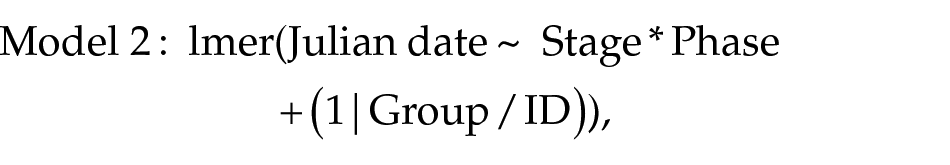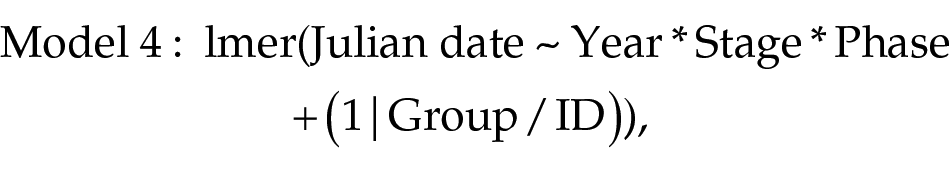Abstract
Because of the complications in achieving the necessary long-term observations and experiments, the nature and adaptive value of seasonal time-keeping mechanisms in long-lived organisms remain understudied. Here we present the results of a 20-year-long study of the repeated seasonal changes in body mass, plumage state, and primary molt of 45 captive red knots Calidris canutus islandica, a High Arctic breeding shorebird that spends the nonbreeding season in temperate coastal areas. Birds kept outdoors and experiencing the natural photoperiod of the nonbreeding area maintained sequences of life-cycle stages, roughly following the timing in nature. For 6 to 8 years, 14 of these birds were exposed to unvarying ambient temperature (12 °C) and photoperiodic conditions (12:12 LD). Under these conditions, for at least 5 years they expressed free-running circannual cycles of body mass, plumage state, and wing molt. The circannual cycles of the free-running traits gradually became longer than 12 months, but at different rates. The prebreeding events (onset and offset of prealternate molt and the onset of spring body mass increase) occurred at the same time of the year as in the wild population for 1 or several cycles. As a result, after 4 years in 12:12 LD, the circannual cycles of prealternate plumage state had drifted less than the cycles of prebasic plumage state and wing molt. As the onset of body mass gain drifted less than the offset, the period of high body mass became longer under unvarying conditions. We see these differences between the prebreeding and postbreeding life-cycle stages as evidence for adaptive seasonal time keeping in red knots: the life-cycle stages linked to the initiation of reproduction rely mostly on endogenous oscillators, whereas the later stages rather respond to environmental conditions. Postbreeding stages are also prone to carryover effects from the earlier stages.
Keywords
On our tilted Earth, animals and plants have to stay in tune with ever-changing environments and, if they live longer than a year, must repeatedly adjust to the seasonal cycles in resources, predators, and parasites, yielding an endless variety of annual schedules (Hut et al., 2013; Wingfield, 2005). Seasonal time keeping is vital. Vertebrates do this by going through repeated sequences of sometimes overlapping life-cycle stages such as breeding, migration, molt, and hibernation (Dawson, 2008; Gwinner, 1986; Wingfield, 2005). In each organism, the scheduling of life-cycle stages should provide a good match with the cyclic environmental conditions encountered by the organism. Some of the stages may require rigid timing, whereas others may best be scheduled conditionally, that is, as a function of previous experiences, the current environmental situation, and individual state (Cornelius et al., 2013).
To stay in tune with environmental cycles, organisms use information from external cues, among which photoperiod is the most reliable and commonly documented environmental zeitgeber (Bradshaw and Holzapfel, 2007; Dawson et al., 2001). However, there is also a strong evidence for endogenous components to the time-keeping systems. When placed in an artificially maintained unchanging photoperiod (most often 12:12 LD), birds of various species continue to express cycles of at least some physiological processes, e.g., development and regression of gonads, molt, premigratory restlessness, and body mass changes related to migration (Gwinner, 1986).
Although the nature of the endogenous oscillators involved remains unknown (Kumar et al., 2010; Wikelski et al., 2008), there are several, not always mutually exclusive, fundamental interpretations of the persisting physiological cycles in an uninformative environment: 1) there is an internal clock(s) similar to the ones of the circadian system, especially evolved to maintain endogenous rhythms with a period close to the calendar year (Gwinner, 1986); 2) animals count days to derive an annual cycle (Farner and Follett, 1979; Gwinner, 1973); and 3) the whole of an animal’s physiology acts as a macro-clock, either based on the subsequent transitions between life-cycle stages (Dawson, 2015; Dawson et al., 2001; Wingfield, 2005) or tracking time passage as a total amount of energy spent (the “energy turnover hypothesis”; Wikelski et al., 2008).
Whatever the actual mechanism of internal time keeping, it is traditionally seen as an integral part of a functional system that adjusts the animal’s annual cycle to its environment (Gwinner, 2003). Endogenous oscillators serve as a basis in determining the timing, and the time course, of seasonal events, ensuring that responses to the external stimuli are adequate and occur at proper times (Chandola-Saklani et al., 2004; Gwinner, 1996; Holberton and Able, 1992; Rani and Kumar, 2013; Wikelski et al., 2008). While there is no doubt that responses to external cues can be adjusted to adapt to changing environments (Hahn and MacDougall-Shackleton, 2008; Wang et al., 2013), no direct evidence for an adaptive response of an endogenous oscillator has been brought forward (Kumar et al., 2010). The latter is probably related to endogenous time keepers generally being understudied for numerous technical reasons (Numata and Helm, 2014; Rani and Kumar, 2013; Wikelski et al., 2008). Nevertheless, there are many examples suggesting that endogenous clocks are subject to natural selection.
First, circannual programs differ among closely related (sub-) species with different migration routes, providing distinct responses to external cues that are appropriate to the particular schedules (Helm, 2006; Helm et al., 2009; Helm and Gwinner, 1999; Maggini and Bairlein, 2010). The endogenous time keepers can also act differently between the sexes when their schedules differ (Budki et al., 2012; Coppack and Pulido, 2009; Maggini and Bairlein, 2010). Second, even within individual animals, phenotype cycles can get out of phase with each other under unvarying environmental conditions (i.e., in the absence of synchronizing information; Cadée et al., 1996; Gwinner, 1986; Helm, 2006; Piersma et al., 2008). This suggests that requirements for temporal precision may vary between different physiological processes. This variation is probably achieved by the differentiation of circannual oscillators within an organism (Pengelley, 1968). From an ecological perspective, such dissociation of time keepers should enhance the degree of flexibility of annual routines (Helm, 2006; Piersma et al., 2008; Wang et al., 2013). Such flexibility may be particularly important in the orchestration of the complex schedules of long-distance migrating birds, in which selection pressure on timing is expected to vary across an annual cycle (Conklin et al., 2013; Cornelius et al., 2013; Winkler et al., 2014).
