Abstract
Trivalent lanthanide ions are known for their ability to interact with calcium-binding sites in various proteins. There is a need to assess the bioavailability of lanthanides and other heavy metals introduced into the body as components of implants or as contrast agents. This study aimed to develop a method to address bioavailability and/or presence of trivalent lanthanide ions by examining electrophoretic mobility in an agarose gel of a plasmid harboring the human metallothionein-II gene (hMT-II). Mobility of the plasmid was specifically altered by a monoclonal antibody raised against the zinc-binding transcription factor that controls the activity of the hMT-II gene. This study showed that the plasmid acquired a lanthanide-specific mobility pattern that allowed the presence of lanthanide ions to be readily determined in a 0.8% agarose gel. These findings suggest that this plasmid/monoclonal antibody combination under selected conditions may be useful in industrial, environmental, and biomedical settings to identify, separate, or capture lanthanide ions in complex mixtures that contain an array of metal ions.
Introduction
Heavy metals may negatively impact living systems by interfering with normal metabolic and physiologic mechanisms (Pudpong and Chantansi, 2015; Balali-Mood et al., 2021). DNA segments have been produced that function as sensors for specific heavy metals (Zhou et al., 2017). Most DNA-based biosensors require extensive DNA manipulation to generate the metal specificity and may be expensive.
Specifically, lanthanides are used in dental devices, orthopedic surgical appliances (screws, pins) as well as anticancer treatment, photodynamic therapy, metastatic bone pain palliation in cancer, hypophosphatemia treatment during renal dialysis, and in magnetic resonance imaging (Alavi, et al., 2015; Sharma et al., 2017; Aime and Caravan, 2009). Lanthanides share the ability to replace calcium ions bound to biomolecules in aqueous environments. Investigators have recently discovered lanthanide-specific binding proteins that require lanthanide binding to accomplish their biological functions (Cotruvo et al., 2018).
Metallothionein genes encode anti-heavy metal metallothionein proteins (Heuchel et al., 1994). The metal-responsive transcription factor-1 (MTF-1 protein) is a major hMT-II gene activator that binds to DNA sequences known as metal response elements (MRE) (Stuart et al., 1985). Therefore, the MTF-1 protein was an ideal target for a monoclonal antibody.
This study investigated the effects of a trivalent lanthanide ion on the electrophoretic mobility of a plasmid containing the human metallothionein-II (hMT-II) gene. The plasmid was targeted by the MRE-binding MTF-1 protein, which in turn could be targeted by the anti-MTF-1 protein monoclonal antibody, forming a visibly reactive complex at or near physiological pH. Electrophoretic mobility of the plasmid was influenced by different levels of supercoiled and relaxed plasmid conformations, depending upon the interactions with the monoclonal antibody, the transcription factor (MTF-1 protein), and metal ions. The supercoiled form of the plasmid is more compact and migrates faster through the agarose gel than the relaxed form.
Materials and methods
Lanthanum chloride (LaCl3; anhydrous beads, ≥99.99%), zinc sulfate solution (2.0 M ZnSO4 in BioUltra molecular biology grade water), anhydrous cobalt chloride (≥99.99%), and anhydrous nickel chloride (≥99.99%) were purchased from Sigma (Milwaukee, WI). Tris-base was obtained from EMD Millipore Corporation (Burlington, MA). An Invitrogen E-gel Power Snap Electrophoresis device and Invitrogen E-gel NGS, agarose gels (0.8% agarose) were purchased from ThermoFisher Scientific (Waltham, MA).
The pUC57 plasmid was obtained from GenScript (Piscataway, NJ). The entire gene for human metallothionein-II was cloned into the pUC57 plasmid, resulting in a new plasmid containing the full length 837-bp human MT-II gene. The human MT-II gene was cloned into the pUC57 plasmid between the BamH1 and EcoRV restriction endonuclease sites within the multiple cloning regions. The size of the predominantly supercoiled plasmid construct (pUC57-MT2) was 3547 bp.
