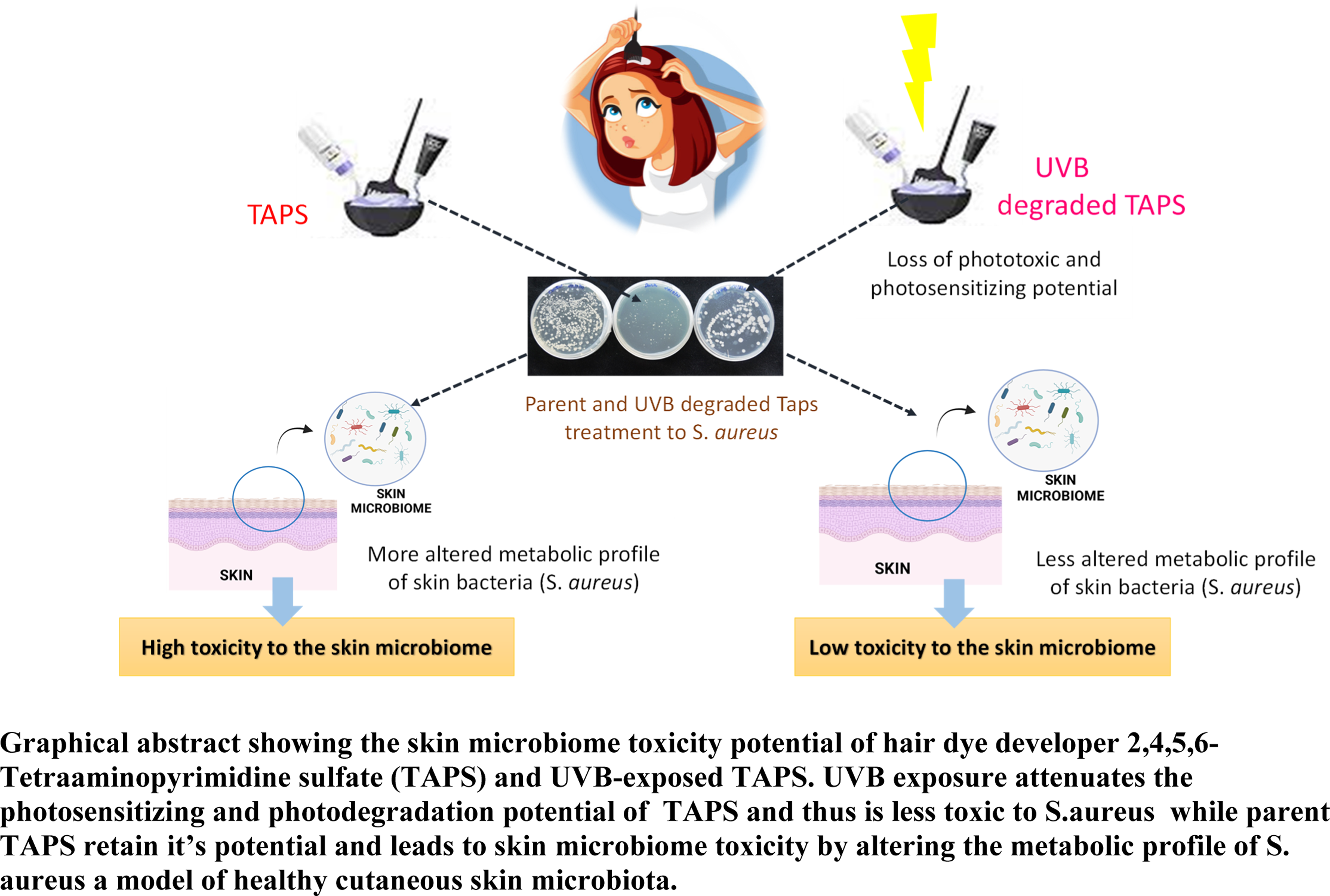
Abstract
Synthetic cosmetics, particularly hair dyes, are becoming increasingly popular among people of all ages and genders. 2,4,5,6-tetraaminopyrimidine sulfate (TAPS) is a key component of oxidative hair dyes and is used as a developer in several hair dyes. TAPS has previously been shown to absorb UVB strongly and degrade in a time-dependent manner, causing phototoxicity in human skin cells. However, the toxic effects of UVB-degraded TAPS are not explored in comparison to parent TAPS. Therefore, this research work aims to assess the toxicity of UVB-degraded TAPS than TAPS on two different test systems, that is, HaCaT (mammalian cell) and
Introduction
The use of hair color to style hair has become an increasing fashion trend in today’s population. Cosmetic industries are more focused on developing hair dyes that are resistant to multiple washes and have long-lasting effects. Among all the hair dye types, permanent or oxidative hair dyes contribute more than 80% worldwide (Platzek, 2010). Oxidizing or permanent hair dyes contain oxidizing as well as alkylating agents as ingredients. The major ingredient, which is frequently used in oxidizing hair dye, is 2,4,5,6-tetraaminopyrimidine sulfate (TAPS). Tetraaminopyrimidine salts are used in hair color as a main intermediate component, which reacts with a coupler to give the final color to hair (Freeman and Peters, 2000). Research stated that the concentration of TAPS should not be beyond 3.4% (Sukakul et al., 2018). It is a class of aromatic amino compounds that are used in the manufacture of hair dye that gives vivid red color to the hair and upon absorption of sunlight shows phototoxic effects. Oxidative hair dye ingredients are known as a potent carcinogenic as well as phototoxic compounds, which generate elevated levels of ROS and cause DNA damage. Mounting evidence documented that long-term exposure to photosensitive chemicals with concurrent exposure to sunlight cause phototoxicity that appears in the form of photosensitization, genotoxicity, phototoxicity/photoirritation skin inflammation/irritation, and cancer (Mujtaba et al., 2021). Our earlier study reported phototoxicity and their molecular mechanism under exposure to UVR where we illustrated the role of TAPS as a photosensitizing agent that may cause dermal toxicity in chronic exposure (Shukla et al., 2022). However, earlier research was restricted only to the safety study of TAPS but did not consider the safety profiling of photodegraded TAPS that formed after UVB irradiation of TAPS. Our previous study demonstrated that the photoproducts generated after the degradation of anthracene are more phototoxic than parent molecules (Mujtaba et al., 2013). Because TAPS remains on the skin for a longer period during hair dyeing, it is important to understand the fate of UVB-degraded TAPS on human skin as well as on skin microbiomes. Under the above background this study has been designed to assess the safety of UVB-degraded TAPS than parent TAPS in two different experimental systems, that is, human keratinocyte cell and
Materials and methods
Chemicals and culture wares
