Abstract
Circadian oscillators are autonomous molecular rhythms that reside in cells to align whole-organism physiology and behavior to the 24-h day. In flies, as in mammals, the oscillator operates in cells that coexpress CLOCK (CLK) and CYCLE (CYC). Recent work in Drosophila has shown that CLK is unique in its ability to generate heterologous oscillators, indicating that Clk gene expression defines the circadian cell fate. Here, using standard in vitro and in vivo techniques, we show that TWIN-OF-EYELESS (TOY; dPax6) regulates Clk expression in small ventrolateral neurons (s-LNvs) that coordinate sleep-wake cycles. Crucially, toy binds multiple sites at the Clk locus, is expressed independent of CLK-CYC in LNvs, regulates CLK protein levels under optimal photoperiodic conditions, and sets clock-speed during endogenous free-run. Furthermore, TOY is necessary for the onset of Clk expression in LNvs during embryogenesis. We propose that TOY contributes to a transcription complex that functions upstream of the oscillator to promote Clk expression in s-LNvs.
Circadian oscillators in flies and mammals are driven by a core heterodimer consisting of the bHLH-PAS (basic-Helix-Loop-Helix-Per-Arnt-Sim) transcription factors CLK and CYC/Bmal1 (Brain and Muscle Arnt-Like 1; Hardin, 2006; Yu and Hardin, 2006; Glossop, 2011). In Drosophila, the oscillator mechanism functions in diverse cell types, including photoreceptors and discrete groups of central neurons (Plautz et al., 1997; Kaneko et al., 1997; Siwicki et al., 1998; Shafer et al., 2006). In all of these cells, CLK-CYC binds to E-box sequences to activate the transcription of several genes (period; per, timeless; tim, vrille; vri, par-domain-protein-1epsilon; pdp1ϵ) whose encoded proteins feed back to either inhibit the activity of CLK-CYC (PER-TIM) or modulate transcriptional activity from the Clk locus (VRI/PDP1ϵ; Hardin, 2006; Yu and Hardin, 2006; Glossop, 2011). It is the ~24-h molecular rhythm generated by this feedback mechanism that constitutes the core oscillator in clock cells.
Currently, the only factors that have been shown to function as direct regulators of Clk are the VRI-repressor and PDP1ϵ-activator. These basic-zipper proteins compete for the same binding sites, V/P-boxes, at the Clk locus (Cyran et al., 2003). In mature oscillator cells, vri and pdp1ϵ rely on CLK-CYC for their expression. Hence, early theories posited that daily rhythms in Clk transcription are maintained by sequential feedback, with acute VRI-repression yielding to chronic PDP1ϵ activation (Cyran et al., 2003). However, recent work has questioned whether PDP1ϵ is necessary and/or sufficient for Clk expression. First, PDP1ϵ does not induce circadian oscillations in neurons that are transformed by CLK, indicating that PDP1ϵ is not sufficient to activate Clk (Kilman and Allada, 2009). Second, analysis of head mRNA from pdp1ϵ-null, pdp13135 flies has shown that the Clk mRNA rhythm is essentially normal under optimal light-dark cycles (LD12:12; 12 h light: 12 h dark). This suggests that in photoreceptors, the predominant oscillator cell-type in the head, Clk expression does not require PDP1ϵ (Zheng et al., 2009). This also appears to be the case in the brain, since CLK protein is present in central oscillator neurons of pdp13135 mutants (Zheng et al., 2009). Furthermore, a minimal-Clk promoter that lacks canonical V/P sites can still drive expression in most subgroups of central pacemaker cells, including the s-LNvs (Gummadova et al., 2009).
To address this PDP1ϵ enigma, we screened the Clk locus for known consensus binding sites to identify other factors that might activate Clk. One sequence that was present at higher frequency than would be expected by random chance was that of the paired-homeobox-6 (Pax6)–consensus bound by TOY and EY. These Pax6 paralogs are better known for their role in eye development (Punzo et al., 2002). However, both are also expressed in numerous neurons throughout the developing and mature brain. For instance, TOY and EY show extensive overlap in expression among Kenyon cells (Kurusu et al., 2000; Furukubo-Tokunaga et al., 2009). Despite this knowledge, the role of TOY or EY in central neurons and mature photoreceptors is poorly understood. This is due, primarily, to the fact that they regulate transcription networks via complex context-dependent codes and integrated feedback (Callaerts et al., 2001; Punzo et al., 2002; Adachi et al., 2003; Kozmik, 2005; Furukubo-Tokunaga et al., 2009; Kumar, 2009; Blanco et al., 2010).
Using a combination of gel-shift assay, immunocytochemistry, and reporter gene analysis, we show that (1) TOY binds multiple sites in the Clk locus, (2) toy is expressed in all LN oscillator cells of adult brain, (3) TOY-binding sites enhance expression of a minimal-Clk promoter in vivo, (4) toy is expressed upstream of the clock mechanism in mature LNvs, and (5) TOY is necessary for Clk expression in presumptive s-LNvs during embryogenesis. Overexpression of TOY (TOY-OE) increases CLK protein levels in mature LNvs and accelerates the s-LNv oscillator, generating short-period locomotor rhythms in DD that reflect CLK-OE behavior. In contrast, reduction in toy dosage causes long-period locomotor activity that is comparable and additive to the effects of reducing cyc. Taken together, these data suggest that TOY functions directly upstream of Clk in the s-LNvs.
Materials and Methods
Electrophoretic Mobility Shift Assay
TOY and EY were synthesized using the rabbit-reticulocyte system (Promega, Madison, WI). Full-length TOY was generated using pOT2-toy cDNA clone GH14454 from the Drosophila Genomics Resource Center (Bloomington, IN) and full-length EY from pBSIIKS-ey cDNA (P. Callaerts, personal communication, 2012). Lysate protein was incubated in TGKD (10 mM Tris, 50 mM KCl, 4 mM DTT, 4% glycerol [v/v]) for 20 min prior to addition of nonspecific competitor (100 ng/µL salmon testes DNA; Ambion, Grand Island, NY), cold-competitor (10-fold), and radiolabeled probe (10k cpm). All probes contained the 17-bp paired-site and flanking bases (10 bp 5′; 7 bp 3′) with exception of +681/2, which was 5′-gaattcgATTAgCTCATGCAATAATTAATttcac-3′ (sense). CD19-2Ains was 5′-gaaggcggtgGTCA CGCCTCAGTGCCCcattctc-3′ (sense). Probes were made as described in Glossop et al. (2003). For competition assays, DIG-labeled probe was used following the manufacturer’s guidelines (Roche, Indianapolis, IN). Antibodies were added post-probe at 1:20 dilution. Reaction products were resolved at 22 °C on 6% native polyacrylamide gels.
Western Blot
TnT-TOY, TnT-EY, and controls, were run on 8% SDS-polyacrylamide gels, transferred to nitrocellulose (Hybond-ECL; Amersham, Pittsburgh, PA), blocked in milk (140 mM NaCl; 10 mM Tris; 0.05% Tween-20 [v/v]; 0.05% Thimerosol [w/v]; 5% milk powder), and probed for TOY or EY. Antibody combinations were guinea-pig-α-TOY (1:5000) with goat-α-guinea-pig-peroxidase (1:1000; Sigma-Aldrich, St. Louis, MO) and rat-α-EY (1:5000) with goat-α-rat-peroxidase (1:1000; Sigma-Aldrich). ECL detection was used (Amersham). α-TOY and α-EY antibodies were generated against the entire C-terminal domain of TOY and EY, respectively. Hence, the N-term starts 2 amino acids after the KWREE residues of the homeodomain (U. Walldorf, personal communication, 2010).
Drosophila Stocks and Transgenics
For mis-expression, fly lines were w;UAS-Gal4/UAS-toy;pdf-Gal4/UAS-cGFP (TOY-OE); w;UAS-Gal4/+;pdf-Gal4/UAS-cGFP (WT); and w;UAS-Gal4/UAS-toy;+/UAS-cGFP (toy-con) or non-UAS-cGFP equivalents. For gene-dosage experiments, cyc01 and toyPw+[LacZ] were crossed into isogenic w;+;+;+. Further details are in Supplemental Figure S5C. −206Clk:Gal4[TEmut] lines were generated as described previously for −206Clk:Gal4[WT] (Gummadova et al., 2009). Sixteen lines were generated (1 on X; 5 on 2; 10 on 3) and crossed into w;+;+;+ prior to analysis. Transgene origins were as follows: pdf-Gal4 (Renn et al., 1999), UAS-Gal4 (lines 7, 12b, and 22; Hassan et al., 2000), UAS-toy (4 and 14; Czerny et al., 1999), UAS-cGFP (Bloomington, IN), toyPw+[LacZ]/ciD (P. Callaerts and U. Walldorf, personal communication, 2009), and −206Clk:Gal4[WT] (Gummadova et al., 2009). All animals were reared at 25 °C in LD12:12.
Co-immunofluorescence
Brains were processed as described in Gummadova et al. (2009). Primary antibodies were guinea-pig-α-VRI (1:2000; Glossop et al., 2003), guinea-pig-α-CLK (GP50; 1:2000; Houl et al., 2008), guinea-pig-α-TOY and rat-α-EY (1:100; U. Walldorf, personal communication, 2004), rat-α-TIM (UPR41; 1:1000; Shafer et al., 2006), and rabbit-α-βGAL (1:1000; Aviva Systems Biology, San Diego, CA). Secondary antibodies were Cy5-α-guinea-pig, Cy3-α-rat, and [FITC;Cy3;Cy5]-α-rabbit, generated in donkey (1:200; Jackson ImmunoResearch, West Grove, PA). Fluorescence was visualized on a Leica sp5 microscope with the following excitation/[emission] parameters (GFP/FITC, 488/[500-535] nm; Cy3, 543/[567-610] nm; Cy5, 633/[650-750] nm). Images were captured as sequential scans (z-sections; 0.5-1 µm thick) and processed using LAS-AF (Leica, Solms, Germany) or ImageJ (http://rsb.info.nih.gov/ij/) software. For comparison between −206Clk:Gal4[WT] and −206Clk:Gal4[TEmut], all lines were raised and brains dissected in parallel before imaging with a fixed laser setting, with exception of Figure 3C, where the gain was increased for TEmut to reveal cells with basal GFP signal.
