Abstract
Patterns of reproductive seasonality in the Carnivora are difficult to study comparatively, due to limited numbers of species for which information is available. Long-term databases of captive populations could overcome this difficulty. We apply a categorical description and a quantitative high-resolution measure (birth peak breadth, the number of days in which 80% of all births occur) based on daily observations in captivity to characterize the degree of reproductive seasonality in the Carnivora for 114 species with on average 1357 births per species. We find that the majority of species retained the birth seasonality displayed in the wild. Latitude of natural origin, delayed implantation, and induced ovulation were the main factors influencing reproductive seasonality. Most species were short-day breeders, but there was no evidence of an absolute photoperiodic signal for the timing of mating or conception. The length of the gestation period (corrected for body mass) generally decreased with birth seasonality but increased in species with delayed implantation. Birth seasons become shorter with increasing latitude of geographical origin, likely because the length of the favorable season declines with increasing latitude, exerting a strong selective pressure on fitting both the reproductive cycle and the interval offspring needs for growth following the termination of parental care into the short time window of optimal environmental conditions. Species with induced ovulation exhibit a less seasonal reproductive pattern, potentially because mates do not have to meet during a short time window of a fixed ovulation. Seasonal species of Carnivora shorten their gestation period so reproduction can occur during the short time window of optimal environmental conditions. Alternatively, other Carnivora species lengthen their gestation periods in order to bridge long winters. Interestingly, this occurs not by decelerating intrauterine growth but by delaying implantation.
Keywords
Seasonality is the phenomenon of recurrent fluctuations in climatic conditions and environmental productivity over the year (Lindstedt and Boyce, 1985). All animals have to deal with changing seasons to some degree. One possibility to cope with the changes in climate and environment is the evolution of corresponding reproductive seasonality (Bronson, 1989). Timing reproductive events is crucial for maximizing reproductive success, as it increases the chances of survival of the offspring (Soto et al., 2004) and enhances future reproduction of both mothers and offspring (Lancaster and Lee, 1965; Thomas et al., 2001).
It is well established from studies of free-living populations that patterns of reproductive seasonality in the Carnivora show an extreme degree of variation. Some species are strictly seasonal breeders, where almost all births occur during a narrow time window of less than 2 months (e.g., Atkinson, 1997; van Dalum, 2013); others are capable of reproducing throughout the year but nevertheless show seasonal peaks in reproductive activity (e.g., Hunter and Barrett, 2012; Larivière and Calzada, 2001); and a third group of species exhibits no apparent seasonal variation at all in reproductive patterns (e.g., Smuts et al., 1978; Wilson and Mittermeier, 2009).
The factors causing this large variation are less well known. To date, few studies have compared seasonality in reproduction systematically between different mammalian species. This is particularly true for the Carnivora, where detecting seasonal reproductive behavior of many species in the wild is difficult for several reasons. Many of these animals are elusive (e.g., Bischof et al., 2014), are nocturnal (e.g., Mudappa, 2013), have a low abundance (e.g., Hawkins and Racey, 2005), live in remote habitats (e.g., Jackson et al., 2006), or give birth in secluded dens that prevent observations and involve substantial risk to both the researcher and the study animal if entered (e.g., Godfrey et al., 2000). Further complicating research efforts, several species have a relatively low reproductive rate (e.g., Brazil and Goudie, 2006; Bunnell and Tait, 1981) and must be studied for many consecutive years.
Long-term databases of captive populations help to overcome the mentioned difficulties, as they provide very precise birth records for many individuals, but it is unclear whether the reproductive seasonality in captive populations reflects the patterns of free-ranging populations (Fairall, 1968). For the Carnivora, the majority of studies published so far provide only some exemplar comparisons of seasonality in reproduction between wild and captive populations based on limited samples. A study including the 8 species of Ursidae found that neither the mean nor median date of estrus and parturition differed between captive and wild populations of the same species within the same climate zone, suggesting that the constant provision of shelter and food throughout the year did not affect reproductive seasonality (Spady et al., 2007). Similar findings were derived from single-species studies (Freeman and Braden, 1977; Painer et al., 2014; Peterson and Bartholomew, 1967; Spotte and Adams, 1981; Zuckerman, 1952). These species retain their seasonal reproduction in captivity, sometimes even if kept in zoos at different latitudes compared with the natural populations. In contrast, some Felids such as the European wildcat (Felis silvestris), leopard (Panthera pardus), or serval (Leptailurus serval), or small canid species such as fennec fox (Vulpes zerda), bush dog (Speothos venaticus), and crab-eating fox (Cerdocyon thous) changed their breeding patterns from seasonal in the wild to nonseasonal or less seasonal in captivity (Brady, 1978; Cociu and Cociu, 1976; Porton et al., 1987; Skinner et al., 2002; Valdespino et al., 2002). Notably, no reports of change to an increased breeding seasonality in captivity seem to exist.
Due to these contrasting results and the limited data available for the Carnivora, the first aim of this paper is to conduct a broad comparison of reproductive seasonality between wild and captive individuals. The studies mentioned above, as well as observations from the wild, mainly provide categories to describe the seasonality of reproductive behavior (e.g., seasonal vs. nonseasonal) or describe the monthly distribution of birth events (e.g., Cociu and Cociu, 1976; Skinner et al., 2002). We used data from Species360, an organization within the international zoo community that has collected stock data over the last 43 years from more than 1000 participating zoos in 90 countries worldwide. These data include the exact dates of birth for almost all individuals born in captivity since 1980, allowing us to describe reproductive seasonality not only in a categorical way (for a comparison with reports on free-ranging populations) but also quantitatively with a continuous measure, the birth peak breadth, which we define as the number of days within which 80% of all births occur (Zerbe et al., 2012).
Using the categorical classification, we first test whether Carnivora species in captivity retain the birth seasonality they display in the wild or change to a less seasonal birthing pattern. Our second aim is to explore which physiological, ecological, or social factors are linked to reproductive seasonality in the Carnivora and therefore to our quantitative measure, the birth peak breadth. We assess the influence of 7 factors on reproductive seasonality in the Carnivora that have been suggested previously in the literature to play a role in birth seasonality: namely, day length (photoperiodism) (Bradshaw and Holzapfel, 2007, for a review), latitude of geographical origin (Valdespino, 2007; Zerbe et al., 2012), hibernation (Ferguson et al., 2000; Sandell, 1990), sexual size dimorphism or mating system (Valdespino, 2007; Zerbe et al., 2012), home range size (Spady et al., 2007), and presence or absence of delayed implantation (Bartholomew, 1970; Ferguson et al., 1996; Lindenfors et al., 2003) and induced ovulation (Larivière and Ferguson, 2003; Sanderson and Nalbandov, 1973). For a detailed description of all the predictions for these factors, see the Supplementary material.
Our third aim is to test whether reproductive seasonality is linked to life-history parameters such as gestation length. One way that large mammals achieve mating and birthing at a beneficial time of the year in seasonal habitats is to shorten their gestation period by accelerating intrauterine growth so that the whole reproductive cycle occurs during the short time window of optimal environmental conditions (Kiltie, 1988; Zerbe et al., 2012). We therefore expect relatively shorter gestation periods (corrected for body mass) for large-sized species of Carnivora with a more distinct seasonal reproduction. The alternative adaptation for small mammals reproducing at high latitudes would be to lengthen their typically short gestation periods to bridge the lag periods between favorable mating and birthing conditions (Jabbour et al., 1997). Apparently, rather than decelerating intrauterine growth and thus achieving longer gestation periods, small mammals typically evolve delayed implantation (Ferguson et al., 2006; Ferguson et al., 1996; Sandell, 1990; Thom et al., 2004). We therefore predict a relatively longer gestation period for Carnivora species with a more distinct seasonal reproduction if we also include species with delayed implantation.
Methods
We used data of approximately 159,000 captive animals representing 114 different species from 13 orders of the Carnivora derived from Species360 data files (Suppl. Table S1). All available data were used regardless of the geographical location of the corresponding zoological institutions (most of which were in the northern temperate zone). The observation interval was between 1 January 1900 and 31 December 2013, and only records with an exact birth date were used. We considered only those species that had an overall birth count of 50 or more. We subdivided years into 73 consecutive 5-day intervals and counted the total number of births within each of these intervals. For leap years, births from 31 December were allocated to the 73rd interval. The number of births for the first interval of the year was calculated as the average of the second and preceding 73rd interval, as there was a salient increase in birth on 1 January, which is most likely an effect of the reporting management of some zoos. Overall on average, we recorded 1357 birth events per species (range 50 to 14,053; Suppl. Table S1).
