Abstract
Endocrine research in animals often entails exogenous hormone administration. Special issues arise when developing administration protocols for hormones with circadian and seasonal periodicity. This article reviews various methods for the exogenous administration of hormones with such periodicities by focusing on melatonin. We discuss that methodological variations across studies can affect experimental results. Melatonin administration techniques used in vertebrates includes infusion pumps, beeswax pellets, oral administration, injections, SILASTIC capsules, osmotic pumps, transdermal delivery, beads, and sponges.
Research in experimental endocrinology began with the development of the classic ablation and replacement technique (Berthold, 1849). Once specific isolates from glands could be extracted, researchers could replace chemical derivatives instead of the entire gland (for review on testosterone, see Soma, 2006). The timing and manner of the administration of exogenous hormones can induce different physiological responses depending on the time of day and season of administration. Special consideration must be given to the administration of hormones with daily, annual, and developmental changes in their endogenous synthesis and release. The synthesis and release of melatonin, a hormone synthesized in most vertebrates in darkness, vary in amplitude based on the time of day and duration at certain times of the year. Specifically, peak melatonin synthesis occurs at nighttime relative to daytime (Wurtman et al., 1963), and the duration of this peak is longer during winter relative to summer, outside of the tropics (for review, see Tamarkin et al., 1985). Melatonin is a unique hormone because it has a direct relationship with light patterns, allowing photoreceptive animals to monitor time of day and year and to coordinate changes in physiology and behavior appropriately.
Although melatonin is the specific focus of this review, the methodologies discussed have implications for the exogenous administration of all hormones with circadian and seasonal periodicity in animal research. Endocrine circadian and seasonal rhythmicity are observed in hormones including, but not limited to, corticosterone (in female rats; Gibbs, 1976), testosterone (in male mice; Daan et al., 1975), or estradiol (in female hamsters; Morin, 1980). Weitzman (1976) reviewed circadian rhythms of adrenocorticotropic hormone, cortisol, growth hormone, prolactin, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) in men. The review presented here on melatonin may be compared to reviews on the modes of testosterone administration that address the relationships between release rate, dosage, and other advantages/disadvantages (Fusani, 2008; Quispe et al., 2015). Researchers who investigate hormone delivery methods for medical use, ranging from sleep medication to birth control, must consider the effects of experimental context (e.g., time of day/year) on research findings. By exploring the range of possibilities in drug delivery, scientists and practitioners alike can customize an administration protocol in research and personalized medicine. Hormone administration for medical purposes can take individualized circadian typology into account (for review, see Adan et al., 2012).
Common methods of melatonin administration in mammals and birds are discussed in the current review, including microinfusion pumps, beeswax pellets, oral administration via food/water, injections, and SILASTIC capsules. Lessons from this review on melatonin administration techniques can assist researchers in designing experiments with other rhythmically released hormones.
Melatonin in Circadian Rhythms
Circadian photic regulation of melatonin production affects its subsequent binding and action. The phototransduction pathway regulates the production of melatonin and includes a key neural site that also binds melatonin in almost all mammals, the SCN of the hypothalamus, or the “master clock” of circadian rhythms (Pevet and Challet, 2011). The mammalian phototransduction pathway is initiated when light activates intrinsically photosensitive retinal ganglion cells (ipRGCs), which contain the photopigment melanopsin. This signal is transduced via the retinohypothalamic tract, which contains monosynaptic and direct axonal projections to the SCN. Axonal projections from the SCN synapse in the paraventricular nucleus (PVN), and preautonomic neurons from the PVN synapse onto sympathetic preganglionic neurons of the intermediolateral cell columns of the spinal cord, ending with noradrenergic projections of the superior cervical ganglion synapsing on pinealocytes. Light inhibits pineal synthesis of melatonin, and the absence of light releases an inhibition of melatonin synthesis (Klein and Weller, 1972; Klein and Moore, 1979). Two enzymes, arylalkylamine-N-acetyltransferase and hydroxyindole-O-methyltransferase, are necessary for pineal melatonin synthesis and are regulated by light via the retinohypothalamic phototransduction pathway (Klein and Weller, 1972; Klein and Moore, 1979). Melatonin binds the SCN in almost all mammals studied to date (Dubocovich and Markowska, 2005), with the exception of the ferret (Mustela putorius furo), spotted skunks (Spilogale gracilis), and mink (Mustela lutreola) (for review, see Bittman, 1993). Melatonin-binding in the SCN affects the electrical activity rhythm of the SCN, facilitating synchronization of peripheral clocks (for review, see Kriegsfeld and Silver, 2006). Thus, melatonin conveys photic information to internal clocks (Axelrod, 1974), and therefore is commonly considered to be the chemical transducer that synchronizes peripheral tissues to the environment (for review of the mechanism, see Pevet et al., 2006; Reiter, 1991).
There is a temporal relationship between a stimulus (e.g., light or melatonin administration) and a response (e.g.,activity rhythms, clock genes, or endogenous melatonin production). Melatonin’s relationship with the circadian pacemaker can be determined by measuring circulating melatonin, core body temperature, and phase-shifts in activity rhythms (Gronfier et al., 2004). Lighting and endogenous melatonin production rhythms must be considered prior to administering exogenous melatonin. Differences in day length are especially relevant when researching seasonal rhythms.
Melatonin in Seasonal Reproduction
Nights are longer in winter outside of the tropics, and the duration of melatonin synthesis and secretion corresponds to the length of the night. The extended duration of melatonin secretion during winter can induce annual physiological changes in seasonal, photoperiodic animals. Melatonin administration techniques are used to study an array of interrelated seasonal phenomena, such as hibernation (Stanton et al., 1987), immunity (Bentley et al., 1998; Champney et al., 1997), oxidative stress (Karuppagounder et al., 2007; Rojas et al., 2008), metabolism (Bartness and Wade, 1985; Prakash et al., 1998), and neuroplasticity (Bentley et al., 1999; Kumari et al., 2015). A critical seasonal phenomenon studied in relation to melatonin is reproduction (for review, see Tamarkin et al., 1985). The duration of the melatonin signal can drive reproductive changes in photoperiodic seasonal breeders (for review, see Bartness et al., 1993). The perceived length of the night can be artificially extended in the lab by administering a relatively long duration of exogenous melatonin. Short-day breeders, such as sheep, goats, and deer (Ovis aries, Capra aegagrus hircus, Odocoileus virginianus) are reproductively active in winter, and long-day breeders, such as Siberian hamsters and domestic horses (Phodopus sungorus sungorus, Equus ferus caballus) are reproductively active in spring/summer. When melatonin was infused at a low concentration for 12 h (10 ng total, 14 pg/min) in juvenile Siberian hamsters housed in 16 h light: 8 h dark (16L:8D) day lengths, testicular regression occurred within 12 days. The same total dose (10 ng) of melatonin infused for shorter periods of time, either 4 h (42 pg/min) or 6 h (28 pg/min), failed to induce testicular regression (Carter and Goldman, 1983). Thus, in this species, the duration of melatonin exposure is critical for timing reproduction appropriately. In short-day-breeding Suffolk-cross ewes housed in long, summer-like days, melatonin ingested before the onset of darkness extended the melatonin signal and induced estrous cycles similar to winter phenotypes (Arendt et al., 1983). In this case of a short-day breeder, a longer duration of melatonin exposure activated the reproductive system.
