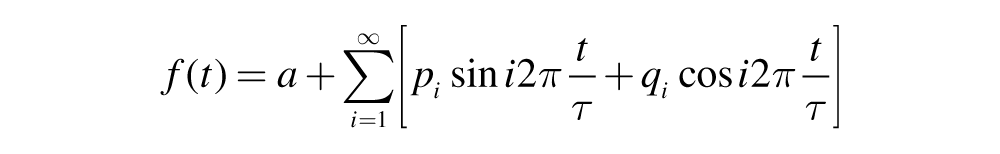Abstract
As both a photoreceptor and pacemaker in the avian circadian clock system, the pineal gland is crucial for maintaining and synchronizing overt circadian rhythms in processes such as locomotor activity and body temperature through its circadian secretion of the pineal hormone melatonin. In addition to receptor presence in circadian and visual system structures, high-affinity melatonin binding and receptor mRNA are present in the song control system of male oscine passeriform birds. The present study explores the role of pineal melatonin in circadian organization of singing and calling behavior in comparison to locomotor activity under different lighting conditions. Similar to locomotor activity, both singing and calling behavior were regulated on a circadian basis by the central clock system through pineal melatonin, since these behaviors free-ran with a circadian period and since pinealectomy abolished them in constant environmental conditions. Further, rhythmic melatonin administration restored their rhythmicity. However, the rates by which these behaviors became arrhythmic and the rates of their entrainment to rhythmic melatonin administration differed among locomotor activity, singing and calling under constant dim light and constant bright light. Overall, the study demonstrates a role for pineal melatonin in regulating circadian oscillations of avian vocalizations in addition to locomotor activity. It is suggested that these behaviors might be controlled by separable circadian clockworks and that pineal melatonin entrains them all through a circadian clock.
The avian pineal gland and its hormone, melatonin, play an important role in maintaining and entraining circadian rhythms (Cassone and Menaker, 1984; Gwinner et al., 1997; Bentley, 2001; Gwinner and Brandstätter, 2001; Brandstätter, 2003). In the house sparrow, Passer domesticus, pinealectomy abolishes free-running circadian rhythms of locomotor activity in constant darkness (DD) (Gaston and Menaker, 1968), and constant light (LL) abolishes circadian rhythms of locomotor activity in intact birds (Binkley, 1976; McMillan, 1972). Rhythmic administration of melatonin restores the rhythmicity of locomotor activity under both conditions above (Lu and Cassone, 1993; Heigl and Gwinner, 1994, 1995; Cassone et al., 2008).
Based upon the widespread distribution of melatonin binding sites in the brains of more than 15 species of birds (Rivkees et al., 1989; Cassone et al., 1995), one would expect many aspects of avian behavior to be controlled by the pineal hormone. The song control system of male but not female oscine passeriform birds contains high-affinity melatonin receptors (Whitfield-Rucker and Cassone, 1996; Gahr and Kosar, 1996; Bentley and Ball, 2000; Jansen et al., 2005), suggesting that this important behavioral system is also regulated by the pineal clock. In European starlings, Sturnus vulgaris, continuous administration of exogenous melatonin attenuates the long-day-induced increase in HVC volume and also decreases the volume of another song control nucleus, area X. This effect is independent of reproductive state (Bentley et al., 1999). In house sparrows maintained in constant light, simulation of the duration of blood melatonin titers in different seasons regulates the size of song control nuclei (Cassone et al., 2008). Rhythmic melatonin administration of both long and short durations (14 h or 8 h per 24 h, respectively) entrains locomotor activity, while control birds receiving vehicle treatment remain arrhythmic. Birds receiving long melatonin durations, which are consistent with blood melatonin durations during short, winter-like photoperiods, have smaller HVC and robust nuclei of the archistriatum (RA) than do those receiving short melatonin durations, consistent with long photoperiods. Birds receiving no melatonin at all also exhibit large HVC and RA. Again, these effects are independent of reproductive state. Together, these effects of melatonin on the song control system point to a role for pineal melatonin on avian vocalizations themselves.
In zebra finches, Taeniopygia guttata, administration of the Mel1B melatonin receptor antagonist luzindole at the beginning of the night shortens the song length produced the following day, while song and motif length do not vary over the course of the daytime. This suggested to the authors a noncircadian role for melatonin in song behavior (Jansen et al., 2005). However, this study did not investigate the effect of luzindole on the rhythmic occurrence of song under different light conditions. In the present study, we test the hypothesis that pineal melatonin regulates the circadian organization of bird vocalization. We investigate song and call separately in addition to locomotor activity to ask whether all 3 behaviors are controlled by separable circadian clockworks and whether rhythmic melatonin release from the pineal synchronizes them through entrainment of a circadian clock.
Materials and Methods
Subjects and Data Acquisition
Unpaired adult male zebra finches (N = 14) were housed in individual cages with finch food (Purina Exact) available ad libitum under LD 12:12 light/dark (12L 12D) (lights on at 6 AM and lights off at 6 PM) conditions before the onset of the experiments. Birds were then transferred into individual cages (42 cm L × 48 cm W × 40 cm H) in isolation cabinets with finch food (Purina Exact) ad libitum under LDm 12:12 light (40 µW/cm2)/dim light (less than 1E-4 µW/cm2) (12L 12Dm) cycles with constant background white noise (average power = 21 dB, frequency range from 0 to 10 kHz, measured within the cabinets). In each cabinet, both locomotor activity and vocalizations were continuously monitored and recorded. Locomotor activity was recorded with an infrared motion sensor located on the top of the cage inside the cabinet. Activity data were acquired by VitalView data acquisition equipment and software (Mini-Mitter Co., Sunriver, OR) throughout the entire experimental period, and activity records were subsequently either double plotted in actogram format or exported as ascii files for further analysis by using ActiView software (Mini-Mitter Co.). Vocalization data were acquired with Rode NT3 cardioid condenser analog microphones in each chamber and then integrated using a MOTU 24 I/O core computer recording system (MOTU, Inc., Cambridge, MA). Vocalizations were analyzed using Raven Pro 1.4 build 34 beta1 software (Cornell Lab of Ornithology, Ithaca, NY). The sampling frequency was 20 kHz, and the sound files were saved in 16-bit wave format with 2-min bins.
