Abstract
This study investigates fire growth and thermal response in clothing retail stores, focusing on hanger arrangement, product density, and fire load. Full-scale burning tests were conducted in a fire room designed for two experimental setups: A1 (single-row hanger, 20 kg of products) and B1 (linear wall unit with circular free-standing hanger, 40 kg). Temperatures, CO, CO2, and O2 concentrations were obtained during tests. Results showed maximum heat release rates of 1991 kW (A1) and 2308 kW (B1), with fire spread to the circular hanger group in B1 at 493 s. CO exceeded critical levels 1.5 min after ignition, O2 dropped to critical levels at 11 min, and CO2 surpassed exposure limits at 4 min. The experiments highlight the influence of product arrangement and spacing on fire propagation, gas emissions, and heat release, providing critical data for performance-based fire safety design and evacuation planning in retail textile environments.
Introduction
In shopping malls, high rental costs per square meter and logistical expenses often lead to increased product density. However, high product density poses significant risks to life and property safety in the event of a fire. In recent years, shopping malls have become an integral part of urban life. However, large-scale fires have highlighted the fire safety risks associated with these structures.
Fires frequently occur in shopping malls worldwide, and some of these incidents result not only in property loss but also in casualties. These events demonstrate that inadequate fire safety measures, high fire loads within stores, and errors in material placement significantly contribute to the occurrence of such fires. Performance-based design is considered one of the most realistic approaches to fire safety solutions in buildings and operational facilities. However, there are certain uncertainties associated with performance-based design. This design methodology depends on the physical characteristics of the structure, the materials used, and the storage conditions of these materials. Consequently, obtaining accurate and reliable results is not possible without a thorough understanding of the building materials and other combustible substances.
To comprehensively analyze the combustion characteristics of flammable materials, it is essential to determine key parameters such as the heat release rate, combustion temperature, and CO, CO2, and O2 emission levels. These parameters not only facilitate a detailed evaluation of combustibles but also serve as critical input variables for fire simulation software, which is widely used in performance-based fire safety design. However, the availability of such data in the literature is highly limited, making it challenging to obtain accurate and reliable information. Therefore, to enhance fire safety, it is imperative to establish a comprehensive database containing combustion-related data. This database should be continuously updated through systematic testing of newly developed materials to ensure its relevance and accuracy over time. Within this scope, various fire tests are conducted using standardized methodologies in the field of fire safety, providing detailed data. Due to the presence of numerous combustible materials in building fires, obtaining fire-related data for each material and product is often not feasible. In this context, it is essential to acquire combustion data for critical flammable materials in textile stores, such as clothing, shoes, display racks, and mannequins. However, studies involving such materials in the existing literature remain quite limited.
Chow et al. 1 conducted experimental studies on potential fires in small clothing stores within shopping malls. These experiments utilized a standardized fire room with dimensions of 3.6 m × 2.4 m × 2.4 m. The researchers systematically ignited vertically arranged upper garments, such as shirts, jackets, and t-shirts, to examine their combustion characteristics. Temperature measurements were collected from 24 different locations within the room, while CO, CO2, and O2 emissions were recorded from the exhaust hood to calculate the heat release rate (HRR). In addition, heat flux values were measured at the doorway. In another experiment, two vertical CD display units were used to conduct similar tests for boutique and CD stores. However, in real-world retail environments, products are arranged in various configurations, and the quantity of flammable materials is significantly higher. Clothing stores not only display t-shirts but also include garments such as dresses, trousers, jeans, and scarves. Under flashover conditions, the ignition of a large volume of combustible materials results in extremely high HRR values. Therefore, the HRR values recorded in the experiments conducted by Chow et al. 1 may be several times lower than those observed in actual fire scenarios. To fully assess all potential combustibles, further experimental studies are necessary.
In addition, in a separate experiment conducted by Chow 2 an office setup was established in a controlled fire room. The study emphasizes the critical need for conducting full-scale burning tests to accurately determine the heat release rates of combustible materials. The research highlights a significant gap in data concerning local combustible products in Hong Kong, which hampers the development of effective fire safety regulations. By integrating measured heat release rates into room fire models, the study aims to simulate probable fire environments more realistically. This approach facilitates the enhancement of fire safety provisions, including improved passive building designs and appropriate active fire protection systems. Ultimately, the findings underscore the necessity of such empirical data for implementing the next generation of performance-based fire safety codes.
Zalok and Hadjisophocleous 3 conducted experimental studies to determine the fire loads in clothing stores, the types of combustible materials, and their impact on fire dynamics. Their research presents a comprehensive investigation into determining design fires for clothing retail environments. The research involved surveying 14 clothing stores in Ottawa and Gatineau, Canada, to ascertain total fire loads (MJ), fire load densities (MJ/m2), and types of combustible materials present. Based on the analysis, stores were categorized into three main types: small-sized stores, stores where wood is the primary combustible, and stores where textiles are predominant. Representative fuel packages for each category were developed, and medium-scale fire experiments were conducted. The results indicated that stores with wood as the main combustible exhibited similar burning characteristics to small-sized stores. In contrast, textile-dominated stores produced twice the peak heat release rate, 50% higher gas temperatures, and three times the toxic gas emissions. These findings underscore the necessity of large-scale experiments and computer modeling to establish realistic and reliable design fires for clothing stores.
