Abstract
Siberian hamsters (Phodopus sungorus) exhibit robust seasonal rhythms of reproduction driven by changes in day length. Day length is encoded endogenously by the duration of nocturnal melatonin (Mel) secretion from the pineal gland. Short duration Mel signals stimulate whereas long duration Mel signals inhibit reproduction. The mechanism by which Mel regulates the reproductive axis has not been fully characterized. In Siberian hamsters, the thyroid hormone triiodothyronine (T3) is thought to be part of the photoperiodic mechanism. The availability of T3 is decreased in hamsters housed in short day lengths, and injections of exogenous T3 stimulate testicular growth in short-day (SD) Siberian hamsters. Thus, T3 acts as a neuroendocrine intermediate between the Mel rhythm and the reproductive axis. The RFamides kisspeptin (Kiss1) and gonadotropin-inhibitory hormone (GnIH) also act as a link between the Mel rhythm and the reproductive axis. Expression of both of these neuropeptides is regulated by photoperiod and Mel. Kiss1 stimulates, and GnIH inhibits, the reproductive axis in long-day housed hamsters. It remains unknown whether T3 acts through changes in RFamide expression in the regulation of reproduction or whether these molecules act independently of one another. We tested the hypothesis that exogenous T3 administered to SD hamsters, a treatment that stimulates testicular growth, would also result in alterations in the patterns of Kiss1- and GnIH-immunoreactivity. Administration of T3 to SD hamsters resulted in significant testicular growth as well as a long day–like pattern of RFamide peptide expression. Thus, exogenous T3 elicited increased numbers of Kiss1-positive cells in the hypothalamic anteroventral periventricular nucleus, decreased numbers of Kiss1-positive cells in the arcuate nucleus, and a greater number of GnIH-positive cells in the dorsomedial hypothalamus compared with SD controls. The results are consistent with the hypothesis that T3 elicits alterations in the reproductive axis through alterations in RFamide peptide expression.
Animals that inhabit temperate latitudes experience predictable seasonal changes in resource availability that affect the likelihood of offspring survival (Bronson, 1988). Many animals, including Siberian hamsters (Phodopus sungorus), have evolved endogenous timing mechanisms that enable them to anticipate such changes and to make seasonal adjustments in physiology and behavior that may help them cope with these seasonal environmental changes (for review, see Prendergast et al., 2002). Thus, these hamsters synchronize their reproductive effort with the environment so that their young are born when conditions are most favorable for offspring survival. Changes in day length are often used as an exogenous cue to track the seasons (for review, see Paul et al., 2008). Day length is encoded by the rhythm of pineal melatonin (Mel) secretion, the duration of which is directly proportional to the duration of the dark period in the light-dark cycle (Carter and Goldman, 1983). Mel secretion is driven by the mammalian circadian clock located in the suprachiasmatic nucleus (SCN) of the hypothalamus (Stephan and Zucker, 1972). In Siberian hamsters, exposure to short days (SD) results in a long duration of Mel secretion that is inhibitory to the reproductive axis, whereas the short duration Mel rhythm exhibited under long days (LD) is stimulatory to the reproductive axis (Carter and Goldman, 1983).
Mel affects reproduction through alterations in the hypothalamic-pituitary-gonadal (HPG) axis, ultimately by altering the secretion of gonadotropin-releasing hormone (GnRH). GnRH acts upon the anterior pituitary to stimulate the secretion of the gonadotropins and thus stimulate the production of sex steroids and gametogenesis in the gonads. SD patterns of Mel secretion inhibit the HPG axis, although the exact mechanism that mediates these effects remains unclear since GnRH-secreting neurons do not express Mel or sex steroid receptors (Clarke et al., 2009). This suggests that there are neuroendocrine intermediates between the Mel rhythm and the HPG axis.
