Abstract
Most animals show rest/activity rhythms that are regulated by an endogenous timing mechanism, the so-called circadian system. The rhythm becomes weaker with age, but the mechanism underlying the age-associated rhythm change remains to be elucidated. Here we employed Drosophila melanogaster as a model organism to study the aging effects on the rhythm. We first investigated activity rhythms under light-dark (LD) cycles and constant darkness (DD) in young (1-day-old) and middle-aged (30-, 40-, and 50-day-old) wild-type male flies. The middle-aged flies showed a reduced activity level in comparison with young flies. Additionally, the free-running period significantly lengthened in DD, and the rhythm strength was diminished. Immunohistochemistry against pigment-dispersing factor (PDF), a principal neurotransmitter of the Drosophila clock, revealed that PDF levels declined with age. We also found an attenuation of TIMELESS (TIM) oscillation in the cerebral clock neurons in elder flies. Intriguingly, overexpression of PDF suppressed age-associated changes not only in the period and strength of free-running locomotor rhythms but also in the amplitude of TIM oscillations in many pacemaker neurons in the elder flies, suggesting that the age-dependent PDF decline is responsible for the rhythm attenuation. These results suggest that the age-associated reduction of PDF may cause attenuation of intercellular communication in the circadian neuronal network and of TIM cycling, which may result in the age-related rhythm decay.
To adapt to daily changes in environment, animals possess a time-keeping mechanism in their body, a so-called circadian clock that generates circa 24-h rhythms. The clock controls a variety of biological activities, such as behavior, physiology, and metabolism, to maximize their functions at a right time of day.
The circadian rhythm deteriorates with age. In mammals, the free-running period, amplitude of the rhythm, and activity amount change as they get older (Froy, 2011). The age-related rhythm changes can be restored by a transplantation of fetal suprachiasmatic nuclei (SCN) of the hypothalamus, which is the center of the clock that regulates behavioral rhythms (Welsh et al., 2010), to older hamsters (Hurd et al., 1995; Viswanathan and Davis, 1995). Expressions of some clock/clock-related genes in the SCN are also found to be reduced with age, although the aging effects are gene and species dependent (Froy, 2011). Thus, the decay in the molecular oscillation in SCN may underlie the age-associated changes in behavioral rhythms. Similar behavioral phenotypes have been reported for the fruit fly Drosophila melanogaster, a genetic model animal (Koh et al., 2006; Shaw et al., 2000). While it is surprising that their clock deteriorates so fast given their short life span (~90 days), this small animal will provide us a great opportunity to investigate the mechanism of the clock aging at molecular and neuronal levels.
Drosophila is one of the best organisms for understanding the mechanism of the circadian clock. The molecular mechanism of the Drosophila clock consists of translational/transcriptional feedback loops composed of clock genes including period (per) and timeless (tim) (Allada and Chung, 2010; Frenkel and Ceriani, 2011; Tomioka and Matsumoto, 2010). The molecular feedback loops exist in a subset of neurons in the brain, the so-called pacemaker neurons that drive bimodal activity rhythms with peaks in the morning and evening (Peschel and Helfrich-Förster, 2011). The pacemaker neurons (~150 cells) are roughly divided into 8 groups: the dorsally located 3 groups of neurons, DN1, DN2, and DN3; the laterally located 4 categories of neurons, LNd, l-LNv, s-LNv, and 5th s-LNv; and those located in lateral posterior region, LPN (Helfrich-Förster et al., 2007; Shafer et al., 2006). They are coupled by neurotransmitters to synchronize each other (Nitabach and Taghert, 2008), but in certain conditions, they are desynchronized and the flies exhibit complex rhythms (Sheeba et al., 2008; Umezaki and Tomioka, 2008; Wülbeck et al., 2008). The pigment-dispersing factor (PDF), the primary circadian neurotransmitter of the Drosophila clock, is expressed only in s-LNv and l-LNv (Helfrich-Förster, 1995). The mutants of Pdf display severe defects in the activity rhythms, showing weak and fragmented rhythms, loss of the morning activity peak, phase advance of the evening peak, and a shorter free-running period under DD (Renn et al., 1999), suggesting the importance of PDF and PDF-positive neurons in the clock network. The axons of l-LNv are projected to the contralateral brain hemisphere through the posterior optic tract (POT), and those of s-LNv project their axons to the dorsomedial area through dorsomedial projections (Dp) (Helfrich-Förster, 1995). The Dp displays circadian changes in its PDF content and morphology (Fernández et al., 2008; Park et al., 2000) and is thought to have synaptic contacts with downstream neurons through the PDF receptor (Im and Taghert, 2010; Shafer et al., 2008). The functional roles of other neurotransmitters remain unclear, however (Hermann et al., 2012; Johard et al., 2009; Umezaki et al., 2011).
In the present study, to investigate the age-related rhythm changes in Drosophila, we examined aging effects on the activity rhythms using young and aged male flies. Aged flies exhibited lengthened free-running periods, reduced rhythm strength, and fragmented activity. These phenotypes were correlated with reduction of PDF levels with age. The amplitude of PER and TIM levels was also deteriorated with age in the pacemaker neurons. Overexpression of PDF prevented the reduction of TIM levels and behavioral rhythm disorder. These results suggest that the reduction of PDF expression is at least partly attributed to the age-associated rhythm decay.
