Abstract
The onset and offset of an activity band in the circadian behavioral rhythm are known to differentially reentrain to shifted light-dark cycles (LD). Differential reentrainment could be explained by different light responsivities of circadian oscillators underlying these phase-markers. In contrast, reentrainment is accelerated by exposure to nonphotic time cues such as timed wheel-running. However, the relationship between the 2 oscillators and nonphotic acceleration of reentrainment is largely unknown. We examined phase-shifts of the mouse behavioral rhythm in response to an 8-h phase-advanced shift of LD and effects of behavioral interventions: maintained in a home cage (HC), exposed to a running wheel (RW) in HC (HC+RW), transferred to a new cage (NC), and exposed to RW in NC (NC+RW). Each intervention was given for 3h from the beginning of the shifted dark period and repeated for 4 days. Following the last dark period, the mice were released into constant darkness (DD). As a result, activity onset and offset were differentially phase-shifted. The activity onset on the first day of DD (DD1) was phase-advanced from the baseline slightly in HC and HC+RW, substantially in NC+RW, but not significantly in NC. The amount of phase-shift was significantly larger in the NC+RW than in the other groups. In contrast, the activity offset was significantly advanced in all groups by 6 to 8 h. The differential phase-shifts resulted in shortening of the activity band (α compression). The α compression was gradually relieved upon exposure to DD (α decompression), and the activity band finally became stable. Interestingly, the magnitude of phase-shifts of activity offset, but not of activity onset, in the following DD was negatively correlated with the extent of α compression in DD1. These findings indicate that the 2 circadian oscillators underlying activity onset and offset are involved in asymmetric phase-shifts and nonphotic acceleration of reentrainment.
Circadian behavioral rhythms in mammals are regulated by the pacemaker located in the suprachiasmatic nucleus (SCN), which entrains to light-dark (LD) cycles through the retinohypothalamic tract (Rusak and Zucker, 1979). The entrainment is best explained by phase-dependent phase shifts of the circadian clock, which is expressed as a phase-response curve (PRC) to light (Daan and Pittendrigh, 1976a). When an LD cycle is suddenly shifted by several hours, the circadian behavioral rhythm shifts its phase to reentrain to the new LD cycle. It usually takes several cycles to complete reentrainment.
Circadian behavioral rhythms in nocturnal rodents are characterized by dual activity bouts during the early and late dark period (Aschoff, 1954). A line of evidence indicates that the 2 bouts are regulated by different circadian oscillators with different sensitivities to light (Pittendrigh and Daan, 1976b; Honma et al., 1985; Elliott and Tamarkin, 1994). The oscillator underlying the activity onset has been called the evening oscillator and that underlying the activity offset the morning oscillator (Pittendrigh and Daan, 1976a). The phase relationship of the 2 oscillators determines the length of the activity band, the interval between the activity onset and offset. Researchers have frequently observed asymmetric reentrainment to a shifted LD cycle (Aschoff et al., 1975; Aschoff, 1978; Kleinknecht and Erkert, 1991). When an LD is phase-advanced, the activity offset shifts immediately, while the activity onset shifts gradually over a transient period of several cycles. The opposite is the case when an LD is phase-delayed. Asymmetric reentrainment could be ascribed to different light sensitivities of the 2 oscillators, but masking effects of the light period sometimes obscures this asymmetry (Aschoff, 1978).
The sensitivity of the circadian oscillator to light is expressed as a PRC. The PRC for a single light pulse in rats is different according to the phase reference (Honma et al., 1985). When the activity onset is used as a reference (onset PRC), the immediate PRC is unimodal, showing only a phase-delay portion; when the activity offset is used (offset PRC), the PRC is bimodal, showing both a phase-delay and a phase-advance portion. The phase-delay portion of the bimodal offset PRC could be a result of phase-delay shifts of the activity onset through an oscillatory coupling between the 2 oscillators. Whatever the mechanism might be, these PRCs predict asymmetric reentrainment; when light is introduced at the advance portion, the activity offset phase-advances immediately, while the activity onset phase-shifts depending on whether light hits the delay portion.
In contrast, several nonphotic treatments such as exposure to a running wheel are known to accelerate reentrainment (Mrosovsky and Salmon, 1987; Reebs and Mrosvsky, 1989a; Yamanaka et al., 2008). The acceleration is phase-dependent (Reebs and Mrosovsky, 1989b) and independent of the number of wheel revolutions (Yamanaka et al., 2016). However, the mechanism of acceleration by wheel-running is not yet elucidated. One possible mechanism is feedback from behaviors to the circadian clock (Aschoff et al., 1973). The free-running period in rats is much shorter when behavioral activity is measured by running wheel than by the so-called nonstress type actogram (Yamada et al., 1988). Timed exposure to a running wheel alters the circadian period of locomotor activity in mice and occasionally entrains the circadian rhythm (Yamanaka et al., 2013). Notably, physical activity is associated with several physiological processes such as changes in energy metabolism, sympathetic activity, and hormone secretion. To understand the behavioral feedback mechanism, we need to know which processes are associated with the feedback. Given the many similarities among the effects of timed wheel-running, restricted daily feeding, and methamphetamine-induced oscillation (MAO), we have proposed that emotional arousal is a possible mediator of the feedback (Honma and Honma, 2009). Our previous findings in methamphetamine-treated animals suggest that the targets of feedback are the extra-SCN oscillators in the brain, which directly regulate behavioral rhythmicity (Natsubori, et al., 2014).
In the present study, we examine the effects of 4 different nonphotic interventions, combinations of wheel-running and exposure to novel environment, during an 8-h phase-advanced LD on reentrainment of the circadian behavioral rhythm. The effects on both activity onset and offset are assessed. Here we demonstrate asymmetric phase-shifts of the activity onset and offset in response to the phase-advanced LD and acceleration of phase-shifts by nonphotic interventions.
Materials and Methods
Animals and Housing
Adult C57BL/6J male mice were used in the present study. They were about 8 to 10 weeks old at the beginning of the experiment. The animals were born and raised in our animal quarter under controlled environmental conditions: 12:12-h LD (lights-on from 0600 h to 1800 h, about 100 lux at the cage level), constant temperature (22-24 °C), and constant humidity (50%-60%). After weaning, 4 or 5 male mice were housed together. They had free access to food and water throughout the experiment. The animal care and experimental procedures were approved by the Animal Research Committee of Hokkaido University (Permission No. 08-253) and performed in strict accordance with the Guidelines for the Care and Use of Laboratory Animals of Hokkaido University.