Previous experiments have indicated that thyroid hormones serve as a link between the Mel rhythm and Mel-dependent seasonal changes in reproduction in multiple species across taxa (Viguie et al., 1999; Yoshimura et al., 2003; Watanabe et al., 2004; Saita et al., 2005; Yasou et al., 2005, 2006; Revel et al., 2006; Freeman et al., 2007; Klosen et al., 2013). Exogenous triiodothyronine (T3) administered to Siberian hamsters housed in SD elicits testicular growth similar in magnitude to transfer to long day lengths (Freeman et al., 2007). Hamsters exposed to SD exhibit reductions in type-2 deiodinase (Dio2) gene expression and increases in type-3 deiodinase (Dio3) gene expression in the ependymal cell layer surrounding the third ventricle (Watanabe et al., 2004; Revel et al., 2006; Barrett et al., 2007; Prendergast et al., 2013), potentially decreasing local availability of T3. Dio2 catalyzes the conversion of thyroxine (T4) to T3 by outer ring deiodination of T4. Thus, a reduction in the Dio2 gene results in a tissue-level reduction of T3 (Yoshimura et al., 2003). Dio3 inactivates both T3 and T4 by inner ring deiodination. The expression of Dio3 is increased in Japanese quail (Yasuo et al., 2005) and Siberian hamsters (Barrett et al., 2007; Prendergast et al., 2013) exposed to SD, likely leading to a reduction in T3 levels. Thus, Mel may affect the HPG axis through its actions on the enzymes Dio2 and Dio3, which in turn determine the availability of T3 in key brain nuclei. Recent results from Prendergast et al. (2013) indicate that increased Dio3 expression serves as the primary regulator of SD-dependent reductions in T3 availability in Siberian hamsters. Reversing the SD-induced decrease in T3 with injections of exogenous T3, either peripherally (Freeman et al., 2007) or in the brain (Barrett et al, 2007), results in testicular growth in photoinhibited Siberian hamsters, although the specific mechanism remains uncharacterized.
Another family of molecules that serve as neuroendocrine intermediates in the regulation of reproduction across taxa are the RFamides, including kisspeptin (Kiss1) and gonadotropin-inhibitory hormone (GnIH; also known as RFamide related peptide or RFRP; see Kriegsfeld et al., 2006; Johnson et al., 2007; Murakami et al., 2008; Bentley et al., 2006, 2010). Both of these peptides are important in the regulation of the HPG axis, and their expression is altered in response to environmental stimuli, including changes in photoperiod (Greives et al., 2007; Mason et al., 2007; Mikkelsen and Simonneaux, 2009; Revel et al., 2009; Roa et al., 2011). Kiss1 and GnIH neurons also express steroid receptors (estrogen receptor alpha [ERα] and androgen receptor), implicating them in the steroid sensitivity of the HPG axis (reviews in Oakley et al., 2009; Bentley et al., 2010).
Kiss1 is a neuropeptide that is encoded by the Kiss-1 gene. Kiss1 expression has been observed across taxa including mammals, birds, amphibians, and fish (see Oakley et al., 2009). Expression in mammals is limited to the arcuate nucleus (Arc) and the anteroventral periventricular nucleus (AVPV), both of which are in close proximity to the third ventricle (Greives et al., 2007; Mason et al., 2007; Revel et al., 2009; Roa et al., 2011). Multiple lines of evidence suggest that Kiss1 acts directly to stimulate GnRH secretion. First, Kiss1 receptors (also known as GPR54) are expressed by most GnRH neurons (Irwig et al., 2004). Second, Kiss1 neuronal projections are in close proximity to GnRH neurons (Smith et al., 2008). Third, Kiss1 acts directly to depolarize GnRH neurons in vitro (Oakley et al., 2009). Along with these direct effects, Kiss1 may also act indirectly on intermediary neurons, such as GABAergic cells, to regulate GnRH secretion (Oakley et al., 2009). GnIH expression is restricted to the dorsomedial hypothalamus (DMH) of Siberian hamsters and rats (Bentley et al., 2010), and GnIH fibers appose GnRH and Kiss-1 fibers (Rizwan et al., 2012). GnIH neurons also express ERα receptors, suggesting that they also serve as sites of steroid feedback (Gingerich et al., 2009). Importantly, injecting Syrian hamsters and Siberian hamsters housed in LD with GnIH reduces circulating LH levels and reduces sexual behaviors (Kriegsfeld et al., 2006; Ubuka et al., 2012), consistent with an inhibitory role regarding the HPG axis. In contrast, exogenous GnIH stimulates the reproductive axis in hamsters housed in SD (Ancel et al., 2012; Ubuka et al.,2012), suggesting that photoperiod history determines the response to this neuropeptide.