Quantification of Protein Levels in Single Neurons
To quantify CLK levels, WT and TOY-OE brains were processed in parallel and imaged using the same laser settings. LNv5 was discernible as a GFP−ve/CLK+ve nucleus among pdf>GFP+ve/CLK+ve LNvs. All LNv nuclei used for quantification were within 15 z-sections of LNv5 (note: minimal bleaching occurred over ±20 z-sections). Values were averaged from 8 WT and 4 TOY-OE brains. For each nucleus, CLK levels were calculated as mean pixel density minus background. For VRI and TIM signal in LNvs, images were collected in parallel using fixed settings and scan number. Channels were converted to grayscale and number of pdf>GFP+ve LNv neurons recorded. VRI and TIM were scored using the GFP channel as a guide, with LNv5 distinguished as a GFP−ve neuron among the GFP+ve LNvs.
Locomotor Activity Rhythms
Locomotor activity of male flies (1-4 days old) was measured using Trikinetics Drosophila activity monitors (Waltham, MA) for 3 to 5 days in LD12:12 followed by constant darkness (DD) at 25 °C. The χ2 periodogram analysis was performed on individual flies for LD or DD, based on 30-min time bins. Animals with power ≥10 and width ≥2 at 95% confidence were deemed rhythmic. Activity plots show normalized data of rhythmic flies, grouped by genotype.
Statistical Analysis
Analysis was 1-way analysis of variance (ANOVA) with Tukey HSD post hoc. Locomotor groups that failed homoscedasticity (p < 0.05; Levene) were analyzed using Welch ANOVA with Games-Howell post hoc. Statistics were done using SPSS software (SPSS Inc, Chicago, IL).
Results
TOY and EY Bind Multiple Sites at the Clock Locus
An initial screen of the Clk locus identified over 20 sequences with high similarity to the paired-domain (PD) consensus bound by vertebrate Pax6 and its fly orthologs TOY and EY (Epstein et al., 1994; Czerny and Busslinger, 1995; Callaerts et al., 1997; Czerny et al., 1999; Punzo et al., 2002; Kozmik, 2005). Electrophoretic mobility shift assays (EMSAs) were used to determine if TOY and/or EY could form protein-DNA complexes with these sequences. A probe based on the Clk+46 sequence showed a clear shifted complex when it was incubated with rabbit-lysate that had been primed to synthesize either TOY or EY (Fig. 1A). In contrast, no shift was observed when the probe was incubated with relevant lysate controls (Fig. 1A). Each shifted-complex migrated identically to a CD19-2-Ains positive control probe (Fig. 1A; Czerny et al., 1999). Mutating 4 bases within the paired site of the Clk+46 probe eliminated the shift produced by either protein, while self-competition with cold competitor reduced both shifted bands >20-fold (Fig. 1A). The TOY-complex was super-shifted by α-TOY antiserum, while the EY-complex disappeared in the presence of α-EY antiserum (Fig. 1A). In contrast, neither antiserum showed cross-reactivity between these protein paralogs (Fig. 1A).
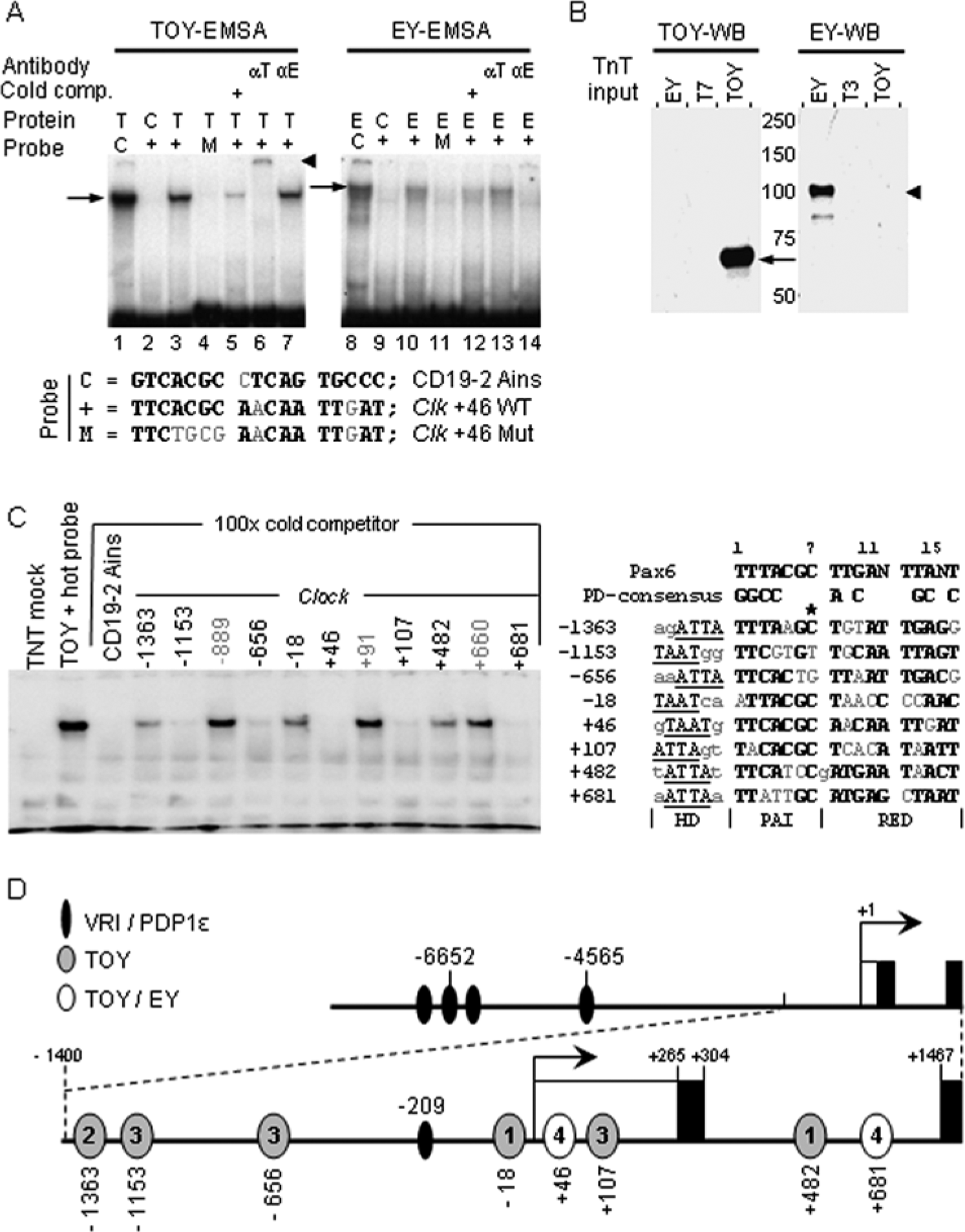
TOY and EY sites in Clk. (A) EMSAs showing TOY (left) and EY (right) bind Clk+46. Probes were wild-type (+), mutant (M), and CD19-2-Ains control (C). Proteins were full-length TOY (T) or EY (E) generated in TnT-rabbit-lysates or unprimed-lysate control (C). Antibodies were anti-TOY (αT) and anti-EY (αE). Cold-competitor was 10-fold excess of Clk+46 (+). Arrows show TOY (left) and EY (right) shifted bands; arrowhead marks TOY super-shift. Experiments were repeated 3 times with similar results. (B) Western blots of protein input for EMSA. Key: TOY, TnT-TOY; T7, lysate control for TnT-TOY; EY, TnT-EY; T3, lysate control for TnT-EY; arrow, full-length TOY; arrowhead, full-length EY. Blots were repeated twice with similar results. (C) Competition assay (left) showing how equimolar concentrations of cold Clk dsDNA compete with labeled CD19-2-Ains for TOY binding. Key: TnT mock, unprimed lysate with labeled CD19-2-Ains; TOY+hot probe, toy-primed lysate with labeled CD19-2-Ains. This experiment was repeated twice with similar results. Sequences (right) show paired-domain (PD) consensus bases in bold and homeo-domain (HD) half-site underlined. Asterisk marks reference base. (D) Schematic of Clk locus showing positions of TOY/EY binding sites relative to transcriptional start site at +1. Numbers in ovals indicate relative affinity for TOY (4 = strongest). Also shown are 100% consensus V/P-binding sites previously identified (Glossop et al., 2003). Key: open boxes, noncoding; filled boxes, coding.
To confirm the presence of TOY/EY in reactions, samples of each lysate were immunoblotted and probed for α-TOY or α-EY immuno-reactivity (IR). The α-TOY antiserum recognized a ~60-kDa protein (predicted = 60 kDa) present in TOY-lysate but absent in control (T7) or EY-lysate (Fig. 1B). The α-EY antiserum recognized a ~100-kDa protein (predicted = 94 kDa) present in EY-lysate and absent in control (T3) or TOY-lysate (Fig. 1B). These data confirm the presence of TOY/EY in EMSA reactions. In addition, the EMSA and Western blot data show that neither antibody cross-reacts with native, or denatured, forms of their respective paralog.