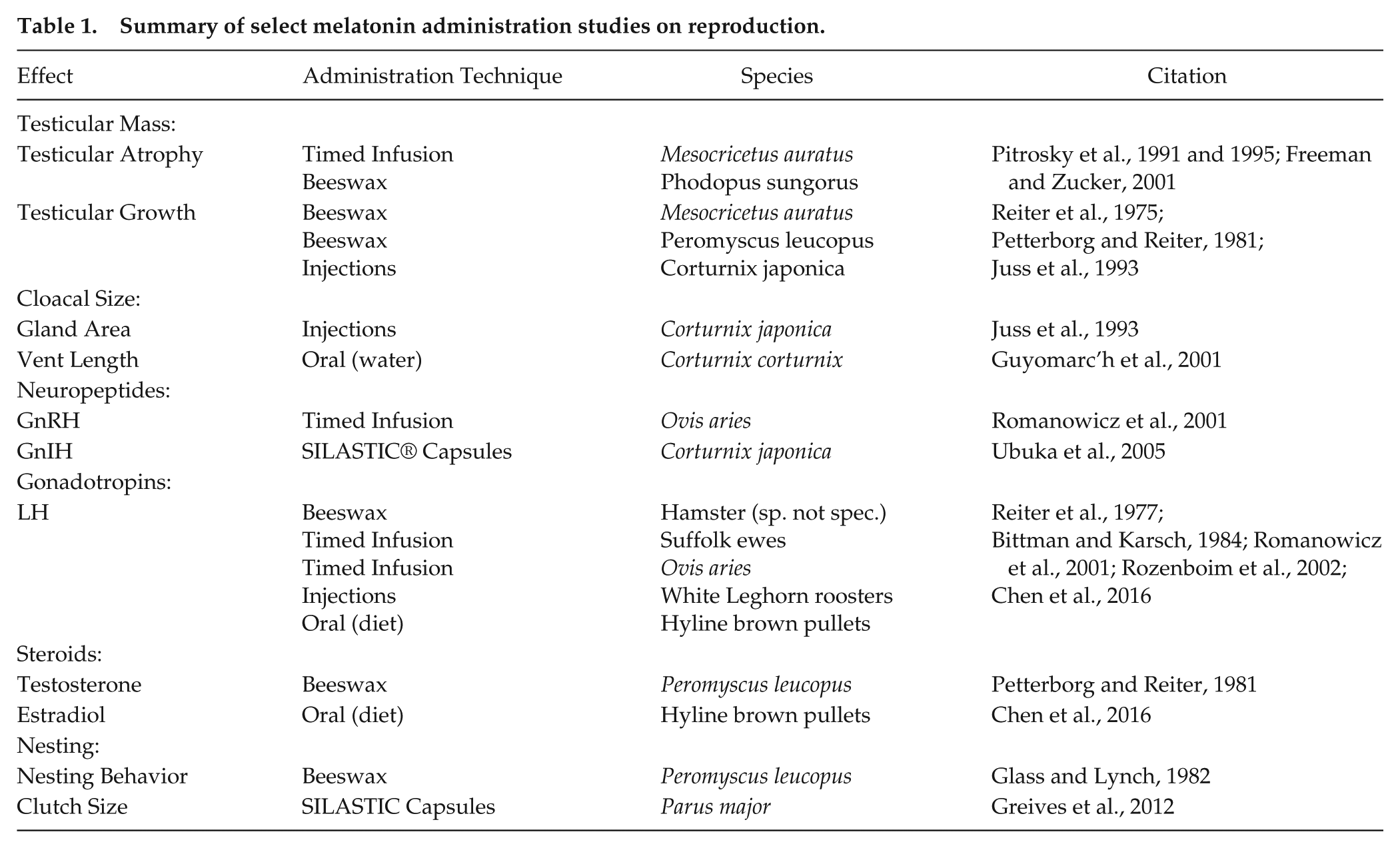
Melatonin’s effects on seasonal reproduction in birds and mammals include changes in testicular mass, cloacal size, reproductive neuropeptides, gonadotropins, testosterone, clutch size, and nesting behavior (see Table 1 for summary). The effects of exogenous melatonin on reproductive development are generally described in a binary fashion: the animal either undergoes a change in gonadal volume or does not. Relative changes in testicular volume of seasonal photoperiodic animals can be on a spectrum, but researchers tend to categorize changes in volume: the testes regressed, failed to regress, were stimulated, or failed to recrudesce. Table 1 shows different species, melatonin administration techniques, and quantified effects on reproductive development. Such methodological differences have generated inconsistent findings on the overall effect of melatonin on avian reproductive physiology.
Summary of select melatonin administration studies on reproduction.
Experimental Design: Lighting and Pinealectomies
Exogenously administered melatonin replaces, extends, or amplifies the endogenous signal. Prior to choosing an administration technique, the experimental context must be set to control for variables that affect endogenous melatonin rhythms. Lighting in a laboratory, field, or semi-natural enclosure can strongly influence the observable effects of melatonin (for review, see Calisi and Bentley, 2009). The following sections address how lighting, an environmental regulator of melatonin production, and the pineal gland, a principal producer of melatonin, can be manipulated to set an experimental context optimized for exogenous melatonin administration.
Lighting
Endogenous melatonin synthesis in the pineal gland is regulated by light (Reiter, 1991; Wurtman et al., 1963). Light exposure of a particular intensity or wavelength during the middle of a dark phase significantly decreases pineal melatonin production (0.186 μW/cm2 in Brainard et al., 1984; 520 nm in Honma et al., 1992). Researchers can regulate light quality and lighting patterns to inhibit or permit the endogenous melatonin signal to complement an exogenous administration regimen.
Animals can be housed in constant lighting conditions, such as constant light (LL) or constant darkness (DD). Rodents housed in constant darkness (DD) usually free-run, and rodents housed in LL usually split locomotor activity (Cheung and McCormack, 1983; Johnson, 1926; Richter, 1922). In constant conditions, DD or LL, light or dark pulses can be used to entrain animals. Entrainment involves aligning the internal clock with a zeitgeber, a “time giver” or external cue that resets the circadian clock. In birds and some mammals, circadian melatonin rhythms persist in DD and are suppressed in LL (Ralph, 1976; Ralph et al., 1971), demonstrating how constant lighting affects endogenous melatonin production.
In the wild, the shift from day to night always involves gradual changes in light intensity. In the laboratory, the shift from light to dark is often extremely abrupt and unnatural, having a square wave pattern. Another example of an unnatural lighting regimen is the use of T cycles, when the length of time between sequential light periods is greater than or less than 24 h (for review, see Merrow et al., 2005). Using T cycles in the context of melatonin research, Kumar and Follett (1993) housed Japanese quail (Coturnix japonica) in 3 consecutive 27-h days (3L:24D) and observed that the phase of the melatonin rhythm advanced 3 h relative to quail housed in 24-h days (3L:21D). Researchers can use T cycles to test if melatonin production has an endogenous circadian rhythm in their model organism. Another lighting manipulation, the skeleton photoperiod, uses multiple L:D cycles in a 24-h period or brief pulses of light during extended periods of darkness. Light pulses can simulate temporal patterns to reflect long or short days, with light acting as a zeitgeber by demarcating time points for dawn and dusk (for review, see Paul et al., 2008). Additionally, skeleton photoperiods can be used to distinguish endogenous circadian rhythms from physiological factors impacted by light/dark. For example, rainbow trout (Oncorhynchus mykiss) were maintained in 3 different skeleton photoperiods (8L:2D:2L:12D; 8L:7D:2L:7D; and 8L:12D:2L:2D), and the concentration of melatonin in blood samples increased in darkness and decreased in light regardless of the time of day, supporting the hypothesis that melatonin secretion in rainbow trout is in direct response to darkness (Alvariño et al., 1993).
Regardless of whether researchers use T-cycles or skeleton photoperiods in their experiments, such square wave light pulses do not simulate the gradual transition in intensity and color associated with dawn and dusk. Gradual transitions in lighting conditions have been used to study circadian rhythms in the laboratory (Boulos, Machi, et al., 1996; Boulos, Terman, et al. 1996) and in the wild (Daan and Aschoff, 1975). In humans, gradual transitions can improve cognitive performance (Gabel et al., 2013) and advance sleep onset (Santhi et al., 2012).
It is not known how shifts between different lighting conditions (sudden or gradual) affect endogenous melatonin production. Decreasing the duration of melatonin infusions can have opposite effects on Siberian hamster reproductive status, depending on the animals’ photoperiodic history (Gorman and Zucker, 1997). When hamsters were raised on short-days, while gradually decreasing the duration of melatonin administration from 10 to 7.5 h, testicular growth occurred, but hamsters raised on long-days exposed to the same infusion regimen showed complete testicular regression (Gorman and Zucker, 1997). Based on the direct relationship between melatonin and lighting, gradually increasing day length, as with gradually decreasing the duration of melatonin infusions, might have similar effects in Siberian hamsters.
The presence or absence of melatonin is influenced by the absence or presence of light, respectively. However, light qualities (e.g., wavelengths and timing of exposure) show varying influences on pineal melatonin synthesis. For example, pulses of 2 different monochromatic wavelengths of light at the same irradiance were administered to Wistar and Long-Evans rats during the midpoint of the dark phase. Green light (520 nm) suppressed pineal and plasma melatonin to levels comparable to the light phase for an extended period, but red light (660 nm) only temporarily suppressed pineal melatonin, with no quantifiable effect on plasma melatonin levels (Honma et al., 1992). A mechanism for wavelength distinction might involve chromatic clocks, and/or non-image-forming opsins of the pineal/parietal organ in vertebrates that sense colors (Spitschan et al., 2017). This hypothesis would need to be tested in blinded non-mammalian vertebrates with extraocular photoreception.
Not only the wavelength but also the timing of a light pulse must be considered, because light pulses administered in the middle and towards the end of a dark period have differing effects on melatonin production (Honma et al., 1992). When Honma et al. (1992) administered green- (520 nm) or red-light (660 nm) pulses early in the night to Wistar and Long-Evans rats, the green-light pulses suppressed melatonin production, but the red-light pulses did not. However, when the green- and red-light pulses were administered later in the night, both wavelengths suppressed melatonin production (Honma et al., 1992).
The timing of light exposure is relevant for wild animals exposed to light-at-night (LAN) in or near urban environments. Artificial LAN has dose-dependent effects on melatonin production and activity rhythms of great tits (Parus major) (de Jong et al., 2016) and seasonally variable effects on melatonin production in European blackbirds (Turdus merula) (Dominoni et al., 2013). LAN also affects activity rhythms of nocturnal grey mouse lemurs (Microcebus murinus) (Le Tallec et al., 2013), though its effect on melatonin production was not studied.
In addition to simulating dawn, day, dusk, and night, lighting can also simulate seasons in a laboratory environment. Studies in Suffolk ewes (Malpaux et al., 1988; Robinson and Karsch, 1987), Siberian hamsters (Stetson et al., 1986), and voles (Microtus montanus) (Horton, 1984) showed that melatonin production can be affected by relative changes in the lighting schedule of an animal or its mother, also known as their photoperiodic history (Bartness and Goldman, 1989).