Following the same method described by Zerbe et al. (2012), we used the birth peak breadth as a quantitative measure of the degree of reproductive seasonality. The birth peak breadth was defined as the smallest number of successive days (calculated as 5-day intervals) in which 80% of all births occurred; in doing so, we allowed the possibility that this time window might stretch from December into January. When more than one such birth peak occurred, we used the window with the highest number of births for further analyses (Suppl. Table S1). To compare the seasonality patterns of birth between wild and captive individuals, we subjectively assigned each species based on its birthing pattern to 1 of the 5 birth seasonality categories as in Zerbe et al. (2012) (Fig. 1): Category 1, narrow peak (less than approximately 60 days), no births for the rest of the year; Category 2, expanded peak (>60 days) or double peak, no births for the rest of the year; Category 3, peak(s) with small number of births throughout the year; Category 4, births throughout the year, undulating pattern (preferred seasons possible); Category 5, constant births throughout the year without a clear preferred season. Data on categorical birth seasonality in the wild were taken from the literature (Suppl. Table S1). Since transitions between seasonality categories are to some degree subjective, we considered a change of more than one category as a true change in the seasonal birthing pattern between free-ranging and captive populations. Free-ranging populations of cheetahs (Acinonyx jubatus), golden jackals (Canis aureus), wild cats (Felis silvestris), ocelots (Leopardus pardalis), jaguars (Panthera onca), and leopards (Panthera pardus) adjust the length of their birthing season to their geographical location (Hunter and Barrett, 2012; Kingdon et al., 2013; Myers et al., 2006; Nowell and Jackson, 1996; Stein and Hayssen, 2013; Wilson and Mittermeier, 2009), which makes it impossible to assign a particular birth seasonality category to them. We therefore excluded these species from the comparison of seasonality patterns between the wild and zoo data. Little is known about the reproductive seasonality for free-ranging Asian small-clawed otters (Aonyx cinereus), Asiatic golden cats (Catopuma temminckii), Cape genets (Genetta tigrina), Northern tiger cats (Leopardus tigrinus), margays (Leopardus wiedii), and leopard cats (Prionailurus bengalensis). These species were therefore also excluded from all analyses comparing wild and captive birthing patterns.
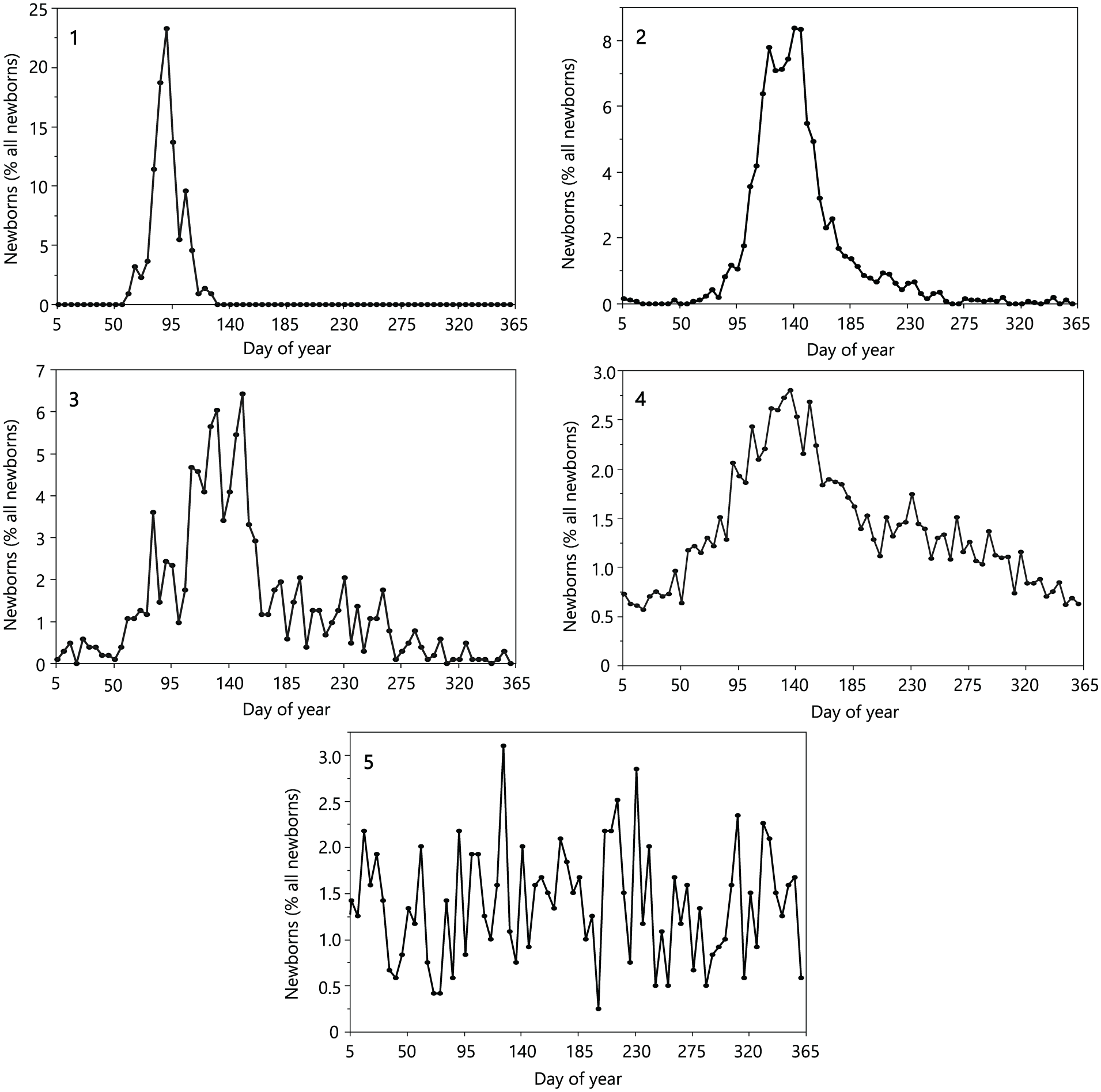
Classification of seasonality categories according to the pattern of birth distributions of captive populations across the year (as divided into 5-day intervals) from 1 = highly seasonal to 5 = aseasonal species: (1) yellow-throated marten (Martes flavigula); (2) snow leopard (Panthera uncia); (3) white-nosed coati (Nasua narica); (4) tiger (Panthera tigris); (5) bush dog (Speothos venaticus).
We retrieved data on body mass, ovulation type, mid-latitude and the latitude range of geographical origin, sexual size dimorphism (expressed as the ratio of male vs. female body mass), mating system (monogamous vs. polygamous), hibernation, home range size, and gestation length, as well as data on the presence or absence and the duration of delayed implantation from the published literature (Suppl. Tables S1 and S2). The red fox (Vulpes vulpes) and swift fox (Vulpes velox) are known to be polymorphic for implantation delays, with delays occurring in only small portions of the species’ range (Hayssen et al., 1993; Larivière and Pasitschniak-Arts, 1996; Lindenfors et al., 2003); thus, they were categorized as not having delays following common practice (Ferguson et al., 1996; Lindenfors et al., 2003; Orr, 2012). We defined the gestation length as the period between implantation and parturition (gestation length without delayed implantation) and the period between conception and parturition (gestation length including delayed implantation) (Suppl. Table S1). We calculated the start of the mating/conception season in captivity and in the wild using data on the start of the birthing period in combination with gestation length. We are aware that this approach disregards potential intraspecific variability of gestation length, as for example documented in domestic dogs (Okkens et al., 1993); however, we consider it acceptable for a broad interspecific comparison. For the comparison of the day length in which mating/conception occurred in the wild and in captivity, only species assigned to the birth seasonality categories 1 and 2 were included (Suppl. Table S3). A second analysis excluding species with delayed implantation was performed, since some studies suggest that in species with delayed implantation, day length controls the timing of the blastocyst implantation and not, or not solely, the date of mating (Boyd, 1991; Enders, 1952; Spady et al., 2007; Temte and Temte, 1993; Woodroffe, 1995). The day length of a specific day of the year, at a specific latitude, was calculated, including civil twilight (which is defined as the time between sunrise and sunset when the center of the sun is 6 degrees below the horizon; light during civil twilight is bright enough to perform ordinary outdoor activities without artificial light), according to Forsythe et al. (1995). For these calculations, the mean latitude of captive populations was assumed to be 45°, as the majority of zoological collections are in the temperate zone.
Statistical analyses were done in JMP 12.0 (SAS Institute Inc., 1989-2016) and in R3.1.3 (R Core Team, 2017). We controlled all analyses for phylogenetic nonindependence using phylogenetic generalized least-squares regressions (PGLS) (Martins and Hansen, 1997) in the “caper” package (Orme, 2013) in R. The phylogeny was based on a composite supertree from Fritz et al. (2009) and is given in Supplementary Figure S1. The red wolf (Canis rufus) is a canid of unresolved taxonomic status (Chambers et al., 2012; Wozencraft, 2005). We therefore retained it here provisionally as distinct and added it to the supertree in Mesquite (Maddison and Maddison, 2001).
To compare the beginning of the mating/conception season in free-ranging and captive populations of clearly seasonal (birth seasonality categories 1 and 2) species, PGLS analyses were performed with day length at conception in the wild as the predictor and day length at conception in captivity as the response variable. Subsequently, the 95% confidence intervals of the corresponding parameter estimates including and excluding species with delayed implantation were calculated.
