Abstract
Within the suprachiasmatic nucleus (SCN)—the locus of the master circadian clock— transcriptional regulation via the CREB/CRE pathway is implicated in the functioning of the molecular clock timing process, and is a key conduit through which photic input entrains the oscillator. One event driving CRE-mediated transcription is the phosphorylation of CREB at serine 133 (Ser133). Indeed, numerous reporter gene assays have shown that an alanine point mutation in Ser133 reduces CREB-mediated transcription. Here, we sought to examine the contribution of Ser133 phosphorylation to the functional role of CREB in SCN clock physiology in vivo. To this end, we used a CREB knock-in mouse strain, in which Ser133 was mutated to alanine (S/A CREB). Under a standard 12 h light-dark cycle, S/A CREB mice exhibited a marked alteration in clock-regulated wheel running activity. Relative to WT mice, S/A CREB mice had highly fragmented bouts of locomotor activity during the night phase, elevated daytime activity, and a delayed phase angle of entrainment. Further, under free-running conditions, S/A CREB mice had a significantly longer tau than WT mice and reduced activity amplitude. In S/A CREB mice, light-evoked clock entrainment, using both Aschoff type 1 and 6 h “jet lag” paradigms, was markedly reduced relative to WT mice. S/A CREB mice exhibited attenuated transcriptional drive, as assessed by examining both clock-gated and light-evoked gene expression. Finally, SCN slice culture imaging detected a marked disruption in cellular clock phase synchrony following a phase-resetting stimulus in S/A CREB mice. Together, these data indicate that signaling through CREB phosphorylation at Ser133 is critical for the functional fidelity of both SCN timing and entrainment.
The suprachiasmatic nucleus (SCN) of the mammalian hypothalamus is the locus of the master circadian clock. Hormonal and synaptic information from the SCN orchestrates the rhythmic gating of various physiological, biochemical, and even behavioral processes. At the center of this clock timing process is a cell autonomous, autoregulatory, transcriptional/translational feedback loop (TTFL), in which an E-box-bound bHLH CLOCK/BMAL1 heterodimeric transcription factor drives the expression of the
A key event in the activation of CREB is its inducible phosphorylation of a serine residue at position 133 (Ser133). This serine site is the target of numerous proline-directed serine/threonine kinases, including protein kinase A, mitogen-stress activated kinase, and Calcium/calmodulin-dependent protein kinase type IV (CaMKIV) (Gonzalez and Montminy, 1989; Matthews et al., 1994; Sun et al., 1994; Enslen et al., 1995; Parker et al., 1996; Deak et al., 1998; Arthur and Cohen, 2000; Naqvi et al., 2014). At the mechanistic level, this phosphorylation event creates a docking site for CBP (
Within the SCN, both CREB phosphorylation and CRE-mediated gene expression are under the control of the circadian clock (Obrietan et al., 1999). Moreover, signaling via the CREB/CRE pathway is implicated in coupling photic input to the induction of
Materials and Methods
Mice
CREB serine 133 to alanine 133 knock-in mice (hereafter referred to as S/A CREB mice) were generated in the lab of J. Simon C. Arthur (University of Dundee, UK). Details of the gene targeting strategy, animal husbandry and genotyping procedures have been previously described (Wingate et al., 2009; Naqvi et al., 2014). The S/A CREB line was bred into the C57BL/6 background for at least 8 generations before their use here. S/A CREB homozygous mice were generated from heterozygous breeding pairs, and the wild-type (WT) offspring were used as reference experimental groups. Consistent with the work of Wingate et al. (2009), we found that S/A CREB homozygous pups were generated at a lower Mendelian ratio than would be expected for a heterozygous breeding strategy. All animals in the WT and S/A CREB groups passed a visual acuity test, which confirmed these systems were intact in S/A CREB mice; visual tests were performed as previously reported (Hansen et al., 2013). To examine cellular level timing, the S/A CREB mouse line was crossed with the
Behavioral Assessment of Circadian Clock Timing and Entrainment
Wheel running activity was used as a measure of circadian timing and clock entrainment. To this end, mice were individually housed in clear polycarbonate cages equipped with running wheels (15 cm in diameter). Wheel rotations were binned into 5-min intervals and collected using the Actiview hardware and software system (Starr Life Sciences; Oakmont, PA). All mice were initially entrained to a 12-h light (300 lux)/12-h dark (LD) cycle for a minimum of 10 days. For experiments that profiled locomotor activity under LD conditions, mice were maintained in this condition for a minimum of 10 additional days. For experiments that assessed free running activity, mice were transferred to total darkness (DD) and allowed to run for up to 50 days.
To examine the rate of re-entrainment to a shift in the noted LD cycle, mice were challenged with a 6-h advance and 6-h delay in the onset of the LD cycle. For these assays, mice were maintained on a stable LD cycle for at least 14 days before the light cycle shift. To assess the phase delaying effects of an early night light pulse, mice were initially dark-adapted for a minimum of 14 days, and then exposed to white light (50 lux, 15 min) at circadian time (CT) 15; animals were then returned to the DD condition for another 21 days.
Assessment of Circadian and Light-entrainment Phenotypes
Tau was determined with the assistance of the Actiview software program. To this end, a linear regression method was used to draw a line through the onset of daily activity for a period of 5-9 days, approximately 14 days after the transition into DD. Average activity profiles were generated using ClockLab Analysis software for 14 contiguous days of LD and DD lighting conditions (ActiMetrics; Wilmette, IL). Other activity parameters were assessed over 14 days of LD or DD conditions with the assistance of ClockLab Analysis software, including phase angle of entrainment, total activity, night/day activity ratios, and average daily bouts of activity. These activity parameters are presented as the mean ±SEM for each experimental group. Analysis was performed using a Student’s
The phase shifting effects of a light-pulse at CT 15 were assessed with the use of the linear regression method described in Daan and Pittendrigh (1976). Regression analysis was performed on 6 days of locomotor actogram data preceding the light pulse and on 6 days of data after the light pulse (starting 3 days after the light treatment). Phase delay values are presented as the mean ±SEM for each experimental group, and significance was assessed via Student’s
Brain Tissue Processing
Animals were killed via cervical dislocation and decapitated under dim red light (<5 lux at cage level) at the noted circadian time points. Brains were isolated and then placed in ice-cold oxygenation media, and cut into 500-µm-thick coronal slices with a Leica tissue slicer (VT1200; Leica; Nussloch, Germany). SCN-containing sections were transferred to a 4% paraformaldehyde solution diluted in phosphate-buffered saline (PBS) for 6 h at 4°C, and then cryoprotected overnight at 4°C in 30% sucrose in PBS (w/v, with 2 mM sodium azide and 3 mM sodium fluoride). Sections were then thin-cut (40 μm) with a freezing microtome and transferred into PBS containing 2 mM sodium azide and 3 mM sodium fluoride, pH 7.4.
Immunolabeling
For immunohistochemical labeling, SCN-containing sections were initially treated with 0.3% H2O2 in PBS for 20 min at room temperature, and then blocked and permeabilized in 10% goat serum/PBS containing Triton-X 100 (0.2%) for 1 h. Sections were then incubated overnight at 4°C with the following antibodies (all raised in rabbit): pCREB (1:1000 dilution, catalog code: 9198, Cell Signaling Technology, Danvers, MA); CREB (1:1000; 9197, Cell Signaling Technology), pERK (1:3000; 9101, Cell Signaling Technology), JunB (1:250; 3753, Cell Signaling Technology) or Period1 (1:1000; provided by Dr. Steven Reppert; University of Massachusetts). Next, tissue was processed using the Vectastain Elite labeling kit, per the manufacturer’s instructions (Vector Laboratories; Burlingame, CA), and the signal was visualized using nickel-intensified diaminobenzidine substrate (Vector Laboratories). The sections were then washed twice for 5 min in deionized water and mounted onto slides with Permount media (Fisher Scientific). Between each labeling step, tissue sections were washed in PBS (3 times, 5 min/wash). Of note, for the pCREB labeling, we intentionally performed an extended period of DAB development, which provided a high baseline level of labeling in the WT tissue. This approach was used to help differentiate labeling in the S/A CREB mice from labeling in the WT mice.