Not all animals synthesize melatonin in darkness. Certain inbred strains of mice have undetectable levels of melatonin at all times of the day (5 out of the 36 strains studied in Goto et al., 1989), and other strains synthesize melatonin with a short peak, which is incapable of conveying seasonal information (2 out of the 4 strains studied in Vivien-Roels et al., 1998). C57/BL6 mice are sometimes used in circadian studies (for example, Guo et al., 2005) despite the absence of melatonin synthesis in the C57/BL6 strain (Goto et al., 1989). Given that some species do not synthesize melatonin, we cannot assume that lighting patterns always translate into melatonin signals. The strains we choose to use in melatonin studies should be selected with caution.
Light has a complex relationship with melatonin production. In general, bright light suppresses melatonin. Lighting patterns (constant vs. L:D ratios, sudden vs. gradual transitions, absolute durations vs. relative changes) and light quality (wavelengths and timing of exposure) can affect endogenous melatonin signals to relay circadian and seasonal information photochemically. For light to affect melatonin synthesis, the experimental animal must be photoreceptive with an intact phototransduction pathway and pineal gland, the primary source of endogenous melatonin.
Pinealectomies and Compensatory Melatonin Synthesis
Removing the pineal gland decreases serum melatonin within the first day post-surgery across vertebrates, as shown in goldfish (Carassius auratus) (Delahunty et al., 1978), Atlantic salmon (Salmo salar) (Porter et al., 1996), sea bass (Dicentrarchus labrax) (Bayarri et al., 2003), tiger salamanders (Ambystoma tigrinum) (Gern and Norris, 1979), ruin lizards (Podarcis sicula) (Foà et al., 1992), Japanese quail (Homma et al., 1967), chickens (Gallus gallus) (Pelham, 1975), rock pigeons (Columba livia) (Foà and Menaker, 1988), European starlings (Sturnus vulgaris), house sparrows (Passer domesticus) (Janik et al., 1992), sheep (Bittman et al., 1983), Syrian hamsters (Mesocricetus auratus) (Vaughan and Reiter, 1986), and rats (species not listed, Bubenik and Brown, 1997; Lewy et al., 1980). In these studies, the serum used to quantify melatonin concentration was collected hours, days, weeks, and/or months after the pinealectomy. The timing between the pinealectomy and serum collection was inconsistent across studies and sometimes not specified. Although we might assume that pinealectomies permanently remove the main source of melatonin, compensatory melatonin-synthesizing tissues can reinstate a detectable nocturnal peak of circulating melatonin. In pinealectomized rats, retinal melatonin production can reinstate diurnal rhythms of circulating melatonin at levels comparable to rats with sham-operated or intact pineals (Yu et al., 1981). Furthermore, the reinstatement of melatonin production rhythms in the Harderian gland of pinealectomized rats (Reiter et al., 1983) suggests that extra-pineal sources of melatonin can compensate for lost pineal melatonin over time. Across studies, there is significant variation in terms of recovery time following pinealectomy and subsequent melatonin administration; for example, from 3 weeks (Heigl and Gwinner, 1995) to as little as 2 h (Kumari et al., 2015). This variation can lead to undetected differences in endogenous melatonin production. Considerations for extra-pineal melatonin synthesis are comparable to experiments determining the “steroid-independent” effects of light on the reproductive neuropeptides of photoperiodic breeders following gonadectomy (for review, see Kriegsfeld and Bittman, 2010). Although gonads are the primary source of circulating sex steroids, neurosteroidogenesis might be impacted in undetectable ways by gonadectomy (for a comparative review, see do Rego and Vaudry, 2016). There is little research on compensatory post-pinealectomy melatonin production, so exogenous administration of melatonin and endogenous extra-pineal melatonin production might confound data in undetected ways.
There is also evidence for rhythmic extra-pineal melatonin production in birds that is not detectable in plasma. For example, a circadian rhythm of melatonin production is observable 2 weeks post-pinealectomy in the Harderian gland of rock pigeons (Vakkuri et al., 1985). In pinealectomized zebra finches (Taeniopygia guttata), circadian rhythms of melatonin production were detected in the gastrointestinal tract (Van’t Hof et al., 1999). Daily variations in melatonin production were also detectable in the eyes of quail (Coturnix sp.), rock pigeons, and domestic chickens (Gallus domesticus) (Pang et al., 1983). Although a small amount of circulating melatonin (13% of control levels) is detectable in pinealectomized and blinded Japanese quail (Underwood et al., 1984), highly localized melatonin synthesis might not be detectable in plasma (e.g., in the eyes and Harderian gland of Single-comb White Leghorn cockerels see Cogburn et al., 1987; in specific hypothalamic nuclei in turkeys [Meleagris gallopavo] see Kang et al., 2007; Kang et al., 2010). Therefore, the concentration of melatonin in plasma is not a reliable proxy for the concentration of melatonin in extra-pineal tissues in birds.
There are a few reasons why pinealectomies have different effects in mammals and birds. The main source of circadian melatonin production in mammals is the pineal gland, and the retinohypothalamic phototransduction pathway relays light cues for pineal melatonin production. Blinded northern grasshopper mice (Onychomys leucogaster) and golden mantled ground squirrels (Spermophilus lateralis) do not entrain to direct sunlight over the course of several months, despite having intact pineal glands (Nelson and Zucker, 1981). Although removal of the pineal gland in photoperiodic mammals inhibits photic stimulation of testicular development (Bartness and Goldman, 1989), pinealectomies in birds have variable effects because there are extra-pineal sources of melatonin regulated by photic input (Underwood, 1989). Even a combination of bilateral enucleation and pinealectomy cannot inhibit photic stimulation of avian gonadal development (Oliver and Baylé, 1982). This finding has been observed in ducks (Anas platyrhynchos) (Benoît, 1935), white-crowned sparrows (Zonotrichia leucophrys) (Gwinner et al., 1971), golden-crowned sparrows (Zonotrichia atricapilla) (Gwinner et al., 1971), Japanese quail (Homma et al., 1972), and American tree sparrows (Spizella arborea) (Wilson, 1991). Extrapineal melatonin synthesis is observable in other higher vertebrates (for review, see Huether, 1993). If the pineal is removed from animals in different reproductive states and/or photoperiods, the results of the experimental treatment differ. In long-day photostimulated quail, pinealectomies decreased ovarian weight and delayed reproductive development, but in short-day quail, pinealectomies had no observable effects on gonadal development (Sayler and Wolfson, 1968). Similarly, removing the pineal from Indian weaver birds (Ploceus philippinus) during short-days led to precocious gonadal recrudescence, and removing the pineal from Indian weaver birds during long-days accelerated gonadal development (Balasubramanian and Saxena, 1973).
Furthermore, the lighting condition in which animals are housed can alter the effects of pinealectomies. Circadian rhythms of rats kept in constant darkness were not impacted by pinealectomies. However, in constant light, pinealectomized and sham-operated rats differentially respond to changes in light intensity in terms of their free run period (Warren and Cassone, 1995).
Lighting and endogenous melatonin production can clearly impact the effects of exogenous melatonin administration. The timing of administration, slow- vs. fast-release capsules, concentration, solvent, duration of exposure, and frequency of administration are all independent variables in melatonin administration methodologies.
Common Techniques of Melatonin Administration
There are species differences in sensitivity to exogenous melatonin. For instance, regularly timed melatonin administration entrains Siberian hamsters but has no effect on the entrainment of Syrian hamsters (for review, see Hastings et al., 1997). Factors that might influence the response of a given species to exogenous melatonin include the animals’ age, sex, and photoperiodic history, and the interactions between these factors. Thus, each of these should be considered before experimentally manipulating melatonin.