Surgeries and Experimental Protocol
After acclimation in the isolation cabinets for 1 week, birds were subjected to either pinealectomy (PINX; n = 7) (Lu and Cassone, 1993) or a SHAM surgery (SHAM; n = 7). Briefly, birds were anesthetized with 90 mg/kg ketamine and 10 mg/kg xylazine, and the head was secured in a Stoelting stereotaxic apparatus. A scalpel was used to make a longitudinal incision in the scalp, and the exposed skullcap was removed using a dental drill and forceps. For PINX birds, the dura mater was then incised and retracted, and the exposed pineal gland grasped and removed. The skullcap was then replaced, and the scalp sutured shut. For SHAM birds, the skullcap was immediately replaced after the removal and the dura mater was left intact. Birds were allowed to recover in the cabinets for 1 week before being subjected to the later experiments.
In the constant dim light (DmDm) (less than 1E-4 µW/cm2) experiment, all birds were placed in DmDm conditions for 11 days, followed by melatonin (200 µg/mL in 0.5% ethanol [EtOH]) administration (10 h melatonin/24 h 0.5% EtOH) through the drinking water only on PINX birds. To control for disturbance, the drinking water was refreshed for SHAM birds twice a day when melatonin water was placed or removed for PINX birds. After 28 days, melatonin administration was stopped, and birds were maintained in DmDm for a week before light conditions were switched back to the previous 12L 12Dm cycles for 10 days. Birds were then subjected to constant light (LL) (40 µW/cm2). On the 10th day of LL, all birds were given melatonin administration as in the DmDm experiment. After 36 days, melatonin administration was stopped, and birds were maintained in LL for an additional 10 days. Birds were sacrificed; brains were removed and sectioned to validate the success of surgery histologically.
Sound Analysis
Song and call were extracted from sound files with the band limited energy detector function of Raven Pro 1.4 Beta software. The song of an adult male zebra finch consists of a specific syllable pattern (motif), which can be repeated several times in a song bout. In this study, we count 1 motif as 1 song unit. The call of an adult male zebra finch is typically composed of single syllables with a frequency modulated down-sweep of a fundamental frequency and several associated harmonics (Supplemental Figure S1). To extract either song or call, we set up the target signal parameters (include the frequency range, duration range, minimum separation time, signal-to-noise ratio, etc.) separately for either song or call for each bird because there are individual differences in both song and call structures. The sonograms of the extracted song and call were made and screened for accuracy, which was greater than 95%. Circadian patterns of song characteristics were not included in this study, but these data are currently being analyzed as part of a separate study.
Rhythm Analysis
The locomotor activity, song, and call data were arranged in hourly total occurrences for all the observation periods. The arranged data for 2-day (48 h) cycles were subjected to cosinor analysis by using CircWave 1.4 time series analysis software (R.A. Hut, Department of Chronobiology, University of Groningen, Netherlands). Briefly, CircWave is an extension of cosinor analysis designed for estimation of circadian components of a data set such as those described here: locomotor activity, song, and calls. It produces a Fourier curve that describes the data better than a simple linear squares cosinor curve by adding as many harmonics to the wave fit as the data allow. This means that the primary wave form (1 sine wave) plus 1 or 2 harmonics describes the data set. The primary wave is described by the following function:
where a = average; i = 1, 2, 3, indicating the fundamental wave, the first harmonic, the second harmonic, …. (i-1)th harmonic; pi is the sine coefficient of the (i-1)th harmonic and qi the coefficient of the (i-1)th harmonic; t = time point value (modulo τ); f(t) is the calculated function value at time point t. The center of gravity of the waveform is taken as the phase (φ) of the rhythmic data set, and period (τ) is taken as the duration between 2 centers of gravity.
Period estimates by this method were validated for locomotor activity by comparing calculations of τ using the periodogram routine within the Actiview software package. These values were not statistically different.
Analytical Statistics
We plotted daily oscillations of different behaviors in actogram format, hourly proportions of total daily behavioral occurrence, or cosinor fitted waves. All the data sets were first tested for normality with Kolmogorov-Smirnov test and then subjected to appropriate statistical analyses. The rates by which arrhythmicity occurred and the rates of entrainment to melatonin cycles in both SHAM and PINX birds were compared among different behaviors by using matched 2-way ANOVA followed by Bonferroni post hoc. The total amounts of behavior under different conditions were also analyzed by using matched 2-way ANOVA followed by Bonferroni post hoc test. One-sample t test was used to analyze whether the phase differences with locomotor activity are statistically significant for both song and call behavior, and then paired t test was used to compare whether the phase differences with locomotor activity are different between song and call with Bonferroni p value correction for multiple comparison. Because the free-running periods of SHAM birds in DmDm conditions were not distributed normally, they were compared among different behaviors by using a Friedman test followed by Dunn’s multiple comparison test. All statistics were performed by using Graph Pad Prism (GraphPad Software, San Diego, CA).
Results
Daily Oscillation under Light Dark Cycles
In 12L 12Dm, diurnal rhythms were observed in locomotor activity, song, and call (Figures 2A and 3). Birds sang, called, and were active primarily during the light phase. However, the waveform of the diurnal patterns was different among the 3 behaviors. Circwave analysis showed that all 3 behaviors were rhythmic (p < 0.05), and ANOVA showed that the oscillations among these 3 behaviors were different (p = 0.0377) with interactions across time (p = 0.0012). Both song and call showed a robust morning peak, while locomotor activity increased gradually at dawn (ZT0) and remained high throughout the day (Figure 1A). Pinealectomy did not affect the waveform of the diurnal patterns of these behaviors under 12L 12Dm conditions (Figure 1B and 1C) nor did it affect the amount of these behaviors (Supplementary Figure S2).
