Abstract
The testis provides not just one but several models of temporal organization. The complexity of its rhythmic function arises in part from its compartmentalization and diversity of cell types: not only does the testis produce gametes, but it also serves as the major source of circulating androgens. Within the seminiferous tubules, the germ cells divide and differentiate while in intimate contact with Sertoli cells. The tubule is highly periodic: a spermatogenic wave travels along its length to determine the timing of the commitment of spermatogonia to differentiate, the phases of meiotic division, and the rate of differentiation of the postmeiotic germ cells. Recent evidence indicates that oscillations of retinoic acid play a major role in determining periodicity of the seminiferous epithelium. In the interstitial space, Leydig cells produce the steroid hormones required both for the completion of spermatogenesis and the development and maintenance of male sexual characteristics throughout the body. This endocrine output also oscillates; although the pulse generator lies outside the gonad, the steroidogenic function of Leydig cells is tuned to a regular episodic input. While the oscillations of the intratubular and interstitial cells have multihour (ultradian) and multiday (infradian) periodicities, respectively, the functions of both compartments also display dramatic seasonal rhythms. Furthermore, circadian rhythms are evident in some of the cell types, although their amplitude and pervasiveness are not as great as in many other tissues of the same organism, and their detection may require methods that recognize the heterogeneity of the testis. This review examines the periodicity of testicular function along multiple time scales.
Rhythms of Spermatogenesis
What defines a male? To comparative biologists, it is the production of large numbers of motile gametes (Ghiselin, 1974). The testis produces spermatozoa continuously during the breeding season, although they must mature in the epididymis and undergo capacitation in the female reproductive tract before they can fertilize an egg. In order to achieve this continuity, spermatogenesis is staggered along the length of the seminiferous tubule. The precise spatiotemporal patterning of cell division and differentiation in the spermatogenic epithelium has been most intensively studied in rodents, ungulates, and primates.
Writing about comparative aspects of spermatogenesis over 45 years ago, Edward Roosen-Runge (1969) wrote, “Spermatogenesis in tunicates, playthelmithes, echinoderms and many other phyla (and classes) has been hardly investigated at all so far.” Among the invertebrates, the situation has not markedly improved as attention has focused consistently on just a few model organisms such as Drosophila melanogaster (Fuller, 1993) and Caenorhabditis elegans (Ellis and Stanfield, 2014) and more sporadically on animals of commercial importance, including some of the lepidoptera (Friedlander et al., 2005), hymenoptera (Sawarkar and Tembhare, 2015), and crustacea (Haley, 1984). Although testicular function in nonamniote vertebrates has been studied more extensively (see Ramm et al., 2014, for review), there has likewise been scant emphasis on rhythmicity apart from description of annual cycles. With few exceptions, therefore, the focus of this review will be on the mammalian testis.
Unlike females, which cease to produce gametes soon after birth, males renew the population of germ cells continuously from the time of puberty until the end of life. This requires not only the production of cells destined to differentiate but also the maintenance of a population of self-renewing stem cells that ensures against depletion of the source of new gametes. In mammals, the mitotic and the meiotic processes occur in compartments separated by Sertoli cell tight junctions that form the blood-testis barrier. This barrier prevents immunoglobulins, hormones, and other molecules from reaching meiotic and postmeiotic germ cells. As the spermatogonia commit to differentiate, they pass as primary spermatocytes to the adluminal compartment. There they undergo meiotic recombination and divide into secondary spermatocytes, which in turn divide once more into postmeiotic round haploid spermatids. These cells then start a process known as spermiogenesis, in which the spermatids differentiate into morphologically mature sperm.
A wave of coordinated events, including mitotic division of the spermatogonia, progression through the stages of meiosis, and release of the spermatozoa (spermiation), travels along the seminiferous tubule, much as thousands of fans form a wave as they rise to cheer in adjacent sections of a domed football stadium. Once the spermatogonia are committed to meiosis, each additional maturational step is extremely well coordinated in time and space with adjacent segments of the seminiferous tubule. A histologist inspecting a cross section of the tubule will see characteristic “assemblies” of germ cells caught at particular stages of meiotic division and differentiation. These constellations of cell types are, by convention, classified as stages of the seminiferous tubule (see Russell et al., 1990; Fig. 1). To pursue the analogy of the wave in the stadium, if each football fan were to release a helium balloon as he rose, three would be at various and characteristic points of ascent by the time the first reached the stadium’s roof, which corresponds to the lumen of the tubule. The period of the spermatogenic cycle, as empirically determined in studies in which the last S phase is labeled by injection of 3H-thymidine, is the time it takes for a cell to advance to the next vertical level of each stage. In our analogy, the period of the wave in the stadium can be determined not only by measurement of the interval that elapses between the successive occasions on which the fan stands and releases the balloon but also by the time it takes for the balloon to reach the altitude characteristic of that phase. The period of the cycle is species specific and genetically determined: the period of the cycle of the seminiferous epithelium ranges from about 8.6 days in mice and Syrian hamsters (Clermont and Trott, 1969) to 10 days in rams (Ortavant, 1958), 11.6 days in macaques (Clermont and Antar, 1973), 13 days in rats (Clermont and Harvey, 1965), 16 days in man (Heller and Clermont 1963; Clermont, 1969), and 17 days in Chinese hamsters (Lok et al., 1982).
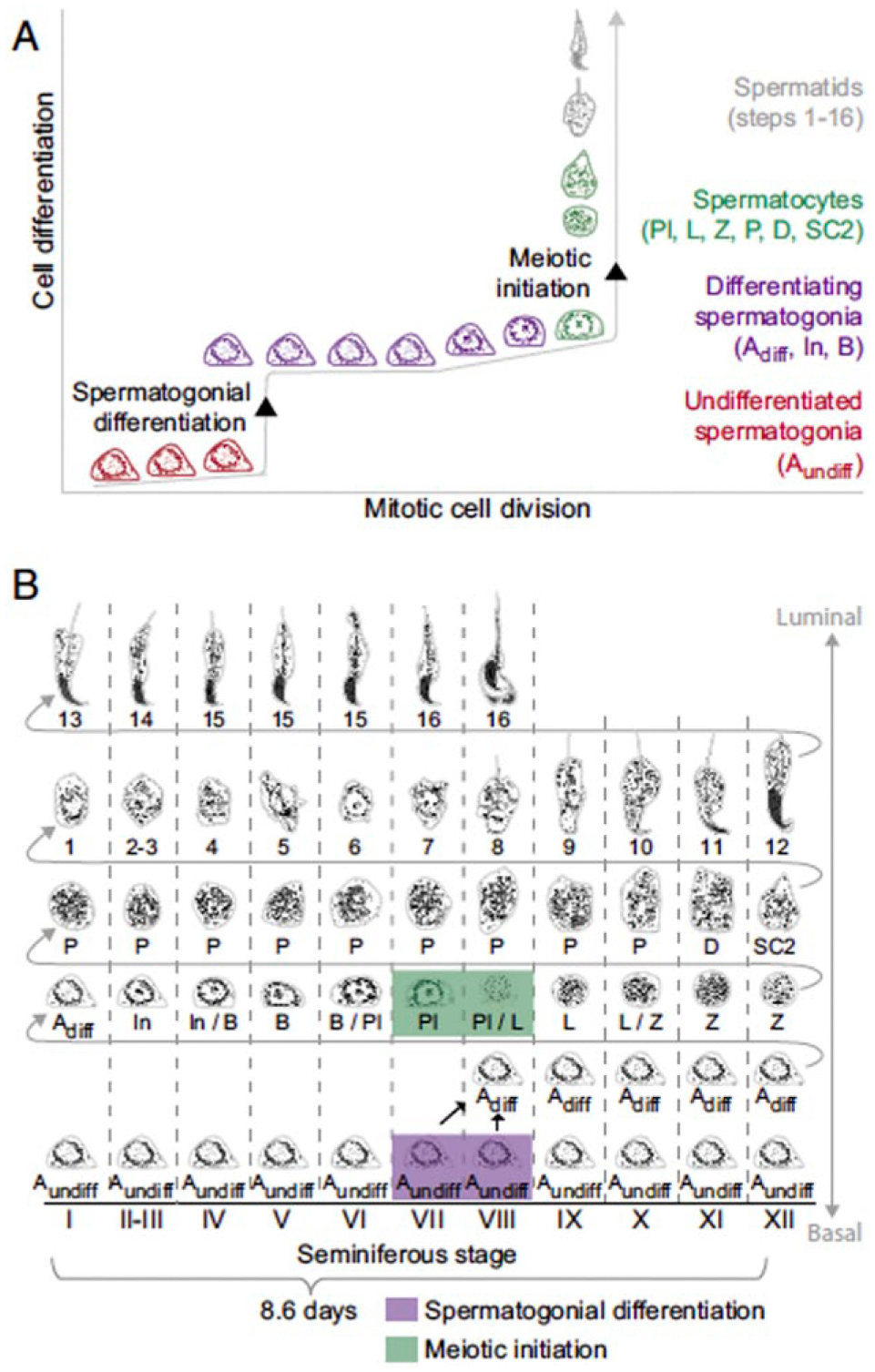
Depiction of (A) spermatogonial cell divisions and (B) the cycle of the seminiferous epithelium in laboratory mice. Note that mitotic divisions of undifferentiated (Aundiff) and differentiated spermatogonia (In, B) occur at intervals of 20 to 60 h, but appearance of these spermatogonial cell types occurs at specific phases of the 8.6-day cycle of the seminiferous epithelium. In histological studies, adjacent regions of the seminiferous tubule are in successive phases of this cycle. The stage of the epithelium is marked by Roman numerals; each association is characterized by a particular assortment of gametes in particular stages of meiotic division (D, diplotene; L, leptotene, P pachytene, Pl, preleptotene) and spermatocyte maturation (SC2, secondary spermatocyte; Z, zygotene). Spermatid differentiation, which occurs after completion of meiosis, is divided into 16 steps. Reprinted with permission from Endo et al. (2015).
The Cell Cycle of Spermatogonia
The meiotic and maturational events, although under strict temporal control, are not themselves rhythmic in the sense that they happen only once in the lifetime of the spermatocyte. In contrast, mitotic divisions of the spermatogonial stem cells (SSCs) and their daughters are rhythmic. Their period, however, is shorter than that of the seminiferous epithelium. The A-single (As) spermatogonia are the stem cells of the rodent testis. They reside in specialized niches that are in close contact across the basement membrane with the interstitium and likely have unique apposition to the vasculature (Yoshida et al., 2007; de Rooij, 2015). Their daughter cells remain connected through intercellular bridges and are designated A-paired (Apr) spermatogonia. These undergo clonal division to generate A-aligned (Aal) spermatogonia, which have the capacity to differentiate into A1 spermatogonia. The population of spermatogonia in the basal compartment of the tubule expands as much as 4000-fold from the initial stem cells, as sequential divisions produce A1, A2, A3, A4, Intermediate, and B spermatogonia, the last of which divide into spermatocytes and enter meiotic prophase. The timing of cell divisions and regularity of the cycle differ between the stages of spermatogonial differentiation; each transition between spermatogonial cell types is closely linked to the stage of the epithelial cycle and the interval between the initiation of the 6 mitotic divisions and the onset of meiosis spans one cycle of the seminiferous epithelium. In rodents, the As, Apr, and Al spermatogonia divide asynchronously with a cell cycle length of 60 to 90 h. Thus, they undergo 2 to 3 meiotic divisions over the course of each cycle of the epithelium and proliferate most actively in stages XII to III (Lok et al., 1982; de Rooij, 2015). The A1 spermatogonia, in contrast, go through S phase in stage VIII (Lok et al., 1982). The A2 through B spermatogonia express the tyrosine kinase receptor c-kit, whose stem cell factor ligand is produced by Sertoli cells in response to follicle-stimulating hormone (FSH). The strategy is similar in primates, as particular stages of spermatogonia divide at specific stages of the cycle of the seminiferous epithelium (Adark cells, proposed to be a stem cell reserve population, at stage V; Apale at stages IX-X; B1 at stage XII; B2 at stage II; B3 at stage IV; B4 at stage VI; Clermont, 1969).
