Abstract
The dual-oscillator model, originally proposed as a mechanism for how vertebrates adapt to seasonal changes, has been invoked to explain circadian entrainment in Drosophila melanogaster. Distinct subsets of neurons have been designated as “morning” and “evening” oscillators that function as regulators of rhythmic activity/rest behavior. Some studies have led to a model in which a subset of 8 “morning” cells (4 bilaterally located small ventral lateral neurons) and another subset of approximately 130 “evening” cells exert different levels of dominance within the circadian circuit in different seasons. However, many studies propose a more integrative neuronal network, with the whole network orchestrating activity/rest rhythms in different seasons, as opposed to hierarchical dominance among neurons. Within the circadian network, our understanding of the role of the large ventral lateral neurons (l-LNv) has thus far been limited to conveying light information to the clocks and as light-activated neurons mediating arousal. In support of the framework of a more distributed model, we report an important circadian clock–related role for the l-LNv in electrical activity–dependent phasing of the evening peak across a range of photoperiods. Further, we propose a model in which l-LNv enable adaptation to seasonal changes by regulating the phase of the evening peak. Additionally, we demonstrate a hitherto unknown role for the small ventral lateral neurons (s-LNv) in the arousal circuit, thus showing that neuronal function is flexible such that certain neurons can play more than one role in distinct circuits.
The daily cycling of environmental factors such as light and temperature creates a periodic environment for organisms, which synchronizes their metabolic, physiological, and behavioral processes to environmental cycles with the aid of endogenous circadian clocks (Allada and Chung, 2010). The cues that provide time information to circadian clocks are numerous, with light being predominant (Sharma and Chandrashekaran, 2005). While in the tropics, photoperiod remains roughly constant throughout the year, it varies dramatically in temperate regions. Although a number of other factors also change with seasons, day length changes are most consistent. Such variations in the environment can be deleterious to organisms if they are unable to time their behavior and physiology appropriately.
The dual-oscillator model (Pittendrigh and Daan, 1976) suggested a mechanism by which animals might cope with such dramatic environmental changes. According to this model, a morning (M) oscillator tracks dawn, and an evening (E) oscillator tracks dusk. With changing photoperiods, the timing of dawn and dusk varies, resulting in corresponding changes in the phase relationship between the 2 oscillators, thus enabling organisms to adjust to varying seasons. The model attempted to provide an explanation for bimodality in activity/rest profiles by positing that M and E oscillators give rise to the morning and evening peaks, respectively, thus rendering the twilight transition periods very crucial (Pittendrigh and Daan, 1976).
In Drosophila, 7 subsets of circadian clock neurons—3 dorsal groups (DN1, DN2, DN3) and lateral posterior neurons (LPN), large and small ventral lateral neurons (l-LNv and s-LNv), and lateral dorsal neurons (LNd)—have been characterized. All LNv except the fifth s-LNv secrete neuropeptide pigment dispersing factor (PDF) (Shafer et al., 2006). One of the earliest studies, which designated neuronal correlates to the oscillators, concluded that PDF+ LNv and PDF– CRY+ (photoreceptor CRYPTOCHROME) neurons were the M and E oscillators, respectively (Stoleru et al., 2004). In parallel, a separate group narrowed the M oscillator to s-LNv (Grima et al., 2004). More recently, the phasing and amplitude of the E peak have been attributed to 3 to 4 NPF+ LNd and PDF– fifth s-LNv (expressing neuropeptide F) (Hermann et al., 2012). Notably, adaptation to seasonal variation was proposed to be a result of a seesaw in the dominance hierarchy between M and E cells, with each setting the phase of the other in winter and summer conditions (Stoleru et al., 2007).
Other studies have hypothesized that the M oscillator (consisting of PDF+ s-LNv) is the “main” oscillator, as it also exerts its influence on the evening peak (Rieger et al., 2006). Indeed, in flies with ablated or electrically silenced LNv and in pdf 01 mutants, the evening peak is phase-advanced compared to controls (Renn et al., 1999; Nitabach et al., 2002). Under constant darkness (DD), oscillation in PER levels (an indicator of a ticking circadian clock) dampens quickly in l-LNv (Shafer et al., 2002), suggesting that the s-LNv regulate PDF signaling to other circadian circuit components. Yet another approach showed that speeding up circadian gene oscillations in s-LNv sped up oscillations in LNd, DN1, and DN3, whereas speeding up DN2 oscillators increased the speed of oscillations in l-LNv alone, suggesting a hierarchical circadian circuit in which l-LNv occupy a subordinate role (Stoleru et al., 2005). These results have mostly disregarded the role of l-LNv in the circadian clock circuit.
While a hierarchical circadian circuit explains some phenomena, a more distributed and plastic clock network has been called for, taking into account mammalian studies (Nitabach and Taghert, 2008; Sheeba et al., 2008c; Meijer et al., 2010). The behavior of anticipating lights-on, considered a hallmark of the M oscillator, was found to persist even in the absence of PDF in the cell bodies of s-LNv; l-LNv, along with other clock neurons, were proposed to govern this behavior (Sheeba et al., 2010). Another study indicated that PDF signaling from l-LNv independent of the classic circadian clock is essential for E cells to anticipate lights-off, especially in the absence of CRY (Cusumano et al., 2009). These results point toward a role for l-LNv in the circadian clock network.
Previous studies have suggested that l-LNv have a role in determining the phase of the evening peak, although without being able to distinguish between l-LNv and s-LNv (Helfrich-Förster, 2009; Yoshii et al., 2009). In the current study, we have explored the role of l-LNv in the distributed clock network in a variety of environments that approximate varying seasons. We investigated the role of l-LNv in the neuronal circuit by perturbing the membrane properties of these neurons in the absence of s-LNv and examined the activity profiles of flies under different photoperiods to reveal potential miscommunication within the clock circuit. Apart from short- and long-day conditions (LD8:16 and LD16:8), we exposed flies to extreme photoperiod conditions (LD4:20 and LD20:4) to cover the full range of seasonal effects on the circadian clock circuit. We were able to assign specific roles to s-LNv and l-LNv in the control of definite components of evening peak in different photoperiod conditions. Further, we show that l-LNv, whose role in the circadian circuit was contentious, can influence the phase of the evening activity peak.
Materials and Methods
Fly Strains
Flies were reared on standard corn medium under light/dark cycles with 12-hour light and 12-hour darkness (LD12:12) at 25 °C and constant humidity (~70%) to minimize confounding effects of rearing environment. To specifically render the s-LNv dysfunctional, we used the pdfGAL4 driver yw;pdfGAL4 (Renn et al., 1999) to express a toxic form of Huntingtin (HTT) protein (w;UAS-HTT-Q128), consisting of 128 glutamine (Q) repeats (Lee et al., 2004), thus giving the genotype yw;pdfGAL4/UAS-HTT-Q128 and designated as s–L+. While pdfGAL4 can drive the expression of toxic neurodegenerative proteins in s-LNv and l-LNv, the s-LNv appear to be specifically targeted, as demonstrated by their lack of PDF and PERIOD expression and breakdown of behavioral rhythm in DD (Sheeba et al., 2008a). The reason for this selective degeneration is yet unknown; however, it allows us to tease apart the function of the 2 LNv subsets. The controls for the pdfQ128 genotype were yw;pdfGAL4/UAS-HTT-Q0, designated as s+L+, with the Q0 construct being a nontoxic form of the HTT protein with no polyQ repeats, and driver controls yw;pdfGAL4/+ (s+L+). With the goal of checking if the number and not the type of LNv is important for activity/rest rhythm, we studied yw;pdfGAL4/UAS-rpr-C14 (s±L±) flies, which showed incomplete ablation of LNv. To assess the role of l-LNv, we used yw;pdfGAL4/UAS-HTT-Q128;UAS-NaChBac1/+ flies (s–LH), which rendered l-LNv alone hyperexcited, as s-LNv were made dysfunctional by HTT (Sheeba et al., 2008a). Genotype of flies with both s-LNv and l-LNv hyperexcited (sHLH) was yw;pdfGAL4/UAS-HTT-Q0;UAS-NaChBac1/+. Additional controls were yw;pdfGAL4/UAS-HTT-Q0;UAS-dORK-NC1/+ (s+L+) and yw;pdfGAL4/UAS-HTT-Q128;UAS-dORK-NC1/+ (s–L+). dORK-NC1 is a nonconducting K+ channel that acts as a control for introduction of the NaChBac ion channel (Nitabach et al., 2006).