An additional site bound by TOY and EY was identified in intron 1, and 6 further sites (upstream, in 5′UTR and intron 1) were found to be targeted by TOY. The relative affinity of all sites for TOY binding was then determined by their ability to outcompete labeled CD19-2-Ains (Fig. 1C), and an updated map of binding sites in the Clk locus is shown in Figure 1D. In addition to these novel TOY sites, 14 other putative sites were screened and found to have weak or no affinity for TOY (Fig. 1C; Suppl. Fig. S1). TOY contains 3 separate DNA-binding domains, the homeo-domain (HD), and bipartite paired-domain (PD) comprising PAI and RED domains (Kozmik, 2005). Comparison between strong and weak targets suggests TOY only binds PD sites in Clk that also contain a 5′-HD half-site, or ATTA, sequence (Suppl. Fig. S1; Kozmik, 2005). In addition, all poor-affinity sequences contain at least 1 of 3 bases in the PD that is not present in any of the strong targets (Fig. 1C; Suppl. Fig. S1). This suggests these bases disrupt TOY binding and may, instead, facilitate binding by other members of the Pax family. In summary, these data show that TOY can bind a specific subset of Pax6-PD sites at the Clk locus, and EY binds a subset of these sites.
toy Is Expressed in All Lateral Neuron Pacemaker Cells
The presence of TOY/EY binding sites at the Clk locus suggests both proteins regulate Clk expression. In adult brain, TOY and EY are expressed in many neurons that do not contain oscillators (e.g., Kenyon cells), suggesting both may be dedicated repressors of Clk (Kaneko et al., 1997; Adachi et al., 2003; Furukubo-Tokunaga et al., 2009). However, microarray analysis has shown that toy mRNA is enriched in LNvs (Nagoshi et al., 2010; Kula-Eversole et al., 2011). Hence, we wanted to determine if TOY protein is present in the nucleus of these and other central pacemaker neurons at times when the Clk promoter is active: between zeitgeber times (ZT) 16 and 4 ([ZT0-12 = lights on; ZT12-24/0 = lights off]; Kula-Eversole et al., 2011).
Initially, specificity of α-TOY antiserum was confirmed by the co-immunosignal for TOY and β-galactosidase (βGAL) in stage 16-17 embryos (E16-17) containing a toyP[lacZ] transgene (toy>βGAL; U. Walldorf and P. Callaerts, personal communication, 2009). This analysis also confirmed that toyP is a severe hypomorph, or null, insertion (Suppl. Fig. S2A). Next, to determine if TOY is in the nucleus of LNvs at times when Clk is being expressed, flies containing an LNv marker (pdf>cGFP) were entrained to LD cycles and brains of 4-day-old adults processed for α-TOY/α-TIM-IR at ZT16, 22, and 4. TOY was present in the nucleus of all pdf>GFP+ve LNv neurons at all 3 time points (Fig. 2A; Suppl. Fig. S2B). Furthermore, TOY was also evident in GFP−ve LNds at times when TIM-IR made it possible to discern these neurons (Fig. 2A; Suppl. Fig. S2B). Intriguingly, TOY+ve oscillator cells were often distinct from TOY+ve nonoscillator cells by virtue of nuclear foci (Fig. 2A). While this was not investigated further, it seems likely that these foci represent TOY aggregates in transcription complexes or proteasomes. Irrespective, it suggests that TOY may have some unique property in oscillator cell nuclei. In all LNs, TIM was predominantly cytoplasmic at ZT16, nuclear at ZT22, and undetectable at ZT4, indicating normal functionality of the oscillator mechanism (Fig. 2A; Myers et al., 1996; Shafer et al., 2002). These data show that TOY is present in the nucleus of LNs at times when Clk is being transcribed.
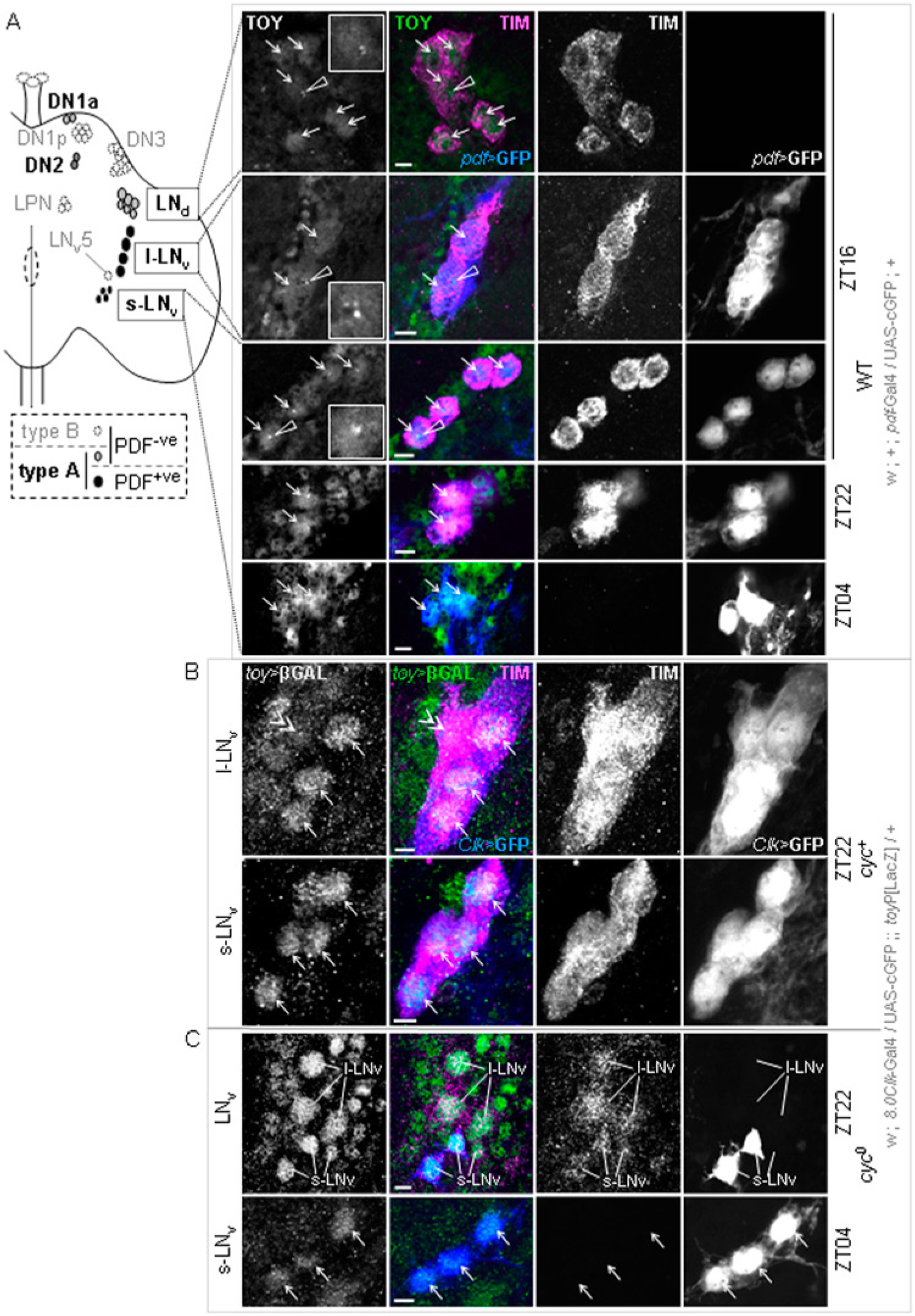
toy expression in LN oscillator cells of adult brain. In all panels, dorsal is up and medial left; scale = 10 µm. (A) Schematic of right hemisphere of a fly brain (left) showing central oscillator subgroups, type A/B division, and PDF+ve cells. Key: LN, lateral neuron; DN, dorsal neuron; v, ventral; d, dorsal; s, small; l, large; 1a, 1 anterior; 1p, 1 posterior; LNv5, PDF−ve LNv; LPN, lateral posterior neuron. Confocal images (right) show TOY-IR in LNs marked with TIM and/or pdf>cGFP at indicated zeitgeber times (ZT). Key: arrows, TOY+ve LN nuclei; open arrowheads and insets, nuclear foci. Images are compressed stacks of 0.5-µm z-sections through the LNs; n > 10 hemispheres from 6 brains. (B, C) Confocal images of LNs in toyP[LacZ]/+ adults showing toy>βGAL, TIM, and 8.0Clk>cGFP in cyc+ (B) and cyc0 (C) backgrounds at indicated ZT. Images are compressed stacks of 0.5-µm z-sections; n > 8 hemispheres from 6 brains. Arrows show toy>βGAL signal in TIM+ve and/or Clk>cGFP+ve LNs. Double arrowhead denotes LNv5. Additional images in Supplemental Figure S2.
The spatial complement of TOY+ve LNs was verified using toyP, since this transgene marks the nuclei of toy-expressing cells with βGAL. Flies containing toyP and an 8.0Clk>cGFP reporter were entrained to LD and 6-day-old adults processed for βGAL-/TIM-IR (Glossop et al., 2003). Strong toy>βGAL-IR was evident in all s-LNv and l-LNv neurons (Fig. 2B; negative control for βGAL-IR in Suppl. Fig. S2C). toy>βGAL was also evident in LNds and the solitary PDF−ve LNv neuron, LNv5 (Fig. 2B; Suppl. Fig. S2D). However, in these neurons, the signal was qualitatively weaker than in PDF+ve LNvs. For LNvs, toy expression occurs independent of the circadian mechanism, since toy>βGAL-IR was evident even in a cyc0 mutant background (Fig. 2C; discussed below). These results confirm that toy is expressed in s-LNv and l-LNv clusters and, to a lesser extent, LNds and LNv5.