Materials and Methods
Fly Strains and Behavioral Analysis
Canton-S was used as a wild-type strain. white (w) was used as a control strain for transgenic lines that are in a w background. w;Pdf-gal4 and y w;uas-Pdf were described previously (Helfrich-Förster et al., 2000; Renn et al., 1999). To obtain w;Pdf-gal4/uas-Pdf flies, virgin females of w;Pdf-gal4 were crossed with males of y w;uas-Pdf without prior backcrossings to w flies. w;Pdf-gal4/+ and w;uas-Pdf/+ flies were also used as controls for the w;Pdf-gal4/uas-Pdf flies. The flies were reared on standard cornmeal-glucose-yeast medium. To obtain flies of desired age, newly emerged male flies were collected within 24 h after eclosion under CO2 anesthesia, and 20 flies of each genotype were housed in plastic vials (28.5 dia. × 95 mm; Fischer Scientific, Pittsburgh, PA) with 5 mL Jazz-mix Drosophila food (Fischer Scientific). They were transferred to fresh food vials twice per week during light phase without anesthesia. The flies were reared and maintained in an incubator (CR-32; HITACHI, Tokyo, Japan) in which temperature was kept constant at 25 °C, and a 12-h light (400-900 lux) and 12-h dark cycle (LD12:12, light 0600-1800, Japanese standard time) was provided by a cool white fluorescent lamp (FLR20S W/M; Panasonic Co, Osaka, Japan) connected to an electric timer.
For activity recordings, 1-day-old (adult flies within 24 h after eclosion), 30-day-old, 40-day-old, and 50-day-old male flies of each genotype (Canton-S, white, w;Pdf-gal4/+, w;uas-Pdf/+, and w;Pdf-gal4/uas-Pdf flies) were used. They were designated as Day1, Day30, Day40, and Day50 flies, respectively. Flies were fed agar/glucose medium. The activity was monitored at 25 °C under LD cycles and subsequently in DD. Light intensity at the animal’s level was approximately 600 lux. Procedures for locomotor activity recordings were the same as those described previously (Umezaki and Tomioka, 2008).
The period of locomotor rhythms was calculated by the chi-square periodogram (Sokolove and Bushell, 1978) with Actogram J software (freely available at http://actogramj.neurofly.de/) (Schmid et al., 2011). The power value was calculated according to the previous report (Umezaki et al., 2011).
Immunohistochemistry
To determine expression patterns of PDF with age, the whole bodies of each age group were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) with 0.1% Triton X-100 for 2.5 h at room temperature (RT). The fixed flies were washed 3 times in PBS with 0.3% Triton X-100 and then dissected in PBS. The brains were blocked in PBS containing 5% normal donkey serum (NDS) and 0.5% Triton X-100 for 2 h at RT and subsequently incubated in the anti-PDF primary antibody at 4 °C for 24 h. The primary antibody, a rabbit anti-Gryllus PDF (Abdelsalam et al., 2008), was diluted 1:6000 with PBS containing NDS and 0.5% Triton X-100. After rinsing 6 times in PBS with 0.3% Triton X-100, anti-rabbit IgG conjugated with Cy3 (Millipore, Billerica, MA) diluted 1:2000 was applied as the secondary antibody at 4 °C for 24 h. After the incubation, the brains were washed 6 times in PBS with 0.3% Triton X-100 and subsequently mounted in Vectashield mounting medium (Vector Laboratories, Burlingame, CA).
The PDF-immunostained specimens were observed with a fluorescent microscope (Eclipse E-600; Nikon, Tokyo, Japan), and images were captured with a laser-scanning confocal microscope (Fluoview300; Olympus, Tokyo, Japan).
For triple immunolabeling, the primary antibodies, rabbit anti-PER serum (1:1000) (Stanewsky et al., 1997), rat anti-TIM serum (1:500) (Yoshii et al., 2005), and the monoclonal antibody nb33 (diluted 1:100) (Veleri et al., 2003), were applied. The last one was directed against the PDF precursor and thus recognized the PDF-expressing neurons (Veleri et al., 2003). Normal goat serum was used for the blocking solution and the dilution for antibody. Alexa Fluor 488 (goat anti-rat), Alexa Fluor 568 (goat anti-rabbit), and Alexa Fluor 647 (goat anti-mouse) (Invitrogen, Carlsbad, CA) were used as secondary antibodies. Images of the triple-labeled specimens were captured and visualized with a laser-scanning confocal microscope (Leica TCS SPE; Leica, Wetzlar, Germany).