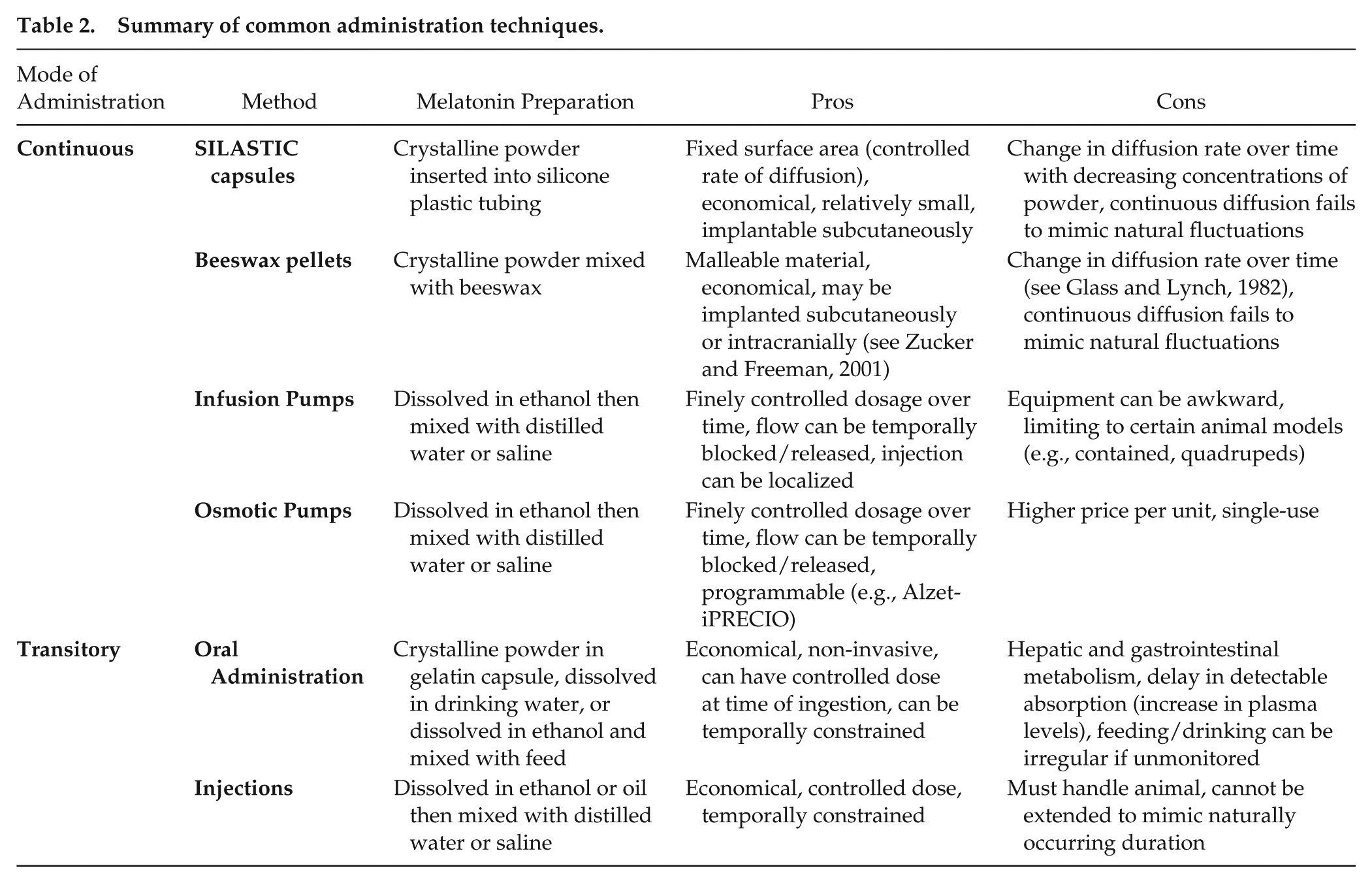
Melatonin and other hormones can be delivered by infusion pumps, beeswax pellets, oral administration, injections, SILASTIC capsules, osmotic pumps, transdermal application, sponges, and beads. Affordability, time-investment, frequency of stressful handling of animals, and physiological relevance can influence the choice of administration method (see Table 2 for summary of common administration techniques). In addition to economic factors, we focus on the duration and concentration of each administration regimen.
Summary of common administration techniques.
Infusion Pumps
Infusion pumps, or microinfusion pumps, can infuse fluids intravenously and subcutaneously with rates as low as 500 nL/h (see Supplementary Materials Table 1 for summary). Small-volume pumps include programmable systems that operate by rotating a screw that slowly depresses a syringe. The syringe is connected to tubing that leads to a gauged needle inserted subcutaneously, intraperitoneally, or intravenously. Infusion pumps administer a drug continuously and limit administration to a specific time frame. Over the course of multiple days or hours, exogenous melatonin can be administered rhythmically with microinfusion pumps by alternating a water-soluble solution containing dissolved melatonin with an oil-based solution within the tubing, closely mimicking the endogenous circadian rhythm of melatonin (e.g., Lynch et al., 1980). The duration of the melatonin signal is the critical parameter for driving reproductive development in photoperiodic, seasonally breeding mammals (for review, see Bartness et al., 1993). The duration of time when there is reduced circulating melatonin (the inter-pulse interval, or IPI) allows mammals to regain sensitivity to the melatonin signal (Bartness et al., 1993). Microinfusion pumps may have several advantages over other administration techniques, because their administration is predictable, temporally constrained, and convenient (Tan et al., 2011). These characteristics of microinfusion pumps complement research on circadian rhythms and seasonality because the hormone signal and the IPI can be temporally constrained in a manner that is physiologically relevant.
Melatonin administered with a microinfusion pump can replace or supplement the endogenous, circadian melatonin rhythm. Microinfusion pumps have been used to replace circulating levels of melatonin in pinealectomized animals housed in constant dim light (in western fence lizards, Sceloporus occidentalis in Hyde and Underwood, 1995; in Syrian hamsters in Schuhler et al., 2002). In the study by Hyde and Underwood (1995), pinealectomized lizards showed arrhythmia in circadian locomotor activity, and exogenous melatonin administration restored rhythmic locomotor activity. In Schuhler et al. (2002), 37 of the 64 pinealectomized Syrian hamsters fully entrained to melatonin infusions, whereas 20 hamsters transiently entrained to the melatonin signal, and 7 hamsters did not entrain at all. In addition to replacing melatonin in pinealectomized animals, microinfusion pumps can be used to extend the endogenous melatonin signal in pineal-intact animals (e.g., Romanowicz et al., 2001). When microinfusion pumps are used to supplement the endogenous melatonin rhythm, researchers should consider how exogenous melatonin administration is impacting endogenous melatonin production.
In studies related to seasonality, microinfusion pumps are used to administer melatonin and testicular volumetric changes are measured to determine how exogenous melatonin administration affects reproductive state (e.g., Pitrosky et al., 1991; Pitrosky 1995; Romanowicz et al., 2001). The rate of gonadal growth or regression varies across species, so the overall length of the administration period must be long enough to allow these physiological changes (4 to 8 weeks for adult male Syrian hamsters in Pitrosky et al., 1991 and 2 weeks for Polish lowland ewes in Romanowicz et al., 2002). Lighting conditions and/or the timing of pinealectomies can also alter the rate of gonadal development thereby affecting the required length of the administration period using microinfusion pumps.
There are some limitations to infusion pumps. Animals attached to external infusion pumps have limited locomotion. Microinfusion pumps and microdialysis (e.g., Ravault et al., 1996) are more commonly used in caged, quadruped animals. Infusions can be used in animals with primarily 2-dimensional locomotion (e.g., mammals and reptiles). Animals caught in the wild must be brought into a lab environment to enable monitored use of microinfusion pumps (e.g., Hyde and Underwood, 1995), but stress-induced physiological shifts likely occur when transporting animals from the wild into different settings (Dickens, Delehanty et al., 2009; Dickens, Earle, et al., 2009). Options for programmable, implantable pumps are becoming available (see Osmotic Pumps section of this review).
Beeswax Pellets
Beeswax mixed with melatonin crystalline powder is one mode of continuous melatonin administration (see Supplementary Materials Table 2 for summary). The compound is mixed, shaped, and implanted. Reproductive development has been the focus of most studies where researchers administered melatonin using beeswax pellets. Only one study measured the rate of release in vitro and assumed a similar rate in vivo (Glass and Lynch, 1982). The rate of release in vitro from a larger pellet (0.8 mm Outer Diameter, OD) on the first week was over 20 times higher than the rate of release in the subsequent 7 weeks. However, the rate of release of a smaller pellet (0.4 mm OD) on the first week was only 6- to 7-times higher than the rate of release during the subsequent 7 weeks (Glass and Lynch, 1982). Therefore, the initial rate of melatonin release is proportional to the volume of the beeswax pellet, and the size of the pellet not only affects the concentration of melatonin that is diffusing out but also the rate of melatonin diffusion (see Fig. 1).
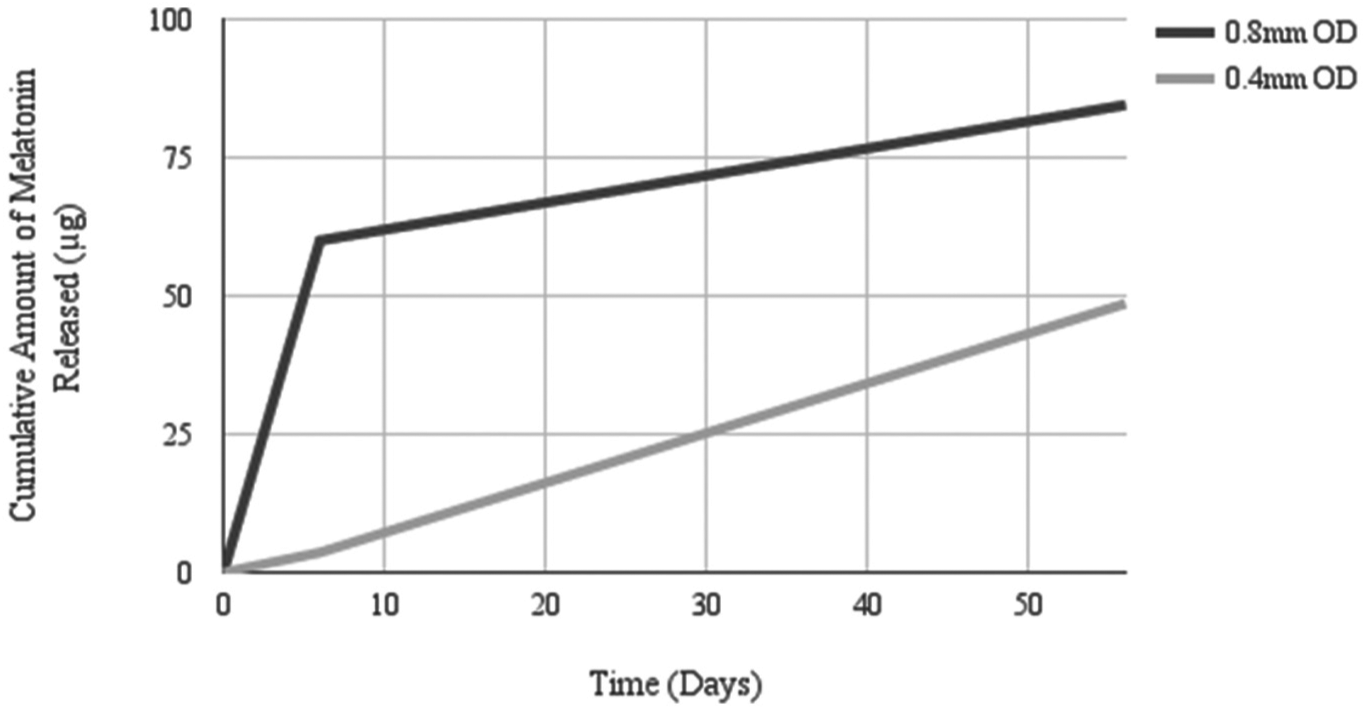
0.8 mm OD vs. 0.4 mm OD beeswax pellets: Estimated rate of melatonin release in vitro. Samples were collected on Day 6 and Day 56 and the rate of release from the preceding days was extrapolated from these collection times. Note that the rate of release is less consistent, and the overall amount of melatonin released is higher in the 0.8 mm OD beeswax pellet. Adapted from Glass and Lynch, 1982.