Activity Recording
Individual virgin males (3-4 days old) were placed in glass tubes and recorded using the Drosophila Activity Monitoring System (TriKinetics, Waltham, MA). Separate batches of flies were recorded for each photoperiod to avoid confounding effects of age. The different photoperiod conditions created in an incubator (DR-36VLC8, Percival Scientific, Perry, IA) were LD12:12, short day LD8:16, very short day LD4:20, long day LD16:8, and very long day LD20:4, with a light intensity of approximately 0.15 W/m2 during the light phase. Photoperiods were created by symmetrically shrinking or expanding day length from both ends of the light duration of LD12:12.
Analysis of Behavioral Data
Activity counts sampled at 5-minute intervals were binned into 15 minutes to obtain a time series of activity data for individual flies. From the 9-day-long time series, the first 2 cycles were excluded for further analysis to eliminate transients. Data for the next 7 days were normalized to total activity during each day, which were then averaged across 7 days to obtain the average activity profile of each individual fly. Such profiles were then averaged across individual flies of each genotype to obtain the average activity/rest profile of that genotype. Normalized actograms of individual flies were batch-analyzed to obtain average actograms for each genotype using ClockLab (Actimetrics, Wilmette, IL). To compare activity levels, we divided the 24-hour day into 4-hour intervals and considered lights-on as ZT0 in all photoperiods. The activity in each bin was normalized to total activity during the 24-hour cycle to obtain the percentage of activity. In the long and short photoperiods, we often found peaks that were mere responses to light-to-dark or dark-to-light transitions and distinct peaks that were likely to reflect the “true” morning and evening peaks. These 2 types of peaks were distinguished based on whether they occurred within a 1-hour window following lights-on or -off, in which case they were designated as “startle” peaks. The “true” peaks in all nonstandard photoperiods were designated as M or E peaks based on the proximity to lights-on or -off transition. To determine objectively whether “true” peaks were present, average activity counts in each photoperiod were binned into 1-hour intervals. Two-way ANOVA was used to check if any 1-hour bin within an interval of 4 to 6 hours before or after lights-on or -off transitions was significantly different from its immediate neighbors. In certain cases when the peaks were broad and it was not possible to identify one bin that was significantly higher than its neighbors, we relaxed the criterion such that a set of 3 successive 1-hour bin values significantly higher than the neighboring values was considered as a true peak. Genotypes that met either of these 2 criteria were considered as displaying nonstartle “true” morning and/or evening peaks. To determine the phase of these peaks, we referred to the 15-minute average activity profiles of individual flies and manually scanned that interval containing the peak (as determined by 1-hour profiles) and identified that 15-minute bin, at which maximum activity count was recorded. The phase values thus obtained for individuals were averaged to obtain phases of the peaks for each genotype in each photoperiod. We calculated the morning and evening anticipation indices in LD12:12 as the ratio of activity in the final 3 hours prior to lights-on and -off transitions to that which occurs in the 6 hours preceding the transitions (Harrisingh et al., 2007). Anticipation indices in photoperiods other than LD12:12 were not calculated, as the activity peaks did not coincide with the transition periods.
Statistical Analysis
All statistical analyses were done using STATISTICA (StatSoft, Tulsa, OK). Percentage activity data were transformed by taking the square root of the arcsine of the fraction of activity at each time point before 2-way ANOVA was carried out for 4-hour interval data of all photoperiods, with the 2 factors being genotype and time interval. A Tukey HSD test was conducted for post hoc analysis. Similarly, for 1-hour activity/rest profiles, data were analyzed by 2-way ANOVA, with genotype and time interval as fixed factors. The 95% confidence intervals for these values were calculated and plotted on the activity/rest graphs to allow for visual hypothesis testing. Morning and evening peak anticipation indices of LD12:12 across genotypes were compared using 1-way ANOVA followed by a post hoc Tukey test (p < 0.05).
Results
Normally Firing l-LNv Are Necessary for Lights-Off Anticipation under LD12:12
All control flies with normal s-LNv and l-LNv (s+L+) showed bimodal activity under LD12:12 (Fig. 1A and 1B) with prominent morning and evening peaks, with both anticipation and startle components (GAL4, Q0, and NCQ0) (Fig. 1B). Anticipation of lights-on or -off is an indicator of the presence of functional circadian clocks and is distinct from the “startle” response (an abrupt increase in activity counts immediately after lights-on or -off), which is shown even by arrhythmic mutants (Wheeler et al., 1993). When both LNv are partially ablated (s±L±) (Suppl. Fig. S1), or only s-LNv are dysfunctional (s–L+), the evening peak was similar to that of s+L+ controls (Fig. 1A and 1B), both in terms of anticipation of and startle due to lights-off (Fig. 1B and 1D). In contrast, the morning anticipation of s–L+ flies (Q128) was significantly lowered compared to their s+L+ controls (Q0) (Fig. 1D, upper panel). When both LNv groups (sHLH or NBQ0) or only l-LNv were hyperexcited (s–LH or NBQ128), activity profiles differed from s+L+ controls (NCQ0 and NCQ128, respectively), and they failed to anticipate lights-off (Fig. 1B, third row). Consequently, their activity 4 hours prior to lights-off (ZT8-12) (Fig. 1C) and their evening anticipation indices were significantly lower than that of s+L+ controls and also lower than s–L+ and s±L± flies (Fig. 1D). The sHLH and s–LH flies also poorly anticipated lights-on, with their morning anticipation indices being significantly lower than s+L+ controls (NCQ0) (Fig. 1D, top). Expectedly, the overall nighttime activity of sHLH and s–LH flies was higher than s+L+ controls (NCQ0) (Fig. 1A-C) (Sheeba et al., 2008a), with this difference being statistically significant during ZT16 to 20 (Fig. 1C). The nighttime activity of s–L+ flies of the NCQ128 genotype was as high as sHLH and s–LH flies (Fig. 1C), probably due to an unrelated genetic background effect since s–L+ flies of the Q128 genotype did not show such behavior (Fig. 1C). Thus, these results suggest that the absence of s-LNv specifically reduces the ability of flies to anticipate lights-on without particularly affecting lights-off anticipation. However, hyperexciting either both LNv or only l-LNv reduces lights-off anticipation (Fig. 1D, lower panel). Thus, these results indicate that under LD12:12, the firing properties of l-LNv modulate the anticipation of lights-off.