For immunofluorescence labeling, tissues were permeabilized and blocked, as described above, and then incubated (overnight, 4°C) with 5% goat serum/PBS containing rabbit anti-c-Fos (1:1500; ABE457, Millipore, Darmstadt, Germany). Sections were then incubated (2 h, room temperature) with Alexa Fluor-conjugated antibodies directed against the IgG domain of the rabbit primary antibody (1:1000; Molecular Probes, Eugene, OR). Sections were mounted with Fluoromount-G (0100-01; Southern Biotech, Birmingham, AL).
Brightfield images were captured using a Micromax YHS 1300 camera (Princeton Instruments, Trenton, NJ) connected to a Leica DMIR microscope. Fluorescence images were acquired using a Leica SP8 confocal microscope. Both fluorescent and brightfield images were analyzed using ImageJ software (National Institutes of Health; Bethesda, MD).
Image Quantitation
Immunopositive c-Fos, JunB and Period1 cells were counted throughout the SCN using ImageJ. Initially, an intensity threshold cutoff filter was applied to all images to remove background labeling, and positive cells above the defined threshold were quantified. The total number of positive cells were averaged from 2 central SCN sections per animal. Mean group data were generated, and statistical significance was assessed using a 2-way ANOVA followed by Bonferroni multiple comparison tests using GraphPad Prism 6.0. Data are presented as the group mean ±SEM.
Cresyl Violet Staining
SCN-containing sections were mounted on gelatin-coated slides and dried. Tissues were rehydrated using a graded alcohol series and then placed in water. Tissues were then stained with 0.3% cresyl violet solution in water for 20 min, and then de-stained with 95% ethanol containing 0.1% glacial acetic acid. Sections were then dehydrated, cleared with xylenes, and mounted with Permount.
Slice Imaging
Twenty six-day old
Slice Analysis
Venus expression for whole SCN and single cells was initially analyzed using ImageJ to generate mean Venus expression values. For the whole SCN analysis, a region of interest was drawn around the SCN and a separate region lateral to the SCN within the hypothalamus was drawn for each slice. These regions were used to measure mean Venus expression for each time frame of the 10-day (pre- and post-serum treatment) imaging experiment. Mean lateral hypothalamus Venus expression was then subtracted from the SCN to generate a normalized Venus value for each hour of the experiment. This process was performed for slices from 5 animals for S/A CREB and WT genotypes. Single-cell Venus expression profiling was performed using the circadian gene expression (CGE) plug-in within ImageJ (Sage et al., 2010). The radius of the cell nucleus was defined as 10-pixels, which determined the region of interest drawn for the CGE plug-in.
Period and amplitude of the SCN and individual cells within the SCN were calculated using MetaCycle (Wu et al., 2016). MetaCycle parameters were as follows;
The phase of individual cells within the total SCN was defined by peak Venus expression within a defined 24-h period before and after serum treatment for 50 cells. The phases of the cells were descriptively presented by generating raster plots in Microsoft Excel. To quantitatively assess the mean phase of cells within a given condition and genotype (i.e., pre/post serum, SA-CREB/WT), Rayleigh plots were generated using the open source MATLAB toolbox CircStat (Berens, 2009). The hour of peak
Results
The studies described here utilized a knock-in mouse model in which the serine residue located at position 133 within the transcription factor CREB was replaced with an alanine residue (Wingate et al., 2009). Alanine does not serve as a phospho-acceptor and thus, the functional contribution of phosphorylation at this residue to CREB-dependent transcription can be assessed. Initially, PCR-genotyping was used to confirm that the heterozygous breeding strategy (S/A CREB+/−::S/A CREB+/−) generated offspring that were homozygous for the S/A point mutation (S/A CREB+/+; Fig. 1A). However, consistent with the data from Naqvi et al. (2014), the expected Mendelian frequency of S/A CREB+/+ offspring (1:4) was markedly reduced, indicating lethality in a subset of the S/A CREB+/+ embryos.
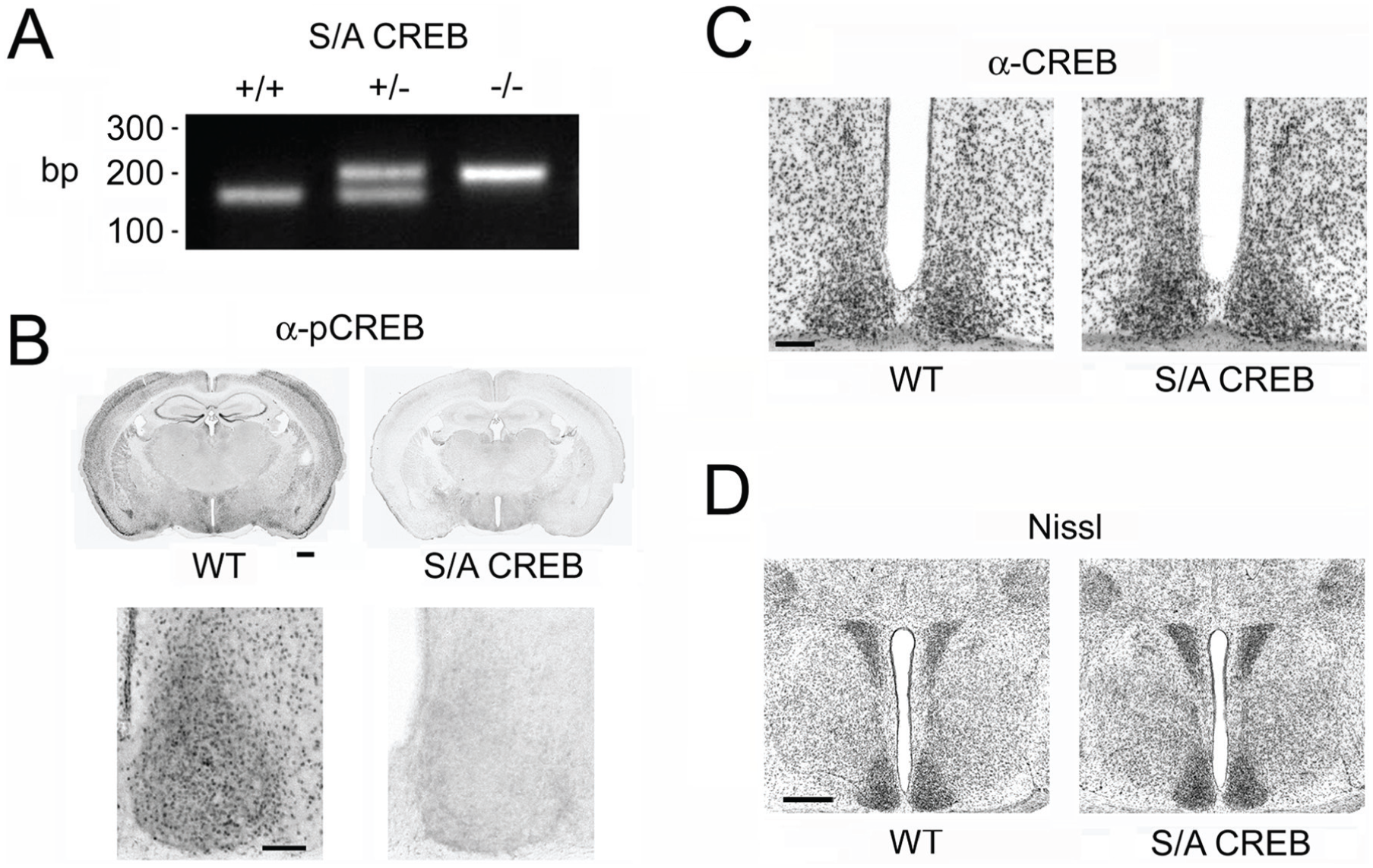
SCN characterization in the S/A CREB mouse line. (A) PCR-based genotyping was used to identify S/A CREB knock-in mice. Mice homozygous for the S/A-CREB transgene (S/A CREB+/+) were generated from crossing S/A CREB heterozygous mice (S/A CREB+/−). The diagnostic PCR results of WT CREB (S/A CREB-/-), S/A CREB+/− and S/A CREB+/+ mice are shown. (B) Immunohistochemical labeling for the phosphorylated form of CREB (pCREB) at Ser133 from WT and homozygous S/A CREB mice (hereafter referred to as “S/A-CREB”). Notably, the expected loss of pCREB immunolabeling was detected in S/A CREB tissue. Bar: 500 microns. Images of the SCN from coronal sections (top panels) are depicted in the bottom panels. Bar: 200 microns. (C) Immunohistochemical labeling for CREB in the SCN in WT and S/A-CREB mice. Of note, CREB expression levels in S/A CREB mice were consistent with the levels observed in WT mice. Bar: 200 microns. (D) Nissl stain from WT and S/A-CREB mice. Bar: 500 microns.