Because the rate of melatonin secretion from a beeswax pellet decreases over time, some researchers will replace the pellet throughout the course of an experiment. Reiter et al. (1975) varied the dose and the frequency of pellet replacement, showing that weekly or bi-weekly replacements with pellets of concentrations greater than 100 µg of melatonin per 24 g of beeswax reversed the effects of bilateral enucleation on reproductive development in male Syrian hamsters. However, this effect was not observed in pellets with lower concentrations of melatonin or for pellets replaced every 3, 6, or 12 weeks (Reiter et al., 1975). The surface area and volume of the pellet were not specified in the paper by Reiter et al., 1975, so the rate of melatonin diffusion cannot be determined. For other studies in which melatonin-beeswax pellets were used, researchers replaced the implant weekly (Reiter et al., 1977; Talbot and Reiter, 1973), bi-weekly (Petterborg and Reiter, 1981) or not at all (Prakash et al., 1998). If the overall length of the study is less than 2 weeks, the researcher can implant the pellet once. For longer studies, weekly replacement is advisable to maintain steady dosage.
The outcome of an experiment depends on where the beeswax pellet is implanted. An adaptive feature of beeswax pellets is their capacity to be implanted in specific nuclei of the hypothalamus. Although subcutaneously implanted melatonin-beeswax pellets have little effect on reproductive development, microimplants of melatonin-beeswax pellets in the anterior hypothalamus (AH) and the suprachiasmatic nucleus (SCN) significantly affect physiology and behavior in adult female white-footed mice (Peromyscus leucopus) (Glass and Lynch, 1982). In male white-footed mice, melatonin-beeswax implanted in the AH decreased testis volume. Subcutaneous (s.c) implants of the same dosage did not have a significant effect (Glass and Knotts, 1987). Freeman and Zucker (2001) implanted melatonin-beeswax pellets in melatonin-binding sites of the Siberian hamster brain for 40 weeks. Hamsters with melatonin-beeswax implants in the SCN, PVN, and nucleus reuniens (NRE) initiated gonadal regression in response to additional timed-daily melatonin infusions while hamsters with s.c. SILASTIC capsules did not respond to the timed-daily melatonin infusions (Freeman and Zucker, 2001). Because melatonin-beeswax implants in the third ventricle did not have effects comparable to implants in the SCN, PVN, or NRE, it was unlikely that melatonin from beeswax implants diffused to adjacent neural sites (Freeman and Zucker, 2001). Thus, subcutaneous implants do not have the same effect as microimplants in the brain.
Continuous peripheral melatonin administration decreases tissue sensitivity to supplementary melatonin. Trakulrungsi et al. (1979) injected melatonin daily in female Syrian hamsters that also had subcutaneous melatonin beeswax pellets. The injections, which would have typically inhibited female Syrian hamster reproductive development, were ineffective in hamsters with beeswax pellets containing 50 µg to 10 mg of melatonin, thus demonstrating a decreased sensitivity to the exogenous melatonin signal. Neural sites exposed to an extended melatonin signal are likely desensitized via downregulation of melatonin receptors in those regions (Gerdin, Masana, Dubocovich, 2004; Gerdin, Masana, Rivera-Bermudez et al., 2004). Furthermore, blocking melatonin receptors with the antagonist luzindole prevented the pre-dawn decrease in melatonin concentrations, suggesting that the nocturnal elevation of melatonin concentrations is downregulated by a feedback mechanism mediated by melatonin receptors in white-footed mice (Peromyscus leucopus) (Bedrosian et al., 2013).
There are many factors to consider when using beeswax implants. Despite their failure to mimic the endogenous rhythm of melatonin production, beeswax implants are still used to study seasonal phenomena such as reproduction. Based on the change in rate of melatonin release in vitro previously discussed (summarized in Fig. 1), the frequency that the pellets are replaced also affects melatonin concentration over time. Weekly or bi-weekly replacement of beeswax pellets is advisable for studies lasting more than 2 weeks. The concentration of melatonin in the studies reviewed here was usually 1 mg melatonin crystalline powder per 24mg beeswax. Other variables, however, such as the volume and surface area of the implanted pellet are inconsistently reported. Variation in the weight and size of the model organism must also be considered in justifying the size and concentration of subcutaneously implanted beeswax pellets. Beeswax pellets can be implanted in specific neural sites, unlike SILASTIC capsules. The pliancy of beeswax seems well-suited for continuous-release microimplants in the brain. Beeswax microimplants in specific neural sites can be difficult to replace, so this mode of administration is not suitable for long-term (>2 weeks) melatonin manipulation in specific neural sites.
SILASTIC Capsules
Since the 1970s, crystalline melatonin has been inserted and sealed inside SILASTIC tubing to be implanted in animals (Campaigne and Dinner, 1970) (see Supplementary Materials Table 3 for summary). The first documented study investigating SILASTIC use in drug delivery was by Folkman and Long in 1964, administering digitoxin, tri-iodothyronine, thyroid I-125, isoproterenol, sodium ethylenediamine tera-acetic acid, or tyrosine in the ventricular myocardium of 36 dogs. SILASTIC is a flexible, inert elastomer composed of silicone rubber and polydimethylsiloxane, produced by Dow Corning Corp. The material is rigid and stable enough to maintain its geometrical shape throughout the duration of an experiment. Although a depleted capsule leaves a foreign body embedded in the tissue, there is typically no evidence of inflammation resulting from the implantation of this material (Kind and Rudel, 1970).
SILASTIC implants are often described as continuous release but certain variables affect diffusion rate. Over time, the diminishing amount of dissolved powder inside the capsule decreases the overall rate of diffusion (Kind and Rudel, 1970). Furthermore, temperature affects the permeability of the SILASTIC membrane and the solubility of its contents (Zhang and Cloud, 2006), so diffusion rate might be mildly affected by fluctuations in core body temperature or by more extreme fluctuations of environmental temperature in studies conducted in the wild (such as Greives et al., 2012; Koresh et al., 2016) or in exotherms. For long-term studies (longer than a few weeks), SILASTIC implants would need to be replaced on a regular basis to maintain continuously elevated levels of circulating melatonin. For all of the studies in the current review, researchers implanted the SILASTIC capsule once and left it for the duration of the experiment. The change in mass of the SILASTIC capsule before and after implantation is sometimes used to calculate the rate of diffusion, as seen in Juss et al., 1993 (posited to be 24 µg/day), but this calculation assumes a constant rate of diffusion over the course of 20 days. Future studies can be conducted in vivo and in vitro to assess the diffusion rate over an extended period for frequently used volumes of SILASTIC capsules (e.g., 1.47 mm ID × 1.96 mm OD × 25.0 mm length), as previously done for beeswax pellets (Glass and Lynch, 1982). These studies would be foundational to determining if SILASTIC capsules should be replaced, and how frequently, in experiments with longer administration periods.
