Abstract
The hypothalamic suprachiasmatic nucleus (SCN), a circadian pacemaker, is present in all mammalian brains. It has a complex organization of peptide-containing neurons that is similar among species, but calcium-binding proteins are expressed variably. Neurons containing calretinin have been described in the SCN in a number of species but not with association to circadian function. The objective of the present study is to characterize a calretinin neuron (CAR) group in the rat anterior hypothalamus anatomically and functionally with a detailed description of its location and a quantitative analysis of neuronal calretinin immunoreactivity at 3 times of day, 0600, 1400, and 1900 h, from animals in either light-dark or constant dark conditions. CAR neurons occupy a region in the dorsal and lateral SCN with a circadian rhythm in CAR immunoreactivity with a peak at 0600 h and a rhythm in cytoplasmic CAR distribution with a peak at 1400 h. CAR neurons should be viewed as an anatomical and functional component of the rat SCN that expands the definition from observations with cell stains. CAR neurons are likely to modulate temporal regulation of calcium in synaptic transmission.
Keywords
The suprachiasmatic nucleus (SCN) circadian pacemaker is recognized to have subdivisions that reflect neuronal cytoarchitecture (van den Pol, 1980), peptide phenotype (van den Pol and Tsujimoto, 1985; Moore et al., 2002; Morin, 2013), patterns of afferent innervation, and distribution of efferent projections (Leak and Moore, 2001; Canteras et al., 2011). The SCN region receiving visual afferents, and containing neurons that produce vasoactive intestinal polypeptide (VIP) or gastrin-releasing peptide (GRP), is designated “core,” whereas the region innervated by nonvisual afferents and containing neurons that produce arginine vasopressin (AVP) is designated “shell” (Moore et al., 2002; Canteras et al., 2011).
Visual afferents also innervate an area dorsal to the SCN, the subparaventricular zone (SPVZ), and one lateral to the SCN, the perisuprachiasmatic area (PSCN; Canteras et al., 2011). The distinction between core and shell is most extensively documented in the rat (Moore et al., 2002; Canteras et al., 2011). There is also an area in the rat SCN lateral and dorsal to core and shell in which neurons do not contain AVP, GRP, or VIP but do contain a calcium-binding protein, calretinin (CAR; Jacobowitz and Winsky, 1991). CAR neurons in the rat SCN were noted briefly in a descriptive study comparing CAR immunoreactivity in the SCN of several rodent species (Marshall et al., 2000). In another study, CAR neuron number and diameter in the rat SCN were compared with the peptide-containing groups (Moore et al., 2002). Neither study was directed to a precise description of the CAR group or its participation in circadian function. In this study, we test the hypothesis that CAR neurons are a distinct functional component of the SCN in the rat by providing a detailed analysis of the location of the CAR cell group and CAR expression at 3 time points over a 24-h period with and without a light-dark cycle.
Car Neuron Localization
SCN borders are typically defined by cell-stained material, and this is shown by the dashed lines in Figure 1. CAR immunoreactive neurons are present throughout the rostrocaudal extent of the SCN. In the rostral SCN, they are predominantly located in the dorsal shell (Fig. 1A, B). At mid-SCN levels, the CAR population extends further into the dorsal shell, with a part of it extending into the PSCN from the dorsal and lateral borders (Fig. 1C, D; Fig. 2). There is a small group of cells extending along the border between shell and core and a few extending into the core. Caudally, CAR cells lie in the dorsal and medial shell and the core. They are also found at the shell-core border extending dorsally and laterally into the SPVZ and PSCN. At the most caudal levels, there are cells in separate populations dorsally and ventrally (Fig. 1F). Thus, in the immunostained material, CAR neurons are present both in and outside the borders of the SCN, as that has been defined previously in cell stains.
