Abstract
Circannual rhythms are innately timed long-term (tau ≈ 12 months) cycles of physiology and behavior, crucial for life in habitats ranging from the equator to the Poles. Here the authors propose that circannual rhythm generation depends on tissue-autonomous, reiterated cycles of cell division, functional differentiation, and cell death. They see the feedback control influencing localized stem cell niches as crucial to this cyclical histogenesis hypothesis. Analogous to multi-oscillator circadian organization, circannual rhythm generation occurs in multiple tissues with hypothalamic and pituitary sites serving as central pacemakers. Signals including day length, nutrition, and social factors can synchronize circannual rhythms through hormonal influences, notably via the thyroid and glucocorticoid axes, which have profound effects on histogenesis. The authors offer 4 arguments in support of this hypothesis: (1) Cyclical histogenesis is a prevalent process in seasonal remodeling of physiology. It operates over long time domains and exhibits tissue autonomy in its regulation. (2) Experiments in which selected peripheral endocrine signals are held constant indicate that circannual rhythms are not primarily the product of interacting hormonal feedback loops. (3) Hormones known to control cell proliferation, differentiation, and organogenesis profoundly affect circannual rhythm expression. (4) The convergence point between photoperiodic input pathways and circannual rhythm expression occurs in histogenic regions of the hypothalamus and pituitary. In this review, the authors discuss how testing this hypothesis will depend on the use of cellular/molecular tools and animal models borrowed from developmental biology and neural stem cell research.
Spring was moving in the air above and in the earth below and around him, penetrating even his dark and lowly little house with its spirit of divine discontent and longing. It was small wonder, then, that he suddenly flung down his brush on the floor, said “Bother!” and “O blow!” and also “Hang spring-cleaning!” and bolted out of the house without even waiting to put on his coat.
In this classic children’s book written by a disenchanted banker, Mole, inherently restless after a winter of inactivity, is triggered into a new way of life by spring cues filtering through the earth above. The combination of an innate sense of seasonal time and a powerful sensitivity to the changing environment is crucial for life on Earth.
At higher latitudes where the seasons are strong and occur predictably, long-term timing processes have evolved to be synchronized by predictable environmental cues, particularly day length (photoperiodic) change. In photoperiodic species, the innate nature of the timing mechanisms is revealed by exposure to constant day length and temperature extended over long periods—analogous to the use of constant light or dark regimens in the study of circadian biology. Under static conditions, complete endogenous long-term cycles of physiology and behavior, alternating between summer and winter phenotypes, may persist for years or even throughout the entire life cycle (Zucker, 2001). The endogenous period (tau) of the circannual oscillation is often close to 10 months and has been well characterized in many long-lived species, including ferret (Herbert et al., 1978), mink (Martinet et al., 1992), sika deer (Goss, 1977, 1984), sheep (Karsch et al., 1989; Lincoln, 2006; Lincoln et al., 2006), various types of ground squirrels (Kenagy, 1980; Licht et al., 1982), chipmunks (Kondo et al., 2006), European hamsters (Monecke et al., 2009), and notably in passerine species of birds (Gwinner, 1986). There is also a very well-characterized insect model (Miyazaki et al., 2005, 2007, 2009). In small rodent species that have shorter life spans (e.g., Syrian and Siberian hamster), exposure to constant photoperiod does not produce free-running circannual rhythms. However, these animals express endogenously regulated reversion to a summer phenotype under constant winter photoperiods (so-called photorefractoriness), and the underlying innate interval timing mechanisms may be qualitatively similar to those in the robustly circannual species (Butler et al., 2010).
The expression of endogenous circannual rhythms is also a feature of life in the equatorial zones, but here their primary role may relate not to anticipating seasonal change but to optimizing individual reproductive success. This can be achieved by breeding synchronously at specific intervals (e.g., 9- to 10-month breeding cycles in sea birds on Ascension Island at the equator that breed at maximum frequency in times of food abundance; Ashmole, 1963) or breeding asynchronously as occurs with male axis deer and African elephants in the tropics, a strategy that reduces competition between rivals (Loudon and Curlewis, 1988; Lincoln et al., 1994; Lincoln and Ratnasooriya, 1996; Poole and Moss, 1981; Poole, 1987). In each case, the generation and timing of the seasonal cycle appear to be intrinsic, and environmental cues (photoperiod, nutrition, social factors) act to entrain the cycle. For a more extensive discussion of circannual rhythms as a normal part of the ontogenetic program, the reader is directed to Lincoln and Hazlerigg (2010).
Hence, while much of our current understanding of circannual rhythms comes from photoperiodic species, there is no absolute link between circannual rhythmicity and photoperiodic responses, and seasonally photoperiodic species can be seen as one major subset of a large group of animals showing cyclical life history characteristics (Lincoln and Hazlerigg, 2010). We argue for the existence of autonomous circannual oscillators that drive seasonal physiological responses. The fundamental issue is to identify the inherent mechanisms that generate the very long time constants of circannual rhythms and to characterize the level of cell and tissue organization. Do circannual rhythms emerge (1) cell-autonomously, (2) through tissue properties of histogenesis, or (3) as a property of multiple interacting physiological systems in the organism at large? In the study of circadian rhythms, this problem is now largely resolved. Circadian clocks are cell autonomous, based on the interaction between about a dozen core clock genes where the component time delays depend largely on the latency in the negative feedback mechanisms that govern gene transcription. The cellular circadian rhythms are in turn synchronized to produce tissue-autonomous circadian cycles and hierarchically coordinated to produce the organism’s circadian timing system (Reppert and Weaver, 2002). What are the equivalent elements for the circannual system?
The Cyclical Histogenesis Hypothesis
We suggest that circannual rhythms emerge from the process of tissue-autonomous, cyclical regeneration that is part of a cyclical life history, governed by the ontogenetic program for each organism. The long time domains are due to the protracted processes of cell differentiation, tissue remodeling, and feedback signaling from within and outside the tissue. These cyclical regeneration processes are seen to occur in multiple seasonally regulated tissues in the brain, pituitary gland, and peripheral tissues with variable phasing to produce each circannually rhythmic phenotype.
In making this proposal, we are mindful of the biology of the mammalian hair follicle (Schneider et al., 2009; Figure 1A). This acts as a semi-autonomous mini-organ, capable of iterated cycles of growth and regeneration, with timescales of months to years. Hair follicle cycles persist throughout the lives of organisms and may run independently (asynchronously) from neighboring hair follicles, demonstrating autonomy. Typically, however, the behavior of hair follicles in a region of skin is coordinated through locally generated morphogenic signals as a local wave of reactivation (Plikus et al., 2008) or as a seasonally synchronous whole-body event activated hormonally, as is the case for the seasonal molt cycle triggered by prolactin or sex steroid hormones (Duncan and Goldman, 1984; Figure 1B).