Experimental Procedure
The mice were housed individually in polycarbonate cages (15 × 25 × 15 cm) placed in a light-proof chamber (40 × 50 × 30 cm) for at least 2 weeks. The LD conditions in the chamber were the same as in the animal room except for light intensity. The light intensity during the light period was about 300 lux at the surface of the cage. The circadian behavioral rhythm was measured with a passive infrared sensor for more than 10 days before LD shifts to confirm full entrainment of the rhythm to the preshift LD. The LD was phase-advanced by 8 h in such a way that the light period on the first day of phase-shift was shortened to 4 h. The shifted LD cycle was continued for 4 cycles and the last dark period was immediately followed by constant darkness (DD), which persisted for at least 3 weeks (Fig. 1A). The first day of DD was designated as DD1.
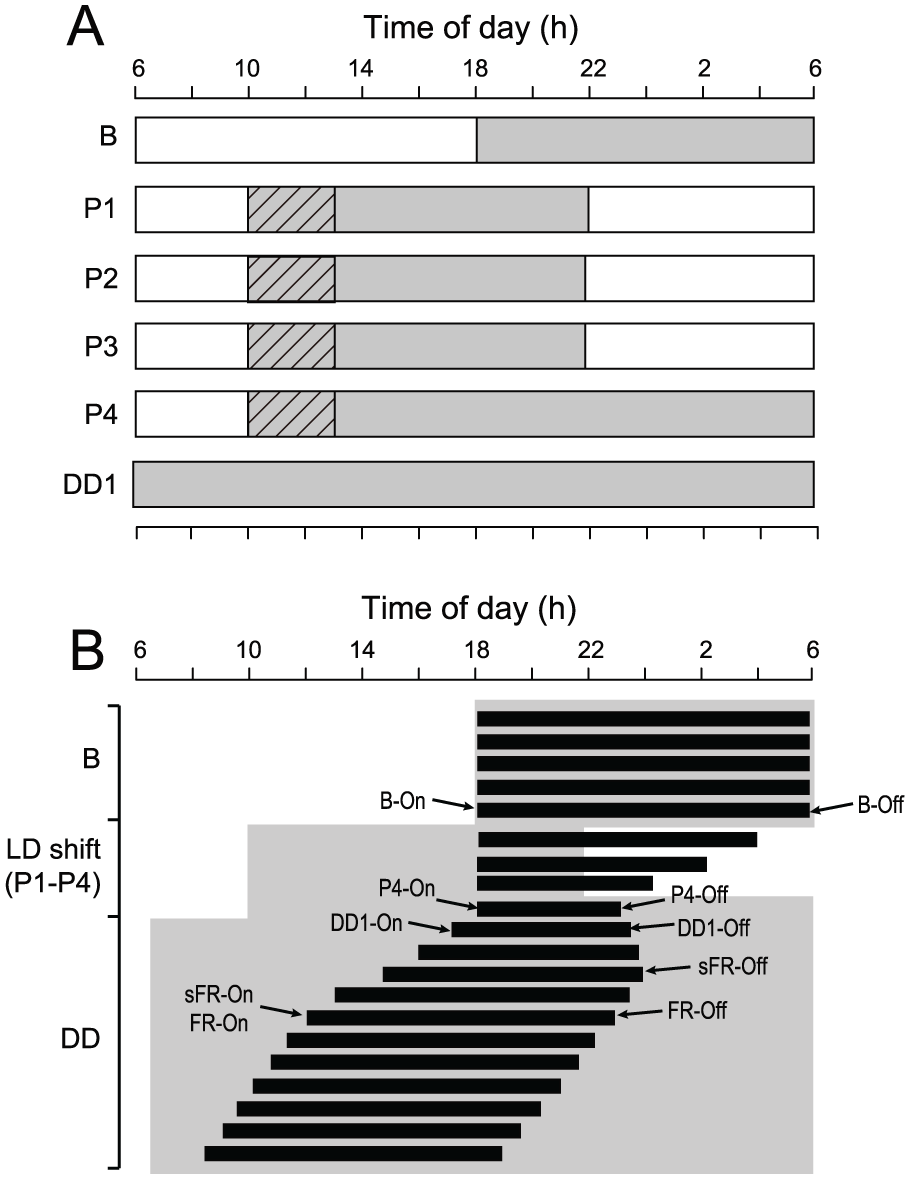
Experimental protocol and schematic drawing of reference phases of the circadian behavioral rhythm. (A) Open and gray areas indicate the light and dark periods, respectively. Shadowed striped areas indicate the period of 3 h of exposure to nonphotic interventions. (B) Closed horizontal bars indicate an activity band of circadian behavioral rhythm. Open and gray areas indicate the light and dark period. B-On (Off) = a phase of activity onset (offset) under the baseline LD. DD1-On (Off) = a phase of activity onset (offset) on the first day of DD. sFR-On (Off) = a phase of activity onset (offset) on the first day that the phase-marker reaches steady state free-running. FR-On (Off) = both activity onset and offset show steady state free-running.
From the beginning of the phase-advanced dark period, mice were subjected to 1 of the following 4 interventions for 3 h: transferred from the chamber for a few seconds and kept in the home cage (HC, n = 9), transferred from the chamber to set a running wheel of 10 cm in diameter (RW) to HC (HC+RW, n = 7), transferred from the chamber and exposed to a new cage (NC, n = 9), and transferred from the chamber and exposed to NC with RW (NC+RW, n = 9). About 1 min later, the animals were returned to the chamber. Three hours later, the same procedure was repeated and mice in the last 2 groups were returned to their home cages. The interventions were performed in the shifted LD for 4 cycles.
Locomotor activity was continuously monitored by a passive infrared sensor attached to the ceiling of the chamber. The sensor detected a change in the intensity of thermal radiation emitted from a mouse in association with a body movement (Abe et al., 2004). The number of movements was fed into a computer system (The Chronobiology Kit; Stanford Software Systems, Stanford, CA). In the HC+RW and NC+RW groups, the number of wheel revolutions was simultaneously counted every 1 min with a micro-switch sensor.
Analyses of the Circadian Behavioral Rhythm
Circadian behavioral rhythms were expressed as double-plotted actographs in 5-min bins, and the phases of activity onset and offset were estimated using Analyze 98 software (Stanford Software Systems) and CLOCKLAB software (Actimetrics, Evanston, IL). The parameters of the behavioral rhythm analyzed in the present study are schematically illustrated in Figure 1B. The activity onset and offset on the baseline day (B) were obtained by averaging those on the 10 consequent days immediately prior to the LD shift. The difference between the 2 phases was defined as activity time (α) (Aschoff, 1981). The phase-shifts in DD showed 2 distinct stages with respect to the rate of phase-shift: an initial fast but steadily slowing stage and a subsequent slow but steady shift. The turning point from the fast to slow stage was defined as separate free-run (sFR), since it was not necessarily the same for the activity onset and offset. sFR was determined first by visual inspection and designated as the temporal sFR (sFRt). To determine the sFR objectively, 2 regression lines with the largest correlation coefficient (R2) were obtained before and after sFRt. The regression line was searched for by incorporating the phase markers around sFRt into the calculation one by one. The day of the cross-point of the 2 regression lines was regarded as the objective sFR. Interestingly, the sFR was not different from the visually inspected sFRt in most cases. The number of days needed for reaching sFR was calculated. The day when both the activity onset and offset started to free-run in parallel was defined as FR. Thus, FR was identical to sFR of either the activity onset or offset. The activity time (α) of the free-running rhythm was obtained by averaging those in more than 3 cycles of those observed after FR.