Cyclic cell division is best understood and most thoroughly modeled in budding yeast, in which cyclin-dependent kinases control checkpoints for progression through the cell cycle (Gerard et al., 2015). The period of division is regulated by signals related to cell size and environmental conditions, so that the period can vary greatly. What controls the periodicity of mitotic divisions of the spermatogonia? Spermatogonial stem cells isolated from mice and rats show average doubling times of 5.5 days and 11 days, respectively (Ryu et al., 2005; Hamra et al., 2005), when cultured in isolation and supplemented with specific growth factors. These studies do not permit an estimate of the period or the regularity of cell divisions. In such in vitro preparations, the synchrony of cell division within the niche provided by the environment of the basal tubule is likely absent. In vivo, the cell cycle is arrested prior to puberty in Go/G1 phase during stages II to VIII. Thereafter, the period of division of spermatogonial stem cells, as well as their decision to self-renew or differentiate, depends on an extensive combination of growth factors produced by the Sertoli cells and the germ cells (see Payne, 2013, and de Rooij, 2015, for review). Despite the suggestion of modulation by developmental and environmental perturbations, the length of the spermatogonial cell cycle may be relatively constant in postpubertal animals. Leydig cells and peritubular myoid cells may regulate these stem cells and inhibit their differentiation through release of colony-stimulating factor 1 (Oatley et al., 2009). Although some cyclin-dependent kinases control spermatogenesis and are made in the testis (Mikolcevic et al., 2012; Zindy et al., 2001), their role in control of spermatogonial division (as opposed to spermatocyte maturation or gonadotropin secretion) is unclear. The initial period of spermatogonial divisions at the onset of puberty is faster than that of the adult. Temperature may regulate the period of the cycle of spermatogonial divisions either in poikilotherms or in mammals before testicular descent.
The Cycle of the Seminiferous Epithelium
The differentiation of spermatogonia and their entrance into the preleptotene (meiotic) stage are the first phase markers of the cycle of the seminiferous epithelium. Control of these events is better understood than the initial division of the SSCs and division of their daughter cells up to the point of differentiation. The membranes of germ cells and Sertoli cells are specialized to allow contact and presumably coordination, not only during the stage at which the spermatogonia initiate their division and commit to differentiation but throughout the process of maturation of the daughter cells (Ventela et al., 2002). The same Sertoli cell retains contact with several dozen daughter cells of the initial division throughout the weeks over which the spermatids mature into spermatozoa. Although they do not often divide in the adult testis, the Sertoli cells undergo cycles of expression of specific genes whose period matches that of the germ cells (Oatley et al., 2011; Sugimoto et al., 2012; Hasegawa and Saga, 2012; Zimmermann et al., 2015). Among these is the FSH receptor, levels of whose transcript peak in stage I of the cycle of the seminiferous tubule in rats and through which the neuroendocrine axis commands tubular function (Heckert and Griswold, 1991; Kliesch et al., 1992; Rannikko et al., 1996). While hormonal regulation is critical, FSH and testosterone determine how many cells survive but not the timing of the divisions or the latency to mature (Clermont and Harvey, 1965; Ortavant, 1958; O’Shaughnessy, 2014). This may reflect the fact that the receptors for these hormones are expressed in the Sertoli cells, not the germ cells.
Elegant experiments involving transplantation of spermatogonia across species indicate that the period of the seminiferous epithelium remains that of the donor (Franca et al., 1998; Zeng et al., 2006). An analogy may be made between these xenograft experiments and classic circadian transplantation studies in which pacemaker properties are confirmed by expression of the phase or period of the donor (Zimmerman and Menaker, 1979; Page, 1982; Ralph et al., 1990). Nevertheless, an intricate set of signals arising from the Sertoli cells coordinates the cycle of the spermatogenic epithelium. Experiments with Sxr mice show that Sertoli cells containing an extra X or lacking a Y chromosome can support meiosis. However, the normal differentiation of germ cells, and perhaps its timing, requires Y chromosome–dependent expression of autosomal genes in the somatic cells of the testis. It is not clear whether this regulation is cell autonomous or depends on paracrine regulation of Sertoli cells by germ cells (Ishii et al., 2007).
Can the spermatogenic cycle be modeled in such a way as to identify the key components that set its period and phase? Study of a variety of oscillations has revealed intricate and often interlocked feedback loops with negative and positive components that are amenable to mathematical modeling (Goldbeter et al., 2012). Examples include spatiotemporal oscillations of Notch1 and delta-like 1 ligand (DII1) expression that govern the addition of somites in vertebrate embryos (Bone et al., 2014) and generation of neural progenitors (Imayoshi et al., 2013; Wang et al., 2015). Although cell autonomous, the nested transcriptional-translational feedback loops of mammalian circadian oscillators provide another illustration of this theme (Gustafson and Partch, 2015). While the extent to which the details of the spermatogenic oscillator resemble these models remains to be determined, it is clear that Notch, Wnt, and FGF signaling pathways participate in spermatogonial division and differentiation. Recent evidence suggests that a local, episodic generation of all-trans retinoic acid (RA) coordinates the differentiation of spermatogonia and their commitment to entry meiotic prophase, as well as contributes to the temporal control of germ cell maturation (Fig. 2). This may reflect a general role of RA, as it may also operate upstream of Notch and FGF in the differentiation of embryonic stem cells into neurons (Chuang et al., 2015).
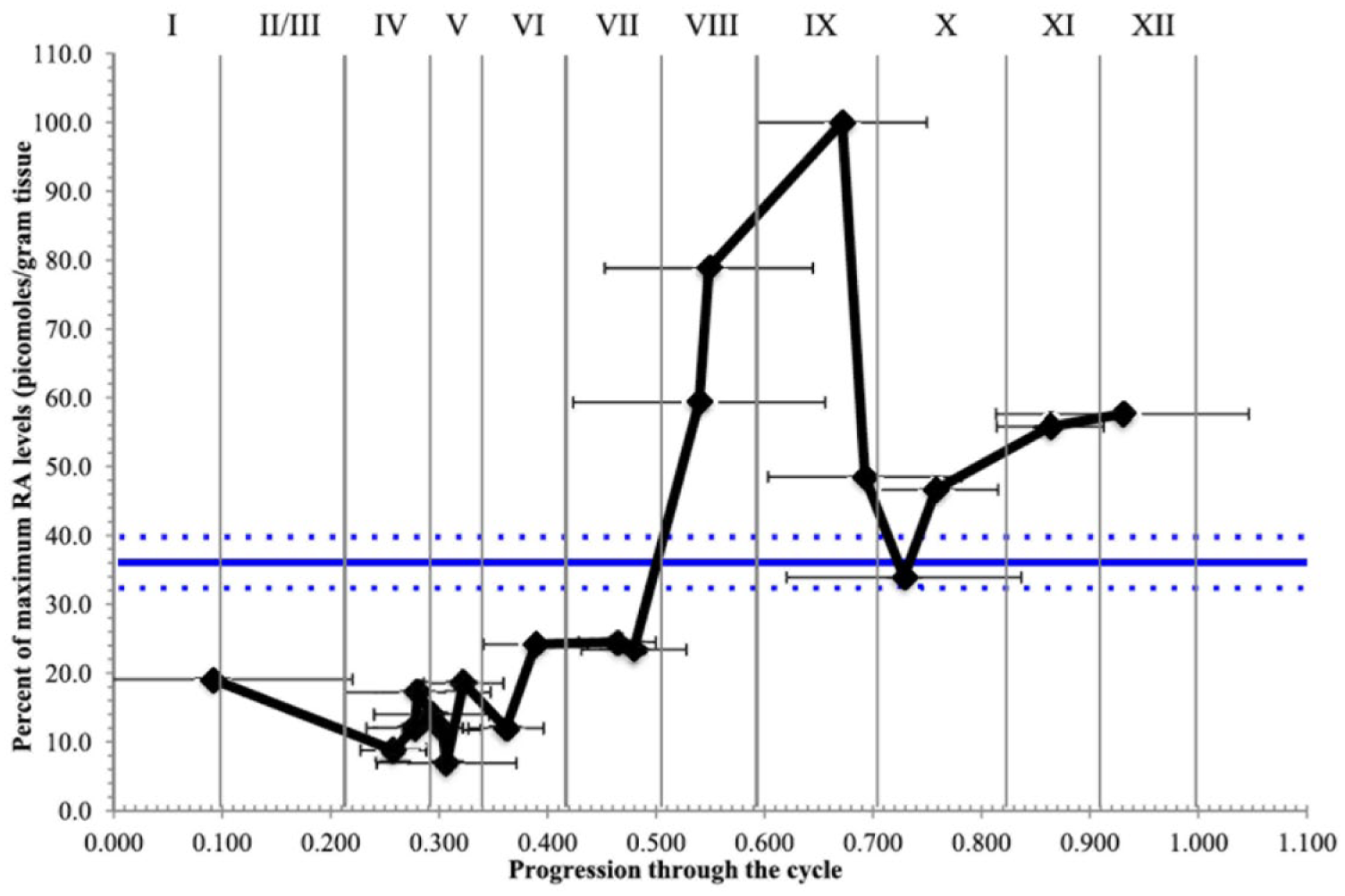
Quantitation of a gradient of retinoic acid (RA) concentrations in the seminiferous tubule. Control (gray line) represents RA levels in mice that did not receive a synchronizing injection of bisdichloroacetyldiamine (WIN 18,446, an inhibitor of retinaldehyde dehydrogenases). Black line depicts RA levels, relative to maximum, as a function of stage of the seminiferous epithelium in animals that received a synchronizing injection between 42 and 50 days prior to sacrifice. RA levels at the peak (stages VII to IX) differ significantly from those at the nadir (stages II to VI). Spermatogonia expressing STRA-8 (the protein product of stimulated by retinoic acid gene 8) were detected by immunocytochemistry in order to confirm staging of the seminiferous epithelium. Production of RA coordinates the seminiferous cycle, controlling the commitment of spermatogonia to differentiate, their progression through meiosis, changes in the blood-testis barrier, and spermiation. Reprinted with permission from Hogarth et al. (2015).
Oscillations of Retinoic Acid Determine Periodicity of the Seminiferous Epithelium
Elimination of dietary vitamin A results in deficiency of RA and the arrest of spermatogenesis. Administration of vitamin A restarts spermatogenesis synchronously at stage VII throughout the testis. Hogarth et al. (2015) have recently quantified patterns of RA concentration in seminiferous tubules of mice in which spermatogenesis is arrested by inhibition of retinaldehyde dehydrogenase and then synchronously restarted by injection of RA (Fig. 2). They find a marked rise in RA immediately after stage VII. Others consider the onset of RA production to occur at stage VII (Sugimoto et al., 2012; Hasegawa and Saga, 2012). Not only does RA control periodic spermatogonial division and germ cell differentiation, but its production by Sertoli cells likely regulates the blood-testis barrier in a stage-specific fashion and thus the access of not only Leydig cell products but also circulating factors that may modulate the spermatogenic cycle (Hasegawa and Saga, 2012). It remains to be determined whether species differences in the period of the spermatogenic cycle arise from differences in the timing of RA metabolism or the potency of particular feedback signals and whether the phase of the cycle can be reset in a phase-dependent manner by acute manipulation of RA.
