Abstract
The suprachiasmatic nucleus (SCN) drives circadian rhythms in behavioral and physiological variables, including the inflammatory response. Shift work is known to disturb circadian rhythms and is associated with increased susceptibility to develop disease. In rodents, circadian disruption due to shifted light schedules (jet lag) induced increased innate immune responses. To gain more insight into the influence of circadian disruption on the immune response, we characterized the inflammatory response in a model of rodent shift work and demonstrated that circadian disruption affected the inflammatory response to lipopolysaccharide (LPS) both in vivo and in vitro. Since food consumption is a main disturbing element in the shift work schedule, we also evaluated the inflammatory response to LPS in a group of rats that had no access to food during their working hours. Our results demonstrated that the shift work schedule decreased basal TNF-α levels in the liver but not in the circulation. Despite this, we observed that shift work induced increased cytokine response after LPS stimulation in comparison to control rats. Also, Kupffer cells (liver macrophages) isolated from shift work rats produced more TNF-α in response to in vitro LPS stimulation, suggesting important effects of circadian desynchronization on the functionality of this cell type. Importantly, the effects of shift work on the inflammatory response to LPS were prevented when food was not available during the working schedule. Together, these results show that dissociating behavior and food intake from the synchronizing drive of the SCN severely disturbs the immune response.
Keywords
In mammals, the hypothalamic suprachiasmatic nucleus (SCN) is considered the master circadian pacemaker, which is entrained mainly by the light-dark cycle. The biological clock drives 24-h (circadian) rhythms in most physiological and behavioral variables, including hormone secretion, body temperature, locomotor activity, and sleep (Hastings et al. 2003). The modern lifestyle, night work and shift work, and light schedules have changed our activity period and food consumption toward the night, promoting the disruption of circadian rhythmicity.
Recent data in the human and animal literature suggest that circadian desynchronization, mainly caused by the exposure to nontraditional work schedules or to “jet lag,” increases the susceptibility to develop pathologies (Evans and Davidson, 2013) such as cancer (Filipski et al., 2006; Schernhammer et al., 2001; Schernhammer et al., 2003; Takane et al., 2002), reproductive disorders (Nurminen, 1998) overweight (Barclay et al., 2012; Karlsson et al., 2001; Salgado-Delgado et al., 2013), diabetes (Karlsson et al., 2005; Morikawa et al., 2005), stroke (Karlsson et al., 2005), and cardiovascular disease (Portaluppi et al., 2012; Tenkanen et al., 1998; Tuchsen et al., 2006).
The effects of circadian disruption on the immune system have been analyzed in rodents subjected to experimental jet lag (chronic shifts of the light-dark cycle). Such a protocol resulted in increased cytokine production and lethality to high doses of lipopolysaccharide (LPS), a component of gram-negative bacteria (Castanon-Cervantes et al., 2010). The rhythms of leukocyte recruitment to tissues under physiological and inflammatory conditions are also disrupted after experimental jet lag (Scheiermann et al., 2012). In mice, shifted light schedules modified circadian expression of clock genes and cytolytic factors in natural killer (NK) cells, as well as their cytotoxic activity, which is associated with cancer development (Logan et al., 2012).
Recently, we demonstrated a direct involvement and an inhibitory role of the SCN in the inflammatory response to LPS (Guerrero-Vargas et al., 2014). Previously, our group demonstrated that in a rat model of shift work, the activation of the SCN is reduced (Salgado-Delgado et al., 2010b). Therefore, we hypothesized that the cause for the development of certain pathologies in shift workers might be a disturbed response of the immune system due to circadian desynchrony. To test this idea we have used an experimental model of shift work in Wistar rats (forced activity during the rest phase) that was developed in our laboratory (Salgado-Delgado et al., 2008). Animals subjected to this protocol exhibit disrupted circadian rhythmicity, consume more food during the rest period, gain more body weight, and show clear signs of metabolic disturbances, such as lipid accumulation (Salgado-Delgado et al., 2010a). Moreover, after this protocol, the liver was notably affected, showing a loss of rhythmic expression in clock and metabolic genes (Salgado-Delgado et al., 2013). The liver plays an important role in the elimination of systemic bacterial infection (Benacerraf et al., 1959; Gregory et al., 1996; Holub et al., 2009) and is the main filter of circulating LPS in the body (Jirillo et al., 2002) as it contains one of the largest macrophage reservoirs (Kupffer cells) capable of recognizing and responding to circulating LPS (Wu et al., 2010a).
Here we evaluated the effects of the “shift work” schedule on TNF-α and IL-6 production after LPS stimulation at 2 different time points. Basal TNF-α protein content in the liver was also analyzed. The effects of desynchronization on liver macrophage (Kupffer cells) response to LPS were evaluated in vitro. Finally, the contribution of shifted food consumption on the increased inflammatory response to LPS in shift work animals was examined.
Methods
Animals and General Housing Conditions
Adult male Wistar rats weighing 210 to 220 g at the beginning of the experiments were obtained from the animal facility of the Faculty of Medicine of the Universidad Nacional Autónoma de México (UNAM). Animals were housed in individual cages and maintained in a monitoring room with a 12:12-h light-dark cycle (LD) that entailed lights-on at 0700 h, defined as zeitgeber time 0 (ZT0), and lights off at 1900 h, defined as zeitgeber time 12 (ZT12). The room was maintained at a controlled temperature of 22 ± 1 °C and had continuous air flow. All rats were housed under specific pathogen-free conditions and given free access to food (Rodent Laboratory Chow 5001, Purina, Minnetonka, MN, USA) and water, unless otherwise stated. Experimental procedures used in this study were in strict accordance with the Mexican norms for animal handling, Norma Oficial Mexicana NOM-062-ZOO-1999, which conforms to international guidelines for animal handling, and were approved by the Ethics Committee in the Instituto de Investigaciones Biomédicas UNAM. All efforts were made to minimize the number of animals used and their suffering.