Phase-shifts were calculated separately for the activity onset and offset between B and DD1 (B-DD1), between DD1 and sFR (DD1-sFR), and between B and FR (B-FR). A correlation between α on DD1 and the amount of following phase-shifts (DD1-sFR) was analyzed.
Statistics
The differences in the circadian parameters (phase and α) among the 4 groups (HC, HC+RW, NC, NC+RW) were analyzed by 1-way analysis of variance (ANOVA) with post hoc Tukey-Kramer test. Changes in the circadian parameters in the time course (B, DD1, sFR, and FR) were analyzed by paired t test. Unpaired t test was used for comparison of 2 values from the independent groups. Pearson’s correlation test was used for evaluation of the correlation between the 2 values. A p < 0.05 was regarded as statistically significant.
Results
Differential Phase-shifts of Activity Onset and Offset Result in Shortening of α
In response to an 8-h phase-shifted LD, both phase and α of the circadian behavioral rhythm were changed (Fig. 2, Tables 1-3). Behavioral activity in most cases became obscure in the first shifted cycle of LD but reemerged afterward. In the HC group, the activity onset phase-advanced only slightly on DD1, whereas the activity offset advanced immediately and substantially (Tables 1 and 2). As a result, α was significantly shortened (α compression) on DD1 (Table 3). Subsequent release into DD phase-advanced activity onset, whereas activity offset phase-delayed, resulting in a lengthening of the activity band (α decompression). The phase-delayed activity offset suddenly turned to a phase-advanced shift at sFR and began free-running with a period shorter than 24 h. Closer inspection revealed that the phase-advance shifts of activity onset also had 2 distinct stages: an initial rapid shift and a subsequent slow but steady shift. The changes in the rates of phase-shift showed individual differences and did not occur simultaneously for the activity onset and offset. The activity offset reached sFR significantly earlier than the activity onset (onset, 7.4 ± 3.3 days; offset, 4.2 ± 1.0 days) (t8 = 3.065, p = 0.016, paired t test) (Table 1). Essentially the same changes in the phases and α values were detected in the groups of nonphotic intervention during the course of reentrainment. Day-to-day activity onset and offset of individual mice in each group are illustrated in Supplementary Figures S1 through S4. P values for the results in Tables 1 through 3 are given in Supplementary Tables S1 through S3.
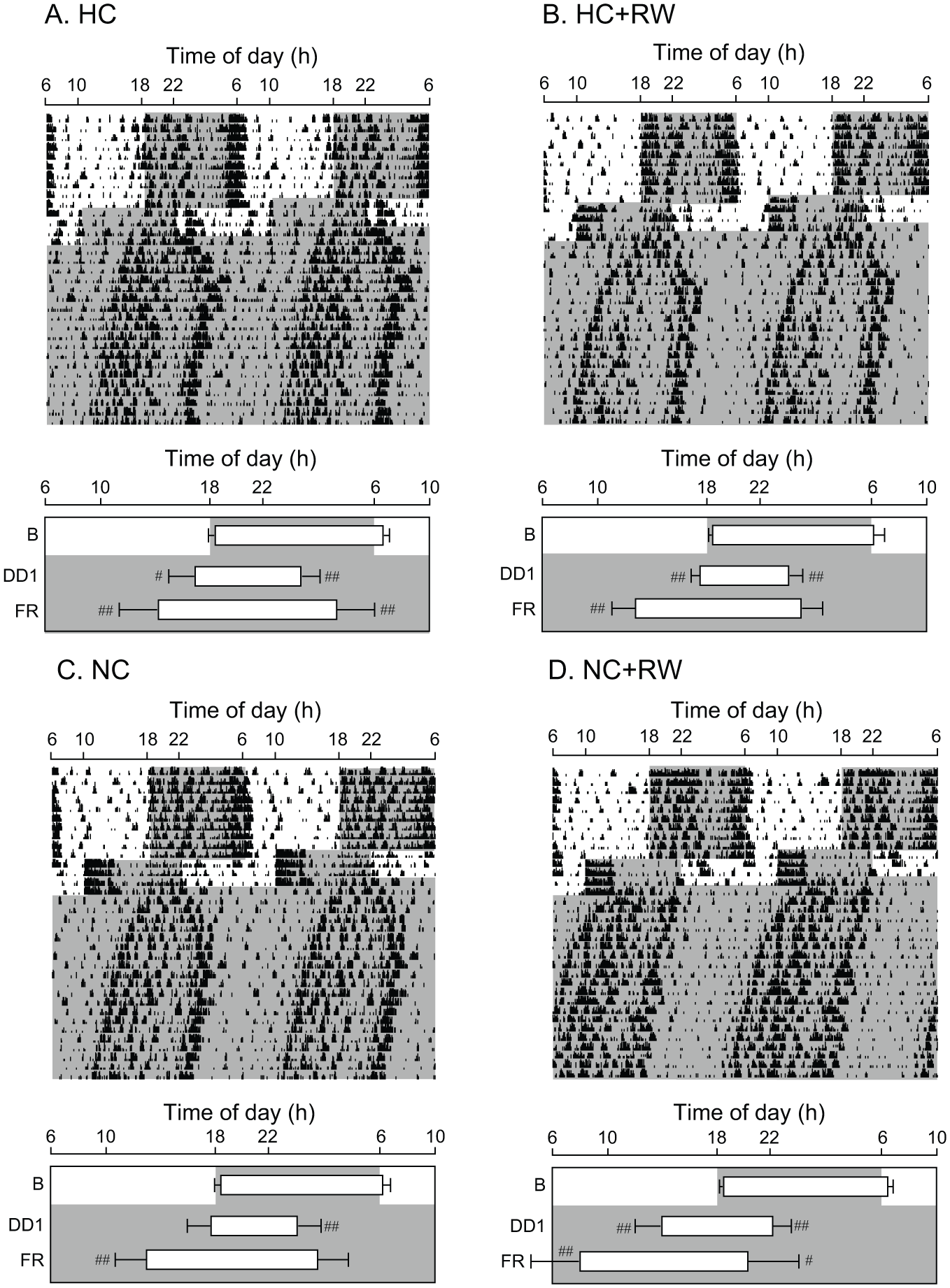
Circadian behavioral rhythms before, during, and after LD shift. Representative double-plot actograms of circadian behavioral rhythms and the mean activity bands in B, DD1, and FR in mice treated with nonphotic interventions during LD shifts. Behavioral activity (counts) was expressed in 5-min bins. Gray area in each graph indicates the dark period. The horizontal columns indicate the mean activity bands. Horizontal bars at the onset and offset of the activity bands indicate SD. #p < 0.05, ##p < 0.01 vs. previous stage by paired t test.
