Abstract
The dorsomedial nucleus (DMN) of the hypothalamus has been implicated in seasonal control of reproduction. Syrian hamsters with DMN lesions, unlike control hamsters, do not undergo testicular regression after transfer from a long day length (14 h of light per day; LD) to a short day length (8 h of light per day; SD). SDs also markedly reduce hamster locomotor activity (LMA). To assess whether the DMN is a component of the neural circuitry that mediates seasonal variation in LMA, neurologically intact males (controls) and hamsters that had sustained lesions of the DMN (DMNx) were housed in an LD or SD photoperiod for 26 weeks. DMNx that prevented testicular regression counteracted decreases in LMA during 8 to10 weeks of SD treatment; steroid-independent effects of SDs did not override high levels of LMA in DMNx males. As in previous studies, testosterone (T) restoration increased LMA in LD but not SD castrated control males. In the present study, T also failed to increase LMA in SD-DMNx hamsters. The DMN is not necessary to maintain decreased responsiveness of locomotor activity systems to T in SDs, which presumably is mediated by other central nervous system androgen target tissues. Finally, DMNx did not interfere with the spontaneous increase in LMA exhibited by photorefractory hamsters after 26 weeks of SD treatment. We propose that DMN is an essential part of the substrate that mediates seasonal decreases in LMA as day length decreases but is not required to sustain decreased SD responsiveness to T or for development of refractoriness to SDs.
Annual changes in biotic and abiotic factors present challenges that affect survival for temperate zone rodents (Bronson, 1989; Sadleir, 1969), most of which exhibit some degree of seasonality that is controlled proximately by day length (DL). The transition from long to short days in male Syrian hamsters (Mesocricetus auratus) is representative of adaptations by numerous male rodents (Prendergast et al., 2002), including gonadal quiescence (Reiter, 1980), marked decreases in plasma concentrations of testosterone and prolactin (Ellis and Turek, 1979; Maywood and Hastings, 1995), increases in fur density (Paul et al., 2007), and cessation of copulatory behavior (e.g., Beery et al., 2007). DL information is transduced into a nocturnal pineal melatonin signal (Bartness et al., 1993; Bittman et al., 1983; Carter and Goldman, 1983), the duration of which is proportional to the length of the night (Darrow and Goldman, 1986). The increased duration of nocturnal melatonin secretion in short day lengths, decoded by pituitary and central nervous system substrates, mediates seasonal transitions in several traits (reviewed in Bartness et al., 1993; Kriegsfeld and Bittman, 2010; Prendergast et al., 2002; Goldman, 2001; Paul et al., 2008).
The timing and duration of locomotor activity (LMA) is an integral component of foraging activity, territorial defense, and the search for mates; locomotor activity in simulated foraging tests is physiologically costly (Vaanholt et al., 2007), and activity synchronization among individuals in a population can reduce the risk of predation (Daan and Slopsema, 1978). The neuroendocrine substrates that mediate seasonal rhythms of locomotor activity are poorly understood. In male Syrian hamsters transferred from long (LD) to short (SD) day lengths and maintained in SDs thereafter, wheel-running activity decreased from approximately 6000 to 2000 revolutions within 3 months and rebounded to LD values approximately 4 months later, mirroring the initial decline and subsequent recovery of serum testosterone concentrations (Ellis and Turek, 1979, 1983). Castrated hamsters transferred from LDs to SDs also exhibited a gradual decline in wheel running followed by recovery to LD values several months later (Ellis and Turek, 1983), thereby establishing steroid-independent modulation of wheel running by SDs. Implantation of testosterone (T) capsules increased LMA and sexual behavior of LD castrates but failed to augment these behaviors in their SD counterparts (Ellis and Turek, 1983; Morin and Zucker, 1978). The sites of action at which T increases LMA are not specified for any of the commonly studied seasonal rodents.
The dorsomedial nucleus of the hypothalamus (DMN) is a critical target tissue through which melatonin controls seasonal rhythms of reproduction in male and female Syrian and Siberian hamsters (Maywood and Hastings, 1995; Maywood et al., 1996; Lewis et al., 2002; Leitner and Bartness, 2011) without compromising melatonin secretion (Maywood and Hastings, 1995; Lewis et al., 2002; Mota et al., 2001). DMN lesions (DMNx) prevented testicular regression in SDs but did not interfere with normal decreases in circulating prolactin concentrations in Syrian hamsters (Maywood and Hastings, 1995). Melatonin treatments that induced gonadal regression in intact males failed to affect testicular function in DMNx hamsters (Maywood and Hastings, 1995).
Melatonin acts on different neuroendocrine substrates to modulate seasonal changes in gonadotropic and lactotropic function (Maywood and Hastings, 1995; Jarjisian et al., 2013; Lincoln et al., 2003; Lewis et al., 2002), but the neural target tissues through which melatonin modulates LMA remain unspecified. Decreases in nocturnal LMA in short days are reversed by pinealectomy (Prendergast et al., 2013). We assessed whether the DMN is a critical component of the neural circuit that mediates seasonal changes in LMA. Specifically, we examined whether (1) DMN lesions that block gonadal regression in short day lengths also counteract short-day decreases in LMA; (2) the DMN is an essential component of the neural mechanism that reduces responsiveness to T in SD hamsters; and (3) the DMN is essential to implement the transition to elevated LMA in photorefractory SD hamsters.
Materials and Methods
Experiment 1
Animals
Male Syrian hamsters from a colony established with stock obtained from Harlan (Indianapolis, IN) were housed individually in translucent polypropylene cages (48 × 27 × 20 cm) on Tek-Fresh Lab Animal Bedding (Harlan Teklad, Madison, WI). Hamsters initially were maintained under a 14L:10D long day (LD) cycle (14 h of light and 10 h of darkness; lights on at 0200 h, PST); groups either remained in LDs or were transferred to an 8L:16D short day (SD) cycle (lights on at 0800 h; see “Photoperiod and endocrine manipulations” below). Room temperature was maintained at 22 ± 2 °C with Harlan 8664 Teklad Rodent Diet and tap water available ad libitum. At regularly scheduled intervals, body mass and the length and width of the hamsters’ right testes were measured externally under light anesthesia induced by isoflurane vapors (Baxter Healthcare, Deerfield, IL). Estimated testis volume (ETV), a measure highly correlated with testis weight and reproductive competence, was calculated as the product of testis length × the square of testis width (Watson-Whitmyre and Stetson, 1985). All procedures were approved by the Animal Care and Use Committee of the University of California, Berkeley (institutional approval number R084-0911C) and conformed to USDA guidelines for the care and use of laboratory animals.