2,4,5,6-tetraaminopyrimidine sulfate (TAPS), Hank's Balanced Salt Solution (HBSS), antibiotic and antimycotic solution, Dulbecco's Modified Eagle's Medium/Nutrient Mixture F-12 HAM (DMEM), chlorpromazine (CPZ), dimethyl sulfoxide (DMSO), nutrient agar/broth (HiMedia), and fetal bovine serum (FBS) were purchased from Gibco, Life Technologies Ltd., UK. PM1-4 and nine MicroPlateTM, IF-0a GN/GP, IF-10b GN/GP, and Biolog edox dye were purchased from Biolog, Hayward, CA, USA. Plastic and culture wares were purchased from Nunc, Denmark.
Radiation source system
1.2 m long UVR emitting tubes (VilberLourmat, France) were used as UV irradiation sources. The UVB intensity has been measured using a microprocessor-controlled RMX-3W radiometer (VilberLourmat, France). Calibrated UVB-detecting probe with a peak at 312 nm was used in the setup. UVB intensity was chosen after a year of dosimetry experiments on our lab's rooftop during the peak hours (11:00 a.m. and 3:00 p.m.) to determine the average daily doses of radiation that would be received by an individual. The UVB intensity used in the experiment was comparable to those of natural solar UVB radiation arriving at Lucknow (26° 51′ 0′′ N latitude, 80° 57′ 0′′ E longitude) (approx. 123 m above sea level). The samples were irradiated with UVB light at a distance of 22 cm from the source tubes at a controlled temperature. The following formula was used to determine the UVR dose
Radiation dose (J/cm2) = Irradiance (mW/cm2) x Exposure time (sec)/1000
Photodegradation analysis of TAPS
Experimentation of photodegradation is carried out in UV equipped chamber at a temperature of 25 ± 2°C with irradiation distance of the sample from the source tube kept at 14 cm. The intensity of UV light was checked periodically with the RMX-3 radiometer study. TAPS was made in MilliQ water of concentration (1 mg/mL) as the main stock. For the photodegradation study, 15 μg/mL was used as a working stock. Beakers with 10 mL solution were kept in UVB for 1–4 h and one beaker was kept in the dark (without irradiation). After every 1 h, samples were taken for absorption spectra of photodegraded TAPS between 200 and 700 nm including both UV and visible spectrum region by Varian Cary 300 Bio UV-visible spectrophotometer. For degradation kinetics of TAPS, absorbance was taken in Simple reads of Cary WinUV software, and the graph was plotted in MS Excel (Chopra et al., 2016)
Photodegradation analysis of TAPS by ultra high-performance liquid chromatography
UHPLC analysis was carried out on a DIONEX Ultimate 3000 instrument (Thermo Fisher Scientific, USA) equipped with a binary pump, column compartment, and PDA detector. The separation was carried out on reverse phase Accucore C18 column (150× 4.6 mm, 2.6 μm, Thermo Fisher Scientific, USA). To study the photodegradation of TAPS, a stock of 15 μg/mL dark and UVB-exposed (1–4 h) TAPS was taken for UHPLC analysis. The chromatographic analysis was performed using a linear gradient mode of the mobile phase. The mobile phase contains 0.1% TFA in water (A) and 0.1% TFA in acetonitrile (B) with 0–6 min (5%–95% of B), 6–6.50 min (95%–5% of B), and 6.50–8 min (maintained at 5% B) at a flow rate of 0.8 mL/min. The chromatographic data were recorded at 282 nm wavelength with a constant injection volume of 20 μL for standard and sample solutions (Negi et al., 2023).