TOY Functions Independently of EY in Lateral Neuron Pacemaker Cells
EMSA analysis indicated that EY can bind the Clk locus, raising the possibility that EY may regulate Clk in LNs. To determine if EY is present in these neurons, pdf>cGFP adults were processed for EY- and CLK-IR at ZT16, 22, and 4 (Houl et al., 2008). Using the Kenyon cells as an internal control for EY-IR (not shown), we were unable to detect EY in any central oscillator neurons, including s-LNvs (Suppl. Fig. S2E).
TOY Sites Enhance Expression of a Minimal-Clk Promoter In Vivo
The −206Clk:Gal4 transgene, comprising Clk[−206 to +1485], contains several TOY-binding sites and drives expression in type A oscillator cells, which includes all s-LNv, l-LNv, LNd, DN1a, and DN2 neurons (Fig. 2A; Gummadova et al., 2009). To determine if the TOY sites contribute to this expression, the highest-affinity sites were mutated. EMSA was first used to identify minimal mutations that disrupted TOY binding (Fig. 3A). These mutations were then incorporated into −206Clk:Gal4[WT] to produce −206Clk:Gal4[TEmut], and 16 independent transgenic lines were generated. Each line was crossed to UAS-cGFP, and a preliminary screen of 3-day-old females in LD (and LL3; constant light from eclosion, to minimize circadian fluctuation) showed a complete lack of Clk>GFP fluorescence in 11 lines and weak, but consistent, signal in 5 lines (1 on chromosome X; 4 on c2; n > 4 brains per line, per condition). The few positive lines and controls were rescreened as 1-day-old females at ZT7 (n > 5 brains per line). In all TEmut lines, weak GFP signal was evident in 2 cells in the region of the s-LNvs. In one exceptional line, 16/2, weaker signal was occasionally evident in 2 additional cells in this region (Fig. 3B; Suppl. Fig. S3). Weak signal was also evident in solitary cells in the region of the LNd and DN2 clusters but was inconsistent even between hemispheres of the same brain (Fig. 3B; Suppl. Fig. S3). As expected, control lines showed robust signal in all type A oscillator cells (Fig. 3B).
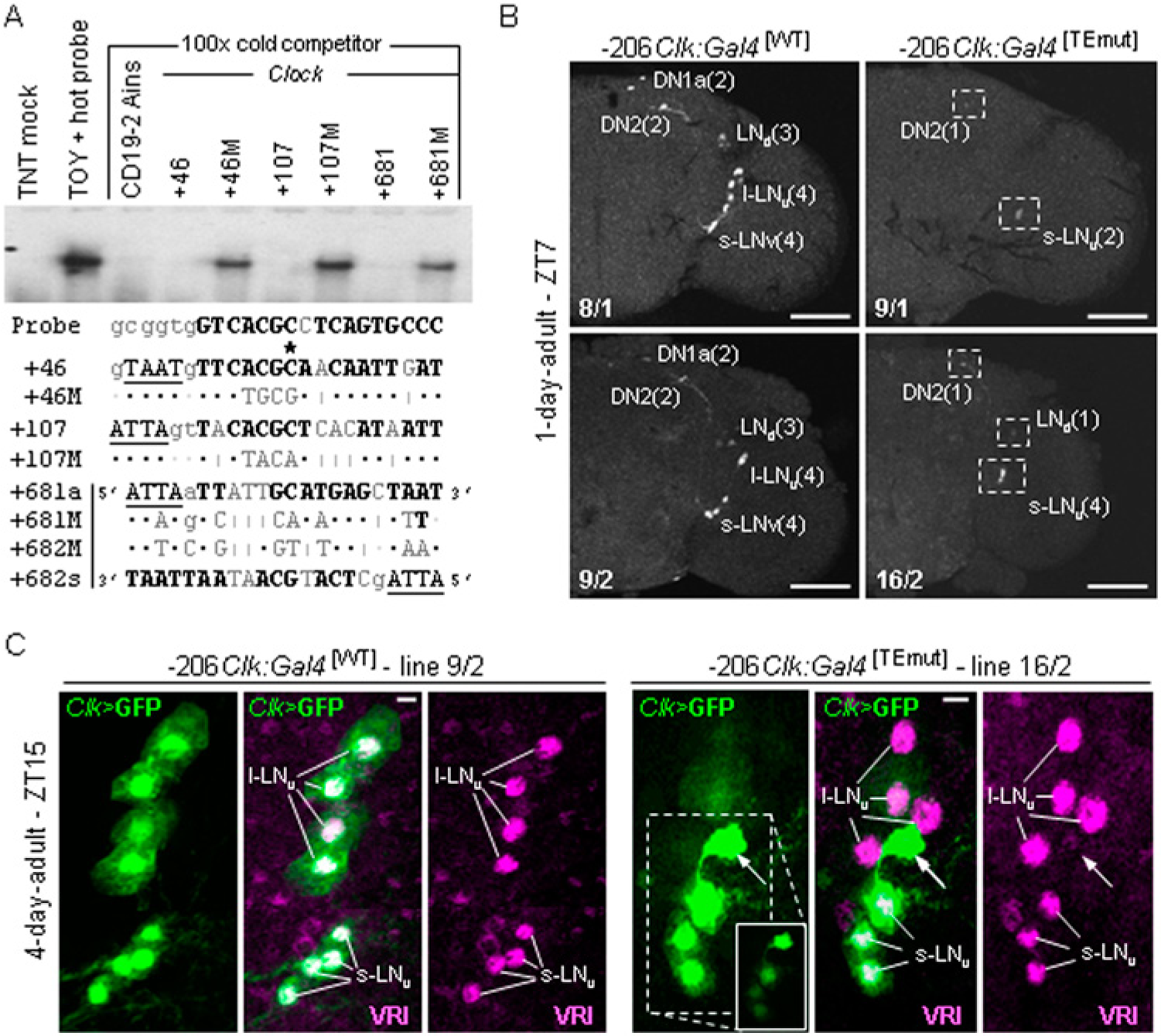
TOY sites enhance expression of the −206Clk:Gal4, minimal-Clk promoter. (A) EMSA showing mutations that disrupt TOY binding to the Clk [+46; +107; +681] sites. (B) Confocal images showing UAS-cGFP reporter signal in the right hemisphere of WT and TEmut. Two WT (8/1; 9/2) and the strongest TEmut inserts on chromosome X (9/1) and 2 (16/2) are shown. Abbreviations are as in Figure 2, with number of GFP+ve neurons per cluster shown in brackets. Brains were processed in parallel at ZT7 (n = 5 brains/line). Similar results were obtained when processed at ZT2. Scale = 100 µm. (C) Representative images for WT[9/2] and TEmut[16/2] showing Clk>cGFP and VRI-IR in brains processed at ZT15. Arrows show GFP+ve/VRI−ve nonoscillator cell. Inset in TEmut shows GFP signal in s-LNvs when gain is set to same level as WT. Scale = 10 µm.
Previous work has shown that removing the −206/−84 region from −206Clk:Gal4[WT] attenuates expression in all type A clock cells of adults. Surprisingly, weak signal in the region of the s-LNvs was due to ectopic expression (Gummadova et al., 2009). To determine if −206Clk:Gal4[TEmut], likewise, drives ectopic expression, TEmut[16/2] and WT[9/2] were crossed to an independent UAS-cGFP reporter and brains of 4-day-old females processed at ZT15 using VRI-IR to mark oscillator cell nuclei (n > 4 brains per line; Glossop et al., 2003). In TEmut, GFP was evident in 2 cells in the region of the s-LNvs. However, only one of these was an oscillator cell as the other was VRI−ve (Fig. 3C). Increasing the gain revealed 2 further GFP+ve/VRI+ve s-LNv clock cells and 1 (of 4) l-LNv neurons (Fig. 3C). In addition, faint GFP signal was detected in the DN1a pair and solitary neurons of the LNd and DN2 clusters. However, the gain level required to visualize these fainter GFP+ve cells also revealed ectopic expression scattered throughout the brain. This was in marked contrast to WT, which showed robust signal in all type A neurons (Fig. 3C).
Taken together, these data show that activity of the TEmut transgene is severely crippled in 16 independent reporter lines. The fact that a few lines show expression demonstrates that TEmut is only able to drive weak expression even when the insert is in a region of accessible chromatin. This indicates that 1 or more of the TE sites is necessary to enhance expression of the minimal-Clk promoter.
Overexpression of TOY Enhances CLK Protein Levels in the LNvs and Selectively Speeds Up the s-LNv Oscillator to Generate Short-Period Locomotor Rhythms
Attenuated expression of the −206Clk:Gal4[TEmut] transgene suggests that TOY enhances Clk expression in type A oscillator cells. To determine if TOY promotes expression from the genomic Clk locus, we turned to overexpression in the context of LNvs. Initially, overexpression was confirmed by driving UAS-toy in all oscillator cells and comparing against controls on Western blot (Suppl. Fig. S4A). Subsequently, TOY was overexpressed specifically in the LNvs using the pdf-Gal4 driver, hereafter referred to as pdf>TOY or TOY-OE (Renn et al., 1999). These flies also contained a UAS-cGFP reporter to co-mark TOY-OE cells and facilitate their morphological analysis. As expected, pdf>GFP was restricted to LNvs, which showed enhanced TOY-IR compared to all other TOY+ve neurons in the brain. Importantly, TOY-OE did not affect gross morphology of the LNvs (Suppl. Fig. S4B). Additionally, since TOY can activate ey (Czerny et al., 1999) and EY can regulate Clk (Fig. 1), we also confirmed TOY-OE does not induce ectopic ey (Suppl. Fig. S4B). Next, brains of TOY-OE were processed to determine if CLK protein levels were elevated in pdf>TOY nuclei relative to an internal control neuron: the PDF−ve/CLK+ve oscillator cell LNv5. Brains were processed at ZT3 since this is the time in a standard LD cycle when Clk mRNA levels peak in s-LNvs (Kula-Eversole et al., 2011). CLK protein levels were statistically significantly higher in s-LNv and l-LNv nuclei in TOY-OE compared to control (Fig. 4A; 1-way ANOVA; F3,78 = 29.93, p < 0.001). Hence, elevated levels of TOY lead to increased levels of CLK in both subgroups of LNv neurons.