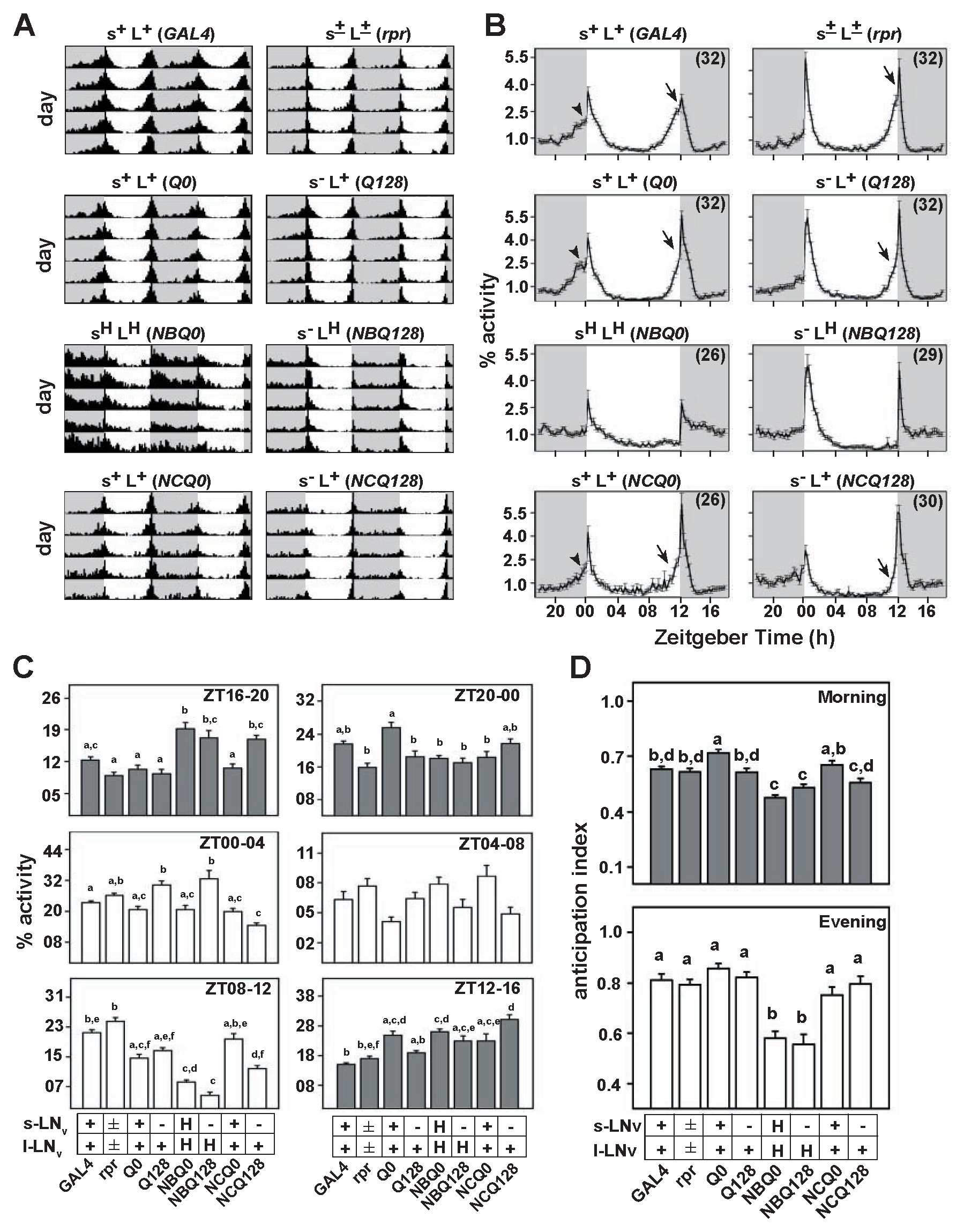
Flies with hyperexcited LNv show reduced anticipation to L/D transition. (A) Average actograms of LNv-modified flies are shown along with their respective controls in LD12:12. The s–LH flies (NBQ128) show reduced nocturnal activity compared to sHLH flies (NBQ0). Actograms of all other genotypes, including s–L+ (Q128) and s±L± (rpr), resemble that of s+L+ controls (GAL4). (B) Normalized activity profiles of the same genotypes each averaged across 7 days are plotted. Number of flies of each genotype is shown in parentheses. The presence of morning and evening anticipation is indicated by slanted arrowheads and arrows, respectively. sHLH and s–LH flies show reduced anticipation to both D/L and L/D transitions. ZT0 is considered as lights-on. The empty and shaded areas in both A and B denote day and night, respectively. (C) Comparison of percentage activity in 4-hour time durations across the length of day and night. X-axis: “+” and “–” indicate presence and absence; “±” indicates partial ablation; and “H” indicates hyperexcitation of s-LNv and l-LNv along with genotype notations. Within each 4-hour interval, different letters above bars indicate that those values are significantly different from each other (ANOVA followed by Tukey HSD, p < 0.05), while bars that have shared letters are not significantly different from each other. Activity levels of sHLH and s–LH flies during ZT8 to 12 and ZT16 to 20 are significantly lower than s+L+ flies. (D) Anticipation of lights-on and -off. Mean anticipation and SEM are plotted.
Electrical Activity of l-LNv Determines Phase, While s-LNv Regulate Amplitude of Evening Peak under Short-Day Conditions
Having examined the role of LNv under LD12:12 conditions, we extended our studies to shorter photoperiods. Previous studies have implicated a dominance of s-LNv in being able to influence oscillators in other neurons of the circadian circuit, although their method could not distinguish between s- and l-LNv (Stoleru et al., 2007). Under short day LD8:16, both morning and evening peaks of s+L+ controls occurred during the dark phase (Fig. 2A and Suppl. Fig. S2), with no startle response to lights-on, while startle to lights-off persisted (ZT0-4 and ZT8-12; GAL4, Q0, and NCQ0) (Fig. 2A and 2B). In contrast, flies with compromised LNv such as s–L+ and s±L± or those with abnormally firing LNv such as sHLH and s–LH show very little “true” morning peak activity, although lights-on startle occurred (Fig. 2A and Suppl. Fig. S2). These differences between controls and LNv-affected flies persisted even in extreme short day LD4:20 (Fig. 2C). Thus, the presence and firing properties of LNv, particularly s-LNv, are critical for phase and amplitude of the M peak under short days. The s–L+ and s±L± flies also showed startle response to lights-off, followed by a low-amplitude evening peak approximately 2 hours after lights-off in LD8:16 (Fig. 2A and 2B, ZT8-12; and Table 1). These results are accentuated under LD4:20, where s–L+ flies (Q128 and NCQ128) (Fig. 2C) showed a relatively low-amplitude “true” evening peak around the same interval (ZT8-12) (Fig. 2D and Table 1) as the higher amplitude evening peaks of the s+L+ controls (Q0 and NCQ0) (Fig. 2C and 2D). Thus, under short days, evening peak amplitude is dependent on s-LNv. Under both LD8:16 and LD4:20, sHLH and s–LH flies did not show clear evening peak of activity (Fig. 2A and 2C), with activity remaining similar to that after lights-off startle had subsided. Evening peak of sHLH flies under LD8:16 may have dampened, probably reflecting a broadly spread out activity at night (Fig. 2A), and along with s–LH flies shows significantly lowered activity in the 4-hour time interval after lights-off (ZT8-12) (Fig. 2B). Under extreme short days, sHLH and s–LH flies showed significant reduction in activity during the 8-hour interval when control flies showed evening peak (ZT4-8 and ZT8-12) (Fig. 2D). However, activity of sHLH flies during ZT8 to 12 did not differ from GAL4 and Q0 controls (Fig. 2D), as controls underwent dramatic changes in activity levels, with activity peaking around ZT8 and then dropping steeply, whereas activity of sHLH flies remained at a constant high level (Fig. 2C). Previous studies have shown that sHLH flies exhibit complex rhythms under DD, with one short and one long period (Sheeba et al., 2008d). We speculate that under extremely short LD4:20 photoperiod and to some extent under LD8:16, this long period component may contribute to the broadening of nighttime activity. Since s–LH flies are arrhythmic under DD, it is unlikely that long period components would have contributed to the broadening of evening peak in this genotype. If l-LNv did not influence evening peak, then in s–LH flies, the dysfunction of s-LNv should have led to the generation of a low-amplitude, correctly phased evening peak, but it is diminished in these flies. Thus, we speculate that the membrane properties of l-LNv regulate the phase of evening peak. Since s–LH and sHLH exhibit similar behaviors under short days, electrical activity of l-LNv appears to be sufficient for phasing the E peak, while a reduction in E peak amplitude of s–L+ flies suggests that s-LNv regulate amplitude (Figs. 2 and 4). While startle to lights-on can be altered by the membrane properties of LNv, evening startle is largely unaffected by it, suggesting that this positive masking is not modulated by either of the LNv.