In this study, we explore the possible role of endogenous rhythms in the ecological adaptations of a long-distance migrant, the red knot Calidris canutus islandica. This is a shorebird that migrates across 5000 km between breeding areas in the High Arctic of Greenland and Canada and coastal wintering grounds in temperate northwest Europe (Buehler and Piersma, 2008; Davidson and Piersma, 1992; Davidson and Wilson, 1992). We documented the repeated seasonal changes in body mass, plumage state, and primary molt in 45 captive red knots maintained in captivity for up to 20 years. Most of the time, the birds were exposed to the natural photoperiod of the Dutch Wadden Sea, where they were captured during the nonbreeding phase. In addition, to study the organization of the phenotype cycles, we excluded informative environmental cues by bringing 14 birds under unvarying environmental condition (with respect to daily LD cycles and temperature) for 8 years. We compared rigidity in the timing of onsets and offsets of the 4 life-cycle stages in the absence of external zeitgebers, predicting that phases of life-cycle stages for which precise timing is essential would drift less under unvarying photoperiodic conditions (i.e., indicating a smaller driving role of environmental cues) than life-cycle stages requiring less stringent timing.
Materials and Methods
Ethics Statement
All experiments were performed in accordance with national animal welfare laws and were approved by the Animal Experimentation Ethics Committee of the Royal Netherlands Academy of Arts and Sciences, with coaching from Nanneke van der Wal and Chris Pool.
Study Species
Red knots are quite easy to keep in captivity as long as they are maintained in social groups with access to sediments to probe (i.e., the opportunity to exercise their sensitive bill tip organ; Piersma et al., 1998). Under these conditions, they show strong and persistent seasonal cycles in body mass, plumage, and flight feathers (Cadée et al., 1996), preen wax and other aspects of body composition (Reneerkens et al., 2007), metabolism (Helm et al., 2012; Jenni-Eiermann et al., 2002; Piersma, 2002; Vézina et al., 2011), and aspects of immunocompetence and disease resistance (Buehler et al., 2008; Milot et al., 2014). This species is monomorphic, and working on it in captivity, we consistently failed to find sex-specific differences (e.g., Reneerkens et al., 2007).
The red knots studied here were of the islandica subspecies, the 45 individuals captured with mist nets in November 1994 (groups 1, 2, and 3) and in October 1995 (group 4) at Wieringen in the western Dutch Wadden Sea (52°9′N, 4°97′E) and brought to the captivity nearby NIOZ Royal Netherlands Institute for Sea Research on the island of Texel immediately after the catch. The birds were housed in 8 outdoor aviaries (4.5 by 1.5 m, with a height of 2.5 m) in groups of on average 7 birds. The aviaries had floors covered with Sika epoxy material and wire-mesh walls. The latter ensured that the red knots had access to outside light and experienced ambient temperatures, although the temperature was somewhat buffered in the sheltered cages. We provided the birds with food (trout-food pellets) and fresh water ad libitum.
Experimental Setup
On 26 November 1996, the birds of groups 1 and 2 (7 and 7 individuals) were moved into indoor avia-ries with the same dimensions, with walls made of Trespa laminate panels. Here the birds were kept under unvarying photoperiodic (1000 lux white light) and temperature (12 °C) conditions until 28 December 2004 (Fig. 1). One of the groups (group 1) experienced 12:12 LD, with light on from 7:00 till 19:00 h during the whole period indoors. A second group (group 2), in addition to being exposed to 12:12 LD, due to an instrumental failure, received constant 24-h light between 25 November 2003 and 28 December 2004. Except for the 1 bird that died in 2004, on 1 January 2005 we returned both groups to outdoor aviaries, and the red knots remained there until June 2015, when they were released back into the wild. Thirty-one birds from the control groups 3 and 4 were kept only in outdoor aviaries until release in 3 July 2001 (10 individuals died earlier).
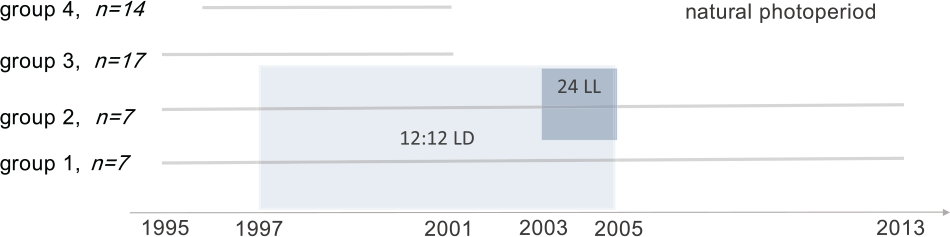
Scheme of the experimental setup. Observations on red knots from groups 1 to 3 since 1995 and from group 4 since 1996 are used in the analysis. Groups 1 and 2 were placed in unvarying photoperiodic and temperature conditions during 1997 to 2004 inclusively. Group 1 experienced 12:12 LD (light-gray shade) for the whole period of the treatment with unvarying photoperiod, while group 2 experienced 24-h light in 2003 to 2004 (dark-gray shade). Red knots from groups 3 and 4 had been kept in natural photoperiod during their stay in captivity until they were released in 2001. Initial sample sizes are given in italics next to the group number.
Groups were not necessarily kept constant in terms of number and composition. In fact, most individuals shared cages with a variety of mates, usually of the same subspecies, but sometimes with individuals belonging to the canutus and rufa subspecies or with great knot Calidris tenuirostris.
