Abstract
Night eating syndrome (NES) is a delayed pattern of energy intake. It is unknown if symptoms associated with this syndrome are accompanied by a delayed pattern of physical activity. This study examines the relationship between physical activity patterns and delayed eating behaviors in children. Children from the QUALITY cohort (n = 269, 45% female, aged 8-11 y) completed the Night Eating Questionnaire (NEQ), which measures NES symptoms on a continuous scale and identifies single NES symptoms. Daily accelerometer data were transformed into mean counts per wear-time minute for each hour of the day. Children with high NEQ scores had higher levels of daily (p < 0.001) and evening physical activity (p = 0.05) and reached 75% of their total daily physical activity 20 minutes later than children with low NEQ scores (p < 0.05). Excessive evening snacking and a strong urge to eat in the evening or at night were the symptoms most related to these physical activity patterns. Children with delayed eating behaviors had higher levels of physical activity in the late morning and evening and a delayed physical activity pattern compared to children with no or fewer symptoms. Future research is needed to determine if physical activity plays a role in the onset or maintenance of night eating.
Childhood obesity is a major health concern, and there is a current need to recognize novel factors implicated in weight gain. The circadian aspect of behavior is emerging as a potential obesity risk factor (Garaulet et al., 2010). Accordingly, night eating syndrome (NES) (Stunkard et al., 1955), a syndrome of misaligned eating and sleeping patterns, is associated with obesity (Gallant et al., 2012b).
Compared to controls, individuals with NES report a delayed eating pattern of approximately 1.5 hours (Goel et al., 2009), such that they start eating later in the day, overeat in the evening (>25% of total energy intake), and/or wake up at night to eat. Aside from the core symptoms of excessive evening snacking and night eating, other important clinical symptoms of NES were recently specified: morning anorexia, onset and maintenance of insomnia, depressed mood, a strong urge to eat in the evening and into the night, and the belief that one must eat in order to fall asleep or resume sleep (Allison et al., 2010). Recently and for the first time, night eating was examined in a population-based cohort of children (aged 8-10 y) (Lundgren et al., 2012) using the Night Eating Questionnaire (NEQ) (Allison et al., 2008). Although no children in this cohort reported the presence of all NES symptoms (Lundgren et al., 2012), the NEQ total scores, a measure of symptom severity, were variable and were associated with a relatively delayed pattern of energy intake; that is, relatively less energy intake was consumed for breakfast and more after the evening meal (Gallant et al., 2012a).
The causes of late night eating in NES are currently unknown, although biological and behavioral models have been hypothesized (Stunkard et al., 2009) including involvement of the circadian system (O’Reardon et al., 2004; Goel et al., 2009). Physical activity could be implicated, as exercise can entrain the circadian rhythm by producing either a phase advance or a phase delay depending on timing (Mistlberger and Skene, 2005; Atkinson et al., 2007). Only 1 study has reported the daily physical activity profile associated with NES, and this was in adults in a 24-hour in-patient study with findings of greater spontaneous physical activity in night eaters (Gluck et al., 2011). It remains to be tested if physical activity patterns are different in individuals with a delayed pattern of energy intake or with specific delayed eating behaviors. The aim of this study was to investigate the relationship between circadian physical activity patterns and delayed eating behaviors in children at risk of obesity.
Materials and Methods
Quality Study
Data were drawn from cycle 1 of the QUALITY (Québec Adipose and Lifestyle InvesTigation in Youth) study, which is a prospective cohort of 630 families that investigated obesity risk factors in children (Lambert et al., 2012). Families were recruited through schools, and eligibility symptoms consisted of a white child aged 8 to 10 years with at least 1 obese biological parent based on body mass index (BMI) (≥30 kg/m2) or waist circumference (>102 cm for men, >88 cm for women). Body weight and height were measured during a clinical testing day. Socioeconomic status was determined by reported family income. All parents signed informed consent forms for their child, and this study was approved by the ethics committees of Laval University and the Centre Hospitalier Universitaire de Sainte-Justine.