Mouse anti-human MTF-1 (H-6) monoclonal antibody (200 µg IgG/mL) was purchased from Santa Cruz Biotechnology (Dallas, TX). This antibody targeted amino acid residues 454-753 within the carboxy-terminus of the human MTF-1 protein. All antibody treated samples were incubated on ice for 20 min with 0.2 µg (200 ng) of monoclonal anti-MTF-1 antibody. The reaction buffer was 100 mM Tris-HCl (pH 8.3).
Pre-cast 0.8% agarose gels were used for the 26-minute electrophoresis procedures. Each gel contained the fluorescent SYBR-Safe DNA gel stain, which allowed monitoring of plasmid DNA bands in real time under ultraviolet illumination, at room temperature. DNA size markers were run in parallel in each gel. Each size marker consisted of the E-Gel 1-kB Plus DNA ladder (Invitrogen, ThermoFisher Scientific).
Results
Agarose gel electrophoresis showed the effects of the trivalent lanthanum ion on the monoclonal antibody-associated electrophoretic mobility of the pUC57-MT2 plasmid (Figure 1). Lane 2 shows the typical migration pattern of the pUC57-MT2 plasmid alone. Lane 3 shows the effect of anti-MTF-1 monoclonal antibody on the pUC57-MT2 plasmid (maintaining a predominantly supercoiled conformation) in the absence of metal ion. Lanes 4 and 5 reveal the distinct zinc ion-dependent pUC57-MT2 plasmid migration pattern when Zn2+ ions are present with the anti-MTF-1 monoclonal antibody. Lanes 6 and 7 show the effects of adding MTF-1 protein in the presence of both Zn2+ ions and anti-MTF-1 antibody. The typical Zn2+-induced pattern was used as an internal control to assure that all components were functioning properly. The zinc-induced or zinc plus monoclonal antibody-induced migration patterns were likely the results of less plasmid being in the supercoiled conformation, causing the relaxed form of the plasmid to migrate slower in the gel matrix. Comparison of effects of 1 mM ZnSO4 and 1 mM LaCl3 on monoclonal antibody-assisted electrophoretic migration of pUC57-MT2 plasmid in the presence/absence of the MTF-1 protein. Lane 1: 1 Kb DNA molecular weight standard ladder. Lane 2: pUC57-MT2 plasmid alone. Lane 3: pUC57-MT2 plasmid + mAb. Lanes 4 and 5: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4. Lanes 6 and 7: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + MTF-1 protein. Lanes 8 and 9: pUC57-MT2 plasmid + mAb + 1 mM LaCl3. Lanes 10 and 11: pUC57-MT2 plasmid + mAb + 1 mM LaCl3 + MTF-1 Protein.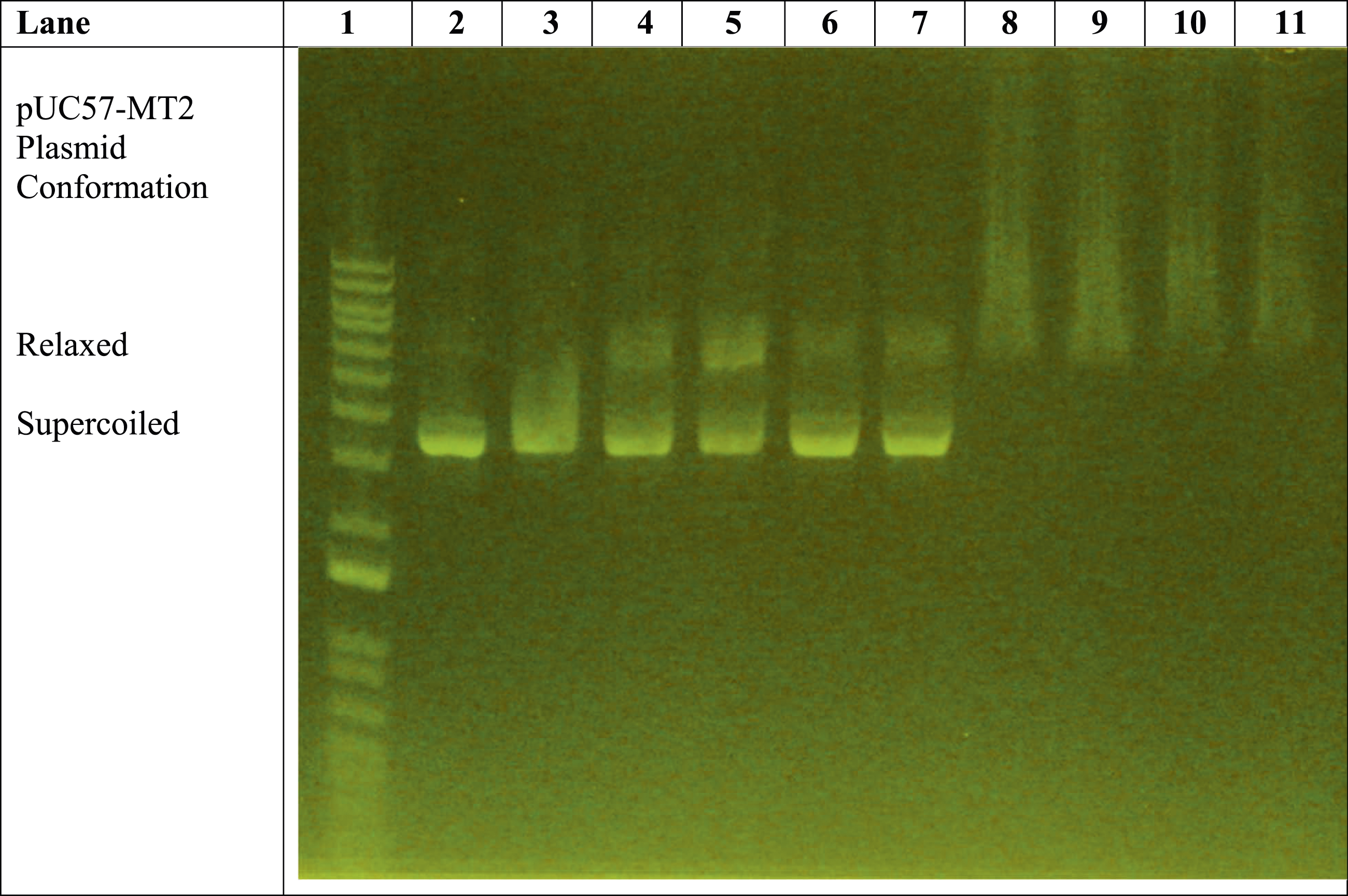
