Abstract
Even though peripheral circadian oscillators in the cardiovascular system are known to exist, the daily rhythms of the cardiovascular system are mainly attributed to autonomic or hormonal inputs under the control of the central oscillator, the suprachiasmatic nucleus (SCN). In order to examine the role of peripheral oscillators in the cardiovascular system, we used a transgenic mouse where the
The cardiovascular system is known to be under circadian control, and traditionally, its daily rhythms are attributed to autonomic or hormonal inputs governed by the SCN (Yamashita et al., 2003). Nonetheless, circadian changes in the heart from gene expression to metabolism to contractile function are intrinsic and independent of autonomic inputs (Bray et al., 2008; Durgan et al., 2005; Durgan et al., 2006; Young and Bray, 2007). We previously showed that at the cellular level, the L-type voltage-gated calcium channels (L-VGCCs) and phosphorylation states of several signaling molecules, such as extracellular signal–regulated kinase (ERK) and phosphoinositide 3-kinase (PI3K)–protein kinase B (AKT), are under circadian control in chick cardiomyocytes (Ko et al., 2010). However, we obtained heart tissue and cardiomyocytes from entrained whole chicken embryos, making it difficult to dissect the significance of peripheral cardiac oscillators on daily rhythms independent of the master clock. Therefore, we set forth to examine the circadian profiles of cardiomyocyte-specific CLOCK mutant (CCM) mice. In these mice, a dominant-negative mutation of the oscillator gene
In addition, using CCM mice is in contrast to the study using a transgenic model with brain tissue–specific disruption of the circadian oscillator (Hong et al., 2007). In the transgenic mice with brain tissue–specific disruption of
Even though there is no apparent pathological problem in CCM mice, as shown in the ultrastructure of CCM cardiomyocytes (Bray et al., 2008), certain changes in the cardiac circadian profiles of CCM mice might have further downstream effects. While wheel-running activity can be used as an indicator of circadian-governed locomotor activity, it also requires higher physical strength and adaptation in contrast to general locomotor activities such as grooming, food intake, or walking. In the present study, we investigated the circadian-driven wheel-running activities of CCM mice and their WT littermates over a 5-week period. We further analyzed the differences in the phosphorylation profiles of several signaling molecules including p42/44 ERK (pERK), p38 MAPK (stress-activated protein kinase), AKT at thr308 (pAKTthr308), and glycogen synthase kinase-3β (pGSK), as well as the protein expression of the L-type voltage-gated calcium channel α1D subunit (L-VGCCα1D) between CCM and WT mice. Our overall results demonstrated that the circadian core oscillator genes in cardiomyocytes play important roles in regulating normal physiological function of the heart, and CCM mice allow us to differentiate between circadian input and output pathways.
Materials and Methods
Both WT and CCM male mice (FVB background) were originally generated at the Children’s Nutrition Research Facility, Children’s Nutrition Research Center, Baylor College of Medicine (Houston, TX). All animal experiments were approved by the Institutional Animal Care and Use Committee of Baylor College of Medicine. All mice were housed under temperature- and humidity-controlled conditions with 12:12-hour LD cycles strictly enforced and fed standard laboratory chow and water ad libitum. Some mice were transferred to Texas A&M University for wheel-running locomotor activity assessment and other experiments.
