Abstract
Steroid hormones including estrogens modulate the expression of daily activity and circadian rhythms, including free-running period, phase angle of activity onset, and response to light. The mechanisms underlying these effects, however, are not fully understood. We tested the hypothesis that estrogen signaling is required for photic responsiveness of the circadian timing system. We used estrogen receptor subtype 1 (ESR1) knock-out mice (ERKO) and nonclassic estrogen receptor knock-in mice (NERKI). ERKO animals are unable to respond to estrogen at ESR1, and NERKI animals lack the ability to respond to estrogens via estrogen response element-mediated transcription but still respond via nonclassical mechanisms. We analyzed behavioral shifts in activity onset in response to 1-h light pulses given across the subjective 24-h day in gonadally intact male and female NERKI, ERKO, and wild-type (WT) littermates. We also examined Fos protein expression in the suprachiasmatic nucleus, the site of the master circadian pacemaker, at 2 times of day. We found a significant effect of genotype on phase shifts in response to light pulses given in the subjective night. Female WT mice had a significantly larger phase response than ERKO females during the early subjective night (phase shift of 98 min and 58 min, respectively; p < 0.05). NERKI females were intermediate to WT and ERKO females, suggesting a contribution of nonclassical estrogen signaling on circadian timekeeping functions. This genotype effect is not observed in males; they did not have a difference in phase shifts following a light pulse at any time point. WT males, however, shifted an average of 47 min less than did females at zeitgeber time (ZT) 16 (ZT 0 lights-on and ZT 12 lights-off). These data indicate that estrogens modify the response of the circadian timekeeping system to light via classical and nonclassical signaling pathways.
Exposure to steroid hormones during development and the presence of circulating hormones in adulthood influence the total amount of daily activity and the pattern of activity across the light-dark (LD) cycle. Ovariectomy of female mice, rats, and degus results in decreased total wheel-running activity relative to intact counterparts, and estradiol replacement to these animals restores the amplitude of wheel-running activity (Albers, 1981; Labyak and Lee, 1995; Ogawa et al., 2003). Estrogens also regulate the daily pattern and consolidation of activity across the light-dark cycle; ovariectomy of female mice increases the relative amount of diurnal activity as compared with intact animals, and estradiol replacement consolidates wheel running to the dark phase (Ogawa et al., 2003; Brockman et al., 2011; Blattner and Mahoney, 2012). Estradiol replacement given to ovariectomized rats, hamsters, and degus advances the onset of the daily activity bout relative to nontreated controls (Morin, 1980; Albers, 1981).
In addition to these effects on the distribution and amplitude of daily activity rhythms in rodent models, estradiol also modifies the endogenous timekeeping mechanism, including the free-running period (tau) and the coupling of activity to the light cycle (Davis et al., 1983). In female rats and hamsters, ovariectomy results in a lengthened tau, and estradiol shortens and restores tau to that of intact animals (Zucker et al., 1980; Albers, 1981). It is likely that circulating estrogens influence the amplitude of the light-induced phase shifts during the dark phase. Ovary-intact and ovariectomized female mice have significant differences in the amplitude and timing of the phase shifts in the subjective day and night, indicating a role for estrogens in modifying the entrainment function of the central clock (Brockman et al., 2011). However, neither the mechanisms underlying this modulation nor the site of estrogen action are known.
The photic phase-response curve (PRC) describes a behavioral or neuronal response to light with the amplitude dependent on the time of day a light cue is given. Typically, animals that are given a light pulse during the early subjective night have a phase delay in the timing of their activity rhythm, while light pulses during the late subjective night advance their activity patterns (Daan and Pittendrigh, 1976; Brockman et al., 2011).The central oscillator, located within the suprachiasmatic nucleus (SCN), maintains intrinsic circadian rhythms in behavior (Nunez and Stephan, 1977; Stephan and Nunez, 1977). Photic information from the retina travels to the SCN via the retinohypothalamic tract and synapses on cells in the “core” subdivision; this region sends photic information to the rhythmic “shell” portion of the SCN. The SCN responsiveness to light cues can be measured by the expression of the transcription factor Fos, a member of the immediate early gene family (Yan, 2009). In parallel with a shift in the timing of activity, light pulses given during the subjective night also induce Fos expression in the SCN (Rusak et al., 1990; Colwell and Foster, 1992; Schwartz et al., 1994). During the subjective day, light pulses have relatively little effect on the timing of activity onset or the induction of Fos expression in the SCN (Mahoney et al., 2001).