Annual Cycle of islandica Red Knots
Of the 6 subspecies of red knot (Buehler and Piersma, 2008), the islandica subspecies migrates the shortest distances, although still covering almost 5000 km one way between breeding areas in northern Greenland and on the Queen Elisabeth Islands and wintering grounds in northwestern Europe. As summarized in Figure 2, in late March they molt from their basic nonbreeding into an alternate plumage and begin to fuel up for the subsequent migration flights. From the Wadden Sea they fly north, make stopovers in either Iceland or northern Norway (Davidson et al., 1986; Wilson and Strann, 2005), and arrive to the breeding grounds in the last days of May and the first of June (Morrison et al., 2005). Adults leave the breeding grounds between mid-July and early August and fly nonstop to staging grounds in western Iceland, where they refuel from late July to early August (Davidson and Wilson, 1992); some may skip Iceland and fly directly back to the Wadden Sea (Dietz et al., 2010). They molt back into the basic plumage in August to September. Wing molt extends from August to mid-October for breeding adults, but it can start earlier and last longer in juveniles and adults that skip migration and spend the summer in the Wadden Sea (Dietz et al., 2013). From October to December, many red knots move westward to Britain and then northwestward within Britain (Davidson, 2002), until their return to the Wadden Sea in March.
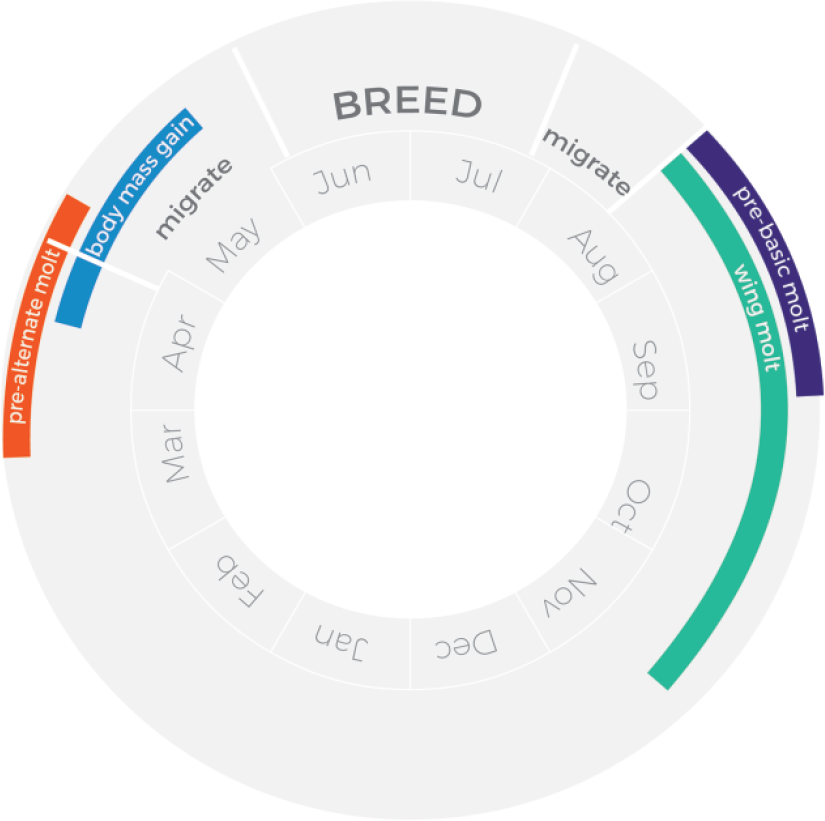
Annual cycle of islandica red knots. See text for interpretation and references.
Life-Cycle Stages in Captivity
Although in the outdoor aviaries at NIOZ the birds experienced the same changes in temperature and photoperiod as faced by their wild conspecifics residing in the Wadden Sea, by necessity they have to skip breeding and thus resemble (the mostly young) birds oversummering in the Wadden Sea (Dietz et al., 2013). Nevertheless, captive red knots continued to show the cyclic expression of body mass, plumage state, and wing molt, presumably facilitated by the ongoing cycles in gonadal maturation and regression (Dawson, 2015). Because of the thick layer of subcutaneous fat that is accumulated in spring exactly when gonadal maturations also take place, the latter are impossible to observe in red knots. Thus, in this study, a high spring body mass gain indicates a preparedness to migrate. Molt of body feathers from the gray basic plumage of the nonbreeding season into the rusty-red contour feather of the alternate plumage accompanies the mass gain in free-living red knots and indicates a preparedness to breed (Vézina et al., 2012). The molt into the basic plumage signals the termination of a breeding condition. The molt of flight feathers occurs on the wintering grounds together with the molt into the basic plumage.
Once a week, usually on Tuesdays, the birds were taken from their aviaries, weighed on an electronic balance (mass to the nearest gram), examined for the state of their plumage and primary-feather molt, and checked for any physical abnormalities (see Milot et al., 2014). The plumage was scored on a 7-point scale, with 1 for the basic and 7 for the full alternate (breeding) plumage. The extent of primary-feather molt (hereafter “wing molt”) was determined with the traditional Ashmole system (Ginn and Melville, 2007): each of 10 primaries (denoted P1 to P10, from innermost to outermost) was assigned a score from 0 (unmolted, old feather) to 5 (fully grown new feather). The total molt score was the sum of the scores for individual primaries and therefore varied from 0 (no growing primaries) to 50 (all 10 primaries had reached a score of 5). Because the final length of the last (P10) relative to the prelast (P9) primary typically varies among individuals (Conklin and Battley, 2012), we considered molt completed at a score of 49. Once birds had reached this point, their molt score was reset to 0 (all primaries old).
Finding Characteristic Switch Points with Continuous Wavelet Transformation
The weekly measurements of body mass and the scores on the progression of wing molt and plumage state comprised individual time series varying in length between 2 and 18 years (Fig. 3). Although there was usually 1 wave or peak per year, rhythmicity sometimes changed, especially in birds kept under unvarying conditions. We were primarily interested in the timing of switches between life-cycle stages and we tried to objectively identify the onsets and offsets of body mass gains, plumage cycles, and wing molts using wavelet transformation (Fig. 3). Wavelet transformation is a widely used technique to analyze periodic time series and is especially recommended for those data in which cycle lengths (frequency) vary over time (Torrence and Compo, 1998). The approach has previously been successfully used to measure the phase and period of behavioral rhythms (Leise et al., 2013).