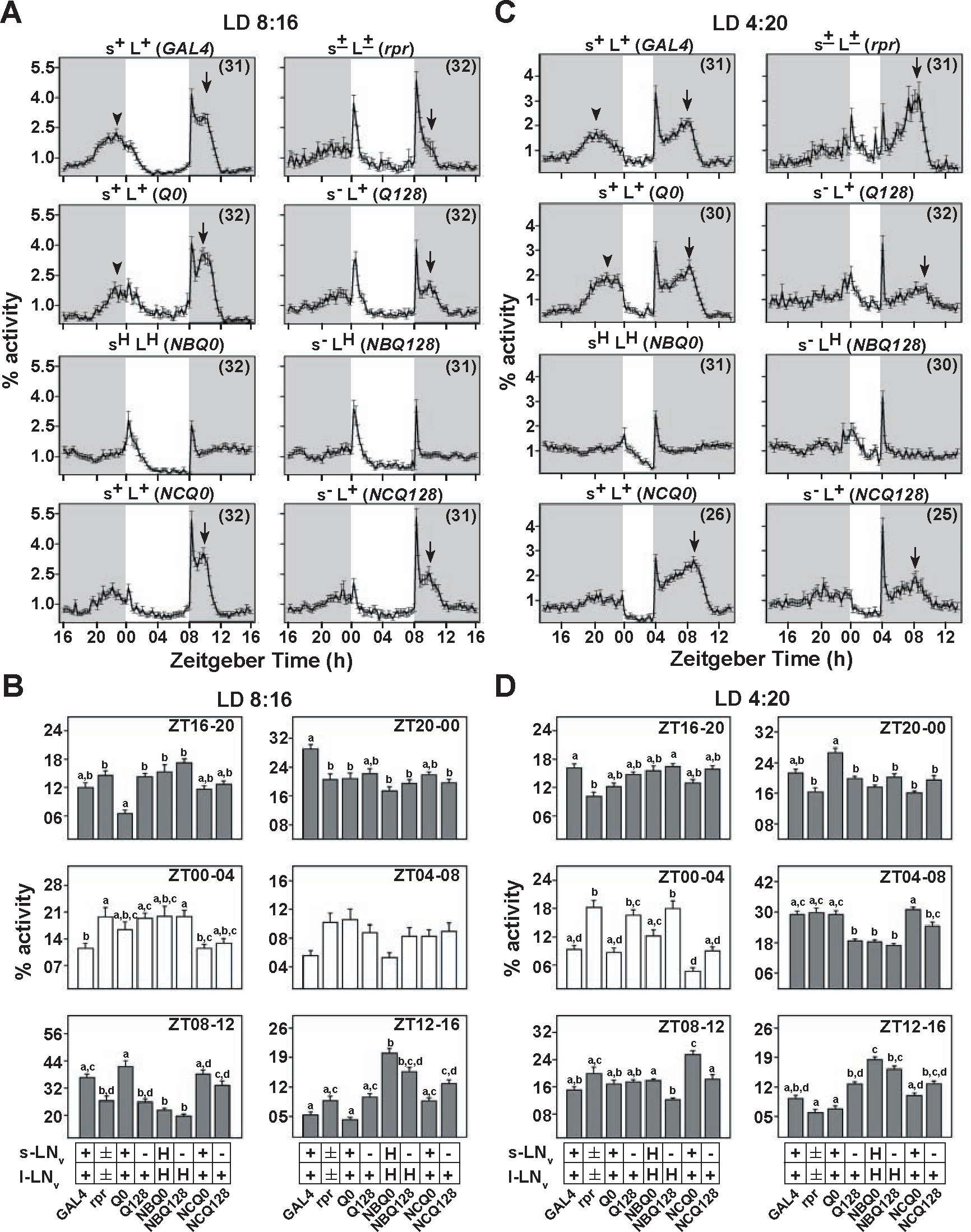
Flies with hyperexcited l-LNv display reduced evening peak under short and very short photoperiods. (A) Normalized activity profiles of different genotypes in LD8:16. sHLH and s–LH flies show relatively fragmented activity during early night, whereas a delayed evening peak is characteristic of control flies. Vertical arrows and arrowheads indicate the presence of nonstartle “true” morning and evening peaks, respectively. The amplitude but not phase of evening peak is affected in s–L+ flies. Partial ablation of LNv (s±L±) dampens evening peak, although not as dramatically as hyperexcitation of LNv. (B) Comparison of activity levels in the same photoperiod across different 4-hour time intervals. During ZT8 to 12, the flies with modified LNv show significantly lesser activity when compared with their respective controls, except for s–L+ (NCQ128). (C) Normalized activity profiles in LD4:20 show that sHLH and s–LH flies show phase-dispersed activity after lights-off as opposed to a correctly phased evening peak seen in controls. Flies with partially ablated LNv (s±L±) show only the delayed evening peak but do not show a morning peak. As in LD8:16, flies with a specific dysfunction of s-LNv (s–L+) show a decreased amplitude of evening peak, although phase is unaffected. (D) Activity levels during different 4-hour intervals show that activity levels during ZT4 to 8 are significantly reduced from their respective controls in all LNv-manipulated flies, except in rpr. All other details are the same as in Figure 1.
