Abstract
Melatonin is endogenously produced and released in humans during nighttime darkness and is suppressed by ocular light exposure. Exogenous melatonin is used to induce circadian phase shifts and sleep. The circadian phase-shifting ability of a stimulus (e.g., melatonin or light) relative to its timing may be displayed as a phase response curve (PRC). Published PRCs to exogenous melatonin show a transition from phase advances to delays approximately 1 h after dim light melatonin onset. A previously developed mathematical model simulates endogenous production and clearance of melatonin as a function of circadian phase, light-induced suppression, and resetting of circadian phase by light. We extend this model to include the pharmacokinetics of oral exogenous melatonin and phase-shifting effects via melatonin receptors in the suprachiasmatic nucleus of the mammalian hypothalamus. Model parameters are fit using 2 data sets: (1) blood melatonin concentration following a 0.3- or 5.0-mg dose, and (2) a PRC to a 3.0-mg dose of melatonin. After fitting to the 3.0-mg PRC, the model correctly predicts that, by comparison, the 0.5-mg PRC is slightly decreased in amplitude and shifted to a later circadian phase. This model also reproduces blood concentration profiles of various melatonin preparations that differ only in absorption rate and percentage degradation by first-pass hepatic metabolism. This model can simulate experimental protocols using oral melatonin, with potential application to guide dose size and timing to optimally shift and entrain circadian rhythms.
Keywords
Mathematical modeling of biological processes may improve understanding of underlying physiology, suggest areas of further investigation, and provide useful predictions. Melatonin is an excellent candidate for modeling because of the growing body of published research and its many current uses, including treatment of jet lag and phase-shift disorders (Sanchez-Barcelo et al., 2010). Current clinical studies with only 1 or 2 dose sizes cannot guide dosing of the dozens of over-the-counter dose sizes and preparations available to the public. We chose to model the circadian phase resetting effects of exogenous melatonin to provide a method for guiding effective timing and dosage of administration relative to preparation.
Melatonin
The hormone melatonin (
Exogenous melatonin is currently used for shifting circadian phase and inducing sleep. Here we focus on the effects on circadian phase. Phase shifts are mediated by melatonin receptors in the SCN (Reppert et al., 1988); the direction and magnitude of phase shifts depend on the initial phase at melatonin delivery (Lewy et al., 1998; Burgess et al., 2008). Phase response curves (PRCs) have been determined for oral doses ranging from 0.5 mg to 3.0 mg (Lewy et al., 1992; Zaidan et al., 1994; Lewy et al., 1998; Burgess et al., 2008; Burgess et al., 2010). These PRCs show relatively weak (type 1) resetting (Winfree, 1980); the point of transition from phase advances to phase delays occurs approximately 1 h after dim light melatonin onset (DLMO), that is, approximately 1 h before habitual bedtime in normally entrained individuals. By comparison, type 1 PRCs to ocular light stimuli (e.g., Khalsa et al., 2003) show this transition point occurring close to the core body temperature nadir, approximately 2 h before habitual wake. The phase-resetting effects of light and exogenous melatonin are therefore approximately 8 h out of phase. Whereas light causes phase delays during early biological night and phase advances during late biological night, exogenous melatonin has the opposite effect. Since light exposure and melatonin production typically occur at different times of day, endogenous melatonin may augment light-based entrainment (Lewy et al., 1998).
Mathematical Modeling
Brown et al. (1997) introduced a physiologically based model of the diurnal variation of pineal and plasma melatonin concentration for calculating melatonin phase and peak amplitude. This model also provided information about melatonin infusion and clearance rates within the pineal gland and blood plasma. To incorporate the ocular light effects on melatonin production and rhythm, this model was integrated with an existing mathematical model of the effects of light on the human circadian pacemaker (St. Hilaire et al., 2007a, 2007b).
Here, we extend this model to incorporate (1) pharmacokinetics of two preparations of oral melatonin and (2) effects of combined endogenous and exogenous melatonin on the circadian pacemaker to predict the phase shift induced by a given dose at any circadian phase.
Material and Methods
In this section, we introduce previously published model equations, our modifications to these equations, and new model equations, which together comprise the current model. Figure 1 is a schematic of our new model. Table 1 summarizes the data sets used.