Desynchronization Protocol (Shift Work)
From Monday to Friday for 8 h (from ZT2 to ZT10), rats were placed in slow rotating drums that are used for sleep deprivation (33 cm diameter, 633 cm wide). Each rotating drum has 4 concentric subdivisions, which allows individual housing of 4 rats. Drums rotate with a speed of 1 revolution per 3 min to force the rats to stay awake. Due to the slow movement of the drums, rats do not need to walk all the time; they can sit, groom, and even lie down. In addition, they can drink and eat freely from a small bottle and pellets hanging from the middle tube (Salgado-Delgado et al., 2008). This procedure was carried out for 5 weeks from Monday to Friday. During weekends, all rats remained undisturbed in their home cages.
Experimental Design
For acclimatization, all rats were monitored in their home cages for 8 days in LD conditions. After this period, rats were randomly assigned to 1 of 4 groups: Control rats (CTRL) were left undisturbed in their home cages during 5 weeks; shift work (SW) rats were exposed for 5 weeks to the shift work protocol; shift work rats without food in the wheel (SW-NFW) were exposed for 5 weeks to the shift work protocol without food access during the working hours; rats fed during the day (FD) were provided with food exclusively 12 h/day during the light phase for 5 weeks.
Surgeries were performed after rats concluded 4 weeks of shift work or FD protocol. All experiments were performed at the end of the fifth week of the shift work or FD protocol. For this study, a total of 103 Wistar rats were used.
Experiment 1
Experiment 1 examined the effect of circadian desynchronization (shift work) on the following:
Basal TNF-α protein content in the liver
Proinflammatory cytokine blood levels after LPS administration
CTRL and SW rats (n = 4-5/group) were sacrificed at 1 of 2 time points: ZT2 (2 hours after lights-on) or ZT14 (2 hours after lights-off) in order to obtain the liver and determine TNF-α protein levels using an ELISA kit. Other series of CTRL and SW rats were cannulated in the external jugular vein and assigned to 1 of 2 groups, LPS administration at ZT2 or LPS administration at ZT14 (n = 5-7/group). These administration time points were chosen based on previous studies (Guerrero-Vargas et al., 2014). At the end of the fifth week of the shift work protocol, each rat received IV LPS (100 µg/kg). Blood samples were collected from the jugular cannula at 0, 40, and 80 min post LPS. In the obtained serum, IL-6 and TNF-α were determined.
Experiment 2
Experiment 2 examined the effect of shift work on the response of liver macrophages (Kupffer cells) to LPS stimulation in vitro. The liver of CTRL (n = 5) and SW (n = 9) rats was perfused at ZT2 through the portal vein with Hanks’ balanced salt solution (HBSS) containing 0.2% IV collagenase from Clostridium histolyticum. Kupffer cells were separated using 2-step Percoll according to Froh et al. (2003); for details, see Kupffer Cell Isolation and Culture. Kupffer cells isolated from CTRL and SW rats were incubated with fresh medium containing 1 µg/mL of LPS or vehicle (HBSS) for 0, 1, 2, 4, 6, and 24 h at 37 °C. TNF-α was measured in the conditioned medium samples of all incubation times using an ELISA kit.
Experiment 3
Experiment 3 examined the effect of feeding schedule on the inflammatory response to LPS of desynchronized rats. A group of rats (n = 7) were assigned to the shift work protocol. However, throughout the 5-week protocol, food was not available in the activity wheels (i.e., from ZT2 to ZT10). Animals had free access to food at the end of the daily shift work schedule (i.e., from ZT10 to ZT2). During the weekends, they remained undisturbed in individual cages and had free access to food and water. A group of shift work rats with no food in the activity wheel (SW-NFW, n = 7), shift work animals fed ad libitum (SW, n = 8), and control rats (CTRL, n = 7) were cannulated in the external jugular vein, and after recovery they received LPS (100 µg/kg) at ZT2. Blood samples were collected from the jugular cannula at 0, 40, and 80 min post LPS. From the obtained serum, IL-6 and TNF-α were determined.
Experiment 4
Experiment 4 examined the effect of feeding schedule on the inflammatory response to LPS. A group of rats were provided with food exclusively for 12 h/day during the light phase for 5 weeks (FD group). Control rats were fed ad libitum (CTRL) and were left undisturbed in their home cages during 5 weeks.
CTRL and FD rats were cannulated and after recovery were randomly assigned to 1 of the 2 groups: LPS (1.6 µg/kg) administration at ZT2 (n = 5-8/group) or LPS (1.6 µg/kg) administration at ZT14 (n = 6/group). This dose was chosen based on previous studies demonstrating a major role of the SCN in the regulation of the inflammatory response to LPS (Guerrero-Vargas et al., 2014).
At the end of the fifth week of the FD protocol, each rat received IV LPS (1.6 µg/kg). Blood samples were collected from the jugular cannula 0, 40 and 80 min post LPS. From the obtained serum, IL-6 and TNF-α were determined.
Surgical Procedures and Intrajugular Cannula Insertion
All surgeries were performed using aseptic procedures. Rats were anesthetized with a combination of ketamine (90 mg/kg, Cheminova, México) and xylazine (10 mg/kg, Procin, PiSA, Atilalaquia, Hgo, México) anesthesia. A polyethylene silicon tube cannula (0.025 inches inner diameter and 0.047 inches outer diameter; Silastic Laboratory tubing; Dow Corning Corp., Midland, MI, USA) was inserted in the internal jugular vein. The cannula was filled with heparin (500 U/mL) as anticoagulant. The outer end of the cannula was fixed in the back between both shoulder blades and clotted with a small needle. Surgeries were performed on Fridays when rats concluded 4 weeks of SW or FD protocol; rats were allowed to recover during the weekend, and the following Monday the working protocol was restarted for a fifth week. All experiments were performed 7 days after cannula insertion—that is, at the end of the fifth week protocols.
Treatment Solutions and Reagents
For all experiments, inflammation was induced by a single IV injection of lyophilized LPS (100 mg, Escherichia coli serotype 0127:B8, Sigma-Aldrich, St. Louis, MO, USA; Lot No. 051M4004) reconstituted with 0.9% sterile pyrogen-free saline (Baxter, México) to derive a 1 mg/mL stock solution that was aliquoted and stored at −20 °C. For the administration, aliquots were thawed and diluted with 0.9% sterile pyrogen-free saline yielding a final concentration of 100 µg/kg or 1.6 µg/kg.
