measure rod sensitivity) remained unchanged compared with control animals. In LD, fluctuations in cone- and rod-system sensitivities were observed; that is, the light thresholds required to evoke the behavioral escape responses were high in the late dark phase (e.g., 0400 h) and low in the late light phase (e.g., 1800 h). However, upon the animals being transferred to DD, the fluctuation in behavioral visual sensitivity was no longer evident. In DD, at all times during subjective day and night, cone- and rod-mediated visual sensitivities were maintained at constant levels, similar to those measured in the late light phase in control animals. This suggests that a circadian signal (e.g., melatonin) from the pineal gland is required to maintain the circadian rhythms of retinal cone- and rod-system sensitivity.
Light and Dopamine
Light produces the most dramatic effect on the expression of core clock genes and the circadian rhythms of visual system functions. In zebrafish, a short period of light treatment may sufficiently entrain the expression patterns of core clock genes (Carr et al., 2006; Hirayama et al., 2007; Ben-Moshe et al., 2014). Because functional expression of core clock genes is required for circadian expression of opsin genes, it is conceivable that alterations in core clock gene expression would interrupt the circadian pattern of opsin expression, which in turn would alter the circadian rhythms of visual system functions (see also Li et al., 2008). Light per se may also modulate the expression patterns of opsin genes. Different spectra of light may exert their effects on the circadian rhythm of opsin gene expression via different pathways. For example, blue light directly excites cryptochromes, which in turn activate protein kinase C- and mitogen-associated protein kinase-dependent intracellular signal transduction and alter the profile of opsin expression. In contrast, white light triggers dopamine-mediated cAMP-dependent protein kinase A and Ca2+ signaling pathways, which then modulate the expression of opsin genes (Yu et al., 2007).
Dopamine has been regarded as a light signal for visual system functions (Dowling, 1991; Morgan and Boelen, 1996; Yazulla et al., 1996), and alterations of dopamine content in the retina directly affect the circadian rhythms of visual system functions (Burnside, 2001; Mangel, 2001; Witkovsky, 2004). The release of dopamine is regulated by an endogenous circadian mechanism and light illumination (Dowling, 1991; Witkovsky, 2004). In zebrafish, light (particularly flickering light) increases the release of dopamine from retinal dopaminergic interplexiform cells (DA-IPCs; see Puppala et al., 2004). Li et al. (2005) examined the role of dopamine in the circadian rhythm of opsin gene expression in zebrafish. They found that dopamine transiently increased the expression of LC mRNA and thereby reset the pattern of its expression. Interestingly, the effect of dopamine on circadian opsin expression was observed only in the late dark phase, when the expression of opsin was low. When the experiment was repeated in the late light phase, no obvious changes in opsin expression were noted. This suggests that dopaminergic signaling is involved in the regulation of the circadian rhythm of opsin gene expression.
Li and Dowling (2000b) also demonstrated that dopamine is required for maintaining the circadian rhythm of retinal neural sensitivity. They recorded the minimum light intensities required to elicit threshold ERG responses at different times of day and night in zebrafish in which the DA-IPCs were depleted by treatment using neurotoxins. In DA-IPC–depleted animals, the absolute cone and rod sensitivities were similar to those measured in control animals, and their ERG waveforms appeared normal in response to light stimuli. However, when tested in DD, no fluctuations in ERG sensitivity between subjective day and night were observed. At all times, the light thresholds required to evoke ERG responses remained at a constant level, similar to the threshold measured in the late light phase in control animals. In DA-IPC–depleted zebrafish, a variety of other retinal functions, such as the course of light and dark adaptation and the transition of cone- and rod- sensitivity, were also interrupted (Li and Dowling, 2000b).
Olfactory Signals from the Olfacto-retinal Centrifugal Pathway
In all vertebrate species examined thus far (teleosts, reptiles, birds, rodents, primates), their retinas receive centrifugal feedback signals from the brain. Depending on the species, the centrifugal signals may originate from different parts of the brain, such as the isthmo-optic nucleus, pretectal cortex, thalamus, and olfactory bulb (Brooke et al., 1965; Demski and Northcutt, 1983; Zuker and Dowling, 1987). In zebrafish, the feedback originates from the terminalis neurons (TNs) in the olfactory bulb. The TNs express glutamate receptors and fire spontaneous action potentials (Ramakrishnan et al., 2010; Appelbaum et al., 2006; Wang et al., 2011). The TNs synthesize several types of neuropeptides, which include gonadotropin hormone–releasing hormone-like hormone (Li et al., 2017). The TN signals are transmitted to the retina through the olfacto-retinal centrifugal (ORC) pathway (Fig. 4). In the retina, the TN axons synapse with DA-IPCs (Li and Dowling, 2000a; Maaswinkel and Li, 2003).