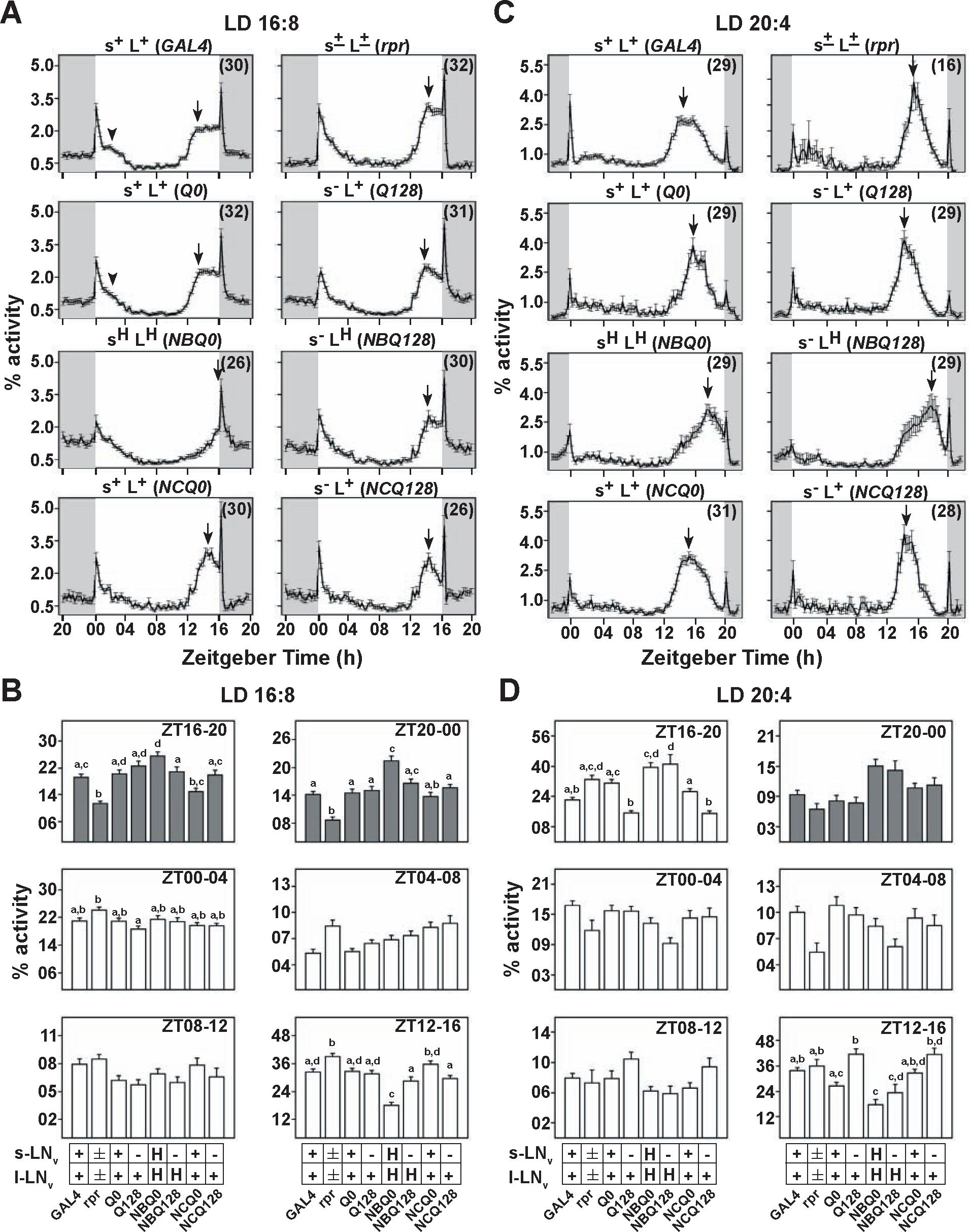
Hyperexcitation of LNv delays the evening peak under long and very long photoperiods. (A) Normalized activity profiles in LD16:8 clearly show the advanced evening peak of controls and s–L+, s±L±, and s–LH flies. However, sHLH flies show a delayed evening peak so that it coincides with lights-off. (B) Comparison of activity levels at different 4-hour time intervals shows that activity in ZT12 to 16 is significantly reduced in sHLH flies when compared with other genotypes. (C) Normalized activity profiles in LD20:4 show an approximately 5-hour advanced evening peak seen in controls, s–L+ and s±L± flies. However, sHLH and s–LH flies show a delayed evening peak so that it occurs approximately 3 hours before lights-off. (D) Activity levels of sHLH and s–LH flies are significantly reduced compared to NCQO control in ZT12 to 16, while it is significantly increased in ZT16 to 20. Other details are the same as in Figure 2.
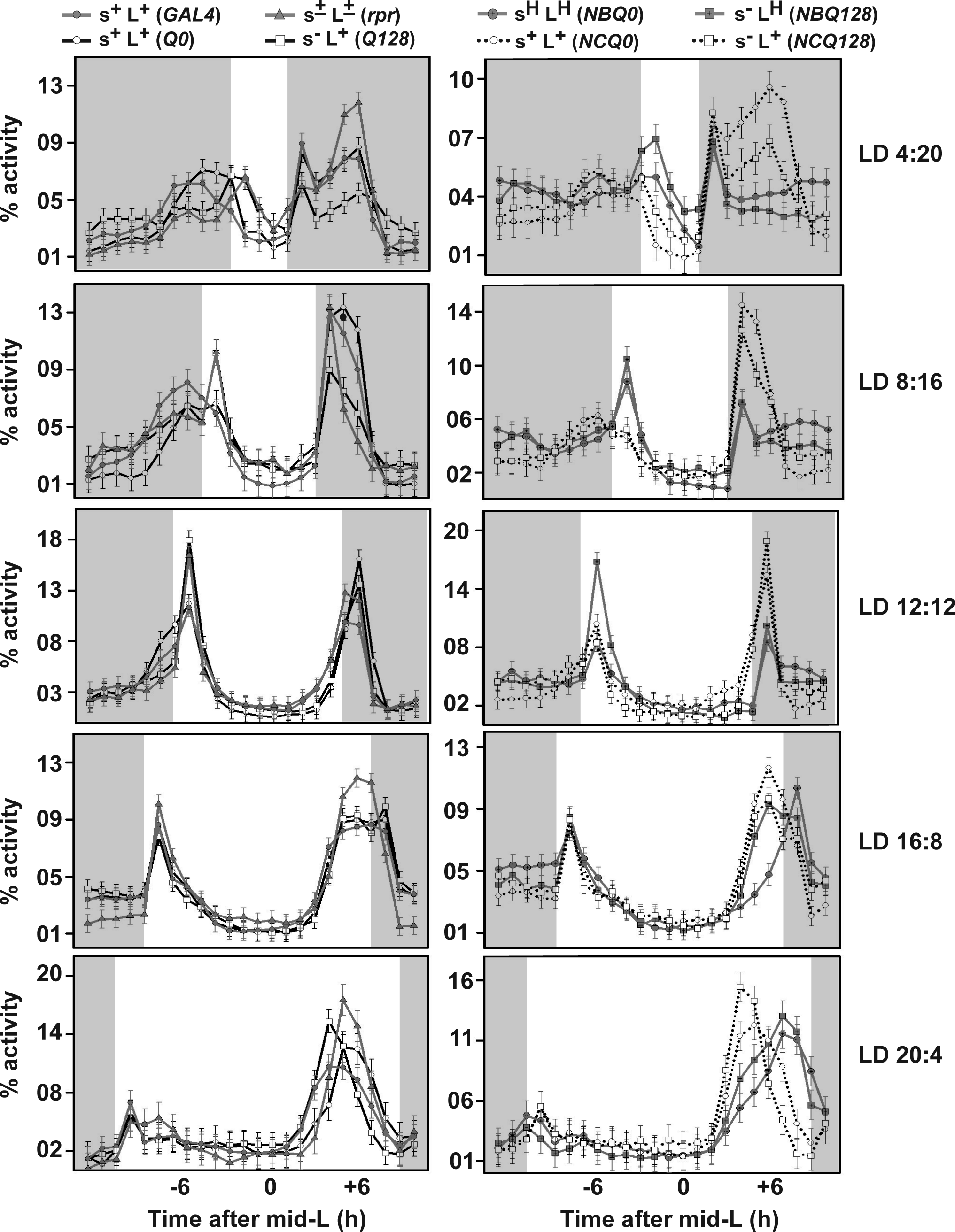
Comparison of activity/rest profiles of different genotypes across different photoperiod conditions. Activity counts that were binned into 1-hour intervals are plotted as percentage activity normalized to total activity during the 24-hour day. The gray highlighted regions represent dark periods of the 24-hour cycle. Error bars represent 95% confidence intervals around the mean. Nonoverlapping error bars within the same genotype across different time points in each photoperiod, except in LD12:12, were used to determine activity peaks. Phasing of evening peak is affected in flies with compromised LNv across all photoperiods, most notably in the sHLH flies.