Cell culture
HaCaT (human skin keratinocytes) was procured from NCCS Pune and maintained in our cell culture facility of Photobiology. Cell lines were grown in DMEM F-12 HAM culture media with antibiotic antimycotic solution (1.5%) and heat-inactivated FBS (10%) filtered with 0.22 µm filter at 37°C, 5% CO2, and relative humidity 95%.
Photocytotoxicity assay
MTT assay
MTT assay is a colorimetric assay based on the reduction of tetrazolium bromide (yellow color) to formazan crystals (purple color) which defines mitochondrial activity. It is a sign of cell viability or cytotoxicity and measures cellular metabolic activity. Cells were seeded in 96 well plates for 70%–80% confluency at 37°C in a CO2 incubator. Media was decanted and treated with two concentrations of UVB-degraded TAPS (10 and 25 µg/ml) and parent TAPS (10 and 25 µg/ml) for 40 min at 37°C and afterward, irradiated under UVB (0.6 mW/cm2) for 1 h. After incubation for 40 min, HBSS was decanted and replaced by MTT dye (5 mg/10 mL) and plates were incubated for 4 h at 37°C in a CO2 incubator. After 4 h, media was discarded and replaced by DMSO [100 μL/well] and placed on a rocker for 10 min (Dubey et al., 2019). Absorbance was taken at 530 nm by a multi-well microplate reader (Synergy LX multi-mode reader, BioTek).
Phenotypic microarray analysis
CFU assay of S. aureus after treatment with dark TAPS and UVB-degraded TAPS