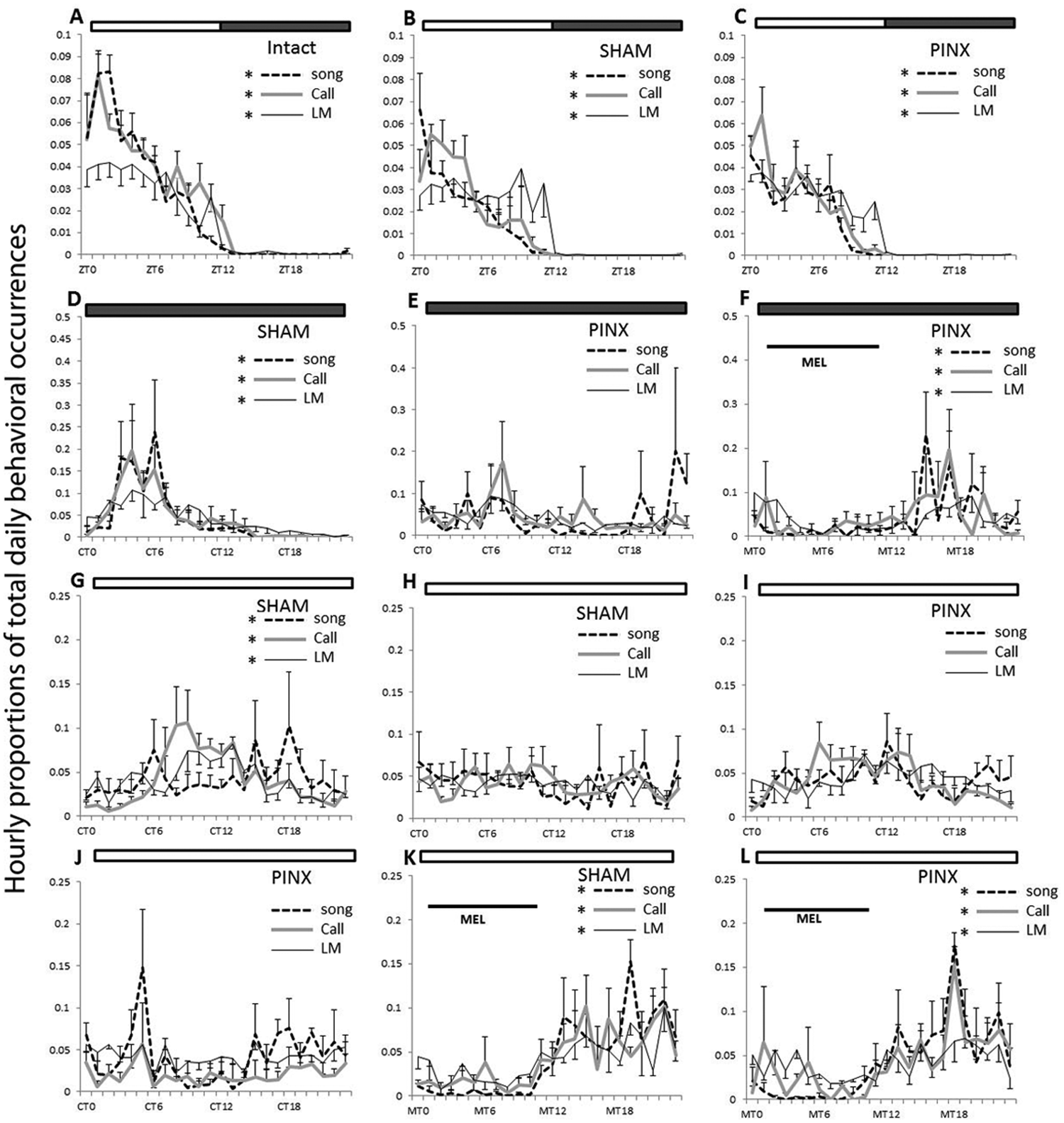
Daily and circadian patterns of hourly behavioral occurrences in locomotor activity (solid black), song (dashed), and call (gray) under different light conditions. Vertical coordinates are measured as hourly proportions of total daily behavioral occurrences normalized for each bird. The bar on the top of each figure indicates 12L 12Dm (white-gray), DmDm (gray), or LL (white) cycles. Horizontal black lines indicate the period of melatonin administration. (A) Daily patterns in intact birds (before surgery) under 12L 12Dm. (B and C) Daily patterns in SHAM (B) and PINX (D) birds under 12L 12Dim after surgery. (D and E) Daily patterns in SHAM (C) and PINX (E) birds under DmDm. (F) Daily patterns during rhythmic melatonin administration under DmDm in PINX birds. (G and I) Daily patterns on the second day of LL conditions in SHAM (G) and PINX (I) birds after transferred from 12L 12Dm. (H and J) Daily pattern on the ninth day of LL conditions in SHAM (H) and PINX (K) birds after transferred from 12L 12Dm. (K and L) Daily patterns during rhythmic melatonin administration in SHAM (I) and PINX (L) birds under LL. Asterisk indicates the presence of rhythmic oscillation for the behavior with matched color, and the position of the asterisk corresponds to the center of the gravity analyzed by cosinor wave fit. Data presented as mean ± SEM. Abbreviations: SHAM = sham-operated; PINX = pinealectomized; ZT = zeitgeber time; CT = circadian time according to locomotor activity; MT =melatonin administration time.
The phases for the centers of gravity of locomotor activity were consistently delayed relative to song (before surgery, t = 1.834, p = 0.1093; after surgery, t = 5.469, p = 0.0009) and calls (before surgery, t = 4.295, p = 0.0036; after surgery, t = 7.338, p = 0.0002), but the difference in phase relationships with locomotor activity between song and call disappeared with pinealectomy (before surgery, t = 4.173, p = 0.0042; after surgery, t = 2.023, p = 0.0828) (Figure 3A and 3B).
DmDm Conditions
SHAM birds continued to express circadian rhythms in all 3 behaviors with free-running periods that were close to but statistically different from each other in that the period (τ) of locomotor activity was significantly shorter than the τ of calls (p = 0.0046) (Figure 1D; Figure 3E). These differences gradually disappeared and likely reflect transients in the locomotor activity rhythms (Figure 3F). Consistent with transients, the phase differences with locomotor activity in 12L 12Dm were maintained for both song (t = 3.454, p = 0.0136) and call (t = 2.45, p = 0.0498) initially, and then gradually disappeared for both song (t = 1.417, p = 0.2921) and call (t = 1.305, p = 0.3218) in DmDm conditions (Figure 3C and 3D).
When released into DmDm, PINX birds gradually became arrhythmic in their locomotor activity, singing, and calling behavior (Figure 1E; Figure 2B), while SHAM birds continued to be rhythmic in all behaviors for the entire experimental period under DmDm but with a circadian free-running period (Figure 1D; Figure 2A). Singing became arrhythmic earlier (within the first day) than did locomotor activity (within 9 days) (p < 0.05) and call behavior (within 9 days) (p < 0.05); there was no difference between locomotor activity and call behavior (Figure 2A). The daily total amount of song and call but not locomotor activity decreased in DmDm compared to 12L 12Dm conditions (p = 0.0313) (Supplementary Figure S2).