Testing our non–mutually exclusive predictions to determine the most important explanatory variables of the birth peak breadth, we included the following explanatory variables in the statistical analyses: adult body mass, mid-latitude of geographical origin, sexual size dimorphism or mating system, hibernation, home range size, and presence or absence of delayed implantation. The variable “ovulation type” could not be included in the initial model selection approach, because the ovulation type was unknown for 39 species. Therefore, we tested the effect of induced versus spontaneous ovulation on the birth peak breadth in a restricted dataset (n = 75). To compare coefficients, all continuous predictors were centered (around the mean) and scaled (by the standard deviation) before incorporation in the models (Schielzeth, 2010), but we present raw data in the figures. Model assumptions of normality and homogeneity of residuals were checked by visually inspecting histograms and qq-plots of the residuals as well as by plotting residuals against fitted values. We checked the variance inflation factor (VIF) on the set of variables included in the full models (Dormann et al., 2013) using the function “vif” in the “car” package in R (Fox and Weisberg, 2011). The VIFs in all models were less than 3, which indicates an acceptable amount of covariance among predictors.
We performed information-theoretic model selection based on minimization of the Akaike information criterion corrected for small sample sizes (AICc) (Hurvich and Tsai, 1989) to determine the most important predictors of the birth peak breadth. We ran the model selection across all possible models built with the 6 explanatory variables mentioned above, including those species having complete variable records (n = 102 for models including sexual dimorphism and n = 101 for models including mating system). As the AICc did not clearly distinguish the most highly ranked model for each of the 2 candidate model sets, we accounted for uncertainty in the models using multimodel averaging (Grueber et al., 2011) including models with ΔAICc < 2. For both sets of candidate models, we calculated ΔAICc as the difference in AICc between the focal model and the AICc of the best-fitting model in the candidate model set, which has a ΔAICc of 0. Estimates of each parameter were averaged across the candidate models (means were weighted by the Akaike weight of a given model). The relative importance of a predictor was obtained by summing the Akaike weights of the models in the candidate model set including the focal predictor, following the method described by Symonds and Moussalli (2011).
To investigate whether reproductive seasonality is linked to gestation length, we tested the effect of birth peak breadth and adult body mass on the length of the gestation period (with [n = 114] and without [n = 113] the duration of delayed implantation as well as with species with [n = 31] and without [n = 83] delayed implantation).
Results
Comparison of Reproductive Seasonality between the Wild and Captivity
A significant positive correlation between the ordinal birth season categories and the continuous variable, the birth peak breadth, was found (PGLS: p < 0.001, n = 114, λ = 0.10, β = 0.017, SE < 0.001, t = 28.74), indicating that the quantitative approach of the birth peak breadth and the categorical approach necessary to classify literature data yielded a comparable ranking of species. The birth peak breadth showed a bimodal distribution suggesting that although intermediate forms of reproductive seasonality exist, both strict seasonality and distinct aseasonality are the most common modes in the Carnivora (Fig. 2). The smallest birth peak breadth was 30 days, the largest 281 days.
Distribution of species according to their reproductive seasonality characterized by the birth peak breadth. The birth peak breadth was defined as number of days (calculated as 5-day intervals) in which 80% of all births occurred.
Fifteen of 114 species (13.2%) changed their pattern of reproductive seasonality between the wild and captivity by more than one category. These were (category free ranging/captive) spotted-necked otter (Hydrictis maculicollis) (1/5), smooth-coated otter (Lutrogale perspicillata) (2/5), giant otter (Pteronura brasiliensis) (2/5), crab-eating raccoon (Procyon cancrivorus) (2/5), white-nosed coati (Nasua narica) (1/3), ringtail (Bassariscus astutus) (1/3), kinkajou (Potos flavus) (3/5), marsh mongoose (Atilax paludinosus) (2/4), common kusimanse (Crossarchus obscurus) (2/4), yellow mongoose (Cynictis penicillata) (2/4), jungle cat (Felis chaus) (2/4), sand cat (Felis margarita) (1/4), black-footed cat (Felis nigripes) (2/4), Geoffroy’s cats (Leopardus geoffroyi) (2/4), and fishing cats (Prionailurus viverrinus) (2/4). All these species changed to a less seasonal pattern in captivity and had their median origin at latitudes of 30.4° or less, indicating that changes in the seasonal pattern of reproduction occurred in low-latitude species only.
Factors Associated with Seasonal Reproduction in Wild Carnivora
The slope of the PGLS regression between the day length at the beginning of the mating/conception season in free-ranging and captive populations of seasonal species did not include 1.0 in the 95% confidence interval for the whole dataset and also not after we excluded species with delayed implantation. This indicates that the day lengths at the beginning of reproductive activity in the wild and in captivity were not identical (Fig. 3 and Table 1). Some of the extreme day lengths at the beginning of the mating season in the wild cannot be reached in most temperate zone zoos, but discrepancies between the wild and captivity were not limited to the upper range of day lengths in the wild (Fig. 3).
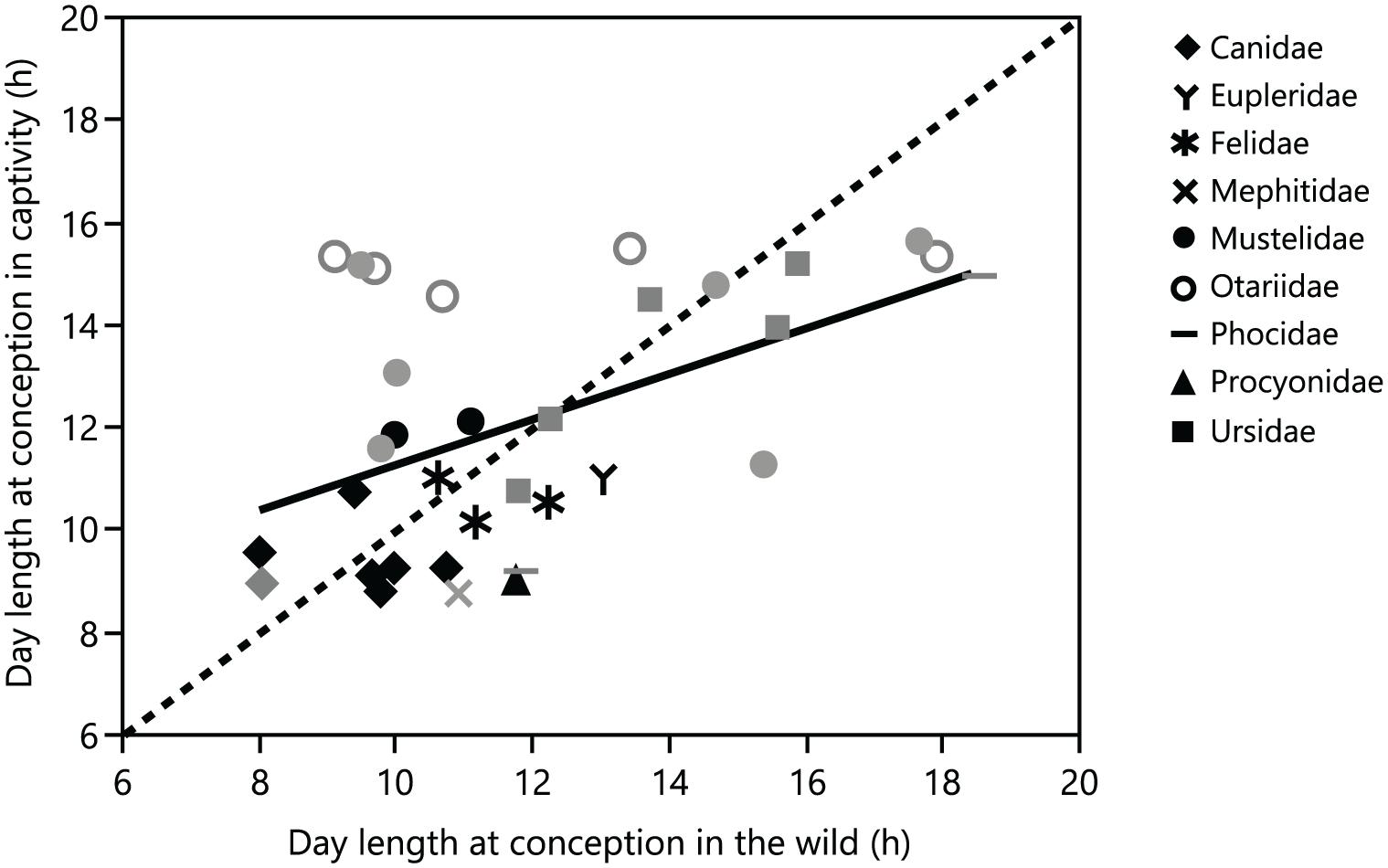
Relationship between the day length at the beginning of mating/conception in captivity and in the wild in 33 seasonal Carnivora species (Suppl. Tables S1 and S3) (gray = species with delayed implantation, black = species without delayed implantation). The linear relationship (solid line) is significantly different from the y = x line (dashed line). For statistics, see Table 1.
Phylogenetic generalized least square regression (PGLS).

PGLS determined according to y = a + bx, with day length at conception in captivity as the dependent variable (y) and day length at conception in the wild as the independent variable (x) (for species selection, see Suppl. Table S3). See Figure 3 for a graphic representation of the data. Results are given with 95% confidence intervals in parentheses.