Statistical analysis
One-way analysis of variance (ANOVA) was used for the interpretation of experiments, which was compared with Dunnett's multiple comparisons test with mean ± SD. Experiments were repeated thrice and significant values were taken as the mean of three results performed independently (***
Results
Absorption spectra and photodegradation of TAPS
Photodegradation is caused by the absorption of photons by the photodegradable compound in wavelength of sunlight, UVR, and visible light. The photodegraded compound absorb light in a particular wavelength and ultimately causes excitation or breakdown into radicals. TAPS showed absorption under UVB by showing strong absorption maxima at 282 nm (dark) (Figure 1(a) ). When TAPS was exposed under UVB with the intensity of 0.6 mW/cm2 for different time intervals (1–4 h) (Figure 1(b)). The results illustrated that compound is photolabile and degraded in a time-dependent manner. Percent degradation and percent remain are also shown quantitatively by degradation analysis at 282 nm by spectrophotometer showing maximum degradation in 4 h (Figure 1(c) and (d)). Photodegradation study of TAPS (15 µg/ml). (a) Absorption spectra. (b) Photodegradation spectra under UVB (0.6 mW/cm2) for 1–4 h. (c) Degradation kinetics showing % remains of TAPS (d) % degradation kinetics of TAPS. Three independent experiments were carried out in triplicate sets with mean ± SD. [***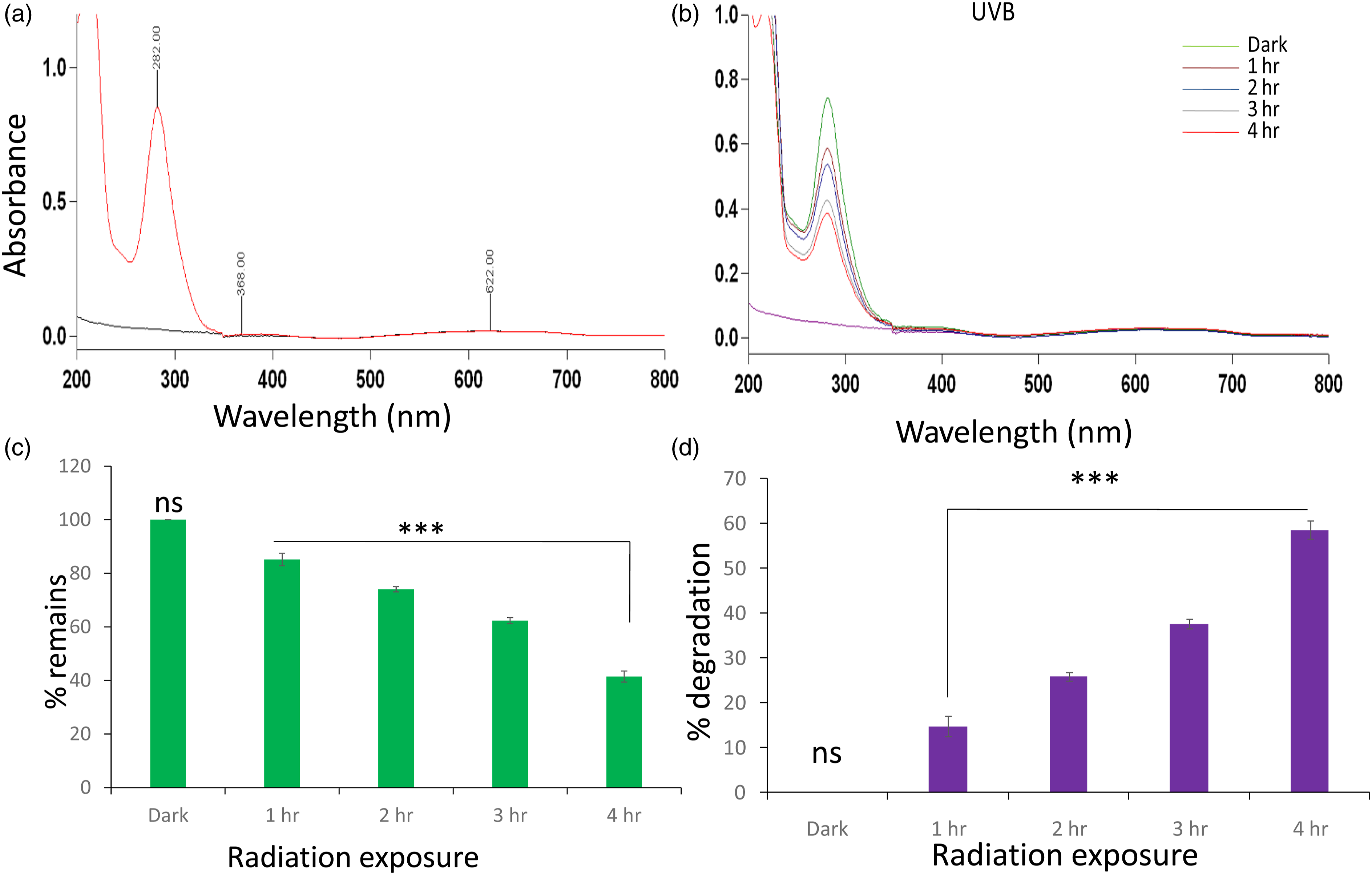