Phase of evening peaks of all genotypes across different photoperiods with respect to lights-off in hours.
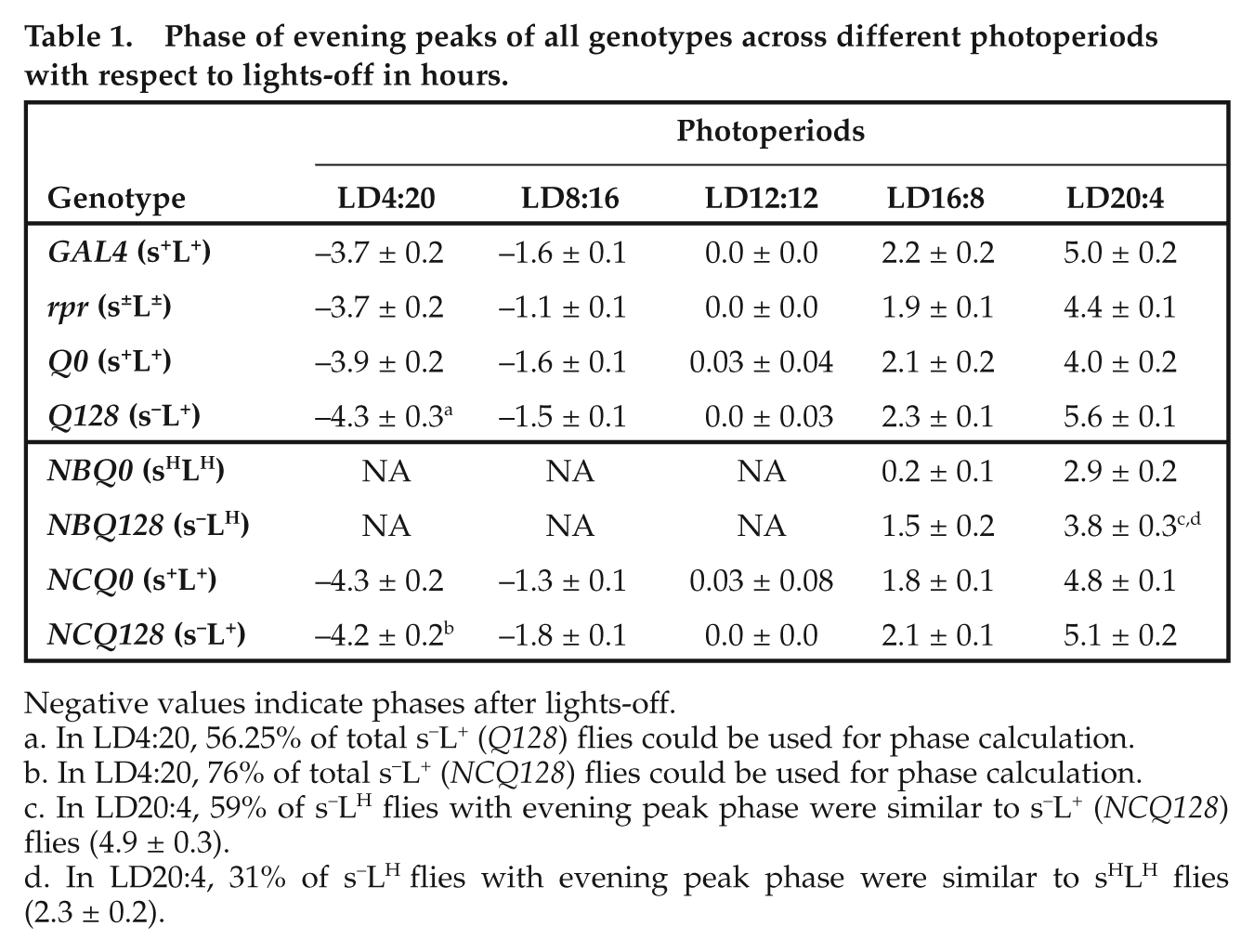
Negative values indicate phases after lights-off.
In LD4:20, 56.25% of total s–L+ (Q128) flies could be used for phase calculation.
In LD4:20, 76% of total s–L+ (NCQ128) flies could be used for phase calculation.
In LD20:4, 59% of s–LH flies with evening peak phase were similar to s–L+ (NCQ128) flies (4.9 ± 0.3).
In LD20:4, 31% of s–LH flies with evening peak phase were similar to sHLH flies (2.3 ± 0.2).
Electrical Activity of s-LNv and l-LNv Modulates Phasing of Evening Peak under Long-Day Conditions
We next tested flies under long days beginning with a photophase of 16 hours. Here, morning and evening activity peaks were almost completely restricted to the light phase (Fig. 3A and Suppl. Fig. S2). Furthermore, all flies including controls showed a clear morning peak, coinciding with lights-on, and startled in response to both lights-on and -off (Fig. 3A). Flies in which s-LNv were compromised (s–L+, s±L±, and s–LH) are similar to controls only in terms of their advanced evening peak phase under LD16:8 (Figs. 3A and 4 and Table 1). Therefore, s-LNv alone are likely not critical for the maintenance of the evening peak under long photoperiods. In contrast, sHLH flies showed a markedly different activity/rest profile compared to all other genotypes, with the evening peak delayed and occurring in conjunction with lights-off (Figs. 3A and 4 and Table 1). Significantly lower activity is exhibited by sHLH flies during ZT12 to 16 compared to all other genotypes (including s–LH), which are at the peak of E activity (Fig. 3B). s-LNv appear to modulate the evening peak phase when hyperexcited, although the absence of s-LNv does not significantly modify it. Thus, when both LNv are hyperexcited, their combined action causes the evening peak to be phase-delayed and aligned with lights-off, suggesting that LNv firing properties are responsible for the phasing of evening peak. With night being only 8 hours long, in this photoperiod, sHLH flies showed a significantly higher activity compared to its controls throughout the night (ZT16-24) (Fig. 3A and 3B). Although s–LH flies exhibited lower nighttime activity compared to sHLH flies, this difference was statistically significant only in the first half of the night (ZT16-20) (Fig. 3B). Taken together, these results demonstrate the role of LNv both in the arousal and circadian circuits under long days since their electrical activity can modify both activity levels at night and phase of the evening peak.
Electrical Activity of l-LNv Regulates Phasing of Evening Peak under Extreme Long-Day Conditions
Based on the results of LD16:8 long-day photoperiod, we surmised that increased firing of LNv could alter phasing of the evening activity peak, which agrees with the predictions from previous studies that have used very different approaches (Stoleru et al., 2004; Rieger et al., 2006). Nevertheless, we subjected flies to an even longer photoperiod (LD20:4) to test their ability to phase their activity peaks when only l-LNv or both LNv are present in the hyperexcited state. Under such extremely long days, we found that s+L+, s–L+, and s±L± flies showed a small morning peak, which is most likely a startle response in conjunction with lights-on, and an advanced evening peak approximately 4 to 6 hours prior to lights-off (Fig. 3C and 3D, Suppl. Fig. S2, and Table 1). Compared to LD16:8, where phase of the evening peak of sHLH flies differed from all others (Fig. 3A), surprisingly, in LD20:4, the activity/rest profiles of sHLH and s–LH flies were not different from each other (Fig. 3C). Here, flies of both genotypes showed a delayed evening peak, such that it occurred approximately 3 hours prior to lights-off, while s+L+ (NCQ0) and s–L+ (NCQ128) controls peaked approximately 5 hours before lights-off transition (Figs. 3C and 4 and Table 1). These differences in phasing of the evening peak were reflected in the 4-hour interval analysis, where the activity levels of sHLH and s–LH flies 4 hours prior to lights-off (ZT16-20) were significantly higher than that of controls (Fig. 3D). In the 4-hour interval between ZT12 to 16, the activity levels of sHLH flies were significantly lower than controls (Fig. 3D). The activity levels of s–LH flies in this interval were not significantly different from controls, possibly because of within-genotype variance in phasing of the evening peak. Indeed, approximately 59% of these flies showed a peak that occurred approximately 5 hours before lights-off (around the same interval as NCQ128 controls) (Table 1), while the remaining flies showed a peak within 3 hours prior to lights-off (around the same interval as sHLH flies). This result indicates that a fraction of s–LH flies were able to align their evening peak similar to controls even in the absence of s-LNv, perhaps due to hyperexcitation of l-LNv. Thus, under the extremely long photoperiod, flies seem to require proper firing properties of both LNv to appropriately phase evening activity peaks. Hyperexcitation of LNv appears to have an overall effect of pushing the evening activity peaks closer to lights-off under long days, probably due to a long period component.