In captivity as well as in the wild, the majority of seasonal species (categories 1 and 2) were short-day breeders where the day length at the beginning of the mating or conception season is decreasing (33/58 species for the day length estimation in captivity and 21/33 species for the day length estimation in the wild).
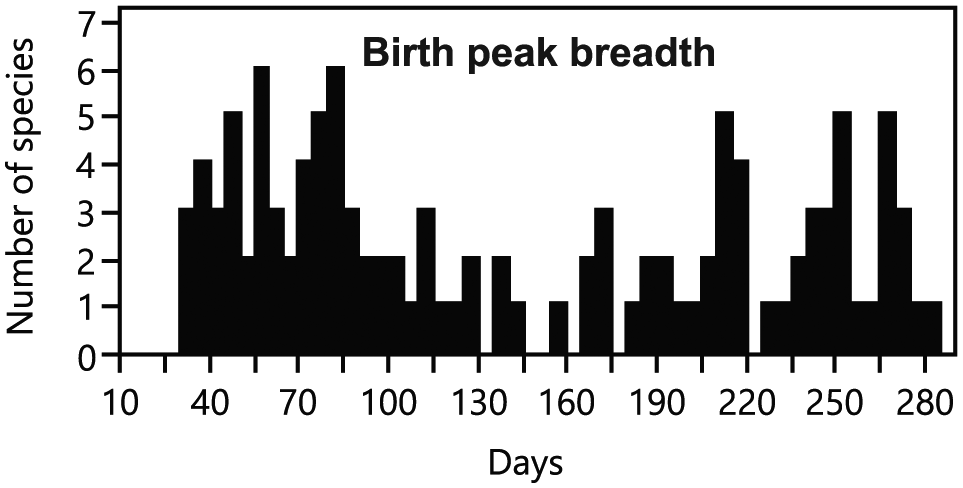
Model selection and averaging showed that the most important variables influencing the degree of birth seasonality were latitude of geographical origin and delayed implantation (relative importance ≥0.69) (Table 2 and Suppl. Tables S4-S6). The birth peak breadth was highly negatively correlated to the latitude of geographical origin, except for one outlier, the sea otter (Enhydra lutris) (Fig. 4). When plotting species on the world map according to their seasonality category, this relationship with latitude is evident (Fig. 5). Results using the breadth of latitudinal origin instead of the mid-latitude of geographical origin are strikingly similar (Suppl. Tables S7 and S8). There was a significant difference in the birth peak breadth between species with and without delayed implantation. Species with delayed implantation demonstrate a much shorter birth peak breadth, indicating that these species have a more seasonal birthing pattern compared with species with no delay in blastocyst implantation (Fig. 6A); species with delayed implantation occurred on average at higher latitudes (Suppl. Fig. S2), and the length of the delay between conception and implantation tended to increase with increasing latitude (Suppl. Fig. S3) and was independent of body mass (Suppl. Fig. S4).
Averaged parameter estimates and their relative explanatory importance for reproductive seasonality (measured as the birth peak breadth in which 80% of all births occurred, n = 102).
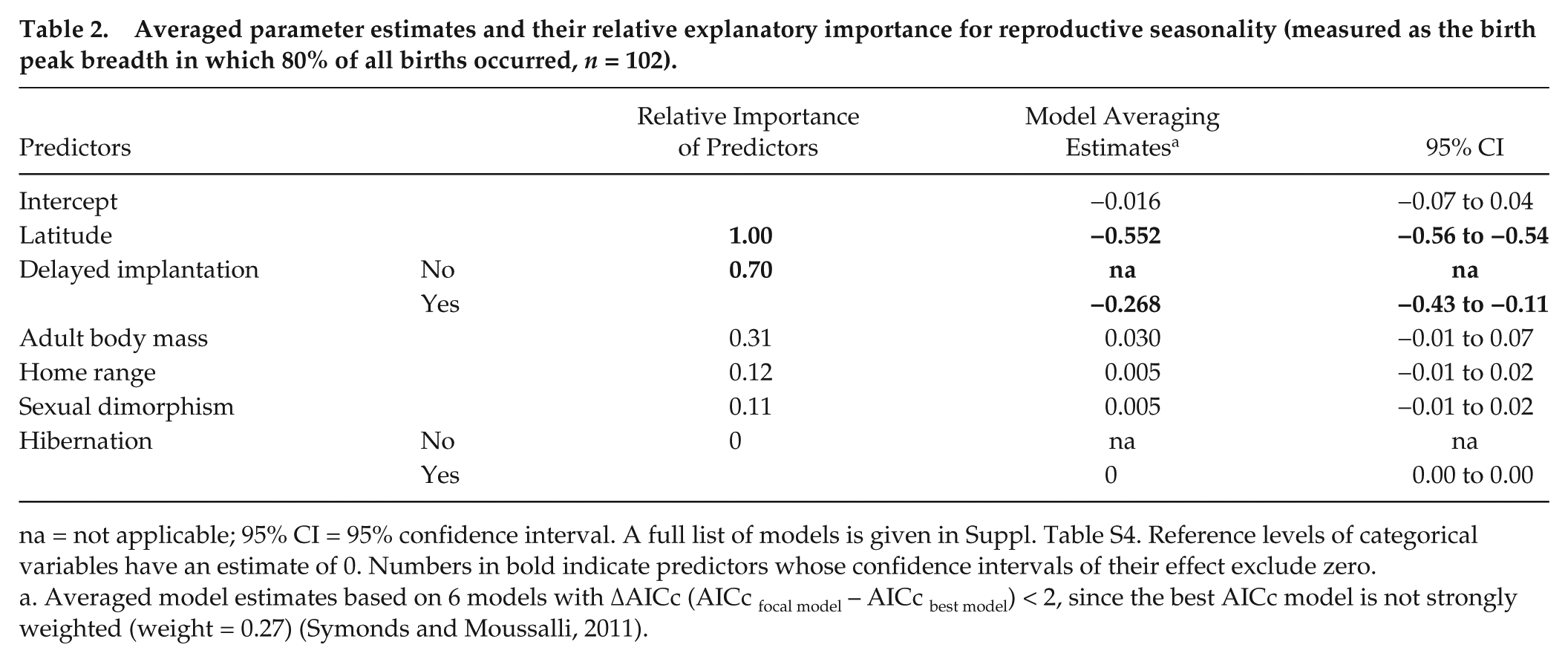
na = not applicable; 95% CI = 95% confidence interval. A full list of models is given in Suppl. Table S4. Reference levels of categorical variables have an estimate of 0. Numbers in bold indicate predictors whose confidence intervals of their effect exclude zero.
Averaged model estimates based on 6 models with ΔAICc (AICc focal model – AICc best model) < 2, since the best AICc model is not strongly weighted (weight = 0.27) (Symonds and Moussalli, 2011).
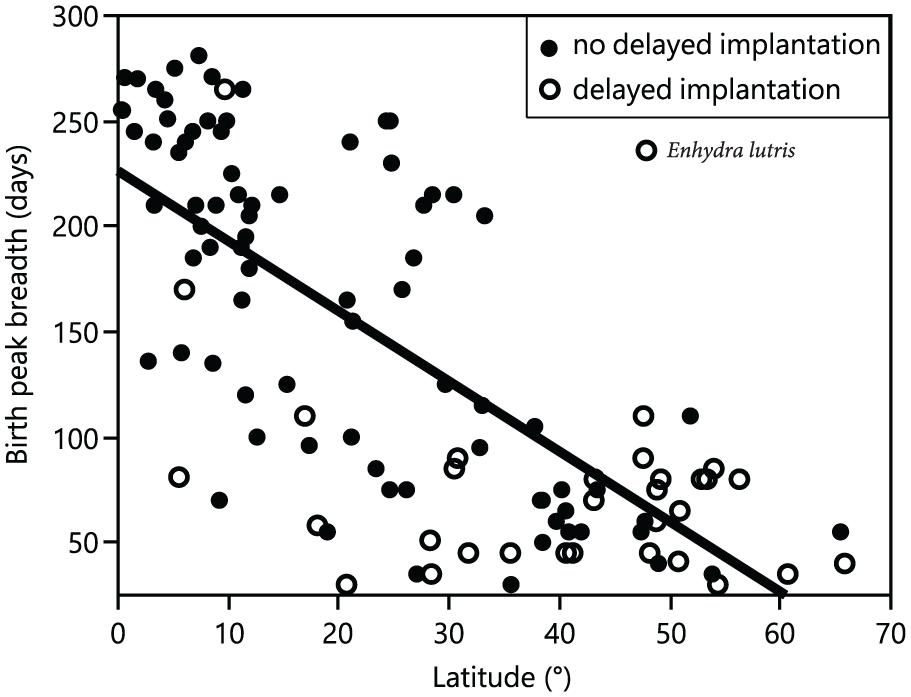
Relationship between birth peak breadth in captivity and the latitude of a species’ origin. The birth peak breadth was defined as the number of days (calculated as 5-day intervals) in which 80% of all births occurred. Species from higher latitudes demonstrate a higher degree of reproductive seasonality when compared with species that originated from lower latitudes. There was one notable outlier, the sea otter (Enhydra lutris), to this pattern. For statistics, see Table 2 and Suppl. Table S4.