Sample Collection and ELISA
Blood samples (300 µL) were collected in Microvette/500 tubes (Sarsted, Nümbrecht, Germany) before LPS infusion (0) and at postinfusion times 40 and 80 min. Samples were centrifuged at 7000 rpm, during 7 min. Plasma was stored in 60-µL aliquots at −80 °C until assay. IL-6 and TNF-α were determined by ELISA according to the manufacturer’s recommendations (Invitrogen, Frederick, MD, USA; KRC3012, Lot 1350343A for TNF-α and KRC0061C, Lot 1358388B for IL-6). Samples were analyzed in duplicate. Detection limits of the assays in plasma were <4 pg/mL for TNF-α and <5 pg/mL for IL-6. The intra-assay coefficient of variation (%CV) was 6.3% for TNF-α and 7.8% for IL-6. The interassay %CV was 2.6% for TNF-α and 3.93% for IL-6. Cytokine levels are expressed as picogram per milliliter.
Tissue Collection
After 5 weeks in the shift work protocol, a group of SW and CTRL rats were sacrificed at ZT2 or ZT14. Rats were deeply anesthetized with an overdose of sodium pentobarbital (Pisabental, PiSA, Atilalaquia, Hgo, México; 65 mg/mL), and part of the left lobule of the liver was quickly removed and immediately frozen at −80 °C.
TNF-α Determination in the Liver
Frozen tissue samples were weighed and placed in saline at a ratio of 250 mg of tissue per 0.5 mL of 0.9% sterile pyrogen-free saline. Samples were homogenized. The final homogenates were centrifuged (12,000 rpm 10 min), and supernatants were immediately assayed for TNF-α using an ELISA kit. Data were obtained as pg/mL and then transformed to pg/mg tissue.
Kupffer Cell Isolation and Culture
Kupffer cells were isolated from the livers of SW and CTRL rats using the method of Froh et al. (2003). Briefly, the liver was perfused through the portal vein with Ca2+- and Mg2+-free HBSS at 37 °C at a rate of 20 mL/min (200 mL total volume) and then with complete HBSS solution containing 0.2% IV collagenase from C. histolyticum (Sigma Aldrich, St. Louis, MO, USA; C5138-1G, Lot 061M8628V) at a rate of 20 mL/min (200 mL total volume). The liver was removed from the animal, placed in a sterile petri dish containing HBSS solution containing 0.2% IV collagenase, and sliced with scissors. Sliced tissue was filtered through a 100-µm nylon sterile cell strainer (BD Falcon, New jersey, USA, 352360, Lot 1293837). Kupffer cells were separated using 2-step Percoll gradient (GE Healthcare, Bio-sciences AB, Uppsala, Sweden). After 15 min of centrifugation at 1800 g, middle layers were collected, washed with HBSS, and resuspended in RPMI-1640 (Gibco, Grand Island, NY, USA; 1879-020, Lot 901692) containing 10% of fetal bovine serum (FBS, Gibco). The purity of the Kupffer cells was determined to be approximately 90% by CD163 (ED-2) staining (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA; SC-5865). The cells nuclei were stained with DAPI (1:3000 dilution; Santa Cruz Biotechnology) (Suppl. Fig. S1). Viability was found to be 98% using the trypan blue exclusion test (data not shown). Kupffer cells were incubated at 37 °C for 24 h under 5% CO2 in 24-well cell culture plates (Santa Cruz Biotechnology; Santa Cruz, CA, USA SC-204444) at a density of 5 × 105/mL and cultured in RPMI-1640 containing 10% FBS.
Kupffer Cell Treatment with LPS
Kupffer cells were incubated overnight in RPMI-1640 containing 10% FBS. On the following day, the cells were incubated with fresh medium containing 1 µg/mL of LPS for 0, 1, 2, 4, 6, and 24 h at 37 °C. Supernatants were then harvested and kept at −70 °C until assayed. TNF-α in the culture medium was measured using an ELISA kit.
Data Analysis
All data are presented as mean ± standard error of the mean (SEM). IL-6 and TNF-α plasma levels after LPS administration at ZT2 or ZT14 were compared with a 2-way ANOVA for repeated measures for condition (CTRL vs. SW or CTRL vs. FD) and time after LPS as a factor of repeated measures. This was followed by Bonferroni’s post hoc test for multiple comparisons. TNF-α plasma levels after LPS stimulation in isolated Kupffer cells from CTRL and SW were compared with a 2-way ANOVA for condition (CTRL vs. SW) and time after LPS administration as a factor of repeated measures. IL-6 and TNF-α plasma levels after LPS administration at ZT2 in CTRL, SW-NFW, and SW rats were compared with a 2-way ANOVA for condition (CTRL, SW-NFW, or SW) and time after LPS administration as a factor for repeated measures followed by Tukey’s post hoc test for multiple comparisons. Statistical comparisons were performed using Graphpad Prism version 6 for Macintosh. Threshold for statistical significance was set at α = 0.05.
Results
Shift Work Affected Basal TNF-α Protein Levels in the Liver
To investigate the effects of the experimental shift work schedule on basal TNF-α protein levels in an organ that plays an important role in the immune response, the livers of CTRL and SW rats were recovered at ZT2 or at ZT14.
In the livers of SW and CTRL rats, similar TNF-α protein levels were measured at both ZT2 and ZT14 (p = 0.05; Fig. 1); however, the 2-way ANOVA indicated a significant difference between conditions (CTRL vs. SW, F1,13 = 4.38, p = 0.046) with lower values for SW rats. No significant interactions between condition and ZT were obtained (F1,13 = 0.010, p = 0.918).
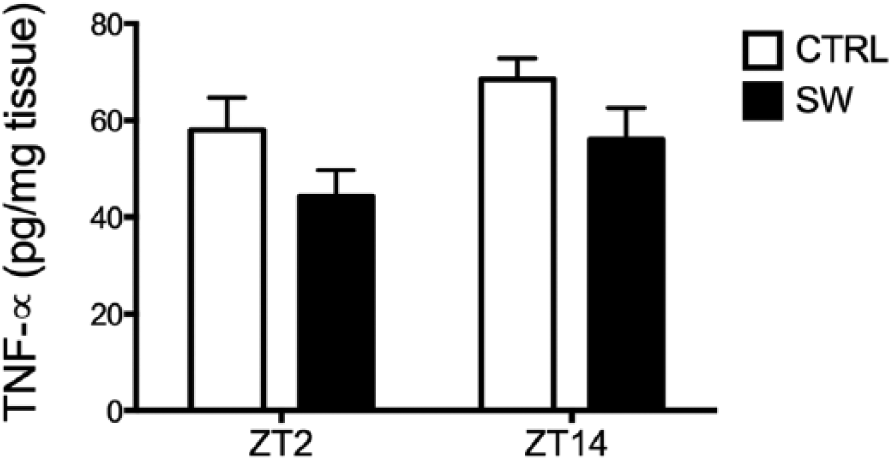
Basal TNF-α protein levels in the livers of control (CTRL) and shift work (SW) rats at ZT2 and ZT14 (n = 4-5/group). Values are expressed as mean ± SEM. Two-way ANOVA and post hoc Bonferroni testing.