It remains to be determined whether T3-induced stimulation of the reproductive axis involves alterations in RFamide peptide expression. We tested the hypothesis that exogenous T3 will elicit changes in the expression of Kiss1 and GnIH in addition to eliciting testicular growth in SD male hamsters.
Materials and Methods
Subjects
Male Siberian hamsters (originally supplied by Irving Zucker at the University of California, Berkeley) were gestated and raised in 16L:8D (16L; light offset at 1700 h CST), weaned and separated by sex at 17 to 19 days of age, and housed individually in polypropylene cages (29 × 18 × 13 cm) at 22 ± 1 °C for the duration of the experiment. Animals had access to ad libitum food (8640 Teklad 22/5 Rodent Diet, Teklad Diets, Madison, WI) and water.
T3 Injections
T3 injections consisted of 0.5 µg of 3, 3′, 5 triiodo-L-thyronine (product #564605, Sigma-Aldrich, St. Louis, MO) dissolved into 0.1 mL of physiological saline, while control injections were 0.1 mL of saline. Injections were administered s.c. between 1500 and 1600 h CST.
Adult Siberian hamsters (n = 19) were housed in either LD (n = 4) or SD (n = 15; 10L; light offset 1700 h CST) photoperiods for 5 weeks, a sufficient interval for the 10L groups to exhibit reproductive responsiveness to short day lengths (estimated testicular volume [ETV] <200 mm3; see below). One group of SD animals received daily T3 injections (n = 9), while the other 2 groups were subject to saline injections to serve as LD (n = 4) and SD (n = 6) controls. Based on the response of SD hamsters to identical T3 injections in a previous experiment in which the maximal testicular response to T3 was observed after 3 weeks of injections (Freeman et al., 2007), treatments in the present experiment were administered for 3 weeks. Body mass and ETV were determined weekly during the treatment period.
Reproductive Response
Estimated testis volume (ETV; width2 × length of left testis) was used to assay the gonadal response to changes in photoperiod (Gorman and Zucker, 1995) and in response to T3 injections. ETV was determined with analog calipers to the nearest 0.1 mm while animals were under light anesthesia with isoflurane vapors (IsoSol, Vedco Inc., St. Joseph, MO). All procedures were approved by the Animal Care and Use Committee at the University of Memphis.
Histology
At the termination of the experiment, brains were removed and prepared for histology by spinning immersion (cf. Kramer et al., 2008). Sections of 30 µm were cut on a sliding microtome, and all tissue was processed in a single IHC run (cf. Kramer et al., 2008). The brains were stained using anti-Kiss1 antibody (1:2000, Kisspeptin, T4771, lot #040380-5, Bachem, Bubendorf, Switzerland) with 3,3′-diaminobenzidine (DAB) as the chromogen. The Kisspeptin antibody has previously been verified for use in Siberian hamsters (Greives et al., 2007) and recognizes both Kiss1 and GnIH, but due to the specific and nonoverlapping localization of cell bodies that express each peptide in the brain, we were able to distinguish Kiss1 cells from GnIH cells (Revel et al., 2009). The tissue was observed under a light microscope to quantify the number of cell bodies positive for each neuropeptide. RFamides were quantified by counting the number of immunopositive neurons in every fourth section of each animal (cf. Greives et al., 2007).
Statistical Analyses
Both body mass and ETV were analyzed using repeated-measures ANOVA. Quantification of RFamides in the T3-treated hamsters was restricted to those hamsters that exhibited stimulation of testicular growth, as defined by ETV greater than 400 mm2, in response to T3 injections (LD saline [n = 4], SD saline [n = 6], and SD T3 [n = 4]). RFamide expression was analyzed using 2-way ANOVA. Post hoc analyses consisted of Fisher’s PLSD test. Differences were considered significant if p < 0.05.