Discussion
Previous studies have shown that l-LNv play an important role in governing morning activity under LD12:12 in the absence of s-LNv (Sheeba et al., 2010). We demonstrate that l-LNv are important in setting the phase of the evening peak in a wide variety of photoperiods (Fig. 4), ranging from extreme short days (LD4:20) up to extreme long days (LD20:4). Furthermore, our results suggest that s-LNv can modulate nighttime activity in some photoperiods, especially during the first half of night (ZT8-12 in Fig. 2D and ZT16-20 in Fig. 3B) (Suppl. Fig. S2), where s–LH show significantly lower activity compared to sHLH flies.
Although we employ chronic ectopic expression of non-Drosophila proteins using the GAL4-UAS method, it is limited to a very small number of neurons and does not produce either lethal or sublethal outcomes (as ascertained by life span assays) (data not shown). Previously, we have shown that l-LNv do not exhibit any detectable changes in electrophysiological properties up to 20 days of age even when both LNv are expressing HTT-Q128 (Sheeba et al., 2010). However, when NaChBac is chronically expressed, the changes in membrane properties do alter neurotransmitter profiles, as visualized by the constitutively high levels of PDF in projections from s-LNv (Nitabach et al., 2006). Thus, the chronic alteration in membrane properties besides changing firing rate and amplitude may cause other yet unknown changes in neuronal circuit properties. Therefore, we acknowledge that l-LNv may not be physiologically completely normal when expressing HTT-Q128 and/or NaChBac.
While our results show that s-LNv are important in phasing the morning peak under LD12:12 and extremely short-day condition, in other photoperiods, they appear to be dispensable (Q128 and NCQ128) (Fig. 4) for this function. In addition, we find that under short days, in control flies, startle response to lights-on is highly reduced (GAL4, Q0, and NCQ0) (Figs. 2 and 4). We propose that this is due to the inhibitory action of normally firing s-LNv and that this inhibition is lost when there is a partial or complete loss of s-LNv (rpr and Q128) (Fig. 4), suggesting that under short-day conditions, they have an inhibitory role on light-mediated immediate increase in activity. When hyperexcited, l-LNv, which are arousal neurons that strongly respond to lights-on, appear to dominate this effect of s-LNv (Fig. 4). Previously, Rieger et al. (2003) have reported the separation of morning startle and nonstartle peaks under short days in wild-type Canton-S flies, which have functional s-LNv. We have also found similar results for the s+L+ controls in an experiment when the same flies were subjected to different photoperiods as they aged (Suppl. Fig. S3).
Our study reveals a previously unknown secondary role for s-LNv in the arousal circuit. It also reveals previously undetected differences in nighttime activity between flies with small and large hyperexcited LNv and those with only hyperexcited l-LNv (Parisky et al., 2008; Shang et al., 2008; Sheeba et al., 2008a). We speculate that s-LNv form the link between l-LNv and other limbs of the sleep circuit, thus bridging an obvious anatomical gap.
We find that under short-day conditions, l-LNv are critical for setting the phase of the evening peak. Two separate studies have shown that phase of the evening peak under LD12:12 is determined in at least 2 ways. One study suggests that light input perceived by l-LNv and communicated via PDF to LNd and the fifth s-LNv determines the phase of the evening oscillator, even in the absence of CRY (Cusumano et al., 2009). However, they show that these oscillators can set their own phase even in the absence of PDF, using visual inputs perceived via CRY. A second study suggested that CRY is responsible for gating of PDF signaling (Zhang et al., 2009). The importance of PDF in regulating the period and phase of clock neurons has previously been demonstrated using pdf 01 mutants. Moreover, these mutants are unable to normally adapt to long-day photoperiods (Yoshii et al., 2009). This is also consistent with studies on mutants with increased PDF projections in the accessory medulla (Wülbeck et al., 2008, 2009). These results are in congruence with our own, and we speculate that l-LNv hyperexcitation causes greater PDF secretion in the accessory medulla, where l-LNv and s-LNv have close contact (Helfrich-Förster et al., 2007). This is plausible since hyperexcitation has been demonstrated to cause constitutively high PDF secretion from the s-LNv projections in the dorsal protocerebrum (Nitabach et al., 2006).
Here, we show that under short-day conditions, the membrane properties of l-LNv critically affect phase of the evening peak. Even under LD12:12, firing properties of l-LNv exert influence on the evening peak since hyperexcitation of l-LNv alone is enough to reduce anticipation of lights-off (Fig. 1B and 1D). Interestingly, a previous study has shown that light information signaled from the compound eyes is important for entrainment to extremely long and short photoperiods, while CRY is specifically required for entrainment to short photoperiods (Rieger et al., 2003). Our results point toward an important role for electrical activity–mediated signals from l-LNv influencing activity profiles across almost all photoperiods. In support of our claim that l-LNv communication is important, a recent study showed that flies that are doubly mutant for pdfr and cry, or alternatively pdf and cry, show activity/rest profiles that are strikingly similar to those in which LNv are hyperexcited, where the evening peak is highly diminished under short days (Im et al., 2011). Interestingly, pdfr single mutants showed a phase-advanced evening peak even under short-day conditions, suggesting that downstream neurons are capable of producing evening peak in the presence of functional CRY, albeit with an erroneous phase. However, in the absence of both cry and pdfr, flies became incapable of producing the “true” evening peak, similar to our finding that upon hyperexcitation of l-LNv, E peak is diminished (Fig. 4, top 2 rows). Intriguingly, pdfr/cry double mutants did not exhibit a “true” evening peak even in a long photoperiod, whereas pdfr single mutants displayed a severely phase-advanced evening peak. In comparison, LNv hyperexcited flies showed a delayed evening peak, whereas flies with dysfunctional s-LNv, despite hyperexcitation of l-LNv, showed a phase of evening peak similar to that of the control under LD16:8. Thus, under long day, unlike short day, communication of both s-LNv and l-LNv with their target neurons is paramount in the generation of correctly phased evening peak.