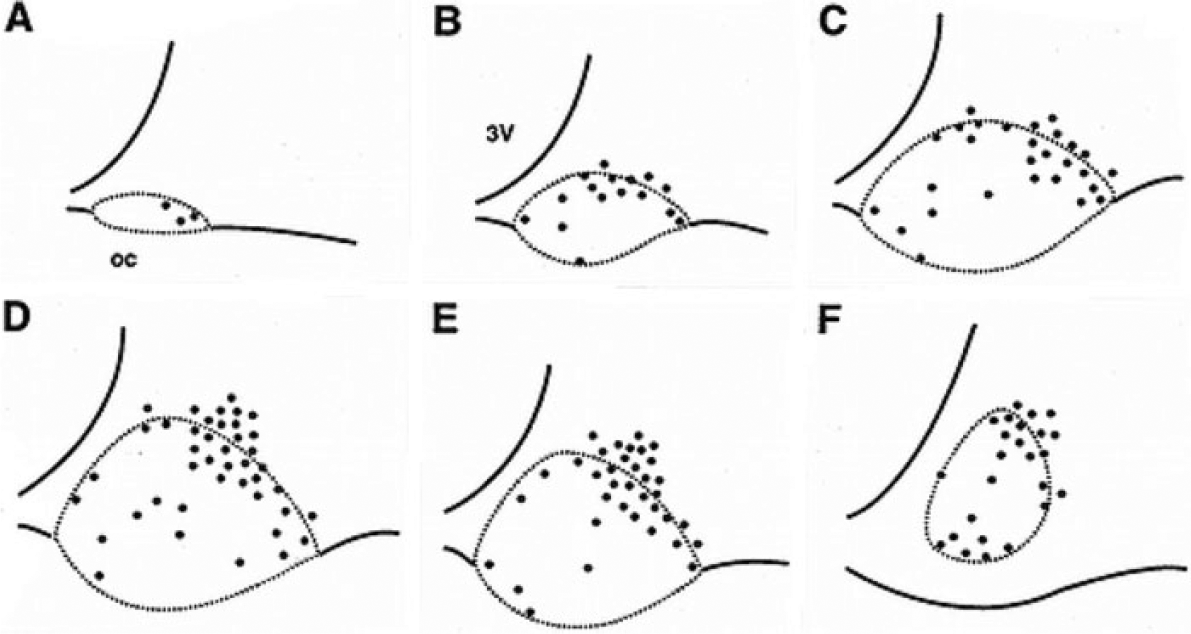
Diagrams of the distribution of calretinin neurons (dots) in 6 coronal levels through the suprachiasmatic nucleus and adjacent perisuprachiasmatic area.
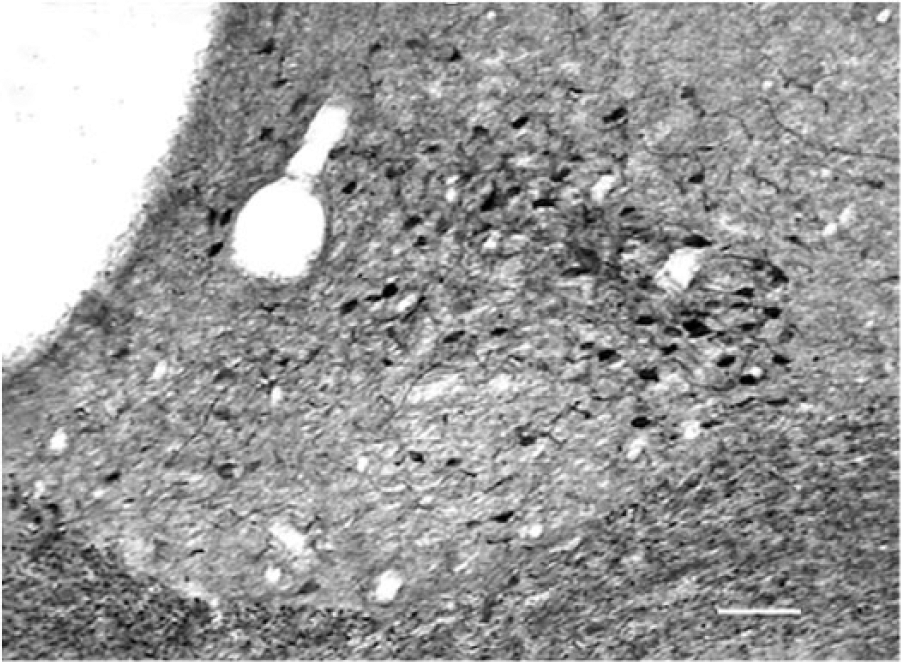
Coronal section at level equivalent Figure 1D through the periventricular chiasmal hypothalamus showing the pattern of calretinin immunoreactivity in the dorsolateral suprachiasmatic nucleus extending into the perisuprachiasmatic area and surrounding the suprachiasmatic nucleus dorsally and laterally. Scale bar = 75 µm.