Wheel-Running Locomotor Activity
All mice between 20 to 28 weeks old were housed individually in cages equipped with running wheels in the animal facility at the Texas A&M University Health Science Center. The procedure used for the wheel-running activity study was approved by the University Laboratory Animal Care Committee at Texas A&M University. All mice were fed standard laboratory chow and water ad libitum. Mice were entrained to 12:12 LD cycles for 5 weeks and then kept in constant darkness (DD; to allow for free-running circadian rhythms) for another 3 weeks. Wheel-running activities were continuously recorded and stored in 10-minute bins, and data were collected and analyzed using ClockLab software (ActiMetrics, Evanston, IL) (Allen et al., 2005). The phase angle of entrainment (Ψ) was assessed under LD conditions. During entrainment, the onset of activity for a given cycle was identified as the first bin during which an animal attained 10% of peak running-wheel revolutions. To measure Ψ, least-squares analysis was used to establish a regression line through the daily onsets of activity during the period of LD entrainment, and then the number of minutes before (positive) or after (negative) the time of lights-off in the LD cycles was determined for each animal. For each animal, the steady-state circadian period (t) of the activity rhythm in DD was determined by χ2 periodogram and fast Fourier transform. The total activity count was calculated by averaging the number of wheel revolutions per 24 hours over the 5-week interval of analysis. An activity bout was defined as a period during which wheel-running activity never dropped below 10 counts/bin for more than 20 minutes. These criteria for bout threshold and maximum intrabout interval were used to determine the number of activity bouts per day as well as bout duration (length) and size (average counts per bout). Comparisons between CCM and WT mice were made using the Student
Preparation of Mouse Heart Samples
Mouse heart samples were collected at both Baylor College of Medicine as well as Texas A&M University. Heart ventricles were isolated from 22-week-old animals at ZT 0, 6, 12, and 18. Heart tissues were subsequently freeze powdered and stored at −80 °C. Ventricular RNA was isolated from powdered ventricular tissue using a standard method (Chomczynski and Sacchi, 1987) with Total RNA Isolation Reagent (Molecular Research Center Inc., Cincinnati, OH) and used for quantitative real-time reverse transcription polymerase chain reaction (Q-PCR) assays. Ventricular protein was isolated from powdered ventricular tissue using a standard method with slight modifications (Gibala et al., 2000). Briefly, protein extraction buffer (50 mM Tris-HCl, 150 mM NaCl, 2 mM EGTA, 2 mM EDTA, 2 mM NaF, 2 mM NaPPi, 1% Nonidet P40, 1% glycerol, 5% SDS, 10 mM Na3VO4, 1 mM PMSF, 6.4 µM benzamidine, 0.15 µM aprotinin, 1 µM leupeptin, and 0.73 µM pepstatin A) was added to frozen ventricular powder (25 mg tissue/mL buffer) and immediately homogenized. The homogenates were subsequently centrifuged at 15,000
Immunoblot Analysis
Samples were separated on 10% SDS-PAGE gels and transferred to nitrocellulose membranes as described previously (Ko et al., 2009a; Ko et al., 2007; Ko et al., 2010). The primary antibodies used in this study were anti-VGCCα1C (Alomone, Jerusalem, Israel), anti-VGCCα1D (Alomone), anti–phospho308 AKT (pAKTthr308, Cell Signaling Technology, Danvers, MA), anti–phosphoGSK-3β at ser9 (pGSK, Cell Signaling Technology), anti–phospho stress-activated protein kinase p38 (p38, Cell Signaling Technology), a monoclonal antibody specific for diphospho extracellular signal–related kinase (pERK, Sigma, St. Louis, MO), and a polyclonal antibody insensitive to the phosphorylation state of ERK (total ERK, used for internal and loading control) (Santa Cruz Biochemicals, Santa Cruz, CA). Blots were visualized using appropriate secondary antibodies conjugated to horseradish peroxidase (Cell Signaling Technology) and an ECL detection system (Pierce, Rockford, IL). Relative protein expressions for all proteins involved in this study are reported as a ratio to total ERK because total ERK remains constant throughout the day. The ratio of pERK to total ERK, p38 to total ERK, pAKT to total ERK, pGSK to total ERK, and VGCC to total ERK was determined by densitometry using Scion Image (NIH, Bethesda, MD). For each set of experiments, either the first time point or lowest ratio value of the WT was arbitrarily set to 1. All measurements were repeated at least 3 times.