To quantify the staining intensity of PER, TIM, and PDF, the fluorescence intensities were quantified from digital images of more than 10 brains of each genotype and age with the ImageJ software (freely available at http://rsb.info.nih.gov/ij/). For scoring PDF staining intensity in the terminal of the axons projecting from s-LNvs, we overlaid 6 confocal images, including the terminals of Dp, and selected a horizontal part of the terminal area using a rectangle of 2.5 × 20 µm2 (Umezaki et al. 2011). The calculation of the staining indices was performed with the method described by Yoshii et al. (2009a). While the laser intensities for PER and TIM stainings were kept constant throughout the scanning, the intensity for nb33 was adjusted depending on the age and on the pictures to obtain clear images of the PDF neurons. Therefore, the nb33 images were only used for marking the PDF neurons but not for the quantifications.
Results
Effects of Aging on Locomotor Activity Rhythms in Wild-Type Flies
To investigate aging effects on the clock in the fruit flies, we recorded locomotor activity rhythms in a total of 111 wild-type (Canton-S) male flies in LD and subsequently in DD. While the young flies showed robust activity rhythms in LD, Day40 flies showed weak and fragmented activity rhythms (Fig. 1A,B). Both morning and evening peaks were dampened as they aged, showing reduced amplitude of the peaks in Day40 flies (Fig. 1C,D). In DD, the free-running period lengthened and the power of the rhythm was significantly reduced in the old flies compared with the young flies (Fig. 1E,F). The average of the free-running period of the Day1 flies (during days 10-20) was 24.4 ± 0.1 h and those of the Day30 (during day 40-50) and Day40 flies (during days 50-60) were 24.8 ± 0.1 and 25.5 ± 0.3 h, respectively (Suppl. Table S1). The percentage of the rhythmic flies was also significantly reduced from 100% in Day1 flies to about 74% and 40% in Day30 and Day40 flies, respectively (p < 0.05, χ2 test; Suppl. Table S1). These results suggest that the flies show age-dependent changes in the circadian activity rhythms.
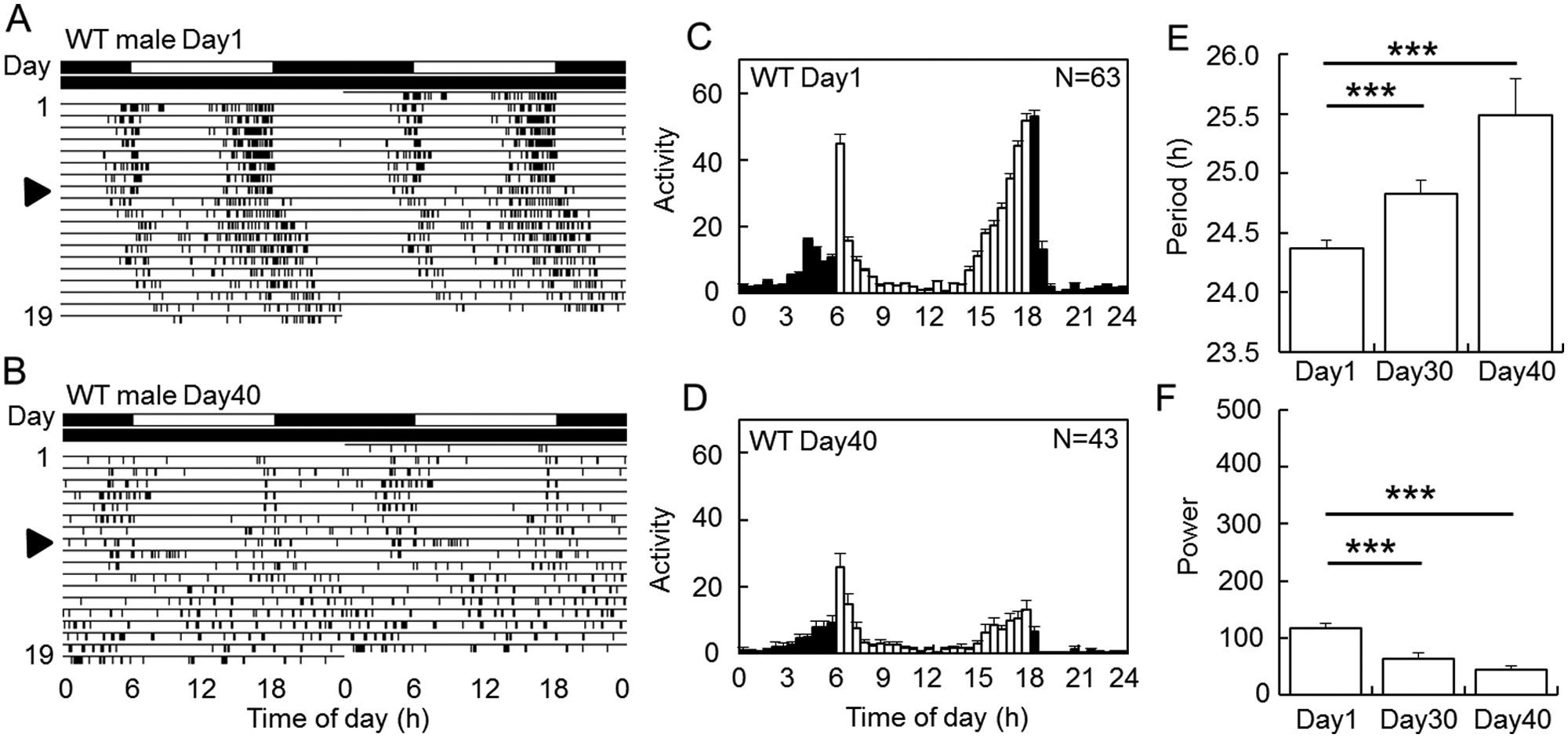