Despite evidence that estrogens modulate the timing (phase) and amplitude of physiological rhythms, wheel-running activity is the only factor for which the activational effects have been described. Circulating estrogens acting at estrogen receptor subtype 1 (ESR1, formerly known as estrogen receptor α) regulate the amount of total locomotor activity; ESR1 knock-out mice (ERKO) have decreased total daily wheel-running activity relative to wild-type (WT) counterparts, and estradiol replacement does not induce an increase in wheel running (Ogawa et al., 2003; Blattner and Mahoney, 2012). Estrogen actions are mediated through at least 2 distinct nuclear receptors: ESR1 and estrogen receptor subtype 2 (ESR2, or estrogen receptor β) as well as more recently characterized “nonclassical” pathways, including membrane-associated estrogen receptors (Zucker et al., 1980; Hall et al., 2001; Kousteni et al., 2001). In nuclear receptor–mediated transcriptional activation, estrogens bind to receptors, and the ligand-receptor complexes form dimers and act as transcription factors. In the “classical” pathway, the hormone-receptor dimer binds to the estrogen response element (ERE) on regulatory regions of target genes to alter gene transcription. In “nonclassical” mechanisms of estrogen signaling, the hormone-receptor dimer initiates a second messenger pathway that leads to transcriptional regulation at non-ERE domains and activates additional downstream signaling (McDevitt et al., 2008). Studies with transgenic mice strongly suggest that the action of estrogen on these nonclassical pathways contributes to overall activity levels (Blattner and Mahoney, 2012).
The characterization of ESR1 and ESR2 protein or mRNA has been described for neonatal rat SCN cell culture and adult rat, mouse, and human tissue (Shughrue et al., 1997a, 1997b; Su et al., 2001). In adult mice, there is higher expression of ESR2 relative to ESR1. The ESR1 population extends throughout the rostral-caudal extent of the nucleus and is present to a greater extent in the shell than the core of the SCN (Vida et al., 2008). This is somewhat inconsistent as Mitra et al. (2003) did not detect any ESR1 protein expression and a relatively small amount of ESR2 immunoreactivity in the mouse SCN (Mitra et al., 2003). The human SCN also contains estrogen receptors with significantly greater ESR1 and ESR2 expression in females relative to males (Kruijver and Swaab, 2002; Kruijver et al., 2003). It remains unknown whether the relatively small number of estrogen receptors found within the SCN plays a significant role in modulating circadian rhythms or if estrogen receptor populations outside of the SCN are more important. For example, in ovariectomized rats, estradiol implants in the medial preoptic region of the hypothalamus increase wheel-running activity, whereas lesions of this region result in an attenuation of the effect of estradiol. This region contains a relatively large number of ESR1-containing cells (King, 1979; Fahrbach et al., 1985; Ogawa et al., 2003).
Here we sought to determine (1) the receptor signaling mechanisms by which estrogens modulate the response of the circadian timekeeping system to the LD environment and (2) whether estrogen actions have an impact at the level of the primary circadian clock within the SCN. We tested the hypothesis that the ability to respond to estrogens, specifically at ESR1 and via nonclassical activation, influences the entrainment of the circadian system to light. To test this, we quantified the behavioral (phase shifts) and neuronal response to light pulses in 2 transgenic mouse models with impairments in responsiveness to circulating estrogens. ERKO (estrogen receptor knock-out) mice lack ESR1 but retain a functional ESR2 (Dupont et al., 2000). In “nonclassical” estrogen receptor knock-in (NERKI) mice, a modified ESR1 receptor with a mutation in the ERE-binding domain is inserted (Jakacka et al., 2002). In these mice, estrogenic action via ERE-mediated transcription is eliminated, but estrogens retain the ability to act via other transcriptional pathways as well as at intact ESR2. The NERKI model allows us to determine underlying mechanisms of estrogens on behaviors that are not mediated by ERE-DNA binding.
We used both male and female transgenic mice for several reasons. First, sex differences in daily activity and circadian rhythms have been reported in both humans and laboratory animals (Zucker et al., 1980; Duffy et al., 2011). Second, in mice, androgens have a significant effect on activity amplitude and distribution across the day. It is possible that some effects of testosterone are via its aromatizable form of estradiol. Last, estradiol exposure in development (Brockman et al., 2011) and in adulthood (Ogawa et al., 2003; Blattner and Mahoney, 2012) has an impact on the expression of behavioral rhythms in both sexes. We quantified the phase-response curve in both male and female transgenic mice. We want to elucidate the molecular mechanisms by which estrogens shape the development and expression of circadian rhythms. Here we use transgenic mice with disruptions in estrogen signaling to establish a baseline for future studies by determining the contributions of genomic and nongenomic signaling mechanisms.