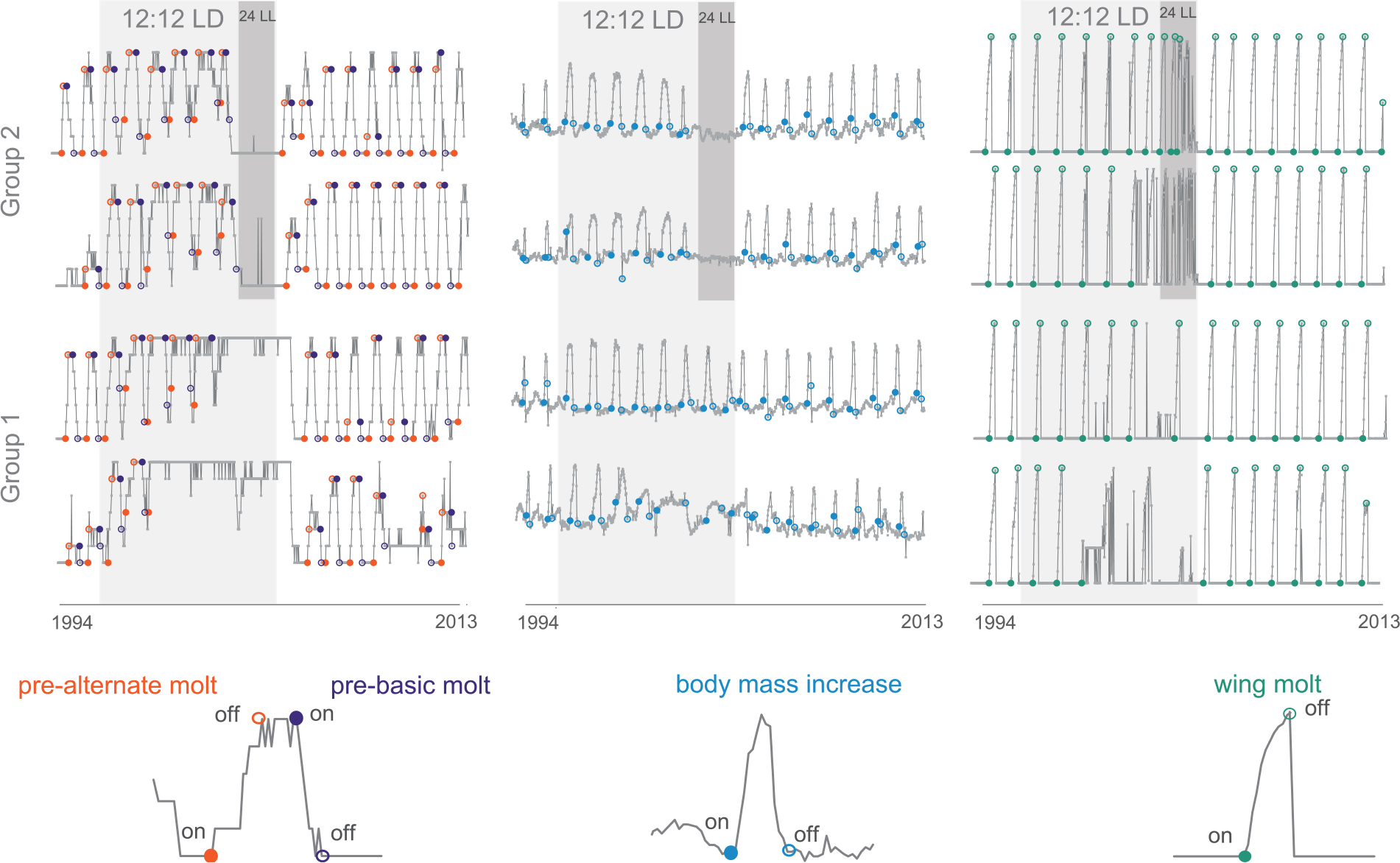
Annual cycles of body mass and molts in captive red knots. An example of time series of plumage state (left), body mass (middle), and wing-molt scores (right) in 4 captive red knots from groups 1 and 2. The measurements were taken with intervals of 7 days (gray dots). Onsets (closed circles) and offsets (open circles) of prealternate (coral-red) and prebasic (plum) plumage molt, spring body mass increase (blue), and wing molt (green) are presented on both the time series and the 1-year fragments from them (below). Periods of treatment with unvarying photoperiod are shown as shaded rectangles (light gray for 12:12 LD and dark gray for 24 LL).
Continuous wavelet transformation (CWT) is obtained by moving a continuously scalable window that contains an oscillatory function (“mother” wavelet) along a time axis, multiplying it by the signal, and integrating the product of the multiplication over all times. The choice for a particular mother wavelet depends on the purpose of the analysis. The scale parameter, in the definition of wavelet transformation, is the inverse of frequency (the number of occurrences of a repeating event per unit time). Therefore, wavelet function at scales larger than unity represents compressed versions of the initial function (oscillating at a higher frequency), while at smaller than unity scales, it provides dilated versions of it (oscillating at a lower frequency). The product of CWT at particular scales captures oscillations in the raw data of corresponding frequencies and can be used to characterize their phase and period (see Polikar, 1999, 1995, for details).
All analyses were performed in R (R Development Core Team, 2015). We used R packages biwavelet (Gouhier, 2013) and pracma (Borchers, 2015) for wavelet transformation and exported the MATLAB (2010) code from Leise et al. (2013) into R (Supplementary Online Material 1, with reproducible code available at https://github.com/eldarrak/red_knot_annual_cycles/blob/master/Supplementary1.rmd). We transformed the time series of each life-cycle stage for each individual red knot using the complex-valued Paul continuous wavelet function as a mother wavelet. We chose Paul, as it provides better time resolution than the more commonly used Morlet and DOG functions (De Moortel et al., 2004).
For the wavelet-transformed time series of each individual red knot and each life-cycle stage, we found the points of sign reversal and local maximum of the amplitude by using the method, earlier applied by Leise et al. (2013). We plotted the found characteristic points over nontransformed time series and visually assessed them for relevance. We removed the few uninformative points produced by noise in the data. Under unvarying photoperiodic conditions, after several years the phenotype cycles often became undefinable. In the analyses, we therefore used only onsets and offsets of cycles that were visually distinct.
Onsets of the body mass increase were defined as dates at which the amplitude reversed sign from negative to positive. Correspondingly, dates of change from positive to negative were assigned offsets of body mass peak. The completion of wing molt we defined as the date at which the amplitude of the wavelet-transformed time series reached a local maximum.
Unlike cycles of body mass, the cycles of plumage state included extended periods during which plumage state was stable: full alternate plumage and full basic plumage (Fig. 3). As a result, points of intersection of wavelet-transformed time series of plumage scores with the zero line did not coincide with the actual onsets and offsets of molt. To define true onsets and offsets of prealternate and prebasic plumage molt, we looked for the points of start and end of rapid plumage state change. Scoring plumage state is accurate only to within the nearest point on the ordinal scale, and this caused noise in the data. In addition, some individuals did not always reach minimal or maximal plumage scores, which complicated the definition of the onsets and offsets of the prealternate and prebasic molt. To cope with these problems, for each cycle of each individual, we searched for the most encountered score between onset of prealternate molt and offset of prebasic molt (intersection with zero line) defined with CWT. This score and higher was used to indicate a full alternate plumage.