Quantitative Real-Time RT-PCR
The method used for Q-PCR analysis was described previously (Ko et al., 2007; Ko et al., 2010). Mouse ventricles collected at 4 different time points for Q-PCR were described above. There was 300 ng of total RNA used to quantify the mRNAs of
All data are presented as mean ± SEM (standard error of the mean). One-way ANOVA followed by the Tukey post hoc test for unbalanced
Results
Disruption of Cardiac Clock Did Not Affect the Circadian Rhythm of Wheel-Running Behavior but Hampered Physical Endurance
Wheel running is a circadian event in mice. It demands higher cardiac workload than routine activities such as grooming, foraging, and walking. Because there is no difference in the circadian rhythms of regular physical activities detected by radiotelemetric infrared beam counts between CCM mice and their WT littermates (Bray et al., 2008), we set forth to examine whether interruption of
WT (Fig. 1A) and CCM mice (Fig. 1B) double-plotted actograms in LD (A1, B1) and DD (A2, B2) show no statistical difference in phase angle of entrainment (Ψ; WT = 2.06 ± 0.89 hours, CCM = 1.67 ± 0.88 hours) or steady-state circadian period (t; WT = 23.52 ± 0.06 hours, CCM = 23.96 ± 0.28 hours; WT:
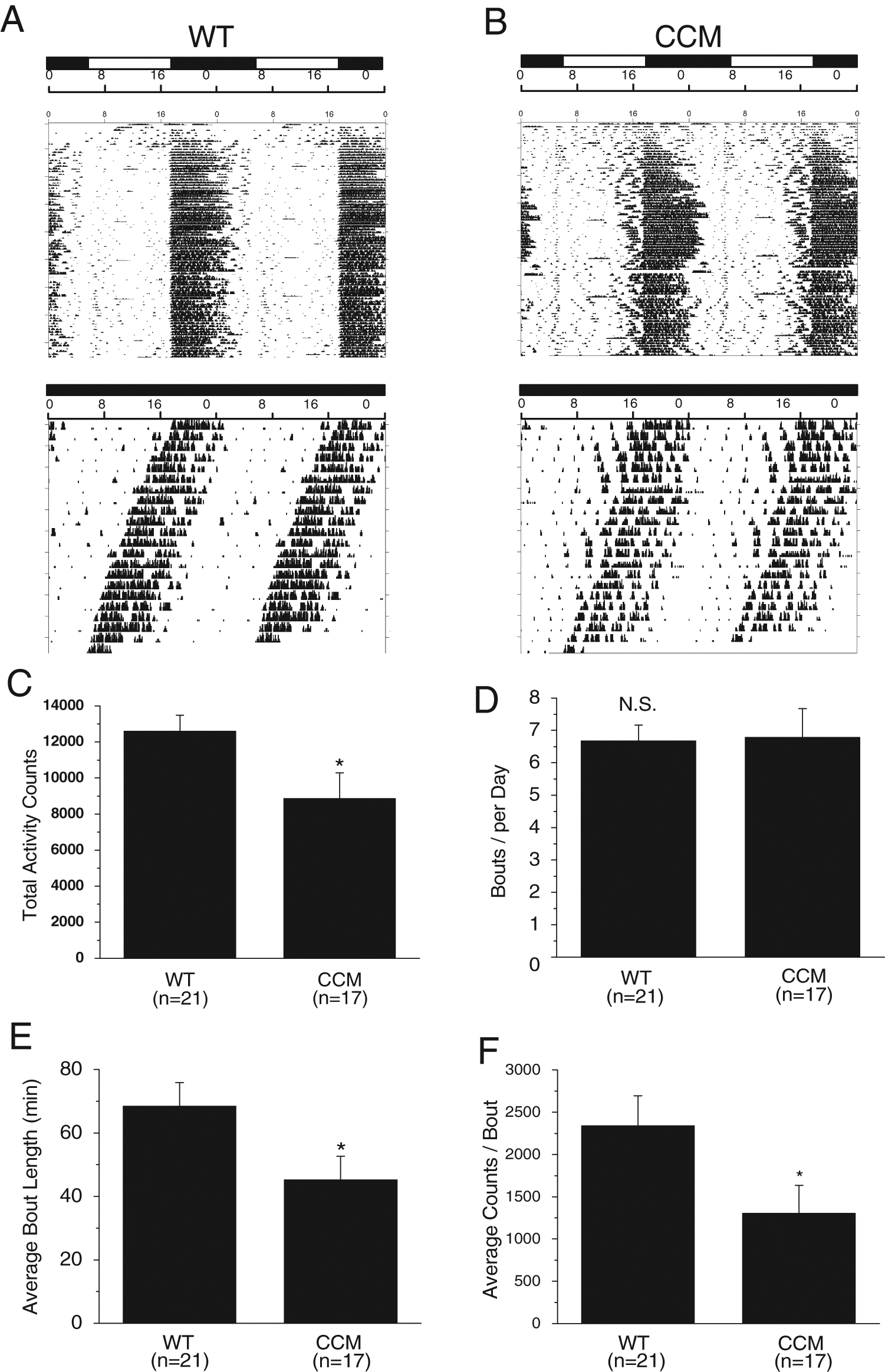
Wheel-running locomotor activity rhythms in CCM and WT mice. (A and B) Double-plotted wheel-running actograms from a WT mouse (A) housed in LD (top panel) and in DD (lower panel) and from a CCM mouse (B) housed in LD (top panel) and in DD (lower panel). When mice were in LD cycles, the