The remaining lanes (i.e., Lanes 8–11) in Figure 1 reveal the effect of the presence of trivalent La3+ ions on the pUC57-MT2 plasmid. Lanes 8 and 9 illustrate the La3+-induced pUC57-MT2 plasmid electrophoretic migration pattern in the presence of the monoclonal antibody. In contrast, Lanes 10 and 11 show the effect of the MTF-1 protein with the La3+ and anti-MTF-1 monoclonal antibody. This resulted in a La3+-specific plasmid mobility pattern. The results of additional experiments with zinc, cobalt, and nickel further demonstrate the ability of trivalent lanthanum to alter the electrophoretic mobility of the plasmid in a lanthanum-specific manner (Figures 2–4). Comparison of effects of 1 mM ZnSO4 with 0.1-1 mM LaCl3 on monoclonal antibody-assisted electrophoretic migration of pUC57-MT plasmid without the MTF-1 protein. Lane 1: 1 Kb DNA molecular weight standard ladder. Lane 2: pUC57-MT2 plasmid alone. Lane 3: pUC57-MT2 plasmid + mAb. Lanes 4 and 5: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4. Lane 6: pUC57-MT2 plasmid + mAb + 0.1 mM LaCl3. Lane 7: pUC57-MT2 plasmid + mAb + 0.2 mM LaCl3. Lane 8: pUC57-MT2 plasmid + mAb + 0.4 mM LaCl3. Lane 9: pUC57-MT2 plasmid + mAb + 0.6 mM LaCl3. Lane 10: pUC57-MT2 plasmid + mAb + 0.5 mM LaCl3. Lane 11: pUC57-MT2 plasmid + mAb + 1 mM LaCl3. Zinc versus lanthanum ion competition assay. Lane 1: 1 Kb molecular weight standard DNA ladder. Lane 2: pUC57-MT2 plasmid alone. Lane 3: pUC57-MT2 plasmid + mAb. Lane 4: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4. Lane 5: pUC57-MT2 plasmid + mAb + 1 mM LaCl3. Lane 6: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + 0.1 mM LaCl3. Lane 7: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + 0.2 mM LaCl3. Lane 8: pUC57-MT2 plasmid + mAb +1 mM ZnSO4 + 0.4 mM LaCl3. Lane 9: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + 0.6 mM LaCl3. Lane 10: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + 0.8 mM LaCl3. Lane 11: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4 + 1 mM LaCl3. Zinc, cobalt, and nickel versus lanthanum ion competition assay. Lane 1: 1 Kb molecular weight standard DNA ladder. Lane 2: pUC57-MT2 plasmid alone. Lane 3: pUC57-MT2 plasmid + mAb. Lane 4: pUC57-MT2 plasmid + mAb + 1 mM ZnSO4. Lanes 5 and 6: pUC57-MT2 plasmid + mAb + 1 mM NiCl2. Lanes 7 and 8: pUC57-MT2 plasmid + mAb + 1 mM CoCl2. Lane 9: pUC57-MT2 plasmid + mAb + 1 mM LaCl3. Lanes 10 and 11: pUC57-MT2 plasmid + mAb + 1 mM LaCl3 + 0.5 mM NiCl2 + 0.5 mM CoCl2 + 0.5 mM ZnSO4.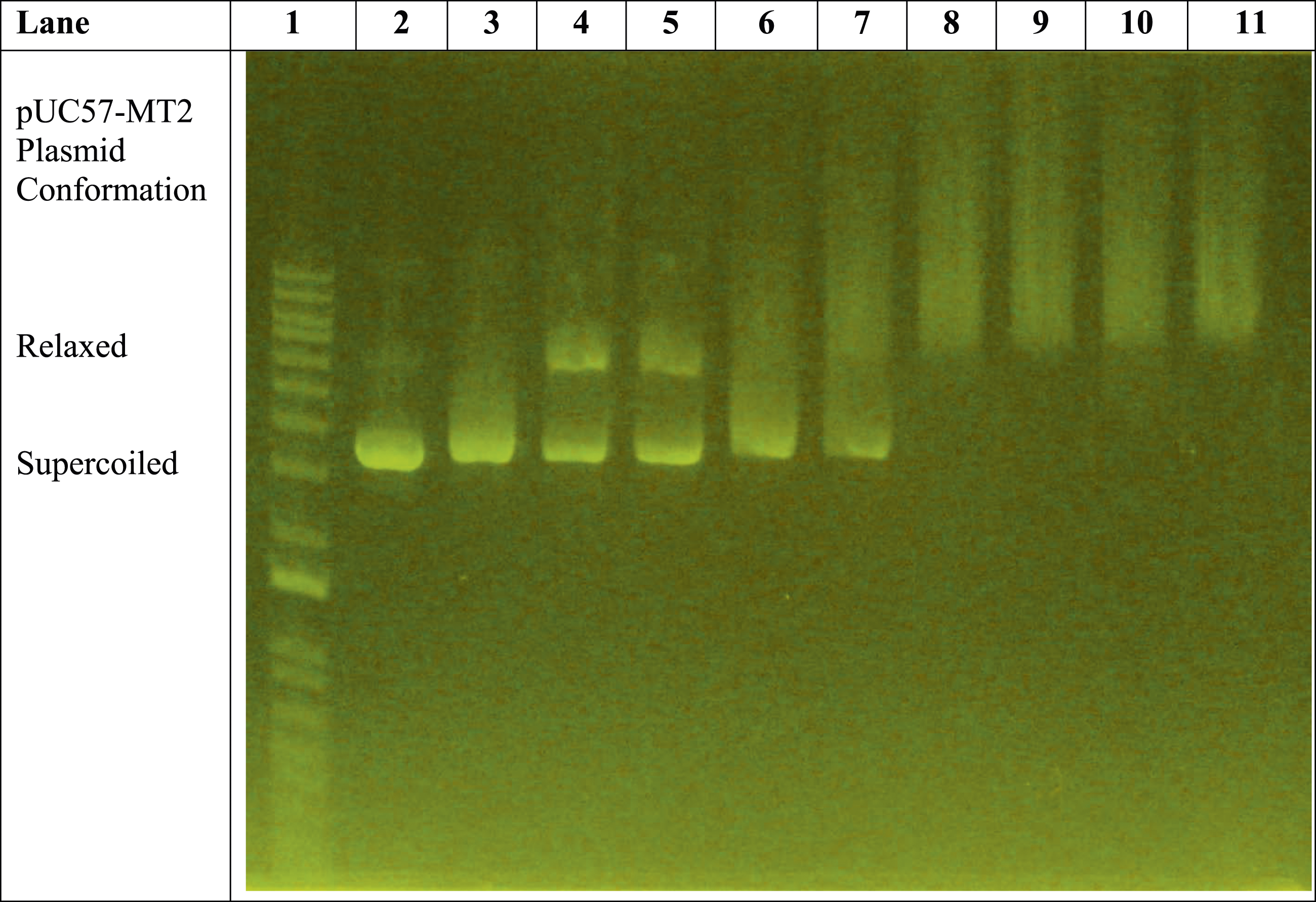
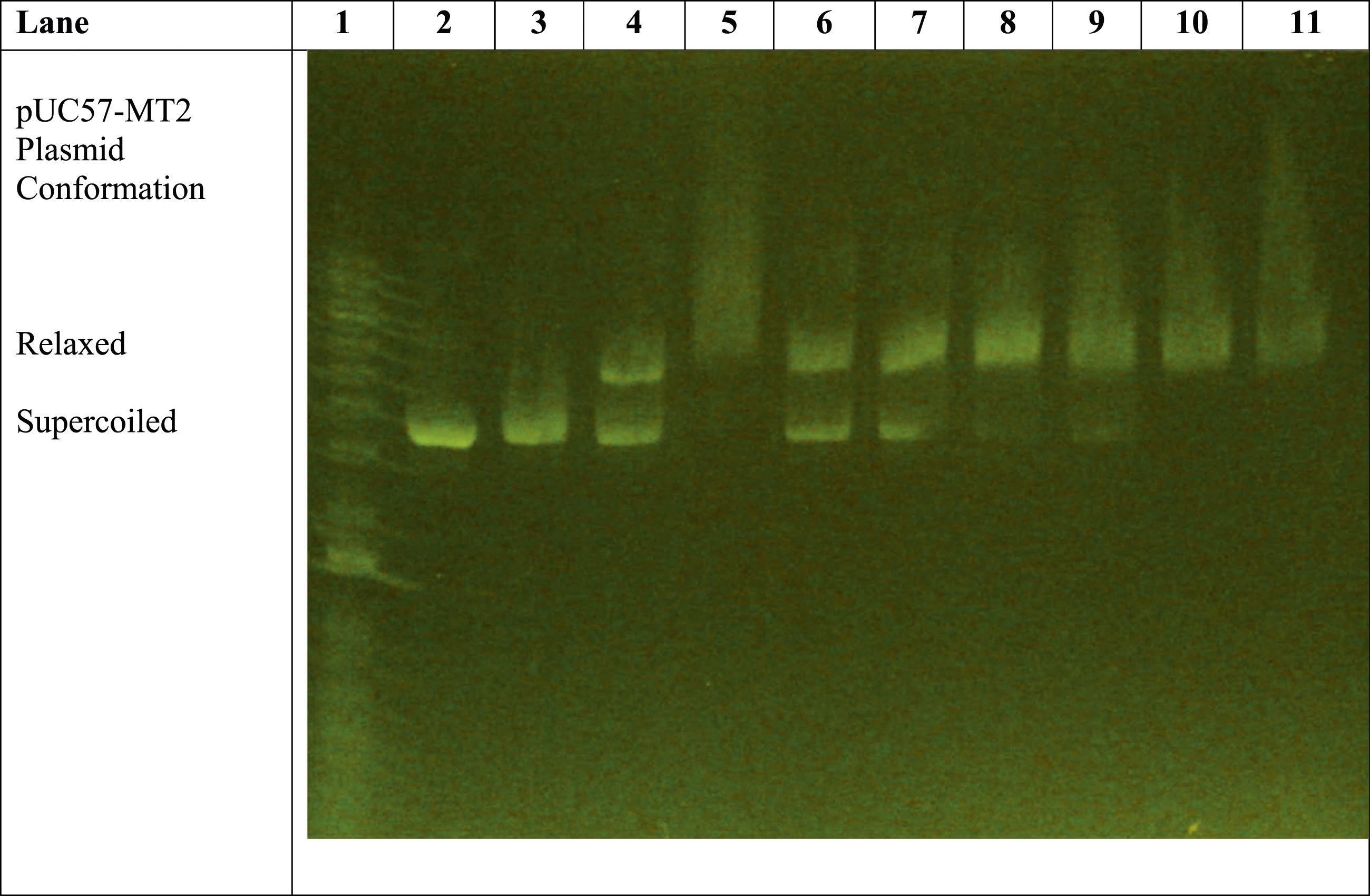
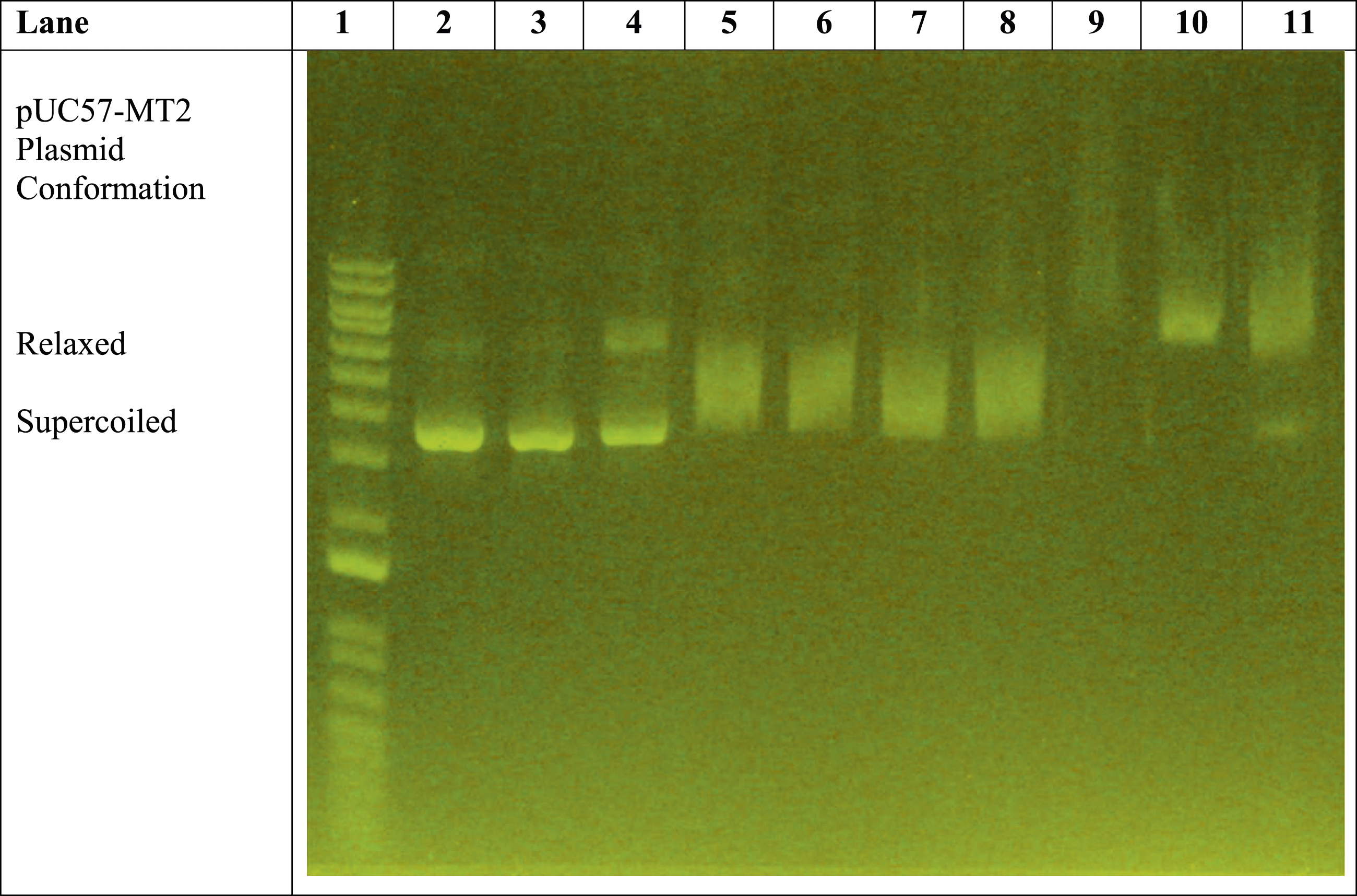
Figure 2 shows a
Figure 3 illustrates the zinc versus lanthanum ion competition assay. Lanes 2 through 5 are essentially internal controls that assure that all components are functioning properly. Lanes 6 through 11 reveal the Zn2+ versus La3+ competition assay with reactions containing 1 mM ZnSO4 and varying concentrations of LaCl3. The results indicate that LaCl3 at 0.4 mM nearly completely converted the plasmid to the relaxed conformation, even in the presence of 1 mM ZnSO4.
Figure 4 is a different version of the divalent cation versus lanthanum ion competition assay. Three divalent heavy metal ions are tested against LaCl3 simultaneously.
Discussion
The ability of a polyclonal antibody to induce metal-specific changes in electrophoretic mobility of a plasmid containing the human metallothionein-II gene was reported previously (Wooten et al., 2016). When considering the effect of ZnSO4, there are some significant differences between the monoclonal antibody used in the current study and the polyclonal antibody of 2016 (Wooten et al., 2016).
The influence of Zn2+ ions on the mobility of the pUC57-MT2 plasmid in the presence of the anti-MTF-1 monoclonal antibody was highly distinctive (Figure 1). When the plasmid was run alone, a highly reproducible pattern emerged, showing very little shifting from a supercoiled to a relaxed conformation. When the plasmid was mixed with anti-MTF-1 monoclonal antibody, another highly reproducible shift pattern was generated (Figure 1, Lane 3), resulting in a significantly more diffuse mobility corresponding to a modest transition from supercoiled to relaxed conformation. When the plasmid was mixed with the anti-MTF-1 monoclonal antibody and 1 mM ZnSO4, a characteristic electrophoretic shift occurred (Figure 1, Lanes 4 and 5), resulting in a distinct binary band pattern.