Similarly, the most encountered score between the CWT-defined offset of prebasic molt and onset of prealternate molt the next year, and lower scores, were considered as full basic plumage. We took the last date of full basic plumage for the real onset of prealternate molt and the first date of full alternate plumage for its real offset. Accordingly, the last date of full alternate plumage was taken as the real onset of the prebasic molt and the first day of full basic plumage as its real completion. The time series of body mass, plumage state, and wing molt, with defined onsets and offsets, for each individual are presented in Supplementary Online Material 2 (https://github.com/eldarrak/red_knot_annual_cycles/blob/master/Supplementary2.pdf). For further analysis, we presented the obtained onsets and offsets of the life-cycle stages as the number of days since January 1 of the year when they took place (Julian date). Because onsets and offsets of single life-cycle stage can behave differently under manipulated conditions (Gwinner, 1986), we considered them separately in the analysis.
Statistical Analysis
Phenotype cycles under natural photoperiods
First, we tested for nonlinearity in timing of all the life-cycle stages across years in the control groups 3 and 4. To detect possible thresholds in the time series, we applied segmented regression (Muggeo, 2008, 2003) separately for each phase (onset and offset) of each life-cycle stage.
where Yearcap stands for year since capture and Julian date for the day since 1 January when the onset or offset occurred.
To test for possible long-term linear trends in the timing of the onsets and offsets of life-cycle stages, we used observations on red knots from groups 1 and 2 from the period 2008 to 2013, when the birds apparently stabilized after the end of treatment with unvarying photoperiod. We applied linear mixed-effects regression (lmer, from package “lme4”; Bates et al., 2015), with Julian date as the response variable and year (continuous), life-cycle stage (body mass peak, prebreeding and prebasic plumage molts and wing molt), its phase (onset and offset), and interactions between the parameters as explanatory variables. Individuals’ ID nested in a group ID were included as random intercepts. We simplified the model that included all the explanatory variables and their interactions by removal of nonsignificant terms (P > 0.05), following a stepwise backward procedure (Zuur et al., 2007). In this study, the same procedure was used for all model simplifications. The variable Year was eliminated from the model during the selection procedure, and the final model was
where Julian date is the day since 1 January, when a phase of a life-cycle stage occurred; Stage refers to a life-cycle stage (body mass peak, prealternate, prebasic, or wing molt) and Phase to onset versus offset of the stages, Group stands for the group a bird belonged to, and ID for a bird’s ID.
The start of linear phase drift in unvarying photoperiods
In 12:12 LD, onsets and offsets of all the observed life-cycle stages drifted linearly toward later dates. Some of them became visibly delayed immediately after transition to an uninformative photoperiod, and others sustained at natural timing for several years. To determine the year when phases started to drift, we used generalized additive mixed-effects regression (gamm, package “mgcv”; Wood, 2011), with date as the response variable, spline of year as a fixed explanatory variable, and individual nested in groups as random intercepts.
We applied this model to onsets and offsets of each event, separately (Fig. 4A), and determined the beginning of the drift as the year when the rate of change (first-order derivative) of date switched from negative to positive. If no switch was detected, we assumed that the drift started in the year when the 12:12 LD experiment started.
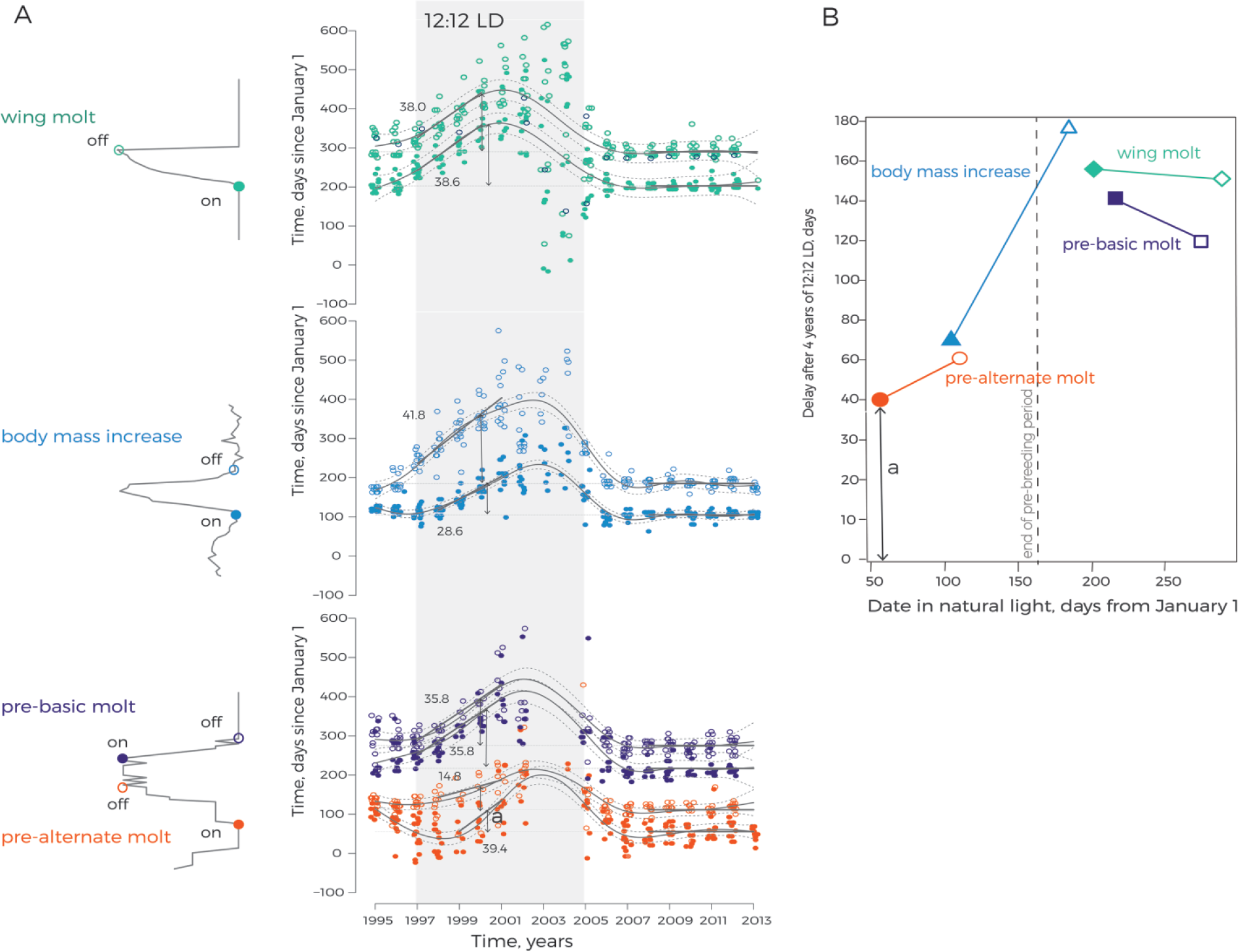
Phase drift of life-cycle stages in captive red knots under unvarying photoperiodic conditions. (A) Timing of spring body mass increase, prealternate and prebasic plumage state, and wing molt in red knots from groups 1 and 2 across their stay in captivity. Onsets (closed dots) and offsets (open dots) of each stage for each individual red knot are plotted together with predicted mean values (curved solid gray lines) and 95% confidence intervals (curved dotted lines, gamm in model 3), predicted mean values (straight solid gray lines) of onsets and offsets in natural photoperiod during 2008 to 2013 (lmer in model 2; the dark lines for this period are continued with thinner lines for earlier years), and linear drift in 12:12 LD during 1997 to 2001 (to 2000 for wing molt, model 4). The numbers next to the lines represent predicted mean values of the rate of linear drift of corresponding phases of life-cycle stages. Two-sided arrows represent cumulative delay after 4 years in 12:12 LD, calculated as the difference between predicted values of onsets and offsets in 2000 under natural and unvarying photoperiods (model 2 and model 4, respectively). One of the arrows (marked with the letter “a”) is projected on panel B. The period of treatment with 12:12 LD is shown in gray shade. (B) Seasonal trend in the persistence of the free-running physiological cycles in captive red knots maintained under unvarying 12:12 LD conditions. Accumulated during 4 years under unvarying conditions delays in onsets (closed symbols) and offsets (open symbols) of spring body mass increase (triangles), prealternate (circles), prebasic (squares), and wing molt (diamonds) are plotted as median dates (in days since January 1; derived from model 2). Delays in the phases within particular life-cycle stages are connected with lines.