Physical Activity
Children’s physical activity was measured using 7-day accelerometry (Actigraph LS 7164 activity monitor, Actigraph LLC, Pensacola, FL) during the week following the baseline clinical testing day. The Actigraph monitor is a waist-mounted, uniaxial accelerometer that detects vertical accelerations (magnitude and frequency) and was set at 1-minute movement epochs for the current study. A valid day was defined as ≥10 hours of recorded activity time per day (Troiano et al., 2008). Children were excluded from the analysis if they had <4 valid days (Colley et al., 2011). Nonwear time was identified by ≥60 minutes of consecutive zeros with 1 or 2 consecutive interruptions (1-2 minutes), with activity counts >0 but ≤100 counts/min (Troiano et al., 2008). Nonwear time was subtracted from wear time. Hourly counts (i.e., 1200-1259 h) were divided by hourly wear time (after nonwear time was removed) to give hourly counts per minute for each hour of the day for all 7 days. Mean counts per minute for each hour were calculated from available valid days (“0900” represents activity from 0900-0959 h). Total daily physical activity levels were estimated with the equations developed by Evenson and colleagues (2008), as recommended for the Actigraph monitor (Trost et al., 2011), where sedentary activity was <100 counts/min, light activity was >100 but <2296 counts/min, moderate activity was ≥2296 but <4012 counts/min, and vigorous activity was ≥4012 counts/min. The Actigraph monitor has been validated in children with a sensitivity of 77% and 68% for moderate and vigorous physical activity, respectively (Evenson et al., 2008). Nocturnal rest phase, the time in hours at night when the accelerometer was inactive, was estimated by subtracting the accelerometer start time (the time of the first activity counts [>50 counts/min]) from the previous night’s accelerometer stop time (the time of the last daily activity counts [<50 counts/min]) for each day (except for the first day). Data on physical activity patterns were presented in absolute (counts/min) and relative terms (% physical activity counts per wear time/4-hour period). For each day, the child self-reported his or her participation in swimming, cycling, or diving, as these activities would not be captured by the Actigraph accelerometer. In addition, the child reported the number of organized sports teams of which he or she was a member.
Delayed Eating Behaviors
Delayed eating behaviors were assessed with the NEQ. This questionnaire, which has been validated in children (Gallant et al., 2012a), was administered in 2008 to 2009 to children over the telephone by trained interviewers. The NEQ measures night eating symptoms through 14 close-ended questions on 5-point Likert scales, with 2 items related to morning appetite, 2 items related to depressive symptoms, 2 items related to sleep troubles, 3 items related to excessive evening eating, 1 item related to the level of consciousness of nocturnal ingestions, and 4 items related to night eating behavior. The NEQ score is the sum of these 14 items (maximum of 52). As suggested in adults (Allison et al., 2008), the question pertaining to excessive evening snacking (i.e., consuming ≥25% calories after the evening meal) was adjusted by using data from multiple-pass dietary recall (described by Gallant et al., 2012a). As previously reported, no children in this cohort met the criteria for NES (Lundgren et al., 2012), but the NEQ total score ranged from 0 to 21. For analysis, children were categorized by a median split of the NEQ score (high score: 8-21; low score: 0-8; mutually exclusive groups). The presence or absence of each NES symptom was identified by item frequency/severity responses on the NEQ as follows: excessive evening snacking: ≥25% total energy intake after the evening meal; night eating: eat upon awakening at least sometimes; morning anorexia: no morning appetite at all and/or eat for the first time after noon; a strong urge to eat in the evening or during the night: strong evening cravings for food at least often and/or very much or extreme cravings to eat upon awakening; the belief that one must eat in order to sleep: at least somewhat of a belief they must eat in order to return to sleep; sleep troubles: at least usually have trouble getting to sleep and/or wake up at night at least 1/week; depressed mood: at least feeling somewhat blue and/or an evening-worsening mood.
Statistical Analysis
Repeated-measures MANOVA controlling for nocturnal rest phase was used to test if physical activity patterns differed between children scoring high on the NEQ and children scoring low on the NEQ and between children with or without each NES symptom. Post hoc Student t tests were used to determine the hours that physical activity counts were statistically different between groups. The relative (%) distribution of physical activity between groups was assessed by grouping physical activity counts into 4-hour blocks covering the relevant daily periods (morning, afternoon, and early and late evening). ANCOVA was used to test for significant differences between the proportions of total physical activity that was undertaken during each of these periods while controlling for nocturnal rest phase (h).
Results
Forty-six children were excluded because they did not have 4 valid days of accelerometer time (≥10 hours), leaving 269 children for analysis. There were no statistically significant differences in the mean age, mean BMI, sex distribution, or NEQ scores between children with and without 4 valid days (data not shown). There were no differences in the reported occurrence of swimming, diving, or cycling between the low and high NEQ score groups.