As expected, immunolabeling for the Ser133 phosphorylated form of CREB revealed a complete loss of antigenicity in S/A CREB homozygous mice (from here onward, mice that are homozygous for the S/A knock-in are referred to as “S/A CREB” mice) (Fig. 1B). These data are consistent with previous work, which used Western analysis to show that pCREB antigenicity was eliminated in cortical cultures from S/A CREB mice (Wingate et al., 2009). In contrast, total CREB levels in the SCN appeared to be consistent between the S/A CREB mice and WT (C57BL/6) mice (Fig. 1C). Thus, deletion of Ser133 phosphorylation did not affect the expression of the alanine-mutated form of CREB.
Through gross morphological assessment of the SCN and surrounding hypothalamic tissue via Nissl staining, we did not detect any marked differences between the S/A CREB mice and WT littermates (Fig. 1D). Likewise, visual acuity tests, tests of arousal, and locomotor activity tests did not detect any marked abnormalities in the S/A CREB mice (data not shown). Together, these findings indicate that the elimination of the Ser133 residue did not result in marked developmental or neuroanatomical abnormalities within the SCN.
Wheel running activity was used to assess SCN clock timing and the entraining effects of light. Initially, mice were placed on a 12 h light/12 h dark (LD) cycle and activity during the light and dark phases was assessed. Actograph analysis revealed striking differences in both the amount and distribution of locomotor activity between the WT and S/A CREB mouse lines. As expected, WT mice exhibited consolidated and long-lasting bouts of activity during the early part of the dark phase (Fig. 2A and 2B). In contrast, S/A CREB mice exhibited fragmented bouts of activity that were distributed throughout the dark phase (Fig. 2A and 2B). The phase angle of entrainment of locomotor activity following lights-off at ZT12 was significantly delayed in S/A CREB mice (Fig. 2C1). Consistent with this delayed phase angle of entrainment, often, the first extended bout of activity was not observed until several hours into the dark phase (Fig. 2A). As a corollary, acrophase analysis revealed that peak locomotor activity in S/A CREB mice was significantly delayed, by approximately 2 h, relative to WT mice (mean acrophase: WT: CT16.28 [±0.11] h, and S/A CREB: CT18.26 [±0.19] h;
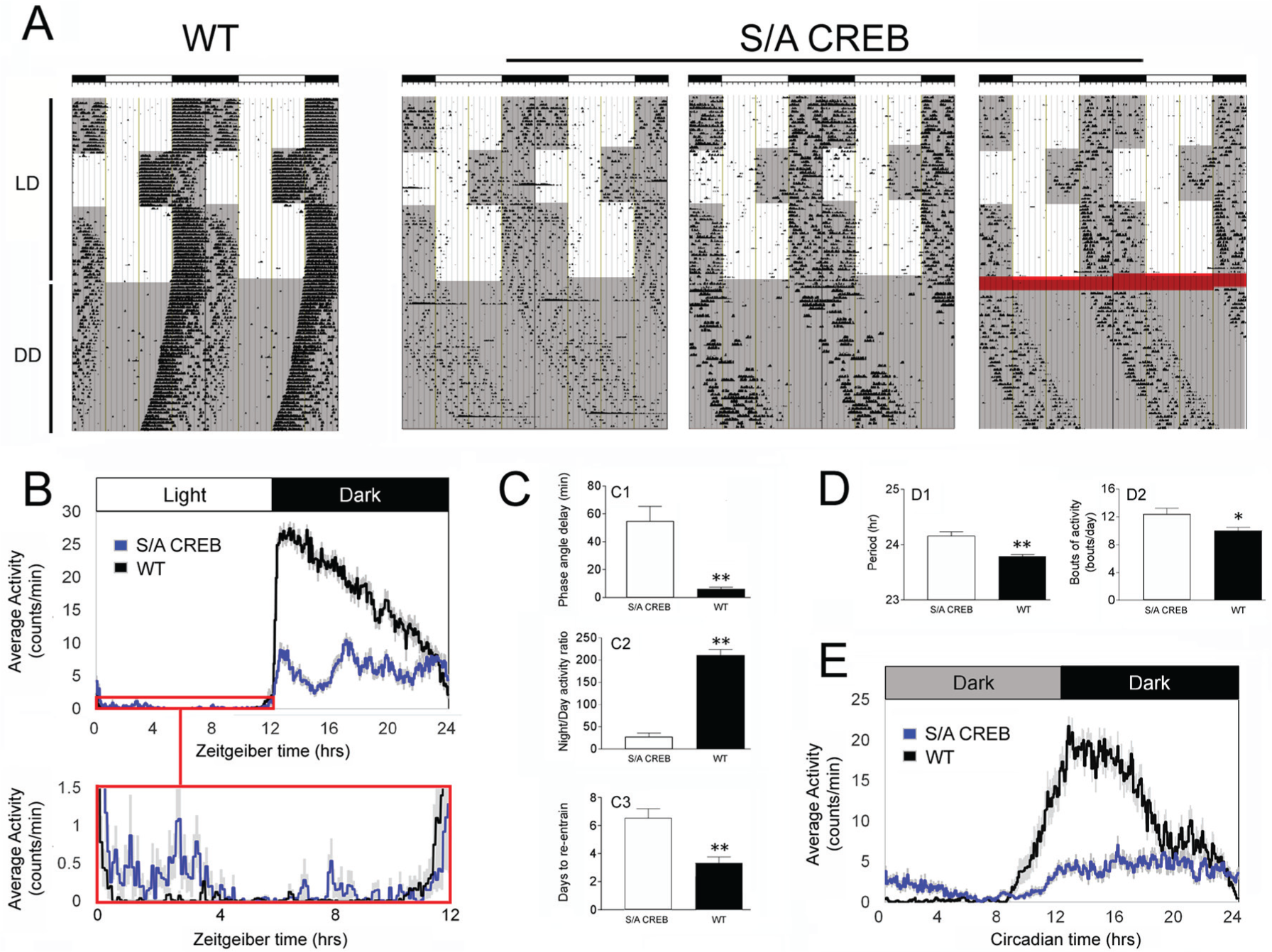
Light- and clock-gated locomotor activity of S/A CREB mice. (A) Representative double plotted actographs of wheel running activity from one WT and 3 S/A CREB mice. Darkened regions of the plots denote periods when the lights were off. Mice were initially entrained to a 12:12 h light/dark (LD) condition, then the light cycle was phase-advanced, and phase delayed by 6 h, and finally, mice were transfer to constant darkness (DD). The S/A CREB mice exhibited variable activity onset under the LD condition, and a long tau phenotype under DD. The red highlight in the far-right trace denotes a period of disrupted/intermittent data collection. (B) Mean daily activity profiles of WT and S/A CREB mice in LD; the boxed region (red) in the top panel is magnified in the lower panel. With the expanded scale, a marked relative increase in daytime activity in S/A CREB mice can be visualized. (C) Quantification of the mean (±SEM) phase angle of entrainment (C1), the night/day activity ratio (C2), and the days to re-entrain to the 6 h phase advance (C3) for WT and S/A CREB mice. (D) Mean circadian period (D1), and the average daily bouts of activity (D2) in DD are presented for WT and S/A CREB mice. (E) Average daily activity profiles of WT and S/A CREB animals in constant darkness. Data in B to E were averaged from 12 to 13 animals per genotype. **