Hypothalamic lesions
Surgery was performed under ketamine cocktail anesthesia (21 mg ketamine, 2.4 mg xylazine, and 0.3 mg acepromazine per milliliter; 0.34 mL/100 g body mass injected intraperitoneally) supplemented with isoflurane vapors as necessary. Hamsters received subcutaneous injections of 5% buprenorphine (0.2 mL per animal; Hospira Inc., Lake Forest, IL) perioperatively and 12 h postoperatively. DMNx were placed using a Radionics Model RFG-4A radio frequency lesion generator system. Coordinates were 0.53 mm posterior to bregma, 0.4 mm lateral to midline, and 7.8 mm ventral to dura mater, with the skull level between bregma and lambda. Lesions were produced by delivering 25 mV for 15 sec through an electrode insulated with epoxy except for 0.5 mm at the electrode tip. For sham operations, the electrode was lowered to a depth 1 mm dorsal to the DMN, without applying current.
Photoperiod and endocrine manipulations
Hamsters recovered from surgery in LDs. Approximately 5 weeks later, LMA recordings were initiated (see “Activity recording” below) and continued for the remainder of the experiment. Eight to 10 weeks after lesion placement (experimental week 0), hamsters either were transferred to SDs (SD-Sham, n = 12; SD-DMNx, n = 21) or remained in LDs (LD-Sham, n = 10; LD-DMNx, n = 11) for the remainder of the study. ETV and body mass were determined at regular intervals between weeks 0 and 10. All hamsters were castrated on week 11 and treated with subcutaneous implants of T beginning on week 13. The timeline of experimental manipulations is shown in Figure 1A.
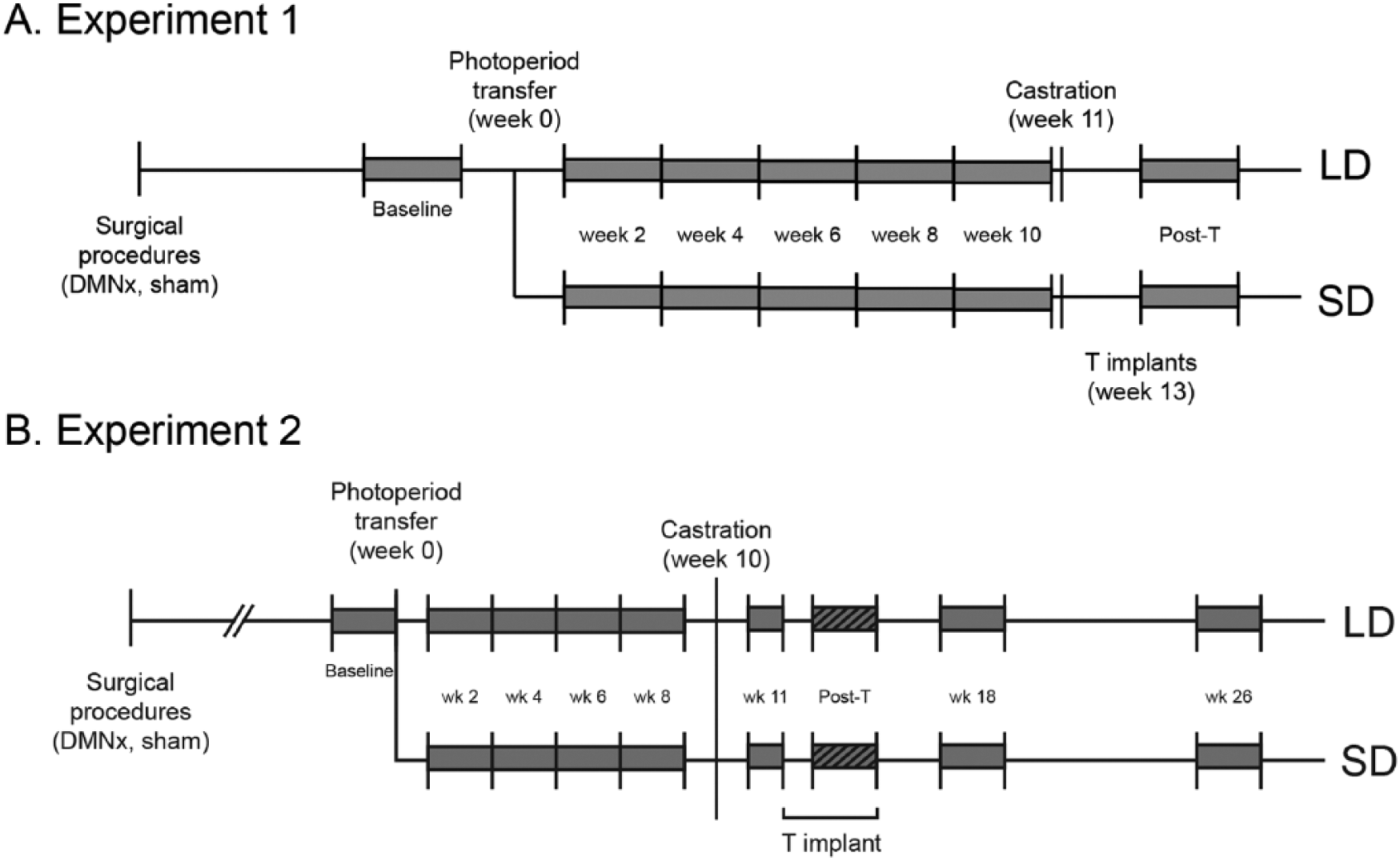
Schematic representation of the timing of interventions. See “Methods” for details. Shaded gray areas indicate intervals during which locomotor activity data were analyzed.
Castrations
Hamsters were anesthetized with isoflurane vapors and castrated through a midline incision in the abdominal cavity. Incisions were closed internally with sterile sutures and externally with 9-mm wound clips. Buprenorphine was administered perioperatively and 12 h postoperatively, as described above.
Testosterone treatments
Silastic capsules (Dow Corning, Midland, MI; ID 1.98 mm, OD 3.18 mm) were filled to a length of 4 mm with crystalline T (Sigma, St. Louis, MO); the ends of the capsules were sealed with silicone rubber cement. Capsules were incubated in a physiologic saline solution for 24 h prior to subcutaneous placement in the interscapular area of isoflurane-anesthetized hamsters via a small incision closed with a wound clip. Capsules of this size generate serum T concentrations of 1 to 3 ng/mL (Campbell et al., 1978; Arteaga-Silva et al., 2005).
Blood sampling
Under isoflurane anesthesia, ~1.0 mL of blood was withdrawn from the retro-orbital sinus between 1300 and 1500 h at weeks 10, 12, and 15, with capillary tubes. Samples were centrifuged at 3500 rpm for 20 min, and serum samples were stored at −80°C prior to radioimmunoassay for prolactin (PRL).