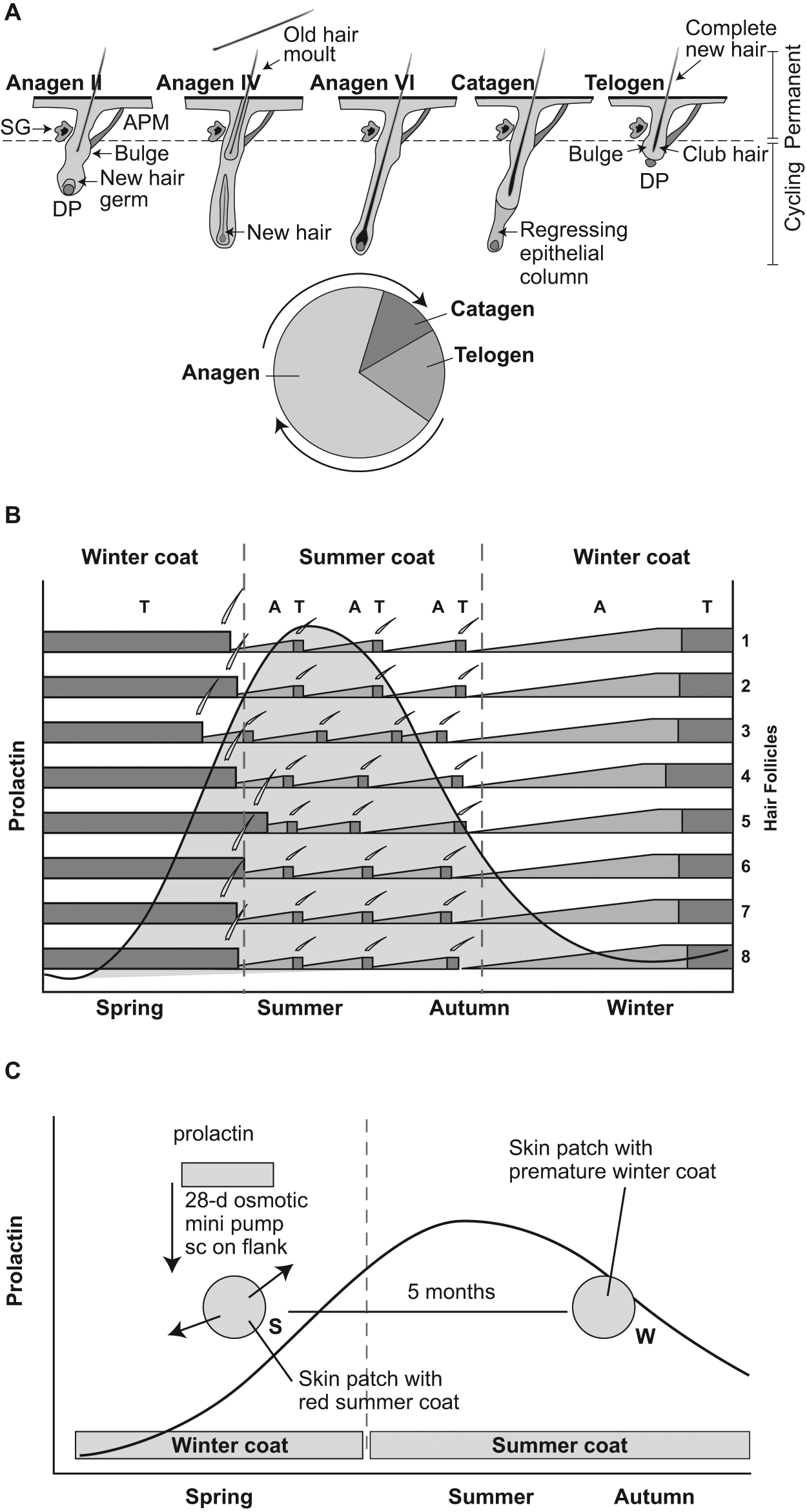
The hair follicle as a tissue-autonomous rhythm generator based on cyclical histogenesis. (A) The growth phase of the follicular cycle (anagen) is initiated by stem cell activation in the bulge region at the base of the hair due to changes in the microenvironment of the bulge niche (arrow; Schneider et al., 2009). Once the growth of the new hair is complete, the germinal epithelial cells at the base of the hair shaft regress, isolating the dermal papilla (DP) to produce the mature phase (catagen) and then the quiescent phase (telogen). The total period of the hair follicle cycle is 2 months to <2 years, and the relative time in anagen, catagen, and telogen varies markedly between species and between skin regions and seasons within species. SG = sebaceous gland; APM = arrector pili muscle. (B) Model for seasonal changes in the hair follicle cycle in sheep or red deer. Hair follicles (illustrated as horizontal bars 1-8) show limited local synchronization and cycle repeatedly during the summer; high prolactin levels are permissive to this state. Declining prolactin in the autumn synchronizes follicles throughout the pelage and leads to an extended anagen (A), producing a long winter coat. Follicles then remain quiescent in telogen (T) during the winter. Synchronous reactivation of the follicles produces the conspicuous spring body molt. The overall circannual period is a composite of multiple histogenic cycles in the hair follicle. (C) Experiment in which female red deer were given a small osmotic mini-pump implanted under the skin of the flank delivering prolactin for 28 days. Treatment in spring induced the local activation of anagen and the development of a skin patch with red summer coat (S), contrasting with the longer gray winter coat on the rest of the body. This skin patch developed a winter coat (W) some 5 months later, long after the end of prolactin treatment, demonstrating tissue-autonomous control of the cycling between the two pelage phenotypes (after Loudon and Jabbour, 1994).
The intrinsic cellular and molecular control of the hair follicle cycle is well understood based on transgenic mouse models (Schneider et al., 2009). The cycle is initiated by stem/progenitor cell proliferation in the bulge region of the follicle (Figure 1A). Progeny cells migrate to the dermal papilla (DP) and activate regrowth of a complete new hair fiber (anagen phase) and casting of the old. During the cycle, paracrine signals are generated that affect adjacent follicles to propagate a regenerative wave (Plikus et al., 2008). The wnt/β-catenin signaling pathway, coupled with low levels of bone morphogenic proteins (BMPs), promotes anagen, while rising BMP levels cause growth arrest (catagen) and transition to the quiescent telogen phase. Return to anagen requires diminution of BMP levels, and so the hair follicle cycle can be thought of as a limit cycle oscillation between the opposing forces of wnt/β-catenin and BMP signaling (Plikus et al., 2008).
These locally acting feedback signals both dictate the periodicity of the individual follicular cycle and allow for local coordination of waves of hair growth within skin regions (Plikus et al., 2008).
In seasonal species, the hair follicle can produce more than one hair phenotype (e.g., summer or winter fibers) with different hair characteristics and cycle periodicities (Figure 1B). Short cycles typically produce short summer hair fibers and are grown asynchronous in the skin, while long cycles produce long, stable winter coats, when all follicles enter an extended period of telogen lasting many months over winter. The annual pelage cycle is thus the composite of more than one tissue cycle alternating between summer and winter programs. This cyclic phenomenon has been investigated in red deer by short-term administration of prolactin into the skin using osmotic mini-pumps to locally alter the hair follicle cycle (Loudon and Jabbour, 1994; Figure 1C). This treatment induced localized, premature regrowth of a summer coat in winter animals around the implant. Remarkably, the affected skin patch then remained out of synchrony with the rest of the body’s molt pattern for at least 5 months and in late summer prematurely developed a gray winter phenotype on an animal with a red summer coat. This indicates that skin tissue has intrinsic control of both the hair follicle cycle and the long-term cycle of switching between summer and winter phenotypes (a circannual clock). While prolactin secretion normally acts as the systemic signal that coordinates pelage changes to the seasons, local mechanisms in the skin generate cycles over long time intervals (Figure 1B,C).
Generalizing from the hair follicle, we conceptualize circannual rhythm generation as a phenomenon involving cyclic tissue growth and remodeling (Figure 2). The prediction based on this example is that some of the genes that regulate the cycle in the adult will be same genes that regulate the initial development of the tissue in the embryo. Thus, events of the circannual cycle recapitulate ontogeny throughout life, producing a cyclical life history. We regard this as the norm for vertebrates (Lincoln and Hazlerigg, 2010).