UHPLC analysis of TAPS
The photodegradation study of TAPS was also confirmed by UHPLC analysis. The chromatogram of the dark, which did not receive any irradiation, had the highest peak area (mAU = 335,626.993), whereas the UVB irradiated TAPS showed a decrease in peak area in a time-dependent manner in UVB 1 h (mAU= 333,111.576), UVB 2 h (mAU = 232,447.678), UVB 3 h (mAU= 228,922.685), and UVB 4 h (mAU= 8237.306), respectively (Figure 2(a)–2(e)). These results further validate the photodegradation of TAPS by spectrophotometry. UHPLC analysis of TAPS gets photodegraded in a time-dependent manner. (a) Chromatogram of dark, (b) UVB 1 h, (c) UVB 2 h, (d) UVB 3 h, and (e) UVB 4 h under the intensity of 0.6 mW/cm2.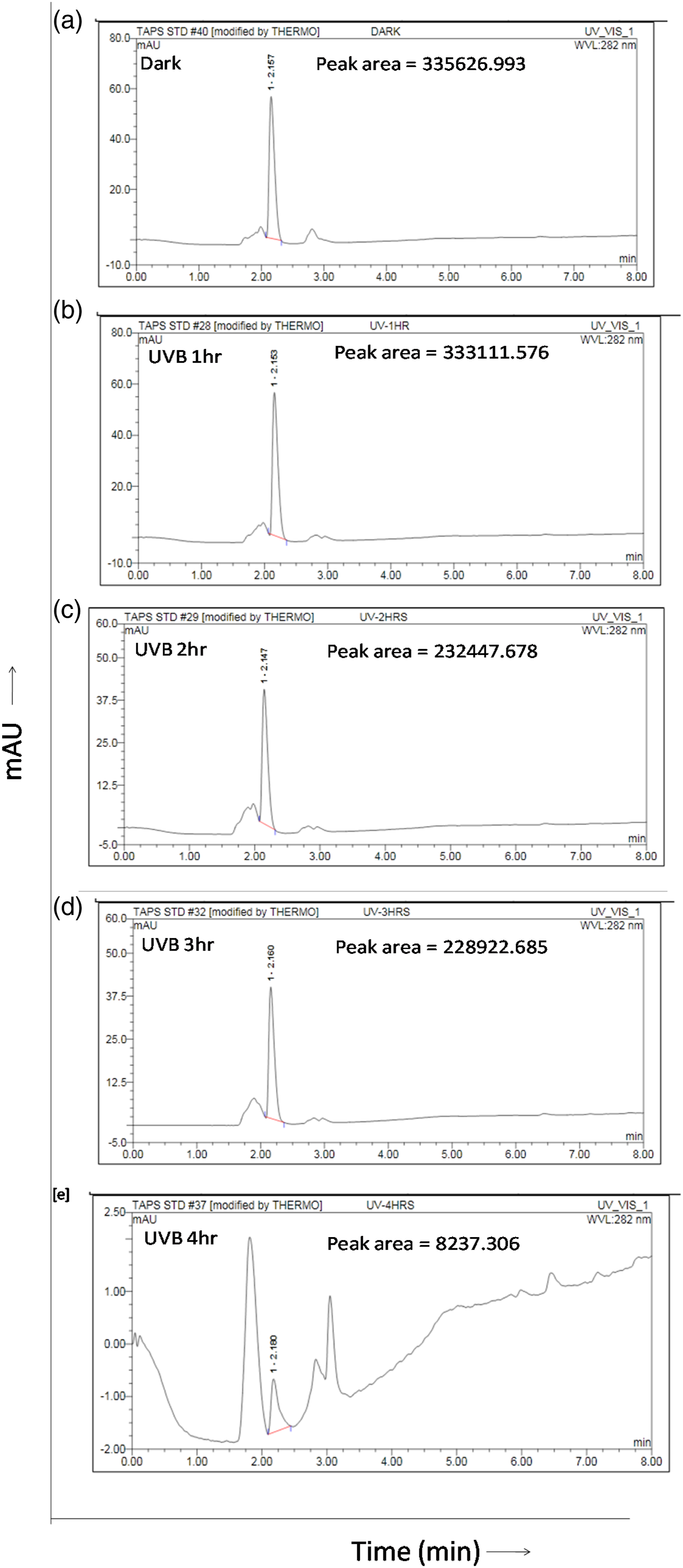
Cytotoxicity assay
MTT assay was used to determine the phototoxicity of TAPS under UVB (0.6 mW/cm2) for 1 h. Results of TAPS showed a greater reduction in cell viability at concentrations of 10 μg/mL (51.2%) and 25 μg/mL (36.7%) than UVB-degraded TAPS 10 μg/mL (72.2%) and 25 μg/mL (67.6%) under exposure of UVB radiation (Figure 3). Our results showed that parent compound is more toxic than UVB-degraded TAPS. There was no significant difference found in cell viability of