Abstract
Seasonal trade-offs in reproduction and immunity are ubiquitous in nature. The mechanisms that govern transitions across seasonal physiological states appear to involve reciprocal switches in the local synthesis of thyroid hormone. In long-day (LD) summer-like conditions, increased hypothalamic triiodothyronine (T3) stimulates gonadal development. Alternatively, short-day (SD) winter-like conditions increase peripheral leukocytes and enhance multiple aspects of immune function. These data indicate that the localized effects of T3 in the hypothalamus and leukocytes are photoperiod dependent. We tested the hypothesis that increased peripheral T3 in SD conditions would increase aspects of reproductive physiology and inhibit immune function, whereas T3 injections in LD conditions would facilitate aspects of immune function (i.e., leukocytes). In addition, we also examined whether T3 regulates hypothalamic neuropeptide expression as well as hypothalamic and splenic proinflammatory cytokine expression. Adult male Siberian hamsters were maintained in LD (15L:9D) or transferred to SD (9L:15D) for 8 weeks. A subset of LD and SD hamsters was treated daily with 5 µg T3 for 2 weeks. LD and SD controls were injected with saline. Daily T3 administration in SD hamsters (SD+T3) resulted in a rapid and substantial decrease in peripheral leukocyte concentrations and stimulated gonadal development. T3 treatment in LD (LD+T3) had no effect on testicular volumes but significantly increased leukocyte concentrations. Molecular analyses revealed that T3 stimulated interleukin 1β messenger RNA (mRNA) expression in the spleen and inhibited RFamide Related Peptide-3 mRNA expression in the hypothalamus. Moreover, there was a photoperiod-dependent decrease in splenic tumor necrosis factor–α mRNA expression. These findings reveal that T3 has tissue-specific and photoperiod-dependent regulation of seasonal rhythms in reproduction and immune function.
Seasonal rhythms in reproduction and immunity are common in temperate species (Nelson, 2004; Stevenson and Prendergast, 2015). Seasonal temporal strategies are important as they coordinate energetic activities across the year; winter is generally associated with reduced energy availability but requires the highest energy demands (Nelson et al., 1998; Sheldon and Verhulst, 1996). Seasonal rhythms in hormones involved in energy balance, such as leptin, have been implicated as mediators of yearly trade-offs according to energetic demands (Drazen et al., 2001; Demas, 2004). Thyroid hormones are also key regulators of energy balance and form an integral part of the neuroendocrine regulation of seasonal rhythms (Ebling, 2014). Thyroid hormones are similarly potent immunomodulators (Dorshkind and Horseman, 2000). Thus, thyroid hormones are poised to mediate seasonal trade-offs in energetic demands involved in timing reproduction and immune function.
Several neuropeptides located in the hypothalamus are involved in regulating the neuroendocrine timing of seasonal reproduction, such as gonadotropin-releasing hormone (gnrh; Stevenson et al., 2012), kisspeptin (kiss; Greives et al., 2007), and RFamide-related peptide 3 (rfrp3; Tsutsui et al., 2013). The annual change in day length is a predictive proximate cue that drives seasonal variation in the timing of reproduction (Stevenson and Ball, 2011). Increased summer day length (LD) triggers the local synthesis of triiodothyronine (T3) in the mediobasal hypothalamus, leading to gonadotropin secretion and subsequent gonadal development (Yoshimura et al., 2003; Rani and Kumar, 2014). Moreover, increased hypothalamic T3 is proposed to regulate the expression of several neuropeptides involved in the control of seasonal reproduction, such as RFRP3 and KiSS1 (Henson et al., 2013). Hypothalamic RFRP3 expression in both Siberian and Syrian hamsters is greater in LD compared to short-day conditions (SD; Ubuka et al., 2012; Mason et al., 2010, respectively). T3 implants or exogenous T3 injection in SD hamsters have been shown to induce growth of the gonads (Freeman et al., 2007; Barrett et al., 2007; Klosen et al., 2013). Daily T3 administration also acts in the hypothalamus to trigger increased anteroventral periventricular nucleus KiSS1, decreased arcuate nucleus KiSS1, and increased RFRP3 expression (Henson et al., 2013). It is clear that thyroid hormones are an integral component of the neuroendocrine circuitry that regulates seasonal reproduction; however, our understanding of the local role of triiodothyronine in the timing of annual rhythms in immune function is in its infancy.