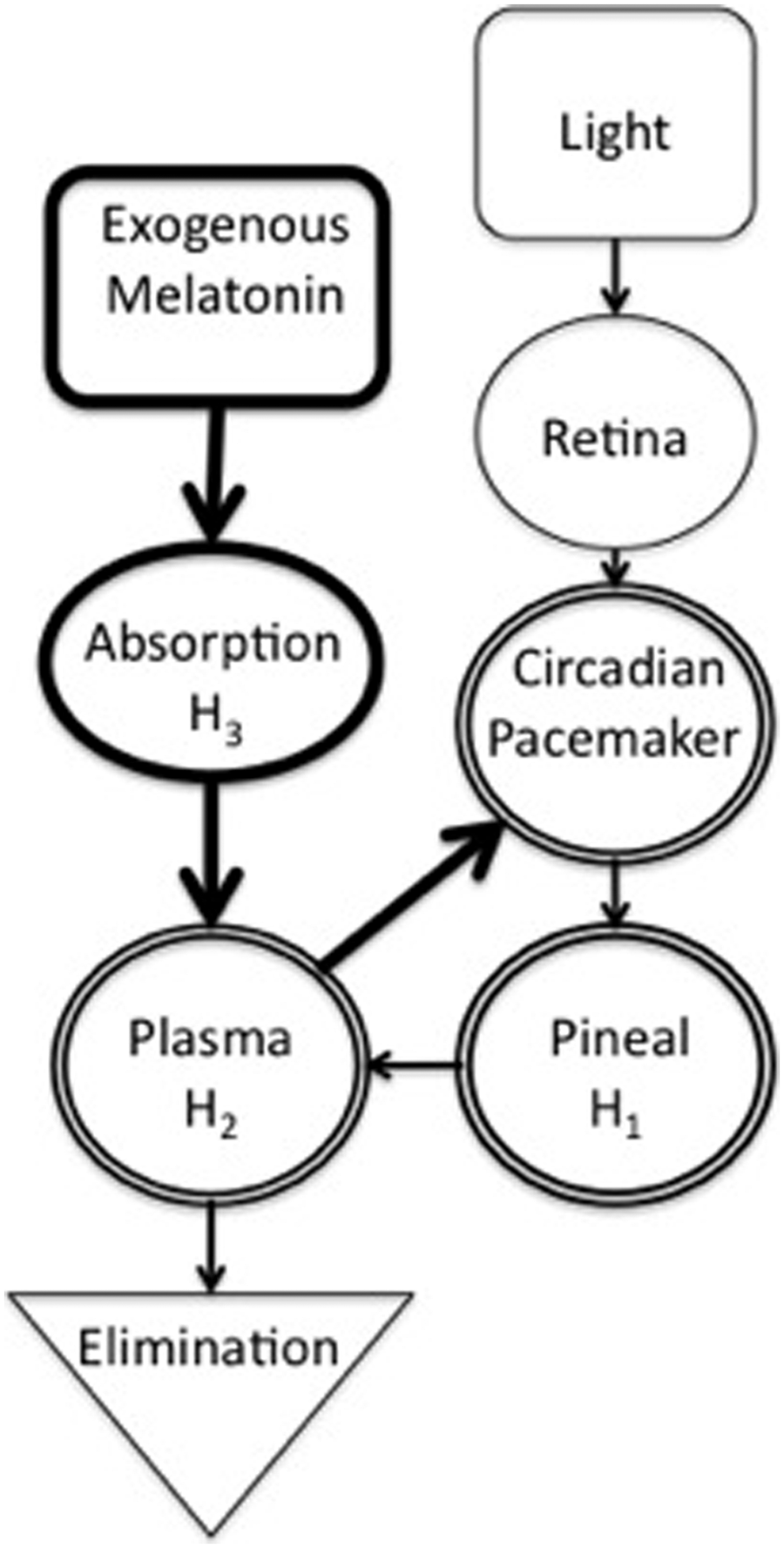
Schematic diagram of the multicompartment melatonin model. A drive generated by light is transmitted to the circadian pacemaker via the retinohypothalamic tract, generating light-dependent phase resetting. Light also suppresses endogenous melatonin production at the pineal via the circadian pacemaker. Melatonin synthesis in the pineal is initiated by signals from the circadian pacemaker. Melatonin then diffuses into the plasma. Melatonin in the plasma has a phase-resetting effect on the circadian pacemaker. Plasma levels are increased by exogenous melatonin doses and decreased by elimination via the kidneys. Before reaching the plasma and acting on the circadian pacemaker, exogenous melatonin goes through an absorption stage during which ~90% of the dose is degraded. Bold lines indicate model additions from previously published work. The double lines around the circadian pacemaker, pineal, and plasma compartments indicate that together they comprise the pathway of endogenous melatonin production.
Summary of data sets used in this article.

Model of Light’s Effects on the Circadian Pacemaker
The Kronauer et al. (1999) model of the effects of light on the human circadian pacemaker includes a light preprocessor in the retina, Process L, and the circadian pacemaker in the SCN, Process P. When light reaches the retina, it activates photoreceptors that project via the retinohypothalamic tract to SCN neurons.
In the model, photoreceptors exist as “activated” or “ready” (since transition time between states is negligible). Photoreceptors are activated at rate
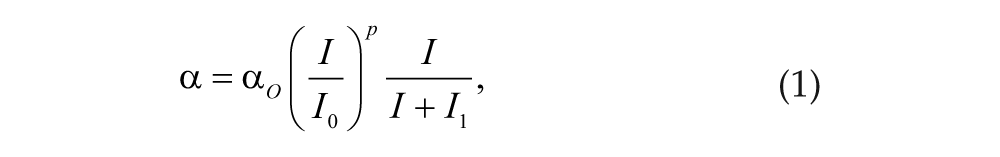
where
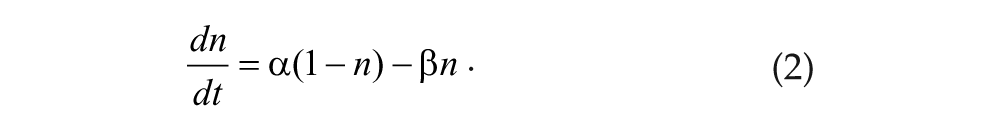
The resultant photic drive,
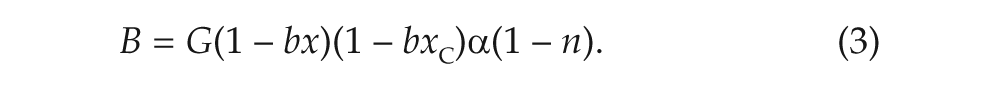
In these equations,
Two coupled differential equations model Process P (St. Hilaire et al., 2007b):
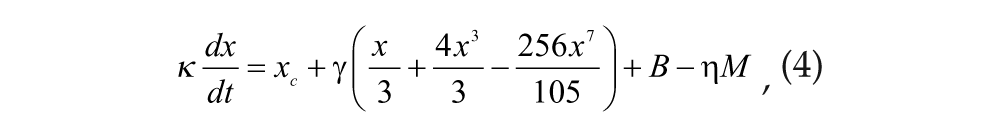
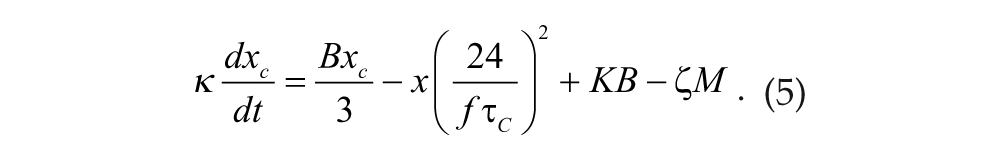
Parameter κ modifies photic drive
Constants η and ξ in Equations 4 and 5 modify the strength of the melatonin drive,
Oscillator phase, φ, uses the standard polar definition
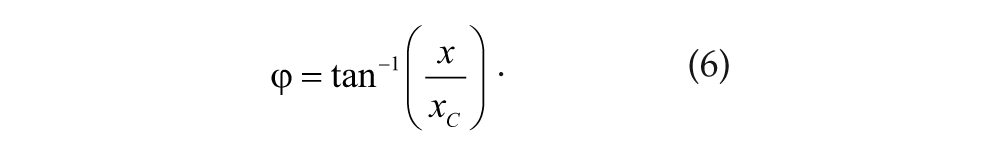
Endogenous and Exogenous Melatonin
The multicompartmental melatonin concentration model includes the pineal gland (
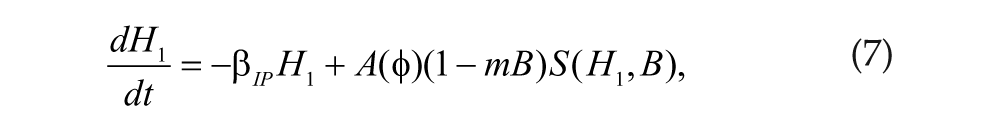
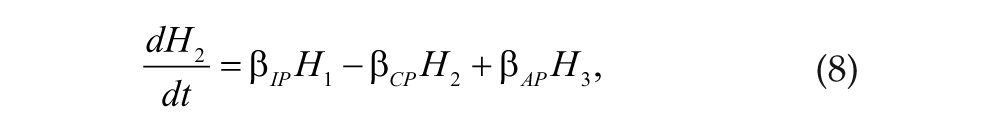

During biological night, the SCN induces the pineal gland to produce melatonin, which infuses into plasma at rate β
At the time of a dose delivery, the value of
Serotonin to melatonin conversion is catalyzed by arylalkylamine N-acetyltransferase (AA-NAT). This enzyme-catalyzed melatonin precursor is then O-methylated by hydroxyindole O-methyltransferase (HIOMT), producing melatonin. This circadian phase dependent process is modeled by