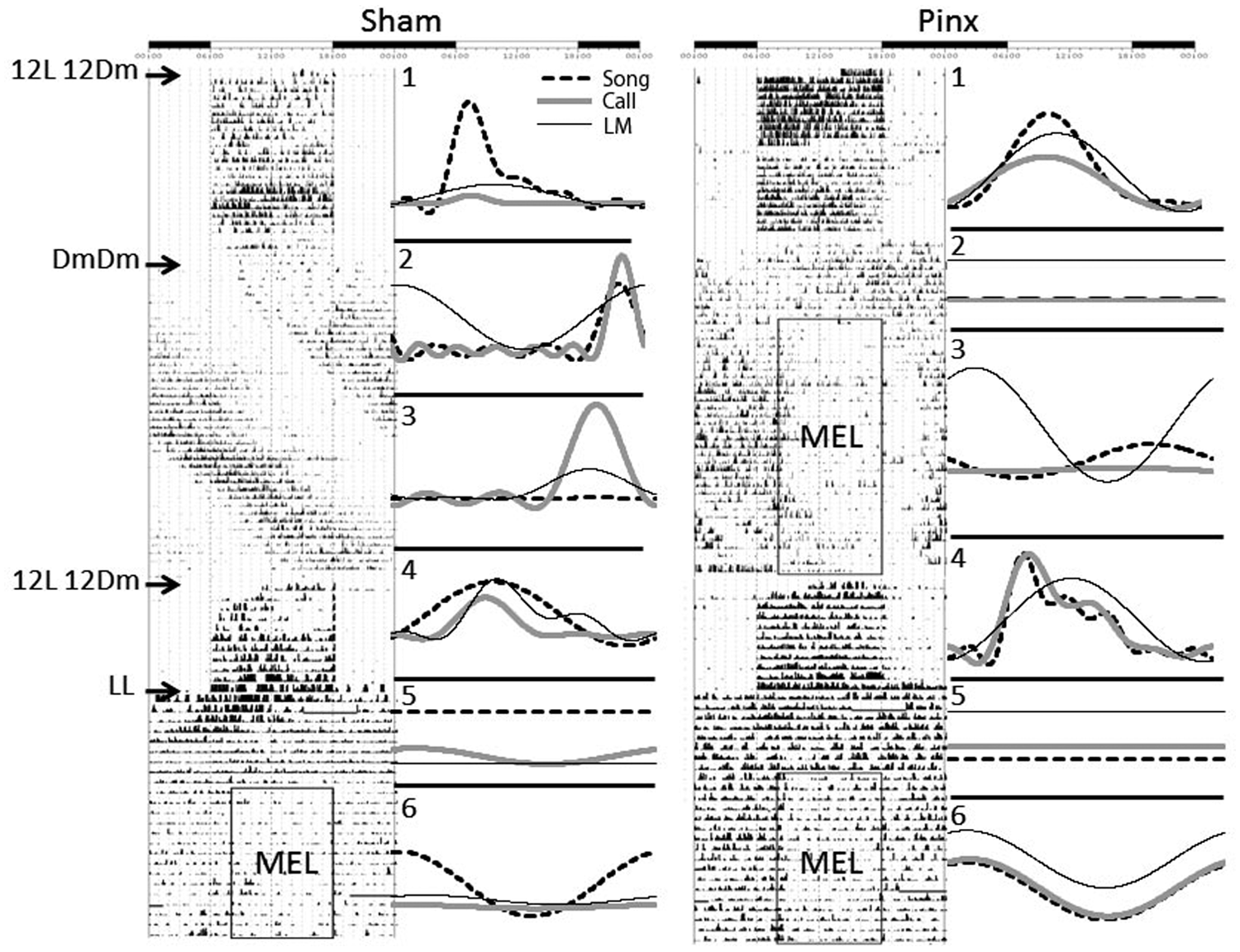
Effects of light conditions and rhythmic melatonin administration on the circadian rhythms of locomotor activity, song, and call in 1 SHAM (A) and 1 PINX (B) bird. Single-plotted actogram on the left of each figure indicates locomotor activity, and the rectangles indicate the period of melatonin administration. The series of subfigures (1-6) on the right of each figure indicate the oscillation of locomotor activity (solid black), singing (dashed), and calling (gray) with cosinor-wave fitting in the conditions corresponding to a certain day in the conditions to the left on the actogram. The bar on the top of each figure indicates the day (white) and night (black) phase in the 12L 12Dm conditions. Abbreviations: DD = DmDm; LD = 12L 12Dim; Mel = melatonin administration; PINX = pinealectomized; SHAM = sham-operated.
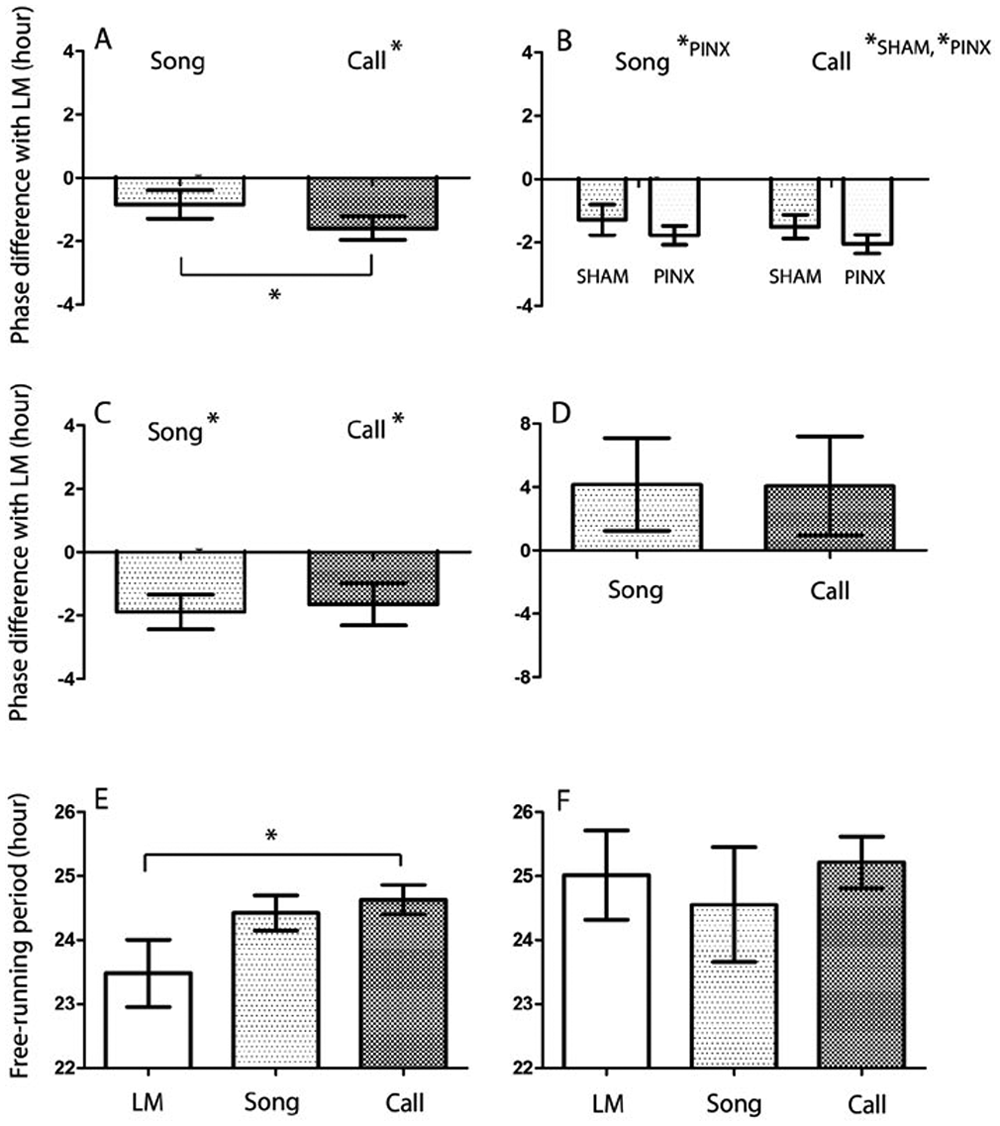
Phase relationship and free-running periods of locomotor activity, song, and call in SHAM birds under DmDm conditions. (A-D) Phase differences of locomotor activity with song and call before surgery in 12L 12Dm (A), after surgery in 12L 12Dm (B) for both SHAM and PINX birds, and for SHAM birds in early DmDm (C) and late DmDm (D). (E-F) Free-running periods of SHAM birds for all 3 behaviors in early DmDm (E) and late DmDm (F). Data presented as mean ± SEM. Significant differences were indicated by asterisk. In A-D, asterisk on the top-right of behavior illustration indicates that the corresponding behaviors have different phase relationships with locomotor activity (positive and negative values indicate phase advance and phase delay, respectively). Abbreviations: LM = locomotor activity; PINX = pinealectomized; SHAM = sham-operated.