Unlike seasonal rhythms in reproduction, immune function does not exhibit an overall potentiation or involution during any phase of the yearly cycle (Nelson, 2004; Sheldon and Verhulst, 1996). Instead, the immune system displays a collection of trait-specific enhancements at specific times across the year. Short winter-like photoperiods (SD) lead to enhanced immunosurveillance capacity (e.g., leukocyte proliferation; Bilbo et al., 2002; Stevenson et al., 2014). However, in some rodent species, humoral immune function is suppressed following adaptation to SD conditions (Drazen et al., 2000; Drazen et al., 2001). Thyroid hormones have been implicated in the regulation of multiple aspects of immune function, including T3-enhanced T-cell–dependent (type IV) delayed-type hypersensitivity reactions (DTH) skin responses (Chandel and Chatterjee, 1989), mitogen-induced splenocyte proliferation, and blood leukocyte concentrations (Chatterjee and Chandel, 1983). In Siberian hamsters, SD leukocytes have increased intracellular T3 concentrations; the local synthesis is likely involved in the timing of SD enhanced immune function, such as memory T-cell–dependent adaptive immune responses (DTH; Stevenson et al., 2014). Furthermore, exogenous T3 in LD conditions induced an increase in peripheral leukocyte concentrations to values similar to SD hamsters (Stevenson et al., 2014). These data indicate that in addition to regulating the neuroendocrine control of seasonal reproduction, T3 has a powerful effect on enhancing trait-specific aspects of immunity.
These patterns indicate a dual role of thyroid hormones for the seasonal control of reproduction and immune function: LD hypothalamic T3 stimulates neuroendocrine mechanisms that trigger gonadal development, and SD enhances leukocyte-T3–mediated components of immune function. The goal of this article was to examine the sufficiency of T3 to drive photoperiodic-dependent switches in immune and reproductive function in LD and SD conditions, respectively. To examine this goal, we sought to establish whether daily peripheral administration of T3 for 2 weeks in LD and SD conditions would induce inverse effects on reproduction and immunosurveillance. We hypothesized that T3 would facilitate measures of immune function in LD and stimulate reproductive development in SD animals. Siberian hamsters (Phodopus sungorus) are a common laboratory animal used to investigate seasonal biology. A simple switch in day length from LD (15 h light) to SD (9 h light) triggers a range of genetic and physiological changes leading to gonadal involution and immune plasticity (Heldmaier et al., 1981; Bartness and Wade, 1985; Bilbo et al., 2002; Prendergast et al., 2013; Stevenson et al., 2014). To assess the sufficiency of triiodothyronine on regulating neuroendocrine control of seasonal reproduction and immune function, we examined photoperiodic and daily injections of T3 on hypothalamic expression of 2 neuropeptides, gnrh and rfrp3, and downstream effects on body mass and testicular volume. We also surveyed the number of circulating leukocytes and proinflammatory cytokine expression in the hypothalamus and spleen, well-established measures of immune function (Dhabhar et al., 1995; Yellon et al., 1999; Bilbo et al., 2002). Finally, we investigated whether daily T3 injections would in part regulate the expression of 2 proinflammatory cytokines: interleukin-1β (il1β) and tumor necrosis factor–α (tnfα).
Materials and Methods
Subjects
Adult male hamsters (n = 31; 3-6 months old) were randomly selected from a colony maintained at the University of Aberdeen. Hamsters were housed in polypropylene cages illuminated for 15 h/d (15L:9D). Food and water were provided ad libitum; cotton-nesting material was available in the cage. All procedures were approved by the Animal Welfare and Ethics Review Board at the University of Aberdeen and conducted under the Home Office license (70/7917).
Study Design
Hamsters were group housed in LD (n = 15) or transferred to SD (9L:15D; n = 16) cabinets for 8 weeks (Arrowmight, Hereford, UK). LD and SD hamsters were then divided into 2 treatment groups that received daily saline controls (LD+S and SD+S) or T3 injections (LD+T3 and SD+T3). The final treatment group sample sizes were LD+S (n = 9), LD+T3 (n = 6), SD+S (n = 8), and SD+T3 (n = 8). Body mass and estimated testes volume (ETV; see below) were measured to confirm SD-induced weight loss and gonadal involution (PRE). Body mass was determined using aeADAM scales (Adam Equipment PGL2002; Adam Equipment, Milton Keynes, UK) and measured to ±0.1 g. To evaluate the effect of exogenous T3 on peripheral physiology, as well as hypothalamic and splenic messenger RNA (mRNA) expression, hamsters were given daily subcutaneous (s.c.) injections of 5 µg T3/100 g (T2877; Sigma-Aldrich, St. Louis, MO) for 2 weeks. This dose and injection regimen was selected based on previous work that revealed a significant increase in circulating leukocytes in LD male hamsters (Stevenson et al., 2014). Peripheral T3 readily crosses the blood-brain barrier, mediated in part by transporters such as monocarboxylate transporter 8 (MCT8; Heuer, 2007). Hypothalamic MCT8 exhibits photoperiodic variation and is proposed to facilitate the local concentration of T3 in brain regions implicated in the neuroendocrine control of seasonal rhythms (Herwig et al., 2013). SD and LD control hamsters received s.c. saline (0.1 mL) for the same 2-week period. Body weight measurements and ETVs were collected after 2 weeks of daily injections (POST).