Variation of rate of linear phase drift in unvarying photoperiods
With this test, we assessed variation in the rate of linear drift among phenotype cycles and their phases. As revealed in the previous step (model 3), phases of the phenotype cycles started to noticeably drift after a different number of cycles in unvarying conditions. Meanwhile, after 5 years of free run, in 2002, phenotype cycles in many individuals became disrupted, with undistinguishable onsets and offsets. Therefore, for each phase of each life-cycle stage, we used observations between the first year of drift and 2001, inclusively (2000 for the onset and offset of wing molt). We applied a linear mixed effects regression, with the same initial set of variables, as in model 2. The full model was also the most parsimonious, as revealed by backward selection:
where Julian date is the day since 1 January, when the onset or offset of a life-cycle stage occurred; Stage refers to a life-cycle stage; Phase refers to onset versus offset of the stages; Group stands for the group a bird belonged to; and ID represents for a bird’s ID.
Variation of cumulative phase delay in unvarying photoperiods
Assuming that variation in slopes and time lag before start of drift should result in a distinct amount of delay among onsets and offsets of the life-cycle stages, we compared the mean Julian dates of onsets and offsets of each life-cycle stage in 2000 predicted in model 4 with the dates of corresponding events during 2008 to 2013 (when the cycles stabilized after transition from unvarying to ambient photoperiods) predicted in model 2. To look for a seasonal pattern in the obtained difference values, we plotted each of them on the mean date of corresponding phase of a life-cycle stage in ambient light in captivity (for the period 2008 to 2013, derived from model 2, Fig. 4B).
Results
The captive red knots consistently expressed cycles of body mass, plumage state, and wing molt for up to 19 years (Fig. 3). Under the natural light conditions of the Wadden Sea, the timing of life-cycle stages roughly followed their timing in nature. However, as indicated by the breakpoints, detected with segmented regression (model 1), the onset and offset of prealternate molt were delayed during the period between capture and transition to 12:12 LD. After return from the unvarying conditions, the timing stabilized within 3 years, and from 2008 to 2013, there were no trends for either onsets or offsets in groups 1 and 2 (P > 0.05, model 2, Fig. 4A).
In the unvarying 12:12 LD photoperiodic regime, the cycles of body mass, plumage state, and wing molt continued but with a progressing delay (Fig. 4). From the sixth year (2002) onward, the patterns of change in body mass and wing molt started to be chaotic in most of the birds. Prebasic molt became gradually less complete in the course of 12:12 LD. As a result, birds retained the full or nearly full alternate plumage for most of the time from 2001 until the end of the treatment under unvarying conditions (Fig. 3). Interestingly, during their exposure to continuous light, all 7 birds of group 2 stopped expressing body mass cycles. The cycles of molt into breeding plumage and wing molt also became disrupted and incomplete. Upon return to natural photoperiodic conditions, the birds from both experimental groups resynchronized cycles of all the 4 traits within 3 years.
The number of cycles during which timing of life-cycle stages in red knots under unvarying conditions followed the timing maintained under natural cyclic photoperiods varied between the life-cycle stages and their phases (Fig. 4A; model 3). The onset of prealternate plumage molt kept the starting values for 2 years and became delayed in the third year after transition to 12:12 LD. The offset of prealternate molt and the onset of the body mass increase started to drift in the second year, whereas delays in the offset of body mass increase and the on- and offsets of prebasic plumage and wing molts were noticeable already in the first year. Even accounting for the time lag before start of drift, the rate of the drift in 12:12 LD varied among life-cycle stages and their phases (P > 0.05, model 4, Fig. 4A, Table 1). The rate of delay was 39, 15, and 29 days/year for the onset of prealternate molt, offset of prealternate molt, and onset of body mass peak, respectively. In contrast, the offset of spring body mass peak and the on- and offsets of postbreeding life-cycle stages (i.e., prebasic and wing molt) became delayed in the first year after transition to 12:12 LD. The rate of delay was 36 days/year for the onset and offset of prebasic molt, 42 days/year for the offset of body mass peak, and 39 and 38 days/year for the onset and offset of wing molt, correspondingly.