After the circadian patterns of all 3 behaviors were abolished in PINX birds, we introduced a rhythmic melatonin regimen (10 h melatonin per 24 h) through the drinking water. Melatonin administration restored and entrained the circadian rhythms of locomotor activity, song, and call (Figure 1F). Locomotor activity was entrained more quickly (around 4 days) than either singing (around 16 days) (p < 0.05) or calling (more than 25 days) (p < 0.05), and singing was entrained more rapidly than was calling (p < 0.05) (Figure 4D). In addition, melatonin administration did not change the daily total amount of locomotor activity, singing, and calling (Supplementary Figure S2). After melatonin administration was discontinued, the rhythms of all 3 behaviors were maintained for a few cycles before becoming arrhythmic again (Figure 5), indicating that they were entrained to the regimen rather than simply masked.
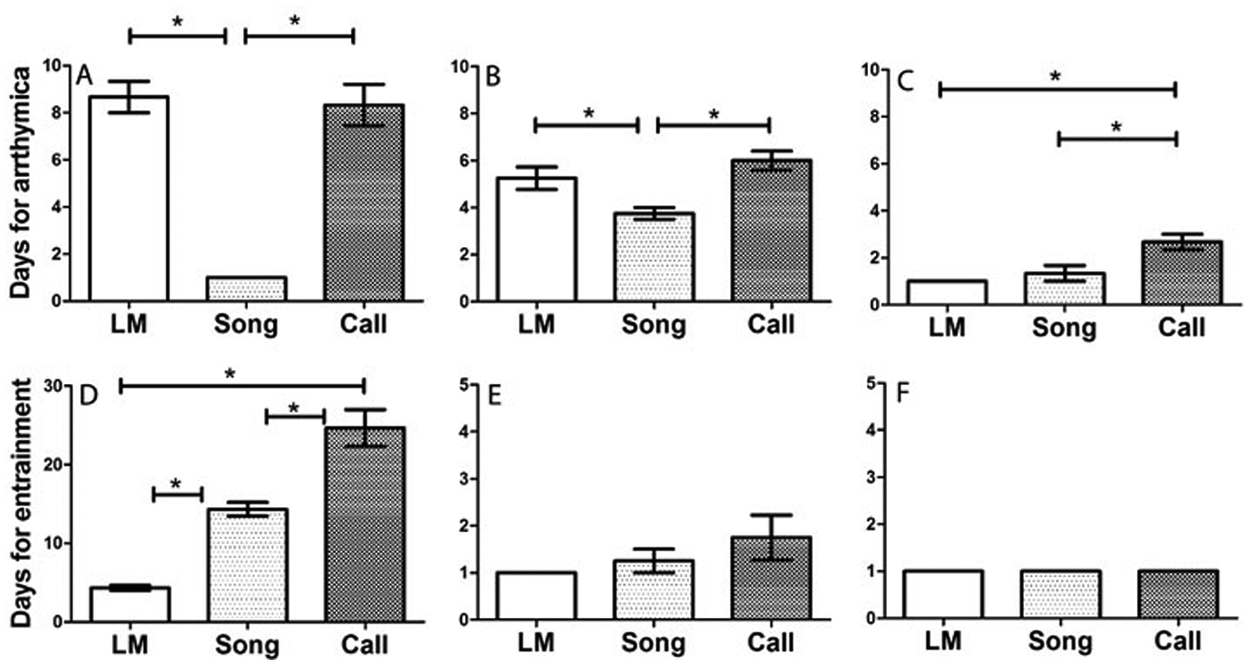
Days for arrhythmicity and melatonin entrainment of locomotor activity, song, and call under different light conditions. (A-C) Rates for arrhythmicity after the exposure from LD to DmDm in PINX birds (A) and from LD to LL in both SHAM (B) and PINX (C) birds. (D-F) Rates for entrainment to the rhythmic melatonin cycles in DmDm in PINX birds (D) and in LL in both SHAM (E) and PINX (F) birds. Data presented as mean ± SEM. *p < 0.05. Abbreviations: LM = locomotor activity; PINX = pinealectomized; SHAM = sham-operated.