The membrane receptor for retinal binding protein that controls the uptake of retinol from circulation is stimulated by retinoic acid 6 (Stra6; Kawaguchi et al., 2007). This is just one of several genes whose expression depends on RA and which likely participates in feedbacks that determine the period of the rhythm of RA concentrations. Controversy surrounding the particular steps of RA metabolism that oscillate and the enzymes that are regulated may reflect the multiple cell types and differences in procedures used to synchronize or arrest spermatogenesis. The synthesis of RA from retinol takes place by 2 oxidative steps, controlled by retinol dehydrogenases and retinaldehyde dehydrogenases acting sequentially. Elizondo et al. (2000) found that RA regulates transcription of retinaldehyde dehydrogenase. Retinaldehyde dehydrogenase 2 made in step 6 spermatids is believed to contribute heavily to intertubular RA synthesis; the RA thus formed acts in step 7 to 8 spermatids. Sugimoto et al. (2012; Fig. 3) propose that RA not only regulates expression in Sertoli cells of enzymes that control its own metabolism but also that postmeiotic germ cells control the availability of RA to both Sertoli cells and spermatogonial stem cells. Specifically, they posit that round spermatids favor storage and/or degradation of RA, while late pachytene spermatocytes increase RA production. Thus, the germ cells would limit the rise of RA to a restricted window by providing “wait” and “go” signals in order to coordinate the progression of the stages of spermatogenesis. Degradation of RA occurs under the control of cytochrome P450 enzymes, which are also suggested to participate in feedback regulation of RA. In contrast, Hogarth et al. (2015) found no stage-specific change in synthetic (RALDH) or degradative (Cyp26) activity, although retinol dehydrogenase (Rdh10) and dehydrogenase/reductase (Dhrs4) showed stage-specific expression. Although RA levels were closely matched to stimulated by retinoic acid 8 (Stra8) expression and nuclear translocation in spermatogonia and spermatocytes, the cellular source of RA was not identified.
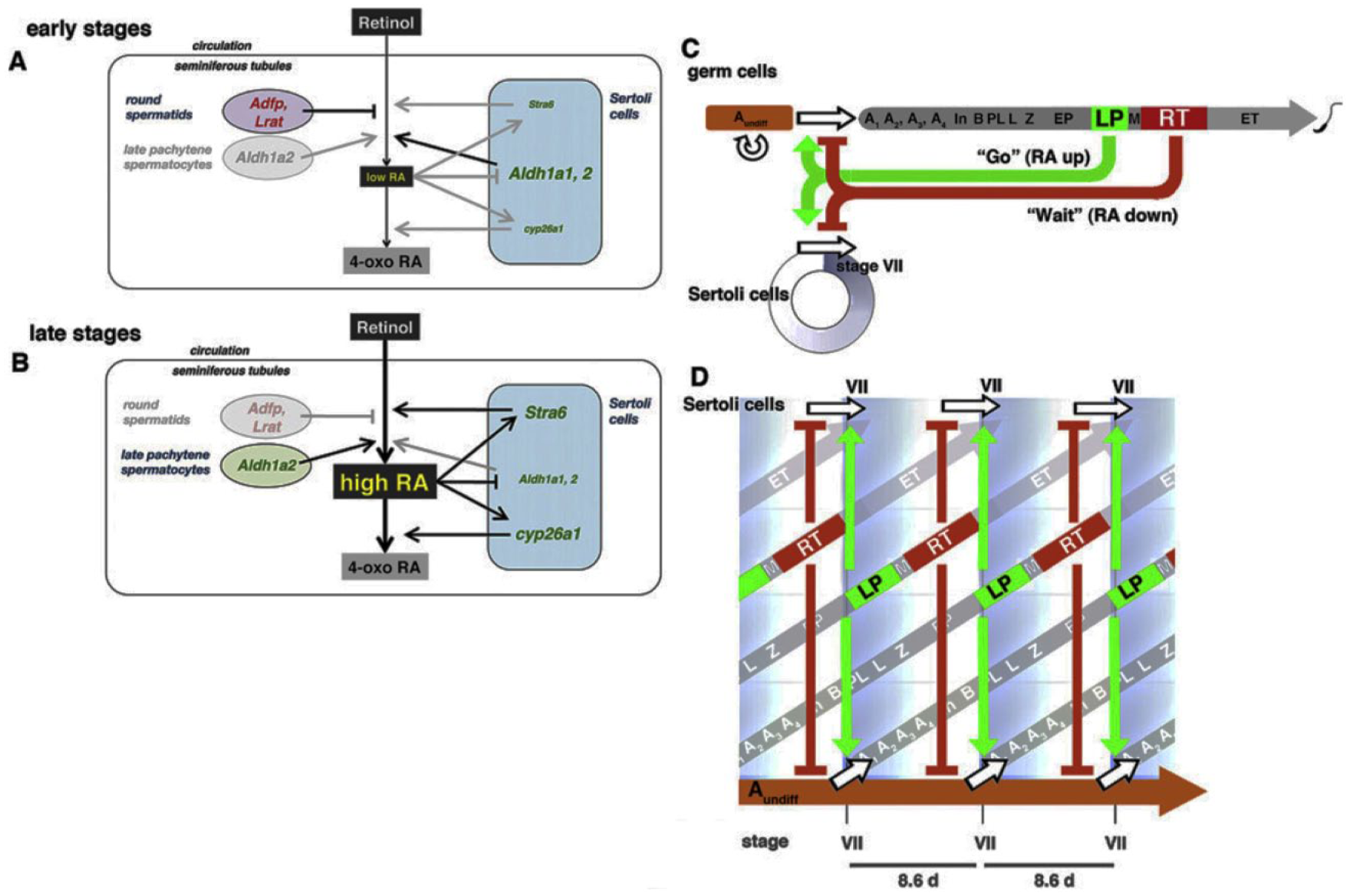
In contrast to the cell autonomous transcriptional-translational feedback loops that underlie circadian rhythms, the generation of oscillations of retinoic acid (RA) that control the spermatogenic wave relies on intercellular communication. This figure illustrates the mechanism proposed by Sugimoto et al. (2012; reprinted with permission). (A) Low levels of RA production from retinol in the seminiferous tubule as the round spermatids express adfp (adipose differentiation-related protein) and Lrat (lecithin:retinol acyltransferase) to promote storage of RA and thus antagonize effects of aldehyde dehydrogenases (aldh1a2) released by Sertoli cells. (B) In the absence of round spermatids during stages VII to XII, RA levels rise as Aldh1a2 production by late pachytene spermatocytes predominates and Stra6 from the Sertoli cells promotes the uptake of substrate. (C) Late pachytene spermatocytes and round spermatids are proposed to exert “go” and “wait” influences on spermatogonial differentiation and Sertoli cell cycle progression. (D) Germ cell differentiation and Sertoli cell progression are coordinately regulated by germ cell signals in order to control RA concentrations and thus the stage of the seminiferous epithelium.
The activation of transcription of specific genes by RA, and hence its cellular actions, is mediated by RAR and RXR nuclear receptors, each of which is subdivided into 3 subtypes. Thus, the periodicity of the seminiferous epithelium may also be regulated by factors that modulate expression, affinity, or availability of these receptors. RARa in Sertoli cells is necessary for the initiation of spermatogonial differentiation, and RA of Sertoli cell origin promotes spermiation. RA synthesized by spermatocytes acts via RAR to induce the onset of meiosis (Vernet et al., 2006; Raverdeau et al., 2012; Endo et al., 2015). Raverdeau et al. (2012) distinguish between actions of RA on spermatogonial commitment and on meiotic initiation. In the former process, RA produced by Sertoli cells acts through activation of RAR alone. In the latter, RA acts cell autonomously through RAR/RXR heterodimers. A prediction of this model is that manipulation of the relevant enzymes will alter the period or reset the phase of the spermatogenic cycle. A suitable strategy might involve use of inducible promoters, much as was employed in experiments to identify core circadian clock components (Aronson et al., 1994).
The identity and roles of genes whose transcription depends on RAR and RXR and whose products mediate critical events at stages VII to VIII of the spermatogenic cycle remain to be fully determined. The protein products of Stra8 and Mafb appear to control critical events in spermatogonial differentiation and likely participate in feedbacks that determine the rhythmicity of RA production. Interestingly, mice deficient in Stra8 experience an overproduction of presumptive spermatogonia (Endo et al., 2015). Although it is not known whether the period of their cell cycle is affected, this suggests that divisions of the stem cells through the type A spermatogonia may be independent of RA. STRA-8 likely mediates both the RA-induced spermatogonial differentiation and the meiotic initiation that occurs 1 cycle later (8.6 days in the mouse) at stages VII to VIII.
The ability of RA to promote both spermatogonial differentiation and the initiation of meiosis may depend on fluctuating competency of the target cells, such that a temporal coincidence of the signal and the responsiveness contribute to the coordination of the spermatogenic wave. RA can accelerate spermatogonial differentiation across a range of stages (II-VIII), but its ability to advance meiotic initiation is more limited (to stages VI-VIII; Endo et al., 2015). Administration of RA at intervals of 8.6 days can drive a normal spermatogenic cycle (i.e., staging of the seminiferous epithelium and meiotic progression resemble those of vitamin A–replete mice) (Endo et al., 2015). As there is no evidence for an independent oscillation in the germ cells, one hesitates to refer to this as entrainment; the 8.6-day interval may represent the time lag to achieve competencies at successive stages of the seminiferous cycle. An alternative possibility is that a rhythm within the target cells periodically opens a window of competence to respond to RA. It would be of interest to vary the period of RA administration to examine whether the phase of spermatogonial differentiation or entry into meiosis varies relative to the injection.
Interestingly, vitamin A is required for several events that occur in the germ cells of the different stages that mark the stage VII assemblies. The presence of RXRa in nuclei of step 7 and 8 round spermatids (Vernet et al., 2006) suggests that RA provides paracrine modulation and synchronization between meiotic commitment and postmeiotic maturation of the germ cells. Thus, local timed peaks in RA concentration may serve to coordinate the spermatogenic wave not only at the points of spermatogonial division and entry into meiosis but also for the ultimate release of mature sperm into the tubular lumen at spermiation. In this way, local production of RA at stage VII would set period as well as ensure the coherent progression of the seminiferous epithelium through cellular assemblies.