Phases of activity onset and offset (mean ± SD hours in local time).
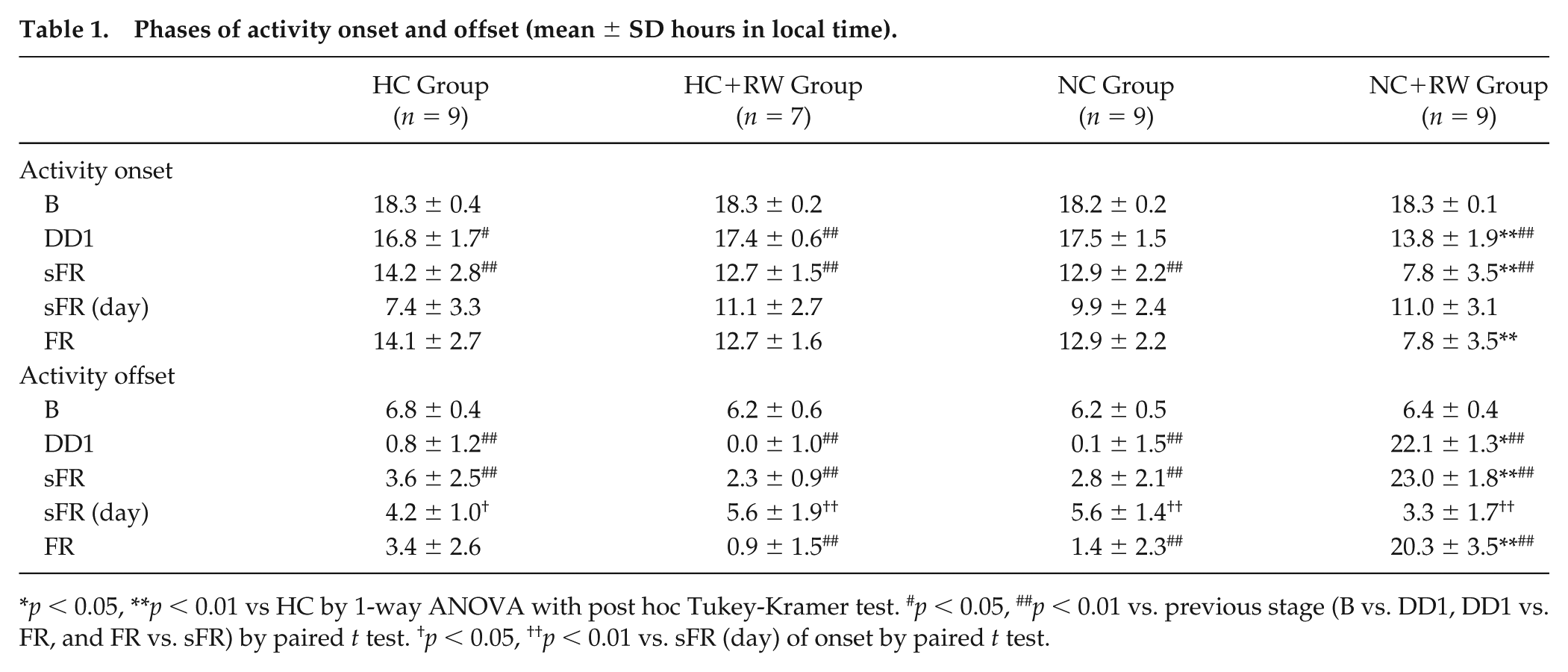
p < 0.05, **p < 0.01 vs HC by 1-way ANOVA with post hoc Tukey-Kramer test. #p < 0.05, ##p < 0.01 vs. previous stage (B vs. DD1, DD1 vs. FR, and FR vs. sFR) by paired t test. †p < 0.05, ††p < 0.01 vs. sFR (day) of onset by paired t test.
Phase-shifts of activity onset and offset (mean ± SD hours).
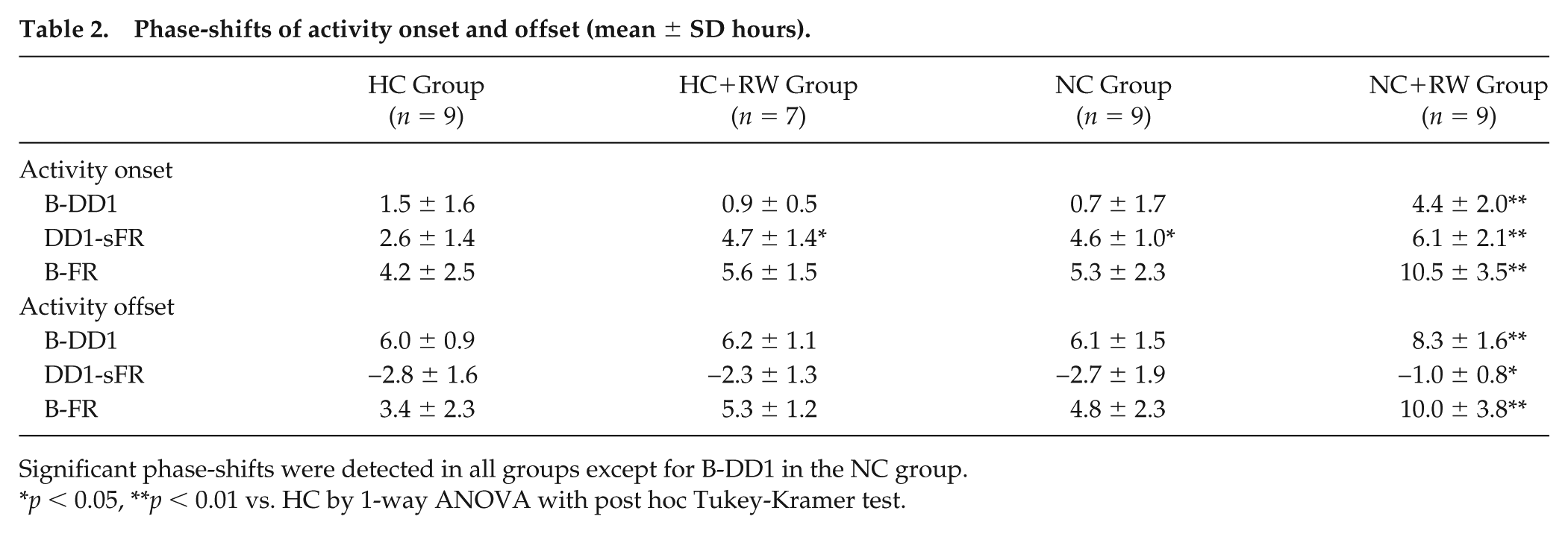
Significant phase-shifts were detected in all groups except for B-DD1 in the NC group.
p < 0.05, **p < 0.01 vs. HC by 1-way ANOVA with post hoc Tukey-Kramer test.