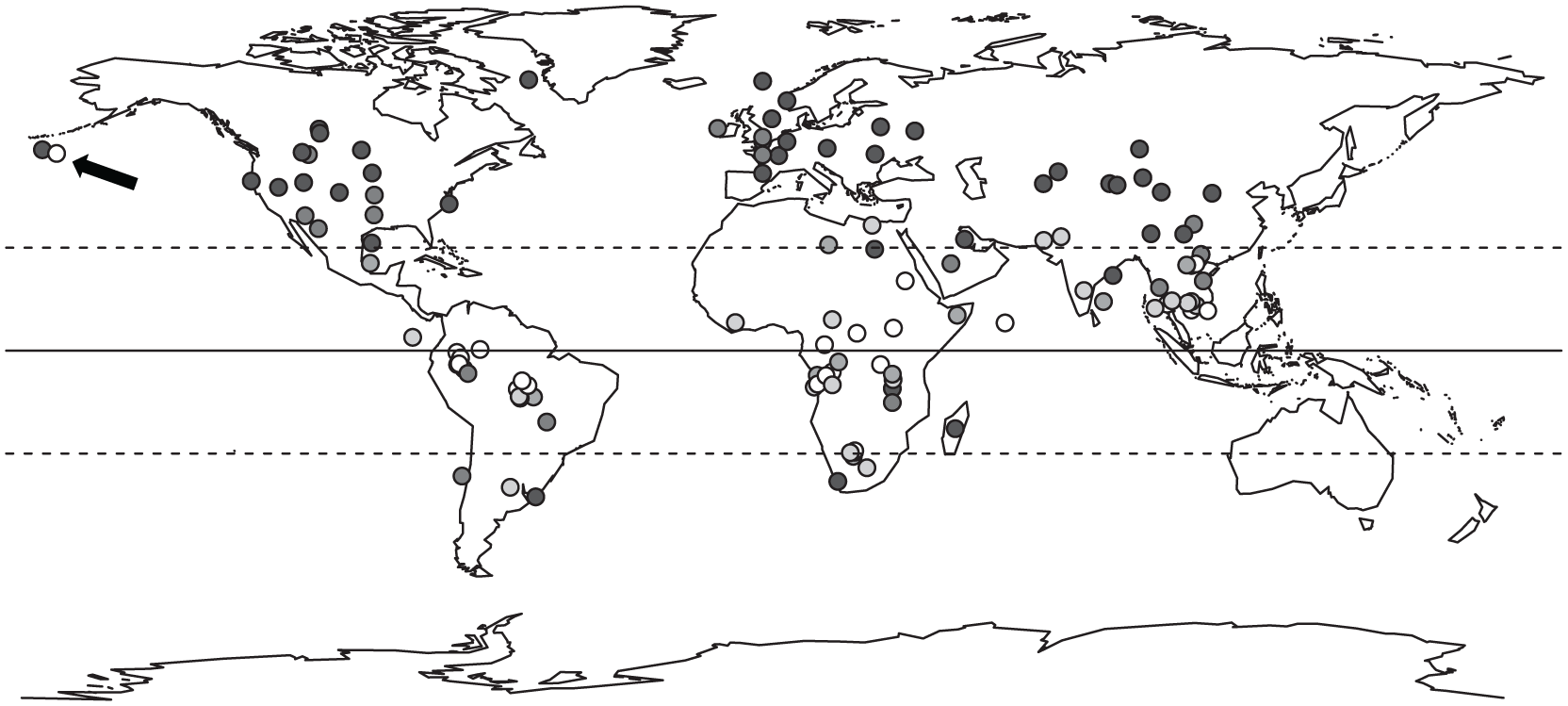
Distribution of Carnivora species across the world. Location of dots indicates the median of the natural origin; gray shading codes categorical seasonality. The degree of gray shading of dots (from white to black) correlates negatively with the length (number of days calculated in 5-day intervals) of the birth peak breadth in which 80% of all births occur. White dots, aseasonal species, birth peak breadth = 231-281 days; light gray dots, birth peak breadth = 181-230 days; gray dots, birth peak breadth = 131-180 days, dark gray dots, birth peak breadth = 81-130 days; black dots, seasonal species, birth peak breadth = 30-80 days. The notable outlier, the sea otter (Enhydra lutris), is indicated with an arrow.
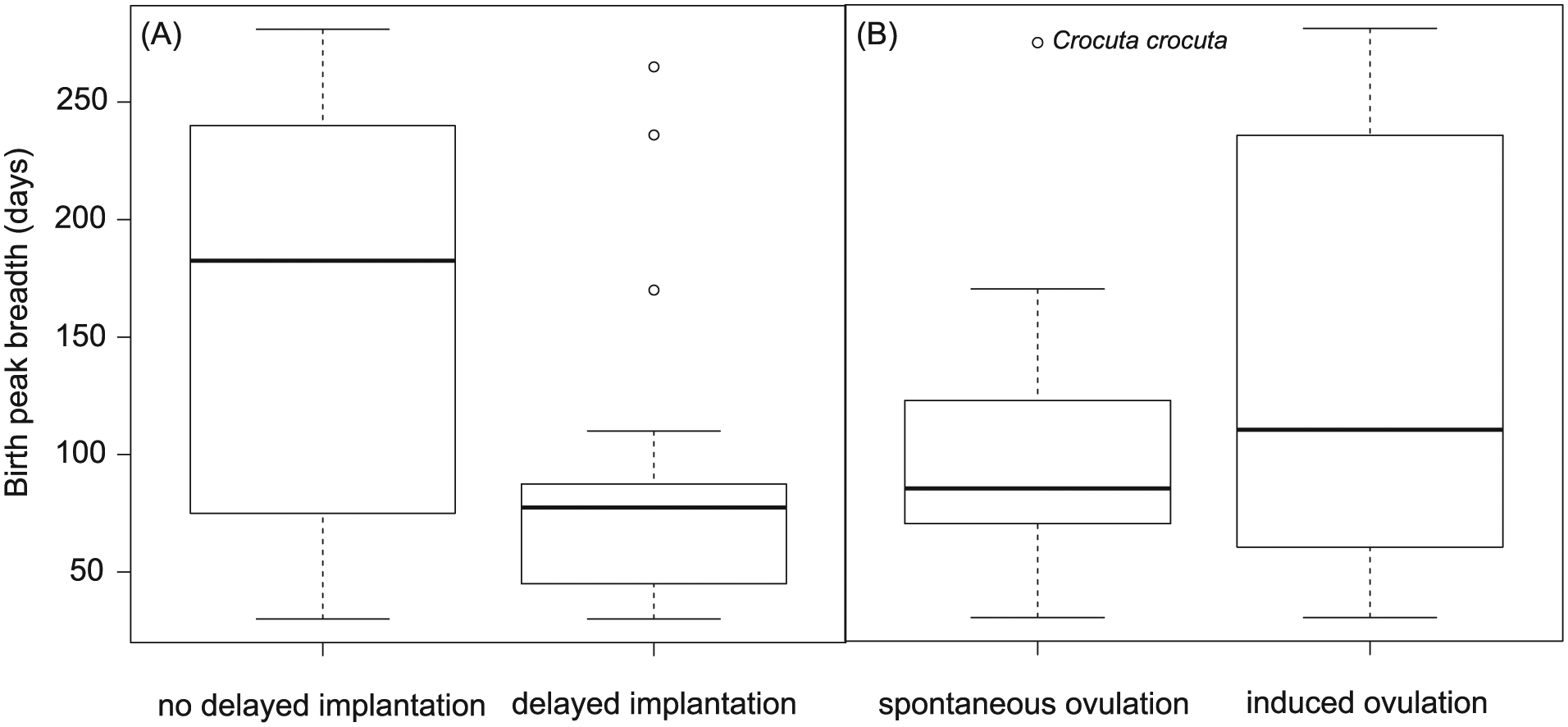
The length of the birth peak breadth in which 80% of all births occur (number of days calculated in 5-day intervals) in Carnivora species sorted according to (A) delayed implantation (without n = 82; with n = 32) and (B) induced ovulation (without, i.e., spontaneous ovulators, n = 52; with n = 23). Depending on availability of information (Suppl. Table S1), the same species may occur in (A) and (B). Note the outlier with a much higher birth peak breadth in the spontaneous ovulators (the spotted hyaena, Crocuta crocuta). For statistics, see Table 2 and Suppl. Table S4.
After removal of one outlier with an absolute Studentized residual larger than 3 (Jones and Purvis, 1997) (the spotted hyaena, Crocuta crocuta) and correction of the analysis for latitude, species with induced ovulation exhibited a more expanded birth peak breadth than species that ovulate spontaneously (relative importance = 0.61; induced ovulation estimate, mean = 0.25, 95% CI = 0.02-0.48) (Suppl. Tables S9 and S10). This finding suggests that species with spontaneous ovulation have a more seasonal birthing pattern than do species with induced ovulation (Fig. 6B).