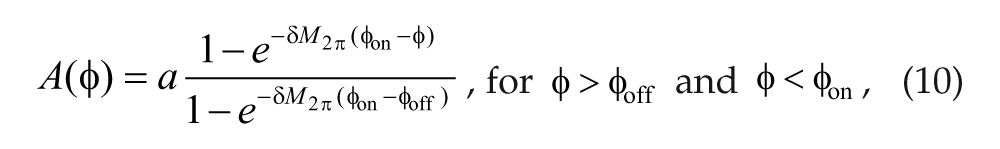
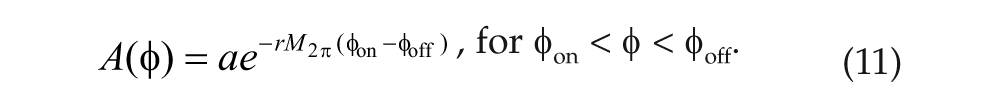
The phases φon and φoff are those at which melatonin synthesis is turned on and off. The function
The melatonin drive to the SCN represents input to melatonin receptors from both endogenous and exogenous melatonin blood plasma. It is modeled by the sigmoid function,
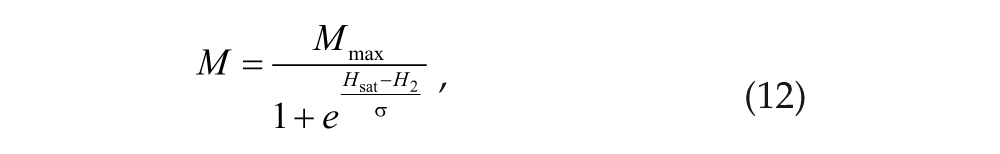
where
A sigmoid function was chosen to simulate receptor saturation. The value of
After fixing a nominal value for
The values of
Pharmacokinetics Methods
Melatonin’s pharmacokinetics are determined by 3 parameters: ε (preabsorption metabolism), β
Exogenous melatonin pharmacokinetics are preparation dependent; therefore, a single parameter set will never fit all data. Aldhous et al. (1985) showed that for an equal dose of 2 mg in gelatin capsules, corn oil, and a slow-release pill, different plasma concentration profiles resulted, presumably because of different degrees of preabsorption metabolism and rates of absorption. Comparison of gelatin preparations used by Aldhous et al. (1985) and Wyatt et al. (2006) reveals further differences. Wyatt et al. (2006) used a gelatin capsule in which 5.0 mg of melatonin was suspended in cellulose, whereas Aldhous et al. (1985) used a gelatin capsule in which 2.0 mg of melatonin was suspended in lactose. The 2 studies show different times to peak and half-lives. These differences cannot be accounted for by changing dose size and instead must be accounted for by changing pharmacokinetic parameters, because the model’s solution to a single dose is a biexponential equation, in which time to peak and half-life are independent of dose size. To illustrate the model’s ability to reproduce different plasma melatonin profiles, we modeled a 2-mg dose for both the Aldhous and Wyatt gelatin preparations by fitting values of ε and β
PRC Methods
We simulated the Burgess et al. (2008) PRC protocol to fit the model’s sensitivity to exogenous melatonin. The protocol began with 7 days of habitual sleep schedules, followed by 5 days on a 4.0-h ultradian sleep-wake and light-dark cycle alternating between 1.5 h of permitted sleep with lights off and 2.5 h kept awake in room light (>150 lux). The ultradian cycle minimized phase-shifting effects of light by evenly distributing light exposure across circadian phases. For the first three 24-h days of this ultradian cycle, participants were given a 3.0-mg gelatin-based capsule of oral melatonin (Burgess et al., 2008) or a placebo at the start of a wake episode at the same clock time each 24-h day. A PRC was constructed based on the last day’s phase assessment. Values of parameters
Model parameters.