Activity time (α) (mean ± SD hours).

p < 0.05 vs. B, #p < 0.05, ##p < 0.01 vs. previous stages (B vs. DD1 and DD1 vs. FR) by paired t test.
Acceleration of Phase-shifts by Nonphotic Interventions
The circadian phases and the magnitudes of phase-shifts in the HC and the 3 nonphotic intervention groups are demonstrated in Tables 1 and 2. Compared with the HC group, the activity onset and offset at DD1, sFR, and FR were significantly advanced in the NC+RW group (Table 1). The phase-shifts at B-DD1, DD1-sFR, and B-FR were statistically significant in all 3 groups except for the phase-shift of activity onset in the NC group at B-DD1 (Table 2). Compared with the HC group, the phase-shifts were significantly larger in the NC+RW group in all 3 stages for both the activity onset and offset. A slight but significantly larger phase-shift was detected in the HC+RW and NC groups for the activity onset in DD1-sFR. These results indicate that NC+RW strongly accelerated the phase-shifts of both the activity onset and offset during the course of reentrainment. HC+RW and NC also facilitated the phase-shifts of activity onset to some extent.
Compression and Decompression of α
During the course of reentrainment, α was significantly changed in all groups (Table 3). The α at DD1 was shortened (α compression) by almost half to B and lengthened at FR relative to DD1 in all groups. The α at FR was also significantly lengthened in HC and NC+RW relative to B.
Correlation between α in DD1 and Subsequent Phase-shifts
Figure 3 illustrates the correlations between α at DD1 and the subsequent phase-shifts (DD1-sFR) for the activity onset and offset. For this calculation, the data from all groups were used together. A significant correlation was found for the activity offset (r = 0.606, p = 1.44E-04) but not for the activity onset (r = 0.019, p = 0.918). Namely, longer α at DD1 was associated with smaller phase-shifts of the activity offset, but not of the activity onset, in the following free-run (DD1-sFR). These findings indicate the impact of α compression at DD1 on the following phase-shifts of activity offset.
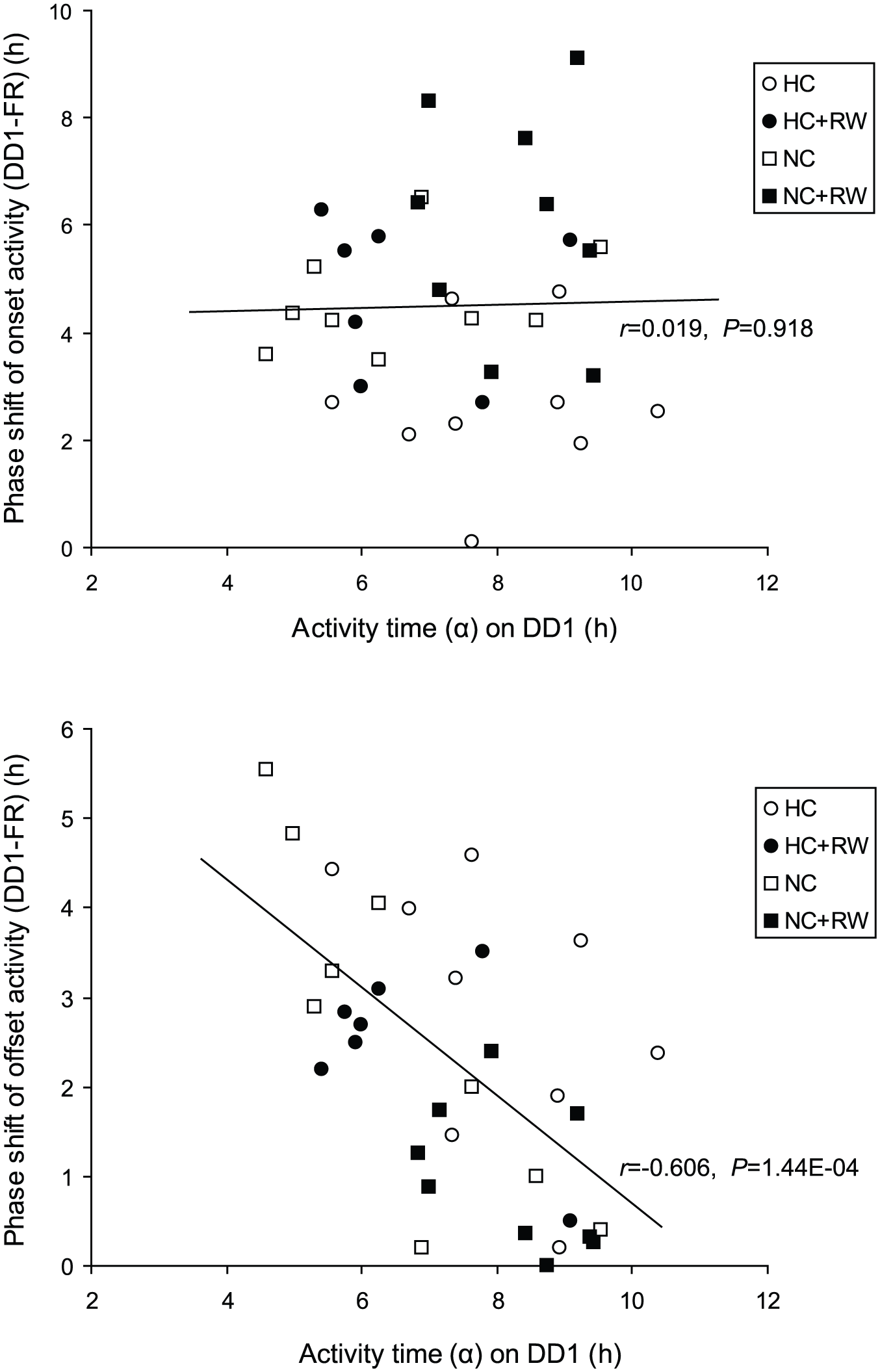
Correlations between α at DD1 and the magnitude of phase-shifts (DD1-sFR). Upper and lower panels indicate the correlations in activity onset and offset, respectively. An oblique line in each panel indicates a linear correlation line with a correlation coefficient and statistical evaluation. The signs in the insert column indicate the types of nonphotic intervention for individual data.