SILASTIC capsules are used for a range of melatonin research topics. Studies on circadian rhythms (Hau and Gwinner, 1994, 1995; Marumoto, Murakami, Kuroda et al., 1996), photoperiodism and reproduction (Bentley et al., 1998; Greives et al., 2012; Juss et al., 1993; Ubuka et al., 2005), and neuroplasticity (Bentley et al., 1999) use different species, different protocols for lighting and different volumes of SILASTIC capsules. The administration period, or the duration the capsules are left implanted, also varies across studies. Researchers of one study compared control animals subcutaneously implanted with SILASTIC capsules, with or without melatonin, with animals implanted with beeswax pellets in the brain (Freeman and Zucker, 2001). SILASTIC capsules are typically implanted subcutaneously. We found only one study in which the SILASTIC capsule was inserted intravaginally (Nowak and Rodway, 1985). In only a few studies, the main sources of melatonin are removed (pineal in rats Marumoto, Murakami N, Kuroda, et al., 1996; pineal and eyes in Japanese quail Ubuka et al., 2005). Continuous melatonin supplementation via SILASTIC capsules decreases the time it takes for animals to resynchronize circadian locomotor activity to a changing light schedule (Hau and Gwinner, 1994, 1995; Marumoto, et al., 1996). Results in reproductive studies, however, are not consistent enough to draw broad conclusions. In the wild, female great tits (Parus major) with melatonin-filled SILASTIC capsules delay the timing of the first clutch of eggs (Greives et al., 2012) and male great tits are more susceptible to cuckoldry because melatonin delays their onset of daily activity, permitting early morning extra-pair mating by the females (Greives et al., 2015). SILASTIC capsules are stable enough to tolerate conditions in the wild. The change in diffusion rate of SILASTIC capsules over time and the effects of continuous administration on an animal’s sensitivity to a hormone should be considered in long-term studies. Regardless, SILASTIC is one of the most commonly used continuous-release implantable devices for hormone administration.
Oral Administration: Food and Water
The least invasive technique for hormone administration included in this review is oral melatonin administration (see Supplementary Materials Table 4 for summary). Melatonin is ingested and absorbed through an animal’s digestive tract. A variety of plants and plant-based products have high levels of melatonin (phytomelatonin), including cherries (Burkhardt et al., 2001), walnuts (Reiter et al., 2005), wine (Fernández-Mar et al., 2012) and several others (for reviews see Garcia-Parrilla et al., 2009; Reiter et al., 2001; Reiter et al., 2007). Adult male humans consuming fruits with detectable levels of phytomelatonin show a dose-dependent increase in serum melatonin (Sae-Teaw et al., 2013). The list of sources of phytomelatonin and the ingredients of various chow or feed provided ad libitum are typically not described in the methods of studies focusing on melatonin. Although there are exciting prospects for comparing different sources of phytomelatonin, this review focuses on experiments with controlled melatonin concentrations by dissolving or mixing a measured amount in the vehicle of water or food.
Most, but not all, researchers dissolve melatonin crystalline powder in ethanol and dilute the solution in drinking water (Guyomarc’h et al., 2001; Heigl and Gwinner, 1995; Kumari et al., 2015) or mix it with food and let the ethanol evaporate (Chen et al., 2016; Lewis et al., 2006). Researchers who dissolve melatonin crystalline powder in ethanol should note that ethanol consumption rapidly and robustly suppresses plasma melatonin (Ekman et al., 1993) and salivary melatonin (Rupp et al., 2007) in a dose-dependent manner. Melatonin crystalline powder was also fed to deer (Odocoileus virginianus) in gelatin capsules stuffed into apple slices (Bubenik and Smith, 1987).
The dosage of orally administered melatonin corresponds to detectable increases in plasma melatonin, which suggests that dosage has a linear relationship with absorption through the digestive tract. However, in the animal studies reviewed here, only Chen et al. (2016) administered more than one dose and measured circulating levels. Researchers should test if different doses of an orally administered hormone correlate with circulating levels of the hormone.
Researchers using oral administration techniques must consider digestive tract metabolism as well. First, molecular circadian oscillators regulate everything from food intake, appetite, metabolism, and digestion (for review, see Waterhouse et al., 1997). Circadian and seasonal changes in metabolism in white-tailed deer, Odocoileus virginianus (Freudenberger et al., 1994) affect absorption of orally administered melatonin. The effects of seasonal fluctuations in melatonin duration on feeding were observed in European sea bass (Dicentrarchus labrax), in which orally administered melatonin affected both the proportion of macronutrients and total amount of food consumed (Rubio et al., 2004). Usually, administration of melatonin via animal feed involves weighing the animal and/or the food tray to check for consistency in food consumption and body mass. However, total food consumption does not account for physiological changes in digestion and metabolism. Secondly, melatonin is subject to a high hepatic extraction ratio (Lane and Moss, 1985). Unlike injected melatonin, orally administered melatonin is metabolized by the liver, which decreases its bioavailability. Furthermore, the GI tract synthesizes melatonin at local concentrations up to 500- times higher than the nocturnal levels of plasma melatonin (Bubenik, 2001). Melatonin in the upper GI tract acts as a free-radical scavenger and protects the mucosal layer from irritation caused by GI tract lesions (for review, see Konturek et al., 2007). Orally administered melatonin can affect endogenous GI melatonin synthesis, along with how melatonin is catabolized in the GI tract before reaching detectable levels in plasma. Because melatonin changes gastrointestinal physiology (Motilva et al., 2001), the effectiveness of a given dose and the sensitivity of the GI tract likely change with frequent administration. Lastly, using rat GI segments, melatonin absorption is highest in the ileum and rectum and lowest in the stomach (Tran et al., 2009). Melatonin is classified as a low-permeability drug when it is administered without vehicles (Tran et al., 2009). Due to the impressive variation across vertebrate digestive systems (Stevens and Hume, 2004), melatonin absorption probably varies with the anatomical and physiological properties of the GI tract of a given species. A comparative study of melatonin GI absorption across vertebrates would help guide the research using food or water as an administration method.
Oral administration also occurs with pharmaceutical use in humans. Melatonin pills are available over-the-counter in the United States and are unregulated. Melatonin pills affect circadian rhythms (Lewy et al., 1992), depression (Dalton et al., 2000), senescence-induced insomnia (Rikkert et al., 2001), jet-lag (Arendt et al., 1986; Herxheimer et al., 2002; Suhner et al., 1998), shift-work and blindness (Arendt et al., 1997). The fixed variables in these studies include the timing of ingestion, the dose, and the rate of release of melatonin from the pills. These variables are collectively optimized to facilitate sleep without interfering with productivity. The same variables can be controlled and tested in animal studies; e.g., conducting cognitive behavioral tests in addition to taking physiological measurements to determine the optimal timing and dosage of orally administered melatonin.
The mode and vehicle of oral delivery determine the rate of release of a given hormone. The duration of the elevated amplitude of circulating melatonin is different for melatonin administered in gelatin capsules, corn oil, or slow-release pills, suggesting that the mean elimination half-life of melatonin is impacted by its vehicle (Aldhous et al., 1985). The pharmacokinetics of oral melatonin delivery can be altered by how the capsule is coated (Lee et al., 1995). The chemical properties of different solvents and vehicles can affect hormone elimination half-life and thus should be taken into consideration for experimental design.
Injections
The delay between the time of administration and the subsequent increase in plasma melatonin is shorter for melatonin administered via injection relative to melatonin administered orally (Kennaway and Seamark, 1980) (see Supplementary Materials Table 5 for summary). Subcutaneous melatonin injections in sheep and goats produced transient peaks in plasma melatonin, and oral administration (via drinking water or in food) maintained elevated levels of circulating melatonin for up to 7 h after administration (see Fig. 2 adapted from Kennaway and Seamark, 1980). Replication of this type of study in different species can be used to choose an appropriate administration technique.