Output rhythms in locomotor activity are driven by the s-LNvs in DD (Rieger et al., 2006). Previous work has shown that overexpression of Clk leads to ~1-h shortening of locomotor period under DD conditions due to acceleration of the s-LNv oscillator (Kadener et al., 2008). Hence, to confirm that TOY-OE is increasing CLK protein levels in s-LNvs, pdf>TOY flies were tested to see if they had a short-period locomotor rhythm consistent with CLK-OE. Indeed, TOY-OE flies showed wild-type entrainment during LD but a statistically significant 0.5- to 1-h shortened period in DD compared to controls (Table 1; Suppl. Figs. S4C-D). Locomotor activity plots confirmed the daily advance in morning and evening activity peaks (Fig. 4B; Suppl. Fig. S4C). Hence, overexpressing TOY in s-LNvs generates a CLK-OE behavioral phenocopy. Further-more, the persistence of the short-period rhythm in DD demonstrates that toy interacts with the core oscillator mechanism in the s-LNvs even after several days of constant darkness.
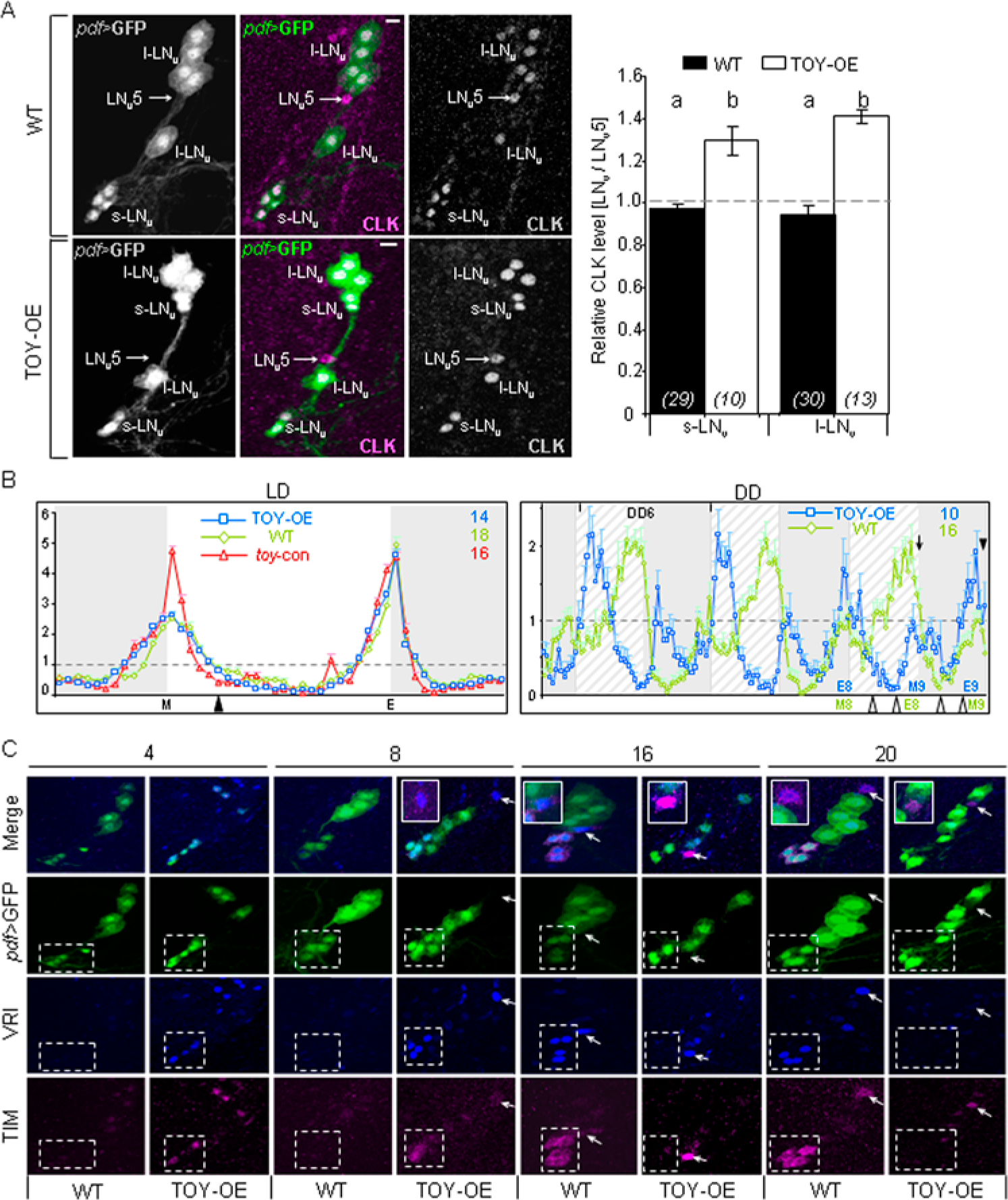
Overexpression of TOY increases CLK protein levels and accelerates the s-LNv molecular oscillator. (A, C) For hemisphere images, dorsal is up and medial left. (A) Comparison between WT and TOY-OE showing quantitative increase of CLK levels in TOY-OE neurons (s-LNv and l-LNv) compared to a PDF−ve/LNv5 internal control neuron (arrows). Images for WT and TOY-OE are shown (left) with quantitative analysis of CLK levels in LNv neurons relative to LNv5 (right). Key: scale bar = 10 µm; error bars = SEM; a-b (p < 0.001; Tukey HSD). (B) Normalized group locomotor activity plots for TOY-OE, driver control (WT), and responder control (toy-con). LD shows synchronized morning (M) and evening (E) peaks of activity during entrainment and arrowhead denotes time at which samples were taken for A. DD shows advanced morning (M) and evening (E) activity peaks for TOY-OE flies, leading to ~12-h advance in M9 (arrow; compare with WT; E8) and E9 (solid arrowhead; compare with WT; M9), which now occur during DD8. LD background indicates day (white; ZT0-12) and night (gray; ZT12-24/0); DD background indicates subjective day (hashed; CT0-12) and subjective night (gray; CT12-24/0). x-axes = 30-min time bins; y-axes = average activity per 30 min ± SEM. Numbers in top right show raw activity average per 30 min. Open arrowheads indicate times at which brains were processed for C. toy-con is not shown in DD to make comparison of the anti-phase rhythms for TOY-OE and WT clearer. (C) TOY-OE selectively advances the s-LNv oscillator in DD. Confocal images of WT and TOY-OE brains in the region of the LNvs taken at indicated time points (CT4, 8, 16, 20; open arrowheads in B) during DD8 and processed for VRI-/TIM-IR. Images show compressed z-stacks (0.5 µm). Dashed boxes outline s-LNvs, while arrows and insets indicate LNv5. n > 5 hemispheres from 3 brains per time point. Images for LD are shown in Supplemental Figure S4E.
Overexpression of TOY in LNvs causes short-period locomotor rhythms.
n = number of flies; %R = percent rhythmic through LD and DD; period is mean ± SEM. Data from different transgene combinations are pooled relative to ±UAS-GFP. Statistical significance values for period versus genotype: LD (–GFP:[F2,13.96 = 1.97, p = 0.18]; +GFP[F2,73.70 = 1.50, p = 0.23]) and DD (–GFP:[F2,36 = 53.01, p < 0.001]; +GFP[F2,63.47 = 200.01, p < 0.001]). Groupings in DD are a-b (p < 0.001; Tukey HSD), c-d, c-e, and d-e (p < 0.001; Games-Howell). For representative actograms and power/width values, see Supplemental Figure S4.
To confirm the acceleration of the molecular oscillator in s-LNvs, brains of 13-day-old TOY-OE and control were dissected across LD8 and DD8 and processed for VRI-/TIM-IR. In s-LNvs, the protein rhythms for VRI and TIM persist during DD, so monitoring both proteins provides better temporal resolution of oscillator status. In brief, VRI is nuclear-localized and present between ~ZT/CT9-22 (peak = ZT/CT15; CT = circadian time in DD, based on previous ZT cycle in LD), while TIM accumulates in the cytoplasm between ~ZT/CT15-21 and then enters the nucleus and degrades by ~ZT/CT24 (Glossop et al., 2003; Shafer et al., 2002; N. Glossop, unpublished data). For this analysis, it was necessary to distinguish s-LNvs at all times, so the TOY-OE line containing UAS-cGFP with a ~22.5-h locomotor period was used.
During LD8, VRI and TIM protein rhythms were in phase in s-LNvs of WT and TOY-OE, with both proteins being undetectable at ZT4, 8 and readily detected at ZT16, 20 (Table 2; Suppl. Fig. S4E). By DD8, VRI and TIM profiles were unchanged in WT, whereas in TOY-OE, both proteins were present in subjective day (CT4, 8) and undetectable in the majority of s-LNvs during subjective night (CT16, 20; Table 2; Fig. 4C). Hence, in TOY-OE, the molecular oscillator in s-LNvs is in anti-phase to that of WT, due to a ~12-h advance over 8 days of DD.
Overexpression of TOY selectively accelerates the s-LNv molecular oscillator.
n = number of brain hemispheres scored per time point; ZT = zeitgeber time; CT = circadian time. Columns show number of pdf>GFP+ve cells and whether these were VRI+ve/TIM+ve as indicated on left; boxes highlight peak VRI/TIM signal in s-LNvs (clear) or LNv5 (gray). Images for all 16 data points are in Supplemental Figure S4.