Estimated Testes Volume
ETVs were used to determine the response to photoperiod and T3, in brief; ETV is calculated by testis width squared multiplied by length. Hamsters were lightly anaesthetized with isofluorane/oxygen vapors (4% induction, 1% maintenance), and testes were measured using analogue calipers. This approach to assess reproductive state is widely used, as it is strongly correlated with actual testicular volume (Gorman and Zucker, 1995). Testicular volumes of hamsters indicated reproductive competence (LD) or gonadal involution (SD).
Leukocyte Analyses
At the termination of the study, hamsters were anesthetized with isoflurane gas (4%), and 500 µL whole blood was collected via the right retroorbital sinus, using Natelson tubes coated with sodium heparin. A 30-µL aliquot of blood was mixed with 3% acetic acid at a 1:20 dilution. Leukocytes were counted under a light microscope (Zeiss Axioskop, Cambridge, UK), and values were obtained according to methods reported previously (Stevenson et al., 2014, Prendergast et al., 2003).
Quantification of Hypothalamic and Splenic RNA Expression
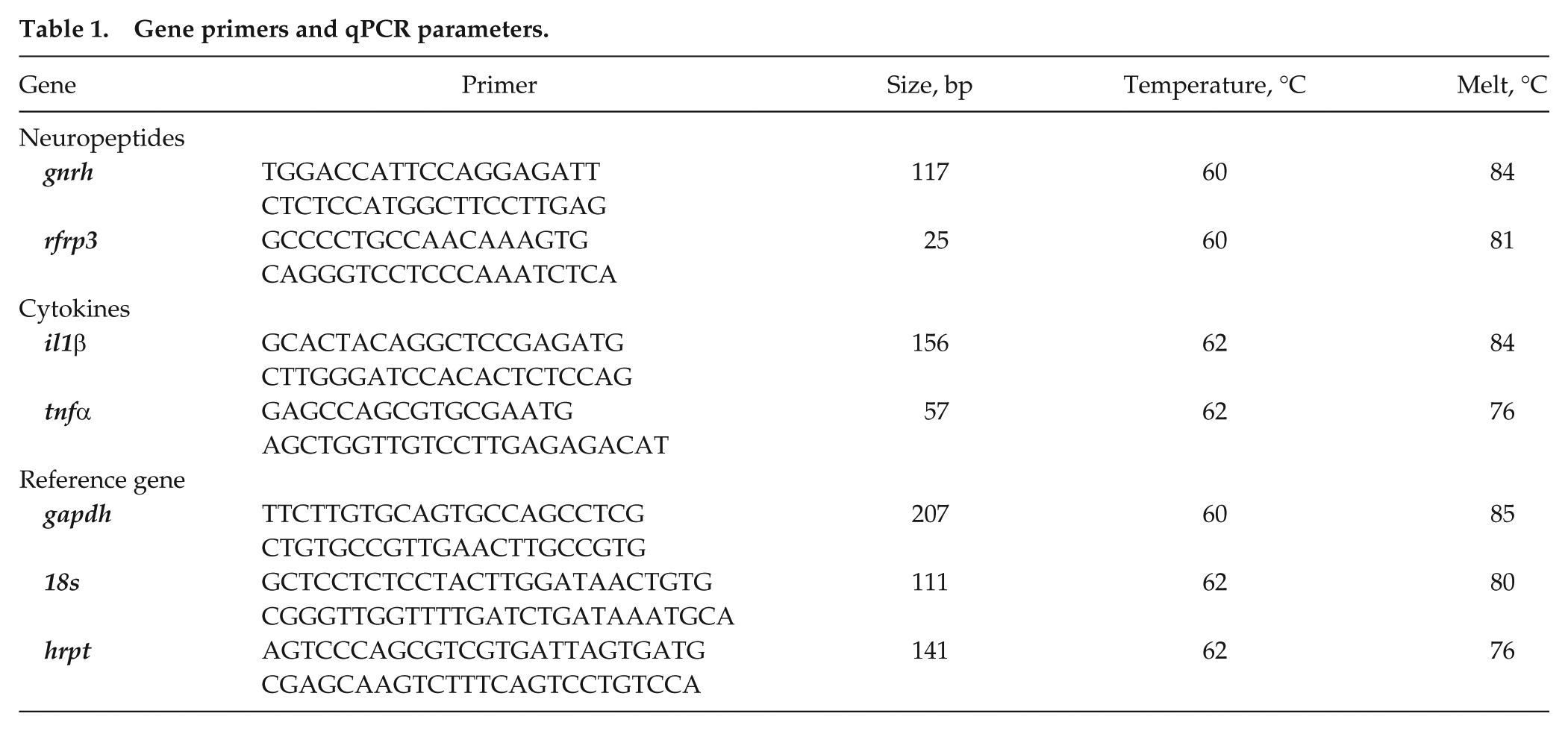
Hypothalamic and spleen tissues were rapidly dissected as previously described (Stevenson and Prendergast, 2013) and frozen in powder dry ice. RNA was extracted from tissues using Trizol (Thermo Fisher Scientific, Waltham, MA). Nucleic acid concentration and quality were determined by a spectrophotometer (Nanodrop; Thermo Fisher Scientific). Complementary DNA (cDNA) was synthesized using Superscript III (Invitrogen, Carlsbad, CA), and cDNA was stored at −20 °C until quantitative polymerase chain reaction (qPCR) was performed. All cDNA tissue samples were run in triplicates. qPCRs for mRNA expression in hypothalamic tissue were performed using BIORAD CFX96 (BIORAD, Hertfordshire, UK), and splenic mRNA was assayed by using Roche LightCycler 480 (Roche, West Sussex, UK). qPCR parameters were as follows: an initial denature at 95 °C for 30 secs, then 39 cycles of 95 °C for 10 sec, with annealing dependent on target mRNA (see Table 1) for 30 secs, and then an extension at 72 °C for 30 sec. A melting curve analysis was added to determine the quality and specificity of each