Locomotor activity rhythms of wild-type (WT) male flies of different ages in light-dark (LD) cycles and in subsequent constant darkness (DD). (A, B) Typical actograms of Day1 and Day40 wild-type flies. The arrowheads indicate the day when the flies were transferred to DD. The second day of DD corresponds to 10th (A) and 50th day (B), respectively. The lighting conditions are indicated by black (dark) and white (light) above actograms. With aging, the rhythm became weak and the free-running periods in DD lengthened. (C, D) Mean activity profiles of Day1 (C) and Day40 flies (D) under LD cycles. White and black columns represent mean activity levels in 30 min during the light and dark phases, respectively. N indicates the number of flies used. Vertical bars indicate SEM. Total activity levels were reduced with age. (E, F) Mean free-running periods (E) and power values (F) (mean
PDF Level Is Reduced in Aged Flies
A null mutant of Pdf, Pdf01, displays some phenocopies of behavioral rhythms observed in old flies: the mutant flies show weak and fragmented rhythms, and half of the flies eventually become arrhythmic in DD (Renn et al., 1999). We thus hypothesized that the reduced and fragmented activity rhythm might be attributed to decline in the PDF level. To examine this hypothesis, we verified PDF levels of young and old flies with immunostaining using anti-PDF antibody (Fig. 2).
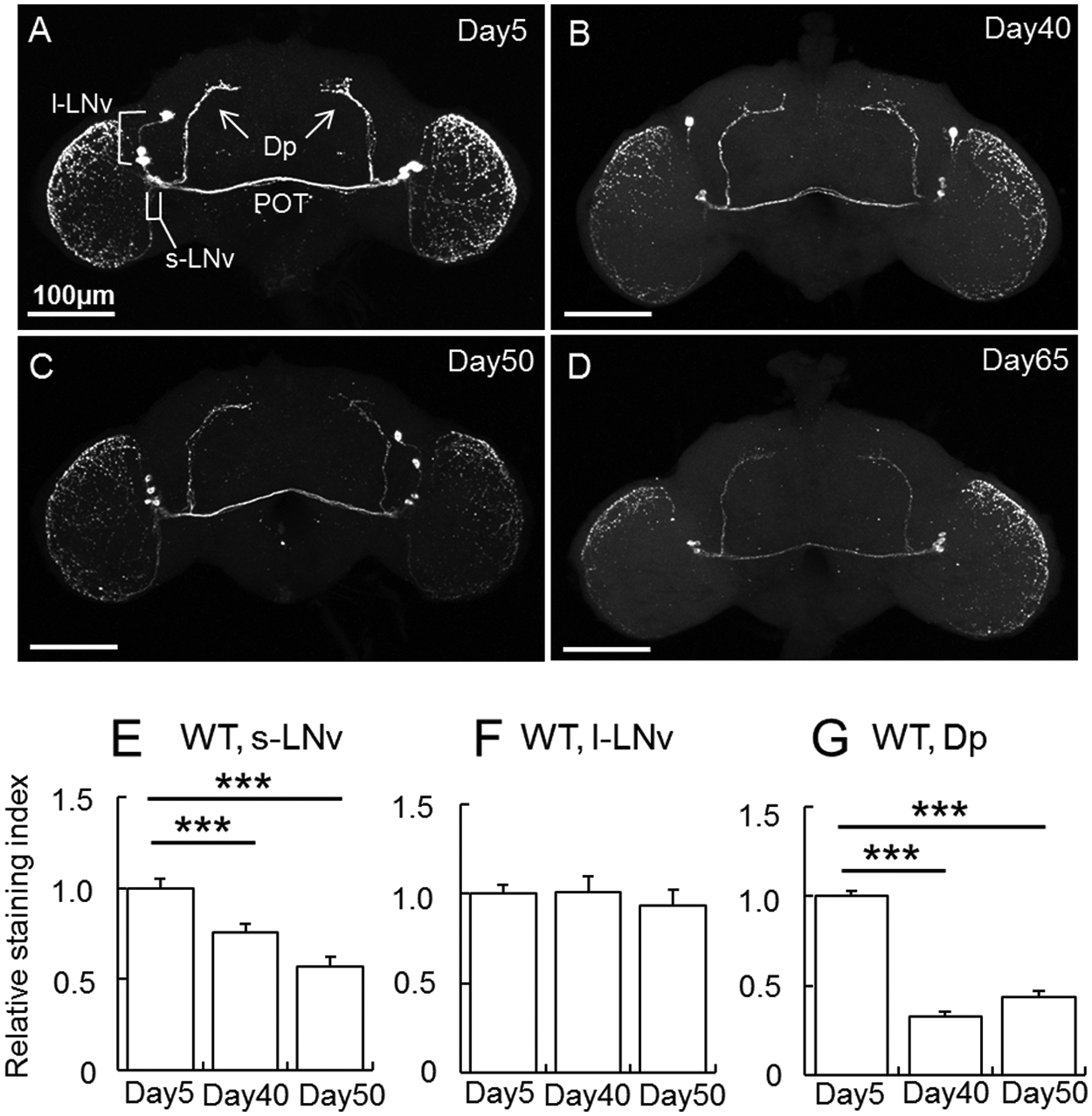
Age-associated changes in the PDF immunoreactivities in wild-type flies. (A-D) PDF immunoreactivities in Day5 (A), Day40 (B), Day50 (C), and Day65 (D) wild-type flies. All brains were sampled 2 h after light-on (ZT 2). POT = posterior optic tract. (E-G) Quantification of PDF levels in cell bodies of s-LNv (E) and l-LNv (F) and dorsal projection (Dp) of s-LNv (G) of wild-type flies of different ages (mean
In the young (Day5) flies, strong PDF signals were observed in s-LNv and l-LNv (Fig. 2A). As flies got older, the signals were reduced in s-LNv cell bodies and their axonal projection, called dorsal projection (Dp) (Fig. 2B-E). The same was observed in the axonal terminals of Dp (Fig. 2G), the axonal projection of l-LNv in the POT, and the optic lobe (Fig. 2A-D). A slight reduction of PDF signals was also observed in