Abstract
Recent demonstration of the histone acetyltransferase activity of the Clock gene greatly expanded the regulatory role of circadian clocks in gene transcription. Clock and its partner Bmal1 are responsible for the generation of circadian oscillations that are synchronized (entrained) to the external light cycle. Entraining light often produces long-lasting changes in the endogenous period called aftereffects. Aftereffects are light-dependent alterations in the speed of free-running rhythms that persist for several weeks upon termination of light exposure. How light causes such long-lasting changes is unknown. However, the persistent nature of circadian aftereffects in conjunction with the long-term effects of epigenetic modifications on development and various aspects of brain physiology prompted us to hypothesize that the histone acetyltransferase CLOCK was required for circadian aftereffects. The authors exposed Clock knockout mice to 25-hour light cycles and report that these mice retain the ability to display circadian aftereffects, indicating that Clock is dispensable for this form of circadian plasticity.
Chromatin remodeling shapes transcription profiles that underlie neuronal plasticity, learning and memory, early life modulation of adult behavior, and responses to drug exposure (reviewed in Meaney and Ferguson-Smith, 2010). Chromatin remodeling is also integral to circadian clock function. The Clock gene is the only canonical clock gene with histone acetyltransferase activity, and Clock acetylates several histone and nonhistone residues on genes including Bmal1, its partner in circadian transcription (Asher and Schibler, 2006; Doi et al., 2006; Grimaldi et al., 2009). Thus, the interplay between Clock and Bmal1 as regulators of the circadian program and Clock-mediated acetylation regulates the transcriptional mechanism underlying the expression of overt circadian rhythms (Grimaldi et al., 2009).
Circadian aftereffects are light-induced alterations of the free-running period following exposure to non–24-hour light schedules (T cycles) (Pittendrigh and Daan, 1976). Because circadian aftereffects are a form of experience-dependent long-term phenotypic plasticity, and because Clock influences chromatin events involved in circadian rhythmicity, we hypothesized that circadian aftereffects are dependent on Clock by virtue of its effects on chromatin modifications. We tested this hypothesis by exposing Clock knockout (Clock−/−) mice (Debruyne et al., 2006) to a 25-hour T cycle (T25) known to induce aftereffects in wild-type mice.
We measured the endogenous free-running period, entrainment to a 25-hour T cycle, and induction of aftereffects in 6 male Clock−/− and 6 male wild-type (Clock+/+) mice (Appendix). We confirmed that Clock−/− mice did not show staining for the CLOCK protein in the SCN (Fig. 1). As shown previously (Debruyne et al., 2006), Clock−/− mice are rhythmic, free run with a period significantly shorter than Clock+/+ mice (Fig. 2 and Table 1), display reduced locomotor activity (average of 54% decrease, p < 0.001), and show significantly advanced phase angles of entrainment under both LD (LD1 Clock−/−: −1.70 ± 0.10 h v. Clock+/+: −0.05 ± 0.1 h, p < 0.00001; LD2 Clock−/−: −1.85 ± 0.22 h v. Clock+/+: −0.20 ± 0.17 h, p < 0.001) and during T25 (Clock−/−: −3.20 ± 0.39 h v. Clock+/+: −0.31 ± 0.15 h, p < 0.0001). Except for the initial LD condition, we report that Clock−/− mice display lower circadian amplitudes throughout the experiment (average of 60% decrease in amplitude, p < 0.01), a finding not reported by Reppert’s group (Debruyne et al., 2006). All mice entrained to the T25 (Fig. 2) and started to free run in DD with a phase predicted by their respective phase angles during entrainment to the T cycle (bottom of each actogram in Fig. 2). All mice displayed circadian aftereffects following T25 (Fig. 2 and Table 1) and had identical free-running periods following the T cycle (Table 1). Thus, we conclude that CLOCK, and by extension its acetyltransferase activity, is dispensable for the expression of circadian aftereffects.

Clock−/− mice do not express CLOCK. Representative photomicrographs of a Clock+/+ (top) and a Clock−/− (bottom) SCN section stained with a rabbit anti-CLOCK primary. Magnification, 10x.