PRL radioimmunoassay
A low serum PRL concentration in SDs is a useful marker for identifying photoresponsive hamsters (e.g., Maywood and Hastings, 1995; Bae et al., 1999). PRL was assayed using the hamster PRL kit from the National Hormone and Peptide Program (NHPP), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), and Dr. A. F. Parlow. Assay details and results for these hamsters are described by Jarjisian et al. (2013). DMN lesions that block gonadotropic, but not lactotropic responses to SDs (Maywood & Hastings, 1995; Lewis et al., 2002; Jarjisian et al., 2013), are considered effective because the lactotropic response to SD confirms that pineal function has not been compromised.
Activity recording
LMA was monitored continuously beginning 3 weeks prior to week 0 (baseline period) with passive infrared detectors mounted on plastic brackets positioned above the wire-bar cage lids. Movement in the cage across 3 or more of 27 zones activated a closed-contact relay to Dataquest III software (Data Sciences, St. Paul, MN). Data were collected in 10-min bins and exported as ASCII files to Clocklab (Actimetrics, Evanston, IL) to generate actograms and to Microsoft Excel for parsing and further reduction.
Data reduction
Mean daily (light phase + dark phase) activity counts were quantified over several experimental epochs: baseline (10 days prior to photoperiod transfer on week 0) and for 14 days beginning on each of weeks 2, 4, 6, 8, and 10, after photoperiod manipulation and before castration (Fig. 1A). Mean daily activity counts also were compiled for a 7-day interval during the first weeks after castration (week 11) and for a 17-day interval beginning 1 day after hamsters received T implants (week 13, Fig. 1A).
Statistics
To correct for individual differences in sensitivity and/or positioning of motion sensors, activity counts were expressed as a percentage of each individual’s baseline activity value. Changes in activity between baseline and week 10 were compared across treatment groups with repeated-measures ANOVA. Within-group changes in LMA in response to (a) photoperiod (from baseline through week 10), (b) castration (week 11), and (c) T implants (week 13) were compared using paired t tests on uncorrected activity data. All pairwise comparisons between groups were performed with Tukey’s tests on planned comparisons (Statview). All values are presented as mean ± SEM. Differences were considered significant if p ≤ 0.05, with 2-tailed tests, except where noted otherwise.
Experiment 2
Animals
Housing conditions and procedures were similar to those in Experiment 1, except hamsters were obtained directly from Harlan. The timeline of interventions is shown in Figure 1B.
Hypothalamic lesions
Hamsters housed in the LD photoperiod received either radiofrequency or sham lesions that targeted the DMN (tooth bar +5 mm above the ear bars, 2.4 mm anterior to bregma, ±0.4 mm lateral to midline, and 7.5 mm ventral to dura). Current was delivered to heat the electrode tip temperature to 80° C for 15 sec. The experiments were conducted at intervals separated by 3 years by a different experimenter with coordinates determined with the skull level in Experiment 1 but not in Experiment 2.
Photoperiod manipulations
Eight weeks after lesions were produced, hamsters either remained in LDs (14L:10D) or were transferred to SDs (8L:16D), with dark onset held constant between the 2 photoperiods. Activity recordings began prior to surgery to assess within-hamster surgical effects and continued for ≥26 weeks to evaluate LMA during the interval of neuroendocrine refractoriness to SDs.
Activity monitoring
LMA was monitored continuously throughout the experiment with passive infrared detectors as in Experiment 1.
Surgical manipulations
Hamsters were castrated after 10 weeks of LD or SD treatment. LMA recording continued for an additional 25 weeks to assess the relative contribution of steroid-independent versus steroid-dependent effects on activity profiles in the 2 photoperiods, before and after hamsters became refractory to short day lengths. Similar to Experiment 1, hamsters in Experiment 2 received a 4-mm T capsule on week 13, and the capsule was removed on week 15.
Functional assessment of DMN lesions
Post hoc group assignment was determined by paired testes weight and quantitative histological assessment of lesions. All SD-Sham hamsters exhibited gonadal regression (paired testis weight = 0.8 ± 0.1 g). Hamsters in the SD-DMNx group with lesions that impinged on the paraventricular nucleus (PVN) were excluded from subsequent analysis. Of 15 SD-DMNx designated hamsters without damage to the PVN, the lesion blocked testicular regression in 5 hamsters—only data from these individuals are considered here. Final group sample sizes were SD-DMNx (n = 5), SD-Sham (n = 20), LD-DMNx (n = 14), and LD-Sham (n = 7).
Data reduction
LMA data were condensed into 10-min bins for quantitative evaluation. To assess the effects of the lesions, 3-week pre- and postsurgery blocks were defined as ending 5 days before surgery and beginning 13 days after surgery, respectively. To assess the effects of photoperiod and lesions on LMA, mean daily (light phase + dark phase) activity counts were quantified for baseline (1-14 days prior to the LD-SD transition), week 2 (days 8-21 after transition), week 4 (days 22-35), week 6 (days 36-49), and week 8 (days 50-63). Mean daily activity counts also were compiled for a postcastration interval (6-15 days after castration, and before T implantation), a T implant interval (23-32 days after castration, 8-17 days with the implant), and 2 longer term postcastration points, one at 8 weeks after castration (18 weeks after SD onset) and one at 16 weeks after castration (26 weeks after SD onset). All hamsters in SDs are presumed to be refractory after 26 weeks of treatment (e.g., Bae et al., 1999; Reiter 1972; Piekarski et al., 2012).
Results
Experiment 1
Histology
Effective lesions extended from no more than 400 µm posterior of the termination of the PVN, bordering the dorsal termination of the VMN, and encompassed at least 75% of the DMN, centered around the third ventricle, and extended laterally from 75% through the VMN to slightly lateral of the termination of the VMN (illustrated in Jarjisian et al., 2013).
Effects of DMN lesions on photoperiod-induced changes in LMA
Activity data from 1 SD-Sham hamster that failed to exhibit gonadal regression and 9 hamsters with ineffective lesions that exhibited gonadal regression in SDs were excluded from subsequent analyses. Data from 3 of the SD-DMNx hamsters with effective lesions were excluded because they failed to exhibit decreased PRL responses in SDs, indicative of lesion-induced disrupted pineal function (cf. Maywood and Hastings, 1995; Lewis et al., 2002). Final sample sizes were LD-Sham, n = 10; LD-DMNx, n = 11; SD-Sham, n = 11; and SD-DMNx, n = 9.