To assess the impact of the S/A CREB mutation on period (tau) of the SCN oscillator, mice were transferred from the LD cycle to total darkness (DD) and allowed to free-run for over 20 days (Fig. 2A). WT mice exhibited an average tau of 23.78 h, which is consistent with published data on C57BL/6 mice (Schwartz and Zimmerman, 1990) (Fig. 2A and 2D1). In contrast, S/A CREB mice exhibited a long tau phenotype of 24.34 h, which was significantly different from WT mice (Fig. 2D1). Furthermore, S/A CREB mice had significantly more fragmented activity during DD (Fig. 2D2), and their overall activity in DD was reduced relative to WT mice (Fig. 2E; mean wheel rotations/5 min binned period: WT: 40.68 [±6.78] and S/A CREB: 17.36 [±5.24];
To further test the idea that S/A CREB mice exhibited disrupted light entrainment, we assessed the re-entrainment rate to a 6 h advance and a 6 h delay in the light cycle. Here, we show that WT mice exhibited a ~3.4-day entrainment to the 6 h advance in the light cycle (Fig. 2A and 2C3). In contrast, a large percentage of S/A CREB mice showed difficulty resetting to a new light cycle (Fig. 2A). Along these lines, even after 10 days in the new light cycle, ~ 40% of S/A CREB mice (
To further address the light entrainment properties of the S/A CREB mice, we used an Aschoff type I paradigm (Aschoff, 1965), which allowed us to test the resetting response to an acute phase delaying (early night) light pulse. To this end, mice were dark-adapted for 21 days, and then exposed to a light pulse (50 lux, 15 min) at circadian time 15 (CT15). With this paradigm, WT mice exhibited a phase delay of 97.39 (±8.34) min; in contrast, S/A CREB mice exhibited a significant relative reduction in the phase-delaying effects of light, as their average shift was only 27.69 (±6.10) min (Fig 3A and 3B). Late-night, phase-advancing light pulses were not utilized, given the relatively weak phase-resetting effects of late-night light observed in C57BL/6 mice, coupled with the weak and fragmented DD free-running rhythm of S/A CREB mice. Mock light treatment at CT15 did not markedly alter the clock phasing of either WT or SA-CREB mice (data not shown). Together, these data reveal a critical role for CREB phosphorylation at Ser133 in the coupling of light to entrainment of the circadian clock.
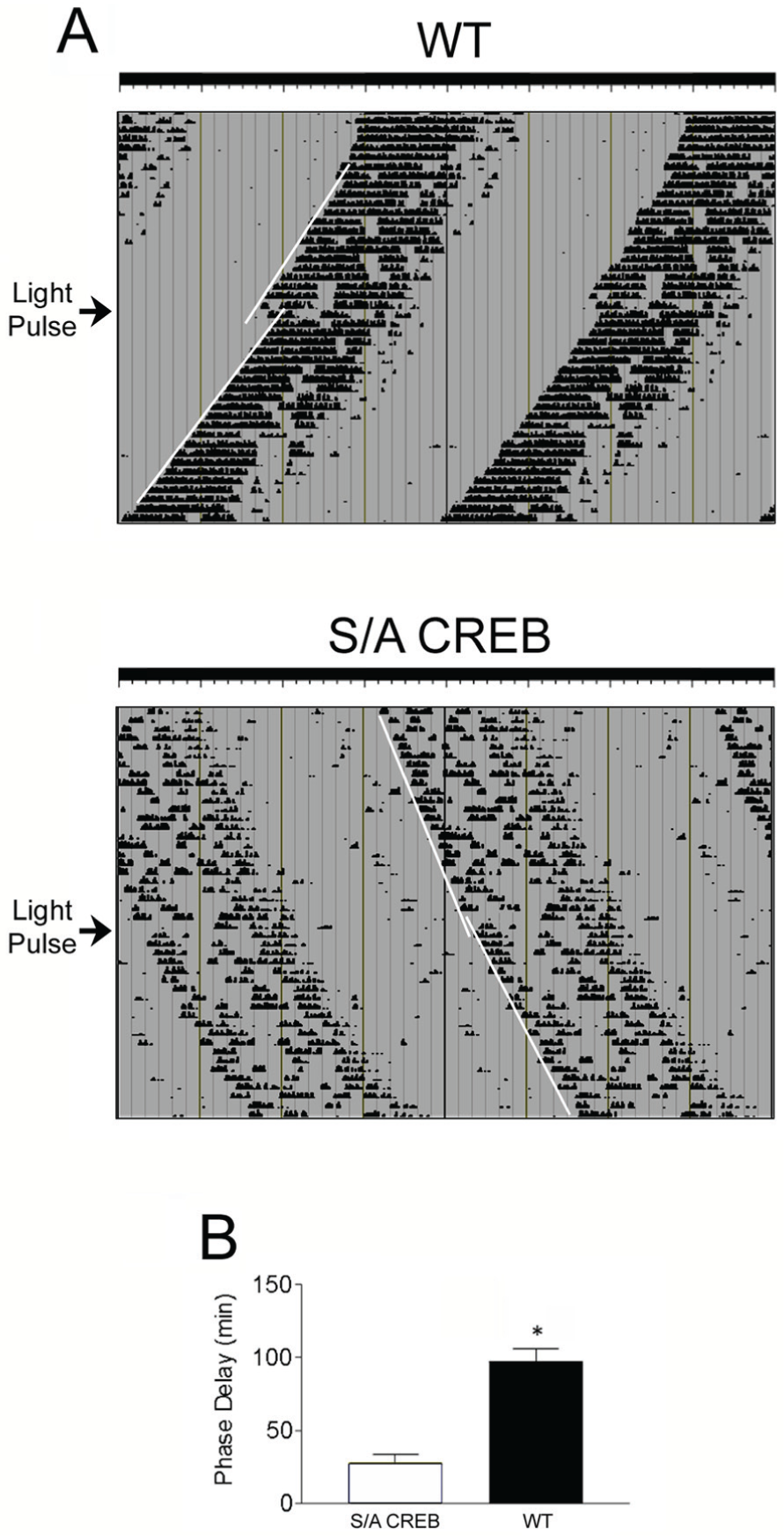
Light-evoked phase delaying is attenuated in S/A CREB mice. (A) Mice were dark adapted and exposed to light (100 lux, 15 min) at CT 15 and allowed to free run for an additional 14 days. The arrow to the left of each trace denotes the day of the light pulse, and the regression lines denote activity onset before and after the light pulse. (B) Mean (± SEM) phase delay, in minutes, is depicted for the WT and S/A CREB mice. S/A CREB mice had significantly attenuated entrainment relative to WT mice.
Next, we assessed the effects of the S/A mutation on inducible gene expression in the SCN. To this end, we profiled the expression of c-Fos, JunB and the core clock protein Period1 (Figs. 4 and 5). Mice were initially dark-adapted for 2 days, exposed to light (50 lux) for 15 min at CT15 and then sacrificed either 45 min later (to profile c-Fos and JunB) or 4 h later (to profile Period1). These time courses are in accordance with the reported light-evoked expression profiles for these 3 proteins (Aronin et al., 1990; Rusak et al., 1990, 1992; Kornhauser et al., 1992; Yan and Silver, 2004). In WT mice, light triggered the expected increase in all 3 immediate early genes. Interestingly, the evoked expression of all 3 genes was significantly blunted in S/A CREB mice (as measured by the number of immunopositive cells for each protein). Notably, fold-induction of light-evoked c-Fos expression was reduced by ~63% in S/A CREB mice, relative to fold induction observed in WT mice (Fig. 4A). Similarly, a ~43% relative decrease in JunB induction was observed in S/A CREB mice (Fig. 4B). Likewise, the fold photic induction of the core clock gene Period1 was reduced by ~43% in S/A CREB mice, relative to the fold induction in WT mice (Fig. 5A). Finally, basal expression of cFos, JunB, and Period1 (at CT15) were not significantly different between S/A CREB and WT animals.