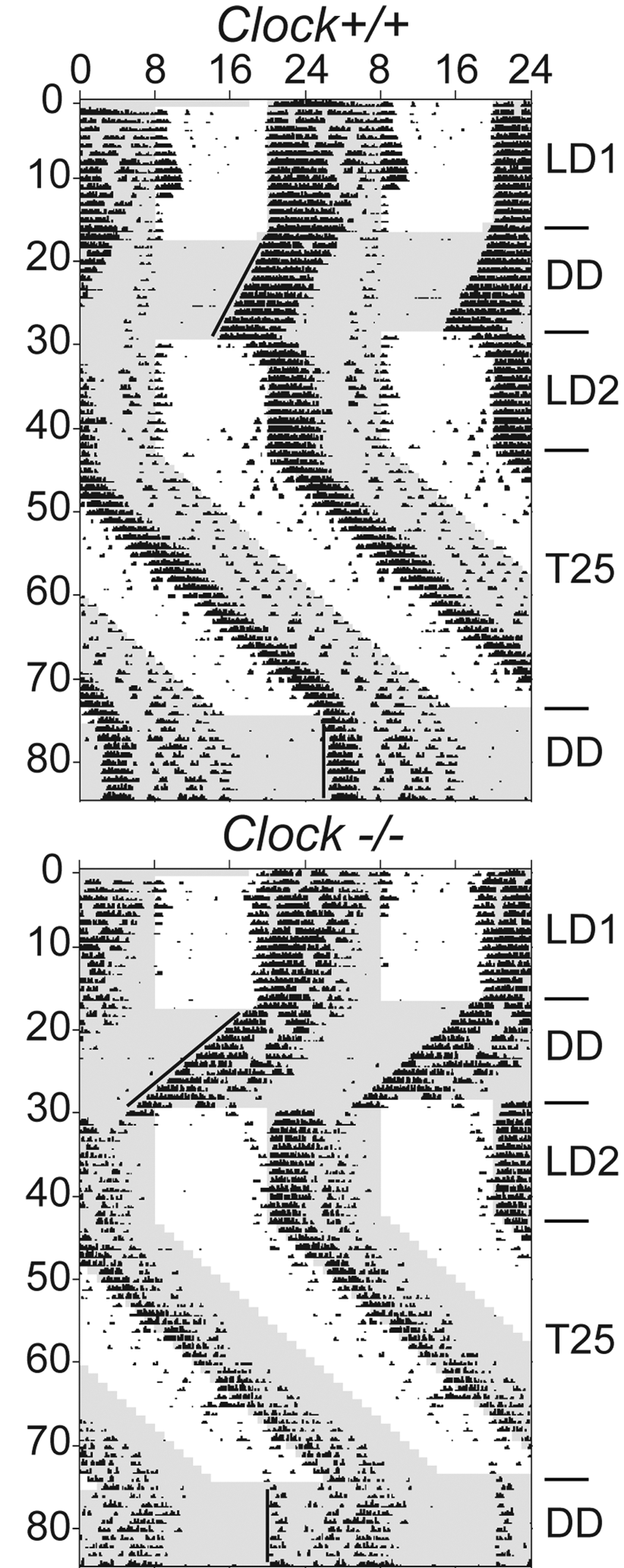
Clock−/− mice entrain to 25-hour T cycles (T25) and show aftereffects. Representative double-plotted actograms for a Clock+/+ (top) and a Clock−/− mouse (bottom). Black regression lines over activity onsets in DD conditions illustrate the period of free-running rhythms and the aftereffect induced by the T cycle. Gray shaded area represents times of darkness. LD = 12-hour light:12-hour dark cycle; DD = complete darkness; T25 = 12-hour light:13-hour dark cycle.
Circadian period in DD and during aftereffects.