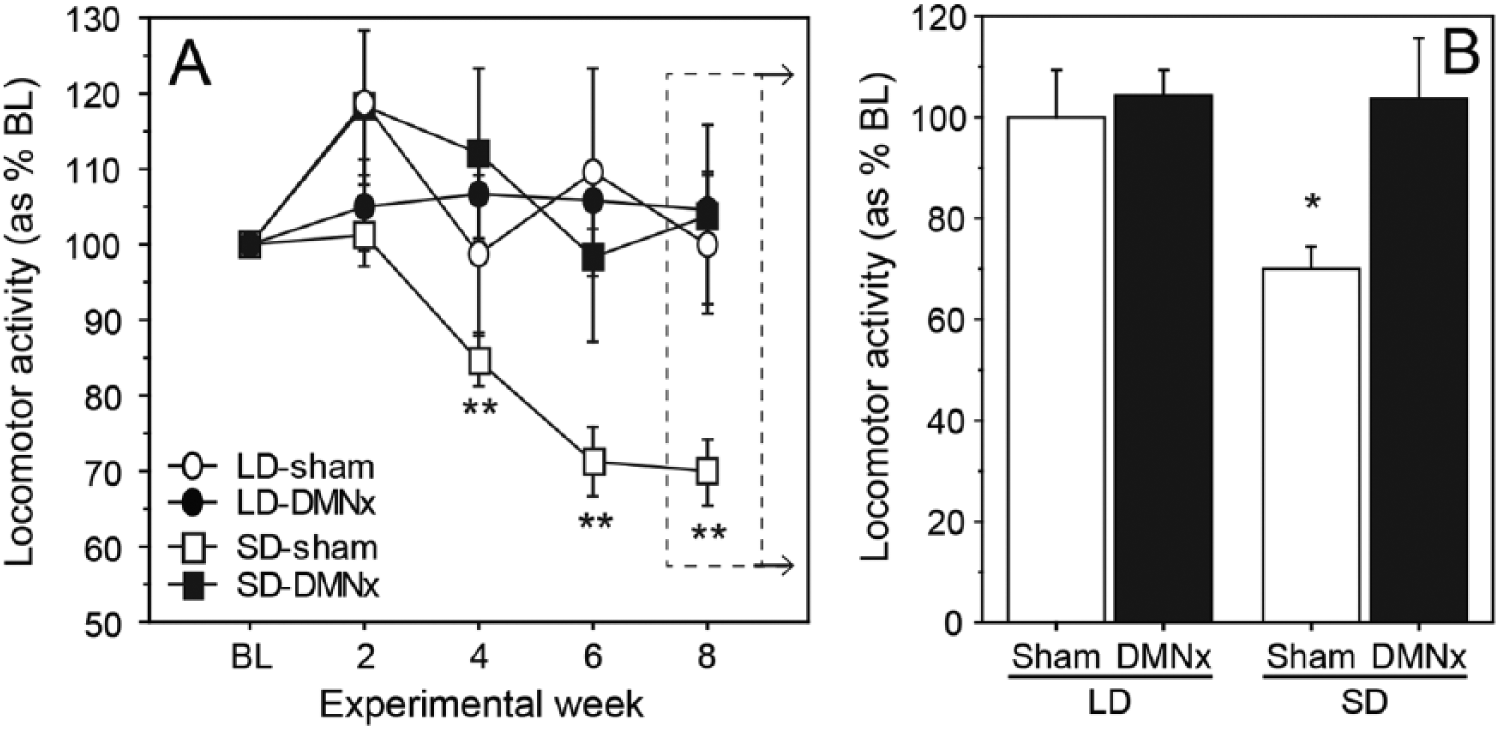
Mean daily activity levels were comparable across groups in LDs prior to photoperiod interventions (Baseline: F3,37 = 0.45, p > 0.70; p > 0.30, all pairwise comparisons). Photoperiod and lesion treatments interacted to affect LMA (repeated-measures ANOVA, photoperiod × lesion: F5,185 = 3.51, p < 0.005; Fig. 2A). LMA did not change between baseline and week 10 in LD hamsters (LD-Sham: t9 = 0.19, p > 0.80; LD-DMNx: t10 = 1.63, p > 0.10, Fig. 2A). Activity decreased by 32% in SD-Sham hamsters (t10 = 3.87, p < 0.01) but did not decrease significantly in the SD-DMNx group (t8 = 1.23, p > 0.20).
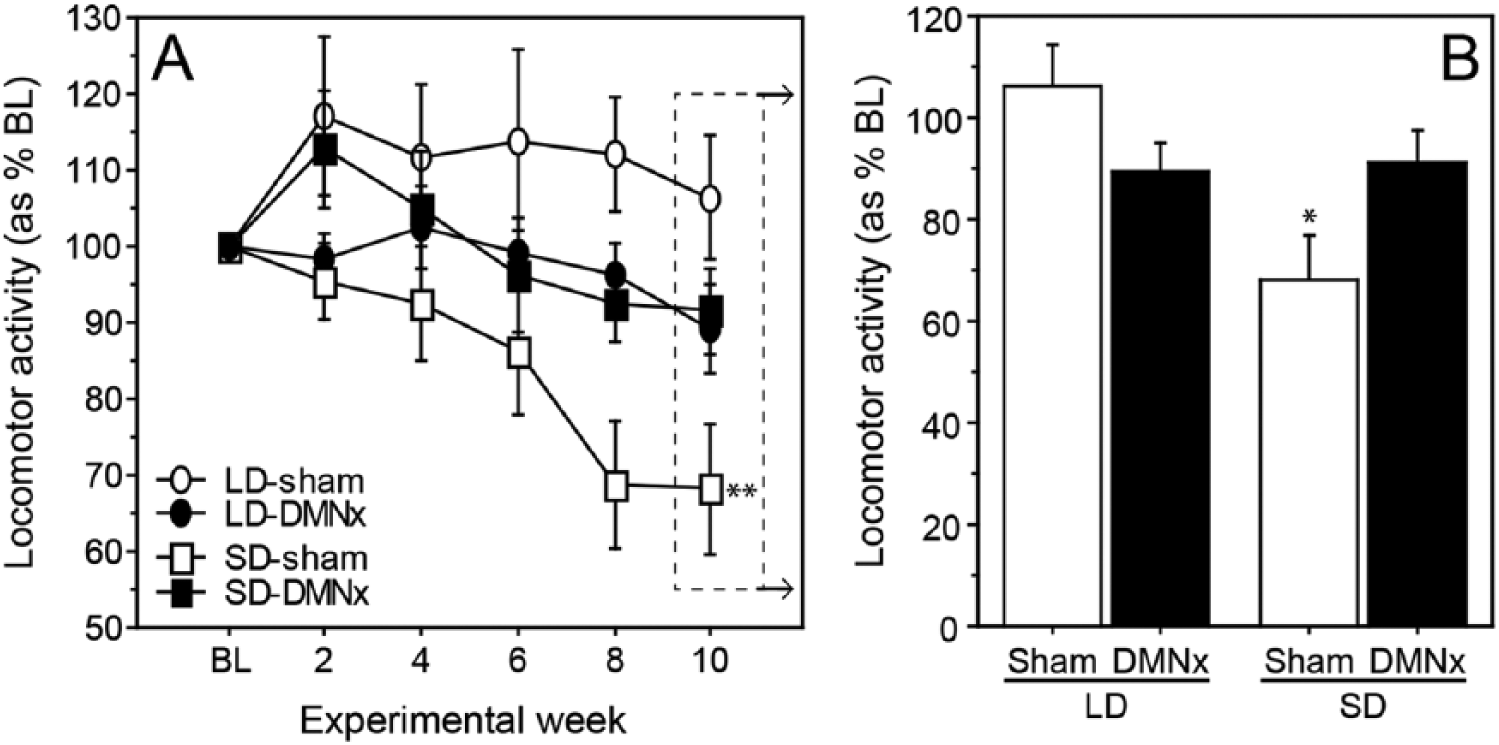
DMN lesions block inhibitory effects of SDs on LMA. (A) Mean ±
Photoperiod and lesion treatments interacted to affect activity levels on week 10 (photoperiod × lesion: F1,37 = 7.62, p < 0.01; Fig. 2B). LMA was significantly lower in the SD-Sham than the LD-Sham group (t19 = 3.21, p < 0.005; Fig. 2B). Photoperiod did not affect LMA in DMNx hamsters (LD-DMNx vs. SD-DMNx: t18 = 0.27, p > 0.75). Activity values of LD-DMNx and SD-DMNx hamsters did not differ significantly from those of LD-Sham hamsters (t17 = 1.72, p > 0.10 and t17 = 1.45, p > 0.15, respectively). Activity levels of SD-Sham hamsters were lower than those of both LD-DMNx (t20 = 2.04, p = 0.055) and SD-DMNx (t18 = 2.16, p < 0.05) groups.