Rates of Drift (Days/Year, Predicted Population Mean, and SE) toward Later Dates of Onsets and Offsets of the Life-Cycle Stages in the Experimental Red Knots under Unvarying Photoperiod a
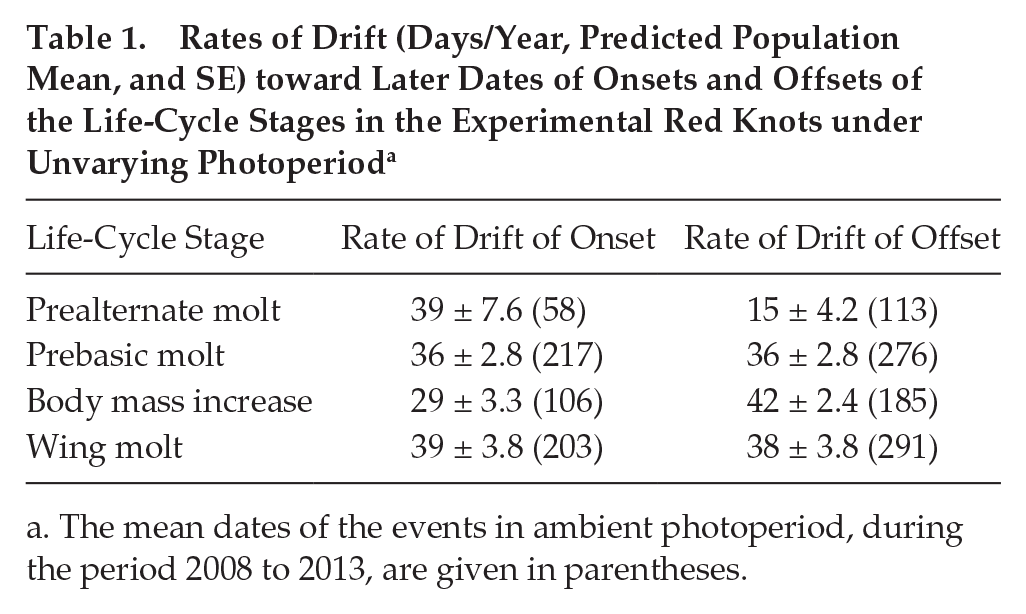
The mean dates of the events in ambient photoperiod, during the period 2008 to 2013, are given in parentheses.
As a result of variation in both the time before the beginning of drift and the rate of drift, the cumulative shift in timing estimated on the fourth year of 12:12 LD differed between phenotype cycles and between their onsets and offsets. This cumulative shift was smaller in phases of prebreeding life-cycle stages compared with those occurring later in the season (Table 1; Fig. 4B).
Discussion
The varying predictability of environmental features and the vagaries of individual circumstances force animals to balance the scheduling of life-cycle stages between extreme rigidity and conditional flexibility. Based on the observed long-term phenotype cycles of individual red knots kept either under natural or unvarying photoperiodic conditions, corroborated by our knowledge of their natural history, we suggest that the endogenous time keepers play an important role in maintaining this balance.
Red Knots Use Photoperiod for Time Keeping
Just as in the much more thoroughly studied passerine birds (Gwinner, 1986), the few papers that have addressed time keeping in shorebirds (Cadée et al., 1996; Piersma, 2002; Piersma et al., 2008) all provide evidence for the use of a combination of endogenous clocks and external cues. Also in the present study, red knots apparently used photoperiod to maintain cycles of body mass and molt equivalent to those in the wild. This is because transferal to unvarying conditions brought about shifts in the phenotypic cycle phase, whereas under natural photoperiods, the timing of the cycles roughly comprised the timing in the wild.
However, the timing of some life-cycle stages was altered in the absence of temporal cues. This may have been due to the elimination in captivity of several important stages from the annual cycle (i.e., northward and southward migration and breeding) and therefore a relaxation of the temporal constraints on their time schedule (Dietz et al., 2013). Of course, it is also possible that the lack of a migration to the High Arctic, where the birds would have experienced a rapid increase in day length up to 24:0 LD followed by its rapid decrease, may have influenced the expression of any endogenous cycles. The up to three years long delays in the onset of prealternate plumage molt and body mass gain after capture, and after return from unvarying to ambient photoperiods, may indicate the adjustment to novel conditions (see data on the long-term decline of corticosterone in captivity in Piersma and Ramenofsky, 1998).
Red Knots Use an Endogenous Time Keeper
Under unvarying photoperiods, red knots maintained cycles of annual processes for at least 4 years, apparently based on endogenous oscillators. Moreover, some of these cycles remained entrained (with a phase length of 12 months and timing of the events very close to the timing under natural photoperiods) for 1 or several rounds after transition to unvarying conditions. Previous experiments revealed that, contrary to most of other bird species, the free-running cycles in Calidris species are longer than 365 days (Cadée et al., 1996; Piersma, 2002; Piersma et al., 2008). Also in the present study, red knots molted and deposited body stores with a delay that accumulated in the course of their stay in unvarying conditions. Duration of body mass peak, prealternate molt, and stay in breeding plumage gradually increased in 12:12 LD, suggesting a facilitating effect of photoperiod on these life-cycle stages. The duration of wing molt remained constant, but its onset and offset drifted toward later dates. Under constant 24-h light, all 4 traits became disrupted. Upon return to the outdoor aviaries, the red knots fully resynchronized timing of all the life-cycle stages within 3 years (Fig. 4A), which, again, suggests an important role of changing day lengths in the adjustment of schedules to the annual environmental cycle.
Adaptive Variation in Reliance on Endogenous Rhythm
As also indicated by other studies, the persistence of physiological cycles under uniform conditions varies greatly among taxa, among individuals of the same species, and even among life-cycle stages within a single individual (Gwinner, 1986; Helm et al., 2013; MacDougall-Shackleton et al., 2015; Piersma et al., 2008). We will not be the first to suggest that this variation is most plausibly explained by distinct selection pressures on the timing of different life-cycle stages, depending on the particulars of specific life histories. Free-running cycles are especially persistent in bird species which lack exposure to clear photoperiodic cycles (Gwinner, 1986; Helm, 2006). For example, nonmigratory tropical residents that in nature experience only subtle changes in day length, even though some of them appear to be sensitive to the slightest modulation in photoperiod (Goymann et al., 2012; Hau et al., 1998; Numata and Helm, 2014), under unvarying conditions continue expressing physiological cycles for many years (Gwinner and Dittami, 1990). In long-distance migrants that experience varying seasonal photoperiodic regimes, endogenous cycles last longer under unvarying conditions than in closely related species migrating less far (Gwinner, 1986; Helm, 2006; Helm et al., 2013).