Endocrine Rhythms of the Testis
Pulsatility of Androgen Secretion
The regular episodic secretion of steroid hormones represents another rhythmic function of the testis (Sanford et al., 1974; Steiner et al., 1980; Winters and Troen, 1986; Price et al., 2000; Schlatt et al., 2008; Schuler et al., 2014). Sampling of the gonadal vein reveals oscillations of serum androgens with a period of approximately 1 h, and pulsatility is evident in jugular samples collected in rams and rhesus monkeys despite dilution of testosterone in the systemic circulation (Fig. 4). Peaks of T almost always occur shortly after episodes of luteinizing hormone (LH) secretion (Sanford et al., 1974; Schlatt et al., 2008). A pulse generator in the hypothalamus and preoptic area controls the release of GnRH into the hypothalamo-hypophyseal portal system, resulting in spikes of LH secretion that decay with the half-life of this glycoprotein. The basis of the oscillation lies in the neuroendocrine regulation rather than the testis itself. Thus, this is not an instance of entrainment of a gonadal oscillator: testicular androgen secretion can match the frequency of LH presentations over a wide range, and the phase of testosterone release is similar whether pulses of gonadotropin fall at 1- or 2-h intervals. The function of the Leydig cells is nevertheless attuned to the pulsatile delivery of LH, and this aspect of testicular rhythmicity is thus relevant to consideration of timing in the testis. As is the case for the gonadotropes, whose production of LH depends on the pulsatile pattern of the driving input of GnRH (Belchetz et al., 1978), the LH response of the testis is blunted when stimulation is delivered continuously (Veldhuis et al., 2012a, 2012b). Presumably, pulsatile delivery of LH prevents downregulation of its receptor and/or desensitization of the signal transduction cascade within the Leydig cells. The secretory patterns of FSH to which the testis is exposed are less notably pulsatile, in part because of the longer half-life of this glycoprotein hormone, due to a basal level of secretion not coincident with LH pulses (Padmanabhan et al., 1997) and also because of modulation of its secretion by protein hormones of the testis (inhibin and activin; see Makanji et al., 2014, for review). In addition, the existence of a separate FSH-releasing hormone, or control of FSH by a different GnRH variant, is suspected (Padmanabhan et al., 2003). Nevertheless, expression of the beta subunit of FSH is differentially regulated by the frequency of GnRH pulses (Ciccone et al., 2010).
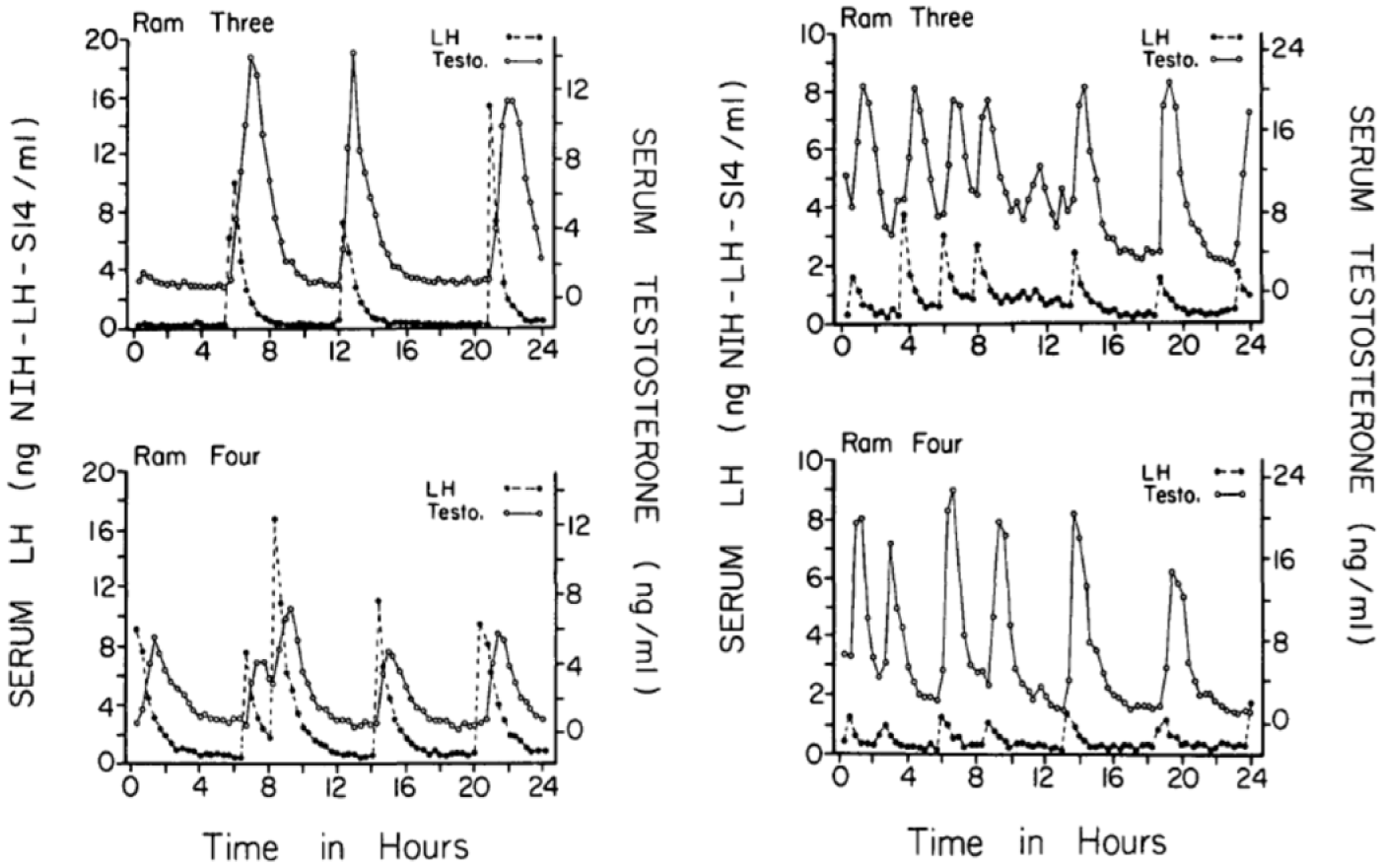
Pulsatile pattern of concentration of luteinizing hormone (LH) and testosterone in jugular blood collected from rams at 20-min intervals. The operation of a pulse generator in the hypothalamus and preoptic area results in an oscillatory release of the steroid hormone from the testis. Note that the frequency of LH and T pulses is about half as great during the nonbreeding season (August, left) as in the breeding season (January, right). Reprinted with permission of the Endocrine Society from Sanford et al. (1974).
Circadian Rhythms of Androgen Production
Daily rhythms of endocrine function have been widely reported in mammals (see Turek and van Cauter, 1994, for review), and the secretory output of the testis is no exception. Once- or twice-daily peaks of serum testosterone concentrations have been reported in laboratory mice and rats held on an LD cycle, although the question of whether they persist in constant conditions has received scant attention (Kinson and Liu, 1973; Mock et al., 1978; Kalra and Kalra, 1977; Keating and Tcholakian, 1979; Sayegh et al., 1990; Waite et al., 2009; Thorpe et al., 2012). Thus, the circadian basis of testicular secretory rhythms, let alone dependence on the central pacemaker, is not established. In monkeys, serum testosterone levels change across the course of the day, apparently reflecting a variation in the frequency of LH pulses (Goodman et al., 1974; Schlatt et al., 2008). The phase of peak serum T differs somewhat between species, and the amplitude of this diurnal rhythm changes with age (Schlatt et al., 2008; Tenover and Bremner, 1991; Cooke et al., 1993; Urbanski and Sorwell, 2012). Rhythms of binding globulins may contribute to oscillations in serum levels of androgen (Simoni et al., 1988; Plymate et al., 1989). Androgens influence glial and synaptic function in the ventromedial core of the mouse SCN and regulate the induction of Period gene expression and behavioral phase shifts in response to light pulses (Karatsoreos, 2011; Butler et al., 2012). Evidence that the episodic pattern of androgen secretion influences its neuroendocrine effects (Zwart et al., 1997; Rhim et al., 1993) suggests a potential for interaction between brain and gonadal oscillations that remains to be explored.
Circadian Oscillators in the Testis
Circadian rhythms are deeply ingrained in cell function. It remains uncertain whether eukaryotic rhythms evolved solely from the symbiotic incorporation of prokaryotic circadian mechanisms or whether Archaea also contributed critical elements along with their considerable innovations in RNA splicing and regulation of gene expression (Koonin, 2010). Regardless of the evolutionary origin of circadian rhythmicity in eukaryotic cells, it is clear that such oscillations predate the evolution of multicelluarity. In this context, perhaps the discovery that individual cells in peripheral organs display circadian rhythmicity should not have surprised us to the extent that it did (Balsalobre et al., 1998; Rosbash, 1998). Other reviews in this series make it clear that such rhythms are the norm rather than the exception. Thus, findings that an organ lacks the capacity for cell-autonomous rhythmicity might startle us. Nevertheless, attempts to find molecular oscillations in the testis have met with inconsistent results. Indeed, publications describing the incidence of circadian rhythms in peripheral organs typically qualify their distribution as “nearly” ubiquitous, with the testis being the principal exception.
Circadian Rhythms in Homogenates of the Testis
Reports of negative data notwithstanding, it is clear that circadian rhythms occur in the testis. With the exciting finding in 1998 that homologs of the Drosophila gene Period are expressed outside the SCN pacemaker (Balsalobre et al., 1998), efforts began to describe the tissue distribution of core clock gene expression, and the testis was among the organs studied. Zylka et al. (1998) used Northern blots to describe rhythms in the abundance of Per1 and Per3 transcripts in the mouse testis, with peak expression at circadian time (CT) 6 and CT9 to CT15, respectively. Little Per2 messenger RNA (mRNA) was detected. Bioluminescence of testis slices from Per2::luc mice revealed circadian rhythmicity ex vivo (Fig. 5A), and similar recordings were made in testis transgenic rats expressing a destabilized luciferase under control of a Per2 promoter (Nishide et al., 2013). Such recordings leave open the question of which cell type(s) may be responsible for the rhythm.