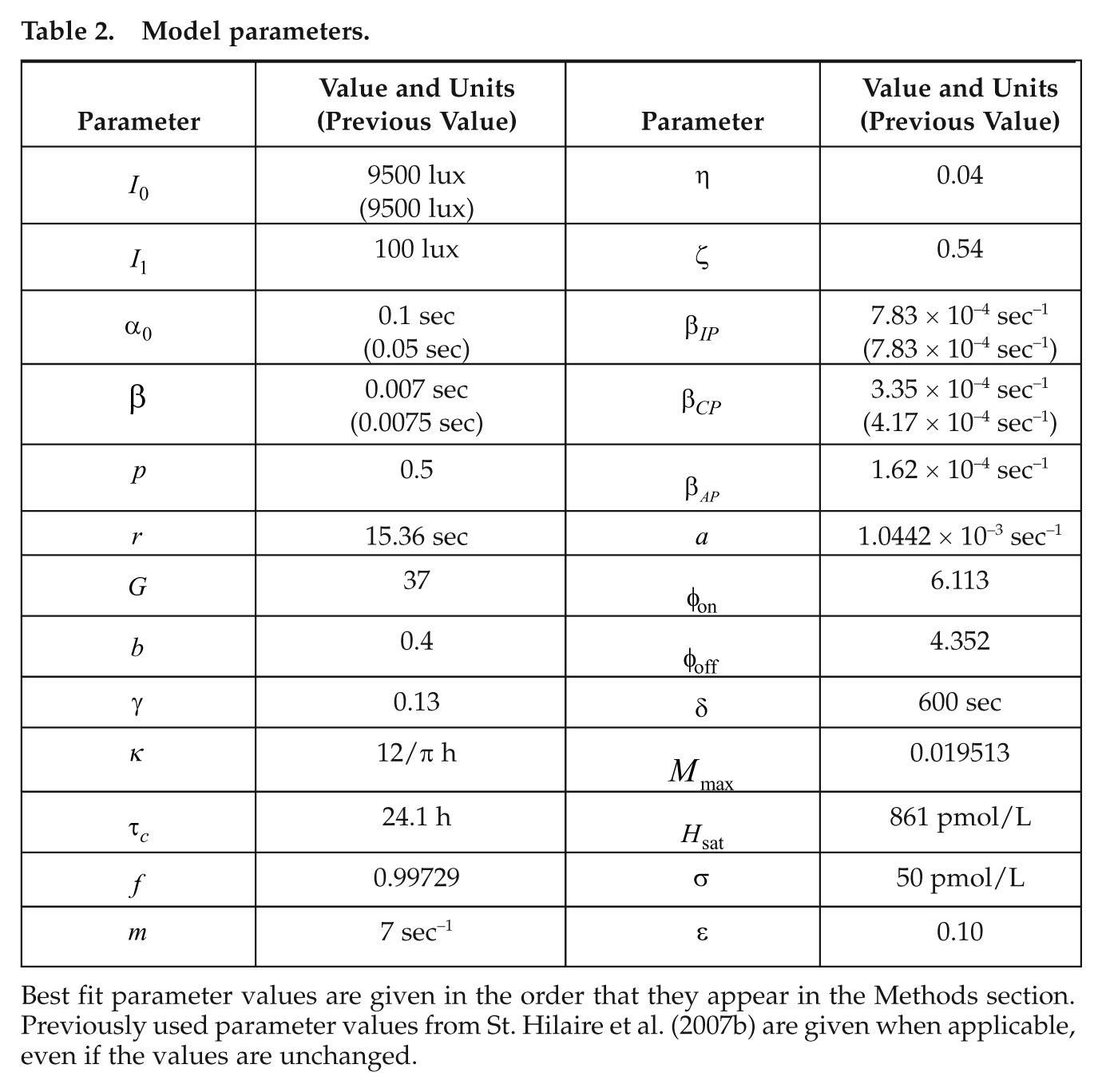
Best fit parameter values are given in the order that they appear in the Methods section. Previously used parameter values from St. Hilaire et al. (2007b) are given when applicable, even if the values are unchanged.