Results
Testis and Body Mass Response
There was a significant effect of treatment on ETV (repeated-measures ANOVA; F2,16 = 31.7, p < 0.0001, Fig. 1). Hamsters administered exogenous T3 exhibited significantly larger testes than SD control hamsters administered saline (Fisher’s PLSD, p = 0.03). The LD saline control group exhibited significantly larger testes than either SD group (Fisher’s PLSD, p = 0.0001 for both comparisons). Body mass did not differ significantly among groups (repeated-measures ANOVA; F2,16 = 0.89; mean average body mass ranged from 29.5 to 32.9 g; p > 0.4).
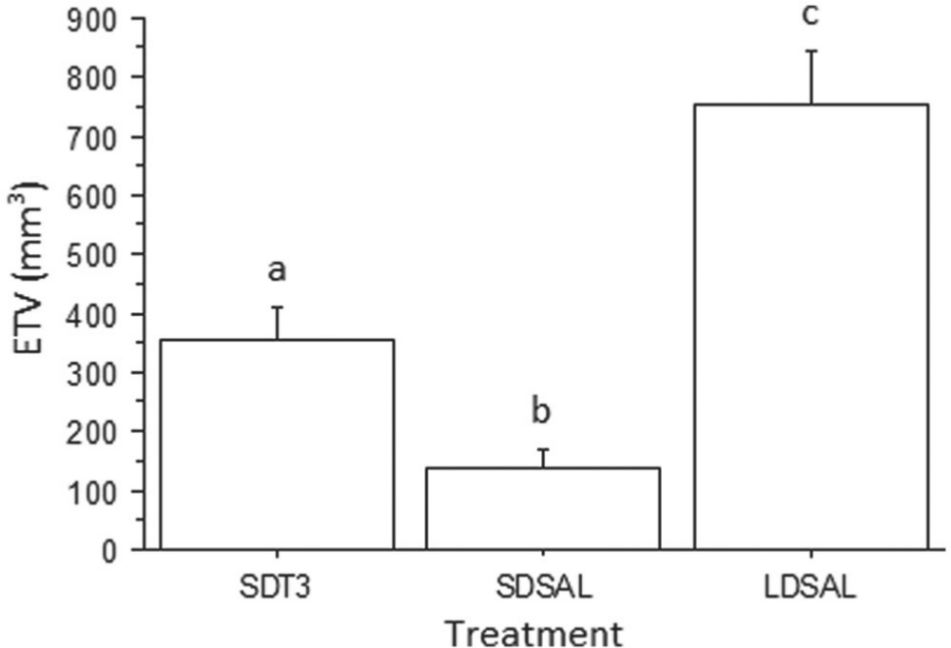
Mean (±1 SE) estimated testicular volume (ETV) following 3 weeks of treatment. Different letters indicate significant difference between treatments (p < 0.0001). SDT3 refers to short-day hamsters treated with T3; SDSAL and LDSAL refer to the short- and long-day control groups injected with saline vehicle, respectively.
RFamide Peptide Expression
AVPV and Arc
There was a significant effect of treatment on RFamide peptide expression in the Arc (ANOVA; F2,11 = 14.66, p = 0.0002, Fig. 2A) and AVPV (ANOVA; F2,11 = 7.862, p = 0.0046, Fig. 2B). SD-housed control hamsters exhibited significantly fewer RFamide-positive cells in the AVPV than the LD controls (Fisher’s PLSD, p = 0.0015). SD hamsters injected with T3 exhibited significantly more RFamide-positive cells than the SD saline control group (Fisher’s PLSD, p = 0.020). SD-housed control hamsters exhibited significantly more RFamide-positive cells in the Arc than the LD controls (Fisher’s PLSD, p = 0.0061). SD hamsters injected with T3 exhibited significantly fewer RFamide-positive cells in the Arc compared with the SD saline control group (Fisher’s PLSD, p = 0.0035).