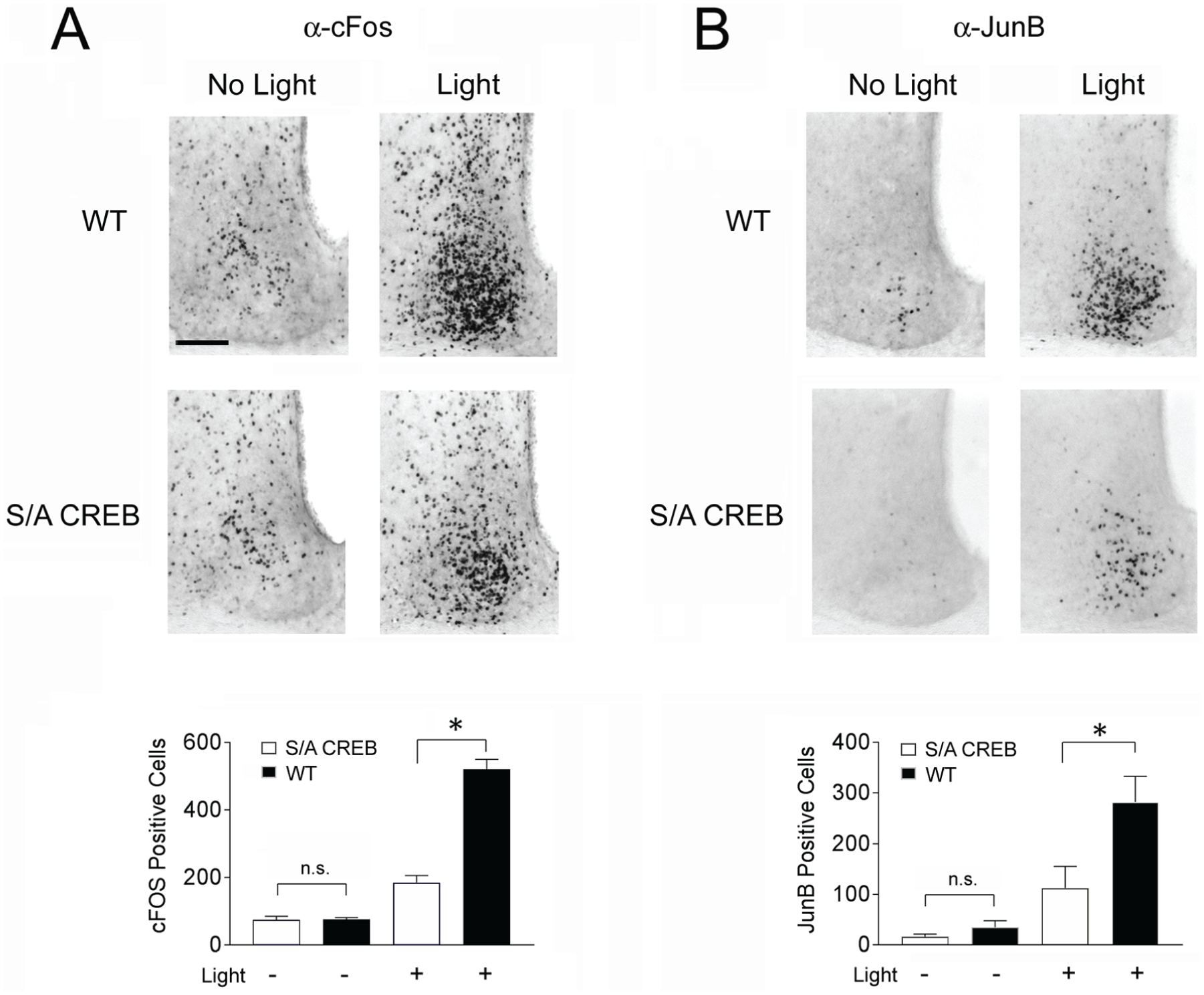
Light-evoked immediate early gene expression is attenuated in S/A CREB mice. For the analysis of cFos (A) and JunB (B), WT and S/A CREB mice were pulsed with light (50 lux, 15 min) at CT15 and sacrificed 45 min later. Representative images of the SCN reveal marked light evoked cFos and JunB expression in WT mice and a reduced relative level of induction in S/A CREB animals, as determined by the total number of immunopositive SCN cells. Quantification of the mean (±SEM) number of cFos- and JunB-immunopositive cells is provided in the lower panels. Bar: 100 microns. Data were analyzed via a one-way ANOVA followed by the Bonferroni multiple comparison test; post-hoc testing compared the WT and S/A CREB light-pulse conditions. *
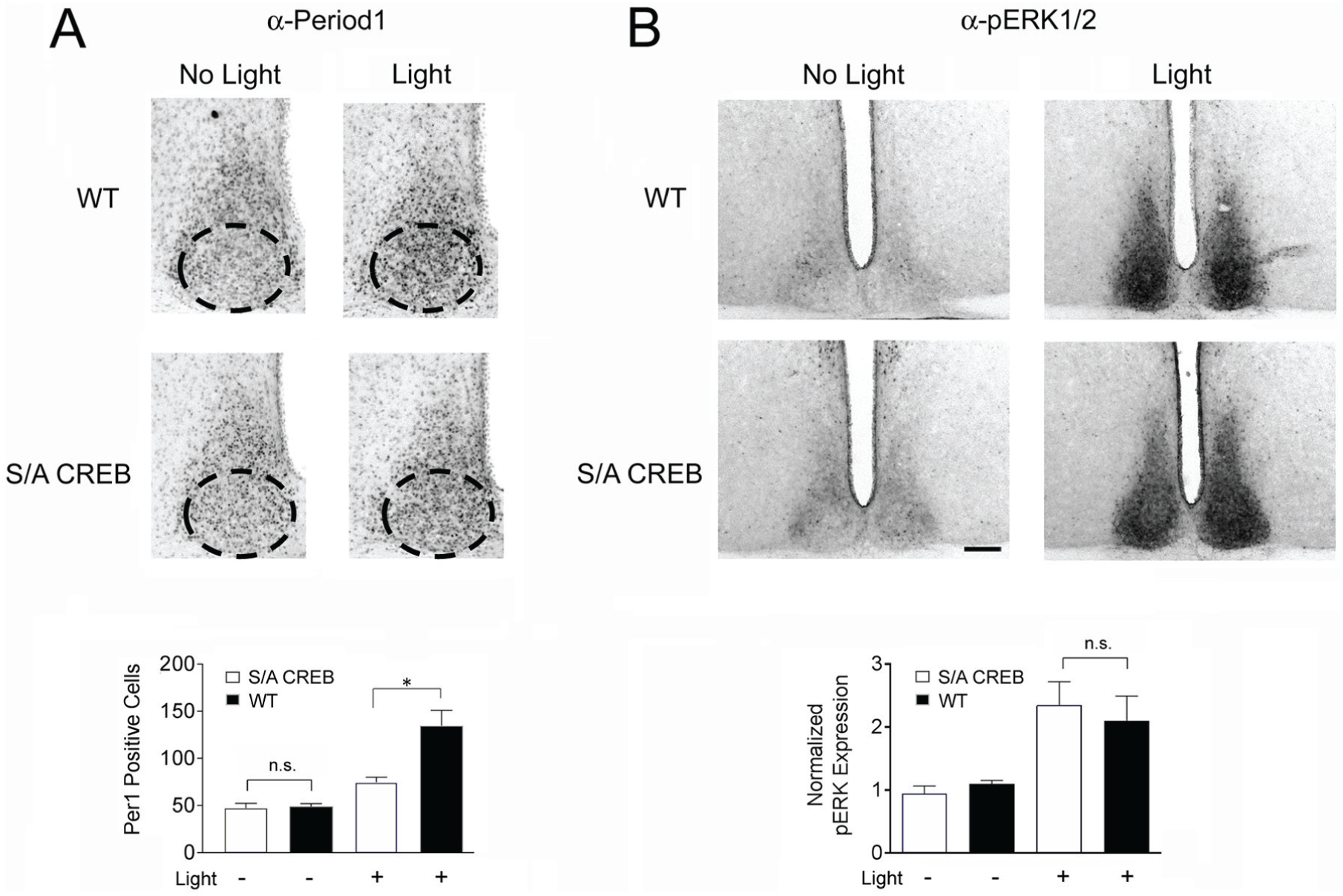
Light-evoked Period1 expression is attenuated in S/A CREB mice. (A) Top panels: WT and S/A CREB mice were pulsed with light (50 lux, 15 min) at CT15 and sacrificed 4 hours later. Within the ventral SCN (denoted by the dashed ovals: top panels), light evoked an increase in Period1 immunopositive cells, an induction that was attenuated in the S/A CREB SCN compared to WT SCN. Bottom panels) Quantification of the mean (+ SEM) Period1 expression within the ventral SCN is quantified. Data were analyzed via a one-way ANOVA followed by the Bonferroni multiple comparison test; post-hoc testing compared the WT and S/A CREB light-pulse conditions. *:
As a control experiment, we also tested whether the S/A CREB mutation could be affecting upstream synaptic circuits or the light responsiveness of SCN neurons. To this end, we profiled the photic response properties of the p44/42 mitogen-activated protein kinase (MAPK) pathway. Of note, the MAPK pathway is highly sensitive to light stimulation during the night-time domain and is a key regulator of Ser133 CREB phosphorylation (Obrietan et al., 1998, 1999). For this experiment, mice were dark-adapted for 2 days and then exposed to light (50 lux, 15 min) at CT15, and the tissue was labeled for phosphorylated forms of ERK1 (tyrosine-202 and threonine-204) and ERK2 (threonine-185 and tyrosine-187). These dual phosphorylation events are essential steps in the activation of the MAPK cascade. In both WT and S/A CREB mice, robust ERK activation was detected, relative to mice that were not exposed to light; further, the level of induction was not significantly different between WT