l-LNv cell bodies (Fig. 2A-D). These results show that PDF level is reduced in an age-dependent manner. However, quantification for the signal intensity in l-LNv cell bodies revealed no significant difference between the young and aged flies (Fig. 2F). This is probably because the signal intensities in l-LNv cell bodies were saturated in the young flies, so that the immunostaining-based quantification could not discriminate the differences.
PDF Overexpression Partially Suppresses Age-Dependent Rhythm Changes
The next question is whether the age-associated reduction of PDF causes the rhythm changes. We thus overexpressed PDF using the Gal4-UAS system (Brand and Perrimon, 1993). In the control strains and Pdf-gal4/+ and uas-Pdf/+ flies, the age-dependent rhythm deterioration was more or less reproduced (Suppl. Figs. S2 and S3). However, in Pdf-gal4/+ flies, the age-dependent reduction of the activity level was not observed and the night-activity was somehow elevated in the old flies, and in uas-Pdf/+ flies, a higher number of flies remained rhythmic (Suppl. Tables S1 and S2). These might be effects of genetic background. Young PDF-overexpressing (Pdf-gal4/uas-Pdf; Pdf>Pdf) flies showed robust activity rhythms, and their free-running periods were slightly shorter than 24 h (period = 23.8 ± 0.0 h, n = 23; Fig. 3A,D and Suppl. Table S1), as was reported previously (Helfrich-Förster et al., 2000). Interestingly, the free-running periods were not altered with age in the PDF-overexpressing flies (Fig. 3A,B,E), and the strength of the rhythm was maintained at high level even in Day40 flies (Fig. 3A,B,F). The age-dependent reduction of activity amount was observed in the PDF-overexpressing flies, even though Pdf-Gal4/+ flies did not show it with age (Fig. 3C,D and Suppl. Table S2). PDF immunostaining revealed that even in the old PDF-overexpressing flies, PDF levels stayed at a level similar to the young flies in both s-LNv and l-LNv (Suppl. Fig. S1). The PDF level in axonal processes was also significantly higher than that of aged wild-type flies (Fig. 2 and Suppl. Fig. S1). These results suggest that PDF reduction with age is responsible for the rhythm changes in period and strength.