Using these parameter values, we simulated 2 independent data sets. First, we simulated an experiment by Burgess et al. (2010), which is identical to the 2008 experiment except for the use of 0.5-mg instead of 3.0-mg doses. Then, to investigate the effects of a light-dark cycle on the melatonin PRC, we simulated the Lewy et al. (1992, 1998) protocol in which baseline DLMO was assessed on day 1, followed by a week of protocol acclimation with placebo. On day 7, pretreatment DLMO was obtained followed by 2 days of placebo (days 8 and 9), 4 days of 0.5 mg melatonin (days 10-13), and posttreatment DLMO measurement on day 14. With the exception of DLMO measurement days (1, 7, and 14), subjects were at home in uncontrolled and unrecorded light exposure environments. For individuals previously adapted to dim light (e.g., in inpatient experiments), the dynamic range of the circadian phase shifting response to light falls between 0 and 1000 lux (Zeitzer et al., 2000); we therefore simulate light intensities from this range. Light-dark cycles simulated had 16 h of 5, 50, or 500 lux with 8 h of 0 lux in each simulated 24-h day. The parameter set was estimated using the least-squares data fitting method described above.
Results
Pharmacokinetics Results
Simulated and experimental (Wyatt et al., 2006) pharmacokinetics results for 0.3-mg and 5.0-mg data are shown in Figure 2. For the gelatin capsule preparation modeled here, we use ε = 0.1, β
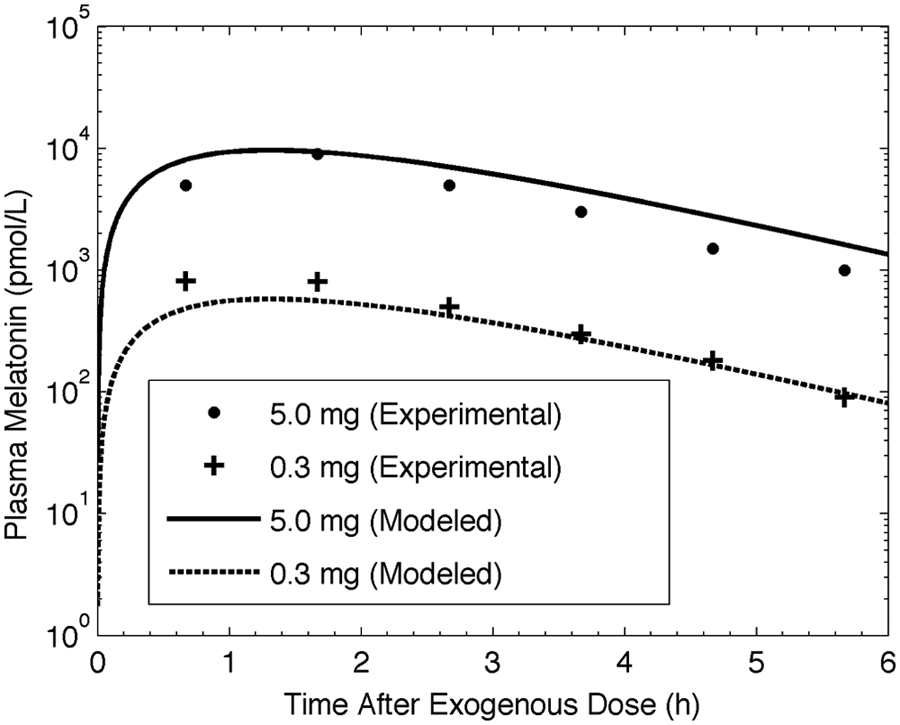
Experimental (symbols, from Wyatt et al. 2006) and modeled (lines) plasma melatonin concentrations over 7 h following 5.0-mg and 0.3-mg doses of oral melatonin.
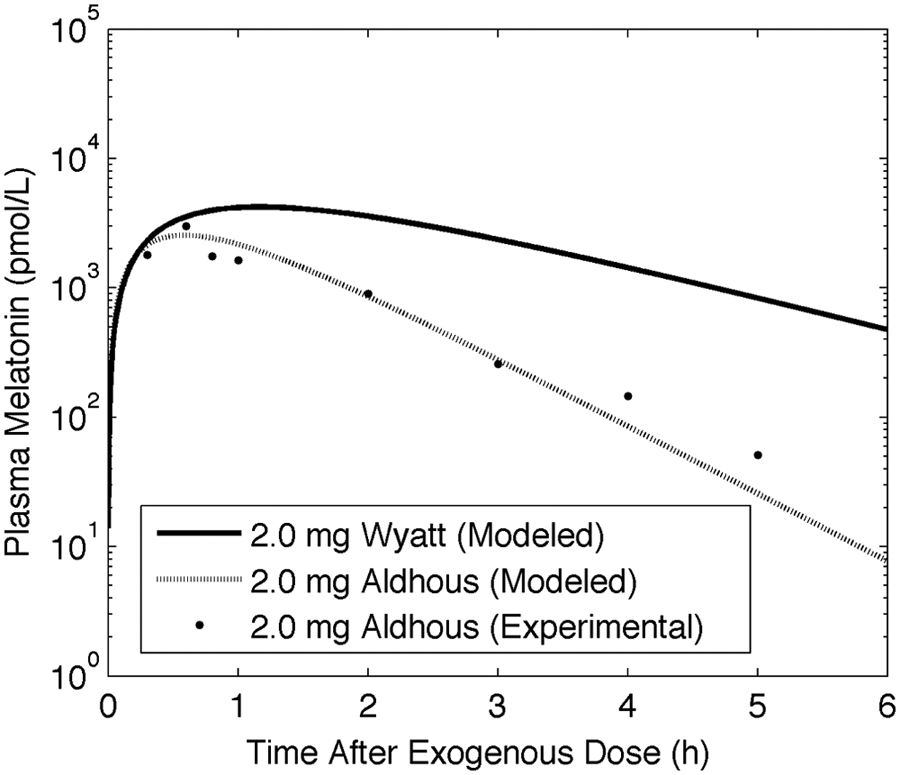
Simulated pharmacokinetic profiles following the Aldhous (1985) (dashed line) and Wyatt (2006) (solid line) 2.0-mg gelatin-capsule melatonin doses. Experimental data from Aldhous et al. (1985) indicated by symbols.