Methods
Animal Breeding and Care
Adult intact (4- to 7-month-old) NERKI, ERKO, and WT male and female mice were used for these experiments. ERKO and NERKI mice were obtained from well-established genetic lines. ERKO mice (ESR1–/–) were derived from an inbred colony of ERaL3–/+ mice (Dupont et al., 2000) with animals kindly supplied by Dr. Pierre Chambon (IGBMC, France). NERKI mice (ESR1–/AA) have a mutant form of the ESR1 receptor (AA) and were obtained from a colony established at the University of Illinois with animals kindly supplied by Dr. J. Larry Jameson (University of Pennsylvania, Philadelphia). NERKI mice were originally created as described (Jakacka et al., 2002). Both NERKI and ERKO lines have been bred onto a congenic C57BL/6J background and maintained on this background for the current experiment. ERKO mice are maintained with heterozygous breeding pairs. NERKI mice are maintained by pairing heterozygous males (ESR1AA/+) with 1 copy of the modified ESR1 with heterozygous ERKO females (ESR1+/–), as NERKI heterozygote females are infertile. All genotypes of tail biopsies were confirmed by polymerase chain reaction (PCR) containing the 3 primers: P1 (5′-TCG CTT TCC TGA AGA CCT TTC ATAT-3′), P2 (5′-CCA TTG TCT CTT TCT GAC ACA TGC-3′), and P3 (5′-GCA AAT AGC GGG AGA TCT AAT TCT AGA TAC-3′). These primers amplify products that are 320 bp (NERKI), 280 bp (WT), and 190 bp (ERKO) in size. Control mice are WT littermates of NERKI and ERKO mice. Breeding animals and litters (<21-day-old pups) were maintained on Teklad 8626 rodent diet. At the time of weaning (postnatal day 21) and throughout the rest of these studies, animals were maintained on Teklad 2016 diet, which contains low soy estrogens (isoflavones) in the range of nondetectable to 20 mg/kg. Food and water were given ad libitum.
Mice were maintained in 12-h light/12-h dark (LD) cycles unless otherwise indicated, and the light intensity in the cages ranged from 220 to 360 lux (average 290 lux). Animals were individually housed in cages (28 × 16 × 12 cm) equipped with a metal wheel affixed to the top of the cage. Wheel revolutions were registered by a magnetic switch and recorded in 10-min bins of activity. Wheel-running activity was recorded and visualized using VitalView and ActiView, respectively (MiniMitter, Bend, OR). Stage of female reproductive cycle was not monitored during the studies. In rats, hamsters, and degus, changes in estrous cycle stage correlate with the timing and amplitude of activity (Morin et al., 1977; Albers et al., 1981; Wollnik and Turek, 1988; Labyak and Lee, 1995). In mice, estrous cycle stage has no predictable effect on daily activity levels. We are thus able to make comparisons between cycling intact animals (Koehl et al., 2003; Kopp et al., 2006; Blattner and Mahoney, unpublished data). All animal procedures were approved by the Institutional Animal Care and Use Committee of the University of Illinois and were conducted in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
In these studies, we compared gonadally intact animals to establish their baseline circadian rhythms. Circulating hormone concentrations for NERKI, ERKO, and WT animals have been previously reported and are relatively similar to one another, allowing us to compare between intact animals. In intact female WT mice, the levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) do not differ from those of ERKO and NERKI animals. Further, in NERKI, females’ estradiol concentrations are not significantly different compared with WT littermates (Jakacka et al., 2002; McDevitt et al., 2008). In ERKO females, circulating serum levels of estradiol and LH are increased somewhat compared with WT females (Couse et al., 1995). In intact ERKO males, testosterone levels are slightly elevated relative to WT mice, but there is no difference in FSH and LH levels (Eddy et al., 1996). NERKI and WT males do not differ from one another with respect to testosterone concentrations (McDevitt et al., 2007).
Determination of Phase Response to Light Pulse
The behavioral shift in activity that results from a light pulse given at different times of the subjective day is called a phase response and can be plotted as a phase-response curve. A modified Aschoff type II procedure was used, which enables us to pulse animals with light before their free-running rhythms drift significantly from their entrainment to the LD cycle (Mrosovsky, 1996). Animals were entrained to a 12:12 LD cycle and then placed in constant darkness (DD) prior to a 1-h light pulse. The light pulse was given at different times during their second day in the subjective day or night, which corresponds with the former zeitgeber times (ZT) 0, 4, 8, 12, 16, and 22 (ZT 0 = lights-on, ZT 12 = lights off). The time of the light pulse thus corresponded to 24, 28, 32, 36, 40, or 46 h in DD, respectively (Mrosovsky, 1996). Following the light pulse, animals were returned to constant darkness for 7 days. For each time point, a sample size of the behavioral tests for males was WT 5 to 7, NERKI 7 to 8, and ERKO 14 to 18. For females, the sample sizes were WT 10 to 14, NERKI 5 to 7, and ERKO 7 to 9. Sample sizes for each group (following removal of outliers) are listed in the figure legends.
The phase response, measured in minutes, was calculated as the difference between the prepulse and postpulse regression lines on the day following the light pulse. The prepulse regression line was determined by the onset of activity for 3 to 5 days prior to the light pulse. The postpulse regression line was determined for 7 days in DD, disregarding the day immediately following the light pulse to avoid transition effects. Onset of activity was defined as the first of at least 3 consecutive 10-min bins of activity that were not separated by more than two 10-min bins of inactivity before the next recorded activity bout. The phase of activity onset does not differ between intact females of any genotype. In males, ERKO and WT have a phase angle of activity onset that differs by 8 min (–9 ± 1.3 vs. –17 ± 2.6 SEM, relatively) (Blattner and Mahoney, 2012). Regression lines were determined by eye by 2 investigators that were blind to the genotype and sex of the animals. The calculated phase shifts did not differ between the 2 investigators.