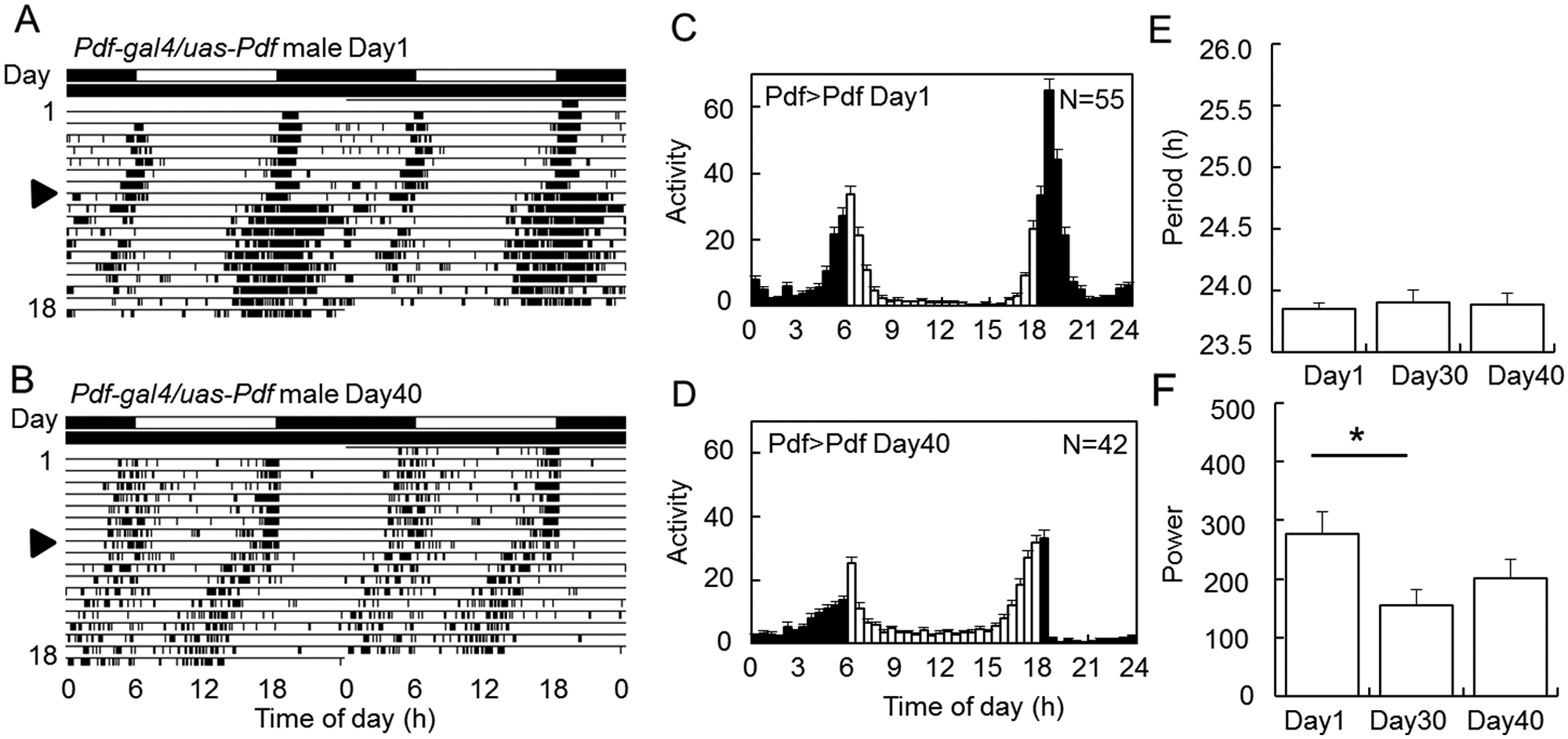
Suppression of the age-related rhythm changes by PDF overexpression. (A, B) Typical actograms of PDF-overexpressing flies (Pdf-gal4/uas-Pdf). (C, D) Mean activity profiles of Day1 (C) and Day40 (D) flies under LD cycles. (E, F) Free-running periods (E) and power values (F) (mean
Effects of Aging on Expression of Clock Proteins
To elucidate the effects of aging on the molecular clock mechanism, we examined clock protein levels with immunostaining using anti-PER and anti-TIM antibodies in each clock neuron group—that is, s-LNv, 5th s-LNv, l-LNv, LNd, DN1a, DN1p, and DN2 (Fig. 4 and Suppl. Figs. S4 and S5). In the young wild-type (Canton-S) flies, PER and TIM levels were high at zeitgeber time (ZT) 20 but low at ZT10 in LD (Suppl. Fig. S5). The amplitude, as the difference in staining intensity between the two time points, deteriorated with age in all cell groups (Suppl. Fig. S5A,B). The same reduction of PER was observed in the PDF-overexpressing flies as well (Fig. 4C and Suppl. Figs. S4B and S5C), while, interestingly, the reduction of TIM level was prevented in most of the cell groups in the PDF-overexpressing flies, although the effects were not the same among the cell groups (Fig. 4D and Suppl. Figs. S4B and S5D). For instance, in all LN and DN2 groups, the age-associated reduction of TIM level was almost completely suppressed, while in DN1 groups, the suppression occurred but was limited to a certain range. These findings indicate that PDF overexpression suppresses the age-related reduction of TIM but not PER levels in the cerebral clock neurons.