PRC Results
To verify that a phase response is indeed present, we simulated zero phase response (i.e., a horizontal line) at all phases. The adjusted
Simulated and experimental PRC data for the 3.0-mg dose are shown in Figure 4. Exogenous melatonin administered around the time of DLMO causes negligible phase resetting. Doses given prior to DLMO (left of 0 h in the figure) cause phase advances, whereas doses given after DLMO (right of 0 h in the figure) cause phase delays. The model thus predicts type 1 resetting to 3.0 mg of exogenous melatonin.
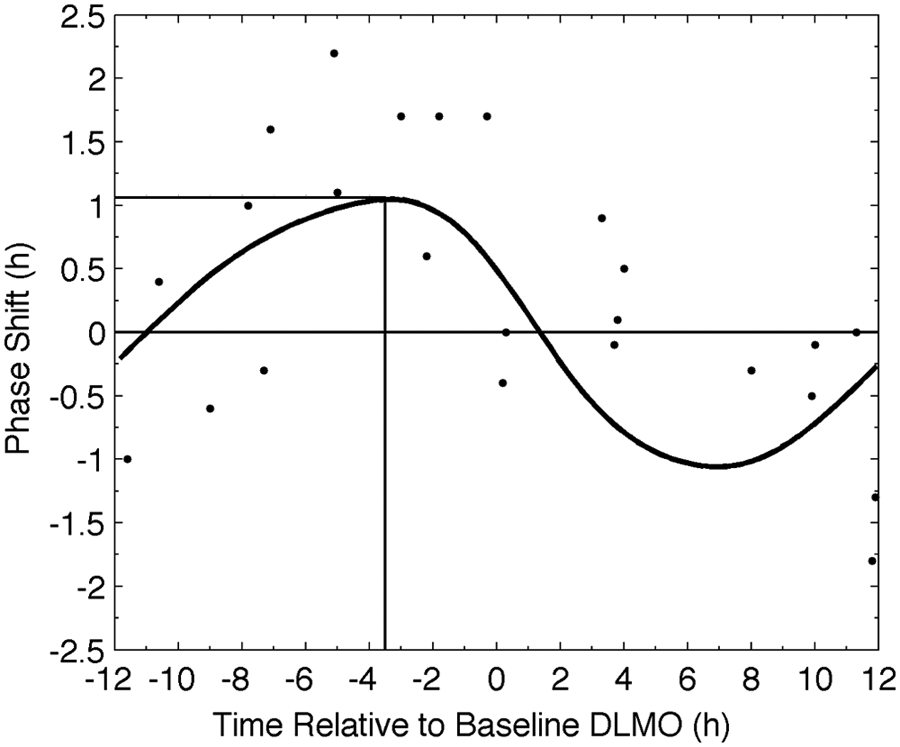
Melatonin PRC for 3 consecutive daily doses of 3.0 mg of oral melatonin. Data extracted from Burgess et al. (2008) are represented as symbols; model output is represented by the solid curve. The vertical line marks the stimulus time required for maximum phase advance (horizontal line). The thin, solid line across the figure demarks a phase shift of 0 h.
In a more recent experiment, Burgess et al. (2010) showed that compared with a 3.0-mg dose, a 0.5-mg dose of the same preparation results in a slightly lower peak that occurs at a slightly later phase. The model correctly predicted these findings without modification to model parameters (Figure 5).
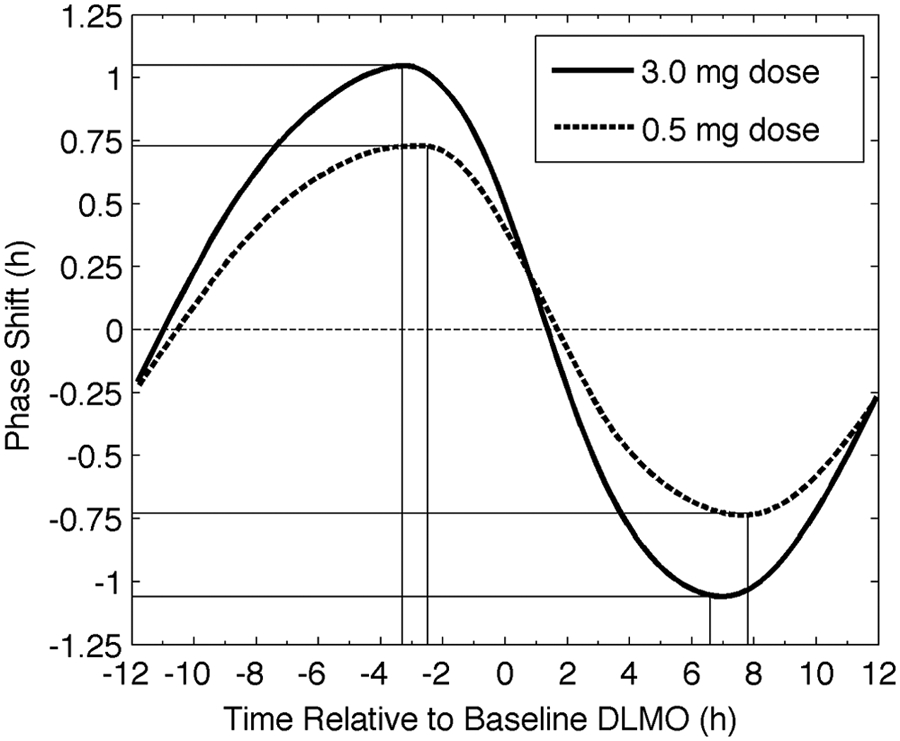
Simulated PRCs for 3 consecutive daily doses of 3.0 mg (solid line) and 0.5 mg (dashed line) of melatonin. The vertical lines mark timing required for the peak phase advance and phase delay (horizontal lines) for each PRC. The horizontal straight, dashed line at zero shows zero phase shift.