Our study provides strong evidence for the existence of a plastic circadian neuronal network by demonstrating that electrical activity of l-LNv enables flies to deal with varying day lengths by appropriately changing the phase of the evening activity peak with respect to lights-off. The phases of the morning and evening peaks do not follow the dawn and dusk transitions in a dedicated fashion, as posited by the original dual-oscillator model; instead, we propose that the phase relationship between the M and E oscillators is flexible only up to a certain limit. Thereafter, the oscillators become inefficient in tracking dawn and dusk signals under shrinking or expanding photoperiods, as seen in our study and by others (Rieger et al., 2003; Stoleru et al., 2007; Rieger et al., 2012). We propose that l-LNv maintain the phase relationship between oscillators located in s-LNv, LNd, and fifth s-LNv across photoperiods (Fig. 5). Since l-LNv are light responsive (Sheeba et al., 2008b), we speculate that l-LNv represent a group of neurons that are capable of measuring day length, as they fire continuously in the presence of light. In conclusion, we propose a vitally important role for l-LNv in bringing about an alteration in the phase of the evening peak. We opine that the circadian clock network has distinct but flexible roles for neurons belonging to each subset, such that according to the environmental conditions, they interact to bring about overt behavior. Altogether, the significance of l-LNv in the clock circuit and a secondary role of s-LNv in the arousal circuit demonstrate the ability of neurons to transcend circuits and play critical roles apart from their primary functions.
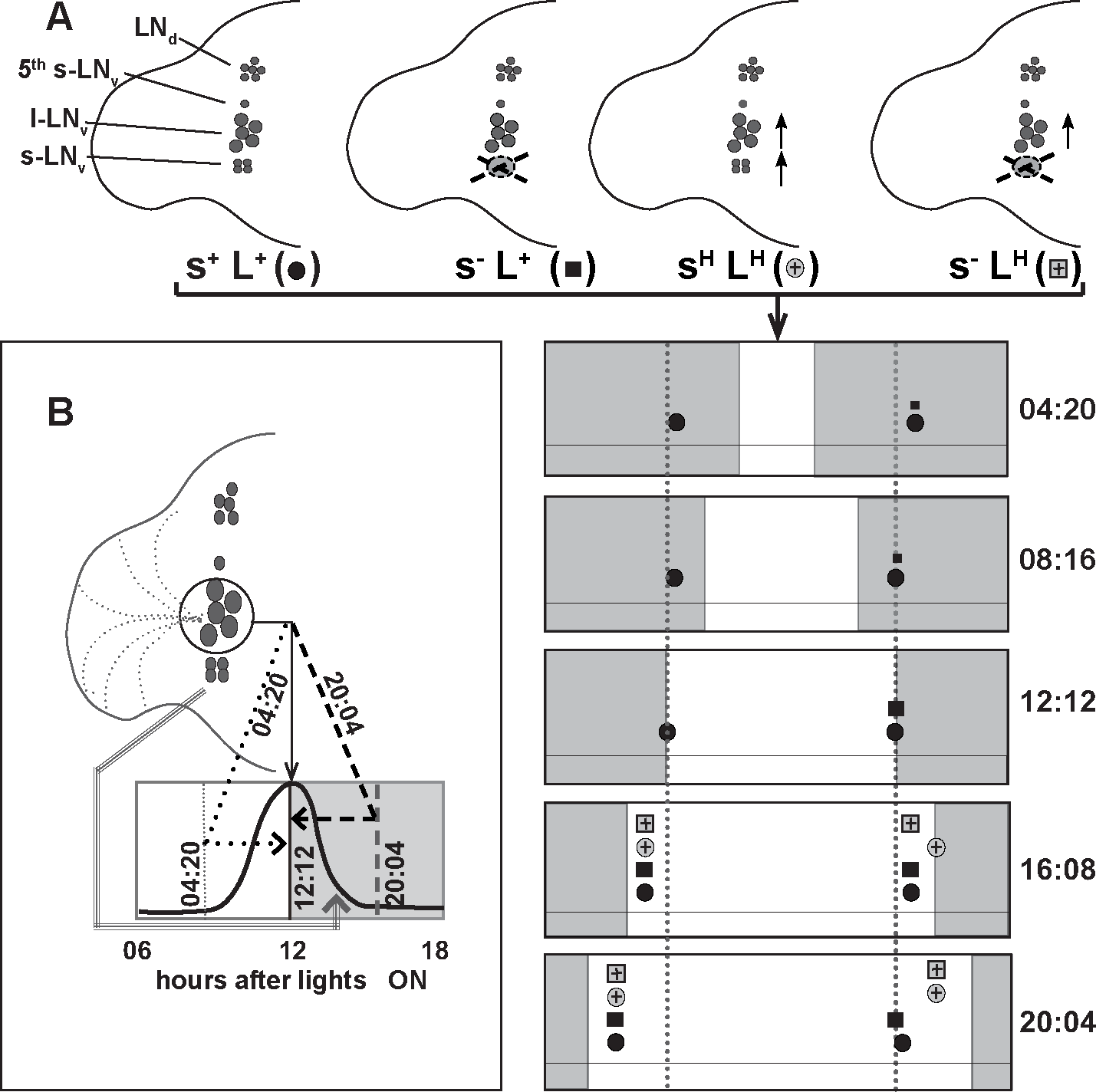
A proposed model for the role of lateral neurons in the adaptation to seasonal variations. (A, upper) Schematic representations of various perturbations to LNv (s+L+) control; (s–L+) dysfunctional s-LNv, normally firing l-LNv; and (sHLH) hyperexcited both LNv and (s–LH) dysfunctional s-LNv, hyperexcited l-LNv. Solid-line cell boundaries indicate normally firing neurons, while dotted lines indicate dysfunctional neurons. Vertical arrows next to a group of neurons indicate that the group is hyperexcited. (A, lower) Different photoperiod conditions are indicated by the white (day) and gray (night) shaded regions. Symbols of different genotypes indicated in the upper panel are used to indicate the presence and phases of “true” morning and evening peaks of genotypes across different photoperiods. Dotted vertical line is aligned to lights-on and -off in LD12:12 to aid the comparison of peak phase in other photoperiods. In short photoperiods (LD4:20 and LD8:16), only controls exhibit “true” morning and evening peaks. The s–L+ flies exhibit a low-amplitude evening peak (indicated by smaller sized squares) at the same phase as the control evening peak. Neither sHLH nor s–LH display prominent morning or evening peaks, indicated by the absence of their symbols on top of the photoperiod bars. In LD12:12, control and s–L+ flies display anticipation of lights-off and thus the “true” evening peak. Only controls show anticipation of lights-on and thus the “true” morning peak. In long-day conditions (LD16:8 and LD20:4), all genotypes show a morning peak close to lights-on. The evening peaks of the controls and s–L+ and s–LH flies are phase-advanced as compared to that of sHLH, whose peak is close to lights-off especially in LD16:8. In LD20:4, the evening peaks of both s–LH and sHLH flies are delayed when compared with the controls and s–L+ flies. (B) Model proposing that l-LNv determine the phase of the evening peak (solid, dotted, and dashed arrows) and s-LNv modulate early night activity levels (gray arrow). Empty and shaded areas denote 12 hours around lights-off under LD12:12. Dotted, solid, and dashed vertical lines denote the lights-off transition under LD4:20, LD12:12, and LD20:4 regimes, respectively. Dotted arrows and dashed arrows indicate the repositioning of the evening peaks under short (LD4:20) and long (LD20:4) photoperiods, respectively. Under LD4:20, E peak is delayed by about 4 hours after lights-off, while under LD20:4, E peak is advanced to occur approximately 4 to 5 hours before lights-off.
Footnotes
Acknowledgements
The authors thank Todd Holmes and Troy Littleton for fly lines. They also thank 3 anonymous reviewers for critical comments on a previous version of the article. Vijay Kumar Sharma, Pavitra Prakash, and other laboratory members are gratefully acknowledged for comments and suggestions, as are Rajanna and Muniraju for assistance. This work was supported by a Ramanujan Fellowship (Department of Science and Technology) to V.S. The Jawaharlal Nehru Centre for Advanced Scientific Research provided financial assistance to S.P. in the form of a fellowship.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