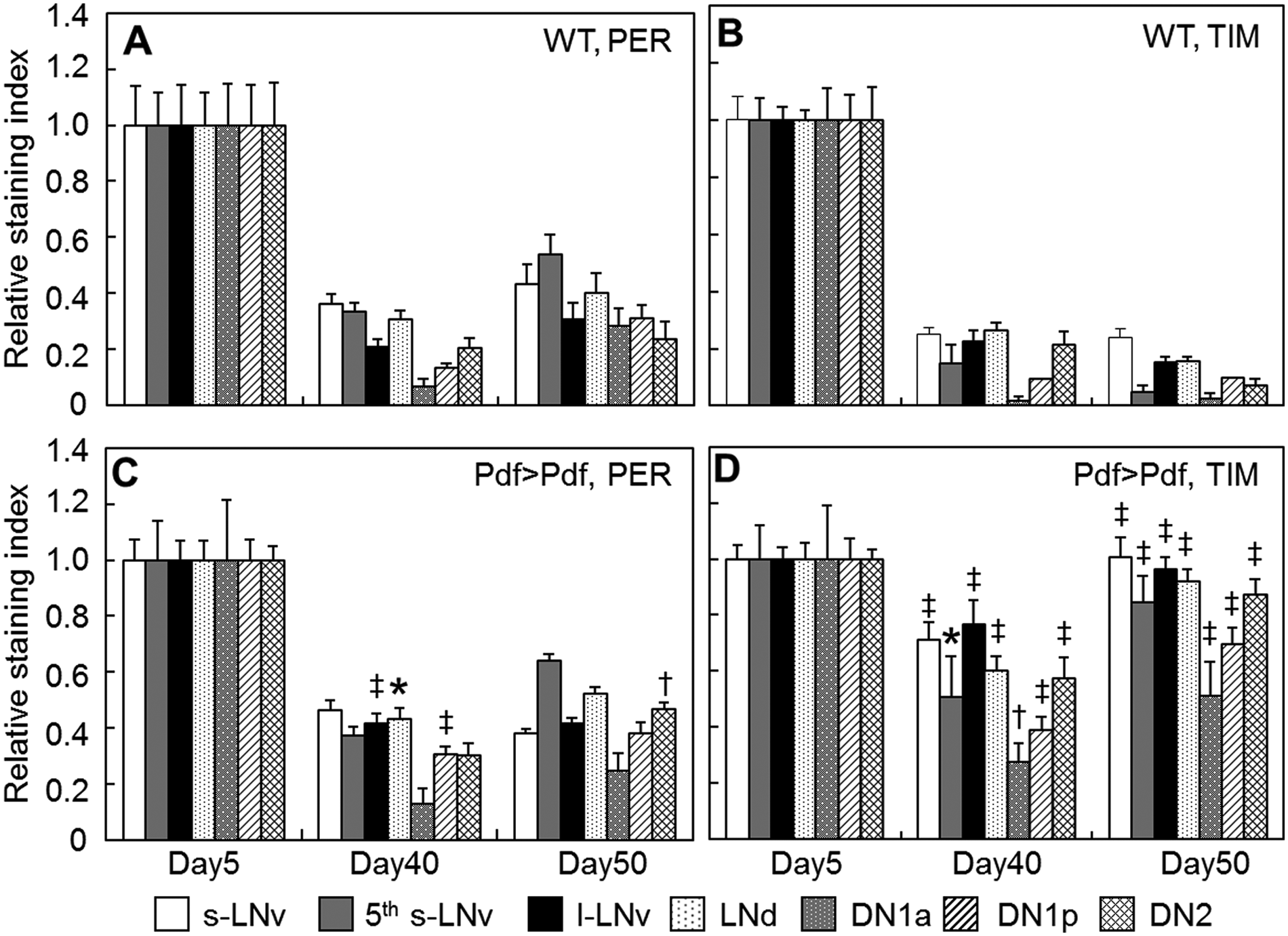
Age-associated reduction of PER and TIM expression levels in wild-type and Pdf-gal4/uas-Pdf (Pdf>Pdf) in a subset of clock neurons at nighttime (ZT20) in Day5, Day40, and Day50 flies. The PER and TIM staining intensities were normalized by values at ZT20 of Day5 flies. Vertical bars indicate SEM. PER and TIM levels declined with age, whereas the TIM reductions were suppressed in PDF-overexpressing flies. Data were compared between wild-type (WT) and Pdf>Pdf flies at ZT20 in each time point by t test. *p < 0.05. †p < 0.01. ‡p < 0.001.
Discussion
Circadian rhythms decay with age in their robustness in a variety of animals (Froy, 2011; Hofman and Swaab, 2006; Pittendrigh and Daan, 1974; Watari and Arai, 1997), but the underlying mechanism is not well understood. The fruit fly D. melanogaster has high potential to serve as a model animal to investigate the aging of circadian rhythms, because of its short life span and abundant information on its circadian clock. In the present study, we have demonstrated that the Drosophila circadian rhythms decayed with age; aging lengthened the free-running period in constant conditions, deteriorated the robustness of the rhythms, reduced activity amount, and eventually led to arrhythmic behavior. These findings are consistent with 2 reports published very recently (Luo et al., 2012; Rakshit et al., 2012). Thus, the aging mechanism of the clock is now able to be dissected at a molecular level in this simple but excellent model animal.
Here we found that some aging phenotypes were correlated with an age-dependent PDF reduction. PDF is a main circadian neurotransmitter in Drosophila and expressed only in a subset of clock cells: s-LNv and l-LNv (Helfrich-Förster, 1995; Renn et al., 1999). The expressed PDF directly targets other non-PDF clock neurons and also provides feedback to PDF neurons through PDF receptors that are expressed in most PDF-negative and PDF-positive clock neurons (Im and Taghert, 2010). Thus, PDF works as an intercellular messenger among the clock neurons (Klarsfeld et al., 2004; Lin et al., 2004; Peng et al., 2003; Yoshii et al., 2009b). The phenotypes of the aged flies showed some similarities to Pdf null-mutant or Pdf knockdown flies (Renn et al., 1999; Shafer and Taghert, 2009): the rhythm strength was weaker than wild-type flies. But not all age-related phenotypes are reproduced in these mutant flies; the free-running period lengthened in old flies, whereas it shortened in Pdf01 mutants. Our results showed that the aging effects on the rhythm strength and free-running period were reduced by increasing PDF amount in the old flies. Therefore, the age-related reduction of PDF would be key to the circadian aging phenotypes. However, it still remains to be elucidated whether transcriptional changes are involved in the age-related changes in PDF levels and whether the releasing rate of PDF corresponds to the staining/accumulation level.