reaction. Quantification of mRNA expression levels was accomplished with iQ Sybr Green Supermix (BIORAD). We used PCR Miner (Zhao and Fernald, 2005) to calculate reaction efficiencies (E) and cycle thresholds (CTs). According to the Minimum Information for Publication of Quantitative Digital PCR Experiments (MIQE) guidelines, samples that had efficiency values below 0.8 or above 1.2 were excluded from analyses (Bustin et al., 2009). The expression levels of hypothalamic transcript were determined using glyceraldehyde 3-phosphate dehydrogenase (gapdh) as the reference. For splenic transcripts, we used the average of 18s and hrpt cycling time, as these were found to be the most stable and unaltered housekeeping genes in these tissues. Fold change in expression for all target transcripts was calculated using 2−ΔΔCt.
Gene primers and qPCR parameters.
Statistical Analyses
Repeated 2-way analysis of variance (ANOVA) was conducted to examine the effects of T3 on body mass and ETVs. Leukocyte counts and hypothalamic and splenic mRNA expression were analyzed using 2-way ANOVAs. Post hoc analyses were performed using Fisher’s least significant difference (LSD) method. Log transformation was performed on mRNA values when violations of normality were observed. Statistical analyses were performed using SigmaStat 12.0 (Systat Software, San Jose, CA) and difference considered significant when p < 0.05.
Results
Photoperiod and T3 Regulation of Peripheral Physiology
Repeated 2-way ANOVA revealed a significant interaction of photoperiod and T3 treatment on hamster body mass (F = 20.72; p < 0.001; Fig. 1A). Post hoc analyses confirmed that saline-injected controls did not exhibit a significant change in body mass after 2 weeks of daily injections (p > 0.08 for LD+S and SD+S). Daily T3 injections were found to significantly reduce body weight in LD+T3 (p < 0.001) and SD+T3 (p < 0.001).
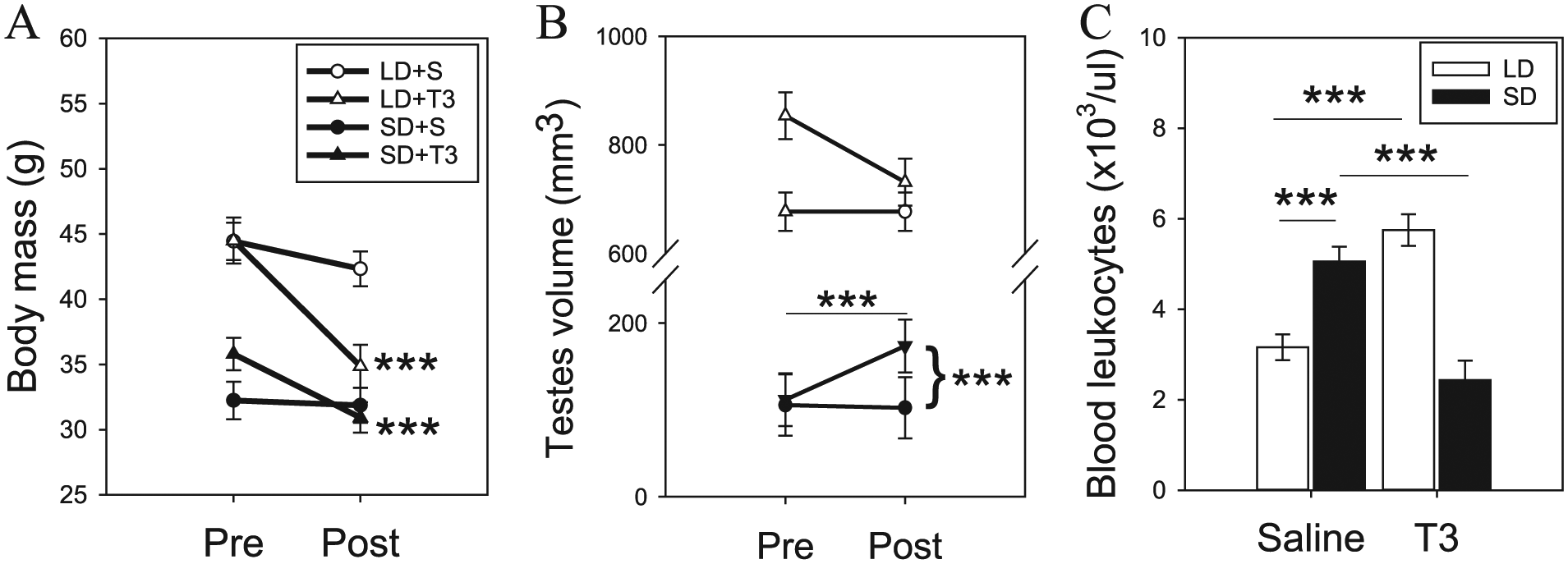
Photoperiod and T3 regulation of body mass, testicular volume, and blood leukocytes. (A) Short-day (SD) treatment significantly reduced body mass after 8 weeks (PRE); daily T3 for 2 weeks (POST) significantly decreased mass in hamsters. (B) SD significantly reduced testicular volume, and T3 treatment induced a slight increase after 2 weeks. (C) T3 significantly reduced leukocytes counts in SD hamsters to levels that were equivalent to long-day (LD) hamsters. Conversely, T3 treatment in LD hamsters significantly increased leukocytes. Asterisks denote significant differences. ***p < 0.001.