Since Lewy et al. (1998) did not control light exposure, and light is a stronger phase-shifting stimulus than is melatonin, the simulations of that protocol were performed with 4 different light levels during the wake episodes (Figure 6). As light intensity decreases, the PRC peak occurs earlier and at higher amplitude, similar to the effects observed when increasing exogenous melatonin dose (Figure 5). Higher light intensities reduce melatonin-induced phase shifts due to light’s entraining effects, which may lock the rhythm to a stable phase.
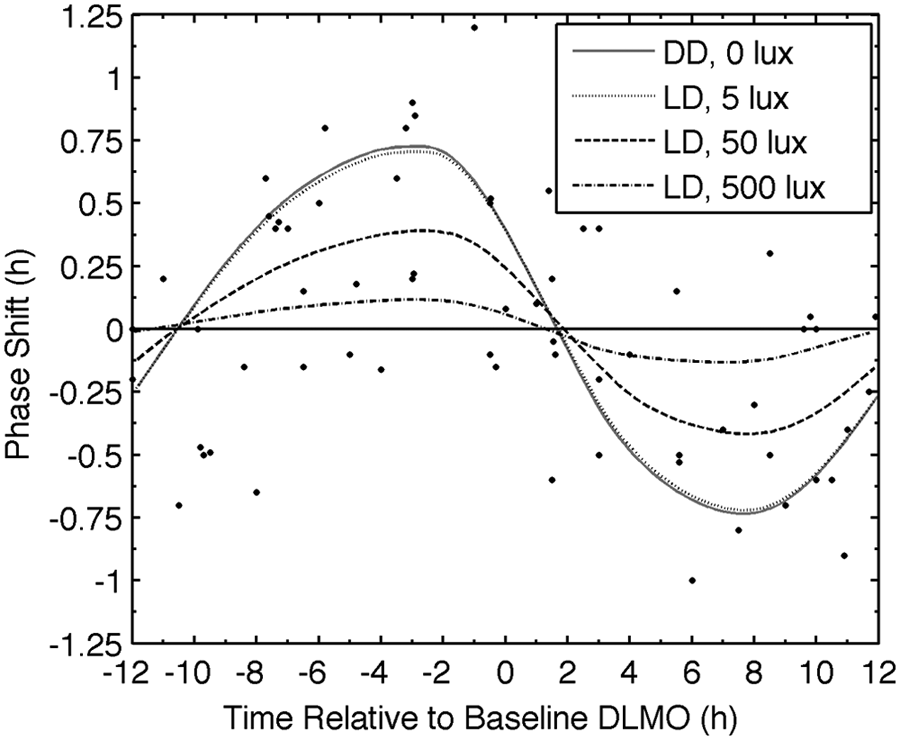
Simulated PRC for 4 consecutive doses of 0.5 mg of exogenous melatonin. Symbols represent experimental data (Lewy et al. 1998). The 4 curves show results from constant darkness (DD; 0 lux) and 3 light levels (5, 50, and 500 lux) within a light-dark (LD) cycle of 16 h light and 8 h dark. Note that PRCs for DD and 5 lux conditions overlap. The horizontal straight, solid line at zero shows zero phase shift.
Discussion
This article presents the first physiologically based mathematical model of diurnal variations in melatonin that includes the phase-shifting effects of exogenous doses of melatonin. The model was shown here to accurately reproduce melatonin’s pharmacokinetics and PRC.
Many equations presented in the Materials and Methods section were adapted from a previous model (St. Hilaire et al., 2007b), as is appropriate when a model is extended to add additional terms. In Equation 7, light suppression of melatonin is approximated by a simple linear function. The functional form used for suppression makes little difference here, as we are modeling phase shifts. However, in future we may opt to use a more realistic saturating suppression function.
Although model parameters presented in this article were optimized to fit the data of Burgess et al. (2008), the model fit at least 1 other PRC (Lewy et al., 1998). The Lewy et al. (1998) PRC was similar to but lower in amplitude than that of Burgess et al. (2008). We showed that this difference possibly can be explained by the uncontrolled light exposures that participants received in this study. Although there were likely interindividual variations in daily light exposure, we showed that a 16:8 LD cycle of 50 lux during wake and 0 lux during sleep fits the Lewy et al. data (
We showed that the model simulates different oral melatonin preparations by varying the fraction of melatonin metabolized preabsorption, ε, and the rate of absorption, β
One prediction of our model is that different preparations result in different PRCs. We note that although preliminary studies attempted to demonstrate this effect (Aldhous et al., 1985), the data sets are highly variable. Because of large interindividual variability in same-dose response, an ideal pharmacokinetic data set would measure the pharmacokinetics of different preparations in the same subjects. To our knowledge, no such data set exists.