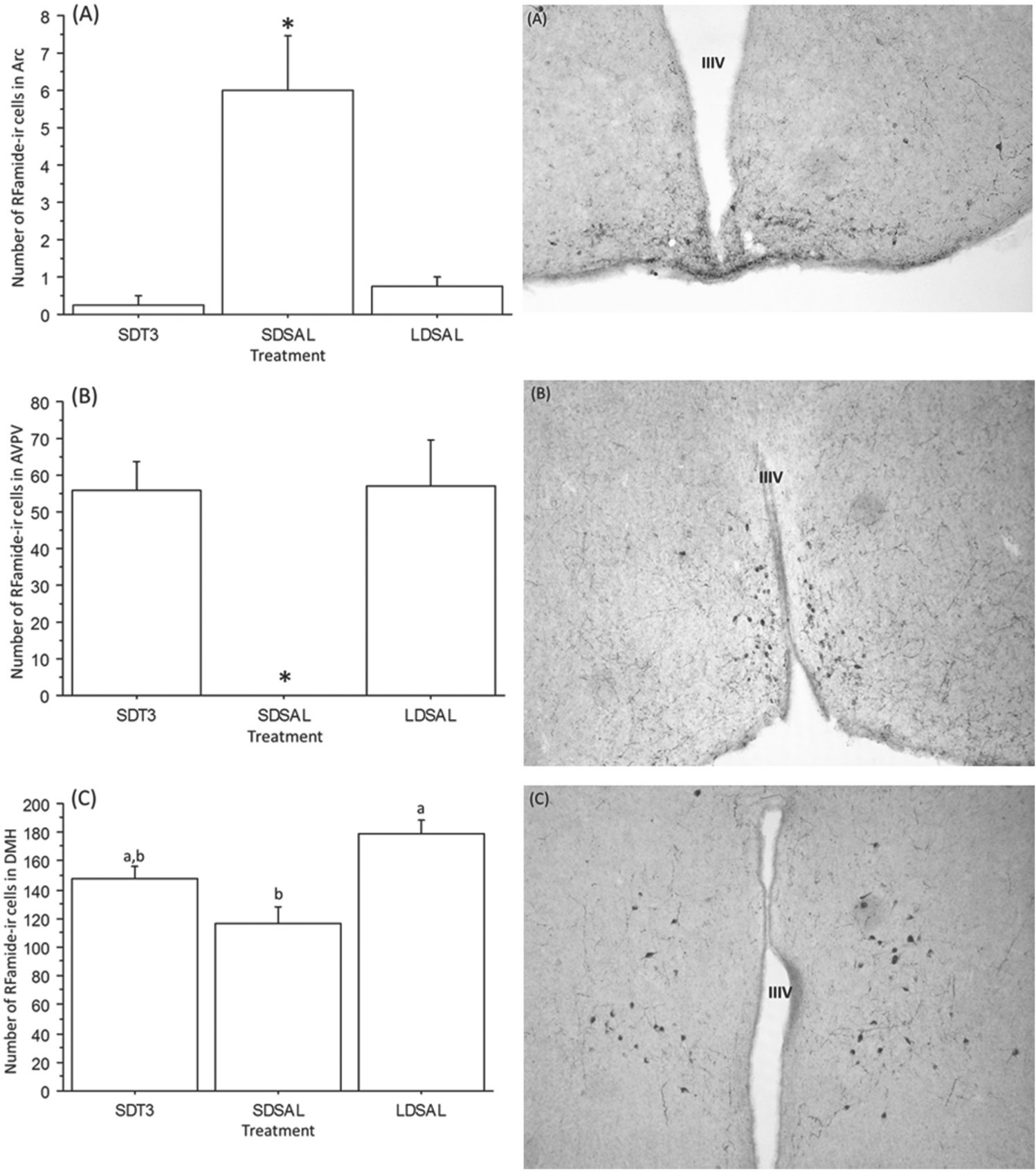
(Left) Number of RFamide-ir cells (±SE) in the arcuate nucleus (Arc) (A), anteroventral periventricular nucleus (AVPV) (B), and dorsomedial hypothalamus (DMH) (C). Bars with asterisks or different letters differ significantly (p < 0.006 for each comparison); (B) AVPV SDSAL (mean = 0.167 ± 0.167). SDT3 refers to short-day hamsters treated with T3; SDSAL and LDSAL refer to the short- and long-day saline-injected control groups, respectively. (Right) Representative photomicrographs illustrating RFamide-ir cells in the (A) Arc, (B) AVPV, and (C) DMH. IIIV refers to the third ventricle. Adobe Photoshop was used to rotate, crop, and adjust the brightness of images.