Gestation Length and Reproductive Seasonality
As expected, body mass was positively correlated with gestation length, except in the dataset comprising only species with delayed implantation (Table 3 and Fig. 7A). The relative gestation period (corrected for body mass and defined as the period between conception and parturition) showed a negative correlation with the birth peak breadth in the overall dataset, indicating that more seasonal species had a longer gestation period if species with delayed implantation were included (Table 3 and Fig. 7B). By contrast, in species without delayed implantation, a positive correlation between the relative gestation period and the birth peak breadth was found (Table 3 and Fig. 7C). Similar results were found when gestation period length was related to latitude (Suppl. Fig. S5, Suppl. Table S11).
Results from phylogenetic generalized least squares regression models testing for the effect of reproductive seasonality on the relationship between log (body mass) and log (gestation length).
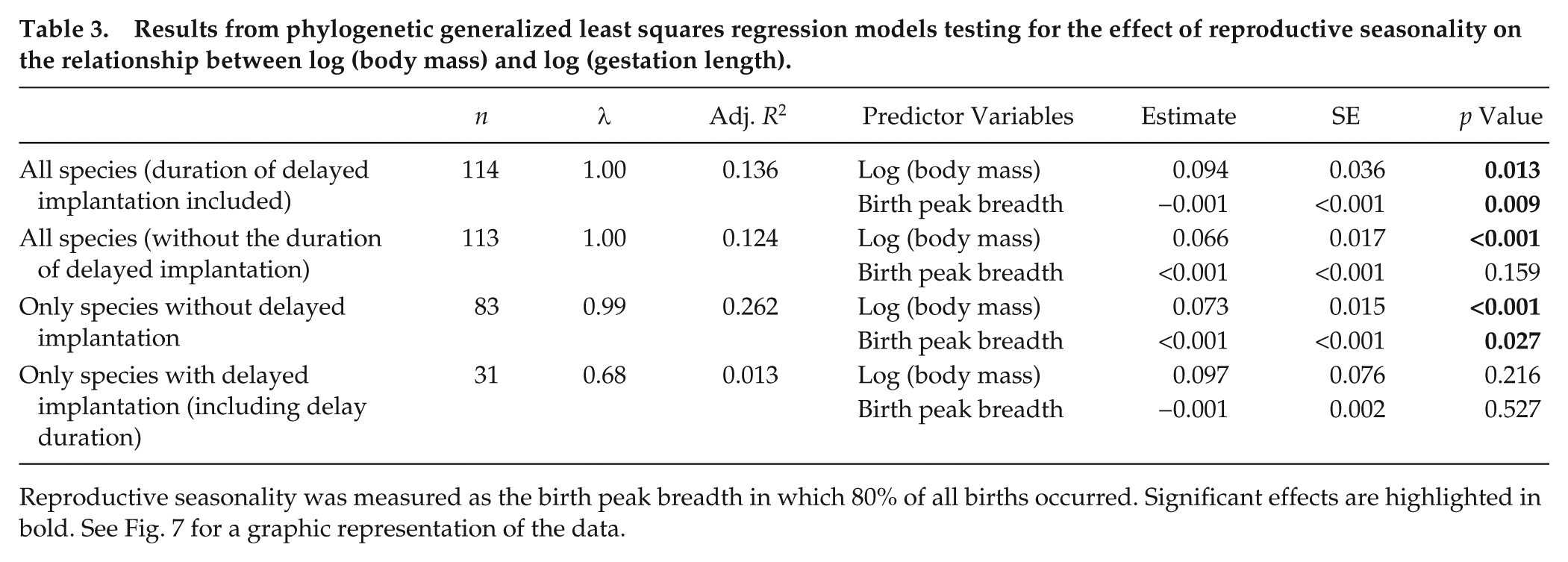
Reproductive seasonality was measured as the birth peak breadth in which 80% of all births occurred. Significant effects are highlighted in bold. See Fig. 7 for a graphic representation of the data.
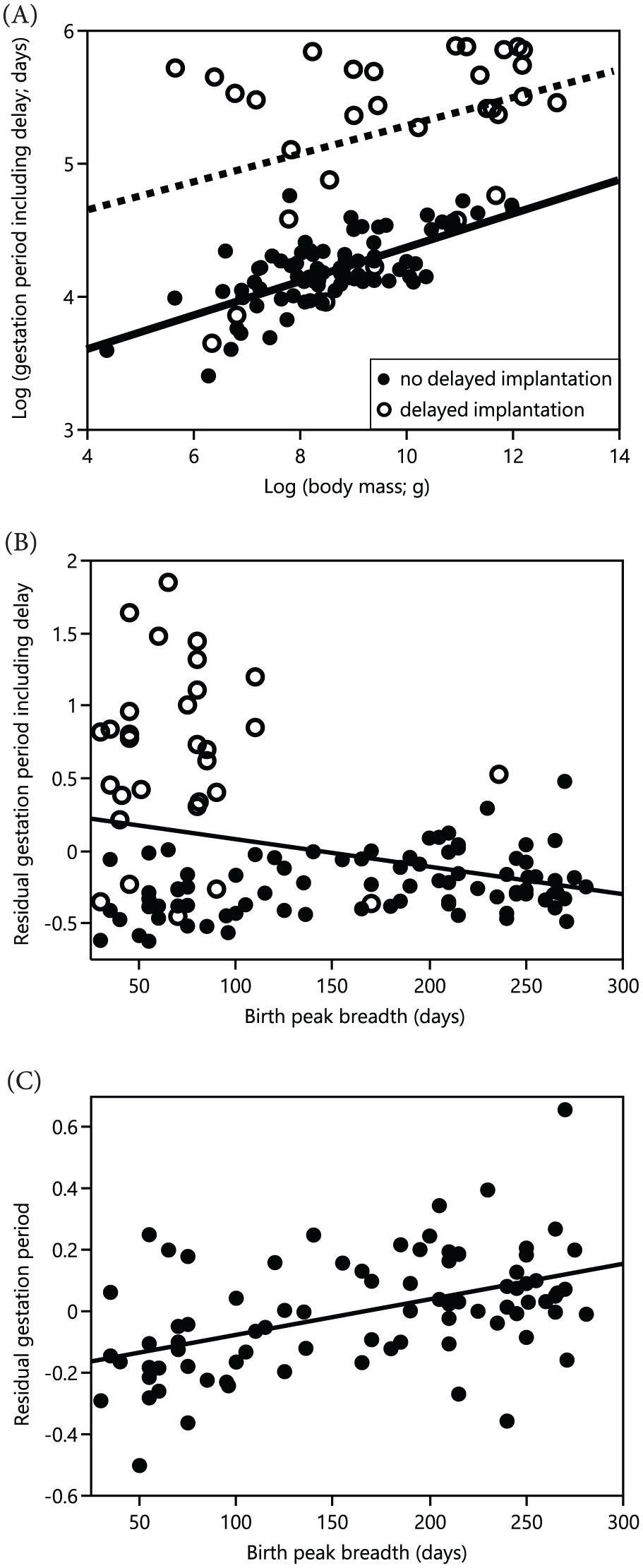
(A) Relationship between the length of gestation period (duration between conception and parturition) and body mass. (B, C) Relationship between relative gestation period including the duration of delayed implantation (corrected for body mass) and the birth peak breadth (B) in the overall dataset and (C) in species without delayed implantation. The birth peak breadth was defined as the number of days (calculated as 5-day intervals) in which 80% of all births occurred. For statistics, see Table 3.
Discussion
We applied both a traditional categorical description (seasonality categories 1-5) and a quantitative measure (the birth peak breadth, i.e., the number of days in which 80% of all births occur) to characterize the degree of reproductive seasonality in the Carnivora. With a sample size of 114 species, we found that the majority of species in the order of Carnivora (86.8% of all examined species) retained the birth seasonality displayed in the wild also in captivity. Our results add to another comparative study on reproductive seasonality in 110 wild ruminant species (Zerbe et al., 2012) demonstrating that for most species, data on birth season length from captive populations can be considered a good metric of birth season length in free-ranging populations. We further show that latitude of geographical origin, delayed implantation, and induced ovulation were the main factors linked to seasonality in reproduction in Carnivora species. Our results support our prediction that life-history adaptations to seasonality in Carnivora comprise either a shortening of the gestation length or a lengthening of the gestation period using delayed implantation; both act as important adaptations for species living in seasonal habitats to ensure birthing and mating at beneficial times of the year. However, in contrast to our expectation and unlike results for ruminants (Zerbe et al., 2012), our result did not provide evidence for a photoperiodic signal consisting of an absolute day length for the timing of the initiation of mating or conception in the Carnivora. For species with a sufficient sample size, additional analyses that account for differences in the actual latitude of the different zoological institutions holding those species would likely reveal a more detailed pattern of photoperiod-driven seasonality.