Fos Activation in the SCN
Mice were entrained to 12:12 LD prior to going into DD for at least 24 h. They were given a 1-h light pulse at times corresponding to either ZT 4 (28 h in DD) or ZT 16 (40 h in DD), as described above. Animals were perfused immediately following the 1-h light pulse (ZT 5 or ZT 17, respectively, n = 3-5 animals/time point/genotype). Brains were collected, stored overnight in a 4% paraformaldehyde solution (0.1M PB, pH 7.2), and then transferred to a 20% sucrose solution (0.1M PB, pH 7.2). Tissue was sectioned in 40-µm slices in 3 series for analysis. Due to the availability of transgenic animals, we elected to compare between genotypes within a time point (ZT 4 and 16) rather than have nonpulsed controls. To detect Fos in the SCN, 1 series of tissue from all animals (WT, NERKI, and ERKO males and females pulsed at ZT 4 or ZT 16, n = 3-5/group) was processed simultaneously. Free-floating sections were incubated in rabbit polyclonal anti–c-Fos (sc-52) antibody (lot #G1108, 1:20,000 dilution; Santa Cruz Biotechnology, Santa Cruz, CA) containing 0.3% Triton-X overnight at room temperature on a shaking platform. They were then incubated in biotinylated goat anti-rabbit antibody (1:400 dilution; Vector Laboratories, Burlingame, CA) for 1.5 h at room temperature. Finally, sections were incubated in horseradish peroxidase–conjugated avidin-biotin complex following the manufacturer’s directions (Vector Laboratories) for 1.5 h at room temperature. The chromogen reaction was carried out at room temperature in a 50-mL solution containing 10 mg diaminobenzidine in Tris buffer (pH 7.6, 0.0002% DAB) (Sigma-Aldrich, St. Louis, MO), and 0.0006% H2O2. Between each step, free-floating sections were rinsed in 0.01M phosphate-buffered saline (PBS, pH 7.2) for 3 × 5 min. Sections were then mounted on gelatin-coated slides, air dried, dehydrated through an ascending alcohol series, cleared in xylene, and cover-slipped with Permount mounting medium.
Photomicrography and Data Analysis
For each animal, the region containing the central SCN of the immunostained sections was photographed, and the captured images were then analyzed with ImageJ software (available at http://rsb.info.nih.gov/ij/download/src/). Central SCN sections were identified based on a mouse brain atlas (Franklin and Paxinos, 2008), published SCN anatomy (Yan, 2009), and cresyl violet–stained series from a separate age-matched littermate. The SCN was divided into core and shell, and Fos-positive cells in these regions were counted from both of the paired nuclei (Yan and Silver, 2004).
Statistical Analysis
For the phase-response curve, behavioral shifts (min) were analyzed with a 2-factor analysis of variance (ANOVA) with time and genotype as independent factors (SPSS). Males and females were analyzed separately. A priori post hoc comparisons were then performed to determine differences between genotypes at each individual time point. The data are presented with ZT 0 replotted as ZT 24 to aid in visualization of the PRC shape. A line of best fit was applied to the data to interpolate between points using a Bezier spline equation (Klasson, 2008).
The number of Fos-positive cells within the core, shell, and total SCN regions was analyzed in an ANOVA with genotype as the independent factor. Each time point (ZT 4 and 16) was analyzed separately because we did not have unpulsed animals with which to quantify the time-dependent effect of light on Fos induction. Because intact males and females have differences in circulating hormones, we elected not to make statistical comparisons between sexes to avoid this confound. For all data sets, outliers were identified by SPSS as 3 times greater than or less than the interquartile range. Differences were significant when p < 0.05.
Results
Phase Response Curve: Genotype Influences Phase Response in Females but Not Males
Mice exposed to a 1-h light pulse during constant darkness had phase shifts in wheel-running activity dependent on the time of the light pulse (Figs. 1 and 2). The amplitude of the phase shifts at different times of day was typical of that of other nocturnal species (Daan and Pittendrigh, 1976). Animals exhibited a phase delay in response to a light pulse given in the early subjective evening (ZT 16) and a phase advance in response to light pulses in the late subjective evening (ZT 22) and at the beginning of the subjective day (ZT 0). There were relatively small phase shifts in response to light pulses given during the subjective day or the typical period of inactivity (ZT 4 and ZT 8). At ZT 16, WT females had a larger phase delay than WT males (phase delay of –98.2 ± 11.7 min, n = 11 in females and –51.4 ± 10.8 min, n = 7 in males).