The effect of 1 mM LaCl3 on the pUC57-MT2 plasmid mobility reveals a significant shift from supercoiled to relaxed conformation (Figure 1, lanes 8–11). Lanes 8 and 9 show the effect of adding LaCl3 to the mixture of plasmid and the anti-MTF-1 monoclonal antibody. There is a complete upward shift in plasmid mobility from a supercoiled to relaxed conformation. The MTF-1 protein caused a modest enhancement of the upward shift in plasmid mobility (Figure 1, lanes 10–11) in the presence of 1 mM LaCl3. The DNA binding domain of MTF-1 protein is located near the N-terminal end of the MTF-1 protein’s amino acid sequence. The region near the N-terminus contains six zinc ion-binding zinc finger structures that participate in DNA binding (Radtke et al., 1995). The anti-MTF-1 monoclonal antibody was raised against the carboxy terminus of the human MTF-1 protein (Gierdroc et al., 2001).
To further characterize the influence of LaCl3 on the electrophoretic mobility of the pUC57-MT2 plasmid, a series of increasing concentrations of LaCl3 was utilized (Figure 2). The influence of 1 mM ZnSO4 on the mobility of the pUC57-MT2 plasmid is clearly illustrated in lanes 4 and 5 versus lanes 2 and 3. Lanes 6 and 7 reveal the effect of 0.1 mM LaCl3 and 0.2 mM LaCl3 on the pUC57-MT2 plasmid’s mobility. Even at 0.1 mM and 0.2 mM concentrations, the effect of LaCl3 on plasmid mobility is distinctly different than that of 1 mM ZnSO4. The plasmid fully shifted from supercoiled conformation (Lane 2) when the LaCl3 concentration reached 0.4 mM (Lane 8).
Since most lanthanides are trivalent with divalent-like ionic radii, they are likely to have higher binding affinities for plasmid-binding sites than divalent ions like zinc, cobalt, and nickel (Figures 3 and 4). Thus, lanthanide binding in an aqueous solution could be assessed by monitoring initial Zn2+ binding, followed by Zinc ion displacement.
Several investigations have shown how lanthanides bind to DNA and cause closed circular DNA molecules to form supercoiled or relaxed conformations in aqueous solutions (Chen et al., 2013; Gibson et al., 2020). Additionally, some antibodies bind specifically to certain forms of DNA, such as antibodies that target negatively supercoiled plasmids, which contain left-handed Z-DNA segments (Nordheim et al., 1982).
Figure 4 (lanes 5 and 6 and lanes 7 and 8) shows that cobalt and nickel ion-induced plasmid mobility patterns are distinct from those of the zinc ion (lane 4) and lanthanum ion (lane 9). Furthermore, when divalent cations such as cobalt, zinc and nickel are present in a combined higher concentration than the lanthanum ion, trivalent lanthanum still induces a lanthanum ion-specific plasmid mobility pattern (Figure 4, lanes 10 and 11).
At least one other plasmid has been shown to display mobility shifts in 0.8% agarose gels when lanthanides were present (Jastrzab et al., 2019). Several different lanthanides were shown to induce relaxed and supercoiled conformations in plasmids in one-dimensional agarose gel electrophoresis, with each lanthanide eliciting non-specific, instead of metal-specific, electrophoretic mobility shifts of the pUC19 plasmid (Jastrzab et al., 2019).
Conclusions
This study reports upon the novel use of an anti-MTF-1 monoclonal antibody and the pUC57-MT2 plasmid to generate metal-specific agarose gel mobility patterns that can be used to clearly identify and separate trivalent lanthanum ions from mixtures of divalent metal ions. This report also demonstrates a new method of assessing the bioavailability of lanthanides and possibly other heavy metals without a need for cell cultures or complex analytical equipment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by an intramural grant provided by the Sam Houston State University College of Osteopathic Medicine.