lights were on at 0600 h and off at 1800 h. (C) The WT mice had significantly higher total numbers of wheel revolutions (total activity counts) compared to CCM littermates. (D) There was no difference between WT and CCM mice in total number of bouts per day (bouts per day). (E) The CCM mice had significantly lower length of time for each bout on average (average bout length in minutes) compared to WT mice. (F) The WT mice had significantly higher counts of wheel revolutions for each bout (average counts/bout) compared to CCM mice. WT:
The Phosphorylation States of Several Signaling Molecules Were under Cardiac Circadian Control
Because CCM mice had significantly lower wheel-running activities than the WT in the quantitative analysis, we next examined whether the circadian rhythms of the phosphorylation states of several signaling molecules important in many cardiac physiological conditions and function, including ERK, p38, AKT, and GSK-3β, were altered in CCM mouse hearts. Both ERK and p38 have been implicated in regulating cardiac contractility and the development of various pathological states such as cardiac hypertrophy and heart failure because they control cell growth and proliferation (Olson and Molkentin, 1999; Sugden, 1999; Szokodi et al., 2008). Also, in human failing hearts, the activity of GSK-3β is greatly inhibited (Haq et al., 2001). The phosphorylation states of ERK (diphosphorylated; pERK) and p38 (phosphorylated p38; p38) displayed diurnal rhythms in WT ventricles (Fig. 2A and 2B). While pERK peaked at