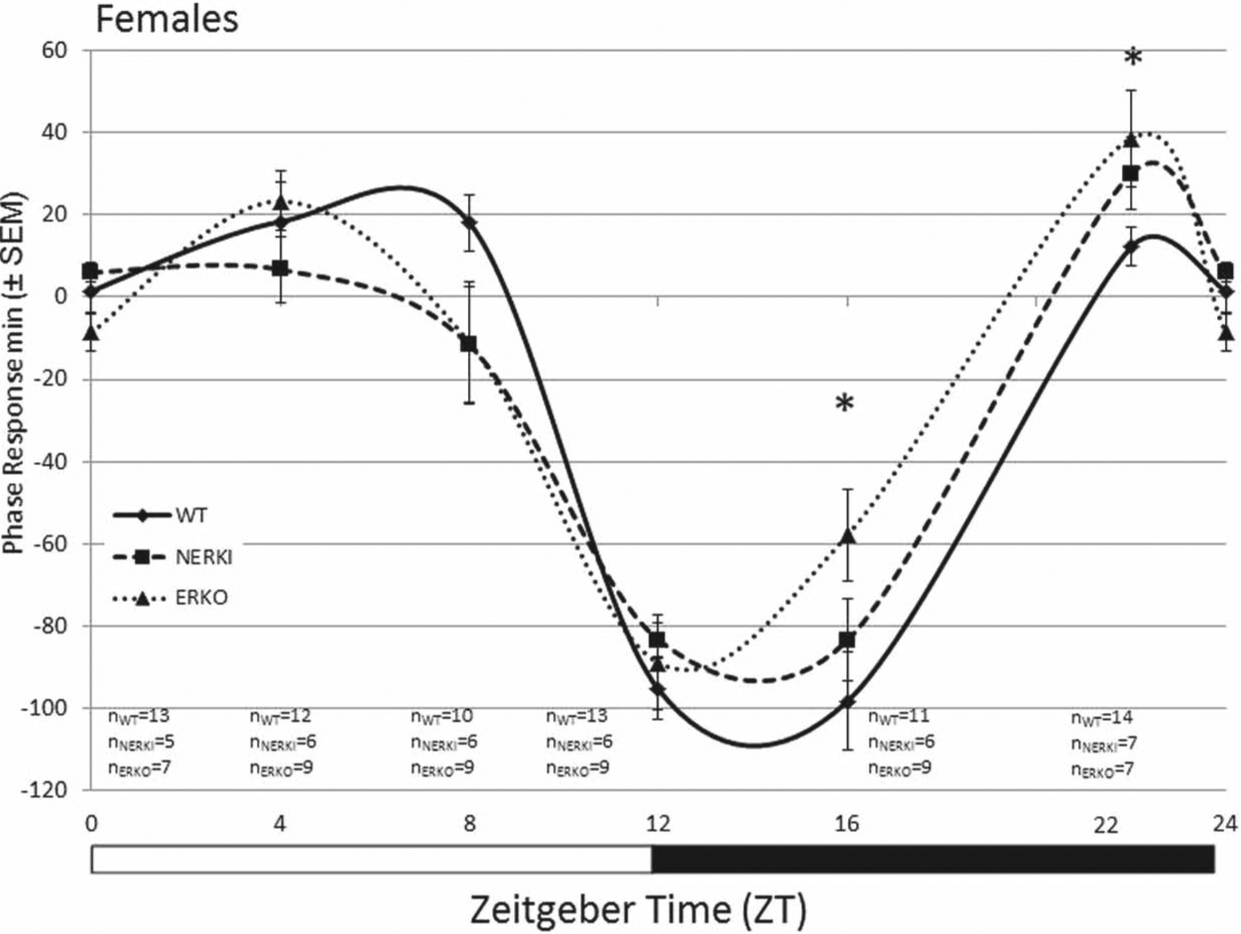
Photic phase-response curve for female WT, NERKI, and ERKO mice (phase shift [min] ± SEM). Animals were pulsed with light for 1 h at ZT 0, 4, 8, 12, 16, and 22, and the phase response (minutes) was calculated as the difference between the pre- and postpulse regression lines. *WT compared to ERKO, p < 0.05. Sample sizes are indicated below each time point: WT (n = 10-14), NERKI (n = 5-7), and ERKO (n = 7-9) for each time point.

Representative actograms from female WT, NERKI, and ERKO mice at ZT 4, ZT 16, and ZT 22. Wheel revolutions were recorded in 10-min bins and data are double plotted.