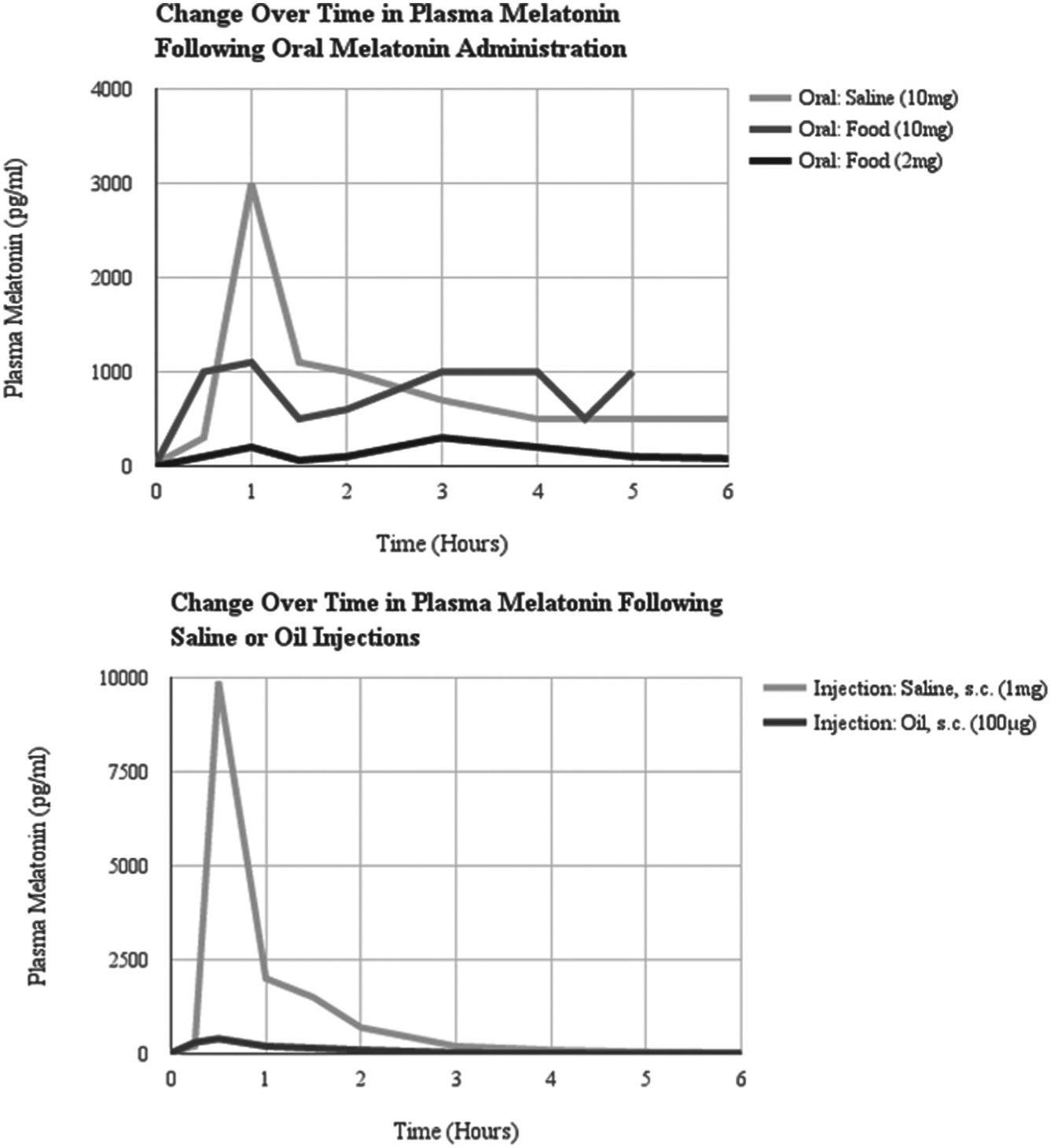
Plasma melatonin concentrations following melatonin administration to 2 Merino cross-bred sheep. Note the duration of the elevated peak in plasma melatonin is shorter for melatonin administered via subcutaneous injections relative to orally administered melatonin regardless of the vehicle (saline or food). The amplitude of the peak in plasma melatonin is lower for melatonin injected with the oil vehicle relative to the saline vehicle. While the concentration of melatonin in the saline injection is 10-times higher than the concentration dissolved in oil, the peak in plasma melatonin is 33-times higher following the saline injection. Adapted from Kennaway and Seamark, 1980.
Variations in dosage and/or frequency of injections can lead to different physiological outcomes. Only a subset of injection studies addressed the effects of dosage on physiological factors, such as changes in gonadal volume or oxidative stress (e.g., Cassone et al., 1986a; Juss et al., 1993; Rozenboim et al., 2002; Karuppagounder et al., 2007). The frequency of injections also affects the experimental results. Less-frequent injections had attenuated effects on testicular volume (Juss et al., 1993) and on plasma LH (Rozenboim et al., 2002). A possible explanation for why variations in dosage and/or frequency of injections lead to different physiological outcomes is that injections of melatonin at supraphysiological concentrations alter the sensitivity of target tissue. Melatonin injections in rats prevented nocturnal increases in endogenous melatonin and downregulated melatonin receptor expression (Gerdin, Masana, Dubocovich et al., 2004; Gerdin, Masana, Rivera-Bermudez, et al., 2004). Additional research on the effects of melatonin injections on melatonin receptor transcription, affinity, or function has yet to be conducted.
We observe some common practices across injection studies. Researchers conducting most of the injection studies did not remove the pineal gland or the eyes. They typically administered the injection(s) before the transition between lights on/off, extending the nocturnal melatonin signal while studying changes in reproductive state (Bartness and Wade, 1985; Champney et al., 1997; Juss et al., 1993; Rozenboim et al., 2002). For animals housed in constant lighting conditions, either constant darkness (Cassone et al., 1986a) or dim light (Cassone et al., 1986b), researchers administered the injection out-of-phase with circadian oscillations of endogenous melatonin production while studying circadian activity rhythms (Cassone et al., 1986a, 1986b).
Less-Common Techniques for Melatonin Administration
Some techniques are not commonly used to administer melatonin or other hormones. This section provides an overview of other technologies that are used or being developed for exogenous melatonin administration: osmotic pumps, transdermal creams, melatonin-loaded beads, and sponges.
Osmotic Pumps
There are numerous types of osmotic pumps used in drug delivery in animal research (for review, see Urquhart et al., 1984; Verma et al., 2004). An osmotic pump has an internal drug reservoir surrounded by an osmotic sleeve and a semi-permeable membrane (Theeuwus, 1987). The outer diameter of osmotic pumps is larger than SILASTIC capsules. When implanted, they cause relatively more local tissue damage than other administration methods (Sallinen et al., 2008). Mini-osmotic pumps can be programmed to administer at a constant volumetric delivery rate (Theeuwes, 1987), so the variability of diffusion rates previously described in SILASTIC capsules is controlled for with osmotic pumps. Furthermore, the release rates in vivo and in vitro are not significantly different (Theeuwus, 1987). Prevo et al. (2000) used ALZET osmotic pumps to administer increasing concentrations of melatonin (0.03%, 0.3%, and 3%) to Sprague-Dawley rats and observed a dose-dependent change in plasma melatonin and 6-sulphatoxymelatonin, a melatonin metabolite, in urine. The constant mass and volumetric delivery of osmotic pumps ensured a controlled release of the same concentration throughout the duration of the experiment, which is not guaranteed with any other method described in this review.
Intermittent drug delivery can be enabled in osmotic pumps by alternating a water-soluble solution with oil within a coiled reservoir, spacing the delivery of the drug of interest (with nicotine, Brynildsen et al., 2016). It is yet to be determined if and how intermittent delivery with lipid-water layers works for an amphipathic molecule like melatonin. Injection and infusion studies dissolve melatonin in solvents ranging from water-soluble ethanol (e.g., Bartness and Wade, 1985) to lipids (e.g., peanut oil; Reiter et al., 1977). Researchers can load hydrophilic and lipophilic solvents in an alternating fashion in the coiled reservoir of an osmotic pump. Because the pump has a constant flow rate, there is a direct relationship between the volume of solvent loaded in the reservoir and the time it takes for its release from the reservoir. Ideally, this setup allows researchers to alternate between a hydrophilic melatonin vehicle and a lipophilic blank vehicle. For example, a low volume of phenolsulfonphthalein solution with dissolved melatonin took 6 h to flow out of the coiled reservoir, and a higher volume of mineral oil took 18 h (Lynch et al., 1980). However, melatonin is amphipathic, and its lipid-solubility and potential diffusion into the mineral oil was not discussed. Future work attempting intermittent melatonin delivery would benefit from testing if its separation is maintained between adjacently placed hydrophilic and lipophilic solvents.
The effects of a drug are enhanced when administered with chemicals that improve its stability and/or permeability (see Transdermal Delivery section). Because melatonin is an antioxidant, research on its role in preventing oxidative damage has been studied through co-infusion with other chemicals via osmotic pumps. Co-infusion was used to study the neurodegenerative effects of iron on dopaminergic transmission in the nigrostriatal system (Lin and Ho, 2000). Melatonin infused alone did not significantly affect dopaminergic transmission, but when it was co-infused with iron, the oxidative damage caused by iron was reduced (Lin and Ho, 2000). Co-infusion would be challenging to conduct accurately and precisely with SILASTIC capsules because, depending on the chemicals, the diffusion rates through the SILASTIC membrane are likely different. Other osmotic pump studies that focus on the protective, antioxidant, free-radical properties of melatonin include studies in mice on post-cardial infarction (programmed at 4.5 mg/kg body weight per day in Sallinen et al., 2008), senescence (4 mg/kg body weight of bolus followed by continuous infusion of 8 mg/kg body weight per day in Kilic et al., 2002), and neurodegenerative damage induced by oxidative toxicity (Dabbeni-Sala et al., 2001). Melatonin can induce these protective effects by acting either as a free-radical scavenger or as a hormone that binds to and activates a melatonin receptor. To determine if melatonergic effects were receptor-mediated, Lemley et al. (2013) administered melatonin and a receptor-antagonist, luzindole, to pregnant sheep using ALZET osmotic minipumps. The catheter of the minipump was placed next to the gravid uterine vascular network and induced a local increase in arterial blood flow which was not observed in sheep simultaneously administered melatonin and luzindole (Lemley et al., 2013). This suggests that these physiological changes are mediated by melatonin’s G-protein-coupled receptor and not by melatonin’s role as an antioxidant.
The use of osmotic pumps to study the effects of melatonin in endocrine functions (e.g., circadian rhythms and photoperiodism) is not as prevalent as research on melatonin’s protective functions (e.g., as a free-radical scavenger and antioxidant). However, osmotic pumps are used to administer other neuropeptides involved in reproduction, such as gonadotropin-releasing hormone (GnRH, Robinson et al., 1993; Ronayne et al., 1993) and gonadotropin-inhibitory hormone (GnIH, Ubuka et al., 2006).
A decade ago, iPRECIO (Primetech Corporation, Tokyo, Japan) entered the market with a refillable and programmable infusion pump for small laboratory animals (Abe et al., 2009). The iPRECIO system allows researchers to start/stop drug administration at controlled doses in several applications (Tan et al., 2011). However, each unit is single-use and only lasts 52 days. The iPRECIO system requires power even in the blocked-flow condition (Johnson and Borkholder, 2016). Although 52 days is adequate for the duration of most studies on melatonin, a study in which the iPRECIO system was used to administer melatonin could not be found at the time of this review. Osmotic pumps offer fine-tuned control over the rate and timing of drug release in an unprecedented way, so it is ideal for administering circadian hormones like melatonin.