Within individuals, in the absence of external zeitgebers, cycles of phenotypic traits that are directly correlated with fitness were more strongly endogenously programmed (Gwinner, 1986). For example, molt cycles are often more persistent under unvarying conditions than other functions (Berthold, 1974; Helm, 2006). The number of free-running cycles has always been used as a measure of their rigidity. Nevertheless, under unvarying photoperiodic conditions, even the most long-lasting phenotype cycles will start to deviate from 365 days (Gwinner and Dittami, 1990). Since patterns of the drift have received little attention, the information on how long different species can maintain natural timing of different life-cycle stages in the absence of external zeitgebers is scarce. In the red knots in our experiment, the onset of prealternate molt, the earliest event of the red knot annual cycle, remained entrained (i.e., kept the starting values) for several cycles, until it came into conflict with the increasingly delayed completion of wing molt. Completion of prealternate molt, as well as onset of spring body mass peak, started drifting on the second year under 12:12 LD. In the only study we know that describes the pattern of drift in 12:12 LD (Gwinner and Dittami, 1990), the free-running gonadal and molt cycles of African stonechats also deviated from natural timing only in the second year of an unvarying photoperiod. It seems, however, that in most other birds that have been placed in unvarying photoperiodic conditions, except for great knots (Piersma et al., 2008), physiological cycles started to drift immediately with the loss of external cues. It is interesting to us that both the African stonechats (an equatorial species) and the red knots (long-distance migrants) have good ecological reasons to have precise internal time keepers.
Nevertheless, timing of postreproductive free-running cycles in the red knots deviated from the timing under natural conditions already in the first year. This can be the consequence of the lack of a synchronizing effect of photoperiod per se and the constraining effect of the extended under unvarying conditions life-cycle stages occurring earlier in the season. After 4 years under unvarying photoperiodic conditions, the onsets and offsets of spring life-cycle stages (i.e., those related to reproduction) were only half as delayed as those of later in the year (Fig. 4B). We interpret this observation as follows: prebreeding life-cycle stages rely greatly on the endogenous oscillators, whereas the later stages rather respond to environmental conditions. This concurs with the idea that in birds, prebreeding phases may be more time selected and therefore more synchronized than those happening after breeding (Conklin et al., 2013; Cornelius et al., 2013). Postbreeding life-cycle stages are also prone to carryover effects from earlier stages, which mirrors the pattern observed in nature: that carryover effects accumulate in the course of the season but can be compensated at the end of an annual cycle (Conklin and Battley, 2012).
Dolnik (1975), Jenni and Schaub (2003), and Bauer et al. (2008) earlier suggested that photoperiod, the most reliable external cue, facilitates the development of spring life-cycle stages, with nonphotic environmental cues taking over in the course of the season and allowing more flexible decisions. On the basis of the present findings, we suggest that the time-keeping system of long-distance migrants acts as a hierarchy of endogenous rhythm, photoperiod-response systems, and systems that perceive and process a variety of nonphotic information, in which each of the levels gets priority according to the required (evolutionarily informed) balance between plasticity and rigidity in timing (Fig. 5). The most precise timers, endogenous clocks, should be especially important when high precision is beneficial. External, and especially nonphotic, factors will be prioritized at stages when plasticity is needed.
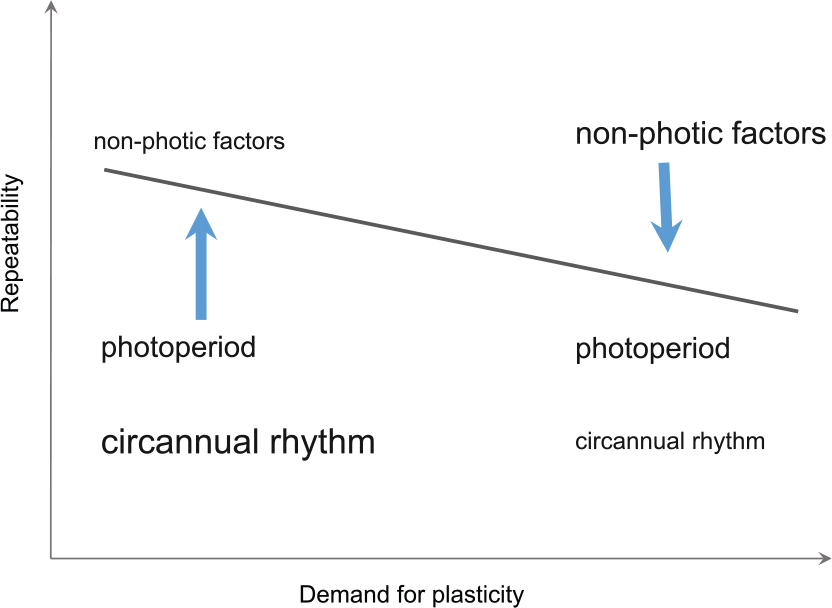
A hypothetical representation of the repeatability of timing of life-cycle stages (gray line) as a function of the demands for plasticity, imposed by environment (i.e., reflecting degree to which environmental conditions are predictable). Arrows indicate the direction of action of prevalent elements of such an adaptive time-keeping system.
Footnotes
Acknowledgements
The birds studied over a period of 20 years in captivity were captured on Wieringen, using the welcome home of Theo Mulder as our base. We thank the late Piet Duiven for organizing these catches and Jan van Gils, Petra de Goeij, and Wim Tijsen for their field help during a memorable evening. We acknowledge the long-term commitment and far-sightedness of NIOZ management that enabled us to carry through this long-term study. The building of the aviaries was facilitated through grants from the Netherlands Organization of Scientific Research (NWO) and internal funding by NIOZ. We owe the indoor facilities to the expertise and dedication of NIOZ engineer Fen Schilling. We are grateful to the late Ebo Gwinner and Barbara Helm, Menno Gerkema, and William Schwartz for guidance and encouragement early and later on. We thank Indic Premananda and Andrea Cimatoribus for excellent advice on use of wavelet transformation and Alistair Dawson and an anonymous reviewer for helpful comments. The analyses and write up were made possible by the Spinoza Premium 2014 awarded by NWO to T.P. E.R. and T.P. were additionally supported by a grant to T.P. from Waddenfonds (“Metawad,” WF209925).