Children with high NEQ scores had statistically higher counts per minute compared to children with low NEQ scores. Consequently, children with high NEQ scores had higher daily amounts of vigorous and moderate and, to a lesser extent, light physical activity (Table 1). This difference remained statistically significant after controlling for nocturnal rest phase (h).
Participant characteristics.
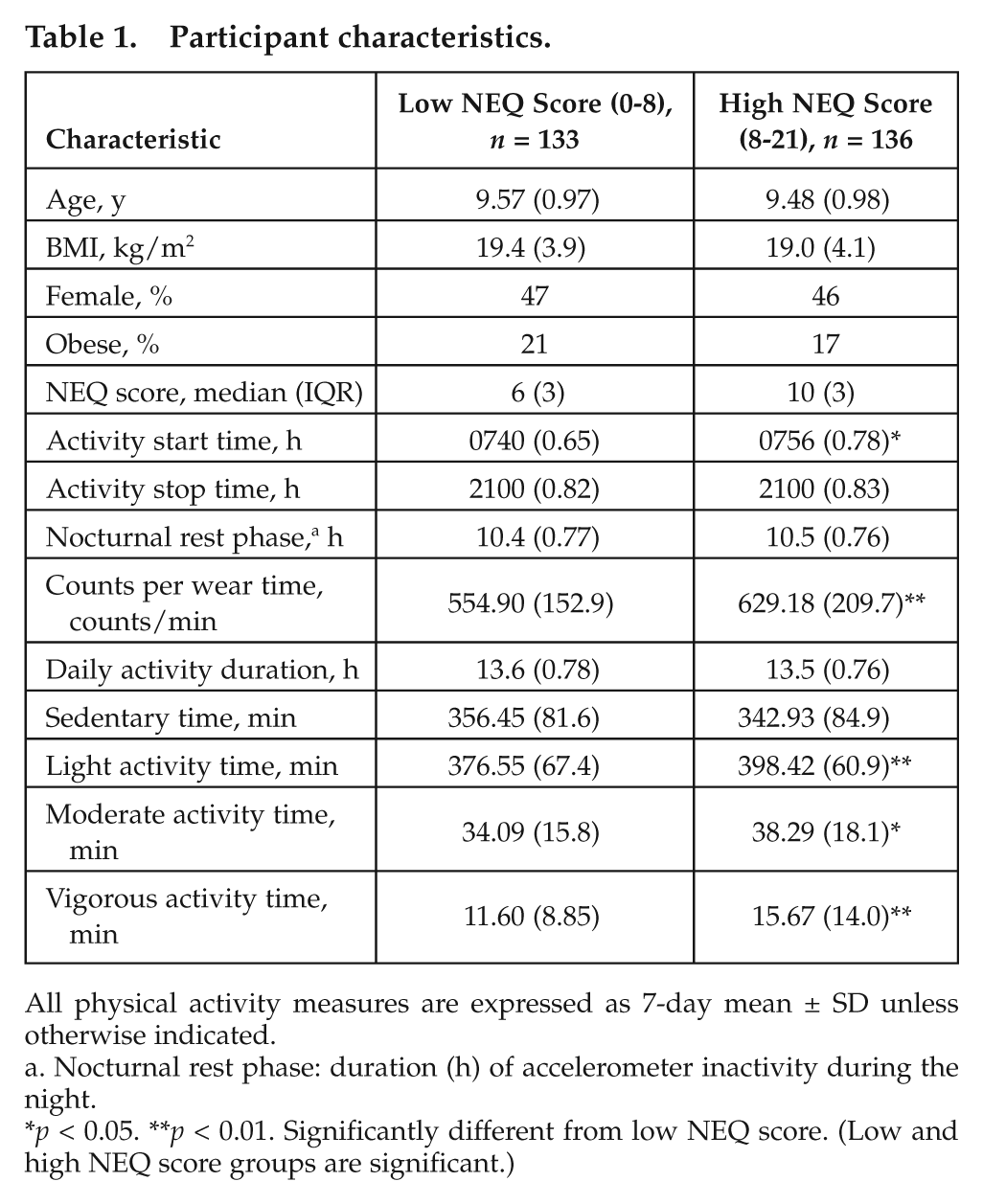
All physical activity measures are expressed as 7-day mean ± SD unless otherwise indicated.