ZT 0 (Fig. 2A), p38 peaked at ZT 12 (Fig. 2B). Interestingly, in CCM mice, pERK was no longer rhythmic (Fig. 2A), while p38 remained rhythmic (Fig. 2B). In WT mice, phosphorylation of AKT at thr308 (pAKTthr308) peaked at ZT 6 (Fig. 2C), while the phosphorylation of its downstream target GSK-3β (pGSK) peaked at ZT 12 to 18 (~6-hour lag behind pAKTthr308) (Fig. 2D). In CCM mouse ventricles, both pAKTthr308 (Fig. 2C) and pGSK (Fig. 2D) were no longer rhythmic. Hence, the phosphorylation states of ERK, AKTthr308, and GSK were under cardiac
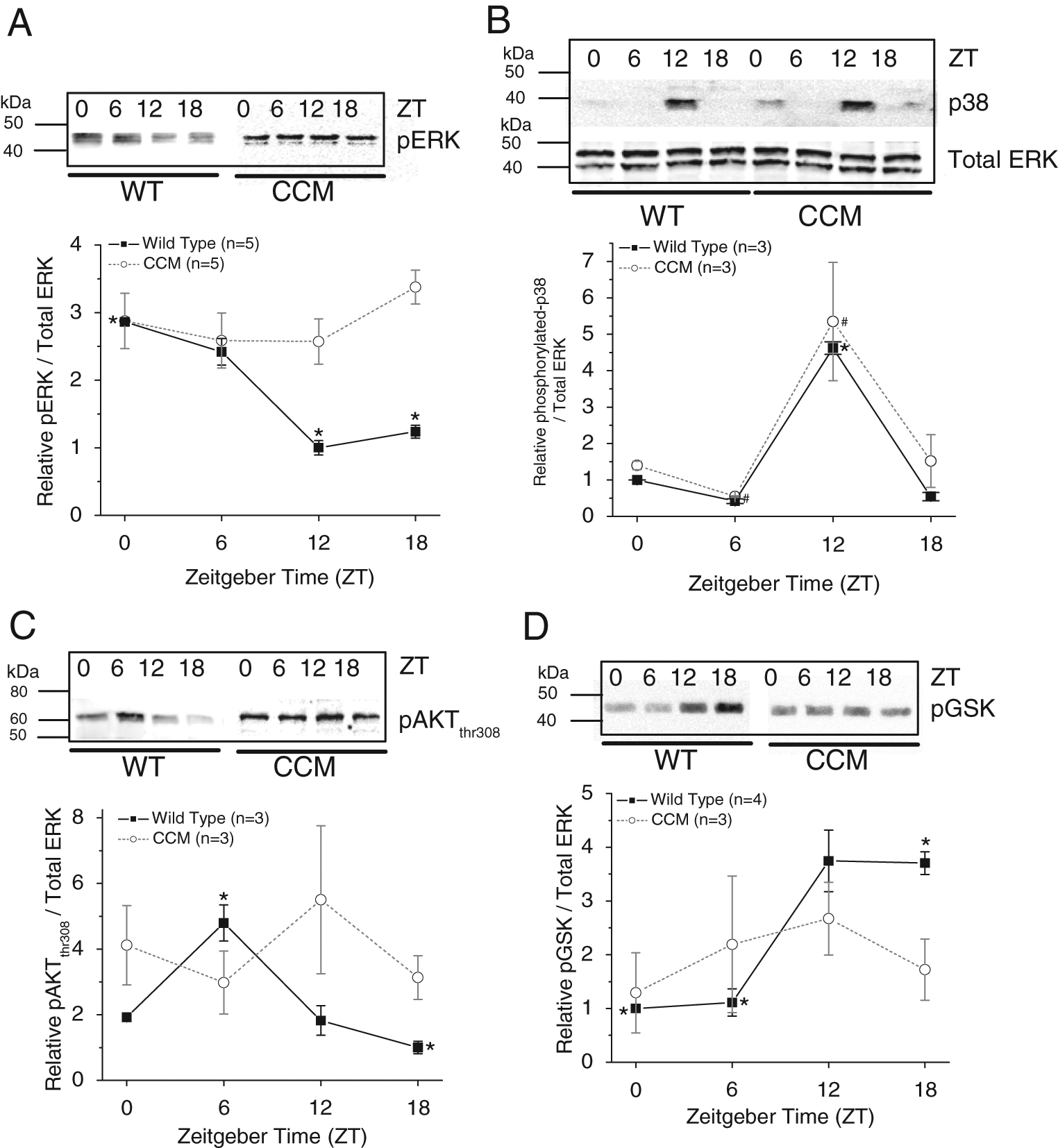
The daily rhythms of protein expression and phosphorylation states in CCM mice and their WT littermates. (A) The phosphorylation of p42/44 ERK was rhythmic in WT mice (
Disruption of Clock in the Heart Dampened the Daily Oscillations of Ventricular Bmal1 and Abolishes Those for VGCCα1D
While heart rates of CCM and WT mice have similar circadian oscillation patterns, overall heart rates of CCM mice are slower than the WT throughout the entire day (Bray et al., 2008). The L-type voltage-gated calcium channel (L-VGCC) consists of a pore-forming α1 subunit and regulatory β and α2δ subunits (Takahashi et al., 1987; Wang et al., 2004). Among major L-VGCCα1 subunits, VGCCα1C (CaV1.2) and α1D (CaV1.3) are the most prevalent in the heart and play important roles in cardiac physiology. In both humans and mice, the α1D subunit is expressed in both ventricles and atria but at lower quantities compared to the α1C subunit (Gaborit et al., 2007; Zhang et al., 2005).