Effects of DMN lesions on testosterone-induced changes in LMA
Responsiveness to T implants was assessed with a repeated-measures ANOVA on total activity counts from week 10 (prior to castration), week 11 after castration but prior to T treatment, and week 13 (during T treatment). There was a significant main effect of photoperiod on the pattern of change in activity over these 3 intervals (F2,74 = 4.35, p < 0.05; Fig. 3). T treatment was predicted to either increase LMA or have no effect, based on earlier studies in which T restored LMA and sexual behavior in male Syrian hamsters (Ellis and Turek, 1983; Morin and Zucker, 1978); therefore, 1-tailed t tests were used in these comparisons. Activity increased in LD-Sham (T treatment vs. week 10: t9 = 3.33, p < 0.005; T treatment vs. Postcastration: t9 = 1.96, p < 0.05; 1-tailed t tests, Fig. 3) and in LD-DMNx (T treatment vs. week 10: t10 = 1.88, p < 0.05; T treatment vs. Postcastration: t10 = 1.85, p < 0.05) but did not increase in SD-Sham (p > 0.05, both comparisons) or SD-DMNx hamsters (p > 0.10, both comparisons; Fig. 3).
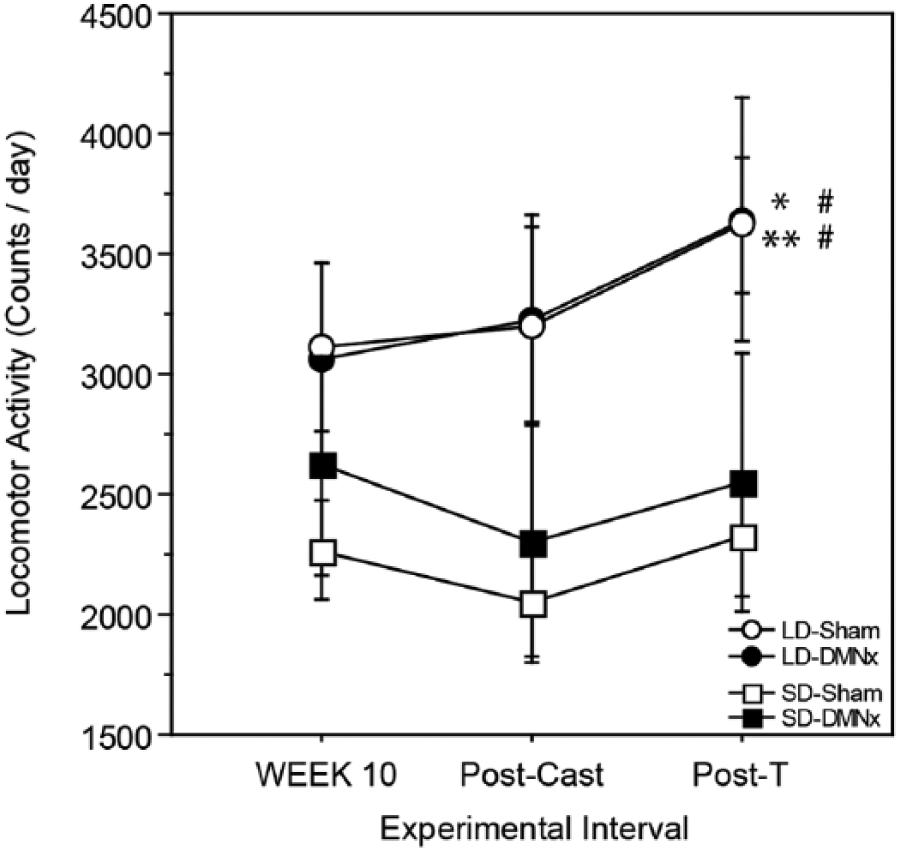
The DMN is not required for SDs to inhibit effects of testosterone on LMA. Mean ±
Experiment 2
Histology
As in Experiment 1, lesions bordered the VMN and encompassed the majority of the DMN (centered at the third ventricle; Fig. 4).