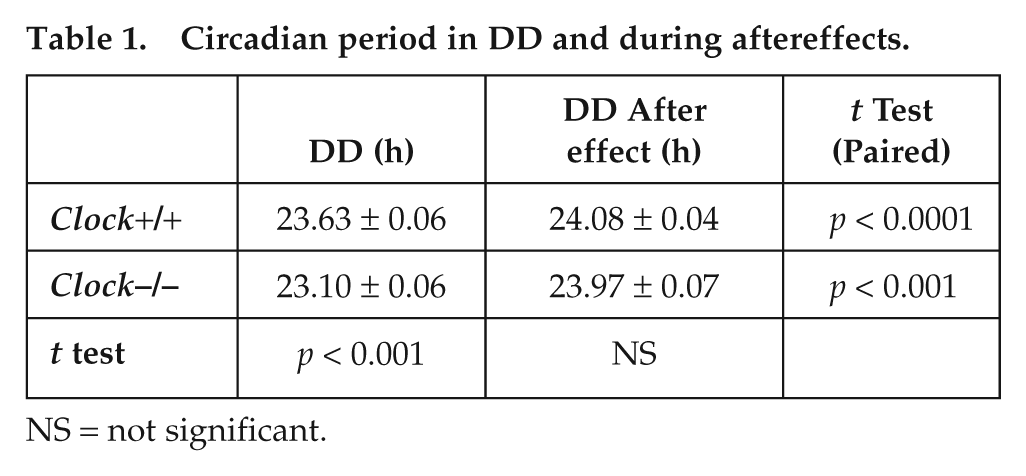
NS = not significant.
Despite the reported role played by the acetyltransferase activity of the CLOCK protein in regulating gene transcription, including regulation of the Bmal1 gene essential for expression of circadian rhythms (Grimaldi et al., 2009), we find that CLOCK is dispensable for circadian aftereffects. A recent report further showed that circadian aftereffects do not depend on any of the 3 Period genes (Pendergast et al., 2010), suggesting that aftereffects are independent from clock gene regulation. These results also suggest that if chromatin remodeling were indeed involved in aftereffects, its mechanism would depend on factors other than the Clock and Period genes.
Interestingly, aftereffects can be maternally transferred when pregnant mice are exposed to T cycles during gestation (Aton et al., 2004). Additionally, photoperiod length, another condition that induces aftereffects (Pittendrigh and Daan, 1976), experienced by neonatal mice changes (imprints) their adult free-running period, showing again that light has long-lasting effects on circadian behaviors (Ciarleglio et al., 2011). Whether maternally transferred aftereffect or photoperiod imprinting requires chromatin remodeling, and whether CLOCK is required for this type of light-induced circadian plasticity, remains to be explored.
It is interesting that Clock−/− mice entrain to T25, which requires daily phase delays of approximately 1.9 hours, despite a previous report showing severe reductions in their ability to phase delay (Debruyne et al., 2006). In our experiment, Clock−/− mice entrained to light cycles with periods significantly longer than their endogenous free-running periods and displayed large, negative phase angles of entrainment under all entrainment conditions. Advanced phase angles of entrainment in Clock−/− mice can be a consequence of both their reduced ability to phase delay as well as their shorter endogenous free-running period. However, this altered phase relationship also complicates the determination of phase shifts obtained by a modified Aschoff type 2 procedure, as employed previously (Debruyne et al., 2006). Determination of a complete phase-response curve for Clock−/− mice would confirm that these mice can indeed phase delay and characterize the shape of their phase responses to light.
The question is still open on whether the acetyltransferase activity of CLOCK is required for circadian behaviors. Finally, it is unclear how phenotypes previously observed in Clock/Clock mutant mice bearing a dominant-negative form of CLOCK, such as sensitivity to drugs of abuse, metabolic syndrome, or even circadian disruptions (King et al., 1997; McClung et al., 2005; Turek et al., 2005), are due to disrupted chromatin remodeling. Future research should make good use of the Clock knockout model to further characterize the requirements for CLOCK-mediated acetylation in the regulation of circadian and other functions.
Footnotes
Appendix
Acknowledgements
The authors thank Dr. David Weaver (University of Massachusetts Medical School) for the gift of anti-CLOCK primary antibody. This work was supported by grants from the Canadian Institute of Health Research (CIHR; #086549) and from the Canadian Foundation for Innovation (CFI) to H.-Y.M.C. In addition, H.-Y.M.C. holds a Canada Research Chair (CRC) Tier II in the Genetics of Biological Timing and an Early Researcher Award from the Ontario Ministry of Research and Innovation.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