Circadian Rhythms in Car Immunoreactivity
The location and number of immunoreactive neurons from LD and DD are nearly identical. The number of cell bodies is greater at ZT-CT 6 than at ZT-CT 14 or ZT-CT 19 (Figs. 3 and 4). Nuclei are apparent in a few cells (Fig. 3, arrows). At ZT-CT 14, there are numerous stained processes with a large diameter diminishing gradually as the distance from the cell bodies increases. These processes have varicosities and appear to be dendrites (Fig. 3B). A quantitative analysis of CAR neurons at ZT-CT 6, ZT-CT 14, and ZT-CT 19 is shown in Figure 4. Comparison of the number of CAR neurons at each of the time points sampled is statistically significant for the LD group (df = 2, F = 87, P < 0.001) and the DD group (df = 2, F = 83, P < 0.001). The differences between the LD and DD groups do not differ significantly at any time point. In summary, CAR neurons are found predominantly in the dorsolateral SCN extending into the PSCN. The number of CAR neurons varies with time of day, peaking at ZT-CT 6, and a cytoplasmic distribution of CAR peaks at about ZT-CT 14.
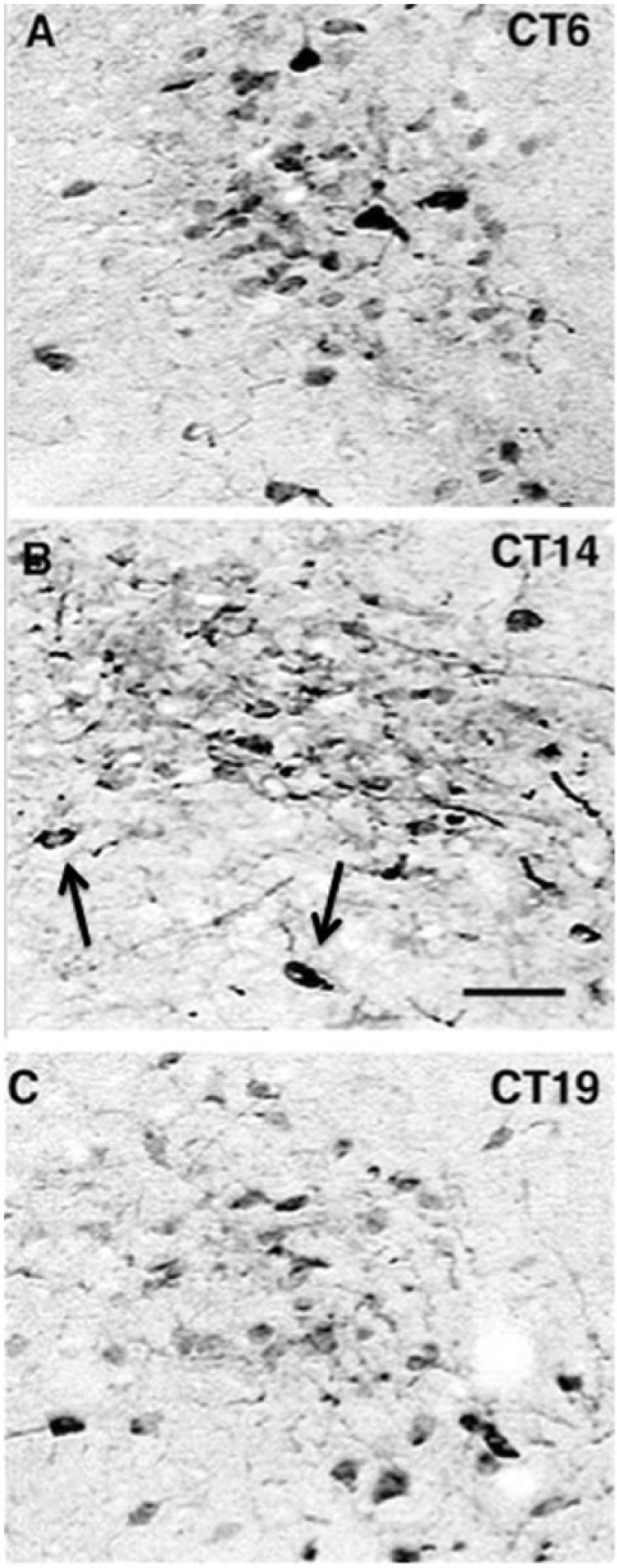
Images showing calretinin (CAR) immunocytochemistry at 3 circadian times (photographs taken from brains of animals in constant dark). The images are from a suprachiasmatic nucleus level equivalent to that shown in Figure 1D in the area of largest concentration of CAR neurons. At CT 6, a large number of perikarya are CAR immunoreactive. At CT 14, the number of immunoreactive cells is reduced but more processes, predominately dendrites, are evident. Arrows designate 2 immunoreactive neurons with nuclei. At CT 19, the number of immunoreactive cells is less than at CT6 with less dense staining. Scale bar = 30 µm for all panels.