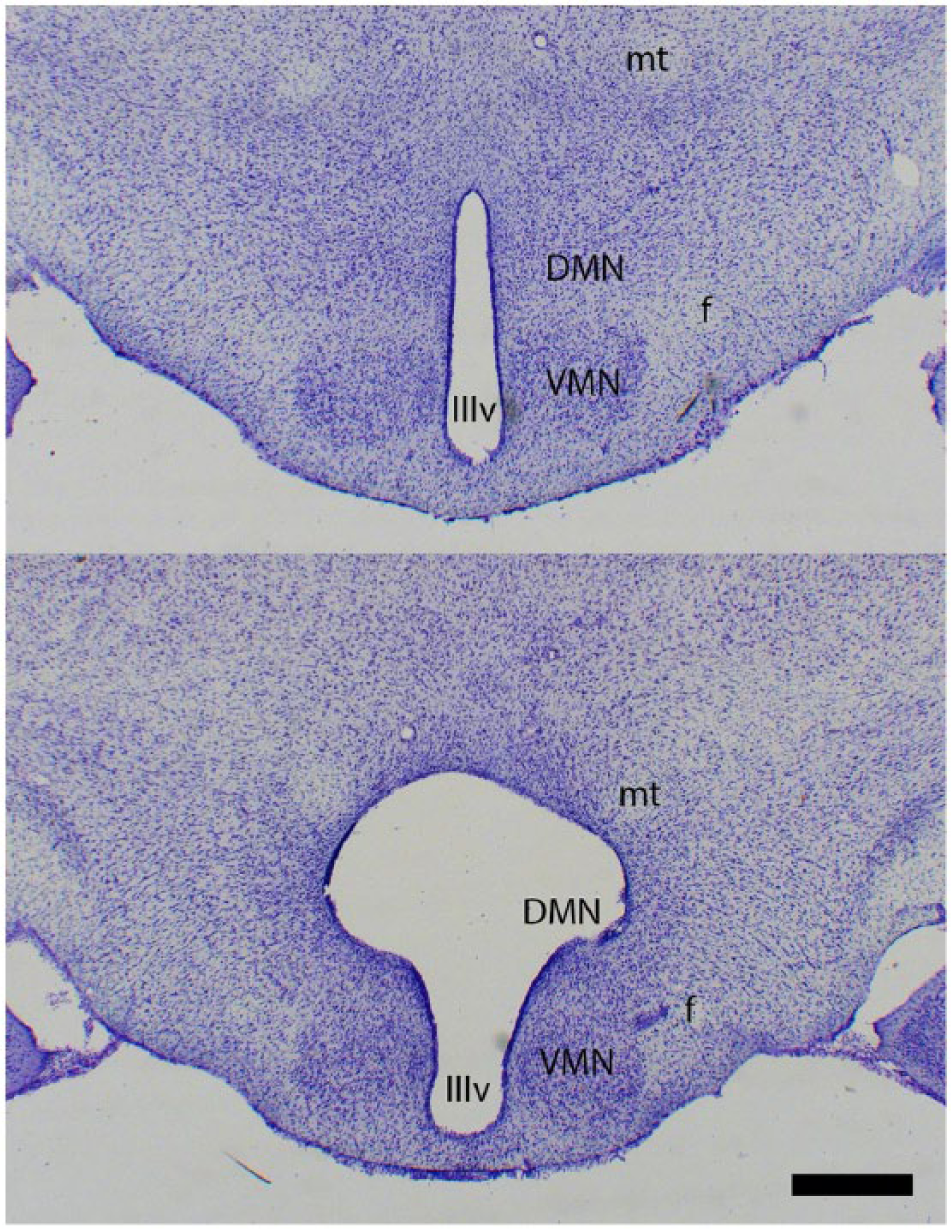
Representative photomicrographs of cresyl-violet-stained coronal sections of an SD-Sham and SD-DMNx hamster showing lesion size and locus. Scale bar = 500 µm. IIIv = third ventricle; DMN = dorsomedial nucleus of the hypothalamus; f = fornix; mt = mammillothalamic tract; VMN = ventromedial nucleus of the hypothalamus.
Acute effects of DMNx surgery on LMA
Prior to photoperiod manipulations, DMN lesions did not alter the amount of LMA. This was true for the set of all DMNx versus sham-operated hamsters (effects of surgery type [DMNx vs. sham], time [pre vs. post lesion], and interactions: all p > 0.50). Additionally, DMNx did not alter mean LMA in the 5 DMNx hamsters with effective lesions that were later transferred to SDs (paired t test, p = 0.70).
Effects of DMN lesions on photoperiod-induced changes in LMA
As in Experiment 1, the amount of activity gradually decreased over the first 8 weeks of SD treatment in sham-lesioned hamsters (Fig. 5A). Hypoactivity was prevented by DMN lesions (SD-DMNx) that also blocked testicular regression. At week 8, SD-Sham hamsters had significantly reduced activity compared with the other 3 groups (p < 0.01, all comparisons; Fig. 5B). Representative actograms for hamsters in each experimental group are shown in Figure 6. During weeks 8 through 14, LMA was reduced and fragmented in the SD-Sham actogram (lower left panel) but not in the SD-DMNx record (lower right panel).
DMN lesions block inhibitory effects of SDs on LMA. (A) Mean ±
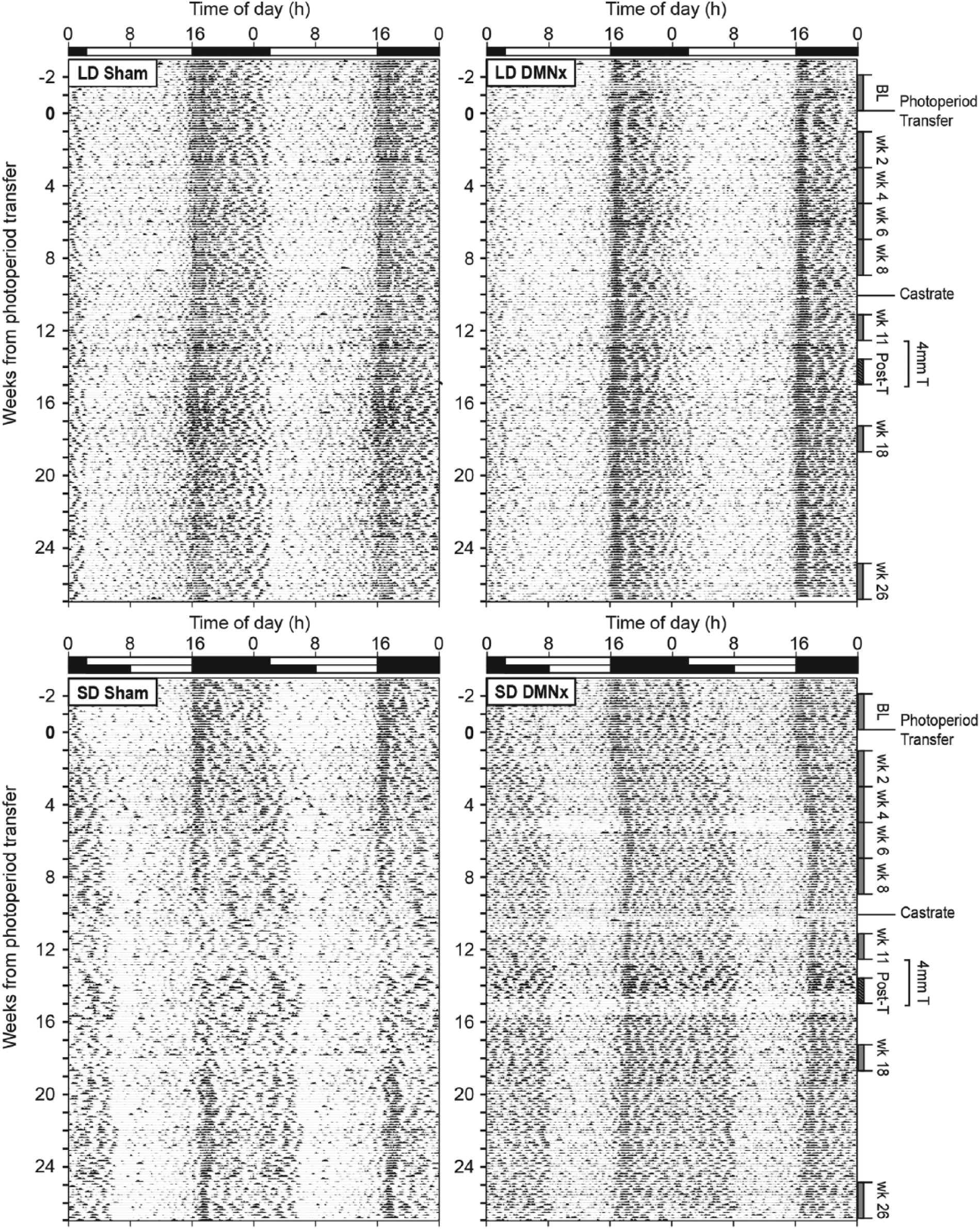
LMA of DMNx and sham-operated hamsters in LDs and SDs. Consecutive days of LMA in 10-min bins double-plotted along each horizontal line. White (light phase) and black (dark phase) bars at the top of each record indicate the prevailing light-dark cycle. Photoperiod transfer occurred at week 0. Data analysis epochs corresponding to Figure 1 are indicated along the right vertical axis. Note the absence of reduced LMA in the SD DMNx actogram (lower right panel) and normal expansion of the active phase in SDs.