Shift Work Increased Cytokine Production after LPS Administration at ZT2
To study the effect of shift work on TNF-α and IL-6 production after an inflammatory stimulus, LPS was given IV at ZT2 or ZT14 to CTRL and SW rats. Basal TNF-α and IL-6 plasma levels, as measured at time 0, were very low or undetectable and were not different between SW and CTRL rats in all cases.
In SW rats, LPS administration at ZT2 triggered significantly higher TNF-α plasma levels (p < 0.001) at both 40 and 80 min after LPS compared with CTRL rats (Fig. 2A). The 2-way ANOVA indicated a significant interaction between condition and time after LPS administration (F2,18 = 11.68, p = 0.0006). IL-6 plasma levels were significantly elevated 80 min after LPS administration at ZT2 in SW rats (p < 0.001) compared with CTRL rats (Fig. 2B). The 2-way ANOVA indicated a significant interaction between the condition and time after LPS administration (F2,18 = 29.92, p < 0.0001).
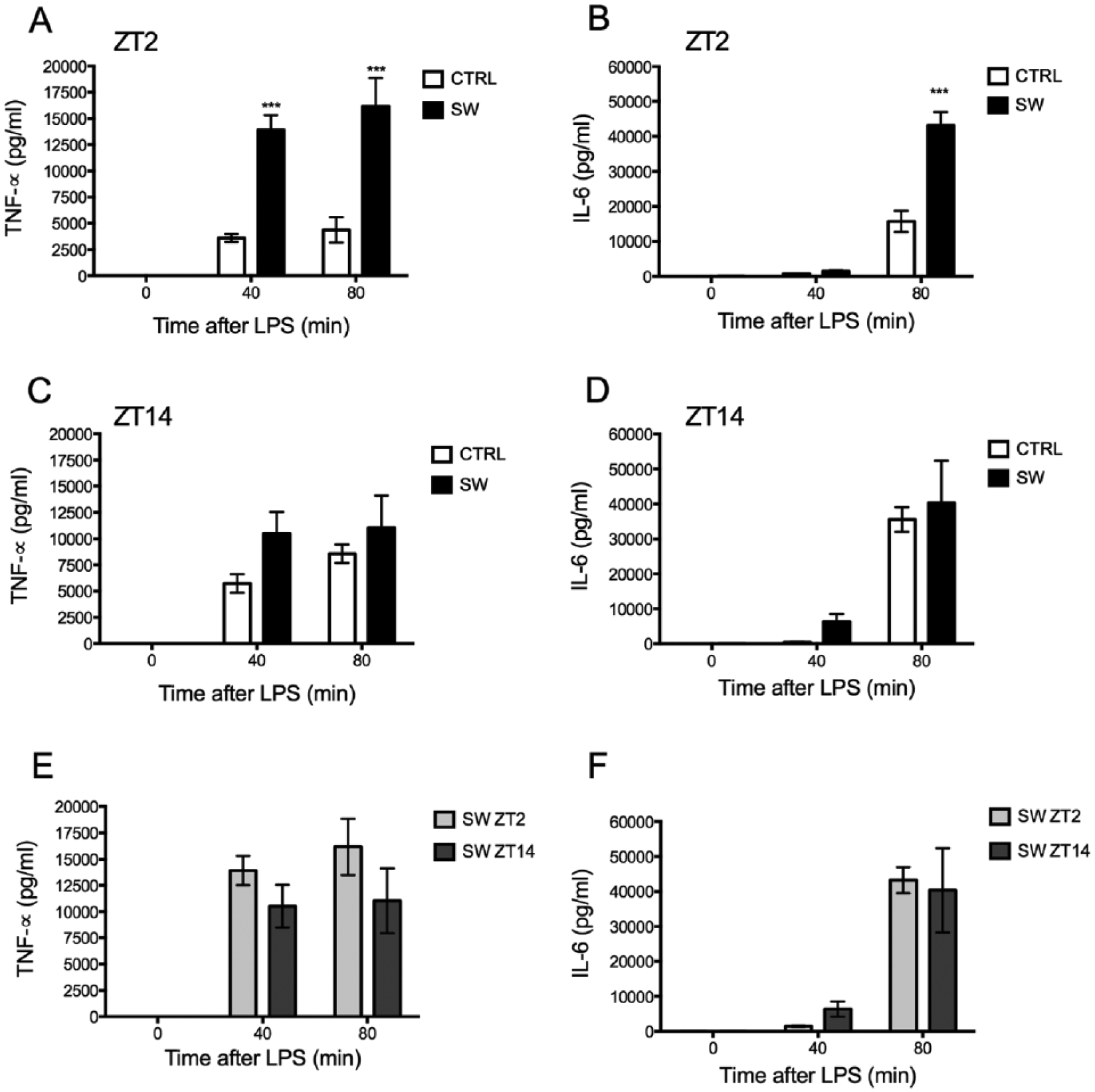
Shift work (SW) increased the inflammatory response to LPS. TNF-α (A) and IL-6 (B) plasma levels were higher in SW rats after LPS stimulation at ZT2 in comparison to control (CTRL) rats (n = 6-7/group). However, TNF-α (C) and IL-6 (D) plasma levels in SW and CTRL were similar after LPS administration at ZT14 (n = 5-7/group). Similar TNF-α (E) and IL-6 plasma levels (F) were measured after LPS administration at ZT14 or ZT2 in SW rats (n = 6-7/group). TNF-α and IL-6 plasma concentrations were measured before and after IV administration of LPS (100 µg/kg) at ZT2 or ZT14. Values are expressed as mean ± SEM. Two-way ANOVA and post hoc Bonferroni testing. ***p < 0.001 significance of difference vs. CTRL.