Daily T3 injections led to a significant increase in ETV only in SD housed hamsters (F = 5.34; p < 0.005; Fig. 1B). Post hoc analyses indicated that both LD and SD hamsters injected with daily saline maintained constant ETV levels (p > 0.85). Similarly, daily T3 injections did not significantly augment ETV in LD hamsters (p < 0.05). In SD, T3 injections resulted in a significant increase in ETV (p < 0.001). These data suggest that T3 has photoperiod-dependent effects on testicular volumes; there was a stimulatory effect only after gonadal involution had occurred.
Two-way ANOVA analysis also revealed a significant interaction on leukocyte concentrations (F = 54.61; p < 0.001; Fig. 1C). Consistent with previous publications, post hoc analyses confirmed that SD hamsters had significantly greater peripheral leukocyte numbers compared to LD controls (p < 0.001). Daily T3 injections had significant photoperiod-dependent effects; LD hamsters had significantly greater leukocyte counts (p < 0.001), whereas under SD, there was a reduction (p < 0.001). These robust findings indicate that exogenous T3 can have opposing effects on leukocyte numbers that are, in part, regulated by photoperiodic history.
Hypothalamic mRNA Expression Is Regulated by Photoperiodic Condition
Next, we examined the impact of photoperiodic condition and exogenous T3 on the expression of reproductive neuropeptides, gnrh and rfrp3, as well as proinflammatory cytokines, il1β and tnfa. We did not detect significant variation in gnrh expression across photoperiodic condition (F = 0.36; p = 0.55; Fig. 2A), T3 treatment (F = 0.22; p = 0.64), or an interaction (F = 0.61; p = 0.44). We observed a significant photoperiodic effect on rfrp3 expression, with reduced levels in SD hamsters (F = 54.87; p < 0.001; Fig. 2B). Furthermore, daily T3 injections also reduced rfrp3 expression (F = 5.20; p < 0.05). There was no significant interaction for rfrp3 expression (F = 0.33; p = 0.57).