Effects of castration on LMA
To evaluate whether maintenance of higher levels of LMA in SD-DMNx hamsters is mediated in part by a steroid-independent mechanism, hamsters were castrated on week 10. By week 12, LMA was reduced compared with the week 8 precastration activity levels in SD-Sham hamsters (Fig. 7A; t19 = 6.8, p < 0.001). There was a nonsignificant reduction in SD-DMNx hamsters (t4 = 2.2, p < 0.10) and no reduction in either LD group (p > 0.80 for both). When plotted as a percentage of original pre-SD baseline values (Fig. 7B), SD-DMNx and both LD groups maintained significantly higher LMA than did the SD-Sham hamsters 2 and 8 weeks after castration (SD-Sham vs. SD-DMNx: t21 = 2.8, p < 0.05 and t21 = 2.9, p < 0.01 for 2 and 8 weeks, respectively; vs. LD-Sham: t23 = 5.4, p < 0.001 and t23 = 7.9, p < 0.001; vs. LD-DMNx: t29 = 8.7, p < 0.001 and t29 = 9.0, p < 0.001).
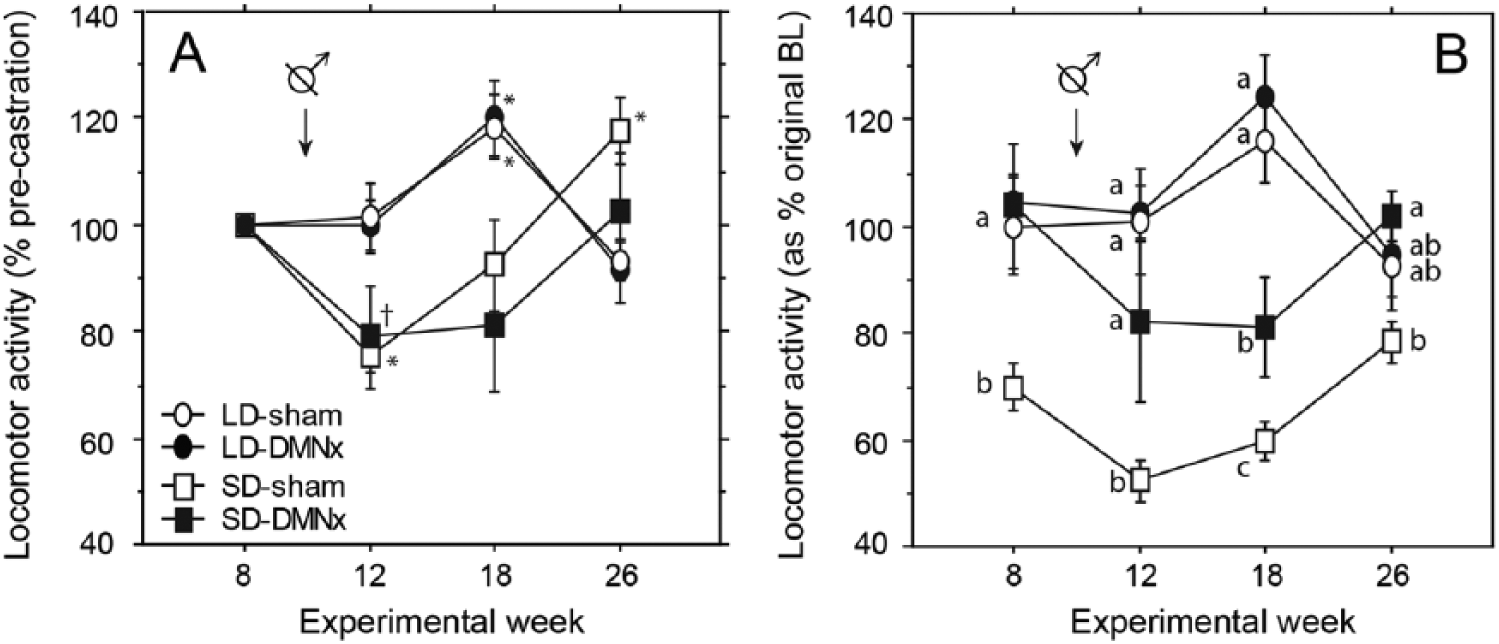
Locomotor responses to castration and after the onset of photorefractoriness to SDs in DMNx and sham-operated hamsters. Mean ±
Effects of DMN lesions on testosterone-induced changes in LMA
From weeks 13 to 15, hamsters in most groups received a 4-mm T-filled or an empty capsule, but all 5 SD-DMNx hamsters received T implants, yielding an unbalanced design; the results were uninterpretable and are not illustrated.