In contrast, ZT14 LPS administration resulted in similar TNF-α (Fig. 2C) and IL-6 (Fig. 2D) plasma levels in SW and CTRL rats at 40 and 80 min after the stimulus. The 2-way ANOVA indicated no significant interaction between condition and time after LPS administration (TNF-α, F2,20 = 0.112, p = 0.29; IL-6, F2,20 = 0.190, p = 0.827).
As we have previously shown (Guerrero-Vargas et al., 2014), LPS administration to CTRL (synchronized) rats at ZT14 triggered higher TNF-α and IL-6 plasma levels in comparison to LPS administrated at ZT 2. Here we confirm this time-dependent response and report in CTRL rats increased cytokine plasma levels at 80 min after the LPS administration at ZT14 (p < 0.001; Suppl. Fig. S2). In contrast, SW rats, with their increased response at ZT2, did not show this daily variation of TNF-α (Fig. 2E) or IL-6 (Fig. 2F) in response to LPS. The 2-way ANOVA indicated no significant effect of ZT (TNF-α, F1,9 = 3.486, p = .0.094; IL-6, F1,10 = 0.021, p = 0.8863) and no significant interaction between time after LPS administration and ZT (TNF-α, F2,18 = 0.894, p = 0.426; IL-6, F2,20 = 0.306, p = 0.739). Therefore, in the next series of experiments, aimed to investigate the cause for this disparate inflammatory response, we studied changes at ZT2, where the differences between SW and CTRL rats were the largest.
Kupffer Cells Isolated from Shift Work Rats Produced More TNF-α in Response to LPS
The liver is one of the main sources of cytokines after LPS is injected into the circulation; therefore, we evaluated the response to LPS of liver macrophages (Kupffer cells) isolated from shift work rats. Based on our in vivo results demonstrating an increased immune response of SW at ZT2, Kupffer cells were isolated from SW and CTRL rats at this time point.
Kupffer cells isolated from SW rats produced more TNF-α after LPS stimulation in vitro in comparison to CTRL Kupffer cells; this was significantly different between groups at 2, 6, and 24 h post LPS (p < 0.05; Fig. 3). The 2-way ANOVA indicated significant interaction between time after LPS administration and condition (F4,48 = 2.695, p = 0.0418). No differences in TNF-α supernatant levels were found between CTRL and SW after HBSS (vehicle) administration (mean values: CTRL, 17 ± 3.5; SW, 23 ± 5.1). The 2-way ANOVA indicated no significant interaction between time after vehicle administration and condition (F4,40 = 0.244, p = 0.911; data not shown).
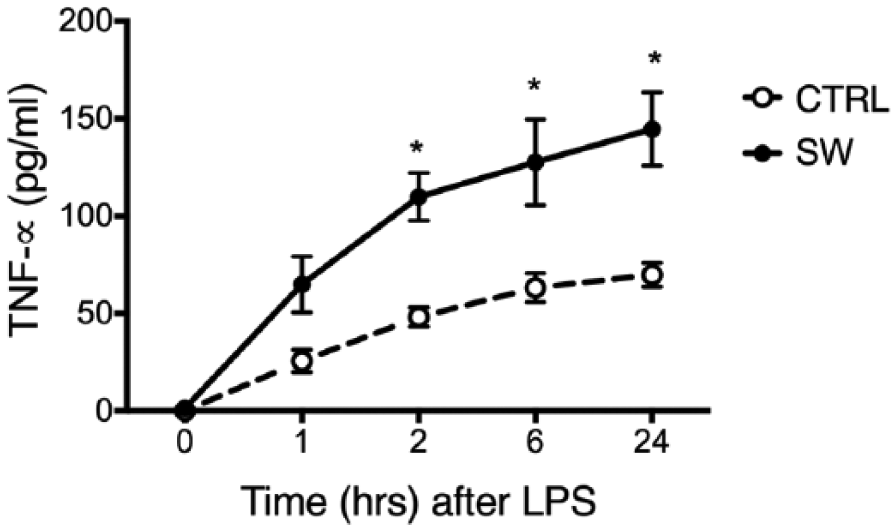
Kupffer cells isolated from shift work rats (SW) produce more TNF-α in response to LPS than Kupffer cells isolated from control rats (CTRL). TNF-α was measured in Kupffer cell conditioned medium containing LPS (1 µg/mL). Open circles represent TNF-α levels from CTRL isolated Kupffer circles and solid circles represent TNF-α levels from SW isolated Kupffer cells. Values are expressed as mean ± SEM. Each point represents the average ± SEM of 5-9 animals per group. Two-way ANOVA and post hoc Bonferroni test. *p < 0.05 significance of difference vs. CTRL.
Avoiding Food Intake during the Working Hours Prevented Increased Inflammatory Response
Previously it was demonstrated that desynchronization and weight gain induced by this shift work protocol were prevented if food intake was not available in the 8-h working period of the rat (Salgado-Delgado et al., 2010a). To investigate whether food intake during the rest phase was contributing to the heightened inflammatory response to LPS observed in SW rats, cytokine production after LPS administration at ZT2 was evaluated in a group of SW rats that had no access to food in the activity wheels (SW-NFW) during the 5-week protocol and consequently were eating mainly during the dark phase.
As observed previously, LPS administration at ZT2 to SW rats induced higher TNF-α plasma levels at 40 and 80 min compared with CTRL rats (p < 0.05). This response was prevented in SW-NFW rats that were only able to eat in their home cages (ZT10-ZT2), corresponding largely with their normal activity phase (Fig. 4A). TNF-α plasma levels in SW rats were also significantly higher compared with SW-NFW animals at 40 and 80 min after LPS administration (p < 0.05). The 2-way ANOVA indicated a significant interaction between time after LPS administration and condition (F4,36 = 4.034, p = 0.0084). No significant differences between the SW-NFW and CTRL groups were found after post hoc testing.