CLK protein levels in LNv5 are wild-type in pdf>TOY flies (Fig. 4A). As such, LNv5 provides an internal control to determine if TOY-OE is causing selective acceleration of s-LNvs. Analysis of VRI and TIM during LD8 showed that LNv5 was in-phase with s-LNvs in WT and TOY-OE (Table 2; Suppl. Fig. S4E). By DD8, LNv5 remained in-phase with s-LNvs in WT, while in TOY-OE, it phase-lagged the s-LNvs by ~4 to 8 h (Table 2; Fig. 4C). Hence, TOY-OE is accelerating the oscillator in s-LNvs even when compared to non-TOY-OE oscillator cells in the same brain.
Reduction in toy and cyc Dosage Causes Additive Lengthening of Locomotor Period
Due to recessive lethality of the toyP transgene and resistance of chromosome 4 to recombination-based mosaic analysis, it is not possible to monitor the effect a toy-null phenotype has on Clk expression in mature pacemaker cells (Furukubo-Tokunaga et al., 2009; Blair, 2003). However, if the toy-Clk interaction has physiological relevance, then a reduction in TOY levels should impair CLK-CYC function, slow down the s-LNv oscillator, and lengthen the locomotor rhythm in a manner consistent with perturbations in Clk or cyc (Allada et al., 1998; Rutila et al., 1998). Initial attempts to reduce TOY levels using pdf>toyRNAi proved inefficient (<20% reduction in LNvs) or lethal in combination with UAS-Dicer. Hence, to reduce TOY levels, a gene-dosage approach was used.
Halving toy copy number had no effect on period length during LD entrainment but lengthened the free-running period of locomotor activity by a modest, yet statistically significant, ~0.4 h compared to background (Table 3; Fig. 5). This was comparable to a ~0.6-h lengthened period when a null copy of cyc was tested in the same isogenic background (Table 3; Fig. 5; Suppl. Fig. S5). More compelling, the effects of toy and cyc were additive, since double heterozygotes had a ~1-h lengthened period (Table 3; Fig. 5; Suppl. Fig. S5). This confirms there is a genetic interaction between toy and cyc in the context of the circadian mechanism and is consistent with TOY promoting CLK-function in pacemaker cells that regulate locomotor activity. Furthermore, this shows that physiological levels of TOY have rate-determining effects on the oscillator mechanism during DD.
Reduction in toy and cyc dosage cause additive lengthening of locomotor period.
n = number of flies; %R = percent rhythmic; period is mean ± SEM. Statistical significance values for period versus genotype: LD 1-way ANOVA [F3,109 = 1.38, p = 0.25]; DD 1-way ANOVA [F3,109 = 44.33, p < 0.001]. Groupings in DD are a-b (p < 0.01), a-c, and b-c (p < 0.001); Tukey HSD. Additional controls in Supplemental Figure S5.
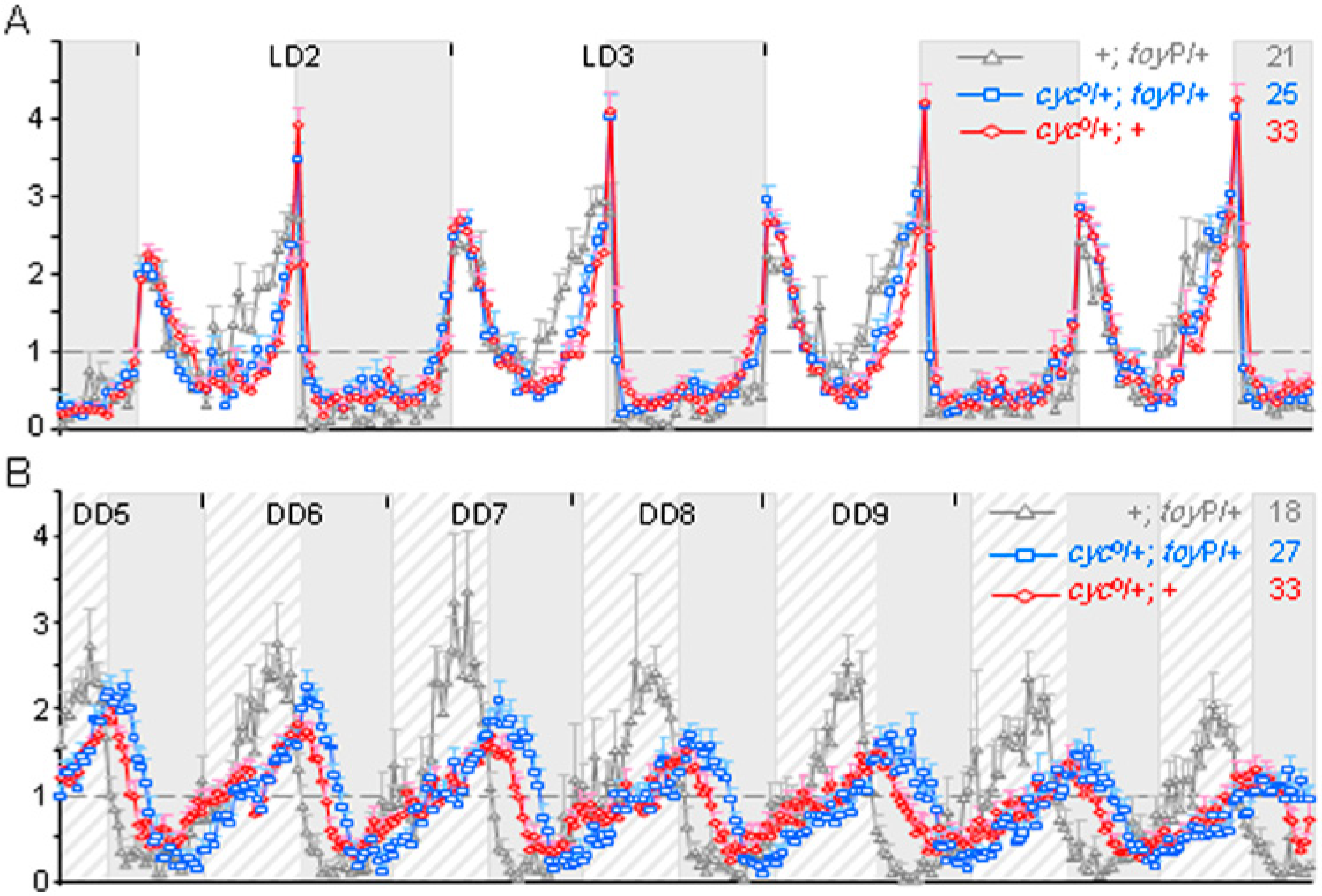
Normalized group locomotor activity plots in LD (A) and DD (B) showing effects of reduced toy and cyc dosage on free-running period. Key: LD and DD background as in Figure 4C; x-axes, 30-min time bins; y-axes, average activity per 30 min ± SEM. Numbers in the top right show raw activity average per 30 min.
TOY Is Necessary for Clk Expression during s-LNv Development
The onset of a functional circadian system occurs during embryonic stage 16 when a handful of neurons first begin to express CLK (Houl et al., 2008). These neurons persist through to adulthood to become the s-LNv and DN1a/2 clusters. Since toyP homozygotes progress to larval stages, it is possible to determine if TOY is necessary for this onset of Clk expression in the s-LNvs.
Analysis of toy>βGAL and CLK in heterozygous wild-type (toyP/ciD) embryos confirmed that toy is expressed in s-LNvs when the Clk promoter first becomes active (Fig. 6A; n = 9 embryos/hemispheres). This analysis also revealed that the DN clusters consist of toy>βGAL+ve and βGAL−ve nuclei at this time (Fig. 6A; arrowheads indicate CLK+ve/toy>βGAL−ve). In stark contrast to wild-type, CLK-IR was only evident in a pair of neurons in the dorsal lobe of toy-null (toyP/toyP) embryos (Fig. 6B; n = 6). One of these CLK+ve neurons was βGAL−ve. Hence, this is one of the DN pairs (Fig. 6B). The lack of CLK-IR elsewhere in the brain shows that the onset of Clk expression in presumptive s-LNvs is severely attenuated in the absence of TOY.