and S/A CREB mice (Fig. 5B). This indicates that the photic response pathway from melanopsin-expressing retinal ganglion cells to the intracellular SCN signaling events upstream of CREB are not markedly altered in S/A CREB mice. Together, the results in this section indicate that CREB phosphorylation at Ser133 plays a key role in light-evoked gene expression in the SCN.
Next, we examined circadian clock-gated expression of the 3 noted genes (c-Fos, JunB and Period1) in the S/A CREB mouse line. The expression of both c-Fos and JunB within the SCN have been reported to be regulated by the circadian clock, with peak levels of each immediate early gene (IEG) expression occurring during the early subjective day (Guido et al., 1996, 1999; Miller et al., 2005). For this experiment, mice were dark-adapted for 2 days, and then tissue was collected at 6-h intervals across the circadian cycle, starting at CT4. In WT mice the expression of both c-Fos and JunB followed the expected pattern, with peak expression within the shell occurring during the early subjective day (Fig. 6A and 6B). Interestingly, in S/A CREB mice, the early day peak in c-Fos and JunB was significantly blunted relative to WT mice (Fig. 6A and 6B).
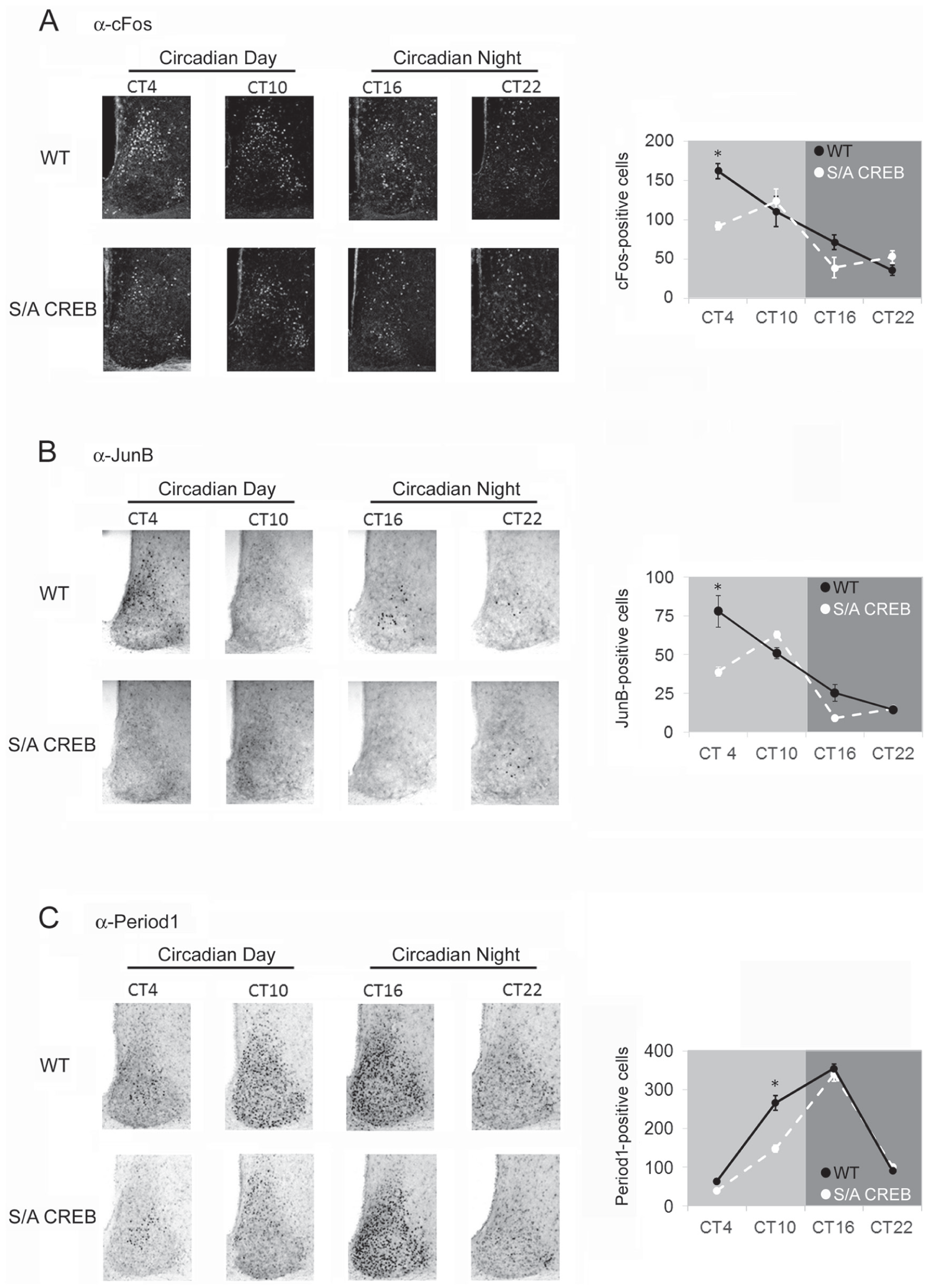
Circadian clock-gated gene expression is altered in S/A CREB mice. Mice were dark-adapted for 2-days and then sacrificed at CT4, CT10, CT16 and CT22. (A) Left panel: Representative immunofluorescence images of c-Fos expression from the SCN of WT and S/A CREB animals. Right panel: cFos-positive SCN cells were quantified and plotted across the circadian cycle; note that, at CT4, the number of cFos-positive cells were significantly reduced in S/A CREB SCN compared with WT animals; *
Rhythms of the core clock protein Period1 within the SCN have been well-documented, with peak levels occurring during the early subjective night (Hastings et al., 1999). Interestingly, in S/A CREB mice, damped Period1 expression was observed during the subjective day (i.e., CT10; Fig. 6C). This reduction was observed throughout the rostro-caudal extent of the SCN. However, by the early subjective night, levels of Period1 in the S/A CREB mice had sharply risen, reaching a level that was not significantly different from the level observed in WT mice (i.e., CT16; Fig. 6C). Together, these data indicate that the S/A CREB mutation leads to damped transcriptional drive during the subjective daytime.