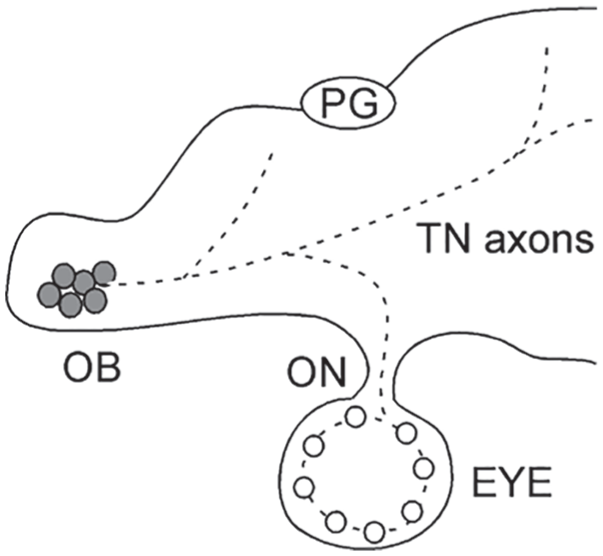
A diagram that shows the olfacto-retinal centrifugal pathway in zebrafish (lateral view, anterior is to the left). The terminalis neuron (TN) cell bodies (gray circles) are located in the olfactory bulb, and they project axons to the brain. Some of the TN axons enter the retina via the optic nerve. In the retina, the TN axons synapse with dopaminergic interplexiform cells (white circles). Dashed lines indicate TN axons in the brain and retina. Abbreviations: OB, olfactory bulb; ON, optic nerve; PG, pineal gland.
Maaswinkel and Li (2003) and Huang et al. (2005b) examined the role of centrifugal input in zebrafish visual system functions. This was done by recording the minimum light intensities required to fire action potentials in retinal ganglion cells (RGCs) or to evoke the visually mediated behavioral escape responses after odor stimulation. In those experiments, amino acids were chosen as the stimulus to activate the olfactory neurons (amino acids evoke robust olfactory responses in zebrafish; see Edwards and Michel, 2002). They found that in response to odor stimulation, the light thresholds required to elicit RGC and behavioral escape responses were decreased. Interestingly, the effects of odor stimulation on retinal and visual functions were only observed in the late dark phase (e.g., 0400 h), when visual sensitivity level was low. When the experiments were repeated in the late light phase (e.g., 1800 h), no obvious changes in RGC or behavioral thresholds were observed. This suggests that circadian clocks are involved in the modulation of centrifugal input in visual system functions.
When the ORC pathway was surgically severed or retinal dopaminergic signaling transduction was inhibited (e.g., DA-IPC depletion or intraocular injection of dopamine receptor antagonists), stimulation of the olfactory neurons produced no effect on RGC activity or behavioral visual sensitivity. Normally, dopamine inhibits the activity of inner retinal neurons, which include the RGCs (Umino et al., 1991). The mechanisms underlying odor-modulated visual system functions may entail the following: stimulation of the olfactory neurons increases glutamate release in the olfactory bulb and activates the TNs, which in turn increase ORC signal transduction to the retina. In the retina, because of the increase of TN input, the release of dopamine from DA-IPCs is decreased (Huang et al., 2005b). This lifts the inhibition of dopamine on RGCs and thereby increases retinal sensitivity (Wang et al., 2011; see also Li, 2019).
Discussion and Concluding Remarks
In zebrafish, brain signals from the pineal gland are essential for maintaining the circadian rhythms of visual system functions. In addition, self-sustained circadian oscillators in the retina are required for the function of circadian vision. The oscillators in the retina are capable of running autonomously, but they can be modified by a variety of factors, such as the expression of core clock genes, light, dopamine (Dowling, 1991), melatonin (Green and Besharse, 2004), and other local neural transmission, including that mediated by GABA and glutamate (Huang et al., 2005a; Moldavan and Allen, 2013). Via the ORC pathway, the retinal circadian oscillators are also influenced by centrifugal feedback signals from the olfactory bulb (Maaswinkel and Li, 2003; Huang et al., 2005b; Wang et al., 2011; Fig. 5).