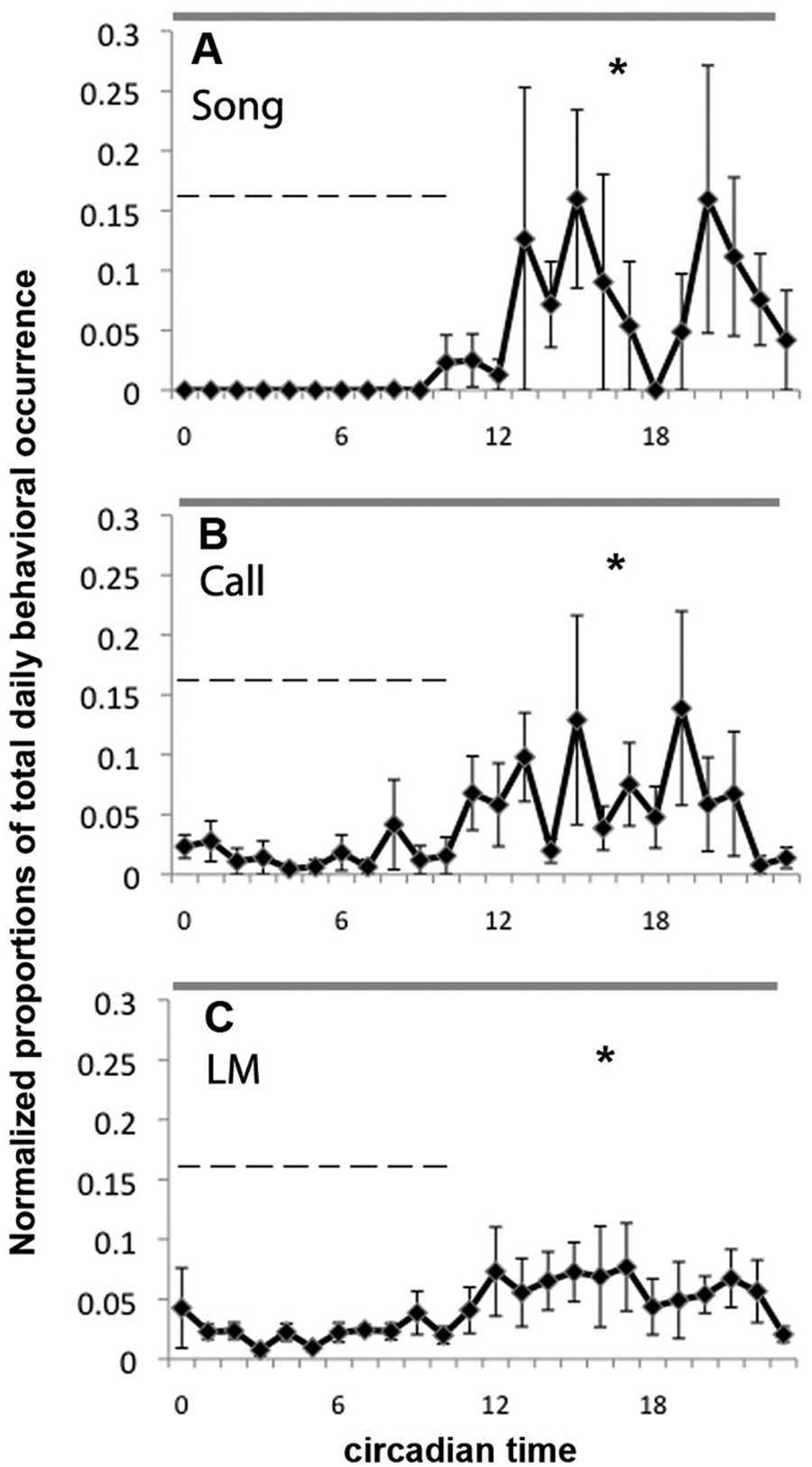
Daily patterns of hourly behavioral occurrence in song (A), call (B), and locomotor activity (C) after the removal of rhythmic melatonin administration in PINX birds. Dashed horizontal lines indicate the subjective melatonin administration period. Vertical coordinates are measured as proportions of total daily behavioral occurrence normalized for each bird. The gray bar on the top of each figure indicates DmDm conditions. Abbreviation: LM = locomotor activity.
LL Conditions
All birds from DmDm experiments were returned to the original 12L 12Dim conditions for 10 days. Rhythmic patterns of locomotor activity, song, and call re-entrained to the 12L 12Dm at the same rates within 1 day of exposure (Figure 2). Then, all birds were placed in LL. Both SHAM and PINX birds gradually became arrhythmic in their locomotor activity, song, and call (Figure 1, G-J; Figure 2). PINX birds became arrhythmic earlier than SHAM birds in their locomotor activity (t = 8.24; p < 0.001), singing (t = 4.69; p < 0.001), and calling (t = 6.47; p < 0.001). In SHAM birds, singing became arrhythmic earlier (within 4 days) than did the locomotor activity (within 6 days) (p < 0.05) and call (within 7 days) (p < 0.05), and there was no difference between the locomotor activity and call (p > 0.05) (Figure 4B). In PINX birds, calling (within 3 days) became arrhythmic later than either the locomotor activity (within the first day) (p < 0.05; n = 3) or song (within 3 days) (p < 0.05), and there was no difference between the locomotor activity and song (p > 0.05) (Figure 4C). Compared to 12L 12Dim conditions, there was no change on the daily total amount of locomotor activity, song and call (Supplementary Figure S2).
After 9 days in LL, birds received rhythmic melatonin administration (10 h of melatonin per 24 h) as in the DmDm experiment. In both SHAM and PINX birds, locomotor activity, song, and call entrained to the rhythmic melatonin treatment within 2 days (Figure 1K and 1L; Figure 4E and 4F). There was no difference in the rate of entrainment either between SHAM and PINX birds or among 3 behaviors in both groups. Melatonin administration did not affect the daily total amount of locomotor activity, song, and call under LL (Supplementary Figure S2).