In females, a 2-factor ANOVA (genotype × time) revealed a significant effect of time on the amplitude of the behavioral phase shift following a light pulse (F5,141, p < 0.001). There were no effects of genotype, but we did find a significant interaction between genotype and time (Fig. 1; F10,141, p = 0.015). Post hoc analysis between genotypes at each time point indicate that light given during the subjective day (ZT 0, 4, 8, and 12) produced shifts in activity that were not significantly different between groups. In the subjective night, however, we found a significant effect of genotype on the degree of phase shifting at ZT 16 (F2,23, p = 0.049) and 22 (F2,25, p = 0.043; Fig. 2). Specifically, the behavioral shifts following a light pulse in WT and ERKO significantly differed from one another (ZT 16, p = 0.039; ZT 22, p = 0.048). WT females had a significantly greater delay in response to a light pulse than ERKO females at ZT 16 (phase delay of –98.2 ± 11.7 min, n = 11 and –57.8 ± 11.2 min, n = 9, respectively) and a significantly smaller shift than ERKO females at ZT 22 (12.1 ± 4.7 min, n = 14 and 38.6 ± 11.6 min, n = 7, respectively). Interestingly, NERKI females had intermediate phase responses to light pulse at these times relative to WT and ERKO females.
In males, a 2-factor ANOVA (genotype × time) revealed a significant effect of time (F2,157, = 4.99, p = 0.008) and genotype (F5,157 = 39.43, p < 0.001) on the amplitude of the behavioral phase shift following a light pulse (Fig. 3). In contrast to females, there was no effect of genotype on the phase shift following a light pulse in males at any time point, with the exception of ZT 0. At this time point, the phase shift of NERKI males (40 ± 15.2 min, n = 8) differed from both ERKO (p = 0.029, 10.62 ± 3.6 min, n = 16) and WT males (p = 0.024, 0 ± 0 min, n = 5).
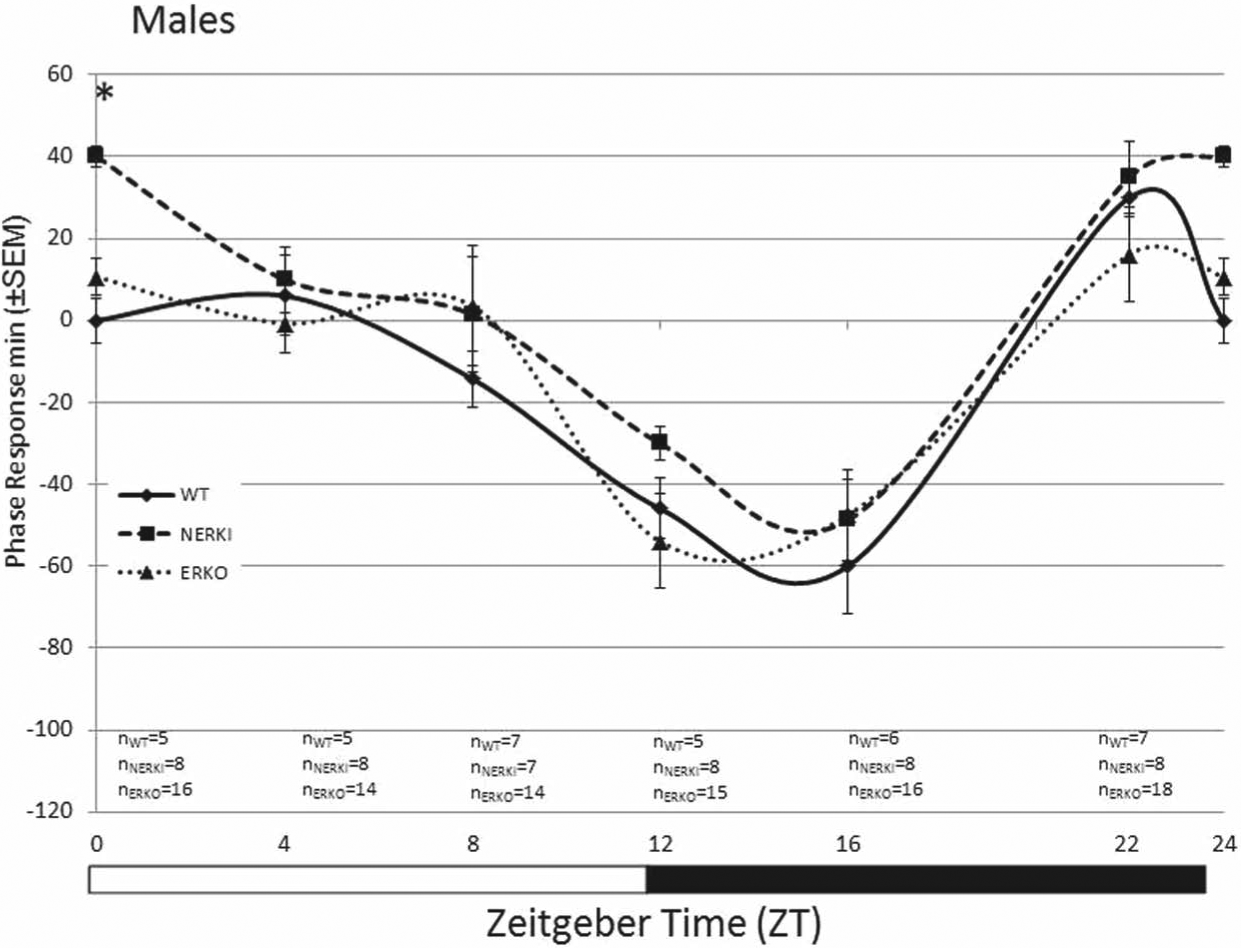
Photic phase response curve for male WT, NERKI, and ERKO mice (±SEM). Animals were pulsed with light for 1 h at ZT 0, 4, 8, 12, 16, and 22, and the phase response (minutes) was calculated as the difference between the pre- and postpulse regression lines. There were no significant differences between groups at any of the tested time points. Sample sizes are indicated below each time point: WT (n = 5-7), NERKI (n = 7-8), and ERKO (n = 14-18) for each time point.