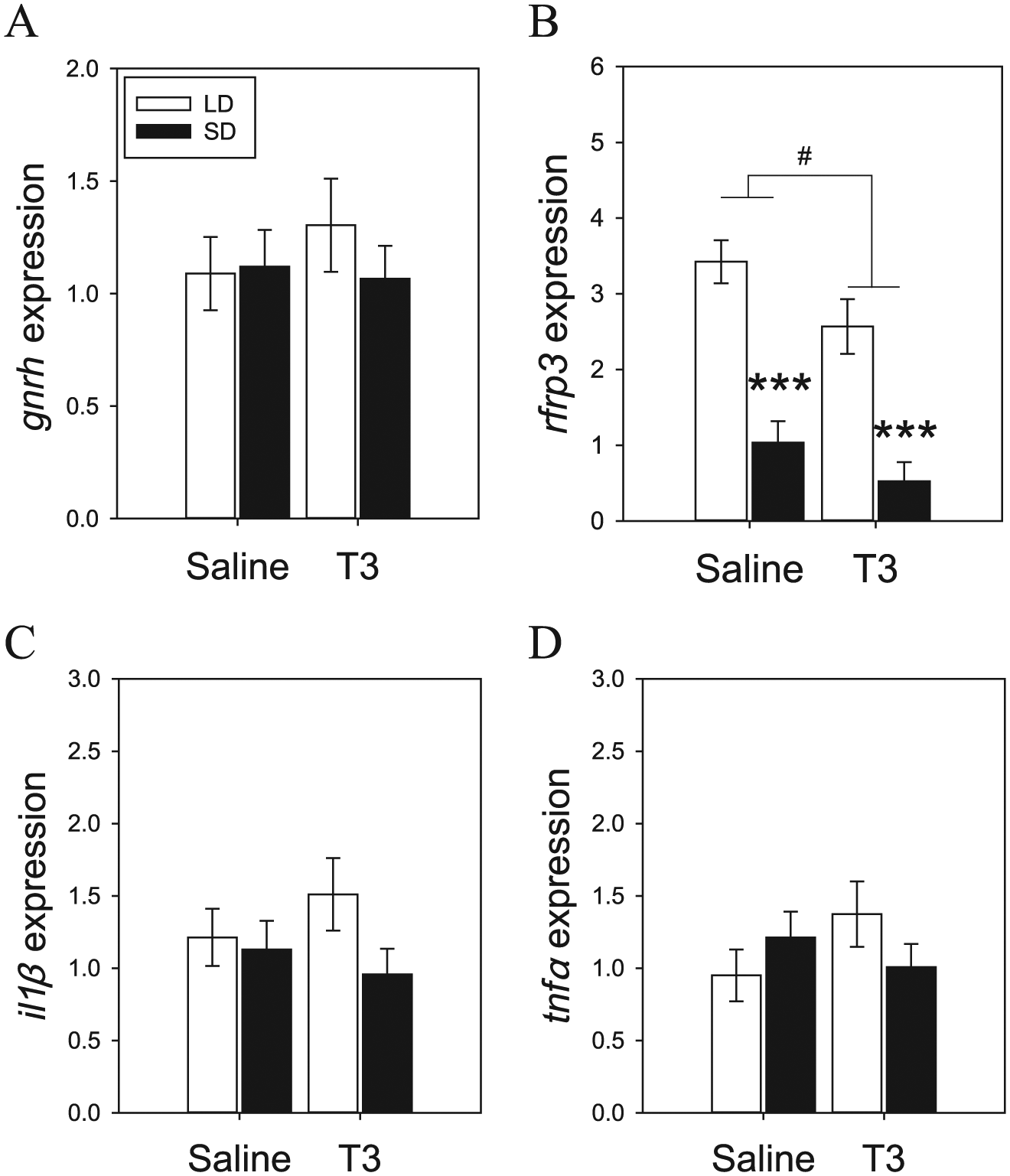
Light- and T3-dependent effects on hypothalamic neuropeptides and inflammatory cytokines. (A) Exposure to short day (SD) or T3 did not significantly affect the levels of gnrh expression. (B) Prolonged exposure to SD as well as daily T3 treatment reduced rfrp3 expression in the hamster hypothalamus. There was no significant effect of light or T3 on (C) il1β or (D) tnfα expression. Asterisks depict a significant main effect of photoperiod condition (***p < 0.001) and main effect of T3 treatment (#p < 0.05).
Analyses of hypothalamic il1β and tnfα indicate that these cytokines are constitutively expressed across photoperiodic conditions and are not regulated by T3. There was no significant photoperiod (F = 2.05; p = 0.16; Fig. 2C), T3 dependent (F = 0.01; p = 0.98), or interaction (F = 1.64; p = 0.21) for il1β expression. Similarly, tnfα expression was not driven by photoperiod (F = 0.08; p = 0.78; Fig. 2D), daily T3 (F = 0.34; p = 0.56), or an interaction of the 2 factors (F = 2.81; p = 0.10). Overall, these data suggest that T3 does not appear to regulate proinflammatory cytokine expression in the hamster hypothalamus.
T3 Regulation of Proinflammatory Cytokines in the Hamster Spleen
We found that il1β expression was significantly upregulated in the hamster spleen after 2 weeks of daily T3 injections (F = 28.12; p < 0.001; Fig. 3A). However, there was no significant photoperiod effect (F = 2.34; p = 0.14) or a significant interaction (F = 0.18; p = 0.67).