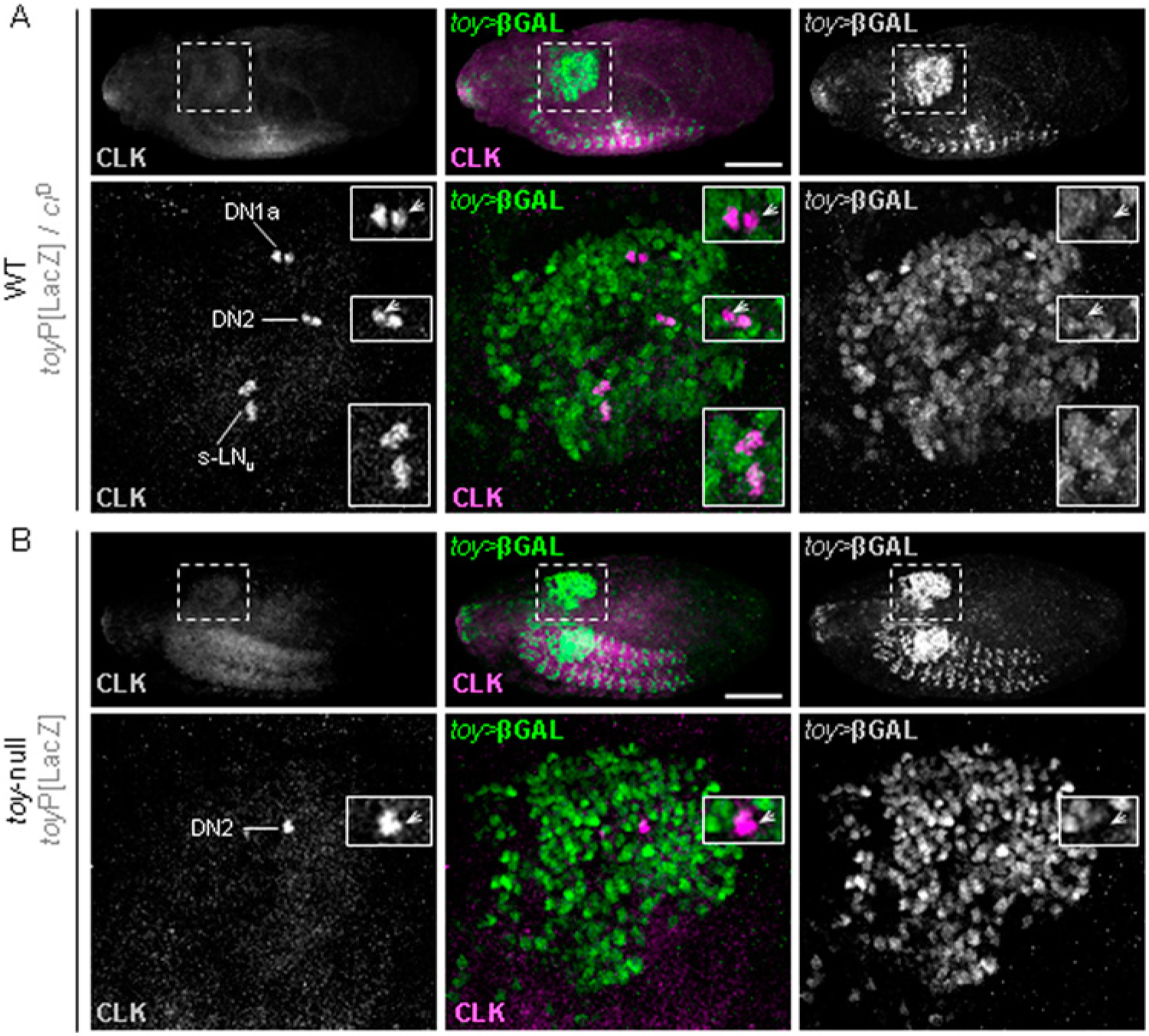
TOY is necessary for the onset of Clk expression in s-LNvs. Images show stage 17 embryos (E17) processed for CLK- and toy>βGAL-IR. Upper panels show embryos and dashed box indicates optic lobe shown in detail in lower panels. Scale = 100 µm. (A) Heterozygous wild-type (toyP/ciD) showing CLK-IR in oscillator neurons and co-IR with toy>βGAL in s-LNvs (bottom inset). DN pairs contain βGAL−ve nuclei (upper insets; arrowheads). (B) toy-null (toyP homozygote) showing CLK-IR restricted to a pair of DNs, one of which is βGAL−ve (arrowhead).
toy Is Expressed Upstream of CLK-CYC in Mature LNvs
Current knowledge of the oscillator mechanism is restricted to regulation that occurs downstream of CLK-CYC. Since TOY is necessary for Clk expression in presumptive s-LNvs, it seems likely this regulation normally persists to maintain the oscillator. If true, this would place TOY in a unique position with regard to oscillator function, as it would provide input rather than PDP1ϵ-like feedback. To address this, the core oscillator was knocked out by crossing the cyc0 mutation into flies containing the 8.0Clk>cGFP and toy>βGAL reporters (Rutila et al., 1998; Glossop et al., 2003). Despite the lack of CLK-CYC in cyc-null flies, basal levels of TIM are still detectable in LNvs toward the end of the night. Hence, TIM-IR was used in conjunction with Clk>cGFP as a dual marker of LNv neurons.
Analysis of adult brains at ZT22 and ZT4 did not reveal any gross difference in the spatial pattern of toy>βGAL+ve cells in cyc0;toyP/+. The level of 8.0Clk>cGFP was predictably lower than in wild-type. Nevertheless, toy>βGAL co-localized with TIM and/or Clk>cGFP in 3 s-LNv and 3 l-LNv nuclei (Fig. 2C). Hence, toy expression persists in LNvs in the absence of functional CLK-CYC. This suggests that the role of TOY, in relation to Clk, is one of input rather than feedback maintenance.
Discussion
Novel TOY- and EY-Binding Sites in the Clk Promoter
Our analysis has revealed that TOY binds multiple sites in the Clk locus and that EY binds a subset of these sites. Previous work has shown that TOY binds PD sites at the eyeless (ey) and sine oculis (so) loci, while EY binds a subset of the TOY sites in so (Halder et al., 1998; Punzo et al., 2002). A common feature of the binding sites at the Clk locus is that they all contain a 5′-HD half-site. This suggests TOY (and EY) bind in a different configuration at the Clk locus to that at ey and so. While it is known that DNA-binding configuration can change the functional properties of Pax factors, it is currently not known why this is (Jun and Desplan, 1996; Yamaguchi et al., 1997; Punzo et al., 2001; Kozmik, 2005). This mechanistic nuance can now be tested in vivo by comparing the regulatory complex(es) TOY forms at the Clk locus with those at ey and so.
In addition to the TOY sites, we identified PD sequences in Clk that are poor targets for TOY. This has revealed 3 bases in the paired box that correlate with poor binding. We suspect these sites are targeted by different Pax factors and, hence, these 3 bases represent key nucleotides for Pax-factor discrimination. Thus, other members of the Pax family may target these sites to regulate Clk expression in TOY−ve cell types.
In the brain, TOY and EY function in parallel and independent regulatory cascades (Kammermeier et al., 2001; Adachi et al., 2003; Furukubo-Tokunaga et al., 2009). There are 4 predicted isoforms of EY, all of which contain a common C-terminal domain. Using a polyclonal antibody generated against the entire C-terminal domain, we did not detect EY in any central clock neurons, indicating that TOY probably functions independently of EY in central pacemaker cells.
TOY Is an Enhancer of Clk in Lateral Neuron Pacemaker Cells
Using standard approaches, we have shown that TOY is a positive regulator of Clk in pacemaker neurons that coordinate daily sleep-wake cycles. The −206Clk[TEmut] analysis shows that TOY sites enhance expression in type A oscillator neurons, including the s-LNvs (Fig. 7A). Importantly, two-thirds of lines containing this reporter showed a complete lack of Clk>GFP fluorescence in adult brain, while a quarter showed weak signal in 1 s-LNv neuron (note: a second putative GFP+ve s-LNv neuron was VRI−ve and, thus, ectopic). In one exceptional line, 16/2, Clk>GFP was evident in additional type A neurons. However, even in this line, expression was weaker than in −206Clk[WT] controls. Critically, the −206Clk[TEmut] transgene still contains low-affinity TOY sites. Hence, even this residual expression may be dependent on TOY.
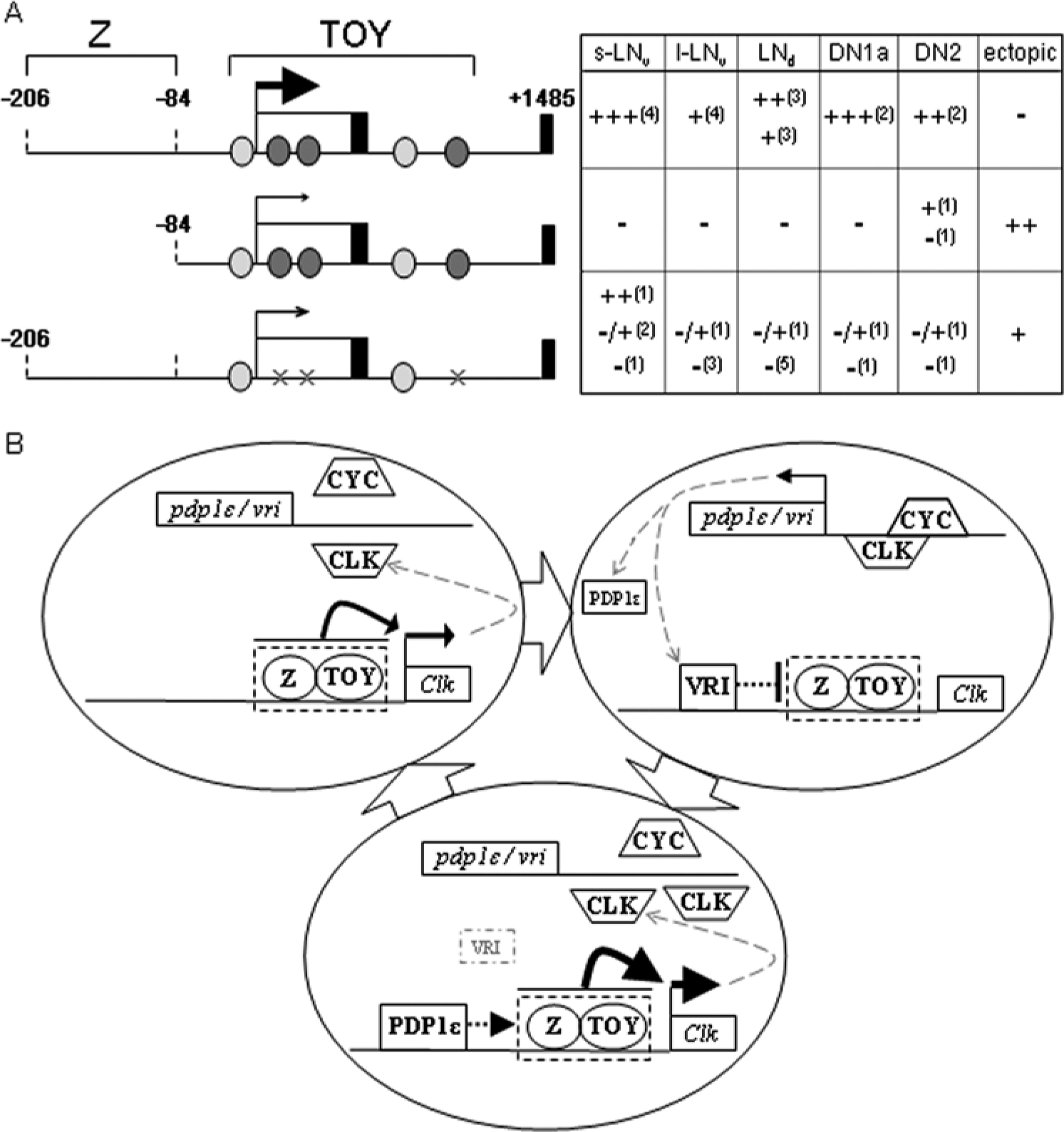
(A) Schematic showing key regions of the Clk promoter that lack canonical VRI/PDP1ϵ sites but support expression in type A oscillator cells. The −206/+1485 region drives expression in type A neurons due to additive, or cooperative, interaction between TOY and unknown factor(s) that bind the −206/−84 region (Gummadova et al., 2009). (B) Model showing Clk gene expression in s-LNvs. Circles denote an s-LNv nucleus with block arrows indicating progressive steps. Clk expression is driven by TOY and factor Z. This mechanism probably initiates expression and explains why Clk is expressed in a CLK-CYC- or PDP1ϵ-null background (upper left). In wild-type, CLK-CYC activates vri and pdp1ϵ. VRI then binds V/P sites to suppress Clk expression, either by inhibiting TOY-Z or by independent suppression of the basal promoter machinery (upper right). VRI suppression of Clk leads to falling levels of CLK-CYC and subsequent decline in VRI. This allows PDP1ϵ to bind V/P boxes and enhance Clk expression, either via TOY-Z or an alternative mechanism (lower). Key: solid arrows, activation; dashed arrow, possible mode of enhancement; dashed blunt-ended line, possible mode of suppression; gray arrows, transcription/translation.