Number of SCN Fos-Positive Cells Is Not Influenced by Genotype
We analyzed the distribution and number of Fos-positive cells in the total SCN and its 2 subregions, shell and core. The number of Fos-positive cells is higher in animals pulsed with light during the night compared to the day (Fig. 4). Within a time point, we did not detect an effect of genotype on the amount of Fos-positive cells for either males or females. This was true for all 3 regions.
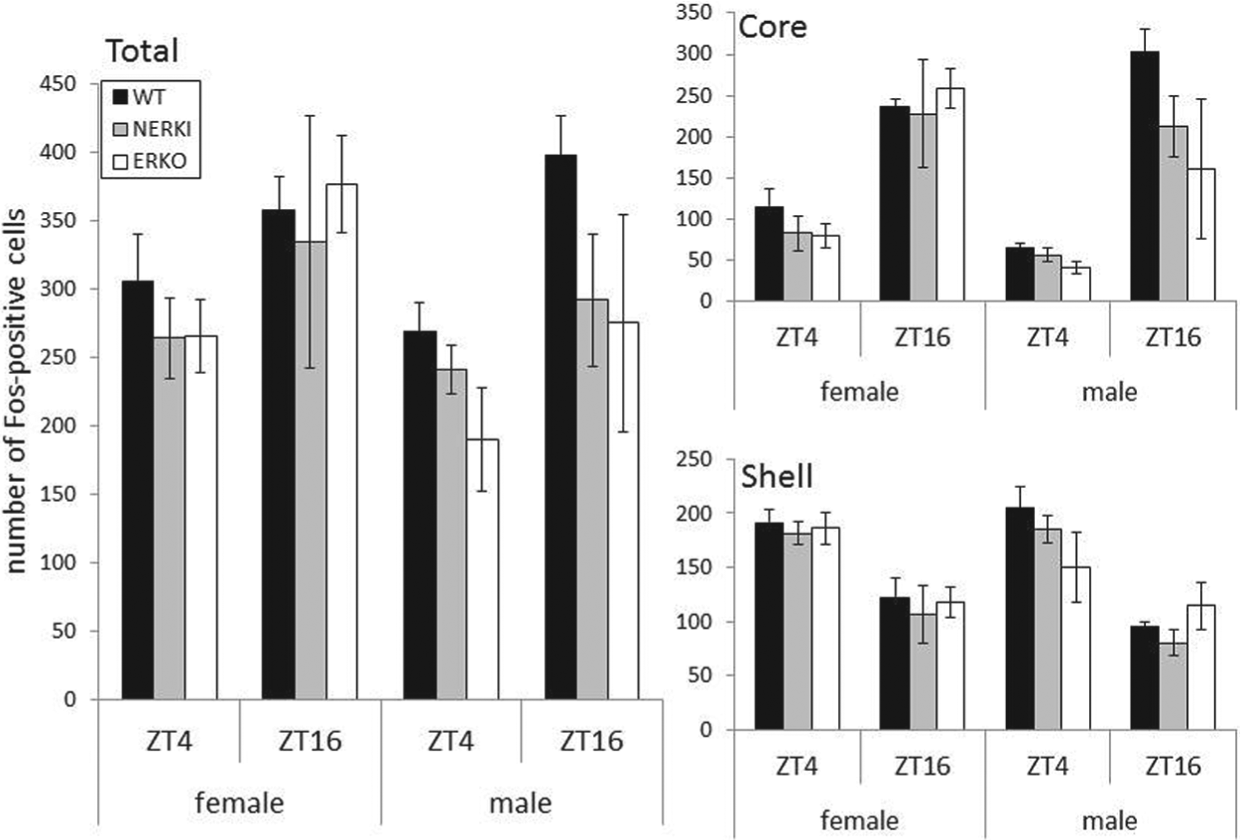
Number of Fos-expressing cells (mean ± SEM) following a light pulse at ZT 4 or ZT 16 for WT, NERKI, and ERKO males and females. Data are from the total SCN (left panel), core (top right), and shell (bottom right) regions of the SCN.