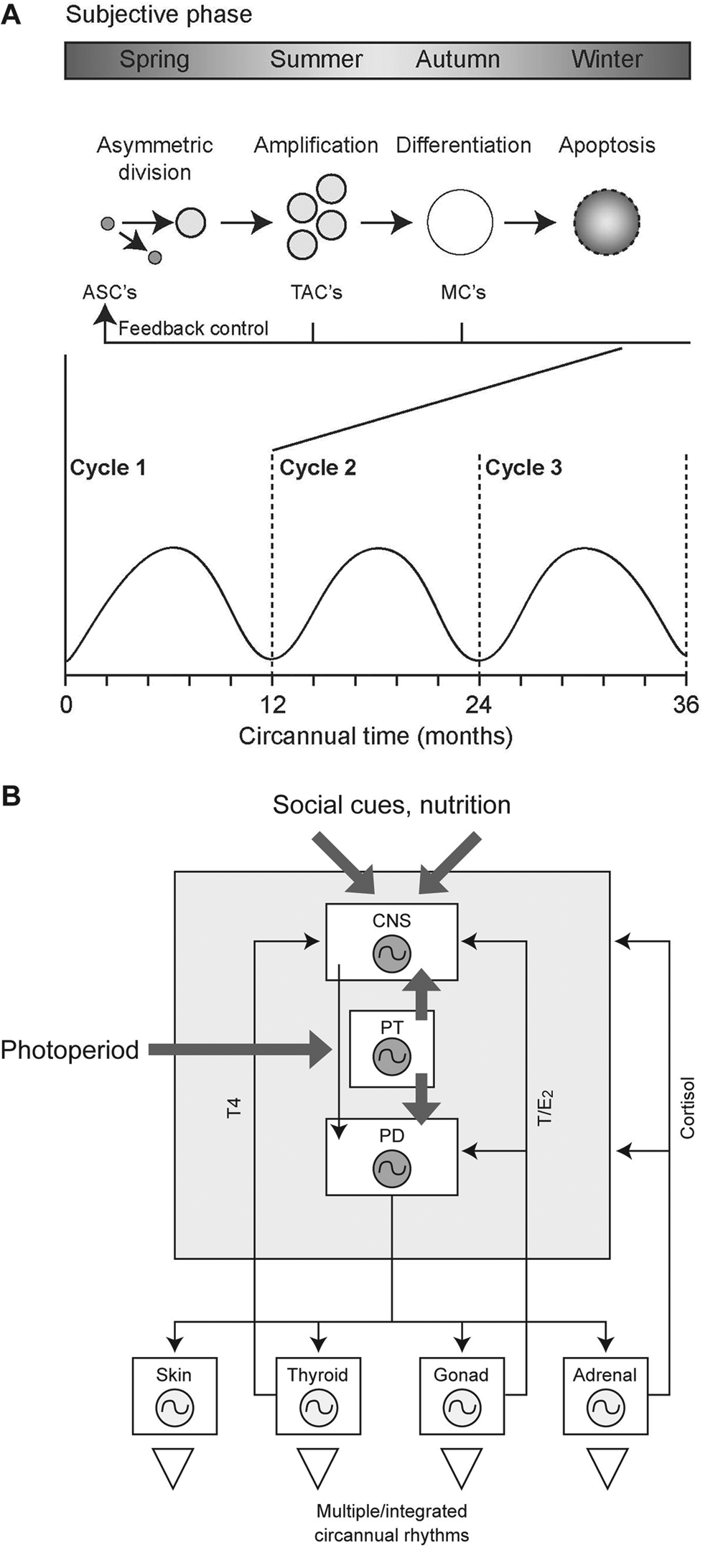
The cyclical histogenesis hypothesis. (A) Generic model for a circannual rhythm generation by cyclical histogenesis. We consider that circannual cycles oscillate between phases of subjective summer (open), with growth and maturation of tissues by histogenesis, and subjective winter (shaded), during which tissues become quiescent. This is driven by pluripotent stem cells located in tissue-specific stem cell niches. Stem cells undergo asymmetric division as a rare event, giving rise to populations of transit amplifying cells (TACs), which undergo repeated rounds of cell division to form a new cell population. Migration and differentiation of these cells then lead to the formation of new functional tissue, as well as providing feedback signals that arrest cell division in the stem cell niche. Withdrawal of negative feedback possibly related to apoptosis initiates a new cycle completion of these multiple phases and gives rise to one circannual cycle (although subcycles are considered possible). ASC = adult stem cell; MC = mature cell. (B) Organism-level circannual coordination as a product of circannual histogenic cycles in multiple tissues. The analogy to circadian organization is drawn (Herzog and Tosini, 2001), and multiple coordinating signals between pacemaker structures in the hypothalamus and pituitary, including thyroxine (T4), glucocorticoid (cortisol) and testosterone (T) / estradiol (E2) feedbacks, are envisaged. Circannual rhythms may be entrained by photoperiod acting at the level of the pars tuberalis (PT) and/or by other cues (social, nutrition etc.) acting via the central nervous system (CNS). PD = pars distalis; circle/oscillator symbol, histogenic cycle.
Within each tissue, we envisage a mechanism in which progenitor cells (adult stem cells, ASCs) act synchronously to initiate phases of cell division (Figure 2A). The resultant progeny (transient amplifying cells, TACs) undergo further proliferation, migrate, and differentiate, yielding mature cells (MCs) that drive seasonal transitions in physiology. Feedback signals generated during the processes of proliferation, differentiation, and/or subsequent cell death are presumed to control phases of progenitor cell activity and quiescence (respectively “subjective summer” and “subjective winter”) and hence the period of the circannual rhythm (tau).
This autonomous cyclical regenerative process is likely to occur in multiple tissues to affect different physiological systems, while overall circannual pacemaker control may depend on dominant signals emanating from the hypothalamus and pituitary gland (Figure 2B). Hence circannual organization at the organismal level is seen as a “circannual clock shop” with a hierarchical organization (Lincoln and Hazlerigg, 2010). This appears similar to the CNS-peripheral oscillator organization of the circadian system in vertebrates (Herzog and Tosini, 2001). However, while circadian timing is clock gene and cell based, we propose that circannual timing is tissue based, as recognized in developmental biology—thus the analogy is limited. Within this framework, we envisage that the local environments surrounding adult stem cells (so-called stem cell niches) are crucial for circannual rhythm generation and that entrainment of circannual rhythms by photoperiod or other inputs operates through the CNS via hypothalamic or pituitary stem cell niches (Figure 1B).
In this review, we focus on two potential stem cell niches as being of key importance: (1) the subventricular zone surrounding the walls of the third ventricle in the basal hypothalamus (SVZ-3v) and (2) the cleft region in the anterior pituitary (marginal zone, MZ; Figure 3). The detailed cellular architecture in these two proliferative zones has been investigated with the aim of identifying the candidate ASCs and TACs (Figure 3B,C). The function of both these niches and evidence linking them to circannual rhythm generation are discussed further below.
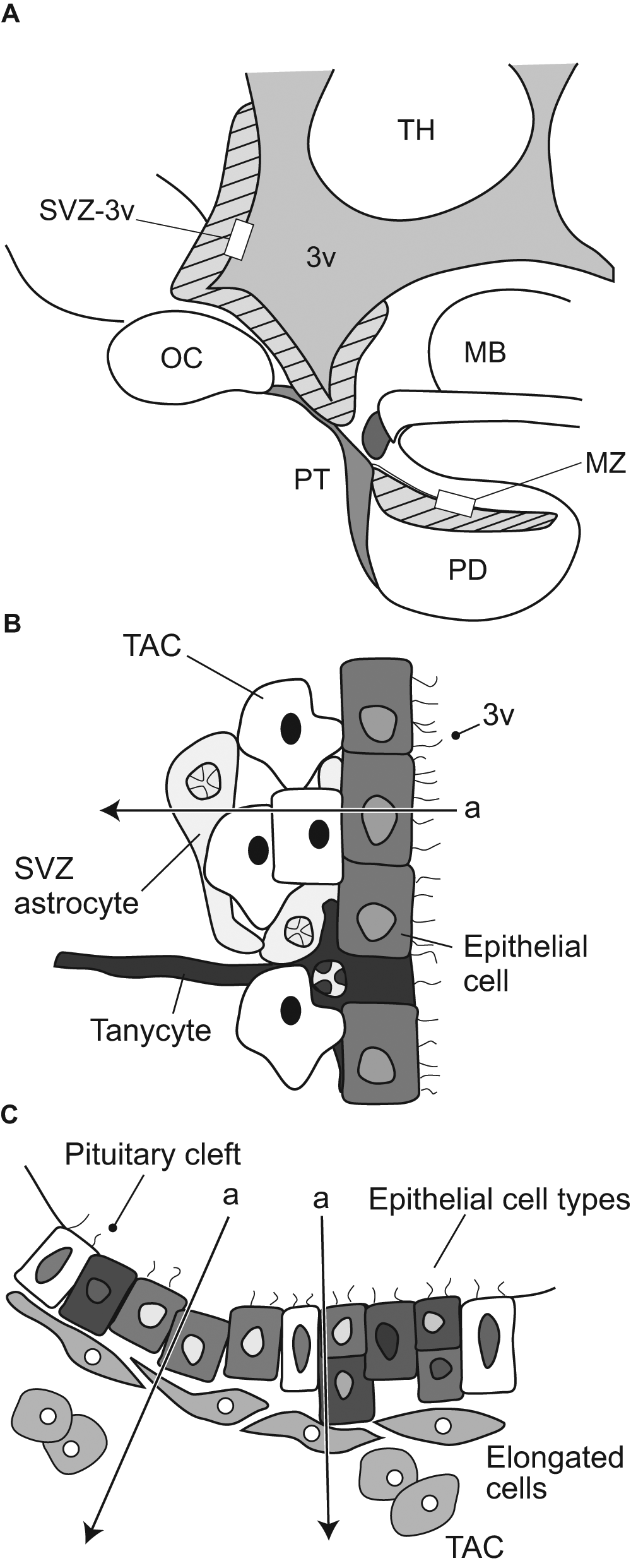
Putative circannual pacemaking stem cell niches in the hypothalamus and pituitary gland. (A) Schematic sagittal view of the sheep hypothalamus and pituitary showing stem cell niches (hatched shading) that potentially serve as pacemakers: the subventricular zone surrounding the basal third ventricle (SVZ-3v) and the marginal zone (MZ) of the pituitary cleft. PT = pars tuberalis; PD = pars distalis; MB = mammillary body; OC = optic chiasm; TH = thyroid hormone. (B) SVZ-3v niche showing cell types immediately adjacent to the third cerebral ventricle (3v). Astrocytes or tanycytes penetrating into the ependymal cell layer of the ventricular wall are potentially neural stem cells, giving rise to neuroblasts and thence transit amplifying cells (TACs), which can then populate neighboring hypothalamic nuclei controlling metabolic and reproductive function (after Chojnacki et al., 2009). (C) Corresponding view of the MZ of the pituitary cleft where some epithelial cells lining the cleft are potential pluripotent stem cells that give rise to TACs that differentiate into new pituitary cells of different cell lineages, and the adjacent elongated cells form part of the pituitary stem cell niche by influencing proliferation of the stem cells (Vankelecom, 2009). Note that both of the histogenic tissues have a polarity (arrow a), which may orientate cell migration.