Effects of DMN lesions on refractoriness to short photoperiods
Following removal of blank and T capsules at week 15, assessments on weeks 18 and 26 determined whether DMNx interfered with the development of refractoriness of LMA to SDs. To increase statistical power for this analysis, groups that received blank and T capsules (from weeks 13 to 15) were combined into a single group, as there was no effect of capsule type on subsequent activity at weeks 18 and 26 (repeated-measures ANOVA: capsule effect, F1,41 = 1.0, p = 0.30; interaction of time by capsule type, F1,41 = 1.0, p = 0.30). LMA increased at week 26 in both SD-Sham and SD-DMNx groups (Fig. 7; see also Fig. 6); the emergence of reproductive photorefractoriness to SD was associated with restoration of LMA levels to LD-like precastration values. On week 26, LMA was comparable in SD-Sham and LD-Sham hamsters (t23 = 1.52, p > 0.10) and in SD-DMNx and LD-DMNx hamsters (t16 = 0.38, p > 0.70). The duration of the active phase (alpha) was expanded in SD-DMNx hamsters from weeks 2 to 26 (Fig. 6, bottom right panel).
Discussion
Ablation of the DMN counteracted SD-associated decreases in LMA. It did so without altering the responsiveness of hamsters to exogenous T and without affecting the normal expansion of the active phase (alpha) in SDs. These observations indicate a role for the DMN in the neural circuits governing seasonal regulation of general locomotor activity.
Normal SD-induced decreases in LMA may involve both steroid-independent and steroid-dependent processes (Ellis and Turek, 1983; Turek and Ellis, 1981). Therefore, the relatively higher LMA levels maintained by SD-DMNx hamsters might be due to the lesion interfering with steroid-independent effects or to the elevated concentrations of circulating androgens maintained by their testes. To test the role of gonadal hormones, all hamsters were castrated, and then all Experiment 1 hamsters received capsules designed to yield T concentrations approximating those of LD males (e.g., Arteaga-Silva et al., 2005). After castration, SD-DMNx hamsters maintained greater LMA than their SD-Sham counterparts, indicating that the DMN may mediate steroid-independent seasonal changes in LMA. LMA did decline after castration, significantly in Experiment 2 but not in Experiment 1, but it did so equally in both lesioned and nonlesioned hamsters, suggesting that gonadal hormones contributed similarly to LMA in all gonadally intact SD hamsters. T implants increased activity in LD hamsters but not SD hamsters, but there was no difference attributable to DMNx. It remains possible that the DMN contributes to diminished responsiveness of LMA to T in SD in neurologically intact hamsters, perhaps as part of a distributed neural network with multiple structures independently each becoming less responsive to T in SDs.
A limitation in the current experiment is that hamsters were castrated and then treated with T 2 weeks later. Residual effects of T may sustain LMA at this point; a longer postcastration interval without T may allow the LMA of SD-DMNx hamsters to approach that of the sham-operated controls. The week 18 time point in Experiment 2 established that SD-DMNx hamsters maintain greater LMA than shams at 3 weeks after implants are removed. Future experiments with DMNx castrated hamsters will be necessary to fully determine the role of the DMN in steroid-independent and steroid-dependent regulation of LMA.
Although the DMN is required for the induction of reproductive quiescence and decreased LMA in SDs, it is not essential for all photoperiodic traits. In particular, PRL secretion and increased negative feedback sensitivity of follicle stimulating hormone (FSH) to T in SD hamsters are unaffected (Maywood and Hastings, 1995; Jarjisian et al., 2013). Distinct mechanisms subserve the several traits that comprise the SD phenotype. Note that SDs can both increase the apparent sensitivity to T (when measured by FSH suppression) and decrease the apparent sensitivity to T (when measured by stimulated LMA).
DMNx blocked the decrease in LMA in SDs but did not prevent expansion of the active phase (alpha), characteristic of SD male activity patterns (Larkin et al., 2002). The duration of nightly melatonin secretion remains unchanged in photorefractory hamsters (Rollag et al., 1980), but this does not prevent the recovery of LMA to LD values. Alpha expansion is a pineal-independent effect (Prendergast and Freeman, 1999), directly controlled by the light-dark cycle, and does not require an intact DMN. These data indicate that photoperiod is being transduced appropriately in DMNx hamsters.
After many weeks in SDs, the LD phenotype of reproductive competence emerges despite continued SDs (Reiter, 1972; Watson-Whitmyre and Stetson, 1988). This has been termed photorefractoriness and may be important to ensure that animals are prepared for reproductive activity in advance of spring breeding conditions (Butler et al., 2010). Although lesions of the DMN prevented gonadal regression, SD DMNx hamsters manifested a nonsignificant decrease in LMA that was completely reversed by week 26 (Fig. 7B); this recovery of LD-typical average LMA suggests that refractoriness of LMA in SD-DMNx hamsters occurs at the normal time. Thus, the DMN is not a necessary component in the circuit underlying photorefractoriness. The extent to which melatonin target tissues other than the DMN contribute to seasonal changes in locomotor activity is unknown and merits investigation. For example, would ablation of the paraventricular nucleus of the thalamus prevent the decline of activity in short days? This is of interest because ablation of this nucleus does not interfere with testicular regression in short day lengths (Purvis and Duncan, 1997).
The DMN has been implicated in the control of body mass, food intake, and ponderal index (Bellinger and Bernardis, 2002). Ablation of the dorsomedial hypothalamus (DMN) disinhibited responses of the hypothalamic pituitary adrenal axis of rats to an emotional stressor, evident by higher plasma concentrations of adrenocorticotropic hormone (ACTH) (Ebner et al., 2012). DMN lesions diminished or eliminated circadian food entrained anticipatory activity (FAA), concurrent with increased SCN activity; subsequent SCN ablation restored FAA (Acosta-Galvan et al., 2011). FAA apparently occurs when the DMH inhibits SCN neural activity, suggesting that FAA originates from a neuronal network that involves interactions between the DMH and SCN (Acosta-Galvan et al., 2011). DMNx effects on testosterone-mediated LMA also may involve the SCN, which is an androgen target tissue (Karatsoreos et al., 2007).
Summary
The present study implicates the DMN as a component of the mechanism that mediates reductions in locomotor activity in short day lengths. The maintenance of testicular activity after DMN ablation likely accounts at least in part for this observation. Future studies of castrated DMNx hamsters will be required to address this question. Although the DMN may be one of the structures rendered unresponsive to the activity stimulating actions of testosterone, it is not a necessary component of this mechanism, nor is it a necessary target for the development of neuroendocrine refractoriness to melatonin that mediates recovery to the long day phenotype (Reiter, 1972; Piekarski et al., 2012).
Footnotes
Acknowledgements
The authors thank Betty Hanson for running the PRL assay in the Diagnostic Endocrinology Laboratory of the Animal Health Diagnostic Center at Cornell University and Chris Tuthill and Kimberly Pelz for expert technical assistance. This work was supported by NIH grant NIH R01 HD-050470 and NSF grant IOS-1257638 to L.J.K.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