Discussion
There is growing evidence that the distribution and amplitude of daily activity and the SCN response to light, including phase-response curves, are modified by the presence of gonadal hormones (Karatsoreos et al., 2007; Brockman et al., 2011). Our major finding here is that estrogens mediate the response to light cues via both classical ERE-mediated transcription and nonclassical pathways. Female mice that developed in the absence of a functional ESR1 (ERKO) had significant alterations in their responsiveness to a photic cue when compared to their WT littermates. The magnitude and difference in the response to a light pulse were time dependent as WT females had a larger phase shift than ERKO females during the early subjective evening (ZT 16), whereas ERKO females had a larger phase shift than WT when given a pulse in the late subjective evening (ZT 22). In the absence of ESR1 action via classical ERE-dependent mechanisms, NERKI mice have a phase shift that is intermediate between that of ERKO and WT animals. Together these data suggest that the estrogenic modulation of photic responsiveness is due in part to its actions at ESR1, at least in females.
There may be sex differences in estrogen receptor modulation of entrainment to light identified in the current study. WT females have a greater phase delay early in the subjective night (ZT 16) than males. Interestingly, in males, there were no genotype effects on the behavioral phase response curve (Fig. 3), whereas there were in females (Fig. 1). It is possible that in males, estrogen signaling mechanisms, either during development or in adulthood, are not essential for circadian rhythm function. Recent literature suggests that androgens have a significant effect on circadian rhythms in males. Gonadectomized male mice have significantly larger phase shifts in response to a light pulse given early in the subjective night than intact animals; replacement of androgens restores the magnitude of the phase shift to that of intact animals (Karatsoreos et al., 2011). Further, male mice have significantly more androgen receptors than females in the SCN (Iwahana et al., 2008). It is intriguing to speculate that there are sex-specific roles for estrogens and androgens in modulating circadian rhythms and daily activity patterns that are mediated by the presence of steroid receptors in the SCN.
We also examined the potential neuronal location for the action of estrogens on circadian function. Sex differences in the magnitude of behavioral phase shifts, coupled with sex differences in steroid receptor expression in the SCN, led us to hypothesize that estrogens modify the response of the SCN to light. Both ESR1 and ESR2 are expressed to varying degrees in the mouse SCN (Mitra et al., 2003; Vida et al., 2008), although the number of estrogen receptors in the SCN is relatively low. Likewise, in humans, there are estrogen receptors of both subtypes in the SCN, with significantly greater ESR1 expression in females relative to males (Kruijver et al., 2003). To date, no research has identified a role for ESR2 in the regulation of timekeeping function. In rats, the expression of transcription factors in the SCN both in response to light pulse and in an LD cycle is mediated by circulating estrogens (Abizaid et al., 2004; Peterfi et al., 2004). We did not detect an effect of genotype on Fos-positive cell number in the SCN of male or female mice. Our data are limited, however, in that we do not have Fos expression from unpulsed animals. It remains possible that baseline gene or protein levels or the magnitude of light-induced gene expression are modified by estrogen. Female rats with circulating estradiol have higher Fos expression in the SCN compared to animals treated with cholesterol (Abizaid et al., 2004). Estrogens may also indirectly modulate circadian functions by acting at brain regions that express estrogen receptors and send their projections to the SCN (de la Iglesia et al., 1999). The medial preoptic region, which is rich with ESR1, is one region that targets the SCN and is implicated in the regulation of activity; animals with lesions of the SCN still respond to estradiol with an increase in wheel running (King, 1979; Fahrbach et al., 1985). However, these previous studies only identified activity amount as being regulated by the preoptic region. Circadian outputs including tau are regulated by the SCN and can be modified by circulating estrogen; thus, the possibility remains that estrogen acts on SCN cells directly to regulate these endogenous functions. It will be interesting to determine if connectivity within the SCN, particularly between the light-responsive core and the rhythmic shell, is altered by developmental or circulating exposure to estradiol.
This study reveals that estrogen signaling pathways affect the photic responsiveness of the timekeeping system, but it does not reveal the target or timing for this estrogen exposure. Changes in circadian function could be due to circulating steroid hormones, developmental effects of estrogen receptor signaling either on the pacemaker or other neural targets, or some combination of these factors. It is also important to consider the impact of global knock-outs or knock-ins on physiology, which may indirectly contribute to the results. Thus, our data need to be interpreted in light of these potential underlying mechanisms. It is likely that both organizational and activational effects of estradiol contribute to endogenous timekeeping functions. Ovariectomized WT female mice, which lack circulating estrogens, have a smaller phase shift in response to a light cue given in the early subjective evening than intact counterparts (Brockman et al., 2011). Further, aromatase knock-out animals that develop in the absence of estradiol have altered PRCs, activity onset, and tau when compared to WT animals. These differences remain following gonadectomy, indicating an organizational effect of estrogen signaling on adult circadian function. Future experiments will aim to distinguish developmental and activational contributions of estrogens to the expression of circadian rhythms in adulthood. Understanding the mechanism of gonadal hormone regulation of circadian rhythms and how hormones contribute to sex differences in daily behaviors in rodent studies will provide insight as to the mechanisms underlying the sex differences in rhythmic properties in humans.
Footnotes
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