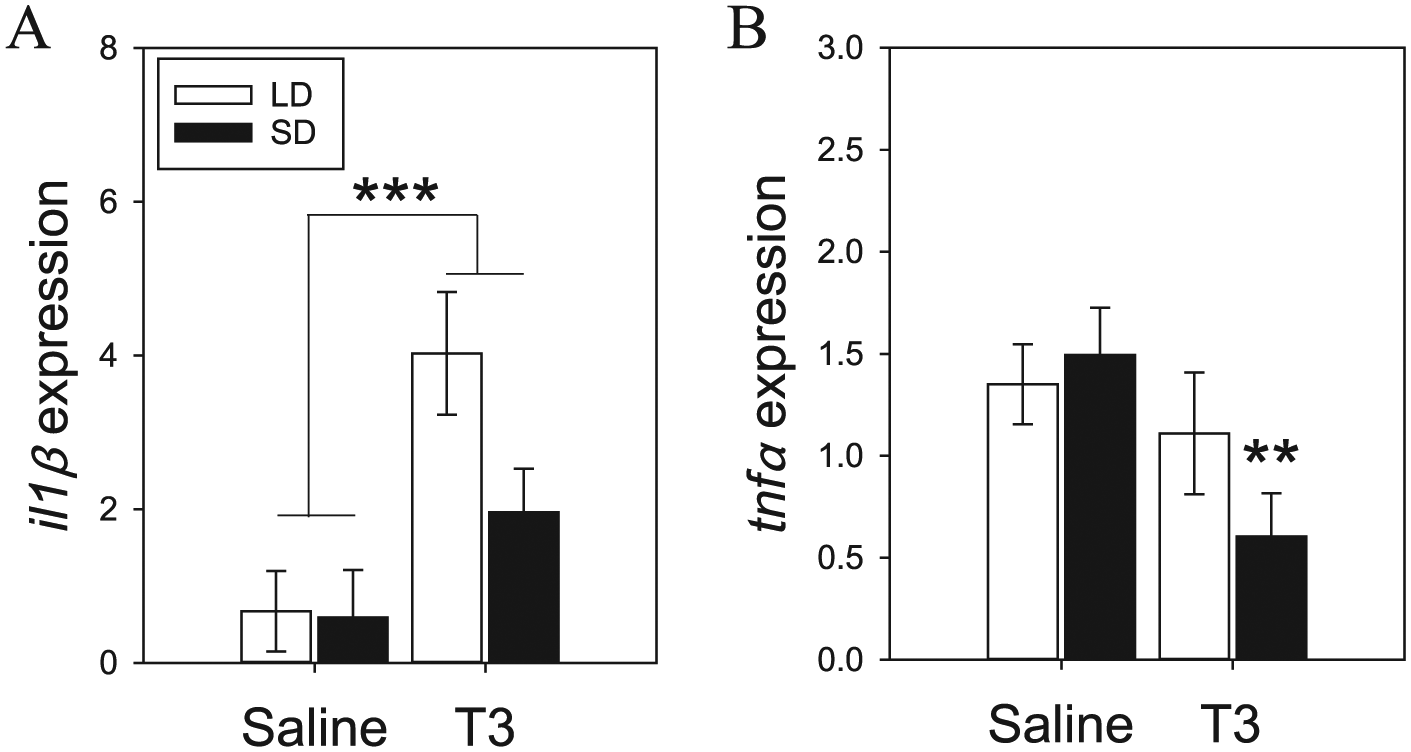
Proinflammatory cytokine messenger RNA expression in hamster spleen. Daily T3 injections resulted in a significant increase in il1β expression (A). T3 significantly reduced tnfα expression, but this was primarily driven by short-day (SD) hamsters (B). Asterisks depict significance at **p = 0.01 and ***p < 0.005. LD, long day.
Two-way ANOVA revealed a significant main effect of daily T3 injections on spleen tnfα expression (F = 5.63; p < 0.05; Fig. 3B). There was no significant main effect of photoperiod (F = 0.58; p = 0.45) or a significant interaction (F = 1.85; p = 0.19). Post hoc analyses indicated that T3-induced reduction in tnfα expression was primarily driven by lower levels in SD+T3 hamster compared to SD+S (p = 0.01). There was no significant difference between LD+S and LD+T3 (p = 0.55).
Discussion
In this study, we demonstrate that exposure to SD reduced body weight and testicular volume, as well as enhanced circulating leukocyte concentrations in adult male Siberian hamsters. Our work corroborates the findings that hamsters maintained in LD photoperiods have greater hypothalamic RFRP3 expression compared to SD-photoregressed animals (Ubuka et al., 2012; Mason et al., 2010). We identified that T3 injections led to a significant inhibition in peripheral leukocyte concentrations in SD hamsters (i.e., SD+T3), a modest increase in ETV, and reduced body mass. Furthermore, daily T3 injections significantly reduced spleen tnfα expression only in SD hamsters. We reveal that T3 categorically decreased hypothalamic rfrp3 expression and increased spleen il1β expression, regardless of photoperiodic condition. These data indicate that exogenous T3 treatment exerts distinct photoperiod- as well as cell- and tissue-specific control over measures of immune and reproduction function.