Supporting Evidence
1. Cyclical histogenesis is a prevalent process in seasonal remodeling of physiology and exhibits tissue autonomy in its regulation
Although the cyclic tissue regeneration concept owes much to the hair follicle, seasonal remodeling of tissues through histogenesis is widely observed in vertebrates and in structures, including the testis, antlers, feathers, neural centers concerned with song learning, and the gut. Additionally, the liver, which is a highly regenerative organ (Forbes et al., 2002), has been shown to be a key component of the circannual rhythm generator controlling hibernation in chipmunks (Kondo et al., 2006).
For our hypothesis, the key feature is not cell proliferation/regeneration per se but rather the degree to which proliferation is synchronized within an organ or physiological system to generate an overt long-term cycle. Direct evidence for this in contexts other than the hair follicle is weaker but includes the finding that limb regeneration in newts shows a marked seasonal variation, which is partly due to intrinsic seasonal variation in the sensitivity of the wound site to neurotrophic signals (Singer, 1974). A similar situation is also found in the antler, where the wound-healing response that initiates antler regeneration can only be triggered during the “summer phase” of the antler cycle (Lincoln and Fletcher, 1976). Intrinsic seasonality in antler tissue is likely in deer where the antlers are regenerated circannually under free-running conditions and periodic antler growth persists in castrated animals where the dominant effect of sex hormones is removed (Goss, 1983). The feather molt in migratory birds has also been shown to be cyclical under constant conditions with a different circannual temporal pattern for wing primaries compared with the body feathers (Piersma et al., 2008), also consistent with local tissue regulation of histogenesis.
Aside from these seasonal examples, the most compelling case of long-term cycles with stem cell origins comes from a family of hematological diseases (leukemias) in which long-term cyclical variations in the titers of different blood cell types are observed (Haurie et al., 1998). Periodic oscillations in numbers of granulocytes with period lengths of up to 100 days have been recorded in patients with periodic myelogenous leukemia and of pancytopenia (in which titers of all blood cell types are affected). Importantly for our hypothesis, these diseases originate from abnormal bone stem cell function linked to specific gene defects. Experimental and modeling evidence indicates that the period length of these oscillations depends on the properties of bone marrow stem cells and their progeny and local signaling between them, rather than feedback cycles involving systemic signals (Haurie et al., 1998).
Extrapolating from these examples, we suggest that long-term cyclic regeneration is an intrinsic property of many tissues and is coordinated by pacemaker processes in the basal hypothalamus and pituitary gland (for which the evidence for histogenesis-based remodeling is discussed in detail later) to govern the seasonal transitions in homeostasis. This “circannual clock shop” then produces the profound difference in the functional state of the whole body between summer and winter.
2. Experiments in which peripheral endocrine signals are held constant indicate that circannual rhythms are not merely the product of interacting hormonal feedback loops
The cyclical histogenesis conjecture proposes that delayed feedback within tissues, particularly in histogenic regions of the hypothalamus or pituitary, is the key to the long time constants of circannual rhythm generation. Modeling circannual rhythm generation illustrates the importance of a delayed feedback element (McGreggor and Lincoln, 2006).
Several lines of evidence suggest that more generalized systemic feedback control may not be necessary for circannual rhythm generation. For example, sheep pinealectomized to remove melatonin as a mediator of photoperiodic entrainment, castrated and given constant-release implants of estradiol, still express circannual rhythms of gonadotrophin secretion (Woodfill et al., 1994). These rhythms can be synchronized by seasonally appropriate programmed daily infusions of melatonin but continue asynchronously in the absence of melatonin. Gonadectomized ground squirrels also express robust circannual rhythms, whose characteristics are modulated by gonadal steroid implants (Hiebert et al., 1998). In circannual rodent species, there is also evidence that ablating major endocrine feedback pathways by thyroidectomy or castration fails to interfere with innate rhythms in hibernation, feed intake, and fat accumulation (Henderson and Demeneix, 1981). Moreover, in hypothalamo-pituitary disconnected (HPD) Soay rams, the surgical destruction of the descending hypothalamic control of the pituitary gland that blocks most seasonal rhythms (gonadal activity, food intake, body weight) fails to abolish circannual prolactin secretion (Lincoln and Clarke, 1994; Lincoln et al., 2006). Pharmacological suppression of prolactin secretion in this model does not affect the phase of the circannual prolactin cycle (Lincoln et al., 2003), indicating that circannual rhythm generation is tissue localizable and independent of the output response.
3. Hormones known to control cell proliferation, differentiation, and organogenesis profoundly affect circannual rhythm expression
Thyroid hormone
Thyroid hormone (TH) is well known for its effects on cell division, proliferation, and organogenesis and most strikingly for its role in regulating metamorphosis in amphibians (Brown, 2005). Here, rising levels of triiodothyronine (T3) are critical for major events at the end of the tadpole stage, including activation of limb buds, tail absorption, and intestinal remodeling (Schreiber et al., 2004). These actions of T3 are controlled locally within each specific tissue by enzymatic conversion of circulating thyroxine (T4) to the more active T3 form; the enzyme type 2 deiodinase (Dio2) activates, while type 3 deiodinase inactivates the system. During the transition from tadpole to adult, precisely timed changes in the relative levels of Dio2 and Dio3 expression determine local T3 bioavailability, and this controls the cycle of histogenesis associated with juvenile development (Brown, 2005).
It has been known since the 1930s that thyroidectomy blocks seasonal breeding cycles in bird species, and equivalent effects were subsequently described in mammals (Follett and Potts, 1990; Moenter et al., 1991; Webster et al., 1991). In sheep, these effects represent a temporally specific and anatomically localized action of TH on the generation of the circannual reproductive rhythm. Thyroidectomy (TX) blocks the transition of ewes to the sexually inactive state of anoestrus normally seen in summer but fails to block the transition to breeding condition associated with autumn (Figure 4). Detailed studies using T4 replacement in TX ewes have revealed a window of sensitivity to T4, which opens in the spring and closes some 6 months later, as part of the circannual cycle (Thrun et al., 1997; Billings et al., 2002). Strikingly, micro-implants of T4 placed in the histogenic regions of the mediobasal hypothalamus, particularly in the premammillary region, are sufficient to mimic the effects of systemic T4 replacement (Anderson et al., 2003; Figure 4B). Furthermore, Barrett and colleagues (2007) have demonstrated in Siberian hamsters that gonadal inhibition induced by short days is prevented by a T3 micro-implant placed in the third cerebral ventricle (3V), similar to effects produced by peripheral T3 treatments (Freeman et al., 2007). These data nicely parallel the findings in quail that central T3 treatments promote a summer breeding phenotype.