Previously, we showed that in chick embryonic hearts, the mRNA and protein levels of both VGCCα1C and α1D are under circadian control (Ko et al., 2010). However, in rats, there is no diurnal rhythm of VGCCα1C protein expression, even though the L-VGCC current density recorded from ventricular cardiomyocytes is rhythmic (Collins and Rodrigo, 2010). Hence, we set forth to investigate whether VGCCα1 subunits were rhythmic in mouse hearts. The mRNA levels and protein expression of VGCCα1C remained constant throughout the day in both WT and CCM mouse ventricles (Fig. 3A and 3B), while the mRNA levels and protein expression of VGCCα1D displayed daily rhythms in WT mice (Fig. 3C and 3D). The protein expression of VGCCα1D peaked at ZT 6, with an approximately 6-hour delay from the peak mRNA level in WT mice (Fig. 3D). The daily rhythms in VGCCα1D mRNA and protein levels were abolished in CCM mice (Fig. 3C and 3D). In addition,
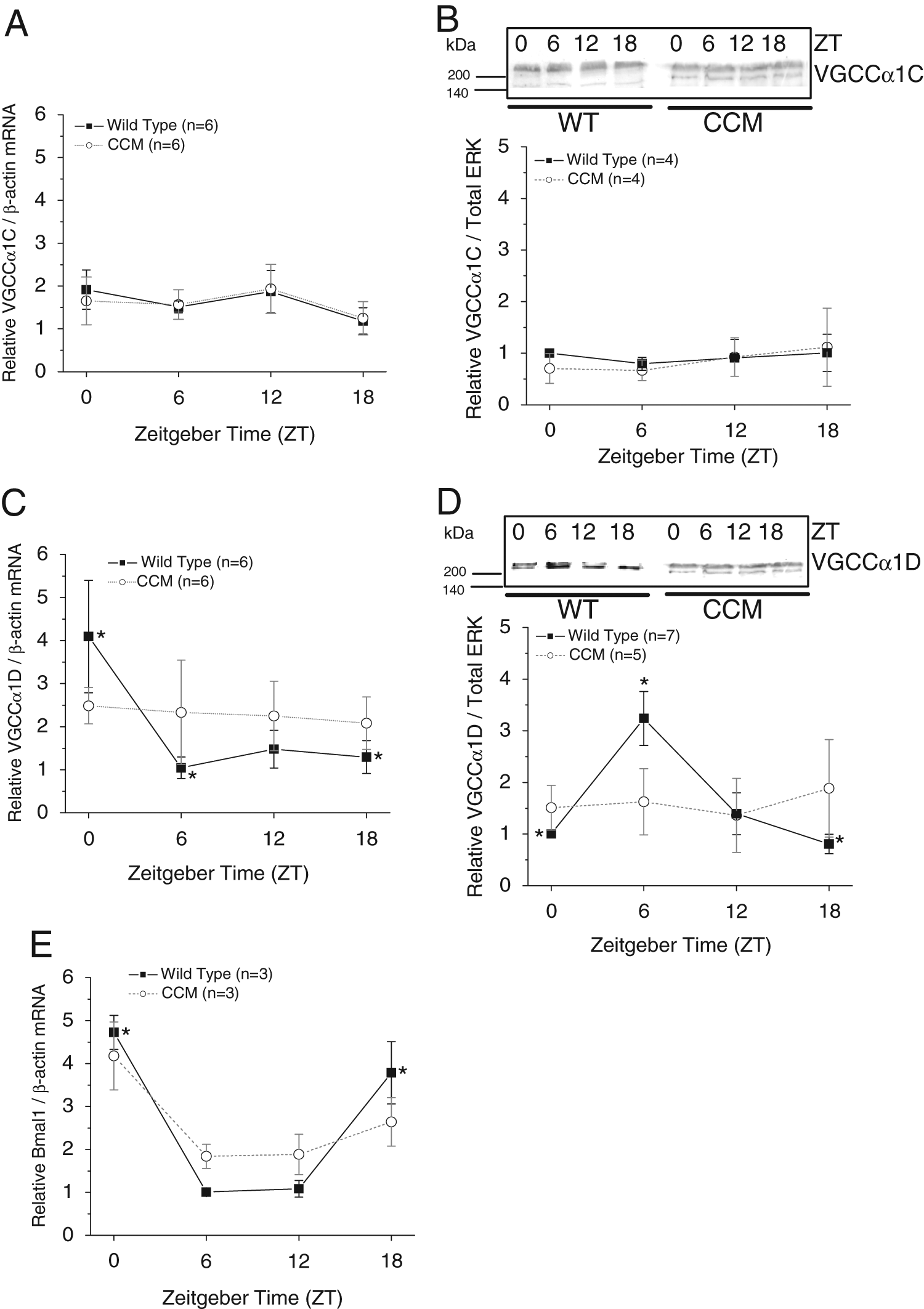
The daily profiles of
Discussion
There are intrinsic circadian changes in cardiac metabolism (Bray et al., 2008; Durgan and Young, 2010; Young et al., 2001), contractile function (Young et al., 2001), and expression of circadian clock genes (Storch et al., 2002; Young, 2003; Young et al., 2001) that are independent of autonomic inputs. Cardiomyocyte responses to starvation or fatty acid overload are also under the control of intrinsic circadian oscillators (Durgan et al., 2006; Stavinoha et al., 2004; Young et al., 2001). Disruption of cardiac circadian oscillators alters cardiac response to insults (Durgan et al., 2010; Virag et al., 2010). Therefore, the intrinsic clocks in cardiomyocytes play critical roles in preparing the heart to anticipate daily workload and synchronizing cardiac metabolism and responses to the environment (Durgan et al., 2005; Young, 2006).