This study permitted the dissection of timescale effects of T3-induced changes in multiple aspects of organismal physiology. Circulating concentrations of T4 and T3 do not exhibit photoperiodic variation and instead maintain constitutive levels in the periphery (O’Jile and Bartness, 1992). Cell-specific expression of thyroid hormone enzymes, referred to as deiodinases (DIO), is critical for regulating intracellular levels of T4 and T3 (St. Germain et al., 2009). Thus, photoperiodic regulation of hypothalamic and leukocyte DIO expression provides the ability to enhance the effects of T3 on localized genomic and physiological function (Prendergast et al., 2013; Stevenson et al., 2014; Stevenson and Prendergast, 2015). T3 injections in SD hamsters induced a 51% inhibition in peripheral leukocytes, a 40% increase in ETV, and a negligible effect on body mass (3% inhibition; within margin of error). Our findings also illustrate that T3 effects on peripheral physiology and immune function are photoperiod dependent. In LD hamsters, body mass was significantly reduced, there was no change in testicular volume, and leukocytes increased to levels comparable to SD controls. These data suggest that the diverse tissues/cells of the peripheral nervous system exhibit photoperiodic- and T3-dependent regulation.
Previous work has illustrated that both central and peripheral exogenous T3 stimulates reproductive development in the SD phenotype (Freeman et al., 2007; Klosen et al., 2013; Barrett et al., 2007). Here, we confirm that daily T3 injections in SD hamsters (SD+T3) significantly increased ETV. However, we observed that T3 reduced hypothalamic rfrp3 expression regardless of photoperiodic condition. Henson and colleagues (2013) demonstrated that the numbers of RFRP3 cells, determined by immunocytochemistry, are greater after daily T3 injections in SD hamsters. Since we observed a reduction in rfrp3 expression in LD+T3, it is likely that T3 has an inhibitory effect on RFRP3 function in the hamster hypothalamus. We propose that T3 inhibits rfrp3 expression, and the greater RFRP3 cell numbers reported by Henson and colleagues (2013) likely reflect an accumulation of protein content due to reduced transport/release from nerve terminals. Our findings also provide evidence that chronic exposure to photoperiod and T3 does not regulate gnrh expression. Indeed, hypothalamic GnRH content exhibits slight variation during rapid transitions from LD to SD conditions (Bernard et al., 1999). It remains a possibility that acute changes in local T3 could augment hypothalamic GnRH expression. These data indicate that T3-mediated effects on the neuroendocrine-gonadal axis are likely due to the reduction in rfrp3 expression leading to gonadal development in SD hamsters. The absence of any effect on testicular volumes in LD is presumably the result of a “ceiling effect” in which the testes are already at, or near, maximum volume.
In this article, we confirm that LD+T3 hamsters have greater blood leukocyte concentrations compared to LD+S (Stevenson et al., 2014) and reveal that T3 effects are photoperiod dependent, as SD+T3 exhibit significantly lower concentrations. We suggest that the reduced leukocytes in SD+T3 are not the result of direct inhibition on immune function by thyroid hormones. Instead, peripheral T3 may be involved in diverting energy demands in SD hamsters to testicular growth and subsequent steroidogenesis (e.g., testosterone). The increased energy demands for reproduction and/or the increased production of testosterone reduce leukocyte counts and, ultimately, immune function (Prendergast et al., 2003; Trigunaite et al., 2015). At a molecular level, we demonstrate that T3 significantly increased splenic il1β expression. T3 has been shown to stimulate il1β expression in human bone marrow stromal cells (Kim et al., 1999), suggesting that T3 can potentiate il1β expression across diverse tissues. Interestingly, we did not detect a significant difference in hypothalamic il1β expression, indicating a degree of tissue-specific regulation. A similar pattern was observed for tnfα expression, as T3 significantly reduced spleen levels in a photoperiod-dependent manner and showed complete absence of an effect on hypothalamic expression. In addition to the spleen, T3 increased tnfα expression in Kupffer (Valencia et al., 2004) and liver cells (Tapia et al., 2003). Thus, T3 has a powerful effect on multiple aspects of immune function (i.e., leukocytes and proinflammatory cytokine expression) that is driven by photoperiodic history and is tissue specific.
In conclusion, our results indicate that localized signaling of T3 differentially regulates seasonal timing in immune and reproduction function. T3 has direct effects on proinflammatory cytokine expression in spleen tissue and hypothalamic rfrp3 expression. In both cases, T3 effects on rfrp3 and il1β are regulated independently of photoperiod condition. However, some exogenous T3 effects are dependent on photoperiodic condition in hamsters, such as enhanced leukocytes in LD and gonadal development in SD. Disruption of T3 signaling likely has long-term consequences on the timing of reproduction and/or immune responses in a seasonally dependent manner (Stevenson et al., 2015). We propose that enhanced peripheral T3 concentrations in the Siberian hamster override the presiding photoperiodic signal, directly stimulate reproductive development in SD, and indirectly lead to the inhibition in leukocyte cells.
Footnotes
Acknowledgements
The study hypotheses and design were conceived by TJS. RB and TJS conducted, collected, and analyzed the data. RB, MD, and TJS wrote the paper. This work was financed by the University of Aberdeen College of Life Sciences and Medicine fund to TJS. We thank Elisabetta Tolla for her technical assistance.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