Discussion
Avian vocalization can be divided into 2 major categories: song and call. Songs are typically multisyllabic, multipart sounds, usually produced only by adult males, whereas calls are usually short, often monosyllabic sounds produced by all ages and both sexes. In nature, song is typically used in territorial and reproductive contexts, while calls are used for general communication such as food begging, alarm, presence of food, and contact (Vicario, 2004). In captive zebra finches, adult male birds not only sing “directed song” to court females but also produce “undirected song,” which is not necessarily addressed to con-specifics (Zann, 1996). Although undirected song and calls are acoustic signals for social communication, both are produced in captivity and even in complete social isolation (Rashotte et al., 2001; Johnson and Rashotte, 2002). The persistence of undirected singing and calling in socially isolated birds leads to the question whether these behaviors reflect a motivational state for the song control system, which may be regulated through a nonsocial pathway. Diurnal rhythms of vocalization have been observed in the wild (Ratcliffe and Otter, 1996; Avey et al., 2008) and in captivity (Ollason and Slater, 1973; Johnson et al., 2002), but these have not been attributed to clock-controlled behaviors. The present study establishes that singing and calling behaviors are indeed controlled by a circadian clock and that pineal melatonin is a critical feature of this regulation.
Circadian Clock Regulation of Song Control System
The central role of the pineal gland and its hormone melatonin in avian circadian pacemaking systems is well documented (Zimmerman and Menaker, 1979; Binkley, 1988; Gwinner, 1989; Takahashi et al., 1989; Cassone, 1990a, 1990b). Our study provides evidence for circadian control of overt rhythms in nonlocomotor behaviors in addition to locomotion and links the circadian clock system to the vocal control system in the zebra finch. Although the role of pineal melatonin in avian circadian rhythms of locomotor activity is indisputable (Binkley et al., 1971; Chabot and Menaker, 1992; Cassone and Menaker, 1984; Lu and Cassone, 1993; Hau and Gwinner, 1994; Murakami et al., 2001), the mechanisms underlying this neurohormonal regulation are poorly understood. The limited knowledge of these molecular mechanisms is due in large part to the fact that the locomotor pathways of the avian central nervous system are relatively unknown (Medina and Reiner, 2000), although somatomotor structures have been identified surrounding song system structures (Feenders et al., 2008; Horita et al., 2010).
In contrast to locomotor activity control, the avian song control system has been studied extensively (Nottebohm et al., 1976; Konishi, 1989; Scharff and Nottebohm, 1991; Vicario, 2004). The song control system of oscine birds is a specialized network of brain nuclei involved in calling, singing, and song learning. This system receives auditory input from ascending, primary auditory pathways, which project to higher order auditory structures in the midbrain, thalamus, and forebrain. Song processing occurs in secondary telencephalic auditory areas, which interact with the anterior forebrain pathway for song plasticity and learning. This pathway includes the HVC in the dorsal forebrain, which projects to Area X, whose projections form a loop between the dorsolateral thalamus (DLM) and the lateral magnocellular nucleus of the anterior nidopallium (LMAN). Then, both HVC and LMAN project to the robust nucleus of the archipallium (RA), which forms the song motor output pathway.
In addition to extensive distributions in the visual system of birds (Cassone et al., 1995; Reppert et al. 1995), high-affinity 2-iodo[125I]melatonin (IMEL) binding and Mel1B melatonin receptor mRNA have been observed in many nuclei of the song control system of oscine birds, including HVC, RA, LMAN, and area X (Whitfield-Rucker and Cassone, 1996; Gahr and Kosar, 1996; Bentley and Ball, 2000; Jansen et al., 2005), and Mel1C melatonin receptor mRNA has been observed in the nucleus of the tracheosyringeal nerve (nXIIts) (Jansen et al., 2005). Further, in house sparrows, we have shown that IMEL binding is regulated by photoperiod (Whitfield-Rucker and Cassone, 1996) and that manipulation of the duration of melatonin titers in sparrows’ blood regulates HVC and RA volume (Cassone et al., 2008). Based upon these observations and the present study, pineal melatonin likely controls rhythmic vocal behavior by directly acting on the song control system. In addition, the responses of singing and calling to rhythmic melatonin administration appear to be entrainment of rhythmic vocal behavior rather than simple inhibition of the behavior, because the different behaviors gradually entrain to the melatonin signal at different rates. This suggests that melatonin controls rhythmic vocal production through a clock system, probably located in the song control system itself.
Circadian Clock versus Sleep in Regulating Song Behavior
Melatonin has also been associated with induction and consolidation of sleep (cf. Ferguson et al., 2010; van Geijlswijk et al., 2010), and since melatonin clearly affects locomotor behavior by causing a quiescent phase in birds (Figure 2), one might conjecture that the effects we see here are due to pineal melatonin’s role in sleep regulation. Song development certainly involves sleep-dependent daily oscillations within song structures with pronounced after-sleep deterioration and daytime recovery, which has been proposed to create opportunities to reshape previously learned motor skills (Derégnaucourt et al., 2005; Shank and Margoliash, 2009). HVC neural sleep activity has also been implicated in song stabilization in adult zebra finches and European starlings (Nick and Konishi, 2001, 2005; Crandall et al., 2007; Brawn et al., 2010).
There are at least 2 interrelated processes associated with these processes: sleep regulation and clock regulation. Most studies favor a predominant role for sleep regulation of song, relegating the circadian clock to a secondary process (Derégnaucourt et al., 2005; Brawn et al., 2010; Rauske et al., 2003). However, these studies did not systematically examine the effects of the circadian clock system on the temporal organization of song or of the learning process, limiting circadian investigation to either day-time singing or night-time sleep.
In contrast to studies favoring homeostatic sleep regulation, the present study points to the direct clock regulation of song behavior. In DmDm conditions, SHAM birds showed the circadian free running rhythms of song production as well as “sleep” suggested by the quiescent phase of locomotion. PINX birds showed arrhythmicity in song behavior and persistent sleeplessness. Rhythmic melatonin administration restored the rhythm of song production but not necessarily the regular sleep phase, although the amount of locomotor activity decreased during the subjective night. The fact that damped rhythms of song, call, and locomotor behavior persist for several cycles following removal of melatonin (Figure 5) punctuates the point that these behaviors are entrained. If they are entrained, this aspect of their responses to melatonin is not consistent with the homeostatic aspects of sleep, although we cannot rule out any role for the circadian component of sleep in these responses to melatonin. Similar results were attained in both SHAM and PINX birds in the LL conditions. Our results suggest that song behavior is directly under the regulation of the circadian clock system and melatonin, which also regulates sleep in parallel.