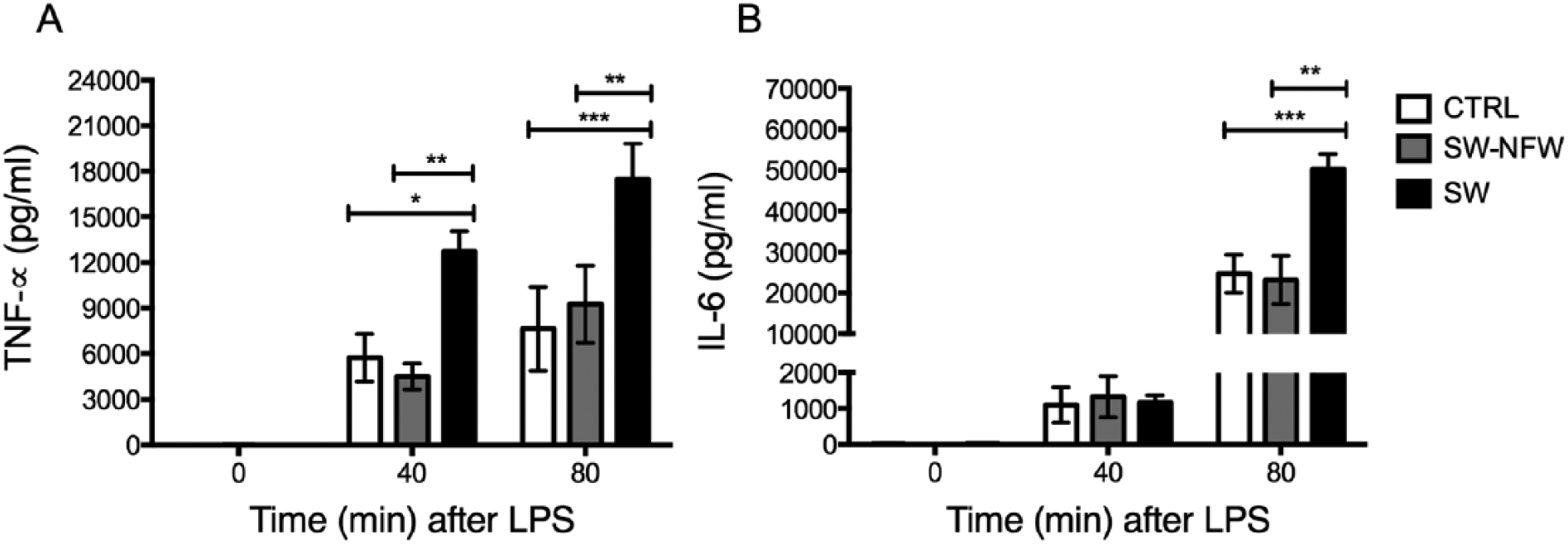
LPS administration at ZT2 to shift work rats without food in the wheel (SW-NFW) triggered similar TNF-α (A) and IL-6 (B) plasma levels in comparison to control (CTRL) rats. TNF-α and IL-6 plasma concentrations were measured before and after IV administration of LPS (100 µg/kg) at ZT2. Values are expressed as mean ± SEM. (n = 5-7/group). Two-way ANOVA and post hoc Tukey tests. *p < 0.05, **p < 0.01, ***p < 0.001 significance of differences vs. shift work rats (SW).
IL-6 plasma levels were also significantly increased in SW rats 80 min after LPS administration compared with CTRL and SW-NFW rats (p < 0.05; Fig. 4B). Restricting food intake to SW rats (SW-NFW) toward the night prevented this response (Fig. 4B). The 2-way ANOVA indicated a significant interaction between time after LPS administration and condition (F2,38 = 10.83, p < 0.0001). No significant differences between SW-NFW and CTRL groups were found after post hoc testing.
Food Intake during the Normal Rest Phase Increased the Inflammatory Response to LPS
To study the contribution of mistimed food consumption on the inflammatory response, LPS was given at ZT2 and ZT14 to a group of rats that were fed exclusively during the day. All animals that received their food ad libitum (CTRL) or during the day (FD) showed very low or undetectable TNF-α and IL-6 plasma levels at ZT2 as well as at ZT14. In contrast, LPS administration at ZT2 induced significantly higher TNF-α (Fig. 5A) and IL-6 (Fig. 5B) plasma levels in FD rats compared with CTRL animals at 40 min (p < 0.01) and 80 min (p < 0.01) after LPS administration. The 2-way ANOVA indicated a significant interaction between time after LPS administration and condition (TNF-α, F2,20 = 7.339, p = 0.003; IL-6, F2,18 = 9.892, p = 0.0013).