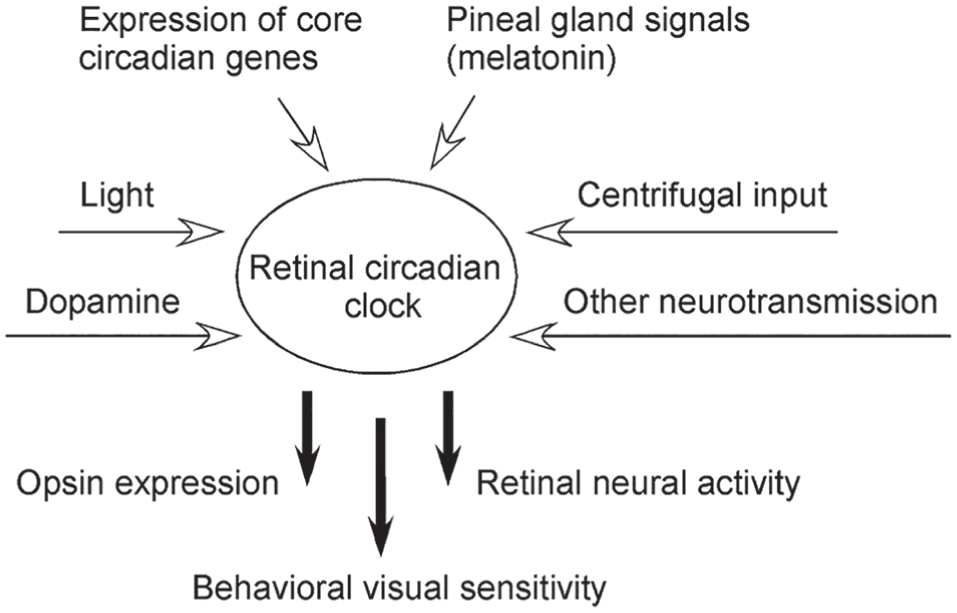
A summary diagram that shows the retinal circadian clock and signals that affect its function. Oscillations of retinal circadian clocks directly regulate the circadian rhythms of visual system functions, such as opsin gene expression, retinal neural activity, and behavioral visual sensitivity.
In zebrafish, circadian rhythms of visual system functions can be entrained by light. In addition to the cone and rod photoreceptor cells, several other cell types such as horizontal cells and intrinsically photosensitive RGCs express opsin-like proteins, which include vertebrate ancient long opsin and melanopsin (Kojima et al., 2000, 2008; Bellingham et al., 2002; Davies et al., 2011; Matos-Cruz et al., 2011). The expression of opsin-like genes may not participate in signal transduction pathways for regulating visual sensitivity, but it does play a role in maintaining circadian rhythms of visual system functions. The circadian rhythms of visual system functions may also be modulated by dopamine or odor stimulation but only in the late dark phase when opsin expression and retinal sensitivity are low. It is possible that under normal physiological conditions (e.g., in normal LD cycles), circadian clocks reduce some nonessential visual activities, perhaps for the purpose of energy saving (e.g., during the dark phase). After the animals have been maintained in prolonged darkness, visual system functions are increased (e.g., opsin gene expression, ERG sensitivity, and behavioral visual sensitivity). As a result, at all times, visual system functions are maintained at high levels similar to those measured in the late light phase.
In summary, this article provides a comprehensive review of the circadian rhythms of visual system functions in zebrafish. It has been demonstrated that both intrinsic and environmental cues may affect the circadian rhythms of visual system functions. In addition to the well-defined circadian clock in the pineal gland, oscillators in the retina also play important roles in the regulation of zebrafish visual system functions. Retinal clocks may reside in different cell types, such as photoreceptors, horizontal cells, and ganglion cells. The clocks run autonomously in 24-h cycles, but they are completely desynchronized. Light efficiently entrains the circadian rhythms of individual clocks. While the data presented in this article were mainly derived from research using zebrafish models, the underlying mechanisms may be applied to other vertebrate species including humans.
Footnotes
Acknowledgements
Some of the works described in this article were conducted in the author’s laboratory. The author’s research was supported in part by grants from the National Institutes of Health (R01EY13147) and the Department of Defense (W911NF-14-1-0555, W911NF-16-1-0316, and W911NF-18-1-0292).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