dark and UVB control cells as well as dark-treated TAPS (10 and 25 μg/mL). The photocytotoxicity potential of TAPS (10 and 25 μg/mL) was assessed on HaCaT cells under UVB (0.6 mW/cm2). Mitochondrial dehydrogenase activity (MTT assay) of UVB-degraded TAPS versus parent TAPS showing a reduction in cell viability. Values are mean ± SD of three experimental results done independently. [***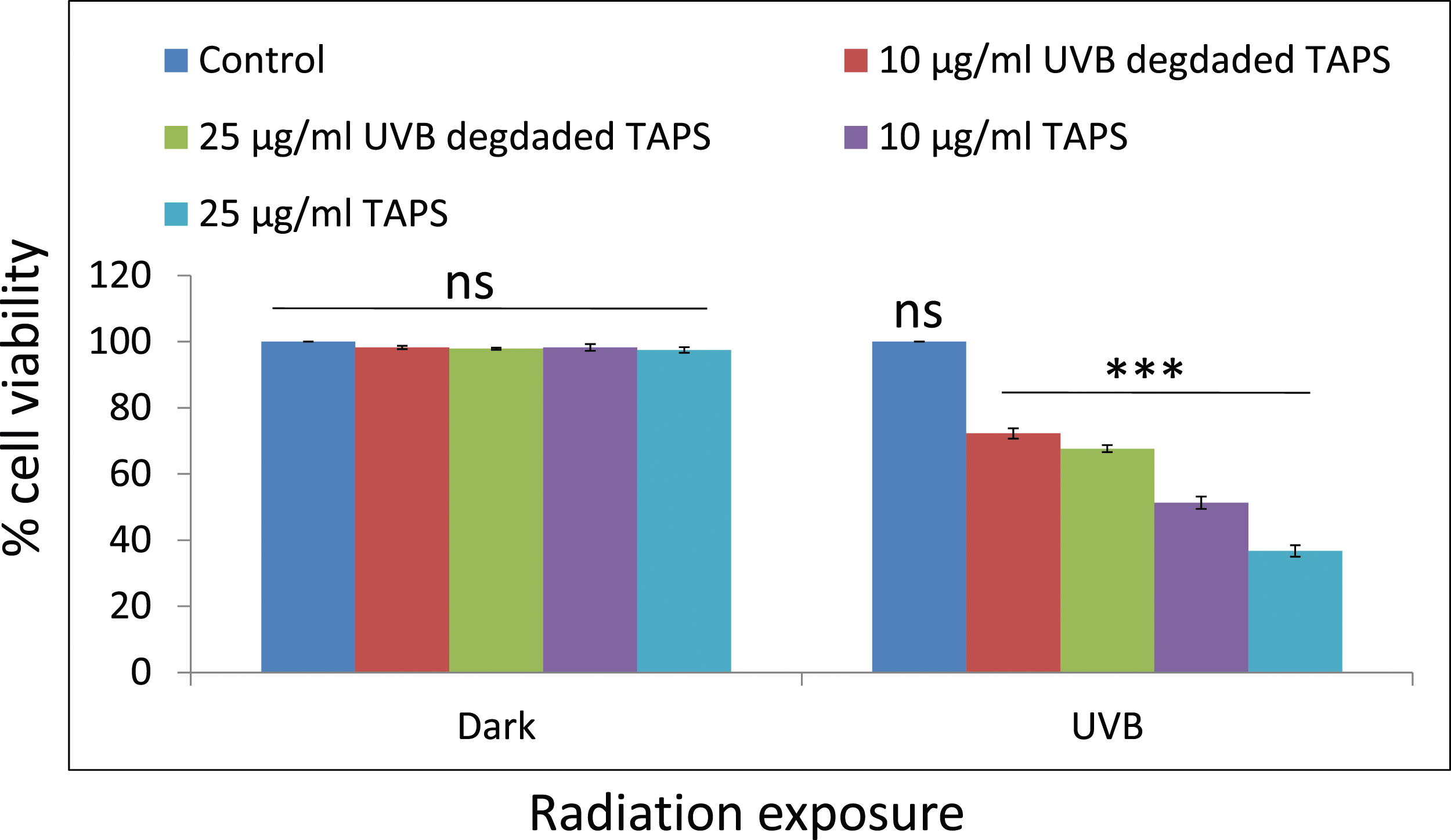
Phenotype microarray
In the present study, the untreated TAPS (15 μg/mL) under UVB-degraded TAPS (0.6 mW/cm2) irradiation for 4 h showing phenotype gained and loss of 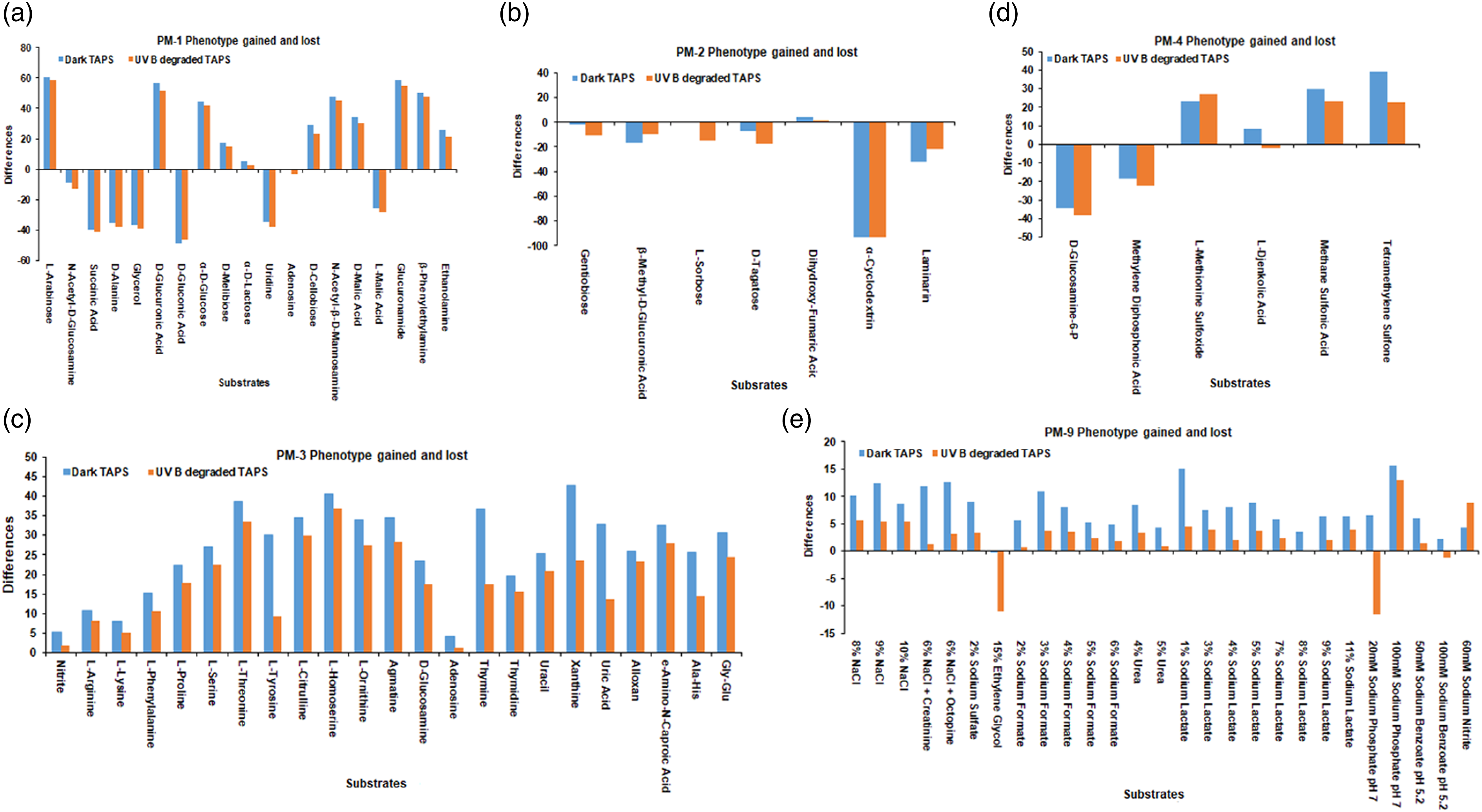
In addition, TAPS treated
Colony forming unit assay
Plate sensitivity test showing colony forming unit (CFU) in TAPS versus UVB-degraded TAPS for 4 h in 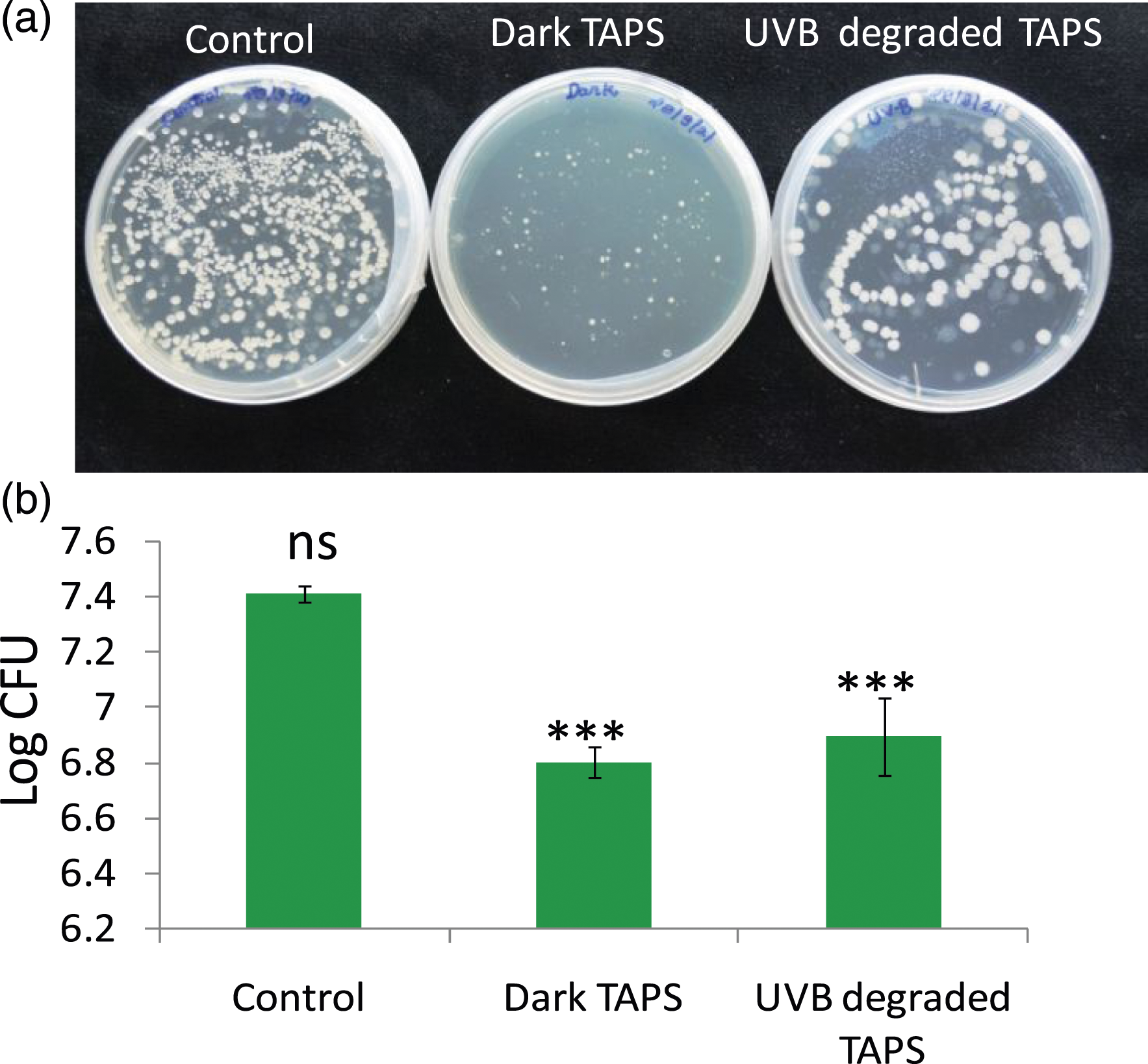
Discussion
TAPS is an essential component of permanent hair dyes that are used as a developer. Skin allergy is very common with chronic exposure to hair dyes and their ingredients (Khan et al., 2019). Our previous study reported that TAPS has strong absorption under UVB. It also induces phototoxicity via ROS generation, DNA damage, and activation of inflammatory pathways (Shukla et al., 2022). This study on TAPS established the photosensitization and phototoxicity potential of human keratinocytes. However, that study failed to discuss