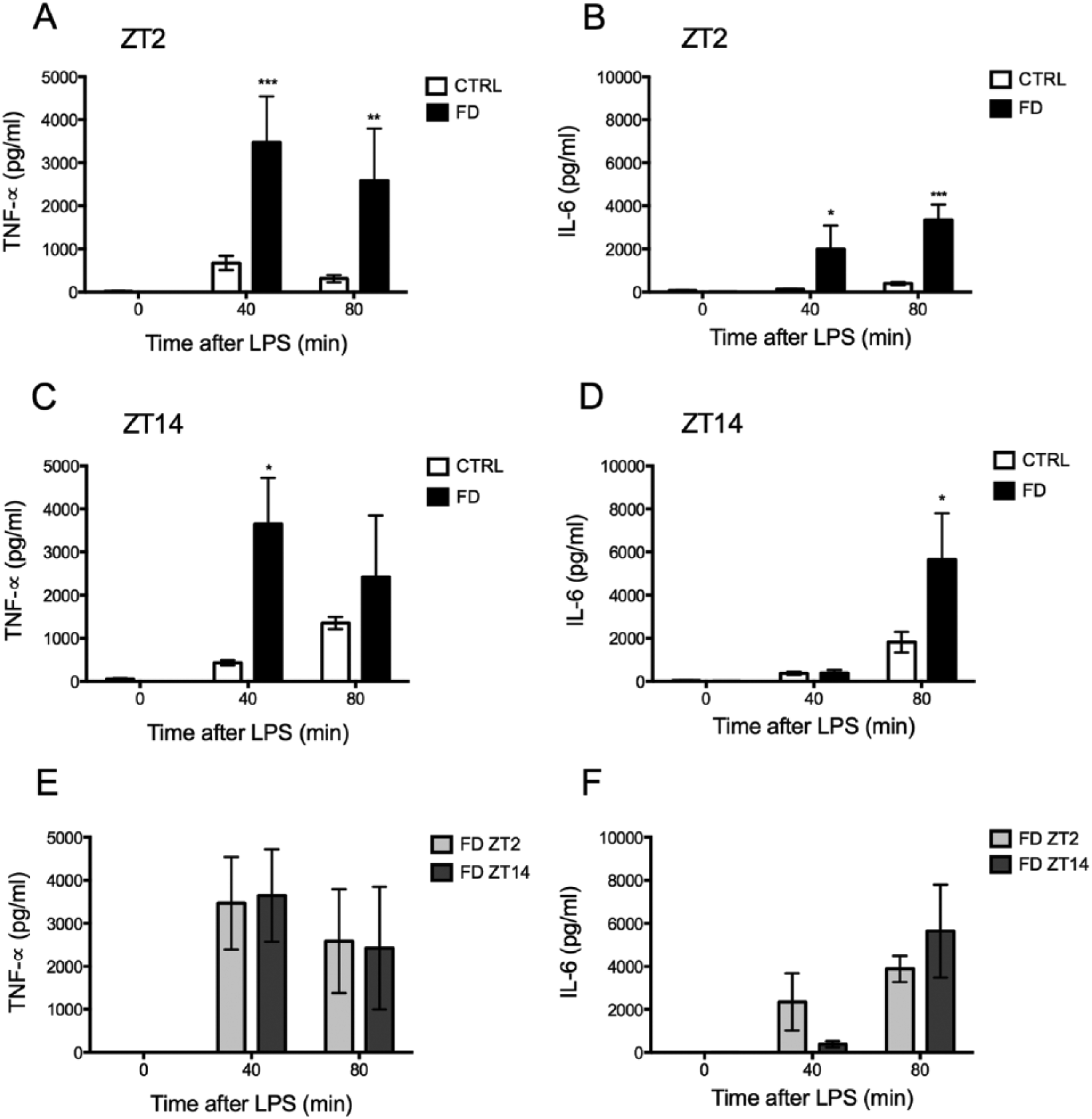
Food during the day (FD) increased the inflammatory response to LPS. TNF-α (A) and IL-6 (B) plasma levels after LPS stimulation at ZT2 were higher in FD rats in comparison to control (CTRL) rats (n = 4-8/group). LPS administration at ZT14 triggered higher TNF-α (C) and IL-6 (D) plasma levels in FD rats (n = 5-6/group). Similar TNF-α (E) and IL-6 (F) plasma levels were measured after LPS administration at ZT2 or at ZT14 in FD rats (n = 5-8/group). Cytokine concentrations were measured before and after IV administration of LPS (1.6 µg/kg). Values are expressed as mean ± SEM (n = 5-7/group). Two-way ANOVA and post hoc Bonferroni testing. *p < 0.05, **p < 0.01, ***p < 0.001 significance of differences vs. CTRL.
LPS administration at ZT14 also induced higher TNF-α plasma levels in FD rats at 40 min (p < 0.05; Fig. 5C) and higher IL-6 plasma levels at 80 min (p < 0.05; Fig. 5D) compared with CTRL rats. The 2-way ANOVA indicated a significant effect of time after LPS administration for both cytokines (TNF-α, F2,18 = 6.403, p = 0.007; IL-6, F2,20 = 10.08, p = 0.0009). Importantly, normal day-night variations in TNF-α and IL-6 plasma levels after LPS administration (Suppl. Fig. S3) were lost in FD rats. LPS administration at ZT2 in FD rats induced similar plasma levels of TNF-α (Fig. 5E) and IL-6 (Fig. 5F) as LPS given at ZT14. The 2-way ANOVA indicated no significant effect of ZT (TNF-α, F1,8 = 2.44e-005, p = 0.996; IL-6, F1,8 = 1.219, p = 0.321).
Discussion
Shift work induces circadian desynchronization and is associated with increased vulnerability to develop disease (Evans and Davidson, 2013). Nonetheless, the mechanisms underlying the effects of circadian desynchronization on the development of disease are not fully understood. Here we demonstrate that circadian desynchronization induced by an experimental model of shift work (SW) in Wistar rats leads to enhanced TNF-α and IL-6 production after LPS stimulation.
Importantly, avoiding food intake during their working hours in SW rats reduced the inflammatory response to LPS to control levels, illustrating that food intake in the day period as demonstrated with our FD group is the main event leading to desynchronization and enhanced inflammatory response.
Circadian Desynchronization Induced by Shift Work Increases the Inflammatory Response to an LPS Challenge
Here we report that LPS administration to SW rats during the light phase (ZT2) resulted in almost 4 times higher TNF-α and 3 times higher IL-6 plasma levels compared with the response of CTRL rats. Importantly, LPS administration at ZT14 induced similarly high cytokine levels in SW and CTRL rats. The explanation for this can be found in our earlier studies demonstrating that high cytokine production in response to LPS is associated with low SCN neuronal activity, a feature of the dark period (ZT14), while low cytokine production is associated with high SCN neuronal activity, a feature of the day period (ZT2) (Guerrero-Vargas et al., 2014). Salgado-Delgado et al. (2010b) demonstrated that shift work rats have low SCN neuronal activity at ZT2 compared with control rats, and in line with the previous observations, this suggests that in shift work animals the SCN does not diminish the cytokine response as efficiently as in control rats. Hereby it is also important to note that the cytokine response of SW at ZT2 is increased in a similar way as in SCN-lesioned rats (Guerrero-Vargas et al., 2014), which strengthens the point that SCN neuronal activity may play an important role in diminishing the inflammatory response to LPS.
Previous studies have demonstrated that the central nervous system influences the immune response via the autonomic nervous system (Felten and Felten, 1988; Martelli et al., 2014; Rosas-Ballina and Tracey, 2009). Since the output of the SCN determines a daily balance of the autonomic output to the organs (Buijs et al., 2013), an imbalance in the autonomic output to immune organs, such as the liver, in SW rats may promote an increased inflammatory response to LPS.
In SW rats, the disrupted rhythm of clock and metabolic genes in the liver, as demonstrated by Salgado-Delgado et al. (2013), may also be partly responsible for the increased inflammatory response, because their influence in immune functions is well documented (Arjona and Sarkar, 2006a, 2006b; Daynes and Jones, 2002; Gibbs et al., 2012; Keller et al., 2009; Liu et al., 2006; Logan et al., 2013; Oishi et al., 2006; Yang et al., 2012).