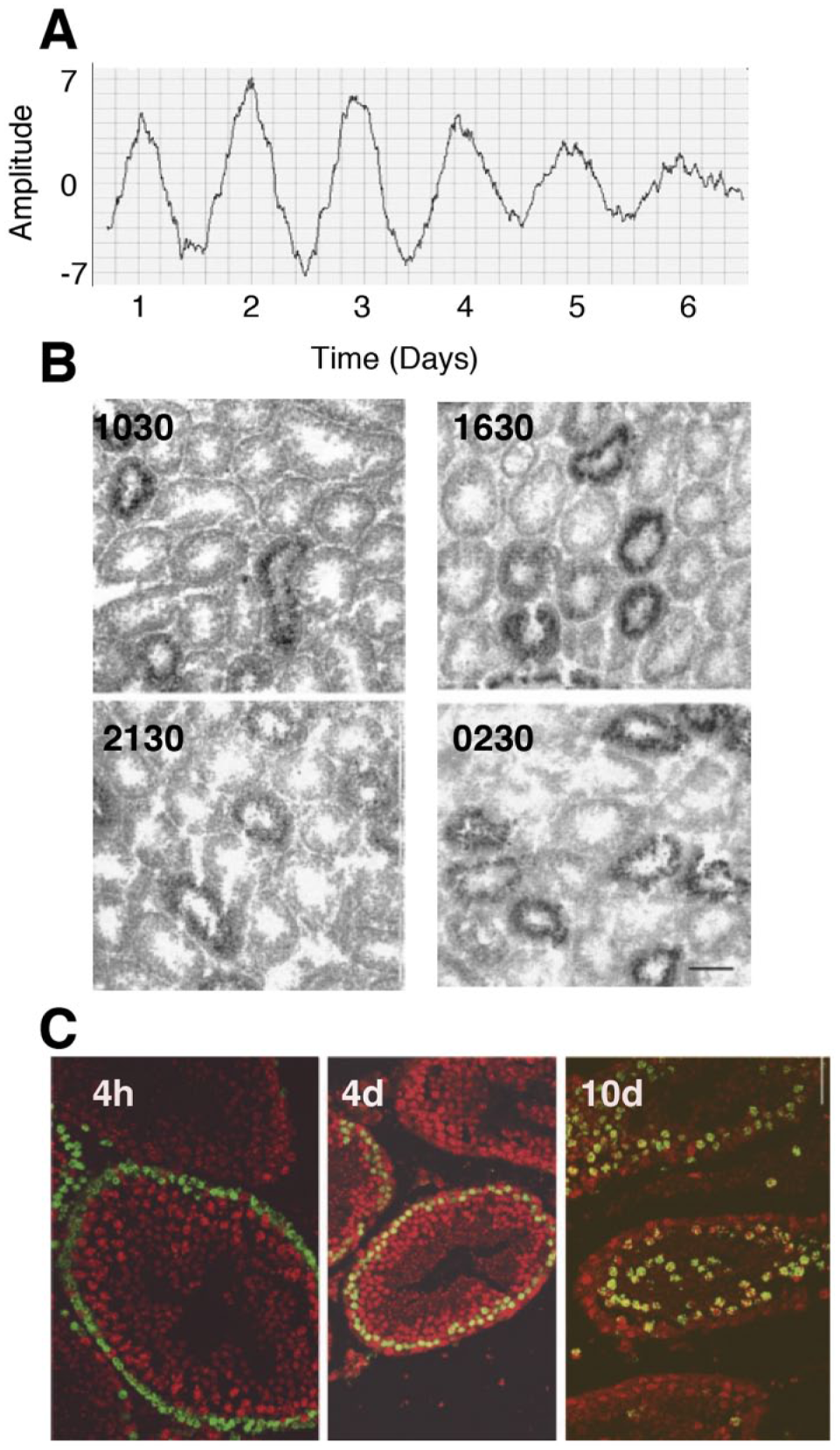
(A) Circadian pattern of expression of the core clock gene Period 2, as reflected by a luminescent reporter, in testis tissue collected from a Per2::luc knockin mouse and incubated for 6.5 days (Bittman, unpublished data). (B). In situ hybridization for Per1 messenger RNA (mRNA) in mouse seminiferous tubule. Note that some segments contain this transcript and others do not at any particular time of day. A distributional (Kolmogorov-Smirnov) analysis indicates that Per1 expression reaches a nadir at circadian time 3 (reprinted from Bittman et al., 2003; with permission of the American Physiological Society). (C). PERir (red) is colocalized with BrdU (green) in leptotene spermatocytes >5 day after injection of this cell birth marker but not in spermatogonia at shorter survival times. This is consistent with studies of embryonic stem cells that suggest that operation of transcriptional-translational feedback loops that generate circadian oscillations does not occur until differentiation. Reprinted from Bittman et al. (2003) with permission of the American Physiological Society.
Nevertheless, quantification of core clock gene transcripts or their protein products in homogenates of testes collected from animals at different times of day has not always provided evidence of circadian rhythmicity. RNase protection assays failed to indicate a rhythm of Per1, Per2, Cry1, Cry2, Bmal1, Clock, or Rev-erb a in mouse testis (Alvarez et al., 2003; Morse et al., 2003). Using quantitative reverse transcriptase polymerase chain reaction (qRT-PCR), neither Alvarez and Sehgal (2005) nor Mazzoccoli et al. (2012) found a daily rhythm of clock gene expression in testicular homogenates of mice held in an LD cycle. In contrast, Bebas et al. (2009) used qRT-PCR to demonstrate a 2-fold rhythm of Per1, Bmal1, and Per2 mRNA, with a nadir late in the light phase, in testes of mice maintained in 12L:12D. Bebas et al. found no evidence for a rhythm of PER1 or BMAL1 in mouse testis, although peaks and nadirs may have been missed because only 4 time points were sampled. Rhythms of clock gene expression were found in accessory organs, including vas deferens, prostate, seminal vesicle, and epididymis, with the expected antiphase relationship. Bmal1 and Per mRNA abundance peaked more or less simultaneously in testis, rather than in antiphase as commonly reported in other organs. These results resemble earlier findings of Tong et al. (2004), who observed free-running rhythms of Per1 and Bmal1 mRNA in hamster testis with coincident peaks during subjective night. A single Clock and 2 distinct Per2 transcripts were also detected in hamster testis, but neither showed a significant variation with time of day. Klose et al. (2011) found a 3-fold excursion of Per1 mRNA in testes of Djungarian hamsters maintained in a light/dark cycle, with an extended peak between zeitgeber times 6 to 18. No fluctuation of Bmal1 transcript levels occurred in the testes of these animals. In a comparative study, Meyer and Lerchl (2014) found significant day-night variation in Bmal1, Per2, and Cry2 transcripts in testes of dwarf and Chinese hamsters maintained on a 16L:8D photoperiod. Per1 was also found to show a significant diurnal variation in Phodopus roborovskii and Cricetulus griseus. As in many other studies, where rhythms of clock transcripts were found, they were of lower amplitude in testis than in other organs of the same hamsters.
What explains the inconsistency of reports that circadian clock gene expression oscillates in rodent testis? Not only do the functions of the interstitial and tubular compartments differ, but the spermatogenic wave and the presence of cell assemblies constituting differentiating cells in various stages of maturation along the length of the tubule open the possibility that cyclic events occurring at various segments are at different phases. The distribution of transcripts may also be uneven between the basal and luminal regions of the tubule due to condensation of the spermatids. At the completion of meiosis, the germ cells elongate. Like space travelers who must jettison weight in order to achieve escape velocity for a journey into orbit (Weir, 2014), they condense both cytoplasm and chromatin. In this process, DNA becomes wrapped in a condensed, toroidal structure around protamines, and heterochromatin is found only in a few regions (largely near the centromeres and telomeres) that preserve histones (Boskovic and Torres-Padilla, 2013). The drastic reduction of gene expression likely includes a suspension of clock gene transcription. This process of DNA condensation probably contributes to a reduction in the amplitude of rhythms of clock gene transcripts when the testis is sampled as a whole.
As is the case for the brain, heterogeneity may lead to inconsistent or ambiguous answers to the question of whether a circadian clock runs in the testis. Different cell types may vary in the amplitude and/or phase of their oscillations. Just as we would not homogenize the entire brain if we sought to understand its rhythmic function, we should hesitate to grind up the testis in order to study circadian rhythms of its cell types and processes. Furthermore, although circadian rhythms arise from cell-autonomous transcriptional-translational feedback loops, coupling of intracellular oscillators is required in order for phase coherence to occur (Liu et al., 2007a, 2007b). In the SCN pacemaker, this is achieved synaptically. Extra-SCN (including peripheral) oscillators ultimately rely upon the coupling of SCN pacemaker cells that is necessary for generation of a combination of autonomic, hormonal, thermal, nutritional, behavioral, and other signals without which the cellular oscillators will lose synchrony. Coupling of testicular circadian oscillations may occur only locally, at least within the tubule. It may also be relevant that the blood-testis barrier limits diffusion of cues, potentially including blood-borne zeitgebers, so that the seminiferous tubules may constitute a compartment that is buffered from, if not completely deprived of coupling and entraining signals.
Histological Approaches Reveal Circadian Function in the Testis
Spatial and temporal patterning of clock gene expression is better indicated by methods that preserve tissue organization. In situ hybridization reveals a striking pattern of Per1 expression in BALB and C57BL6 mice, with high levels of transcript evident in some tubular cross sections but not others at each time of day examined (Bittman et al., 2003; Morse et al., 2003; Fig. 5B). Not only molecular but also statistical methods for investigation of circadian function in the testis must take into account the compartmentalization of function. Analysis of variance, which is most often used to evaluate the total mRNA extracted from whole testis, may not be optimal. Bittman et al. (2003) used the Kolmogorov-Smirnov test to perform a distributional analysis of grain densities. This approach indicated a statistically significant peak of Per1 mRNA at ZT15. In situ hybridization for Clock mRNA documented its presence in spermatogonia and basal regions of the testis. This spatial segregation of Bmal1 in adluminal and Clock in the basal regions led Morse et al. (2003) to suggest that transcriptional regulation of Per1 expression differs between the testis and other peripheral organs. These authors supported their conjecture of alternative regulation of Per1 expression in testis with evidence of control by the transcriptional regulator CREM.
Similarly, immunocytochemistry reveals temporal cell-type specific patterns of clock proteins within and between compartments of the testis. Consistent with evidence for daily rhythmicity of androgen secretion (see above), Alvarez et al. (2008) reported that BMAL-1ir peaks in Leydig cells late in the dark and early in the light phase in testis of mice kept in 12L:12D. Within the seminiferous tubules, immunocytochemical studies have localized PER1-ir to spermatocytes and elongate spermatids, particularly between stages VII and XII, but without evidence of a daily rhythm (Bittman et al., 2003; Morse et al., 2003). PER1-ir was not found until at least 5 days, and more abundantly at 10 to 21 days, after BrdU injection, indicating clock gene expression is low before the leptotene phase of spermatogenesis (Fig. 5C). Similarly, Alvarez et al. (2008) observed PER1-ir in condensing spermatids at all times of day, particularly at stages V to VII and IX to XI. CLOCK-ir was detected in the acrosome of spermatozoa (Alvarez et al., 2008; Perruquetti et al., 2012). Alvarez et al. also found evidence of PER1 in type B spermatogonia. Although immunocytochemical studies of testicular rhythmicity may appear to be discrepant with results of in situ hybridization, the stability and distribution of the protein product are not necessarily predicted by those of the transcript.
Alternative splicing of clock transcripts may lead to expression of variants that have diverse roles and varying degrees of rhythmicity in the testis. Tong et al. (2004) found 2 major Per1 transcripts in Syrian hamster testis that lack both the consensus nuclear localization sequence and the C-terminal CRY1 binding domain. Thus, proteins long associated with circadian function may regulate spermatogenesis through interaction with a variety of molecules that do not regulate transcriptional oscillations and are unlikely to dimerize with CRY. Recent evidence suggests that core clock proteins may regulate rhythmicity of protein abundance in peripheral oscillators through an influence on ribosomal function (Mauvoisin et al., 2014; Lipton et al., 2015). A role in the mouse testis for core clock proteins other than their widely studied function in transcriptional regulation was suggested by Perruquetti et al. (2012), who reported the presence of CLOCK and BMAL1 in the chromatid body, a cytoplasmic granule in pachytene spermatocytes and spermatids that likely modulates translation. de Mateo and Sassone-Corsi (2014) suggest that noncoding RNAs (miRNA and piRNA) act in cytoplasmic germ granules to silence expression of particular transcripts in order to regulate spermatogenesis. These workers found that the morphology of the chromatid body was altered in Bmal1-deficient mice. CLOCK colocalized with MVH, a member of the family of DEAD-box RNA helicases, and BMAL1 with the eukaryotic translation initiation factor EIF4E, which may have a more general role in circadian timekeeping (Cao et al., 2015). CLOCK and BMAL1 are likely to interact, as Clock–/– animals lacked BMAL1 in the chromatid body, and both CLOCK and BMAL1 coprecipitate with MVH.