No Correlation between the Number of Wheel Revolutions and the Magnitude of Phase-shifts
Figure 4 illustrates the total number of wheel revolutions during 4 shifted days in HC+RW and NC+RW and the magnitude of phase-shifts at B-DD1 for the activity onset and offset. Although the total number of wheel revolutions was not different between the 2 groups (19,280 ± 6340 revolutions in 4 days for HC+RW and 22,667 ± 6064 revolutions in 4 days for NC+RW) (t14 = 1.09, p = 0.295, unpaired t test), the magnitude of phase-shift was significantly different for both the activity onset and offset (onset, t14 = 4.53, p = 5.00E-04; offset, t14 = 3.02, p = 0.009, unpaired t test). In addition, no significant correlation was found between the number of wheel revolutions and the magnitude of phase-shifts for either the activity onset or offset (onset, r = 0.239, not significant; offset, r = 0.035, not significant). Thus, there was no obvious relation between the extent of wheel-running and the magnitude of phase-shifts.
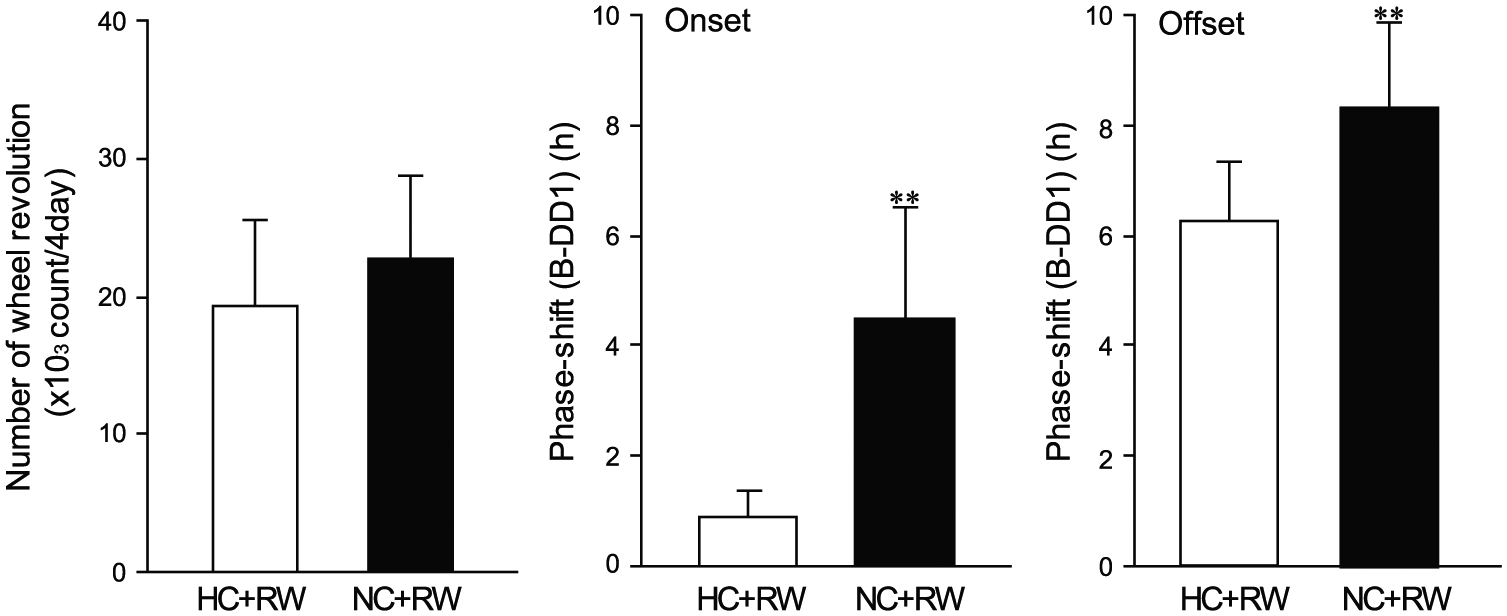
Wheel revolutions and phase-shifts. Total number of wheel revolutions in 4 days (right) and the magnitude of phase-shifts of activity onset (middle) and offset (left) in the HC+RW (white columns) and NC+RW (black columns) groups. Vertical lines indicate SD. *p < 0.05, **p < 0.01 vs. HC+RW by unpaired t test.
Body movements were significantly increased for 3 h after cage exchange in the NC group compared with the HC group (457 ± 161 counts in 4 days vs. 2324 ± 529 counts in 4 days, t16 = 10.14, p = 2.27E-08, unpaired t test). However, phase-shifts were not different between them for either the activity onset or the offset.
Discussion
In response to an 8-h phase-advance shift of LD, the activity offset immediately and substantially phase-advanced, whereas the activity onset moved forward only slightly for the first 4 days, resulting in α compression. Wheel-running in a new cage for 3 h at the beginning of the shifted dark period accelerated the phase-advance shifts of activity onset and offset. After release in DD, the activity onset phase-advanced, while the activity offset phase-delayed, resulting in α decompression. Although nonphotic interventions other than NC+RW showed minor and transient effects, overall phase-shifts were significantly accelerated only by wheel-running in a new cage.
Differential Phase-shifts of the Activity Onset and Offset
Differential phase-shifts of the activity onset and offset in response to an advanced LD result in α compression; α is shortened to about half of the baseline value (Table 3). Previously, the nocturnal increase of N-acetyltransferase (NAT) activity in rat pineal gland was reported to be abolished during phase-advance shifts of LD cycles (Illnerová et al., 1987), which could be explained by α compression and partly by light masking. Masking effects of light seem to be much stronger on the pineal NAT activity than on behavior.
Differential phase-shifts could be explained by differential sensitivities of 2 circadian oscillators underlying these phase-markers. A 2-oscillator hypothesis was advanced to explain split phenomenon (Daan and Pittendrigh, 1976b; de la Iglesia et al., 2000) and photoperiodic changes in the activity band (Aschoff, 1954; Sumová et al., 1995; Refinetti, 2002). Honma et al. (1985) demonstrated different PRCs to a single light pulse in rats by using the activity onset and offset as phase-markers of the circadian behavioral rhythm. The immediate PRC for the activity onset (onset PRC) showed only a phase-delay portion, whereas the PRC for the activity offset (offset PRC) demonstrated both a phase-delay and a phase-advance portion. A phase-delay portion of the offset PRC could be secondary, a result of a phase-delay shift of the onset oscillator underlying the activity onset. A similar PRC was reported for the circadian rhythm in NAT activity in rat pineal gland (Illnerová and Vaněček, 1982). In the present study, light from the shifted LD hit the advance portion of the offset PRC to produce a larger phase-advance shift in the activity offset but only slightly in the activity onset at least by DD1. As a result, α compression took place. The small phase-shift of activity onset could be ascribed to 2 conflicting forces: (1) the phase-delaying impact produced by light at the phase-delay portion and (2) the strong influence of the phase-advanced oscillator underlying the activity offset through the oscillatory coupling. Thus, the differential phase-shifts of activity onset and offset in the present study are understandable in terms of the 2-oscillator hypothesis.