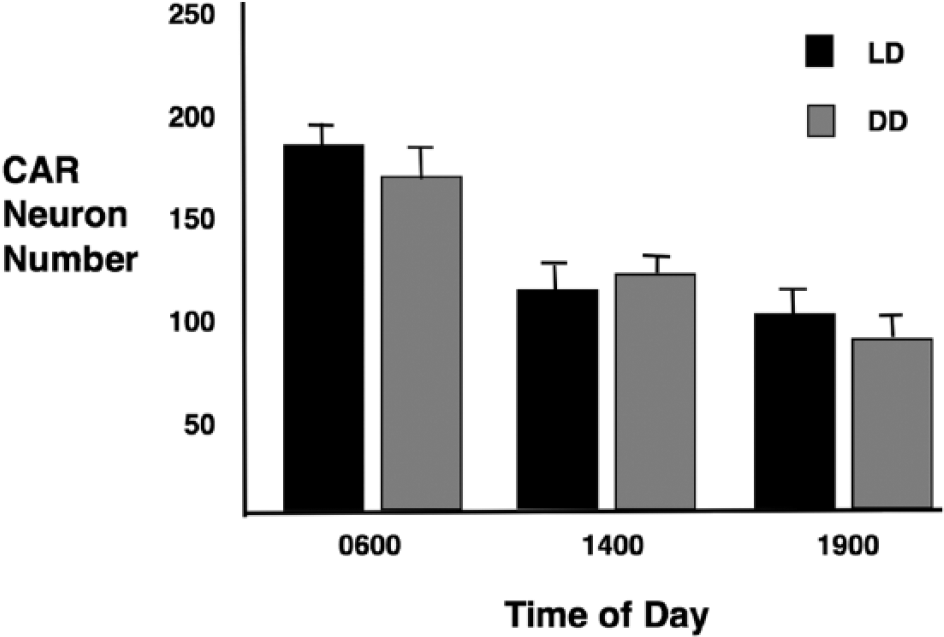
Numbers of calretinin immunoreactive neurons in the suprachiasmatic nucleus as a function of time of day; 0600, 1400, and 1900 h, shown as mean neuron number ± SD for LD and DD conditions.
In this study, we describe a subset of neurons located in the anterior hypothalamus of the rat that contain the calcium-binding protein, CAR, and express 2 circadian rhythms in CAR immunoreactivity. CAR neurons are located predominantly in the dorsal and lateral region of the SCN shell, but some extend medially in the SCN and others laterally into the PSCN. Because all CAR neurons behave functionally like peptide-containing neurons in the SCN (Welsh et al., 2010; Mohawk et al., 2012), we consider them to be part of the SCN shell. This extends the definition of the SCN in the rat by incorporating an area previously considered to be PSCN. Most definitions of the SCN have been based on cell-stained material with relatively thick sections. In these, the dorsal and lateral borders appear distinct, but with thinner sections, the borders become less clear and the lateral SCN merges gradually into the PSCN and SPVZ, making the definition of the SCN more difficult. This is accentuated in immunostained material.