Consideration of the impact of differentiation on circadian rhythmicity may provide insight into the limits of clock function in the mammalian seminiferous tubule. Pluripotent embryonic stem cells do not express circadian rhythms: only at differentiation do epigenetic changes alter transcriptional programs to allow the onset of rhythmicity (Yagita et al., 2010). Critical to this transition may be a shift in patterns of importin expression to allow nuclear localization of PER1 and PER2 so that transcriptional-translational feedback loops can function (Uemura et al., 2014). In female mice, clock gene expression is arrhythmic in oogonia and primary oocytes, and this condition may persist even after fertilization. Amano et al. (2009) found that CRY is present in these cells but suggested that it plays a noncircadian function to arrest expression of clock-controlled genes that participate in the cell cycle. This situation may be similar in the seminiferous tubule, as undifferentiated spermatogonia may conform to this pattern: PER-ir is found in BrdU-labeled cells only 5 days after injection of this marker of cell division, after the commitment to differentiate (Bittman et al., 2003).
Circadian rhythmicity of the steroidogenic compartment may likewise follow a similar pattern in both sexes. The mammalian testis exhibits a division of labor that resembles that of the ovary: under the control of LH, an androgenic population supplies the substrates that cross into a separate, avascular compartment in which aromatization takes place under the control of FSH. This 2-cell model, featuring distinct and complementary roles of the 2 gonadotropins, is essential to coordination of gametogenesis. As reviewed elsewhere in this series (Sellix, 2015), granulosa cells cultured from mouse or rat ovaries of transgenic reporter strains show many cycles of clock gene expression. Circadian oscillations in the corpus luteum, which contains a mixed population of thecal and granulosa cells, has been reported to be stronger than that of the follicle (Chen et al., 2013), although rhythmicity of the ovarian interstitium remains to be studied. As in the ovary, circadian rhythmicity of the interstitial cells of the testis may differ in amplitude from that of the Sertoli cells as well as the gametes. A major difference between the sexes is the relative proportions of the populations of steroidogenic and supporting cells relative to the gametes. In the former, circadian function is readily detectable, while in the latter it may be minimal or absent, at least before differentiation. This may contribute to a sex difference in the amplitude of clock gene expression in the gonads of males versus females.
Among the invertebrates, the question of circadian regulation of testicular function has been addressed in a few insects. In Drosophila, the arrangement of germ line stem cells and cyst progenitor cells that give rise to gonialblast and somatic cells allowed Tulina et al. (2014) to assess daily rhythmicity in both cell types. Immunohistochemical markers suggest that the frequency of mitosis shows a low-amplitude variation with time of day in flies maintained on an LD cycle, although this is not evident using BrDU incorporation as a measure of DNA replication. Furthermore, no rhythms of mitotic divisions were detected in the testes of flies maintained in constant conditions. Despite the fact that mutations that affect the duration of sleep can alter testicular mitotic rate, these data provide no evidence for the operation of a circadian clock in the spermatogenic niche of Drosophila. This may be the general pattern in Drosophila gonads, as efforts to detect clock gene expression using probes for period or timeless failed to support circadian function in the ovary (Beaver et al., 2003).
Relationships between Spermatogenic and Circadian Rhythms
The RA oscillation that coordinates the spermatogenic wave differs from the circadian transcriptional-translational feedback loop in that it is not cell autonomous but requires the integration of synthetic and degradative enzymatic steps that occur in different cell types. The distinction may not be as great as it seems: even in the SCN, considerable heterogeneity is apparent with clock gene expression in the core being light regulated and nonoscillatory in constant conditions, while the shell is capable of coherent free-running oscillations (Hamada et al., 2004). Likewise, in both peripheral circadian and testicular oscillators, cell types differ in the control and generation of the relevant molecular events. In the lung, for example, circadian oscillations are prominent in the Clara cells but less so in other cell types (Gibbs et al., 2014), and entraining signals perturb clock phase through an initial impact on Rev-erb a (Meng et al., 2008). Thus, the situation in the testis, in which the strongest circadian oscillations may occur in the interstitium and cells cooperate to generate a rhythmic RA signal, may not differ in principle from other chronobiological systems.
The dramatic, infradian oscillations of RA that drive the spermatogenic cycle may interact with—and perhaps reset the phase or interfere with the expression of—the transcriptional-translational feedback loops that underlie circadian oscillations. Closely related to the RARs and RXRs through which RA acts to regulate the spermatogenic cycle are retinoic orphan receptors. Circadian rhythms are regulated by these receptors: the transcription of the core clock genes Bmal1 and NPas2 is acutely induced by retinoic orphan receptors and repressed by REV-ERB a and b. RORs compete with REV-ERB a for retinoid response elements in the promoter region of Bmal1. The transcription of these receptors is in turn controlled by PER:CRY heterodimers, completing a feedback loop that contributes to the circadian oscillation. Several isoforms of RORs exist, and their tissue distribution varies. In the testis, RORa is expressed in peritubular cells (Steinmayr et al., 1998). Male mice deficient in RORa, like staggerer mutants that lack RORa, appear to be fertile. While this suggests RORs are unnecessary for spermatogenesis, it is possible that RORg, which is also expressed in testis (Hirose et al., 1994), may support male reproductive function in its absence.
Considerable effort has been devoted to discovery of the endogenous ligands of these receptors. In the case of REV-ERB a, heme is the principal ligand; in the absence of heme binding, REV-ERBs are transcriptionally inactive. Like all cells in the body, testicular cells have some intracellular heme, so that REV-ERBs likely act as constitutive repressors. The endogenous ligands of RORs are unknown; RA is believed to have little affinity for the ligand binding domain, and while 7-hydroxylated cholesterol derivatives may bind RORs, these molecules are more likely relevant to metabolic functions than to male reproduction.
The responsiveness of circadian mechanisms to RA may differ between the brain and the periphery. Navigatore et al. (2013) claimed that vitamin A deficiency shifts phase or blunts rhythmicity of BMAL1 and PER1 in rat hippocampus. In contrast, Shirai et al. (2006a) presented evidence against RA control of clock gene expression in the periphery. They reported that long-term deficiency of dietary vitamin A sufficient to cause a 6-fold reduction in serum retinol levels had no effect on patterns of expression of mPer1, mPer2, Clock, Bmal1, or dbp in the livers of mice maintained on a 12L:12D cycle. Although neither the expression of clock genes in the testis nor the arrest of the spermatogenic cycle was examined in the same mice, this observation suggests a divergence between the spermatogenic and circadian cycles in dependence on RA.
McNamara et al. (2001) found that RAR and RXR can interact with CLOCK protein to inhibit dimerization with BMAL1, thus interfering with transactivation of both core clock genes and clock-controlled genes whose promoters contain E-boxes. RXR-deficient rats show reduced hepatic expression of Bmal1 and Rev-erb a and experience an altered pattern of Cry2 expression (Yang et al., 2010). Shirai et al. (2006b) found that RA modestly transactivates mPer1 promoter-luciferase reporter in NIH 3T3 cells. This effect depended on integrity of E-box elements in the Per promoter. This may reflect a direct action of RA at E-boxes as cotransfection of the cells with RARa converted the action of RA from inductive to inhibitory. Furthermore, RA can markedly amplify the inductive effects of CLOCK:BMAL1 on mPer1, mPer2 and mPPARa expression. RXR may also regulate clock gene expression less directly (e.g., through interaction with peroxisome proliferator–activated receptors that act at the Bmal1 promoter). It is possible that RA effects are indirect—for example, through an influence on the availability of cofactors (e.g., CREB-binding protein/p300) that are shared by several nuclear receptors. Navigatore et al. (2013) argue for a reciprocal regulation of retinoic acid receptors and clock genes, in which RARa, RARb, and RXRb promoters have E-box–like elements, and Per1 and Bmal1 promoters contain retinoid acid and retinoid X response elements.
Functional Significance of Circadian Rhythms in Male Fertility
In contrast to the severe effect of circadian disruption on female reproduction, SCN-lesioned hamsters remain able to sire litters (Eskes, 1984). If anything, fertility of male hamsters is enhanced by SCN lesions in the sense that short days fail to induce testicular regression (Morin and Rusak, 1976). Klose et al. (2011) found that the period of the seminiferous epithelium of Siberian hamsters was not altered by entrainment to light:dark cycles whose period (T) was shorter or longer than 24 h. This suggests that the spermatogenic wave is not based on a circadian oscillator that is entrained by photoperiod. On the other hand, deletion of core clock elements has effects on male fertility that differ between genes and between laboratories.
Evidence has emerged for deficits in male fertility in circadian knockout mice, and explanations have generally been sought at the level of endocrine physiology. Reproduction is compromised (although not completely eliminated) in Bmal1-deficient animals, which lack behavioral rhythmicity and have a shortened life span. Although reproductive failure is most clearly described in Bmal1–/– females, part of the deficit is attributable to the male (Bunger et al., 2000; Boden et al., 2013). Alvarez et al. (2008) described a 70% reduction in sperm count, along with a reduction in tubule diameter and testis weight, but observed that sperm of Bmal1 knockouts were capable of fertilization. Per1/Per2 double knockout and Cry1/Cry2 double knockout mice fail to breed, although as for Bmal1–/– animals, the relative impact of these deletions on males versus females has not been completely described. It is not entirely clear whether reductions of male reproductive function in mice deficient in particular core clock genes are attributable to compromise of function in any particular cell type. Levels of steroid acute regulatory protein (STaR) were reduced in testis, as in ovary and adrenal, of Bmal1-deficient mice. Other steroidogenic enzymes, as well as gonadotropin levels and LH receptor expression, are also altered in Bmal1-deficient animals, but these changes could be secondary to a role of Bmal1 in control of STaR expression. In this respect, infertility in male Bmal1-deficient mice may resemble that in female knockouts, in which suppression of gonadal steroidogenesis leads not to a failure to ovulate but to prevention of implantation (Liu et al., 2014). Given that androgens must act on Sertoli cells in synergy with FSH to prevent apoptosis of the germ cells, compromise of StAR may be sufficient to account for reduced fertility. Other cell types, including peritubular myoid cells, and other enzymatic targets, including DICER, appear to be necessary for spermatogenesis and may be altered upon disruption of core clock loops (O’Shaughnessy, 2014).
ClockΔ19 mutants show a mild decrease in fertility, as evidenced by a reduced size of litters sired, but this is not attributable to a decrease in sperm count or fertilizing capacity in vitro (Doltashad et al., 2006). Liang et al. (2013) achieved a transient knockdown of Clock by intratesticular injection of small hairpin RNA (shRNA). They observed a substantial reduction of litter size and a lower in vitro fertilization rate, as well as a drop in acrosin activity that was not accompanied by a reduction in testicular weight, T level, or sperm production. Interestingly, FSH receptor expression in testis is critically regulated by E-box elements (Rannikko et al., 1996; Heckert and Griswold, 1991; Kliesch et al., 1992; van Dissel-Emiliani et al., 1989; Xing and Sairam, 2001). ChIP experiments have not yet been attempted to determine whether BMAL1:CLOCK heterodimers occupy this element in the FSHr promoter.