Transdermal Delivery
There are a couple of different routes that fall under the category of transdermal delivery. Melatonin administered via transbuccal delivery had no significant effect on sleep quality, calling into question the efficacy of this method (Dawson et al., 1998). Melatonin administered through intranasal delivery required different vehicles (polyethylene glycol 300 in Bechgaard et al., 1999; starch microspheres in Mao et al., 2004). The rate of absorption in nasal passages and the persistence of bioavailability after administration suggest that intranasal administration is a viable alternative to use of melatonin pills in humans. Nasal administration of melatonin in animal research appears to be limited to rabbits and rodents. The researchers using intranasal delivery focused on the chemical properties of melatonin (e.g., diffusion and absorption rate) without considering its physiological effects (Bechgaard et al., 1999; Mao et al., 2004).
Because skin is the primary defense against pathogens, vehicles and enhancers must be combined with melatonin to increase skin permeability (Oh et al., 2001). Franz diffusion cells were used to determine the lag time and permeability for transdermal delivery (hairless mouse skin in Oh et al., 2001 and human cadaver skin in Dubey et al., 2006; Dubey et al., 2007). Dermal permeability, however, can be affected by temperature and other biological factors (Scheuplein et al., 1976).
Transdermal melatonin delivery offers non-invasive and extended melatonin exposure that is ideal for ornithological field research. Free-living birds pose prohibitive challenges in terms of the difficulty of repeated capture for injections, consistent oral administration via food or water across individuals, and continuous-release implants requiring invasive surgery. Melatonin dissolved in Eucerin cream and applied to the naked skins of European stonechats (Saxicola torquata rubicola) significantly increased plasma melatonin concentrations (Goymann et al., 2008). The temporal architecture of plasma melatonin concentrations varied with the concentrations of melatonin applied. The application of cream with 6 µg melatonin led to an increase in plasma melatonin concentration (0.2 ng/ml to 0.4 ng/ml) which was maintained up to 180 min after application (Goymann et al., 2008). However, after the application of cream with 20 µg of melatonin, plasma concentrations of melatonin linearly increased from less than 0.2 ng/ml at 30 min post-application up to an average of 0.7ng/ml at 180 min post-application (Goymann et al., 2008). In garden warblers (Sylvia borin), melatonin cream increased plasma melatonin concentrations during both day and night (Goymann et al., 2008). When the concentration of melatonin in cream applied to warblers was doubled (40 µg) relative to the cream applied to stonechats (20 µg), the plasma melatonin concentrations were 7- to 12-times higher, which suggests that the concentration of melatonin in the cream has a nonlinear relationship with the amount absorbed and circulated (Goymann et al., 2008). Melatonin dissolved in Eucerin cream applied to Sylvia warblers during migration did not affect nocturnal migratory restlessness, or Zugunruhe, during the spring migration (Fusani et al., 2011), but it significantly decreased Zugunruhe associated with autumnal migration (Fusani et al., 2013). Thus, there appear to be seasonal changes in response to melatonin treatment and/or metabolism in this species.
Beads
Small, melatonin-containing beads can be administered orally, intravaginally, or intrarectally. Properties of melatonin-loaded beads, such as trapping efficiencies (how much melatonin can be loaded) and release profiles (rate over time at which melatonin diffuses from the microspheres), vary based on the solution in which the beads are placed (e.g., simulated gastric and GI fluids vs. saline). The trapping efficiencies and release profiles of melatonin-loaded sodium alginate beads, derived from brown algae, were studied in vitro (Lee, Parrott et al., 1996), in GI fluids (Lee and Min, 1996), and in simulated GI fluids (Lee, Parrott et al., 1999). The release rate of melatonin from the sodium alginate beads did not change with increased drug loading at higher trapping efficiencies (Lee, Parrott et al., 1996). Melatonin release was delayed in alginate beads that were coated in a polymer solution, relative to the release profile of plain alginate beads (Lee, Min et al., 1996). The delayed, sustained release of melatonin from alginate beads may offer an alternative vehicle for oral delivery.
Stearyl alcohol, a fatty acid alcohol that forms white microspheres that are insoluble in water, can also load melatonin for oral delivery (Lee et al., 1998). Another way of enabling sustained release of water insoluble components to aqueous solutions is to use sugar spheres coated in Aquacoat, an insoluble and water-permeable polymeric coating that expands and ruptures in aqueous solutions to allow for rapid drug delivery (Dashevsky and Mohamad, 2006). Sugar beads coated with 20% Aquacoat, loaded with melatonin, and orally administered to humans exhibited controlled release of melatonin that maintained plasma concentrations of over 100 pg/ml for as long as 8 h (Lee et al., 1995). Researchers who administer melatonin orally to animals might consider using microspheres instead of other vehicles because microspheres offer sustained melatonin release at relatively constant concentrations.
Sponges
After pre-absorbing melatonin, sponges are typically inserted intravaginally. Sponges as a mode of melatonin administration are used in large livestock such as mares (Thompson et al., 1983) or ewes (Buffoni et al., 2014; Luridiana et al., 2015; Rajkumar et al., 1989). The administration of melatonin from sponges is continuous. However, the concentration and diffusion rate of melatonin is likely not constant. When using sponges to administer hormones it would be relatively easy to calculate the change in diffusion rate over time in vitro, as previously done with beeswax pellets (Glass and Lynch, 1982).
Conclusion
Lessons from this review on melatonin administration methods can be used to design pilot studies for any hormone administration protocol. Firstly, assess the endogenous levels of the hormone of interest, especially if there is a circadian or seasonal component to the study. The amplitude and duration of the peak of hormone secretion can inform the dosage and timing of acute administration via injections. Secondly, determine how chronic exposure affects sensitivity of the animal to the hormone, especially for continuous-release techniques such as beeswax pellets, SILASTIC capsules, or osmotic pumps. The timing and frequency of administration and effects on tissue sensitivity can be ascertained by quantifying changes in receptor expression and binding over time. Because melatonin can downregulate its own synthesis in a receptor-mediated fashion (Bedrosian et al., 2013), researchers who administer exogenous melatonin for extended periods of time should consider such endogenous effects. Lastly, a pilot study for any hormone administration protocol should consider how samples are collected, stored, and analyzed. How plasma samples are collected, stored, and preserved can significantly affect absolute concentrations of melatonin measured (Goymann et al., 2007). Such pilot studies that factor these variables into their experiment can empirically determine how the administration technique of choice affects the responsiveness of the animal and how preservation of relevant samples affects hormone quantification.
Given the importance of amplitude and duration in any endocrine signaling pathway, the task of the researcher is to control for variables that affect how the animal receives an exogenously administered endocrine signal.
Supplemental Material
Supplementary_Materials_-_Table_1_Infusion_pumps-1_(1) – Supplemental material for Melatonin Administration Methods for Research in Mammals and Birds
Supplemental material, Supplementary_Materials_-_Table_1_Infusion_pumps-1_(1) for Melatonin Administration Methods for Research in Mammals and Birds by Dax Ovid, Tyrone B. Hayes and George E. Bentley in Journal of Biological Rhythms
Supplemental Material
Supplementary_Materials_-_Table_2_Beeswax-1 – Supplemental material for Melatonin Administration Methods for Research in Mammals and Birds
Supplemental material, Supplementary_Materials_-_Table_2_Beeswax-1 for Melatonin Administration Methods for Research in Mammals and Birds by Dax Ovid, Tyrone B. Hayes and George E. Bentley in Journal of Biological Rhythms
Supplemental Material
Supplementary_Materials_-_Table_3_Silastic-1 – Supplemental material for Melatonin Administration Methods for Research in Mammals and Birds
Supplemental material, Supplementary_Materials_-_Table_3_Silastic-1 for Melatonin Administration Methods for Research in Mammals and Birds by Dax Ovid, Tyrone B. Hayes and George E. Bentley in Journal of Biological Rhythms
Supplemental Material
Supplementary_Materials_-_Table_4_Oral_Admin-1 – Supplemental material for Melatonin Administration Methods for Research in Mammals and Birds
Supplemental material, Supplementary_Materials_-_Table_4_Oral_Admin-1 for Melatonin Administration Methods for Research in Mammals and Birds by Dax Ovid, Tyrone B. Hayes and George E. Bentley in Journal of Biological Rhythms
Supplemental Material
Supplementary_Materials_-_Table_5_Injections-1 – Supplemental material for Melatonin Administration Methods for Research in Mammals and Birds
Supplemental material, Supplementary_Materials_-_Table_5_Injections-1 for Melatonin Administration Methods for Research in Mammals and Birds by Dax Ovid, Tyrone B. Hayes and George E. Bentley in Journal of Biological Rhythms
Footnotes
Acknowledgements
This material is based upon work supported by the National Science Foundation Graduate Research Fellowship under Grant No. DGE 1106400. Any opinion, findings, and conclusions or recommendations expressed in this material are those of the authors(s) and do not necessarily reflect the views of the National Science Foundation.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