The effects reported here, combined with recent work showing that CREB serves as a nodal point between rhythms in cytoplasmic signaling (e.g, Ca2+, cAMP) and the core clock TTFL, led us to examine the effects of the S/A mutation on both the molecular clock cycling and entrainment properties of the SCN. For this study, we crossed the S/A CREB line with our
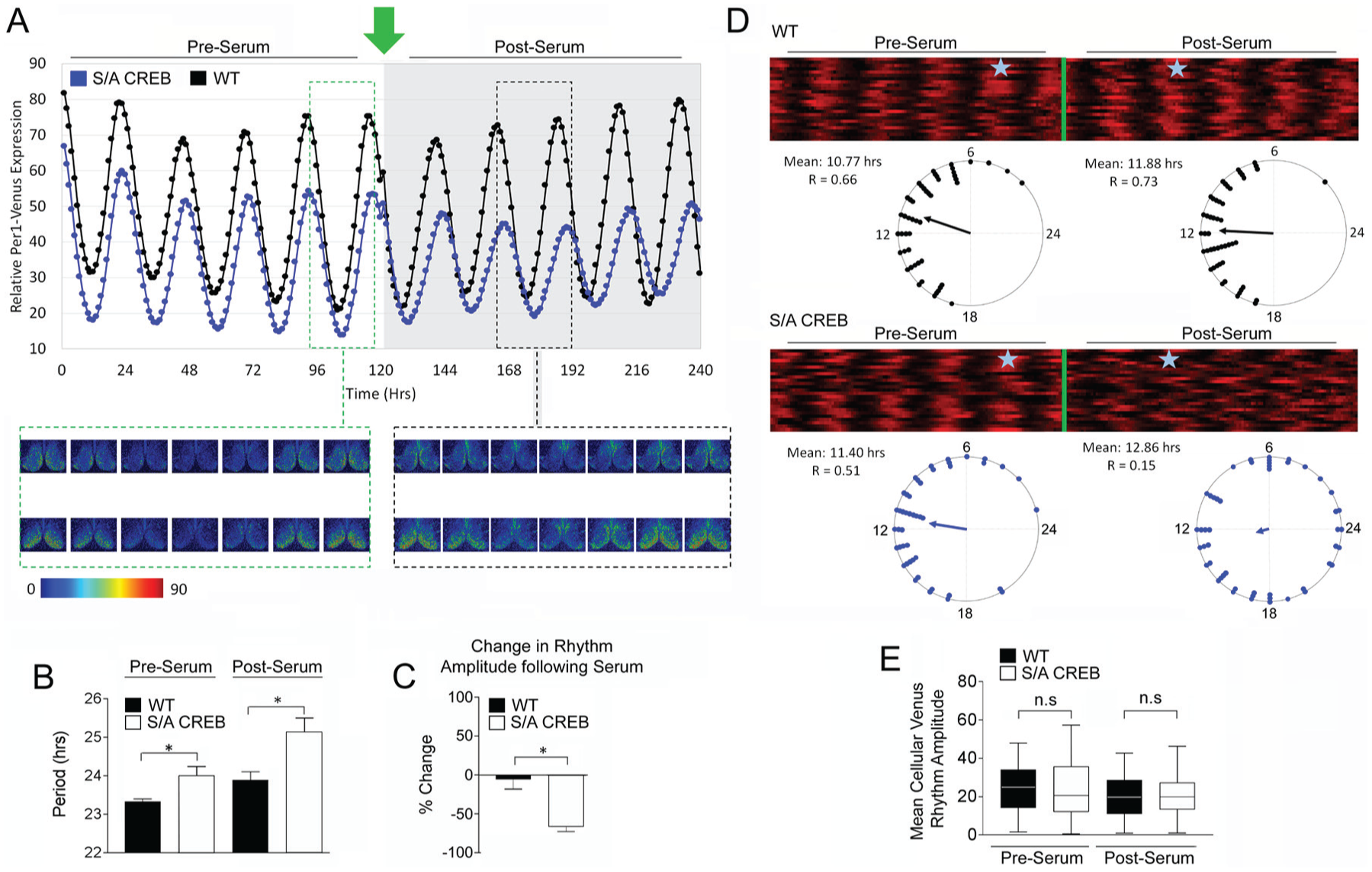
SCN neuronal cell synchrony is reduced in S/A CREB mice.
Following this 120-h baseline recording period, SCN tissue was challenged with a serum shock (20%) approximately 7 h after peak Venus expression, which,
To test whether this damping was the result of a reduction in neuronal cellular oscillator synchrony, we profiled single-cell Venus rhythms in the SCN of WT::Venus and S/A CREB::Venus slices. Raster plot and accompanying Rayleigh plot analyses of the SCN cells demonstrated that before serum treatment, cellular oscillators of both genotypes maintained a high degree of synchrony (Fig. 7D). However, after serum treatment, the phasing of Venus expression in the S/A CREB SCN cells became much more dispersed and desynchronized compared with the Venus expression among the WT SCN cells (Fig. 7D). To determine whether a change in the amplitude of the individual cells also contributed to the dampening of the SCN rhythm amplitude, we measured the single-cell Venus rhythm amplitude. Interestingly, the mean cellular amplitude was not different between the 2 genotypes either before or after serum treatment (Fig. 7E). Together, these data indicate that CREB phosphorylation at Ser133 plays a critical role in the resynchronization/reestablishment of the cellular oscillator network following a resetting stimulus.
Discussion
Overview of the Study: Rationale and Findings
Here, we explored the contribution of CREB phosphorylation at Ser133 to the functionality of the SCN timing system. As noted, CREB phosphorylation is often used to infer induction of CREB-dependent gene transcription. However, to date, the relevance of the Ser133 phospho-acceptor site had not been tested within the context of the circadian timing system. This, combined with work showing that CREB-dependent transcription can be regulated by an array of additional post-translational modifications and cofactors (e.g., CRTCs) (Altarejos and Montminy, 2011; Ch’ng et al., 2012; Nonaka et al., 2014; Parra-Damas et al., 2017; reviewed by Sakamoto et al., 2011), provided us with a compelling rationale to pursue the studies described here.
Initially, our analysis of the gross anatomy of the SCN did not detect any marked changes/deficits in the S/A CREB knock-in mouse line. Further, the expression of CREB in the SCN was similar in S/A CREB and WT mice. This finding is consistent with work from Naqvi et al., (2014), which showed that CREB levels in embryonic fibroblasts derived from S/A CREB knock-in mice were not different from CREB levels in WT embryonic fibroblasts. Interestingly, Naqvi et al., (2014) also reported that the expression of ATF1 and CREM were not altered in the S/A CREB knock-in line. These findings are notable given that the targeted transgenic deletion of CREB can lead to the upregulation of other members of the ATF1 family of bZIP transcription factors (Hummler et al., 1994). Hence, these data indicate that the residual functional activity imparted by the S/A mutant form of CREB is largely sufficient for normal SCN development.