Comparison of Reproductive Seasonality between the Wild and Captivity
The number of studies systematically investigating reproductive seasonality between different species of Carnivora is limited. This is the case because, first, for many species, detecting seasonal reproductive behavior in the wild is difficult and, second, some authors warn against the use of data on birth seasonality from captive populations because these authors believe that the data may differ from the obtained data of the species’ conspecifics in the wild (e.g., Fairall, 1968). Most previous studies on reproductive seasonality in the Carnivora were therefore intraspecific studies or studies restricted to datasets with less than 35 species: Ursidae (Garshelis, 2004; Spady et al., 2007), Canidae (Valdespino, 2007), Felids (Brown, 2011), Pinnipeds (Atkinson, 1997; Boyd, 1991), Martes (Mead, 1994), and various species (Cociu and Cociu, 1976; Fairall, 1968; Skinner et al., 2002). In the present study, we not only increased the species number substantially to 114, covering 13 different orders of the Carnivora, but also applied a quantitative measure for the degree of reproductive seasonality with a high resolution based on daily observations in captivity. Our continuous parameter, the birth peak breadth, correlates well with more traditional categorical descriptions of birth seasonality (e.g., seasonal vs. nonseasonal) but allows more precise descriptions of birth seasonality. This facilitates testing the influence of biological and environmental factors associated with seasonal reproduction.
Factors Associated with Seasonal Reproduction in Wild Carnivora
Day length
One factor that has been repeatedly suggested to influence reproduction in mammals is absolute day length (Bradshaw and Holzapfel, 2007, for a review). Evidence for an impact of photoperiod on reproductive functions in the Carnivora so far stems from intraspecific comparisons and is somewhat controversial. Some studies found that day length affected the timing of estrus and mating (Ben-David, 1997; Bigg and Fisher, 1975; Holcomb et al., 1962) or the timing of embryo implantation and birth (Boyd, 1991; Enders, 1952; Mead, 1981; Temte, 1985, 1993; Woodroffe, 1995). Other studies, including the present one, were unable to find an influence of the exact day length on reproductive activity even in highly seasonal Carnivora species (Coulson, 1981; Crait et al., 2006; Daniel, 1980; Spotte and Adams, 1981; Zuckerman, 1952).
One reason for these conflicting findings could be that besides photoperiodism, many other factors have been proposed to influence the timing of reproductive events in the Carnivora. For instance, sea surface temperature (Coulson, 1981), local climate (e.g., relative humidity, rainfall, air temperature, and windiness) (Mumby et al., 2013; Trites and Antonelis, 1994), a genetic component (Temte, 1991), or the age of the female (Bridges et al., 2011; McNutt and Silk, 2008; Reiter et al., 1981) have been suggested to contribute to timing reproductive events. Furthermore, there is evidence that in numerous species such as gray seals (Halichoerus grypus) (Boyd, 1984), Antarctic fur seals (Arctocephalus gazella) (Boyd, 1996; Lunn and Boyd, 1993), Southern sea lions (Otaria byronia) (Soto et al., 2004), European badgers (Meles meles) (Ferguson et al., 1996; Woodroffe, 1995), American minks (Neovison vison) (Ben-David, 1997), Eurasian otters (Lutra lutra) (Beja, 1996; Ruiz-Olmo et al., 2002), spotted hyaenas (Crocuta crocuta) (Holekamp et al., 1999), and brown bears (Ursus arctos) (Friebe et al., 2014), females have the ability to adjust the timing of embryo implantation and parturition according to resource availability and the female’s nutritional state (especially a female’s body fat stores). Our own finding that 15 species in our Carnivora dataset switched to a less distinct birth seasonality under constant resource provisioning in captivity also suggests that reproduction can be resource-constrained in some species of Carnivora. However, the number of such species was remarkably low, and none of the species mentioned above in this paragraph changed its seasonality distinctively in captivity compared with the wild in our study. Furthermore, the high relevance of the latitude of origin of a given species on birth seasonality, even in captivity with constant food supply, implies a photoperiodic influence on the timing of reproductive activity at least for some species. This photoperiodic influence may be less distinctively linked to a certain day length, as in ruminants (Zerbe et al., 2012), but more to the directionality of day length changes (short-day and long-day breeders) as has been shown in previous studies for several species of our Carnivora dataset (e.g., Asa and Valdespino, 1998; Bigg and Fisher, 1975; Brown et al., 2002; Murphy and James, 1974).
Latitude
Overall, this study confirms that the most important factor influencing reproductive seasonality is the latitude of the geographical origin of a given species. Similar to findings regarding ruminants (Zerbe et al., 2012), this effect was not strongly affected by captivity, suggesting that reproductive patterns are genetically fixed. Many environmental factors are associated with latitude, including photoperiod, temperature, seasonal changes in precipitation (such as rain or snow), and primary productivity (MacArthur, 1972). All these factors heavily influence the availability of seasonal resources to a given species, thereby influencing its optimal timing of conception and parturition. At higher latitude, the length of the favorable season declines, as spring starts later and winter starts earlier in the year (Sparks and Menzel, 2002). This exerts a strong selective pressure on short birthing periods at high latitudes. As expected, we found that reproductive periods became shorter with increasing latitude of the natural habitat, confirming previous studies in the Carnivora, which were based on fewer numbers of species (Spady et al., 2007; Valdespino, 2007).
Animal species living in the tropics may well experience variation in available resources that is not linked to a photoperiodic cue, such as rainy seasons. If their seasonality is not photoperiod-driven but rather resource-driven, one would expect such species to become less seasonal under typical captive conditions with a constant supply of resources. Corresponding to this concept, the 15 Carnivora species in which a difference in reproductive seasonality between free-ranging and the captive population was observed originated from tropical latitudes, and their seasonality became less distinct in captivity. A similar result was found in a comparison of free-ranging and captive populations of wild ruminant species, where only species originating from lower latitudes showed such a change (Zerbe et al., 2012). The low proportion of species that showed such a change in the present Carnivora and the previous ruminant study (13% and 10%, respectively) emphasizes the predominant role photoperiodic cues play in triggering seasonal reproduction in these 2 mammal groups.
One notable outlier to the pattern of increasing reproductive seasonality with increasing latitude was found, namely the sea otter (Enhydra lutris), which has a very wide birth peak breadth despite its high latitude of geographical origin. In the primate literature, it has been repeatedly reported that more complex food manipulations, such as tool use and extractive foraging (e.g., Heldstab et al., 2016), allow species to find and/or access hidden or protected high-quality food sources (e.g., Melin et al., 2014; Navarrete et al., 2016). As extracted food resources are often available year-round, species that are able to exploit them can keep their energy intake relatively constant throughout the year and, hence, can also buffer lean periods in very seasonal habitats (van Woerden et al., 2014). If this finding in primates also applies to the only tool-using species of Carnivora, namely the sea otters (Shumaker et al., 2011), then the resulting constant food availability might allow them to expand their birth window even in their northern high-latitude environment.
An additional explanation for the wide birth peak breadth in sea otters, despite their high-latitudinal origin, is the special marine environment that this species occupies. Living in the ocean provides more uniform conditions throughout the year compared with life on land, because resource abundance is relatively stable across seasons and marine mammals are less affected by adverse weather conditions compared with land-living species, potentially allowing sea otters to give birth year-round (Monson et al., 2000). In particular, sea otters do not live on fish (which may show seasonally varying presence) but mainly consume sedentary invertebrates, which allows the sea otters to have a mostly uniform diet across seasons (Newsome et al., 2009). Furthermore, sea otters are able to carry out all their reproductive behaviors in water (Estes, 1980). This is in contrast to pinnipeds and polar bears (Ursus maritimus) which are also marine carnivores but require land or sea ice to mate, give birth, and/or nurse their young, making them dependent on availability of sea ice or favorable weather conditions on land (Ferguson et al., 2000; Schulz and Bowen, 2005; Tynan and DeMaster, 1997).
Another factor that is supposed to have similar effects as latitude on reproductive seasonality is the altitude of the habitats from which species originate. Due to a lack of sufficient information, we were not able to test for altitudinal effects on birth seasonality. However, as for increasing latitude, the favorable season becomes also shorter with increasing altitude (Körner, 2007; Mooney and Billings, 1961; Sadleir, 1969). We therefore expect the birth season to become more pronounced for species living in high-altitudinal habitats. An example that demonstrates an effect of altitude of a species’ origin on birth peak breadth in addition to the seasonality due to the latitude of origin is the red panda (Ailurus fulgens). With 80% of all births occurring during only 35 days, the red panda is among the 7 species with the narrowest birth peak breadth in our study despite its low latitude of geographical origin (28.38°N). But red pandas live in a narrow altitude band between about 2300 and 4000 m above sea level confined to the Himalaya and Hengduan mountains (Glatston et al., 2015; Roberts and Gittleman, 1984), supporting the prediction that species living at higher altitudes show a higher degree of birth seasonality.