Differential Circadian Regulation of Locomotor Activity, Song, and Call by Pineal Melatonin
Based on previous work (Cassone and Menaker, 1984; Gwinner, 1989; Cassone et al., 2009) and the present study, we propose a schematic avian model for the regulation of locomotor activity, song, and call by the circadian clock system (Figure 6). This model is composed of 2 major parts: the central clock system, itself a multioscillatory system, and downstream overt behavioral rhythms. The central clock system includes major photoreceptive tissues (retina and pineal), encephalic photoreceptors, which collectively mediate the light signal input, and pacemakers (retina, pineal and SCN), all of which interact neuronally and hormonally to maintain the self-sustaining time-keeping system. Although the molecular mechanisms are not as well-established in birds as they are in either mammals or insects (Bell-Pedersen et al., 2005), orthologues of the molecular components in the biological clock (clock genes) are present and rhythmically expressed in avian pacemaker tissues (Bailey et al., 2003, 2004; Karaganis et al., 2008), brain structures, and peripheral tissues (Karaganis et al., 2009). It is therefore reasonable to propose that similar mechanisms are present in birds.
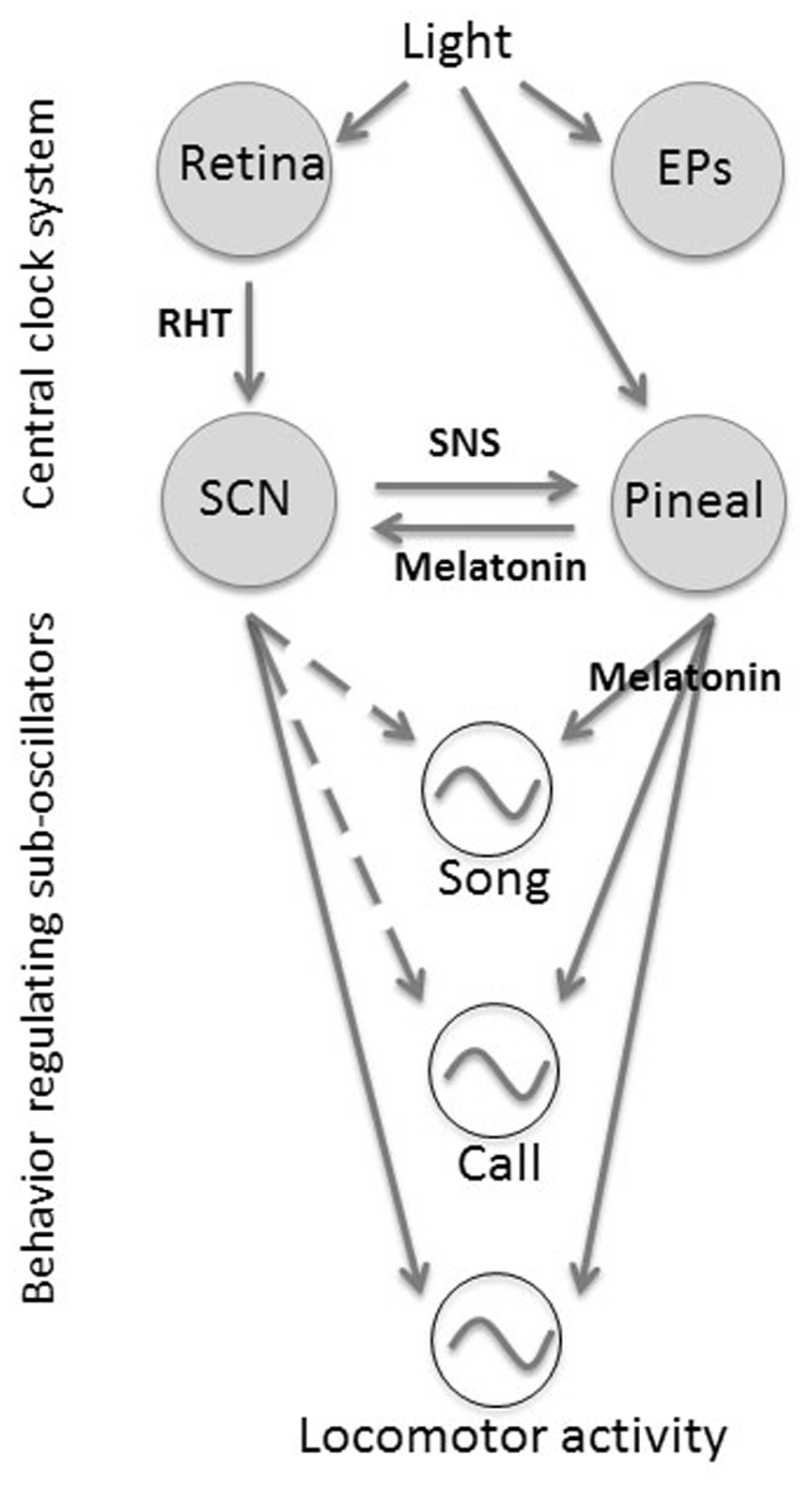
Schematic model of the regulation of the overt circadian rhythms of locomotor activity, song, and call by the circadian clock system. In the model, the light information can be perceived by the central clock system through photoreceptors such as the retina, pineal, and EPs. Within the central clock system, the pineal, retina, and SCN interact with one another through either hormonal melatonin or neuronal innervation, although it is still unknown how EPs get involved in the system. The pineal and SCN can regulate the suboscillators through either melatonin or neuronal innervation, and each suboscillator regulates the overt circadian rhythm of certain behavioral output, such as the locomotor activity, song, and call. Solid arrows indicate the signal transduction pathways, which are strongly suggested by previous and current studies. Dashed arrows indicate pathways for which there is currently no evidence. Abbreviations: EPs = encephalic photoreceptors; RHT = retinal hypothalamic tract; SNS = sympathetic neuron system.
In this scenario, suboscillators, under the influence of pacemaker signals through melatonin and possible neuronal inputs, control overt circadian outputs, such as locomotor activity, song, and call, through entrainment of oscillatory mechanisms within the structures themselves. This predicts that song control structures in the brain express clock genes rhythmically and are entrained by pineal melatonin and/or neural input. This view is supported by our preliminary evidence showing the expression of core clock genes in both mRNA and protein level in song control nuclei of song birds (Harpole and Trivedi, unpublished). The strengths of coupling of these song control oscillators with pineal signal could explain the different rates by which they become arrhythmic and entrain to the hormone. Coordination of these complex, rhythmic behaviors is effected by their mutual, albeit differential, sensitivities to melatonin and to established interactions among the structures themselves.
Footnotes
Acknowledgements
We thank Jennifer O’Brien for data collection; Jiffin Paulose, Jaclyn Malloy, and Ye Li for stimulating discussions; and Melissa Whitfield-Rucker for animal facility and administrative support. This work was supported by the University of Kentucky and NIH P01 NS 39546 to VMC.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.