Nocturnal rest phase: duration (h) of accelerometer inactivity during the night.
p < 0.05. **p < 0.01. Significantly different from low NEQ score. (Low and high NEQ score groups are significant.)
Repeated-measures analyses revealed different patterns of daily physical activity between children with high and low NEQ scores; children with high NEQ scores had higher counts per minute during the afternoon and evening (F18,248 = 1.64, p = 0.05) (Figure 1). Controlling for BMI, socioeconomic status, mean activity morning start time, and total activity duration time did not affect the results (data not shown).
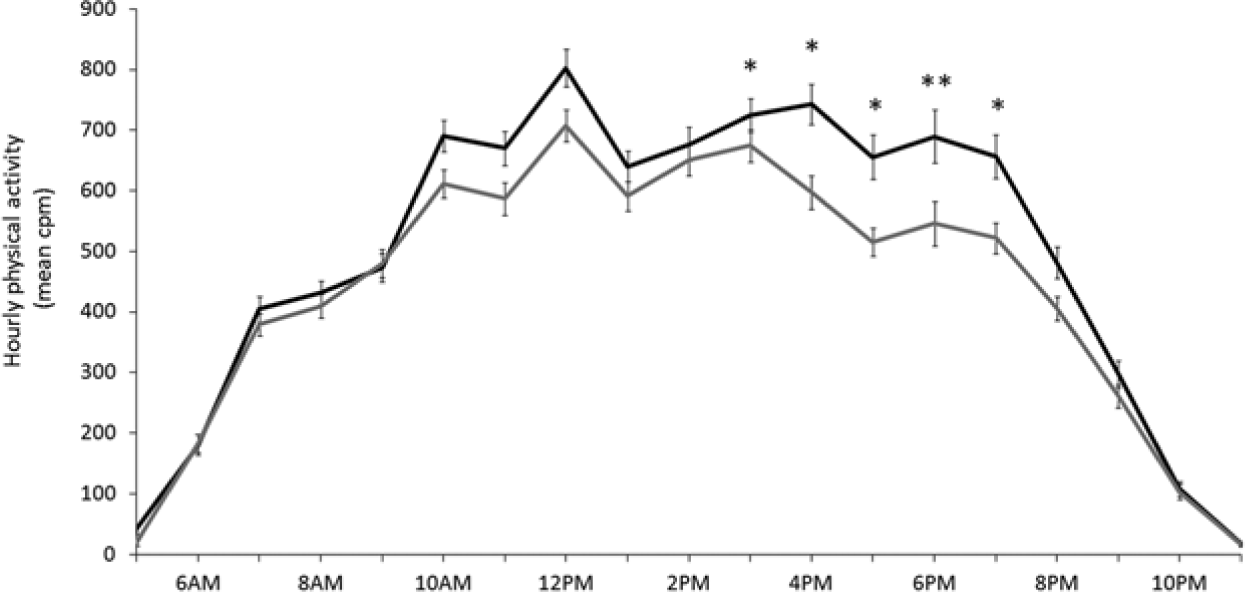
Mean (7-day) counts per minute in children with high (black, n = 136) and low (gray, n = 133) NEQ scores (median split). Repeated-measures MANOVA significant group by time effect: F18,248 = 1.64, p = 0.05. A posteriori analysis: *p < 0.05, **p < 0.01.
The proportion of total physical activity that occurred in the morning was higher in children with low NEQ scores compared to children with high NEQ scores (17.8% v. 16.2%, respectively; p = 0.01). Conversely, the proportion of total physical activity in the evening was higher in children with high NEQ scores compared to children with low NEQ scores (17.0% v. 15.6%, respectively; p = 0.04). Results remained statistically significant after controlling for nocturnal rest phase (h). Children with a high NEQ score attained 75% of their total physical activity approximately 20 minutes later than children with a low NEQ score (1752 v. 1733 h, respectively) (Figure 2).