DMH
There was a significant difference in RFamide expression in the DMH (ANOVA; F2,11 = 8.8, p = 0.005, Fig. 2C). The number of RFamide-ir cells in the DMH was significantly lower in the SD control group than in the LD control group (p = 0.03). Administration of T3 to SD-housed hamsters resulted in RFamide peptide expression that was intermediate to the SD and LD saline control groups; thus, the number of RFamide-positive cells in the DMH of T3-treated SD hamsters did not differ from the number of cells in the LD or SD saline controls (p > 0.06 for each comparison).
Discussion
The results indicate that exogenous T3 resulted in both testicular growth and alteration in RFamide peptide expression in SD-housed hamsters. Thus, T3 administration resulted in recrudescence of the testes and increased the number of RFamide cells in the AVPV and DMH, respectively, and decreased the number of RFamide cells in the Arc. Since Kiss1and GnIH neurons do not co-localize, Kiss1 cells in the AVPV and Arc are distinguishable from GnIH cells in the DMH (Revel et al., 2009). Administration of T3 did elicit the full LD phenotype in regard to RFamide expression in the AVPV but not in the DMH. T3 treatment resulted in intermediate testis size, similar to findings from a previous experiment that used identical T3 injections (Freeman et al., 2007). Our results support the hypothesis that T3 stimulates gonadal growth and extend this finding by indicating that exogenous T3 alters RFamide peptide expression in a manner similar to LD exposure. This result suggests that rather than representing 2 independent neuroendocrine pathways, T3 and the RFamides may represent components of a single pathway in which Mel-induced alterations in T3 availability regulate gonadal status through alterations in RFamide peptide expression. Treatment with T3 resulted in numbers of RFamide cells that were no different from the LD control group in the AVPV and Arc but were intermediate to the SD and LD control groups in the DMH, which were characterized by fewer and more RFamide positive neurons, respectively. This supports the hypothesis that T3 stimulates the reproductive axis via up-regulation of Kiss1 in the AVPV (Oakley et al., 2009). Kiss1 expression in the Arc was reduced by T3 injection as it is in response to LD (Greives et al., 2007). Interestingly, the observed stimulatory effect of T3 injections on the number of GnIH cells was the opposite of what was expected given that GnIH is thought to act as an inhibitor of the HPG axis (Bentley et al., 2010). Thus, it was expected that T3 administration would result in decreased numbers of GnIH cells, consistent with disinhibition of the HPG axis. This seemingly paradoxical pattern of GnIH expression has been noted in other experiments that documented increases in GnIH expression under conditions when the reproductive axis was stimulated. For example, Ancel et al. (2012), Revel et al. (2009), and Ubuka et al. (2012) noted elevated GnIH expression under LD exposure in Syrian and Siberian hamsters, respectively. Ancel et al. (2012) and Ubuka et al. (2012) found that injections of GnIH stimulated the reproductive axis by inducing LH secretion of SD-housed Syrian and Siberian hamsters. Thus, the response to GnIH appears to depend on the individual’s photoperiod history. It is possible that GnIH acts to counterbalance the actions of Kiss1 on the HPG axis; thus, it might be expected that photoperiod-dependent changes in GnIH expression may parallel changes in Kiss1 expression. One recent model of the interaction between Kiss1 and GnIH in the control of reproduction suggests that these peptides act through reciprocal connections to inhibit one another; for example, GnIH neurons project onto kisspeptin cells that express the GnIH receptor GPR147 (Parhar et al., 2012; Rizwan et al., 2012), so it might be expected that their expression changes in parallel.