Even positive results of studies of knockouts and clock mutants must be interpreted cautiously, since genetic manipulations that compromise circadian function may have deleterious effects on immunity, metabolism, longevity, and health. Thus, deficits in fertility may be secondary, although reproduction is less sensitive to deprivation of energetic needs in males than in females (Hamilton and Bronson, 1985). Often overlooked in studies of effects of circadian derangement on fertility is the possibility that aspects of behavioral rhythmicity may contribute to decreases in fecundity. Diurnal variations of male sexual behavior occur in several species and have a circadian basis (Beach and Levinson, 1949; Harlan et al., 1980; Eskes, 1984). At a minimum, mismatches between the timing of arousal in members of the opposite sex may interfere with reproduction simply because males do not encounter females at the phase of their peak receptivity, proceptivity, or attractiveness (Dewsbury, 1968; Richter, 1970; Harlan et al., 1980; Paul et al., 2014). Targeting of genetic deletions to Sertoli, Leydig, or germ cells would be helpful to assess specific roles of the circadian clock in male fertility.
Seasonal Rhythms in the Testis
Arguably, the most striking of all testicular rhythms is the seasonal change in fertility characteristic of most animals of the temperate zone. Beyond their pivotal importance as an adaptation to the fluctuating environment, changes in the testis that occur with the approach of the breeding season may be useful to provide insight into puberty and to understand spermatogonial stem cell renewal after chemotherapy (Mitchell et al., 2009). Both steroidogenic and gametogeneic functions show profound seasonal rhythms in many mammals. In rodents, the seminiferous tubules undergo a marked change in diameter. While a 10-fold reduction in testis weight during the winter is not unusual (Sinha Hikim et al., 1988), regression to as little as 3% of the breeding season weight can occur (Takha et al., 1983). These changes are associated with seasonal fluctuations in angiogenesis in some species (Zayed et al., 1995; Pyter et al., 2005). In mustelids, chiroptera, and some birds, testicular changes are still more extreme: only spermatogonia and Sertoli cells are found within the seminiferous tubules of the seasonally regressed testis. In contrast, seasonal reduction in spermatogenesis in ungulates occurs without changes in the total number of spermatogonia, although tubular diameter and progression of germ cells to com-pletion of meiosis decline (Klonisch et al., 2006; Hochereau-de Reviers, 1992).
Selection strongly favors seasonality. In females, lactation is the most energetically demanding phase of the reproductive cycle and is thus best scheduled to occur in spring and summer. In males, hibernation is incompatible with high serum androgens (Darrow et al., 1988). Thus, seasonal regression of the testis may have adaptive value in males beyond reducing energy expenditure and dangerous exposure to aggressive encounters. Species with long gestation periods (such as sheep and goats, in which pregnancy lasts for 5-6 months) typically achieve greatest reproductive competence in the autumn and winter. In contrast, the short gestation length of rodents is consistent with breeding during the spring and summer. In wild populations, a fraction of “nonresponsive” animals—individuals that remain in breeding condition during the season at which most conspecifics experience reproductive arrest—is documented (Puchalski and Lynch, 1991; Prendergast et al., 2001). This may represent an instance of frequency-dependent selection, in which food resources available during the nonbreeding season can support a fraction of the population, and these individuals will experience disproportional success in years in which clement spring conditions arrive early.
Distribution of Seasonal Testicular Rhythms and Photoperiodism
Although laboratory mice and rats have served admirably to reveal mechanisms of shorter term endocrine regulation and gametogenesis, these species often exhibit little seasonal variation in testis function. Feral rats living in the garbage dumps remain in breeding condition through all but the coldest winters in Nome, Alaska (64°30’N; Davis, 1953; Schiller, 1956). Although nutritional state and energy expenditure are important determinants of testicular function (Bronson, 1989), artificial selection has likely operated to ensure reproduction throughout the year. Thus, responsiveness to cues that ensure restriction of male reproductive function to appropriate times of the year in wild populations is reduced in domesticated and commensal species. In contrast, field mice, voles, and hamsters, like non-domesticated Soay rams (Fig. 6), show dramatic annual arrest of spermatogenesis when exposed to seasonal variations in the environment. While fluctuations in temperature and the availability of food and water unquestionably contribute to seasonal variation in male fertility, more predictable changes in daylength (photoperiod) provide a particularly potent signal to which many species respond (Fig. 6). Indeed, in the classic study that introduced the concept of photoperiodism in vertebrates, Rowan (1925) induced testicular growth in starlings by exposing them to light during the night even though they were maintained in an outdoor aviary on the Alberta prairie in which nighttime temperatures plunged to −34 °C.
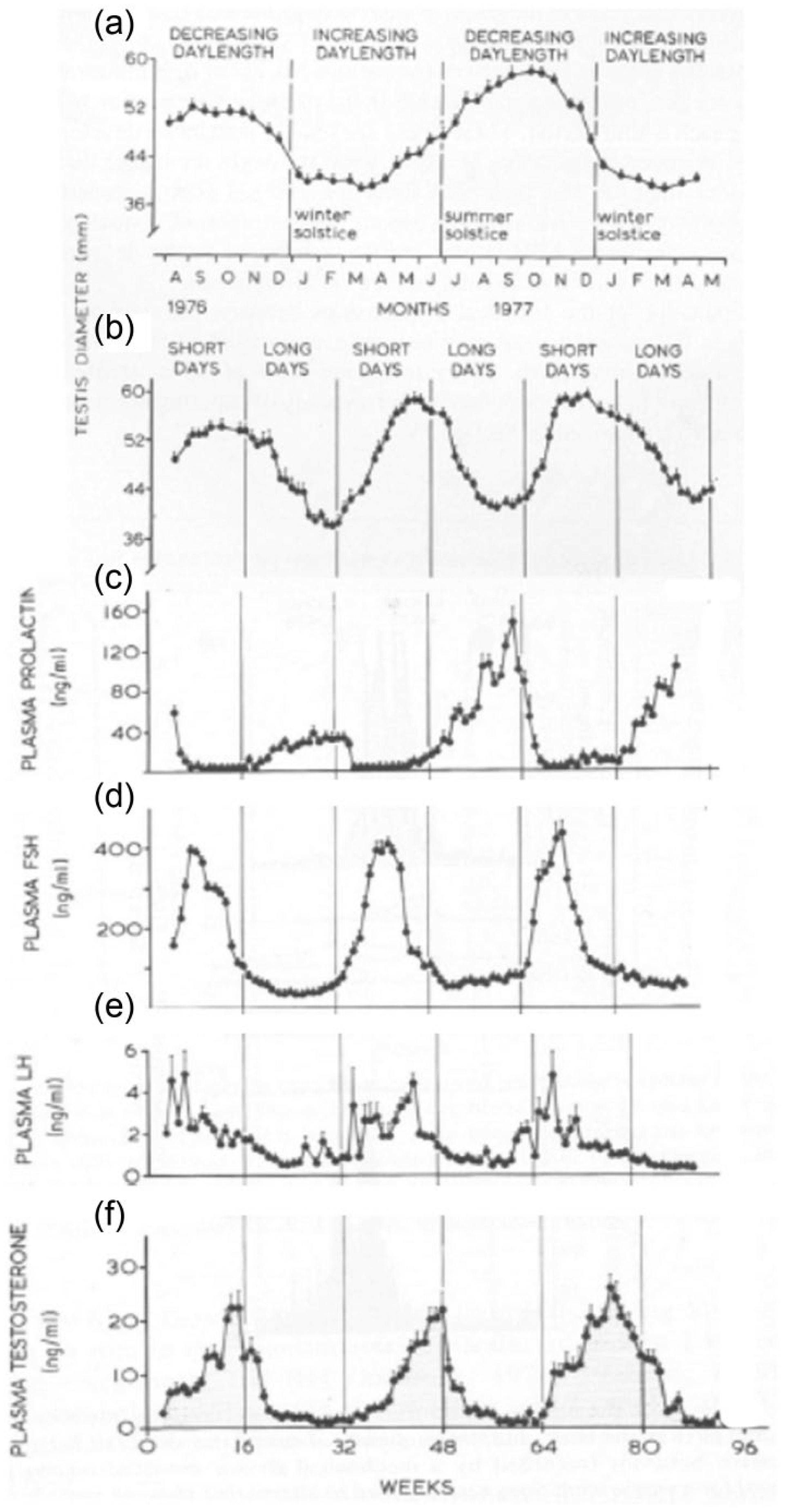
Seasonal reproduction in the Soay ram. (a) The diameter of the testis reaches a peak during the autumn and a nadir in the spring in sheep exposed to a natural photoperiod in Scotland. (b) When animals are subjected to alternating 16-week periods of short and long days (8L:16D and 16L:8D, respectively), the cycle of testicular diameters was accelerated to match that of the plasma concentrations of pituitary hormones (prolactin, follicle stimulating hormone [FSH], and luteinizing hormone [LH]) and of testosterone in the rams exposed to the artificial photoperiods are shown in (c), (d), (e) and (f), respectively. Reprinted with permission from Lincoln and Short (1980).
Siberian and Syrian hamsters have often been used in laboratory studies of effects of photoperiod on testicular regression and regrowth. Exposure of these rodents to winter daylengths triggers testicular regression over the course of several weeks. This process may be reversed by return of animals to photoperiods of greater than 12.5 h (Reiter, 1975; Elliott, 1976; Stetson et al., 1975; Bittman, 1978). If short days are maintained, however, the testes “spontaneously” recrudesce. This reflects a state of refractoriness to short days that ensures that hibernating animals return to reproductive condition before the spring equinox. Thus, animals regain reproductive capacity even when daylengths remain as short as those that initially triggered gonadal regression. Refractoriness to winter photoperiods can be broken by exposure to many weeks of long days, so that a second regression can be precipitated the next fall as daylengths shorten again. It should be pointed out that few myomorph rodents are likely to survive in the wild to experience a second testicular regression; as pointed out by Bronson (1989), predation reduces the life expectancy of voles to the extent that they are regarded as “the potato chip of the prairie.” In contrast, some long-lived species experience multiple breeding seasons. In some ruminants, woodchucks, and European hamsters, not only fertility but also body weight, brown fat mass, and pelage vary on a circannual basis even when photoperiod is held constant (Pengelley, 1974; Hiebert et al., 2000). This indicates that there is no phase of refractoriness to inhibitory photoperiods or that such refractoriness can be broken without the intervention of a photostimulatory regime. Such endogenous annual (i.e., circannual) rhythms are likely entrained by changes in photoperiod (Woodfill et al., 1994; Hiebert et al., 2000; Gwinner, 2003; Saenz de Miera et al., 2014).
Seasonal Changes in Neuroendocrine Regulation of the Testis
Not only the average serum concentrations of gonadotropins but also the frequency of LH pulses decline sharply as the testes regress (Sanford et al., 1974, 2002; Fig. 4). In both summer and winter breeders, short day-induced changes in gonadotropin production are accompanied by a drop in the secretion of prolactin (PRL). In hamsters, the withdrawal of PRL upon exposure to short days may facilitate gonadal regression, as this hormone increases responsiveness of Leydig cells to LH (Bartke et al., 1980; Klemcke et al., 1986; Hair et al., 2002). PRL receptor is expressed in both the Leydig cells and spermatocytes of rams (Jabbour and Lincoln, 1999), and PRL may also act elsewhere in the neuroendocrine axis and on the accessory organs to modulate seasonal changes in reproductive function. Although long day-induced increases in PRL secretion may blunt reproductive inhibition in rams by facilitating the stimulatory effect of LH on testosterone secretion, they also reduce the amplitude of GnRH-induced LH pulses (Sanford and Baker, 2010). Remarkably, long day-induced secretion of PRL is accompanied by increases in testis diameter even in hypothalamo-pituitary disconnected rams. This indicates a direct action of PRL on the testis, as plasma gonadotropins and testosterone drop to undetectable levels due to the inability of GnRH to reach the pars distalis (Lincoln et al., 1996). PRL may support low levels of androgen production even after LH secretion is terminated by ablation of the pituitary stalk (Jabbour and Lincoln, 1999; Sanford and Baker, 2010). PRL is also expressed in Leydig cells, spermatids, and spermatogonia of laboratory mice (Ishida et al., 2010; Maeda et al., 2012). It remains to be determined whether intratesticular expression of PRL occurs in seasonal breeders and, if so, whether its production fluctuates with time of year and has a role in annual rhythms of fertility.