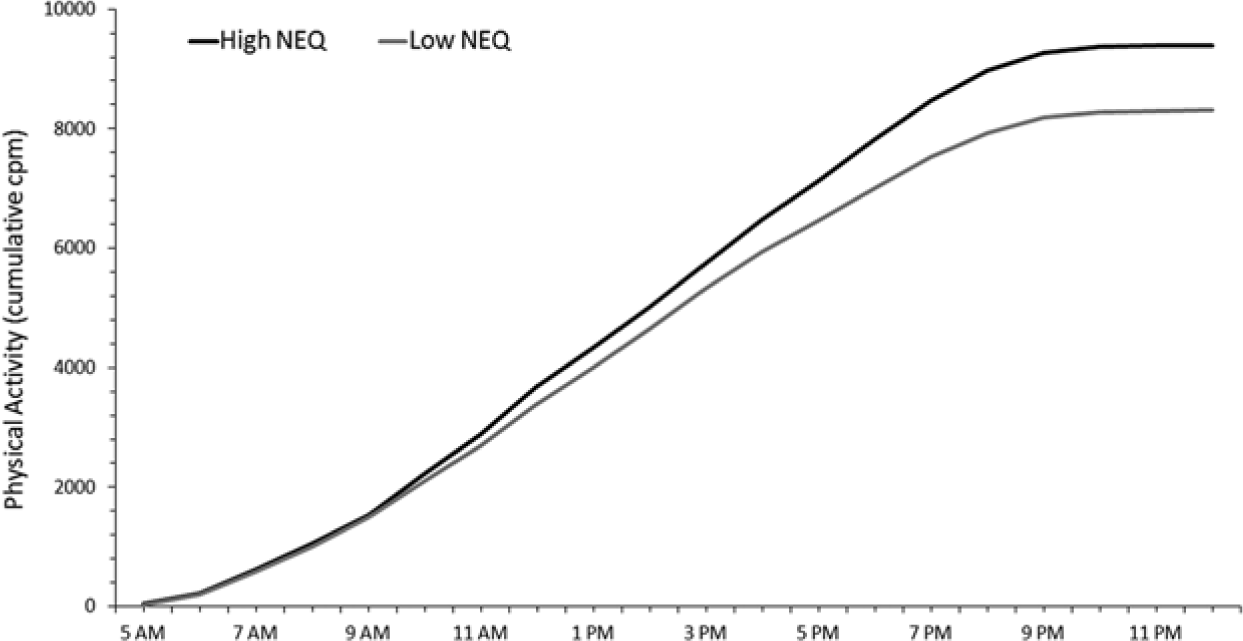
Cumulative mean physical activity counts per minute for children with high (n = 136, black line) and low (n = 133, gray line) NEQ scores. Children with a high NEQ score reached 75% of their total daily physical activity approximately 20 minutes later than children with a low NEQ score (1752 v. 1733 h, respectively).
Different physical activity patterns were observed among children reporting core NES symptoms including excessive evening snacking (F18,248 = 1.72, p = 0.04) and night eating (F18,248 = 2.22, p = 0.004) compared to children not reporting these symptoms (Figure 3). One child reported a need to eat in order to fall asleep, and this child’s physical activity profile was also statistically different from the other children who did not report this symptom (F18,248 = 4.80, p < 0.0001). Children who reported a strong urge to eat in the evening or at night had higher physical activity counts throughout the day compared to children who did not report this symptom (group effect: F1,265 = 4.33, p = 0.04), but the patterns were not significantly different (group by time effect: F18,248 = 1.15, p = 0.30). Repeating analyses excluding this child did not affect the other results. No other NES symptoms were related to physical activity patterns.
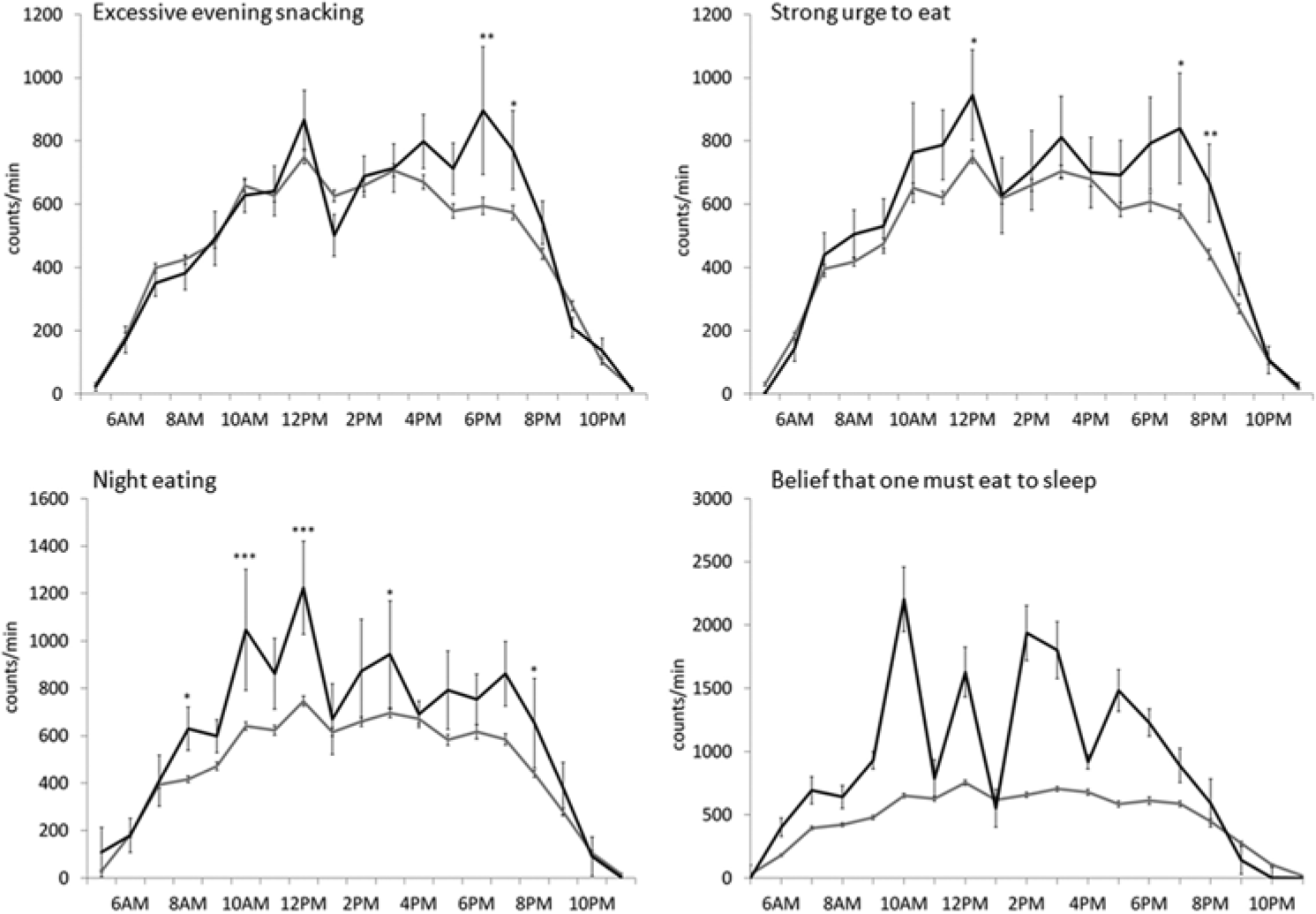