the safety of photoproducts formed via photodegradation of TAPS under exposure to UVB. Therefore, to understand the skin safety of UVB-degraded TAPS, a comprehensive safety study was performed on human and bacterial cell systems. Our results demonstrated that TAPS is photolabile and degrades in a time-dependent manner. Further, the safety profiling of photodegraded TAPS was performed on human keratinocytes in comparison to parent TAPS by measuring the cell viability. Cell viability results showed that UVB-degraded TAPS is less phototoxic than parent TAPS. The results suggested that the photosensitization and phototoxic potential of TAPS is attenuated after degradation under UVB exposure. Additionally, it is well known that skin microbiomes play a very important role in the protection against invaded pathogens (Wang et al., 2018) training the immune system (Eisenstein, 2020) and helping in the degradation of natural products. But as per reports, microbiomes are altered due to exposure to synthetic chemicals on the skin
Conclusion
TAPS degraded by UVB irradiation formed different groups of metabolites that had less detrimental effects on the skin microbiome. However, parent TAPS is more hazardous than UVB-degraded TAPS. Thus, the present study suggests that even though photoproducts may be less toxic than parent molecules, simultaneous exposure of UVR/sunlight should be avoided after hair coloring because light can not only bleach the dye but also promots skin damage via ROS-mediated activation of inflammatory signals.