In seasonally breeding mammals, photoperiodic regulation of reproduction depends on the secretion of melatonin by the pineal gland. It is well established that pineal-dependent action of short days occurs at the neuroendocrine level (Weems et al., 2015), and pineal-mediated effects of daylength upon gonadotropin secretion and steroid hormone feedback are sufficient to explain seasonal reproductive rhythms. The duration of the night is encoded by, and reflected in, the interval over which melatonin is synthesized. Melatonin reaches picomolar concentrations in the systemic circulation, and its targets certainly include pituicytes in the pars tuberalis of the pituitary complex. While circadian rhythms (and the brain’s central pacemaker) are essential to generation of the appropriate durational melatonin signal, the interpretation of this signal (the measurement of melatonin duration) likely depends on a noncircadian mechanism (Bartness et al., 1993). Although considerable progress has been made in determining how photoperiod alters LH pulse frequency, the steps by which the melatonin signal is transduced to altered GnRH release remain to be fully elucidated. The durational melatonin signal regulates expression of Eya2 in the pars tuberalis, leading to changes in monodeiodinase 2 and 3 expression to increase or decrease conversion of T3 to T4, respectively (Stevenson and Prendergast, 2013; Dardente et al., 2014; Saenz de Miera et al., 2013; Wood and Loudon, 2014). This may lead to structural changes in the neuroendocrine axis upon which seasonal rhythms of testicular function depend. Melatonin may also alter the timing—and, more specifically, the temporal coincidence—of Per and Cry expression in its target tissues; a model in which the duration of melatonin determines the opportunity for PER and CRY to dimerize in its target tissues may explain critical aspects of photoperiodic time measurement (Lincoln et al., 2002; West et al., 2013). Diencephalic neurons that express mel1a (also known as MT1) receptors are also likely to mediate some of melatonin’s effects. Melatonin binding sites have been described in the several preoptic and hypothalamic regions, and implantation in the various diencephalic sites may replicate effects of systemic administration (Bittman and Weaver, 1990; Malpaux et al., 1998; Teubner et al., 2008; Leitner and Bartness, 2010).
Clearly, timed administration of melatonin can mimic the neuroendocrine actions of photoperiod to alter patterns of gonadotropin secretion. These effects include not only changes in the ability of gonadal steroids to act as negative feedback regulators of LH but also changes in the frequency of LH pulses after gonadectomy. This does not exclude the possibility that melatonin also acts directly on the testis. Vera et al. (1997) and Valenti et al. (2001) reported specific binding of melatonin in rat testis with a Kd of about 100 to 200 pM, only slightly higher than the affinity of the receptor found in the pars tuberalis. RT-PCR has been used to amplify mel1a from total RNA extracted from the testes of Syrian hamsters (Frungieri et al., 2005). However, acute effects of melatonin on testosterone production in hCG-stimulated hamster Leydig cell preparations appear to be pharmacological, with no concentration-dependent effects in the physiological range (Valenti et al., 1995, 2001; Frungieri et al., 2005). In an examination of possible actions on bovine Sertoli cells, Yang et al. (2014) found that physiological concentrations of melatonin, administered continuously for up to 3 days, could regulate expression of cyclins E and D1, inhibin, and its own receptors (mt1 and mt2) in vitro. Since the duration of melatonin secretion appears critical to transduction of its neuroendocrine effects, it may be worthwhile to check for testicular actions by using appropriate durational signals at physiological concentrations. Given that testicular regression upon photoperiodic manipulation typically takes place over the course of weeks, however, it may be difficult to set up an experimental regime in which Leydig or Sertoli cell cultures are maintained with physiological gonadotropic support. This will be necessary to test effects of exposure to sub-nanomolar concentrations of melatonin of durations that occur upon exposure to inhibitory photoperiods.
Seasonal Changes in Testicular Function
Seasonal regression of the testis is characterized by Leydig cell degeneration, including reduction in the volume of most organelles (Sinha Hikim et al., 1988). The severe reduction of testosterone synthesis in short day hamsters is likely due to a combination of involution and necrotic cell death (Beltran-Frutos et al., 2014a, 2014b). Recovery of androgen production during testicular recrudescence occurs not through mitosis of differentiated Leydig cells but from recruitment of fibroblastic precursors (Hardy et al., 1987; Zayed et al., 1995). Hardy et al. (1992) found that the major androgen produced by the testis differs with season, with 5a-androstane-3a, 17b-diol constituting a larger proportion of the output of the testes of salamanders and hamsters when seasonally regressed and at the onset of recrudescence. In the roe deer, Leydig cell apoptosis is rare even during the anestrous season (Blottner and Schoen, 2005). In rams, testicular regression is accompanied by Leydig cell atrophy and decreased activity of StAR and cytochrome P450 and C17-20 lyase enzymes (Hochereau-de Reviers et al., 1985; Price et al., 2000), although the number of Leydig cells does not decline (Hocherau-de Reviers and Lincoln, 1985). Leydig cell cultures prepared from the seasonally regressed testes of white-footed mice are hyporesponsive to hCG and dibutyrl cAMP, and the production of endogenous opiates in these cells may participate in the arrest of steroidogenesis in short days (Knotts and Glass, 1988).
It is the decrease in the length and diameter of the seminiferous tubules, however, that accounts for the seasonal reduction in testicular weight. The observation by Ortavant (1958) that long day-induced testicular regression in rams is precipitated by a fall in gonadotropin secretion but not a change in the period of the spermatogenic cycle was perhaps the first evidence that hormones determine the survival, but not the division rate, of germ cells. Indeed, Soay rams experience a severe reduction in the diameter of the seminiferous tubules and the number of germ cells completing meiosis as necrotic cells and debris appear during the summer (Lincoln and Short, 1980). In hamsters, a precipitous drop in FSH secretion causes the reduction in Sertoli cell volume and nucleolar regression. In contrast to the interstitium, apoptosis is increased in the seminiferous tubules of photoinhibited hamsters (Fukuta et al., 1994) and deer mice (Young and Nelson, 2000; Young et al., 2001). Food deprivation may accelerate or enhance apoptosis in germ cells (Young et al., 2001). As testicular regression proceeds, spermatogonial proliferation (as evidenced by PCNA immunostaining) drops significantly. This change is followed by 10-fold or greater increases in the incidence of apoptotic cell death not only in Sertoli cells but also in round spermatids, spermatocytes, and spermatogonia (Tiba et al., 1992; Seco-Rovira et al., 2015). Sertoli cells engage in phagocytosis of degenerating germ cells (Seco-Rovira et al., 2014). Secretion of inhibin, predominantly a product of the Sertoli cells, roughly parallels changes in testicular volume and serum levels of FSH and testosterone (Jin et al., 2002; Kirby et al., 1993; Sanford et al., 2002). Expression of Smad3, believed to mediate actions of TGF-b family members including activins and inhibins, is markedly increased in spermatogonia and spermatocytes as short days induce testicular regression in Syrian hamsters (Kano et al., 2001). Conversely, SMAD2 and 3 proteins as well as ActRIIB (activin) receptor decline during the nonbreeding season in ground squirrels (Sheng et al., 2012). Testicular concentrations of polyamines increase during the course of regression as activity of ornithine decarboxylase rises (Frungieri et al., 2005). Ultrastructural analysis indicates a loss of adhesion between spermatogonia and separation of spermatocytes from adjacent cells, followed by nuclear condensation and degradation. Changes in rates of proliferation and apoptosis during the course of testicular regression and recrudescence need not be reciprocal or symmetrical and may reflect the fact that changes in the endocrine signals that impinge upon the testis in the spring and fall are not mirror images.
Photoperiod dramatically affects the expression of core circadian clock genes in the Syrian hamster testis. While testicular haPer1 expression is evident in mid- to late subjective night of hamsters acutely transferred to DD, this transcript becomes arrhythmic in the regressed gonads of hamsters exposed to many weeks of DD (Tong et al., 2004). Conversely, rhythms of Bmal1 expression are absent in hamsters acutely transferred to DD but emerge upon long-term exposure, which results in testicular regression. This may reflect the expression of Per1 in Leydig cells (Alvarez et al., 2008), which atrophy as the testes regress, while Bmal1 may play a noncircadian function in this or another cell type. It is unknown whether these changes in testicular clock gene expression contribute to photoperiodically induced changes in male fertility. Manipulation of these transcripts, perhaps through intratesticular administration of siRNA (Liang et al., 2013) or testis-specific conditional knockouts, would help to answer this question.
New insights into testicular function suggest additional directions for research into the basis of seasonal change. Evidence for the pivotal role of RA in the spermatogenic cycle (Hogarth et al., 2015; see above) indicates that seasonal cues may regulate the enzymes that control RA production or action in Sertoli cells and/or gametes. This possibility has not yet been addressed in seasonal breeders. Similarly, seasonal cues may regulate microRNAs that play critical roles in the stem cell niche (van den Driesche et al., 2014). The role of translational repression in seasonal inhibition also remains to be studied. Peritubular myoid cells, now known to be involved in spermatogenesis and targeted by androgens, may also be targeted in seasonal arrest. Further work is called for to elucidate the proximate endocrine signals that profoundly alter testicular function as the seasons change. The advent of molecular tools, based on the availability of complete genetic sequences for seasonally breeding species such as the Syrian hamster, should make it possible to perform critical experiments.
Perspective
In conclusion, few tissues exhibit the rhythmic complexity of the testis. Spermatogenesis, a core function of the male gonad, exhibits an exquisitely coordinated and periodic pattern of tubular function that depends on both gonadotropic support and rhythmic production of retinoic acid by the seminiferous epithelium. The pulsatile operation of the neuroendocrine axis gives rise to ultradian rhythmicity of androgen production, the physiological significance of which remains to be fully understood but may include consequences for both physiology and behavior. Core circadian clock genes are expressed, but the complexity of testicular organization demands a more sophisticated approach to the study of the temporal and spatial patterns than suffices for the study of other organs. Cell- and stage-specific manipulations of clock gene expression will be essential to gaining an understanding of the functional significance of the circadian clock in this organ. Finally, spectacular seasonal changes in testicular function occur in response to environmental signals and, in some cases, according to a circannual program. The mechanisms of these annual rhythms, which most often reflect powerful influences of daylength, remain to be fully elucidated. A fruitful avenue for future work will be to investigate the interactions between rhythms of testis function across multiple temporal domains, as the mechanisms that generate the spermatogenic wave, rhythms of expression of core circadian and clock-controlled genes, episodes of androgen secretion, and annual changes in male fertility may influence one another.
Footnotes
Acknowledgements
Preparation of this review was supported by funds from NICHD R21-HD078863. I thank Pablo Visconti and Dirk G. De Rooij for helpful comments. This review is dedicated to the memory of Timothy J. Bartness, a good friend and a wonderful colleague.
Conflict of Interest Statement
The author has no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