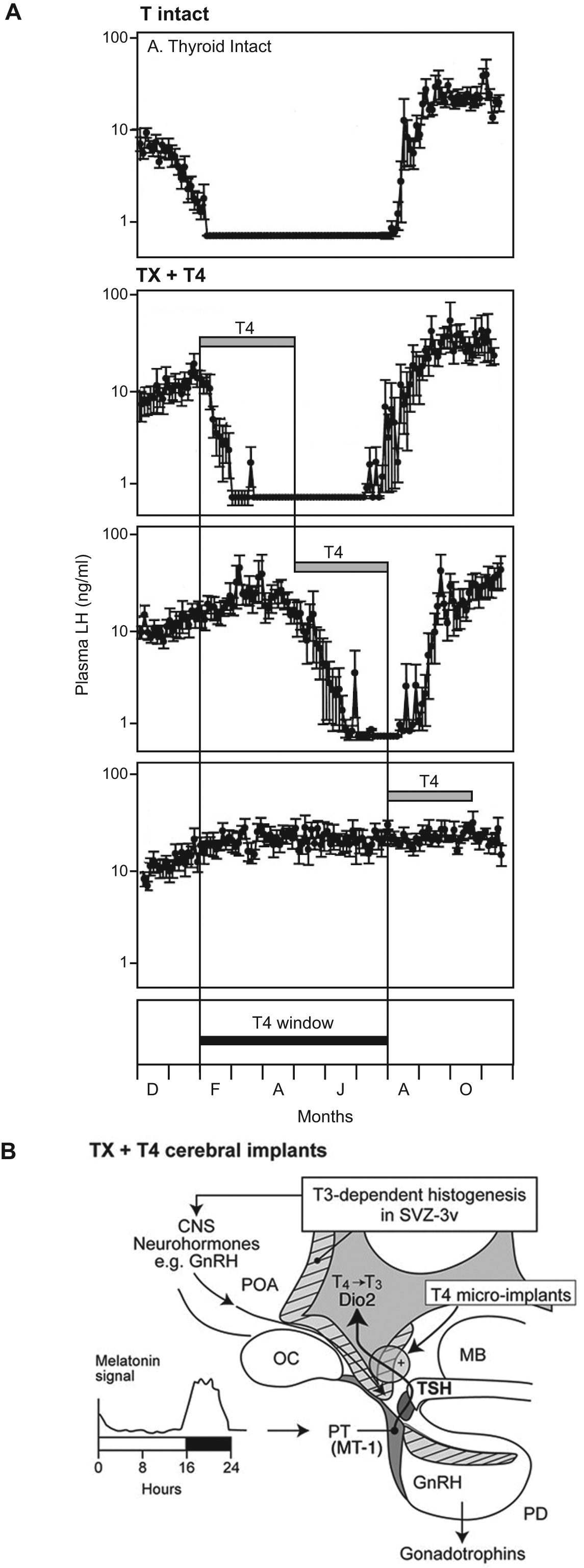
Thyroid hormone role in the expression of circannual gonadotrophin rhythms in female sheep. (A, top panel) Thyroid intact ewes: The normal seasonal profile of luteinizing hormone (LH) secretion comprises a phase of elevated concentrations in the breeding season interrupted by a phase of low levels in the nonbreeding season (anoestrous) (NB data are plotted on a log scale and levels during the nonbreeding season are below the sensitivity limit of the assay). (A, lower panels) Thyroidectomized (TX) ewes: Thyroxine (T4) replacement permits the seasonal shutdown of LH secretion (and anoestrous) when T4 is given in the spring to mid-summer months (middle 2 panels) but not when given later (bottom panel). Hence, a sensitive spring-summer window for T4 actions is defined (solid horizontal bar below; data redrawn from Billings et al., 2002). (B) TX ewes given cerebral micro-implants: T4 micro-implants placed in the premammillary hypothalamus (Circle+, solid arrow) and to a lesser extent the ventral preoptic hypothalamus permit seasonal anoestrous (data from Anderson et al., 2003). B also shows the proposed pathway by which the melatonin signal encoding photoperiod controls the spring-summer window via PT TSH effects on deiodinase 2 (Dio2) gene expression. This is thought to promote T3-dependant histogenesis in the SVZ-3v (putative circannual pacemaker) and ultimately to regulate the activity of the hypothalamic GnRH pulse generator responsible for reproductive control. Based on data from Hanon et al., 2008. CNS = central nervous system; GnRH = gonadotrophin-releasing hormone; MB = mammillary body; MT-1 = melatonin type 1 receptor; OC = optic chiasm; PD = pars distalis; PT = pars tuberalis; POA = preoptic area; SVZ-3v = subventricular zone of the third cerebral ventricle; TSH = thyroid-stimulating hormone; T3 = tri-iodothyronine; T4 = thyroxine.
Most recently, an endogenous control pathway regulating seasonal transitions, analogous to metamorphic regulation in amphibians, has been characterized in the hypothalamus of mammals and birds (Hanon et al., 2008; Nakao et al., 2008; Ono et al., 2008). This depends on the regulation of Dio2 and Dio3 expression in the tanycytes in the ependymal cell layer lining the 3V in the mediobasal hypothalamus, which governs the local availability of T3 in the brain. Increased expression of Dio2 is seen following exposure to long days, with the converse sequence under short days, and spontaneous reversion occurs under prolonged photoperiod indicative of an innate timing mechanism. Photoperiodic control of Dio3 has been less widely reported, but Dio3 expression increases markedly under short photoperiod in the Siberian hamster, suggesting a key role in driving the summer to winter transition in physiology (Barrett et al., 2007). Studies in sheep, mice, and birds also demonstrate that it is thyroid-stimulating hormone (TSH) produced in the pars tuberalis (PT) of the pituitary stalk that acts locally in the hypothalamus to regulate Dio2 expression in the ependymal cells (Figure 4B). Thus, inductive and entraining effects of photoperiod on seasonal physiology are regulated at the level of the PT through the control of TH-dependent mechanisms in the brain. Later we discuss the evidence that the hypothalamic sites of regulated T3 availability harbor a stem cell niche.
Glucocorticoids
The adrenal glucocorticoid axis has far-reaching effects on histogenesis. Synthetic glucocorticoid agonists such as dexamethazone are among the key tools of oncologists because of their antiproliferative effects on tumors and leukemia (Pui and Evans, 2006). Correlations between glucocorticoid levels and histogenesis-dependent processes such as immune and memory function are well documented where intermediate levels produce maximum suppression as an inverted U-shaped function (e.g., McEwen, 1999; Leung and Bloom, 2003).
Recently, we have investigated the impact of the glucocorticoid axis on circannual prolactin secretion in the HPD Soay sheep model by giving the long-acting ACTH secretagogue synacthen-D (Syn-D) to super-activate the adrenal axis (G. A. Lincoln, unpublished results; Figure 5). Groups of HPD animals (n = 8/group) received Syn-D (0.5 mg/animal subcutaneously) or a sham vehicle treatment daily for 16 weeks following a switch from a short photoperiod (SP; 8 h light:16 h darkness) to a prolonged long photoperiod (LP; 16 h light:8 h darkness) designed to activate the expression of the circannual prolactin rhythm. Animals were treated starting either from the initial switch to LP (experiment 1) or starting at 8 weeks in LP when prolactin was already increased (experiment 2; Figure 5). Blood samples were collected hourly at different circannual phases to measure circulating cortisol concentrations and twice weekly throughout the experiment to measure prolactin secretion.