Supplemental Material
Supplemental Material - Toxic potential assessment of hair dye developer 2,4,5,6-tetraaminopyrimidine sulfate exposed under ambient UVB radiation
Supplemental Material for Toxic potential assessment of hair dye developer 2,4,5,6-tetraaminopyrimidine sulfate exposed under ambient UVB radiation by Saumya Shukla, Priyanka Chauhan, Prakriti Gaur, Priyanka Rana, Sunil Kumar Patel, Deepti Chopra, Apeksha Vikram, Gaurav Prajapati, Akhilesh Kumar Yadav, Sumana Y Kotian, Lakshmi Bala, Ashish Dwivedi, and Aradhana Mishra in Toxicology and Industrial Health
Footnotes
Acknowledgments
The authors wish to thank the Director, CSIR-NBRI and CSIR-IITR for his kind support. Saumya Shukla (DST-SERB JRF) acknowledges the DST-SERB (EEQ/2017/000026; GAP-372), New Delhi, India for financial assistance. Priyanka Chauhan (UGC-SRF) is grateful to the University Grants Commission (UGC), New Delhi, India, for Senior Research Fellowship. This manuscript has been approved by institutional ethical committee (CSIR-NBRI_MS/2022/08/07).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science and Technology India; EEQ/2017/000026.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