We showed in this article that our model simulates expected phase shifts with different oral preparations and doses. However, we have not addressed interindividual variability, which may account for some between-studies differences, including differences ascribed to preparations. The variability of PRC data in which each point is from a different individual is suggestive of large differences in responses to the same dose, even at the same circadian phase. Developing methods to enable the model to predict an individual’s response would be valuable. As one step, the fixed 5 L of blood volume used in this version of the model could become a variable modified for each individual.
Melatonin preparation type is important in interpreting the PRC for at least 2 reasons. (1) Slowly metabolized preparations or doses would be expected to activate melatonin receptors across a broader range of circadian phases than just the circadian phase of administration. (2) For plasma concentrations greater than ~1000 pmol/L, melatonin receptors are saturated (Dollins et al., 1994). Therefore, we posit that when melatonin infuses into the bloodstream quickly, causing high melatonin concentrations, a large proportion of that melatonin will be broken down and cleared without acting at the receptor level. Using the pharmacokinetic profile of gelatin capsules from Aldhous et al. (1985) and fitting to the Burgess et al (2008) data, we predicted that Burgess et al. (2008) used gelatin capsules in their study. More direct comparison between the Aldhous et al. (1985) and Wyatt et al. (2006) data is not possible due to the low number of data points in the Aldhous et al. study. After these predictions, we confirmed via personal communication (2011) that both Burgess et al. (2008) and Wyatt et al. (2006) used gelatin melatonin capsules. Although most studies do not report preparation used, a literature search revealed that corn oil was listed more frequently than was gelatin until the mid-1980s, after which gelatin became the primary preparation listed in chronobiology. We note that preparation variation likely exists even among preparations of a single class (e.g., gelatin). Since preparation used is important in interpreting pharmacokinetic and PRC data, we encourage researchers of future publications to provide this information.
Both experimental and modeling results demonstrate a nonlinear dose response in the melatonin PRC, with only about 40% increased amplitude for a 500% larger dose. We note that the amplitude reported by Burgess et al. (2008, 2010) based on a dual harmonic fit indicates a ~4% increase in amplitude (2.8 h in the case of the 0.5-mg PRC and 2.9 h phase shift in the case of the 3.0-mg PRC). Our results may be different because we fit our model to the data points without using a dual harmonic curve.
In addition to the difference in amplitude, the 3.0-mg PRC is phase advanced relative to the 0.5-mg PRC. This result can be understood in terms of time spent with
Light has the strongest effect on the circadian pacemaker, but other stimuli such as meals and locomotion also affect the circadian rhythm (St. Hilaire et al., 2007a). We tested our model with the nonphotic drive of St. Hilaire et al. (2007a) but found it to have a negligible effect on the resultant PRC (data not shown) and therefore excluded it from this work. Nonphotic drives could play an important role in achieving entrainment with and without exogenous melatonin in blind individuals, which we could model in future work. One potential source of variability not modeled here is down-regulation of melatonin receptors after repeated exposure to superphysiological exogenous doses (Lockley et al., 2000). There are not yet sufficient data to speak to the strength of this effect, but it may be important for modeling entrainment by daily doses.
Our present findings suggest 2 model additions of possible merit. The first is to combine our model with a sleep-wake model to include the hypnotic effects of melatonin. One candidate model is that of Phillips and Robinson (2007), which has already been adapted to model the effects of caffeine on sleep (Puckeridge et al., 2011) and has been combined with the circadian model used here (Phillips et al., 2011). The second potential addition to our model is to incorporate the pharmacokinetics and phase-shifting ability of Ramelteon, a new melatonin-like drug designed to be physiologically similar to melatonin but possibly stronger and more receptor selective (Miyamoto et al., 2004).
Aside from light, melatonin is the most widely used chronobiotic agent, with potential applications to jet lag, insomnia, circadian rhythm sleep disorders, shift work, and circadian entrainment of blind individuals. Side effects from low levels of exogenous melatonin are minimal (Herxheimer and Petrie, 2002). However, melatonin is unregulated in the United States, and therefore there are no guarantees of purity or potency of over-the-counter preparations; indeed, potentially dangerous impurities have been reported (Williamson et al., 1998). Very high doses of even pure melatonin can in rare cases have negative side effects, especially with repeated doses, including depression, cognitive alteration, tremor, fatigue, headache, and somnolence (de Lourdes et al., 2000) and may saturate receptors without causing additional phase resetting effects. Therefore, maximizing phase shifting effects via careful timing rather than higher dosage has potential therapeutic benefit.
By creating this dynamic model, we can manipulate the dose time and dose size to predict the best dosing procedure to produce any phase shift. This model therefore provides the initial basis for ultimately predicting and optimizing melatonin use in real-world environments.
Footnotes
Acknowledgements
Research supported by NIH P01-AG009975, K24-HL105664 (E.B.K.), T32-HL07901 (M.S.H.), R01-HL114088, and RC2-HL101340 and by NSBRI through NASA NCC 9-58 HPF01603, HFP02802, and PF02101 (A.J.K.P.).
Conflict of Interest Statement
In 2010-2011, E.B.K. received investigator-initiated research support from Respironics and an unrestricted gift to the BWH from Sony Corporation. In 2011, A.J.K.P. consulted for Zeo, Inc.