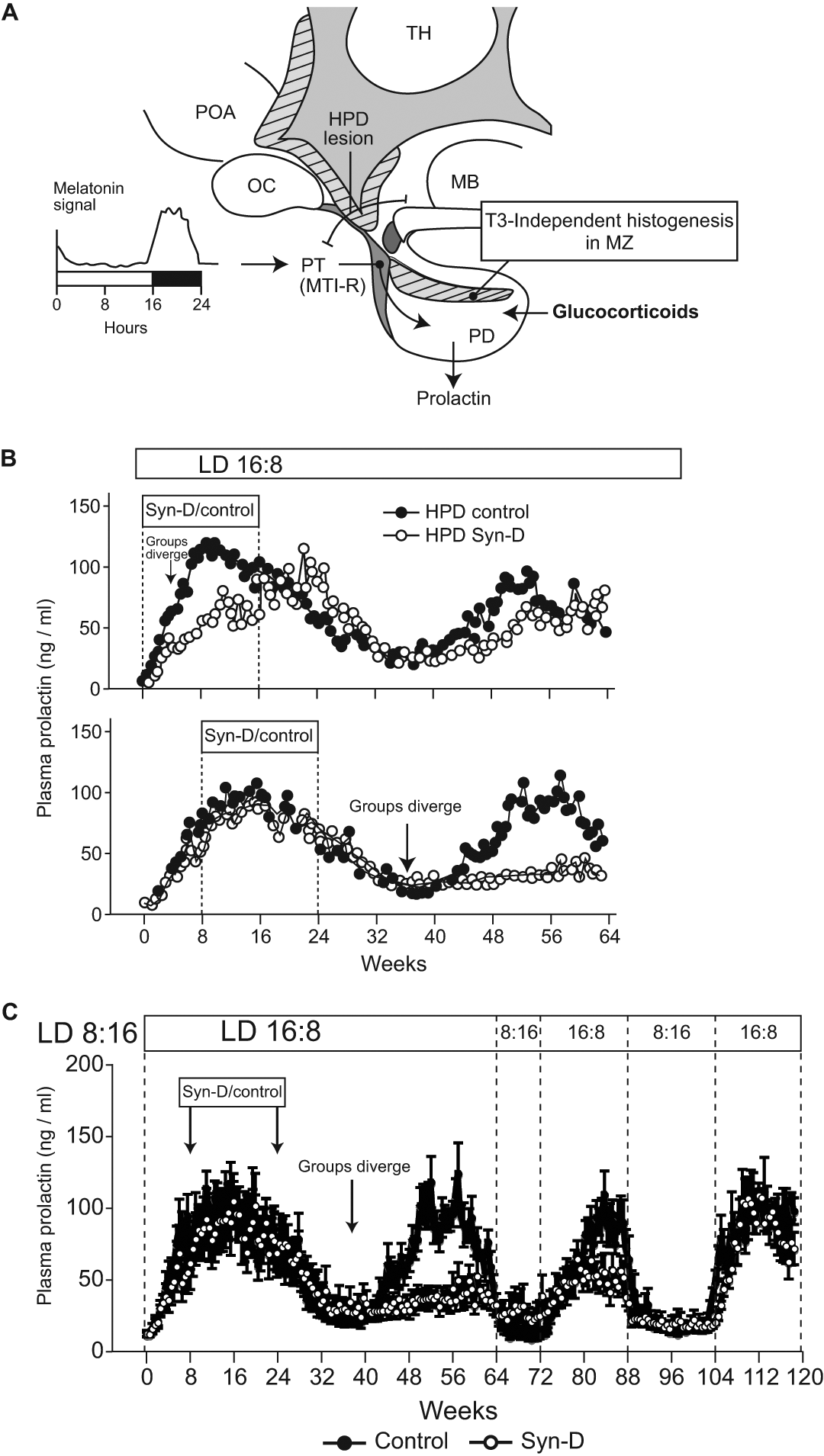
Glucocorticoid role in expression of circannual prolactin rhythms in HPD sheep. (A) Schematic sagittal view of the sheep brain showing the hypothalamo-pituitary disconnection (HPD) procedure; communication between the hypothalamus and pituitary is ablated by destruction of neurosecretory terminals in the median eminence (HPD lesion) and placement of a barrier between the two tissues to prevent restoration of neuroendocrine communication (based on Lincoln and Clarke, 1994). Also shown are the pathway by which the melatonin signal encoding photoperiod acts at the level of the pars tuberalis (PT) to control the secretion of prolactin from the pars distalis (PD; PT arrow) and the way glucocorticoids may act in the pituitary to influence T3-independent histogenesis in the marginal zone (MZ; putative circannual pacemaker). MB = mammillary body; OC = optic chiasm; POA = preoptic area; TH = thyroid hormone; MT-1 = melatonin type 1 receptor. (B) The glucocorticoid secretagogue synacthen-D (Syn-D, a long-acting agonist for adrenocorticotrophin receptors) causes delayed and persistent effects on circannual prolactin rhythm expression in the HPD Soay sheep maintained under long photoperiod (LD 16:8). (B, upper panel) A 16-week treatment with Syn-D during the rising phase of prolactin secretion (subjective summer) has an immediate delaying effect on circannual prolactin secretion (arrow, groups first diverge). (B, lower panel) A similar treatment at the peak of prolactin secretion (mid-subjective summer) has no initial effect on the prolactin profile but largely blocks reactivation of the next circannual cycle, evident at 12 weeks after the end of treatment (arrow, groups first diverge). (C) Recovery from the effects of Syn-D on the circannual prolactin cycle in the HPD model is very protracted, taking repeated switches between short photoperiod (LD 16:8) and long photoperiod (LD 16:8) over a period of 2 years to recover a normal prolactin pattern (G. A. Lincoln, unpublished results; for detailed methodology, refer to Supplementary Online Material). We envisage that these long-term effects are due to the action of high glucocorticoid levels on the histogenic processes in the pituitary gland. Open symbols, HPD controls; closed symbols, HPD + Syn-D treatment; Syn-D/control box, timing of treatment at 0.25 mg Syn-D/d by subcutaneous injection.
Syn-D administration increased blood cortisol levels into the high physiological range (maximum at 8 h and declining to normal values by 24 h—time of next injection), producing a chronic activation of the adrenal axis in all treated HPD animals (data not shown). The effect on the circannual prolactin rhythm was phase dependent, detectable immediately if the drug was given during the “subjective spring” phase of the circannual cycle when prolactin levels were increasing but not detectable until some 3 months later (well after the end of adrenal activation) if Syn-D treatment was delayed until the peak of the prolactin cycle (Figure 5B). Moreover, full recovery from the later Syn-D treatment for the prolactin axis was not seen until 1 to 2 years later, following repeated exposure to 8- to 16-week periods of alternating SP and LP to entrain the cycle (Figure 5C).
Our interpretation of these results is that Syn-D causes a glucocorticoid-dependent suppression of cell division within the anterior pituitary, possibly through induction of cell differentiation, and that the effect depends on the phase of the long-term cycle in pituitary histogenesis. Treatment in the proliferative phase (subjective spring) immediately delays the circannual cycle, while treatment later leads to markedly delayed responses. It is well established also that cell proliferation in multiple anterior pituitary cell types is sensitive to glucocorticoid and estrogen levels (Nolan et al., 1998; Nolan and Levy, 2003, 2009; Levy, 2008), and alterations in the pituitary stem cell niche with either immediate or delayed impact depending on circannual phase could account for the glucocorticoid effects in the HPD sheep.
4. The convergence point between photoperiodic input pathways and circannual rhythm expression occurs in histogenic regions of the hypothalamus and pituitary
The final line of supporting evidence follows on from statement 3: Within the hypothalamus and pituitary, there is good evidence for stem cell niches responsible for regulating long-term cycles.
A TH-regulated circannual stem cell niche in the mediobasal hypothalamus (SVZ-3v)
In the brain of adult mammals, histogenesis/neurogenesis is best documented for the subgranular zone (SGZ) of the hippocampus and for the subventricular zone surrounding the lateral ventricles (SVZ-lv; Ming and Song, 2005). Neurogenesis in the SGZ yields hippocampal granule neurons, necessary for proper memory function (Dupret et al., 2007). Stem cells in the SVZ-lv give rise to neuroblasts that migrate along the rostral migratory stream and maintain olfactory bulb function (Doetsch and Hen, 2005). In male songbirds, seasonal regeneration of the vocal centers responsible for song learning also depends on neurogenesis from progenitor cells in the lateral ventricle, with survival and migration of progeny cells being testosterone dependent (Nottebohm, 2004, 2005); interestingly, melatonin may play a role in this process in birds (Bentley et al., 1999).