CAR neurons in the SCN were first noted by Jacobowitz and Winsky (1991) in a broad description of forebrain CAR neuron distribution. A subsequent study of CAR neurons in the SCN of the rat, mouse, hamster, and Nile grass rat found marked differences in the location of CAR neurons in the SCN among these rodent species and concluded that CAR neurons are not part of a distinct SCN subdivision in contrast to peptide-containing neurons (Marshall et al., 2000). That article, however, does not contain a full analysis of CAR neuron location in any of the species. The general location of CAR neurons in the SCN has been reported in a number of species in lesser to greater detail: 1) in the ventral SCN in the mouse (Silver et al., 1999; Abrahamson and Moore, 2001). 2) throughout the rock cavy SCN (Cavalcante et al., 2008), 3) predominantly ventrolateral and in adjacent PSCN in the hamster (Marshall et al., 2000), 4) ventral in the marmoset (Cavalcante et al., 2008), and 5) dorsal and lateral in the capuchin monkey (Rocha et al., 2014). Each of these differs from the rat, but the presence of CAR in all species is an indication that it plays a significant role in SCN function in many species. One articl3e on the mouse reports a possible rhythm in CAR content with 2 time points in an LD environment (Silver et al., 1999).
In this study, we observed 2 CAR rhythms that are maintained in LD and DD. The first is a rhythm in the number of immunoreactive CAR neurons with a peak at 0600 h. A rhythm in CAR conformation that alters affinity for the antiserum is reported in a study employing the same antiserum. In this study CAR recognition by the antiserum was dependent on the conformation of CAR determined by cytoplasmic calcium (Winsky and Kuźnicki, 1996). The rhythm in CAR immunoreactivity correlates well with other intrinsic rhythms in the SCN, including the rhythms in neuronal firing rate and glucose utilization, all with broad peaks in subjective day (cf. Welsh et al., 2010; Mohawk et al., 2012, for reviews). All 3 rhythms should be viewed as examples of cellular functions controlled by output of the molecular clock.
The second rhythm is in the cytoplasmic location of CAR with prominent labeling of dendrites at 1400 h. It seems likely that this reflects CAR translocation from cell body to dendrites, but dendritic synthesis is a possibility. The alteration of cytoplasmic CAR location implies a mechanism for modulating the timing of local cellular mechanisms of calcium function in synaptic transmission. A circadian rhythm in the neuronal location of another calcium-binding protein, calbindin, was reported with a peak at CT 14 in the hamster (LeSauter et al., 2009). The processes shown in figure 2 in that article have the typical appearance of dendrites. A rhythm in calbindin has also been reported in the mouse lemur SCN (Cayetanot et al., 2007).
CAR has been thought to belong to a small group of Ca++ binding proteins largely bound to cell membranes (Baimbridge and Miller, 1992; Schwaller, 2014). More recent studies place CAR in a larger group with more complex roles in synaptic function. CAR does not now appear be fixed to the cell membrane but may migrate within cytoplasm under varying functional conditions (Schwaller, 2014; Matthews and Dietrich, 2015). In this context, we view the circadian changes in neuronal CAR noted in the present study to indicate that CAR has differing roles in synaptic function over the circadian day. This is likely to reflect differing modulation of synaptic activity, perhaps with a role for CAR as a “sensor” for Ca++-dependent feedback control of neuronal excitability (Arendt et al., 2013).
The varying location of CAR in SCN neurons among mammalian species discussed above is most consistent with the view that CAR expression in the rat SCN has evolved as a specific species adaptation in mechanisms of circadian regulation. The animals used in this study are from an inbred strain of Rattus norvegicus. The closest relative to Rattus norvegicus in the family Muridae is Rattus rattus. These species diverged about 2 million years ago (Verneau, 1998), and their geographical distribution differs substantially, making it likely that the environmental conditions in which each lives differs significantly. It would be interesting to know whether CAR localization is similar in the 2 species to begin understanding environmental determinants of calcium-binding protein type and localization in the circadian timing system.
Footnotes
Acknowledgements
Skilled technical support was provided by Joan C. Speh, Rebecca Danchenko, and Nadine Suhan. Supported by National Institutes of Health grant NS-16304.
Conflict of Interest Statement
The author has no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