Delayed implantation
Carnivora are the best studied group for both the absence and presence of delayed implantation and the frequent appearance of these delays; more than half of the mammalian species known to possess delayed implantation are in this order (Orr, 2012; Orr and Zuk, 2014). One hypothesis on how delayed implantation may be advantageous to mammals is that it allows females to schedule mating, birth, and infant dependency according to the availability of adequate resources in seasonal environments by relaxing the otherwise tight correlation between body mass and gestation length (Fries, 1880; Prell, 1930; Sandell, 1990). In fact, several studies found that species at higher latitudes are more likely to have delayed implantation, which supports the hypothesis that these delays allow females to negotiate seasonal environments (Ferguson et al., 2006; Orr, 2012; Thom et al., 2004). Consistent with these findings, we also found that species inhabiting higher latitudes and hence more seasonal climates have a higher occurrence of delayed implantation than do species living at lower latitudes (Suppl. Fig. S2). As an extension of the seasonality hypothesis for delayed implantation (Thom et al., 2004), we additionally found a trend in the relationship between the length of the delay and latitude, wherein a longer delay was found in areas where winters with unfavorable conditions are longer, that is, farther away from the equator (Suppl. Fig. S3). All these previously mentioned comparative studies, which found a relationship between the latitude of geographical origin and delayed implantation, suggested that delayed implantation may have evolved in response to the timing of breeding restrictions imposed by long winters. Consequently, delayed implantation is expected to be linked to a more seasonal birthing pattern, as most females are expected to time parturition to the same beneficial environmental conditions (in spring). We directly tested and confirmed a relationship between seasonal breeding and delayed implantation using a quantitative measure for birth seasonality, and we conclude that seasonality plays a major role in the evolution of delayed implantation for the majority of Carnivora species. Yet, we cannot fully rule out the possibility that for certain mammalian clades (e.g., pinnipeds), delays also evolved due to other reasons, such as enabling the synchronization of seasonal breeding (Bartholomew, 1970). Nevertheless, in our study, pinnipeds fit the expected birth seasonality pattern based on their latitude of geographical origin.
Induced ovulation
In a comparative study comprising 35 North American Carnivora species, Larivière and Ferguson (2003) showed that induced ovulators had a tendency to live in more seasonal environments, with larger home ranges and lower population density, than spontaneous ovulators. In agreement with this study, we also found that induced ovulation is more common in species living at higher latitudes. This finding suggests that induced ovulation in seasonal habitats is advantageous, because mates do not have to meet during the short time window of a fixed ovulation, which is especially important for solitary species with large home ranges where suitable mates are infrequently encountered (Greenwald, 1956; Llewellyn and Enders, 1954). Incorporating this latitudinal effect, we found that species with induced ovulation exhibit a wider birth peak breadth than species that ovulate spontaneously. A possible explanation for this result is that induced ovulation without a fixed short estrus period allows females who lose their first litter to mate and conceive again within a short interval (Sanderson and Nalbandov, 1973), thereby resulting in a more expanded birth season. This shortening of the interbirth interval after a litter loss is also expected to be beneficial for females living in aseasonal habitats. However, our finding that the ovulation type has an influence on reproductive seasonality needs to be regarded with caution, as data on the pattern of ovulation were not available for several Carnivora species (39 species had an unknown ovulation type in our dataset). Future studies are therefore needed to substantiate our result. If our result is representative for the Carnivora, it emphasizes induced ovulation as an alternative evolutionary strategy for high-latitude species.
The only species of the order Carnivora with an exceptionally wide birth peak breadth for a spontaneous ovulator in our dataset is the spotted hyaena (Crocuta crocuta). However, since induced ovulation is still poorly understood across a broad range of species, it remains to be seen whether spotted hyaena are indeed unique in this respect.
Hibernation, mating system/sexual dimorphism, and home range size that had no influence on reproductive seasonality
Hibernation, mating system/sexual dimorphism, and home ranges size showed no relationship with seasonal reproduction in the Carnivora. We predicted that hibernators would have a more pronounced birth season compared with nonhibernating species, because hibernating species of Carnivora have a shorter active season. Although the birth peak breadth was relatively short in hibernators, comprising only 40 to 110 days, hibernation was not significantly associated with seasonal reproduction. However, our result depends on a relatively small sample of only 7 hibernating species and must therefore be regarded with caution. Mating system/sexual dimorphism had no influence on reproductive seasonality in the Carnivora and was also only marginally associated with the birth peak breadth in ruminants in a previous study (Zerbe et al., 2012). Future studies in other mammalian orders are warranted to determine whether our finding in the Carnivora can be extended to mammals in general.
Home range size was relatively unimportant for seasonal reproduction in ursids (Spady et al., 2007) and also showed no correlation with the birth peak breadth in our study. However, home range sizes show large intraspecific variability in species of the order Carnivora (Garshelis, 2004; Lindstedt et al., 1986; Wilson et al., 2017), and accurately estimating home ranges in this order entails several technical and statistical difficulties (e.g., Powell et al., 1997), making interspecific comparisons difficult. Furthermore, almost no data are available for home range sizes in pinnipeds, except for the ringed seal (Pusa hispida) (Oksanen et al., 2015) and Hawaiian monk seal (Neomonachus schauinslandi) (Wilson et al., 2017), making this group unavailable for interspecific home range size comparisons.
Gestation Length and Reproductive Seasonality
It has been suggested in the literature that large-sized and small-sized mammalian species have different adaptations to reproduce in seasonal habitats: Large species living in seasonal habitats are expected to shorten their gestation period so that parturition and subsequent conception can both occur during the short time window of optimal environmental conditions; otherwise, they would lose one seasonal cycle for reproduction (Kiltie, 1988; Zerbe et al., 2012). The corresponding adaptation for smaller animals to reproduce at high latitudes would be to evolve particularly long gestation periods to bridge long winters with unfavorable conditions (Ferguson et al., 2006; Jabbour et al., 1997; Thom et al., 2004). Unlike ruminants, where delayed implantation has so far been confirmed in only one small species, the roe deer (Capreolus capreolus) (Zerbe et al., 2012), the results of our study quantitatively support both concepts, albeit without the assumed clear body-size dichotomy. Within Carnivora, delayed implantation occurs across the entire body size spectrum (Fig. 7A). Many Carnivora show a shortening of the gestation length in species living in seasonal habitats (Fig. 7C, Table 3, Suppl. Fig. S5, and Suppl. Table S11). In contrast, other Carnivora evolved delayed implantation, allowing the gestation length to be longer than expected for a female’s body mass (Fig. 7A and Table 3).
These findings raise interesting questions about the evolutionary options for changes in life history in mammals. Mammalian gestation periods show a large degree of variation, even within precocial or altricial species (Clauss et al., 2014). Similar-sized species of similar ecology can vary enormously in their gestation period, such as between hyraxes (e.g., Procavia capensis, 230 days) (Millar, 1971) and hares (e.g., Lepus europaeus, 42 days) (Roellig et al., 2010) or between cattle (280 days), horses (330 days), dromedary camels (Camelus dromedarius, 390 days), and okapis (Okapia johnstoni, 450 days) (Clauss et al., 2014), or within domestic species among breeds (Heck et al., 2017, 2018). Within a lineage, a reduction in body size typically does not lead to a proportional reduction in gestation length (e.g., Müller et al., 2011), so that particularly long gestation periods for a given body size have been interpreted as indication for secondary dwarfing (Geist, 1966). The impression arises that gestation period is a comparatively conservative characteristic and that a reduction of gestation period length—such as within clades—represents an innovation (Clauss and Rössner, 2014) that facilitates increased reproductive output or adaptation to seasonally fluctuating resource availability. By which physiological means this acceleration of intrauterine development is achieved remains unexplored. By contrast, evidence for a prolongation of gestation periods, by a deceleration of intrauterine growth, is apparently nonexistent. Those species of Carnivora that require a gestation period longer than expected for their body size as an adaptation for bridging long winters in seasonal environments do not achieve this by a deceleration of intrauterine growth but by delaying implantation. This leads us to hypothesize that growth deceleration may be very difficult to achieve in evolutionary terms, a speculation that derives from the hypothesis of evolutionary escalation (Vermeij, 2013). Selective pressure appears to be able to lead to a selection of faster growth or a break before the growth but not to a selection of slower growth in general. Future studies investigating the physiological mechanisms of these adaptations further are needed to test this hypothesis.
Supplementary Material
Supplementary Material, Supplementary_material – Geographical Origin, Delayed Implantation, and Induced Ovulation Explain Reproductive Seasonality in the Carnivora
Supplementary Material, Supplementary_material for Geographical Origin, Delayed Implantation, and Induced Ovulation Explain Reproductive Seasonality in the Carnivora by Sandra A. Heldstab, Dennis W. H. Müller, Sereina M. Graber, Laurie Bingaman Lackey, Eberhard Rensch, Jean-Michel Hatt, Philipp Zerbe and Marcus Clauss in Journal of Biological Rhythms
Footnotes
Acknowledgements
We are grateful to Karin Isler for constructive comments; Gretchen F. Wagner, Fränzi Korner-Nievergelt, and Filipe C. R. da Cunha for statistical advice; and Gabriella M. Fredriksson, Julia N. Beck, Brian L. Cypher, and Thierry Lodé for contributing birth seasonality data. We gratefully acknowledge all Species360 member institutions for their consistent data contribution to ZIMS. We thank the Swiss National Science Foundation (Grant No. 31003A-144210), the A. H. Schultz Foundation, and the University of Zurich for financial support.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