Neurogenesis in the SVZ-lv of mice depends on TH acting through the type alpha TH-R (TRα; Lemkine et al., 2005), and as we described above, seasonal changes in neuroendocrine function are regulated by cells of the SVZ-3v, which control local T3 availability through deiodinase expression. Moreover, there are clear structural parallels between the SVZ-3v and the SVZ-lv: Both regions comprise cells expressing markers such as GFAP and S100 protein that characterize glial lineages, as well as neuronal progenitor cells. Of particular interest are the tanycyte cells of the SVZ-3v, which may function as neuronal stem cells (Placzek, 2009). These cells have a bipolar morphology, with the perikaryon penetrating the ependymal cell layer of the ventricular wall and contacting the cerebrospinal fluid (CSF), while a single long process reaches out into the surrounding brain tissue (Figure 3B). This morphology is reminiscent of radial glial cells in the SVZ-lv, which are thought to be neural stem cells. These cells derive from the same embryonic glial lineage (Rodríguez et al., 2005). Tanycytes have been subclassified into α1/2 and β1/2 subtypes based on their position along the walls of the third ventricle, the direction in which they project, and ultrastructural characteristics (Rodríguez et al., 2005). Among these cells, the α2 subtype has been suggested to be a potential hypothalamic stem cell type based on their cilial characteristics (Rodríguez et al., 2005).
Neurogenesis in the SVZ-3v has also been reported but is much less extensively studied, and the rates of cell proliferation in this area appear to be lower than in the SVZ-lv (Xu et al., 2005). Interestingly, in frogs, neurogenesis from the SVZ-3v was detected in the summer but not in the autumn, consistent with the concept of a seasonally regulated stem cell niche in this region (Chetverukhin and Polenov, 1993; Polenov and Chetverukhin, 1993). In Soay sheep, we have used acute BrDU treatment to label dividing cells and observed cell proliferation in multiple regions in the mediobasal hypothalamus (MBH), including the ependymal/tanycyte cell layer, the median eminence, and the parenchyma of the arcuate region (D. G. Hazlerigg, unpublished results; Figure 6A), as well as in the PT of the pituitary gland. This pattern of labeling in the brain corresponds well to the region of photoperiodically regulated expression of Dio2 in this species (Figure 6B; Hanon et al., 2008), demonstrating the potential for a TH-dependent modulation of histogenesis in the basal hypothalamus of seasonal species.
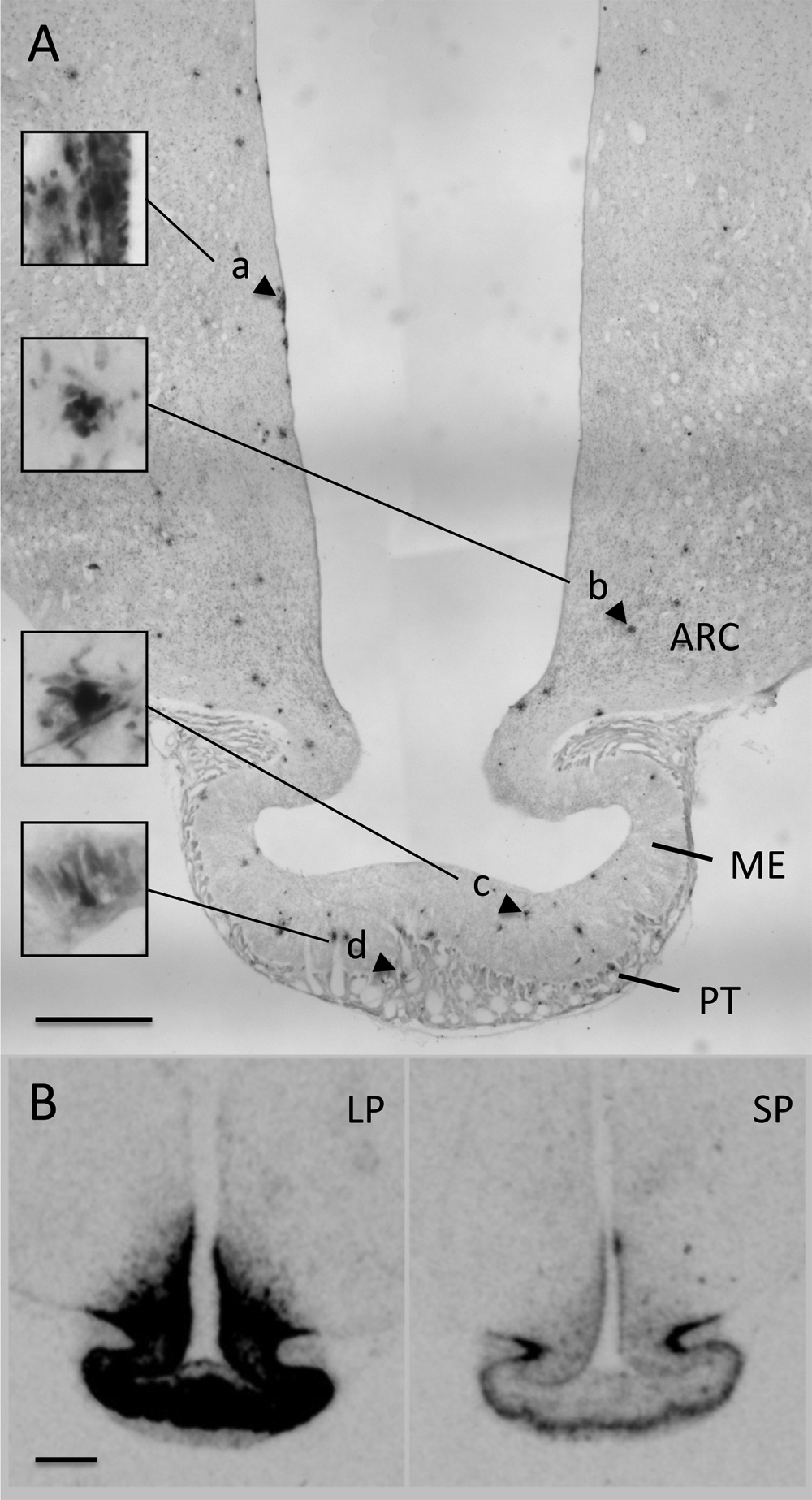
Bromodeoxyuridine (BrDU) incorporation in the photoperiodic control centers of the sheep hypothalamus and pituitary. (A) Low-power (5×) photomicrograph of a coronal section of the mediobasal hypothalamus (MBH) showing diaminobenzidine-stained BrdU-positive cell nuclei in a sheep killed 24 h after intravenous injection of BrdU (for methodological details, see Supplemental Material). Multiple dividing cells can be seen in 3 distinct regions of the SVZ-3v (putative circannual pacemaker): the walls of the 3v (a), the parenchyma of the arcuate nucleus (b), the median eminence (c), and also in the pars tuberalis of the pituitary gland (d; only a narrow strip of PT tissue shown). Scale bar = 1 mm. Insets are 20× magnification images of the cells indicated. (B) Photoperiodic control of type 2 deiodinase (Dio2) RNA expression across the sheep basal hypothalamus—note extensive and intense labeling in the median eminence, ependymal/tanycyte layer, and parenchyma of the mediobasal hypothalamus under LP (exposure to LD 16:8 for 8 weeks) and much less intense labeling restricted to the external zone of the median eminence/PT interface under SP (exposure to LD 8:16 for 8 weeks) (D. G. Hazlerigg, unpublished results). Scale bar = 1 mm. ARC = arcuate nucleus; ME = median eminence; PT = pars tuberalis.
Compelling evidence that the SVZ-3v plays a neurogenic function in mammals comes from a study seeking to understand the prolonged effects of ciliary neurotrophic factor (CNTF) on appetite in mice (Lambert et al., 2001). CNTF delivered into the 3v stimulates cell division in the SVZ-3v, correlating with reduced appetite and body weight, and this persisted for many weeks after the end of treatment (Kokoeva et al., 2005). These persistent effects were blocked by co-infusion of cytosine arabinoside, a drug suppressing cell division. Fate mapping of dividing cells showed that the CNTF treatment generates new precursor cells in the MBH that migrate into the appetite regulatory centers and differentiate into neurones expressing appetite regulatory peptides, accounting for the prolonged effect on energy metabolism. We interpret this striking result as a pharmacological perturbation of the homeostatic process that regulates changes in body weight and other aspects of physiology in the normal animal. In sheep, CNTF-R expression in the mediobasal hypothalamus maps onto the tanycycte/TSH-R-expressing region that we propose is pivotal to circannual rhythm generation, and intriguingly the pituitary PT immediately adjacent to the MBH appears to be a source of CNTF (D. G. Hazlerigg, unpublished results). Thus, CNTF and other neurotrophic factors may coordinate circannual changes through the local induction of histogenesis.