Upon release into DD, the activity onset and offset shift in opposite directions, resulting in α decompression. Interestingly, the amount of phase-shift of the activity offset but not of the activity onset during α decompression is negatively correlated (phase-delay) with the extent of α compression (Fig. 3). This phenomenon could be explained by the mutual interaction of 2 circadian oscillators underlying these phase-markers. Light-induced α compression might produce a force to repel the 2 oscillators from each other. Since α decompression is achieved by small phase-advance shifts of the activity onset and large phase-delay shifts of the activity offset, the coupling force seems bidirectional but differential in direction. The molecular or cellular mechanism of oscillatory coupling is unknown, but the neural network in the SCN could be involved (Ono et al., 2013; Mieda et al., 2015).
Acceleration of Phase-advance Shifts by Nonphotic Interventions
Exposure to a new cage with a running wheel (NC+RW) accelerates the phase-advance shifts of both activity onset and offset (Table 2). HC+RW and NC also accelerate the phase-advance shifts of activity onset to some extent. Previously, we observed similar acceleration of phase-shifts by exposing mice to new cages with running wheels (Yamanaka et al., 2008).
In the literature, the activity onset has been reported to show much larger phase-advance shifts than the present study under similar reentrainment schedules (Hannibal et al., 2008; Cao et al., 2013; Lahouaoui et al., 2016; Miyakawa et al., 2007). The discrepancy is likely due to a difference in the monitoring method of behavior. In all these studies, a running wheel was used for behavior measurement. Actually, reentrainment has been reported to be faster in animals with a running wheel than in those without (LeGates et al., 2009).
The acceleration of phase-shifts by nonphotic interventions could be ascribed to an enhanced light sensitivity of the circadian system, probably at the level of retinal light perception (Panda et al., 2003). Sympathetic activation by enhanced alertness dilates the pupil (Hayashi et al., 2010), which may increase the light input to the SCN and therefore the amplitude of the PRC to light (Nelson and Takahashi, 1991). Although wheel-running was scheduled to the dark period in the present study, the effects might persist for a substantial period (Yamanaka et al., 2013). Perhaps a more plausible mechanism would be changes in the oscillatory coupling between the 2 oscillators underlying the activity onset and offset, as influenced by nonphotic interventions. The circadian period of the rat behavioral rhythm in DD is shortened by wheel-running (Yamada et al., 1988). Timed exposure to a running wheel changes the free-running circadian period of the behavioral rhythm in DD (Yamanaka et al., 2013). These changes in the circadian period could be explained by changes in the oscillatory coupling of a multi-oscillator system (Pittendrigh and Daan, 1976c) The acceleration of phase-shifts is also observed during α decompression (DD1-sFR) in the HC+RW and NC group, in which the acceleration is not detected during LD shifts (B-DD1) (Table 2). These findings indicate that the acceleration of phase-shifts is not only due to increased light sensitivity but also due to the change in the circadian system itself, most likely a change in the strength of oscillatory coupling.
It is a matter of debate whether the effect of nonphotic interventions is due to physical exercise or to other factors associated with body movement. Exposure to a new cage showed an accelerating effect on phase-shifts to some extent. In contrast, the number of wheel revolutions was essentially the same in the HC+RW and NC+RW groups, yet the extent of phase-shifts was significantly different between them (Fig. 4). By contrast, the amount of phase-shifts per se was not changed in the NC group despite a significant increase in body movement. These results suggest an additive effect of wheel-running and novel environment and an involvement of factors other than physical exercise or body movement itself. Enhancement of vigilance or alertness could be a candidate (Mistlberger and Skene, 2004). Gentle handling is reported to mimic the phase-shifting effect of wheel-running in hamsters, which could be explained in a similar way (Antle and Mistlberger, 2000; Mistlberger et al., 2003).
Possible Mechanism of Acceleration of Phase-shifts by Behavioral Interventions
Figure 5 illustrates a working hypothesis for the differential phase-shifts of the activity onset and offset of the circadian behavioral rhythm in response to an 8-h LD shift and the mechanism of nonphotic acceleration of phase-shifts.