Mean physical activity counts per minute for each hour for children with and without delayed eating behaviors (clockwise from top left): (A) excessive evening snacking (black line, ≥25% total caloric intake after the evening meal [n = 18]; gray line, <25% [n = 251]) (group by time effect: F18,248 = 1.72, p = 0.04); (B) a strong urge to eat in the evening or at night (black line, often/somewhat strong cravings/urges to eat after supper or at night [n = 12], gray line, not at all/a little [n = 257]) (group effect: F1,265 = 4.33, p = 0.04; group by time effect: not significant); (C) night eating (black line, eat upon awakening at least sometimes [n = 6], gray line, no night eating [n = 263]) (group by time effect: F18,248 = 2.22, p = 0.004); (D) the belief that one must eat in order to fall asleep (black line, somewhat believe that one must eat in order to return to sleep [n = 1], gray line, no such belief [n = 268]) (group by time effect: F18,248 = 4.80, p < 0.0001).
Discussion
This study aimed to investigate the relationship between daily physical activity patterns and delayed eating behaviors in children at risk of obesity. Results show that children with delayed eating behaviors, as measured by the NEQ, have higher levels of total physical activity that is accentuated during the early evening compared to children with fewer symptoms. Moreover, children with high NEQ scores present a delayed pattern of physical activity compared to children with low NEQ scores. Core delayed eating behaviors (i.e., the presence of excessive evening snacking or night eating) were the NES symptoms most associated with accentuated physical activity patterns in the evening.