Circannual stem cell niches within the anterior pituitary (MZ)
Recent fate mapping studies in mice have defined the marginal zone (MZ) of the pituitary cleft as a pituitary stem cell niche, capable of giving rise to a full spectrum of anterior pituitary endocrine cell types (Gleiberman et al., 2008; Fauquier et al., 2008; Vankelecom, 2009). These cleft cells express the stem cell markers nestin and SOX2 and in vitro retain the capacity for division and pluripotent differentiation into multiple endocrine cells. These cells also express receptors for the brain-derived neurotrophic factor, neurturin, suggesting that this factor may regulate the MZ niche (Garcia-Lavandeira et al., 2009). An attractive hypothesis is that the pituitary MZ generates circannual rhythms in some physiological systems. This may particularly apply to the circannual control of prolactin, which in contrast to the regulation of gonadotrophins is not governed by hypothalamic TH-dependent mechanisms. This is supported by the persistence of circannual prolactin cycles in HPD Soay sheep (Lincoln et al., 2006; Figure 5B,C) and with the described impact of glucocorticoids on circannual prolactin rhythms in the HPD model.
HPD sheep retain normal photoperiod responsiveness for the prolactin axis, and recent work suggests that this involves the relay of photoperiodic information from the melatonin responsive PT to the PD, by a peptide cleavage product of TAC1, which encodes the neurokinins substance P and neurokinin A (Dupré et al., 2010; Figure 5A). Puzzlingly, neurokinin receptors are expressed by corticotrophs but not by mature lactotrophs in the sheep pituitary, suggesting that the circannual regulation of prolactin involves paracrine intermediary mechanisms. One possibility is that TAC1 gene products from the PT influence the function of the pituitary MZ niche and that this leads to recruitment of a new prolactin secretory anterior pituitary cell population. This is consistent with the long latency of prolactin responses in the HPD sheep model (Figure 5B,C).
The possibility that the PT itself undergoes cyclical histogenesis acting as a central pacemaker should not be excluded. BrdU treatment labels cells in the sheep PT (Figure 6A). In mice, nestin labeling (index of progenitor/stem cells) has been found in the PT region, extending along the anterior-ventral face of the PD (Gleiberman et al., 2008). This distribution overlaps with MT-1 melatonin receptor expressing cells in newborn rodents (Johnston et al., 2003), suggesting a link between melatonin sensitivity and histogenic potential. Since the PT (unlike the PD) retains MT-1 melatonin receptors into adult life, we hypothesize that this histogenic function may also be maintained beyond juvenile ontogeny. The general concept that melatonin actions might be exerted through cell developmental pathways is given impetus by the recent finding the eye- and limb-developmental gene eyes absent 3 (Eya3) is a key element of the photoperiodic response pathway in vertebrates (Dardente et al., 2010, Matsumoto et al., 2010). The effects of melatonin on avian song center activity (Bentley et al., 1999) might similarly be accounted for by actions through neurodevelopmental regulatory pathways.
Outlook: Developing And Testing The Cyclical Histogenesis Hypothesis
The basic challenge we face is of localizing and quantifying histogenic cycles in the hypothalamus and pituitary over the long time domain of months and years. We need to define the origins of new cells and the fates they follow and assess the way the processes are temporally regulated to produce a circannual clock. Visualizing dividing cells by labeling newly synthesized DNA with BrdU makes it clear that cell division continues in the basal hypothalamus and pituitary of adult mammals, including seasonal hamsters (Huang et al., 1998) and sheep (Figure 6). The hope is that recent developments in defining the intersection between photoperiodic input pathways and neuroendocrine outputs (Hazlerigg and Loudon, 2008) will help focus attention on histogenic processes in defined hypothalamic or pituitary subregions.
BrdU can be exploited in “pulse-chase” designs, allowing the fates of cells that were dividing during a specific window to be observed. Fate may be defined in terms of cellular localization, as well as differentiated phenotype through dual- or triple-staining immunocytochemistry. BrdU-labeled cells may undergo apoptosis, and in the mouse hippocampus, the majority of dividing cells are lost by this route, and thus regulation of cell survival might be as important as the regulation of cell birth in histogenesis cycle control (Sierra et al., 2010; Dupret et al., 2007). Avian song center development depends on the cell survival–promoting effects of testosterone in male songbirds (Nottebohm, 2005), while the rate of apoptosis is a key factor in periodic hematological disorders (Haurie et al., 1998).
With pulse-chase approaches, a difficulty is that repeated rounds of cell division dilute the BrdU signal, making it difficult to track the fates of progeny cells. To overcome this, two other in vivo methods might be co-opted in studying circannual rhythms (Breunig et al., 2007). First, replication-deficient viral particles, engineered to incorporate reporter genes such as green fluorescent protein into the genome of cells undergoing cell division, offer a route to cell fate mapping without dilution of signal. Second, Cre-recombinase/lox P site (Cre/loxP) recombination, allowing “floxing” of progenitor cells in mice, is a powerful tool for studying histogenesis, allowing both fate mapping and functional targeting of dividing cells and their progeny (Kuo et al., 2006; Breunig et al., 2007).
To adopt Cre/loxP methods, we must revisit an old problem besetting seasonal physiologists—namely, the “nonseasonal” phenotype of the laboratory mouse. Recent work shows, however, that laboratory mice do respond to melatonin at the level of pituitary PT (TSH, circadian gene expression) and at the level of the hypothalamus (Dio2 expression; Ono et al., 2008). On the premise that photoperiodic input and circannual rhythm generation converge in the pituitary and basal hypothalamus, this gives considerable encouragement. The benefits of the Cre/lox P approach make a strategic move to the mouse a compelling objective.
The Conjecture Summary
We present the histogenesis hypothesis Zto explain the long time domains that characterize circannual rhythms. The lack of understanding of long-term rhythms is in contrast to the remarkable progress in the circadian field and may reflect a failure to recognize that histogenic processes running at low rates over months to years are at the core of circannual timekeeping. The key issues are the extent to which tissue regeneration is synchronized in different sites in the body by intrinsic mechanisms to produce protracted cycles and the processes by which the different sites are temporally coordinated to produce seasonal phenotypes (the circannual clock shop). We identify histogenic sites in the hypothalamus associated with the third ventricle (SVZ-3v) and in the pituitary gland associated with the embryonic cleft (MZ) with their discrete stem cell niches as candidate circannual pacemakers. The brain site potentially generates new neurones that enter the hypothalamic centers to control reproduction and energy metabolism, and the pituitary site generates new pituitary cells that have profound systemic effects on the functional activity of body organs. These tissues are seen as regulators of the transformation between the subjective summer and subjective winter phenotype—a form of reversible metamorphosis. Circannual rhythms are expressed by many vertebrates, including fish, amphibians, and reptiles, and we predict that cyclical histogenesis evolved in ancestral species as a strategy to time regenerative processes to the period of food abundance in summer. Hence, it is likely that sets of genes conserved across vertebrates regulate core histogenic cycles. Judging from control of cyclic growth processes in the hair follicle, these will be shared with those regulating embryonic development. Adult stem cells in their specialized tissue niches are seen to recapitulate events of ontogeny, and remarkably, new progeny cells must be born in each cycle to produce sustained circannual rhythms. The important conclusion is that most long-lived organisms have a genetically programmed cyclical life history, revealed under a constant environment. The new challenge is to identify the gene sets that govern the timing of cyclic histogenesis in the adult and confirm their role in circannual timing by tissue-specific ablation.
Footnotes
Acknowledgements
The authors thank the Biotechnology & Biological Sciences Research Council (UK) and the Medical Research Council (UK) for supporting their research, 3 anonymous reviewers for insightful and constructive criticism, and Ronnie Grant for excellent graphics support.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