Focusing on the LNvs, the overexpression data show that TOY enhances CLK levels, demonstrating relevance of the toy>Clk link in the context of genomic Clk gene expression. Furthermore, the toy manipulation data show that physiological levels of TOY interact with the core oscillator mechanism in s-LNvs, with rate-determining effects during DD. This contrasts with results from pdp1ϵ manipulation, which has minimal effect on the free-running period of locomotor activity (Benito et al., 2007; Zheng et al., 2009). Hence, even during constant darkness, TOY appears to be more critical than PDP1ϵ when it comes to setting clock-speed in these neurons.
Naturally, the most direct way to show the necessity of TOY for Clk gene expression would be the complete lack of Clk/CLK in toy-null oscillator cells. Unfortunately, such analysis is not possible in adult. However, we have shown that toy is expressed in presumptive s-LNvs at the time CLK protein first becomes detectable and that CLK is undetectable in these neurons in toy-null embryos. Hence, TOY appears to be necessary for the onset of Clk expression in the s-LNvs. The loss of TOY also affects Clk expression in some, but not all, of the early-born DNs. This conflicts with the −206 Clk[TEmut] data, which show attenuated expression in DN1a and DN2 neurons in adult brain. A possible explanation for this is that TOY may be redundant during the onset of Clk expression in certain DN neurons but becomes necessary, later, to ensure clock maintenance. Alternatively, it may be that some other Pax-factor binds the TE site(s) to drive expression in these DNs. Nevertheless, the combined data from embryo and adult suggest that TOY positively regulates Clk expression throughout the life of the s-LNvs.
Regulatory Hierarchy between toy, Clk, and pdp1ϵ in LNvs
One of the main drives behind this research was the discrepancy in the current model of the oscillator mechanism due to mounting evidence that PDP1ϵ is not necessary for Clk expression. Here we have shown that TOY functions upstream of CLK-CYC in mature LNvs. We propose that TOY combines with an unknown factor (Z, which binds the Clk −206/−84 region), and this probably forms the primary mechanism that drives Clk expression in LNvs (Fig. 7A). Hence, TOY+Z generate sufficient levels of Clk for CLK-CYC to activate the core oscillator genes, including pdp1ϵ. PDP1ϵ then feeds back to the Clk locus to help maintain expression via a semi-redundant mechanism based on cooperative, or additive, regulation (Fig. 7B).
So what is factor Z? Previous work has shown that CLK levels are attenuated in the LNvs of 48-related-2 (Fer2)–null adults. However, it was not determined if this is due to a specific loss of Clk expression or an indirect effect (Nagoshi et al., 2010). FER2 is a bHLH factor so presumably targets E-box (CANNTG) sequences as a dimer. The Clk −206/−84 region contains 3 putative E-boxes (e.g., CAACTGCACTTG). While these sites necessarily differ from the canonical circadian E-box bound by CLK-CYC (CACGTG), they may, nevertheless, be targeted by a FER2-containing dimer. Future work will be required to determine the link between Fer2 and toy with regard to Clk expression in the s-LNvs.
Differentiating between TOY+ve Oscillator and Nonoscillator Cells
The majority of TOY+ve neurons in the adult brain do not express Clk and are, therefore, not oscillators cells. For example, Kenyon cells are not oscillator cells. So why doesn’t TOY activate Clk in these neurons? TOY function is highly context dependent (Blanco et al., 2010). Hence, TOY may only activate Clk in oscillator cells because these are the only TOY+ve cells that also contain factor Z. These cofactors may form stable complexes on the Clk locus, which might explain the unique TOY foci evident in clock cells. Irrespective, previous work suggests that repression is also necessary to prevent Clk expression in certain groups of TOY+ve nonoscillator cells. For example, the −1982Clk:Gal4 transgene drives expression in Kenyon cells (Gummadova et al., 2009). This transgene is a 5′-extended version of −206Clk:Gal4 and, therefore, contains additional TOY sites at −1363, −1153, and −656. Thus, in different cell types, TOY may combine with different cofactors to form alternative enhancer complexes at the Clk locus. Hence, in Kenyon cells, TOY may bind sites in the −1982/−207 region to coactivate with factor Y, while in type A clock neurons, TOY binds the −206/+1485 region to coactivate with factor Z. This potential expression in Kenyon cells is blocked by repressors that bind further upstream in the Clk locus (Gummadova et al., 2009). However, in addition to expression in Kenyon cells, the −1982Clk:Gal4 transgene also supports expression in the visual system (Gummadova et al., 2009). Hence, it seems plausible that in photoreceptors and Kenyon cells, a common enhancer complex may form within the Clk −1982/−207 region, but unique repressors block this in the latter cell type to prevent them from becoming oscillator cells. It will now be important to determine if TOY sites in the Clk −1982/−207 region contribute to expression in retinal photoreceptors and if this also requires the Clk −206/+1485 region.
Novel Insights into a Potential Role for Pax6 (mTOY) in the Mammalian Oscillator
The circadian mechanism is highly conserved from flies to mammals. Hence, the core drive for the mammalian oscillator is provided by the homologues of CLK (Clock, or its paralog, Npas2) and CYC (Bmal1). In contrast to flies, mammalian homologues of VRI/PDP1ϵ do not appear to feed back to regulate Clock/Npas2, and it is currently not known what activates either of these mammalian Clock paralogs (Glossop, 2011). It has been shown that Otx2 and Clock function in an interdependent network during neural plate formation (Green et al., 2001; Morgan, 2002). However, it has not yet been shown if Otx2 is a direct regulator of Clock or if such regulation also applies in the context of mature clock neurons. Intriguingly, Clock-Bmal1 can activate Pax6, the mammalian homologue of toy (Morgan, 2004). In light of our data, we propose that Pax6 may also function upstream of Clock. Hence, Pax6 may combine with coactivators to drive the expression of Clock, with Clock-Bmal1 then establishing positive feedback by upregulating Pax6. Hypothetically, such a mechanism may be relevant in master pacemaker cells of the suprachiasmatic nuclei (SCN) that modulate sleep-wake behavior in mammals (DeBruyne et al., 2007). Alternatively, other members of the Pax family may have evolved to fulfill this function in the SCN while Pax6, instead, regulates Clock (or Npas2) expression in other oscillator cell types, such as retinal photoreceptors. Indeed, the link between toy and Clk in flies suggests that visual photoreception evolved from the genetic machinery whose primitive function was to establish circadian molecular timekeeping.
Footnotes
Acknowledgements
Thanks to Patrick Callaerts, Walter Gehring, Amita Sehgal, Bambos Kyriacou, Drosophila Genomics Resource Center, and Bloomington Stock Center (Bloomington, IN) for antibodies, plasmids, and transgenic fly lines; Bambos Kyriacou for use of the fly monitoring facility at the University of Leicester; Peter March and members of the University of Manchester Bio-imaging Facility; Katherine Edwards, Richard Lyon, Vicki Popova, and Patrick Smith for preliminary and supporting data; and University of Manchester–Life Sciences fly facility: established and supported by the University and Wellcome Trust grant 087742/Z/08/Z. Thanks to Tim Brown for his TauActo periodogram analysis software and Andrew Loudon, Hugh Piggins, Rob Lucas, David Sattelle, Owen Jones, Bea Otalora, and Mino Belle for helpful discussions. Special thanks to Uwe Walldorf for his kind gift of the α-TOY/EY antibodies. This work was supported by an NIH grant NS051280 to PH and University of Manchester RSF award and BBSRC grant BB/D016339 to NG.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