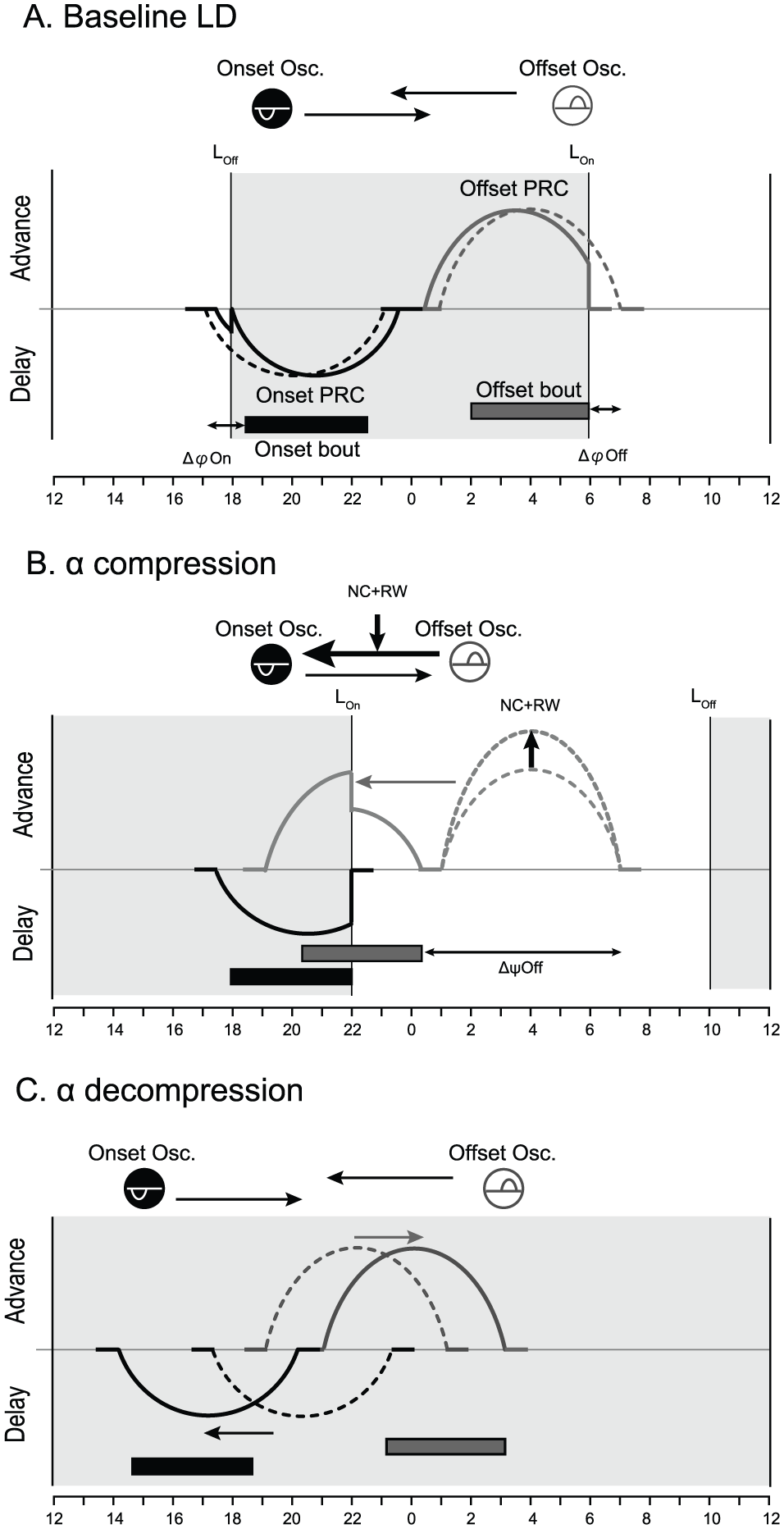
Schematic drawing of a 2-oscillator hypothesis explaining nonphotic acceleration of phase-shifts. (A) Two coupled oscillators underlying the activity onset and offset differentially entrain to LD. (B) Compression of α at DD1 by phase-shifts of the light period and nonphotic intervention. Nonphotic intervention increases the sensitivity of the offset oscillator to light and/or modifies the coupling. The offset oscillator phase-advances together with an advance shift of the phase-advance portion of the PRC. (C) Decompression of α in DD. In each panel, gray and white areas indicate the 12-h dark and light periods of LD, respectively. Black and gray horizontal bars indicate the onset and offset activity bout regulated by the 2 coupled circadian oscillators. Arrows between the 2 oscillators indicate the oscillatory coupling. In this model, each oscillator has a different PRC to light. The PRC for the onset oscillator (black) has only a phase-delay portion, whereas the PRC for the offset oscillator (white) has only a phase-advance portion. Dashed and solid lines indicate the phase position of the PRC for each oscillator. LOff and LOn indicate the time of lights-off and lights-on.
The onset PRC to a single light pulse has a phase-delay portion, while the offset PRC has a phase-advance portion. After an 8-h LD shift, the phase-delaying impact of light at the subjective evening is lost, while the phase-advancing impact of light increases because the light covers the whole advance portion of the PRC (Fig. 5B). Simultaneously, the activity offset or underlying oscillator exerts a phase-advancing impact on the activity onset through an oscillatory coupling. Here, the phase-delaying force by light counteracts the phase-advancing force by the oscillatory coupling, resulting in a smaller phase-shift of the activity onset than the offset.
Upon release into DD, the impact of light is lost and the phase-shifts of both markers are ascribed to the intrinsic energy of the coupled oscillator system. The phase-shifts in DD have 2 stages with different directions and rates. Two factors seem to be involved in these phase-shifts: One is the intrinsic circadian periods of both oscillators, and the other is the coupling force between them. Since the direction and rate of phase-shifts are different for the onset and offset oscillator, the coupling is likely bidirectional (Fig. 5C). The intrinsic periods of the 2 oscillators are not known, but the integrated period is slightly shorter than 24 h under stable coupling.
Wheel-running in a new cage may modify the light sensitivity of the circadian system and the coupling force between the 2 oscillators involved. The circadian period of a coupled oscillator system is also affected by the nonphotic intervention. Overall effects of the nonphotic intervention result in the acceleration of reentrainment to a phase-advanced LD cycle.
It is concluded that differential phase-shifts of the activity onset and offset in response to an 8-h phase-advanced LD are most likely due to different light responsivities of circadian oscillators underlying the 2 phase markers. Nonphotic interventions such as wheel-running in a new cage may increase the sensitivity of the circadian oscillator to light and/or change the coupling force between the 2 oscillators to accelerate reentrainment to a shifted LD. These phenomena could be explained by 2 coupled circadian oscillators underlying the activity onset and offset.
Supplemental Material
Figure_S1 – Supplemental material for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice
Supplemental material, Figure_S1 for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice by Yujiro Yamanaka, Sato Honma and Ken-ichi Honma in Journal of Biological Rhythms
Supplemental Material
Figure_S2 – Supplemental material for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice
Supplemental material, Figure_S2 for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice by Yujiro Yamanaka, Sato Honma and Ken-ichi Honma in Journal of Biological Rhythms
Supplemental Material
Figure_S3 – Supplemental material for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice
Supplemental material, Figure_S3 for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice by Yujiro Yamanaka, Sato Honma and Ken-ichi Honma in Journal of Biological Rhythms
Supplemental Material
Figure_S4 – Supplemental material for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice
Supplemental material, Figure_S4 for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice by Yujiro Yamanaka, Sato Honma and Ken-ichi Honma in Journal of Biological Rhythms
Supplemental Material
Yamanaka_et_al._Supplementary_Data-JBR-17-0089.R3 – Supplemental material for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice
Supplemental material, Yamanaka_et_al._Supplementary_Data-JBR-17-0089.R3 for Two Coupled Circadian Oscillators Are Involved in Nonphotic Acceleration of Reentrainment to Shifted Light Cycles in Mice by Yujiro Yamanaka, Sato Honma and Ken-ichi Honma in Journal of Biological Rhythms
Footnotes
Acknowledgements
This study was financially supported partly by Grant-in-Aid for Science Research (A) from MEXT (No. 20249010) to K.H., Grant-in-Aid for Young Scientists (B) from JSPS (No. 21700650 to Y.Y), and the Creation of Innovation Centers for Advanced Interdisciplinary Research Area Program, MEXT.
Author Contributions
Y.Y. and K.H. designed the experiments. Y.Y., S.H., and K.H analyzed the data. Y.Y., S.H., and K.H. wrote the manuscript.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