There is no difference between WT and CCM mice in quantitative measurements of daily rhythms in routine activities (such as foraging, grooming, sleeping, and walking) using radiotelemetric infrared beam detection (Bray et al., 2008), and the diurnal rhythms of wheel-running activities were also similar between CCM and WT mice (Fig. 1A). These observations demonstrate that the light information from the retina to the SCN and the subsequent circadian entrainment input from the SCN are intact in CCM mice. However, we observed that CCM mice displayed less overall quantitative physical activity. The mice used in the behavior study were 20 to 28 weeks old, with no difference in body weight between WT and CCM mice (data not shown). Thus, neither age nor obesity was a contributing factor to the decrease in physical activity in CCM mice. One possible explanation is that the
Both ERK and PI3K-AKT signaling pathways are important in physiological and pathological states of cardiomyocytes, and in most cases, these pathways regulate cardiac function independently. However, one potential converging point is through phosphorylation of GSK3 because activation of ERK by endothelin and activation of PI3K-AKT through insulin can both lead to phosphorylation of GSK3 (Gonzalez et al., 2007). Therefore, the phosphorylation state of GSK3 could potentially be affected by either PI3K-AKT or ERK or both. These 2 pathways also play important roles in regulating circadian rhythms in various species and organs. p42/44 ERK, a kinase in the MAPK-Erk family, is particularly involved in the circadian entrainment of the mammalian SCN (Butcher et al., 2002; Obrietan et al., 1998) but serves in the circadian output in the avian retina and pineal gland (Ko et al., 2001; Ko et al., 2009b; Yadav et al., 2003). Another MAP kinase, p38, participates in light-dependent phase shifting in
Because the studies of these signaling pathways in circadian inputs/outputs were done in different organs or species, we took advantage of using CCM mice as a tool to access the roles of PI3K-AKT and ERK as circadian input/output pathways. We found that the phosphorylation states of pERK, p38, pAKTthr308, and pGSK were rhythmic in WT mice kept in LD cycles, while they seemed to be suspended at resting period levels in CCM mice kept in LD. The “suspension” of the daily oscillation of these kinase activities correlates to the lower wheel-running activities in CCM mice. Thus, it is possible that the cardiac-specific
Calcium that enters the cardiomyocyte via L-VGCCs triggers a more substantial Ca2+ release from the sarcoplasmic reticulum (Altamirano and Bers, 2007). This Ca2+-induced Ca2+ release in cardiomyocytes underlies the control of cardiac contraction force during excitation-contraction (E-C) coupling (Altamirano and Bers, 2007; Kranias and Bers, 2007). Therefore, L-VGCCs affect E-C coupling, contractile force, and cardiac output in adult animals (Fauconnier et al., 2003; Kubalova, 2003). Previously, Collins and Rodrigo (2010) found that there are diurnal variations in all parameters of E-C coupling as well as L-VGCC currents recorded from rat ventricular cardiomyocytes. However, it is perplexing as to why the L-VGCC current density is higher during the active period, while other E-C coupling parameters including diastolic Ca2+, basal systolic Ca2+, contraction strength, and cell shortening are higher during the resting period. In chicken embryonic hearts, mRNA and protein levels of L-VGCCs (both α1C and α1D) as well as L-VGCC currents are under circadian control, which are higher at night (Ko et al., 2010). Here, we showed that the protein level of L-VGCCα1D, but not α1C, was high during the day (resting period) in WT mice, while in CCM mice, the VGCCα1D protein level remained at a lower constant level throughout the day. We postulate that there could be a species-dependent regulation of the circadian rhythm of L-VGCCα1 subunits and their potential physiological roles, which will require future studies. Taken together, using CCM mice as an animal model, we were able to dissect the circadian light-sensitive input and output pathways in cardiomyocytes. More importantly, we demonstrated that without functional peripheral oscillators, the organism’s health could be under great distress.
Footnotes
Acknowledgements
This work was supported in part by a start-up fund from Texas A&M University and NIH RO1 EY017452 to G.Y.K., NIH PO1 NS39546 to D.J.E., and NIH RO1 HL-074259 to M.E.Y.
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