The mechanism underlying the age-associated changes seems not simple because not all aging phenotypes were suppressed, even if the PDF level of the old flies was comparable to that of young flies. One may also argue that the increased level of PDF has some effects on the physiology of the PDF neurons that eventually affects aging. Indeed, PDF neurons also have neurotransmitters other than PDF (Herman et al., 2012; Johard et al., 2009; Umezaki et al., 2011), which might have some effect on aging. Since electrical activity in the pacemaker cells reinforces molecular oscillations of clock proteins (Depetris-Chauvin et al., 2011; Nitabach et al., 2002, 2005), one may also expect that strengthening the coupling in the circadian network by PDF overexpression may maintain a high level of electrical activity in the pacemaker neurons, which results in better molecular rhythms even in old flies.
In mammals, the vasoactive intestinal peptide (VIP), which is regarded as a functional homolog of PDF, plays a role in the intercellular communication among the pacemaker neurons in the SCN (Aton et al., 2005; Vosko et al., 2007). Like PDF, its receptor VPAC2 is closely related to G-protein-coupled receptors belonging to the B1 subfamily (Hyun et al., 2005; Lear et al., 2005; Mertens et al., 2005). Interestingly, VIP and its receptor also exhibit an age-dependent reduction in their mRNA levels in hamsters and rats (Duncan et al., 2001; Kalló et al., 2004; Kawakami et al., 1997; Krajnak et al., 1998). Thus, Drosophila and mammals share a common trait in age-associated rhythm deterioration, the reduction of the coupling factors in the clock system.
The complexity of the aging effects is also suggested by an elevated TIM level at night by increasing PDF levels in the old flies. PDF affects not only amplitude of clock protein rhythms but also the phase and speed of the oscillation in the pacemaker neurons (Klarsfeld et al., 2004; Lin et al., 2004; Yoshii et al., 2009b), suggesting that the TIM elevation is probably attributed to an effect of PDF.
In general, mRNA levels of many genes decline in the head with aging (Kim et al., 2005). This is the case for CLOCK-CYCLE (CLK-CYC) controlled clock genes: per, tim, vrille (vri), and Par domain protein 1ε (Pdp1ε), except for clockwork orange (cwo) (Krishnan et al., 2009; Rakshit et al., 2012). This fits well to what we found here—that PER and TIM levels in the pacemaker neurons at night decline in the old flies (Fig. 4). We have obtained the same results in 2 different genetic backgrounds (wild-type and Pdf-gal4/uas-Pdf flies) for PER levels, and a similar tendency was observed also in TIM. However, this is totally contradictory to what Luo et al. (2012) reported. They examined PER levels in the pacemaker neurons from flies sampled at 4-h intervals for 24 h in DD and compared the difference between young and old flies. Surprisingly, aged flies still showed robust PER cycling, and no significant age-related changes were found in PER levels. It is very hard to reconcile their data with ours, and it is unlikely that small differences in the experimental conditions result in the opposing results. Interestingly, our wild-type flies (Canton-S) do not live so long (<80 days at 25 °C; Umezaki et al., unpublished observation) compared with the w1118 strain that Luo et al. (2012) used (~90 days at 25 °C) (Koh et al., 2008). per null-mutant (per01) flies shorten the life span and increase age-related neural degeneration (Klarsfeld and Rouyer, 1998; Krishnan et al., 2009). Therefore, it might be that the w1118 strain is more tolerant to the age-related clock deterioration than our Canton-S flies, which may lead to the longer life span of w1118 flies.
In conclusion, we have demonstrated that Drosophila exhibits age-associated deterioration in the circadian rhythm. This occurs not only in behavior but also in PER and TIM expression levels. Since increased PDF levels in old flies suppressed some aspects of age-associated circadian defects, the reduction of PDF with age plays a key role in the age-associated rhythm defects. Our results thus show that the decaying intercellular communication in the circadian network via PDF is one of the factors that cause aging in the clock in addition to damping of CLK-CYC activity (Rakshit et al. 2012). It will be of interest to investigate whether the damping of CLK-CYC activity occurs before PDF reduction or vice versa, which would tell us which primary factor induces the clock damping with age.
Footnotes
Acknowledgements
We thank Paul H. Taghert for providing w;Pdf-gal4 and y w;uas-Pdf flies. We thank Jadwiga Giebultwicz for the TIM antiserum, Alois Hofbauer for the nb33 antiserum, and Ralf Stanewsky for the PER antiserum. We also thank Amita Sehgal for critical reading of an earlier version of the manuscript. This work was supported by the Japan Society for Promotion of Science (JSPS) to Y.U. and K.T. Y.U. was a JSPS Research Fellow.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