As in a previous experiment (Freeman et al., 2007), T3 administration did not elicit complete gonadal regrowth compared with results in hamsters exposed to LD for an extended period; instead, it resulted in an intermediate gonad size, significantly different from both the SD and LD control groups. The degree of gonadal growth in response to 3 weeks of T3 injections was similar to the pattern of gonadal growth elicited by exposure of SD hamsters to 2 weeks of long day lengths, indicating that T3 mimics exposure to a long photoperiod (Kauffman and Zucker, 2002). This partial recrudescence of the gonads could be due to multiple factors. For example, the dose or duration of T3 administered may have been insufficient to elicit full LD gonadal stimulation; although some T3 administered peripherally does enter the CNS (Palha et al., 2002), the amount of the T3 that crossed the blood-brain barrier may have been insufficient. Interestingly, T3 injections also resulted in an intermediate number of RFamide cells in the DMH compared with LD and SD control hamsters, suggesting that testicular stimulation may depend in large part on GnIH activity originating in the DMH. Last, it is likely that multiple cues are necessary for the full transition from the SD to the LD phenotype. For example, the transfer of hamsters from an SD to an LD photoperiod results in the rapid truncation of the pineal Mel rhythm (Paul et al., 2008), which, in turn, actively stimulates gonadal growth (Carter and Goldman, 1983), likely by acting at the NRe (Teubner and Freeman, 2006; Teubner et al., 2008), whereas the present treatment did not alter Mel duration. Future experiments could determine whether stimuli that result in complete gonadal recrudescence also increase the number of GnIH cells in the DMH to LD values.
Previous experiments across taxa indicate that the enzymes Dio2 and Dio3 are involved in T3 production and inactivation, respectively, and that their expression is sensitive to photoperiod and Mel (Watanabe et al., 2004; Yasuo et al., 2005). In hamsters, exposure to short day lengths may result in a decrease in the expression of Dio2 and increases in the expression of Dio3. This expression pattern results in lower T3 availability in short day lengths. A hypothesis consistent with these findings is that elevated T3 supports the long day phenotype whereas the decreased T3 availability elicits the short day phenotype. The present results support this hypothesis in Siberian hamsters; thus, both exposure to long day lengths and T3 injections result in gonadal stimulation and alteration in the expression of RFamide peptides in photoinhibited Siberian hamsters. Similar results were obtained in response to intracerebroventricular injections of thyroid stimulating hormone (TSH) in SD Siberian hamsters (Klosen et al., 2013). Thus, exogenous TSH resulted in testicular recrudescence, possibly mediated by alterations in Kiss1 and GnIH activity. Klosen et al. (2013) postulate that Mel-dependent alterations in TSH secretion from pars tuberalis cells drive alterations in T3 availability, resulting in recrudescence of the gonads via alterations in RFamide activity. The present results are consistent with this model. While the results of this experiment are consistent with the model put forth by Klosen et al., it is possible that alterations in RFamide peptide expression in the present experiment were the result of T3-induced increases in circulating gonadal steroids rather than a direct effect on RFamide neurons (Simonneaux et al., 2009; Greives et al., 2008).
Conclusions
Our results indicate that several weeks of exogenous T3 injections stimulate the reproductive axis of Siberian hamsters similar to the effects of 2 weeks of long day length exposure (Kauffman and Zucker, 2002). Prior results indicate that exposure to LD increases production of Dio2 (Yoshimura et al., 2003) and decreases circulating levels of Dio3 in the brain (Yasuo et al., 2005; Barrett et al., 2007). These alterations result in increased T3 availability, which stimulates the HPG axis. Our results further suggest that the mechanism by which T3 stimulates the HPG axis includes alterations in RFamide peptide expression. Several questions remain unanswered, however. For example, the mechanism by which T3 alters RFamide peptide expression remains unknown. Additionally, it remains to be determined whether the RFamides alter Dio2 and Dio3 expression, thereby regulating T3 availability.
Footnotes
Acknowledgements
The authors thank the animal care staff at the University of Memphis for the care of the animals. They also thank S. Schoech for comments on a previous version of the manuscript. Funds were provided by the University of Memphis.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