Glucocorticoids and the Inflammatory Response of Shift Work Rats
Since glucocorticoids are recognized as modulators of the immune response, their role in the inflammatory response of SW needs to be addressed. We have previously shown that the current shift work schedule does not change the circadian rhythm of corticosterone plasma levels. However, a peak in corticosterone is observed at ZT3 just after the animals are placed in the rotating wheels (Salgado-Delgado et al., 2008; Salgado-Delgado et al., 2010a). Besides inducing inflammation, LPS administration induces a high corticosterone response (Kalsbeek et al., 2012), the most accepted function of which is to decrease cytokine overproduction (Kapcala et al., 1995; Sternberg, 1997). Interestingly, SCN ablation induces even higher corticosterone levels (Kalsbeek et al., 2012) and is associated with an increased inflammatory response to LPS in the same manner as SW rats (Guerrero-Vargas et al., 2014), indicating that high corticosterone levels alone are not sufficient to suppress cytokine production.
Thus, in order to consider the effects of glucocorticoids on the immune response, we need to contemplate that those effects depend on several aspects such as hormone concentration and the moment of exposure; for example, exposure to stress—resulting in endogenous glucocorticoid production—or corticosterone administration prior to an LPS challenge is associated with increased cytokine production in the periphery and increased expression of inflammatory markers in the liver (Frank et al., 2010; Johnson et al., 2002). In contrast, corticosterone administration 1 h after an LPS challenge results in decreased cytokine response in both the brain and the liver (Frank et al., 2010).
Considering our experiments, it is important to note that restricting food availability, as in our SW-NFW group (shift-work, no food in the wheel), resulted in similar corticosterone levels as the SW group that had ad libitum food access (Salgado-Delgado et al., 2010a), yet the SW-NFW group showed a strong decrease in cytokine response compared with SW rats. Altogether this suggests that the increased inflammatory response of shift work rats is not likely to be driven by changes in the HPA axis activation and corticosterone production, and it points to a more complex mechanism.
Shift Work Induces Desynchronized Immune Responses
We have previously shown that the magnitude of TNF-α and IL-6 response after the administration of LPS is time dependent (Guerrero-Vargas et al., 2014). Here we confirm these findings and show that the day-night differences in cytokine response after LPS administration are lost in shift work rats.
In spite of the fact that shift work animals at ZT2 responded with higher cytokine levels after LPS challenge, their basal TNF-α protein levels in the liver tended to be diminished compared with CTRL animals, which suggests an amplified inflammatory response capacity or reaction of these animals to an immune challenge.
This observation is of relevance for human studies, where only certain inflammatory markers in the circulation are reported to be increased, such as leukocyte counts (Sookoian et al., 2007), C-reactive protein (CRP) levels (Puttonen et al., 2011), and IL-6 levels (Khosro et al., 2011). Other studies failed to find effects of shift work on inflammatory markers such as TNF-α (Khosro et al., 2011) and some even reported lower basal levels of circulating TNF-α and IL-β among shift workers (Copertaro et al., 2011). These conflicting data may be related to the heterogeneity of the populations but also may be explained by our present observations, where only under challenging conditions could a higher sensitivity for infection be detected. This enhanced inflammatory response may explain why shift work and circadian desynchronization are strongly associated with negative health consequences, indicating that the normal defense responses are disrupted. This is also suggested by the outcomes of other studies where phase shift schedules exacerbated the development of experimental colitis in mice (Preuss et al., 2008), reduced the survival of animals with cardiomyopathic heart disease (Penev et al., 1998), decreased the survival of rodents after high doses of LPS (Castanon-Cervantes et al., 2010), and resulted in increased tumor development (Filipski et al., 2003; Filipski et al., 2009; Logan et al., 2012).
Food Consumption during “Shift Work Hours” Promotes Inflammatory Response to LPS
Altered food consumption patterns observed in shift work rats are considered the basis of their desynchronization (Salgado-Delgado et al., 2010a). Here, one of the major findings was that the increased inflammatory response to LPS in shift work rats was prevented when food intake during the working hours was avoided and animals were allowed to eat only at the end of their working protocol. This shows that food consumption during the normal rest phase is one of the major factors contributing to the exacerbated inflammatory response to LPS. This holds for shift work and possibly also for other desynchronization protocols such as jet lag, since sleep loss is not associated with the increased immune response observed in jet lag (Brager et al., 2013). Since we have not recorded sleep, we cannot discard that sleep deprivation could have some effects in our shift work model. However, our data on shift work rats without food in the wheel combined with the data on rats receiving food during the day highlight the importance of food consumption in the rest phase as the driving force of the increased inflammatory response to LPS.
The observed effects of mistimed food intake on the inflammatory response have important implications for human health, since altered feeding schedules have been reported in shift workers (de Assis et al., 2003; Lennernas et al., 1995; Pasqua and Moreno, 2004). The present results show that shift work in rats leads to an augmented inflammatory response, while maintaining food intake patterns associated with the normal activity period helps to regulate the inflammatory response.
Footnotes
Acknowledgements
This work was supported by grants DGAPA IG-200314; CONACYT 220598. In addition, the research collaboration with the group of Dra Markus was funded by PROLAB IBRO LARC. Natalí N. Guerrero-Vargas is a PhD student in the Programa de Doctorado en Ciencias Biomédicas, Universidad Nacional Autónoma de México and received a scholarship from Consejo Nacional de Ciencia y Tecnología (CONACYT), México. Natalí N. Guerrero-Vargas, Joselyn García, Rebeca Fuentes, Roberto Salgado-Delgado, María del Carmen Basualdo, and Mara Guzmán contributed to the acquisition of the data. Natalí N. Guerrero-Vargas, Ruud M. Buijs, Carolina Escobar, and Regina P Markus contributed to the analysis and interpretation of the data. Ruud M. Buijs and Natalí N. Guerrero-Vargas initiated and designed the study. Natalí N. Guerrero-Vargas and Ruud M. Buijs wrote the paper.
Conflict of Interest Statement
The authors have declared that no competing interests exist.