Our results are consistent with those of Gluck and colleagues (2011), who reported higher 24-hour spontaneous physical activity in individuals who ate at night. The authors hypothesized that the higher nocturnal activity in night eaters may indicate restless sleep and increased arousal and thus a potential circadian problem. Similarly, results from this study indicate a relative delay in physical activity that is associated with delayed eating behaviors; children with a high NEQ score attained 75% of their total physical activity approximately 20 minutes later than children with a low NEQ score (1752 v. 1733 h, respectively). Whether this delay has clinical significance is uncertain; unlike in a previous study (Ogbagaber et al., 2012), BMI was not implicated in the delay in physical activity observed in this study (results persisted after controlling for BMI, and there were no significant BMI differences between NEQ groups). The present results, in combination with the previous report that children with high NEQ scores show a delayed pattern of energy intake (Gallant et al., 2012a), demonstrate a relative delayed behavior profile in children with delayed eating behaviors. None of these children had NES, and the delays presented herein are small. The implication of physical activity in night eating merits further examination, particularly among individuals diagnosed with NES because an impairment in the circadian system may increase the risk of obesity (Garaulet et al., 2010).
Although the delay in physical activity is short (~20 minutes), it could theoretically be involved in delayed eating behaviors. Exercise is a zeitgeber; that is, it can synchronize the circadian rhythm and can create a circadian phase shift (Buxton et al., 2003; Mistlberger and Skene, 2005; Atkinson et al., 2007). The intensity, duration, and timing of physical activity necessary to generate a circadian phase shift are not known. However, both long, low-intensity activity and short, high-intensity activity can have similar effects on phase shifts (Atkinson et al., 2007). In fact, cycling at 70% VO2 max for 30 minutes creates a phase shift of 1 hour (Edwards et al., 2002). In the present study, it is uncertain if the amount of evening activity observed in children with delayed eating behaviors is enough to produce a zeitgeber effect because the evening physical activity was categorized as light physical activity, using the cut-offs suggested by Evenson et al. (2008). There are no data on the circadian effect of evening exercise (Atkinson et al., 2007), and no studies examined the impact of habitual daily physical activity patterns on circadian rhythm. The latter is important if an effect accumulates over time such that habitual patterns over days (or longer) may have a larger effect on phase shifting than the effects of short-term interventions (Baehr et al., 2003).
Another hypothesis is that higher evening physical activity in children with delayed eating behaviors may increase hunger sensations during this time and thus promote energy intake in the evening. That NES symptoms related to eating are associated with this activity pattern, and not the symptoms related to sleep or mood, also supports this. However, it is important to note that there is also a large variability in dietary compensatory responses to physical activity (King et al., 2012), and a recent review concluded that, although there is some connection between energy intake and expenditure in children, the former is not dependent on the latter (Thivel et al., 2013). Nonetheless, if increased evening activity leads to increased hunger at night, this pattern could lead to more severe delayed eating behaviors.
This study has several limitations. The data are cross-sectional, limiting causal inference. Moreover, no children met all research diagnostic criteria and thus are not representative of individuals who report the full syndrome; however, as in other eating disorders such as binge eating disorder, subthreshold symptoms are a risk factor for developing the full syndrome later in life (Stice et al., 2013). Prospective studies will allow us to understand this relationship better, but for now, there is value in examining the association between the presence of delayed eating behaviors and physical activity patterns. Moreover, data regarding the time of the evening meal were not available, and it is not known if children who reported excessive evening snacking ate their last meal at an earlier time than children who did not report this behavior. Data regarding sleep duration were also not available, and this variable had to be estimated from the accelerometer start and stop times (nocturnal rest phase). Finally, additional measures of the timing of circadian rhythms, such as melatonin levels, would support these associations and would better quantify circadian phase differences.
In conclusion, children with delayed eating behaviors, in particular, excessive evening snacking and night eating, have higher overall physical activity that is accentuated in the late morning and evening, which translates into delayed physical activity patterns compared to children without symptoms.
Footnotes
Acknowledgements
This research was conducted by members of TEAM PRODIGY, an interuniversity research team including Université de Montréal, Concordia University, Université Laval, and McGill University. The QUALITY cohort was funded by the Canadian Institutes of Health Research, the Heart and Stroke Foundation of Canada, and Fonds de la Recherche en Santé du Québec. A.R. Gallant is funded by the Quebec Heart and Lung Research Institute. Dr. Marie Lambert (July 1952–February 2012), a pediatric geneticist and researcher, initiated the QUALITY cohort. Her leadership and devotion to QUALITY will always be remembered and appreciated. The authors are also grateful to all the families that participated in the QUALITY cohort. Finally, they thank Christian Couture for his help with data compilation and Dr. Albert Stunkard, a pioneer of NES, for his help with translating the NEQ.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