CREB and Light Entrainment
Numerous studies have examined the relationship between photic input and the phosphorylation of CREB at Ser133 in the SCN (Ginty et al., 1993; Obrietan et al., 1999; Gau et al., 2002). Hence, photic stimulation during the subjective night but not during the subjective day stimulates marked CREB phosphorylation, and reporter gene approaches reveal that light actuation of CRE-mediated gene expression in the SCN is limited to the night time-domain (Obrietan et al., 1999). Here, we provide evidence that this phosphorylation event contributes to clock entrainment. Along these lines, the genetic elimination of the Ser133 phospho-acceptor site leads to marked deficits in photic entrainment, including a delay in the phase angle of entrainment, a reduced rate of entrainment to a new light cycle and blunted light-pulse-mediated resetting. Further, our data are consistent with work showing that CREB plays a key role in entrainment of the circadian clock. Along these lines, work by Tischkau et al. (2003), which employed a decoy strategy to deplete CREB from native CRE promoters, found that a reduction in CREB-mediated transcription led to a marked attenuation in the phase shifting effect of light. Likewise, our earlier work using an A-CREB mouse strain, which potently represses CREB binding to the CRE site, led to an attenuation of light evoked clock entrainment (Lee et al., 2010). In the work reported here, it was interesting to find that many mice did not exhibit stable resetting to a 6 h advance in the light cycle, even after 7 days on the new cycle. Notably, a subset of the mice that were able to reset did so by phase-delaying to reach the new light-off time point. This phenotype could be a result of the delayed phase angle of entrainment.
The marked alteration in the distribution of locomotor activity across the circadian cycle was a particularly prominent aspect of the S/A CREB phenotype. C57BL/6 mice typically exhibit a robust bout of activity during the first half of the dark period and this activity tails-off in the second half of the night. In contrast, S/A CREB mice exhibited a nearly equal distribution of locomotor activity during the 2 halves of the night. Although some of this redistribution of activity could be accounted for by the negative phase angle of entrainment, if data averaging takes this into account, there is, nevertheless, still more robust activity during the second half of the night in the S/A CREB mice than in the WT animals. Possible explanations for this could include differential S/A CREB-mediated dysregulation of evening (E) and morning (M) oscillator populations, where the functioning of the E population is more affected in the S/A CREB mice. Interestingly, recent work has shown that the rhythms of these 2 distinct populations are important for determining the photoperiod of onset and offset of activity (Inagaki et al., 2007). Alternatively, disruption of CREB signaling may differentially affect transcriptional control mechanisms that are selectively recruited during the first half of the night and the second half of the night, which in turn, could lead to a reduction in early night activity; clearly additional experiments are merited.
In addition to the redistribution of night-time activity, variable low amplitude locomotor rhythms were another characteristic feature of the S/A CREB line. There are several potential explanations for this, including a reduction in the strength of the SCN oscillator, and a reduction in clock-gated output from the SCN. As noted, CREB-mediated signaling affects the expression of several genes (e.g., AVP, VIP, somatostatin) that could shape the functional properties of the SCN (e.g., SCN cellular communication and SCN clock output) (Montminy and Bilezikjian, 1987; Deutsch et al., 1988; Pardy et al., 1992). Further, CREB-mediated signaling is implicated in clock cell communication and, in turn, regulation of TTFL amplitude and the synchrony of cellular oscillators (Lee et al., 2010; Brancaccio et al., 2013). As such, the S/A CREB mutation may reduce the amplitude and the consolidation of locomotor output by damping TTFL oscillations and/or dysregulating the effective coupling of SCN cellular oscillator populations; a phenotypic effect that was detected using slice imaging methods (see below for an additional discussion of this point).
The noted photic entrainment phenotype in the S/A CREB mice was complemented by data showing marked suppression of light-evoked gene expression. The reduction in light-evoked c-Fos and JunB is in line with the well-characterized role of CREB in the inducible regulation of these 2 IEGs (Sheng et al., 1990; Chan et al., 1999). Consistent with this, the 5’-regulatory regions of both c-Fos and JunB have well-characterized CREB binding motifs, and reporter gene assays have shown that the suppression of CREB signaling reduces the inducible expression of both genes (Härtig et al., 1991; Nakajima et al., 1993; Ginty et al., 1994; Phinney et al., 1994). Interestingly, the role of Ser133 phosphorylation at the level of IEG expression was recently characterized using embryonic fibroblasts from the S/A CREB mouse line. Naqvi et al. (2014) found that the inducible expression of numerous IEGs, including NOR1 and Nur77, are blunted by the S/A CREB mutation. Conversely, the expression of other IEGs, including Nur1 and ICER, were not affected by the S/A CREB mutation. Clearly these data speak to the tightly regulated, context-specific role that Ser133 phosphorylation plays in regulating gene expression. Here, it is worth noting that, in addition to reducing transcriptional drive, the disruption of CREB signaling may have subtle effects on the kinetics of IEG expression (see Naviq et al., 2014); hence, a multi-time point analysis would be needed to determine whether an alteration in the temporal profile of gene induction may have contributed to the light-evoked IEG S/A CREB phenotype reported here.
With respect to the S/A CREB light entrainment phenotype, one of the most straightforward mechanistic explanations is centered on a blunted induction of the core clock gene
Circadian Timing and Clock-driven Gene Expression
Clock-gated IEG expression is a well-characterized phenomenon. Along these lines, c-Fos and JunB exhibit a marked increase in expression within the dorsal SCN during the early subjective daytime (Schwartz et al., 2000). Here we found that this early subjective day peak in IEG expression was blunted in the S/A CREB mouse line. Interestingly, the rhythmic expression of c-Fos is reliant on an early-day increase in cAMP within the dorsal regions of the SCN in a manner dependent on RGS16 (
CREB, Cellular Oscillator Synchrony and Circadian Timing
Clock-gated regulation of CREB/CRE transcription, initially reported in
Interestingly, our slice culture data indicated a clear role for Ser133 phosphorylation in cellular level clock synchrony. This effect was revealed by using a serum challenge and monitoring the phasing of individual cellular oscillators. The S/A CREB phenotype, which showed a significant desynchronization of cellular oscillators following stimulation, is similar to the phenotypic effects resulting from a loss of VIP (vasoactive intestinal peptide) signaling (Maywood et al., 2006; Brown et al., 2007; Hughes et al., 2008). As a putative downstream effector of VIP signaling, CREB/CRE-mediated transcription would be well positioned to function as a regulator of clock cell communication. Damped drive at the level of CRE-mediated transcription would affect the expression of a wide range of genes that could underlie the maintenance of clock cell synchrony. Additional experiments that probe the functional effects of CRE-mediated gene expression would likely provide important clues regarding the transcriptional events that underlie the stability of SCN pacemaker circuits.
Finally, it is important to note that the phosphorylation of Ser133 is only one of several mechanisms by which CREB could drive transcriptional activity within the SCN. Indeed, both the phosphorylation of Ser144 and the activation of CRTCs (cAMP-regulated transcriptional co-activators) have also been implicated in clock physiology. Interestingly, Ser144 to alanine knock-in mice were reported to have a light entrainment deficit; however, in contrast with the work reported here, no effect on inherent clock timing or the overall levels of locomotor activity were found in Ser144 knock-in mice (Gau et al., 2002). CRTCs are potent modulators of CREB-dependent transcription that function via a mechanism that is independent of Ser133 phosphorylation. CRTC nuclear translocation (a marker of activation) is gated by circadian time and by photic input (Sakamoto et al., 2013), and CRTCs have been implicated in circadian clock entrainment (Jagannath et al., 2013). Clearly, a more comprehensive series of experiments are needed to effectively parse the ways in which CREB signaling shapes the functional fidelity of SCN timing and clock entrainment. As a step towards this deeper level of understanding, the data presented here supports a critical role for CREB phosphorylation at Ser133 in both clock timing and entrainment.
Footnotes
Acknowledgements
Grant Sponsors: National Institutes of Health (Grant codes: MH103361, NS066345, NS091302) and the National Science Foundation (Grant code: 1354612).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