During store fires, the flame spreads between clothing racks and its propagation to other store products provides critical insights into the fire’s initial growth rate. An example of an experimental study on fire spread is the full-scale fire test conducted by Lee et al. 4 on an intercity train carriage. They investigated that the flame propagation dynamics over time were examined concerning the spacing between sequentially arranged seats and the corridor distance. In addition, the ignition times of the opposing seats following flame spread were analyzed. Poulsen and Jomaas 5 did an experimental study on pool fires for the burning behavior of different wall linings which examines how different wall linings affect the burning behavior of pool fires in enclosed spaces. Experiments conducted in a controlled environment show that combustible wall materials accelerate fire spread and temperature rise, while less flammable materials slow fire propagation. In addition, some wall linings produce significant smoke, impacting indoor safety. The findings highlight the importance of selecting appropriate materials to enhance fire safety in buildings. Grexa et al. 6 studied fire reaction of wood products and other building materials through performance tests conducted. The research analyzes key parameters such as ignition temperature, burning rate, smoke production, and heat release characteristics. The findings indicate that the fire response of wood products varies depending on wood type, density, and surface treatments. While some wood materials exhibit high heat resistance and slow fire spread, they may also generate significant smoke. The fire behavior of other building materials depends on their chemical composition and physical properties. The study highlights the critical role of material selection in ensuring fire safety in buildings.
Altın and Kılıçarslan 7 carried out an experimental investigation involving two full-scale residential room fire scenarios to examine fire behavior and its implications for safety. The study involved a living room and a bedroom, both arranged to mimic typical domestic settings. By determining the fire loads, they quantified fire indices and monitored temperature variations in different vertical zones of each room. Peak temperatures reached approximately 1200°C in the upper zone of the bedroom and around 900°C in the living room. In addition, thermal imaging revealed external surface temperatures, contributing further to the understanding of heat transfer during fires. The findings offer valuable data for improving structural fire safety assessments and the development of fire-resistant building designs.
Kim and Lee 8 performed a numerical analysis to examine the effects of smoke movement between two rooms connected by a lateral opening. The study focused on the development of the upper hot gas layer and its temperature profile over time. Computational simulations were used to observe how room configuration and opening characteristics influence thermal and fluid flow behavior. Their results provide valuable insights for designing safer building layouts by improving predictions of smoke and heat transfer during compartment fires.
In the above literature, it has been observed that studies on convection in stores are limited, and no study has been found on fire propagation between hangers. This study focuses on the characterization of potential fires in retail textile stores, improving evacuation processes, and evaluating product placement in terms of fire safety. The research involves small-scale preliminary studies of experimental setups representing the product groups and fire loads of textile stores operating worldwide. The products selected for the experiments were chosen using a random sampling method from various product groups, with the aim of obtaining homogeneous fuel packages. Fire experiments were conducted on textile products using a room fire test setup accordance with ASTM E2257—17 9 and ISO 9705 10 standards. The “A1” experimental setup represents the experiment with a single-row hanging product wall unit containing a total of 20 kg of flammable products. The “B1” experimental setup represents a configuration with a linear wall unit containing a total of 40 kg of flammable products and a circularly arranged free-standing product unit, with a 50 cm distance between them. The conducted experimental study evaluates fire growth rate, flame egress time from the door, and flame propagation times between product units. Gas temperature measurements were recorded at five different points on the ceiling, while gas temperature, carbon monoxide (CO), carbon dioxide (CO2), and oxygen (O2) emissions were monitored over time in the exhaust duct. The obtained data were used to calculate the heat release rate (HRR), which is presented in this study. In addition, video footage recorded during the experiments was analyzed to assess the combustion behavior of textile products and the flame propagation between display racks. The collected data aims to enable retail textile sector establishments to conduct performance-based and cost-effective fire safety evaluations independently. Moreover, this study seeks to contribute to assessing the impact of textile product placement on fire growth in the context of evacuation time considerations.
Materials and methods
In this study, textile products obtained from a clothing company were used as test samples for physical and chemical analyses. The utilization of sampled products suggests that all product types were included in the experimental process. It has been determined that the average fabric content of clothing purchased from the store is 65% cotton and 35% polyester. The experiments involved a random selection of textile products designed for different seasons (winter, summer, spring, autumn) and various age groups (infants, children, adolescents, and adults). Combustion properties of cotton and polyester used in the experiments are shown in Table 1.
Combustion properties of cotton and polyester used in the experiments.
During the experimental study, temperature, carbon monoxide (CO), carbon dioxide (CO2), and velocity data collected from the ceiling and exhaust duct were used to determine mass flow rate, heat release rate (HRR), and gas emission rates through the following calculation methods. The mass flow rate along the duct was derived from velocity measurements obtained using a bidirectional probe positioned along the centerline. During the experiments, all measured parameters were recorded at sampling interval of 1 Hz. To reduce short-term fluctuations and clarify the underlying signal trend, the data were smoothed using a Savitzky–Golay filtering approach. A second-order polynomial with a 100-point window was applied to effectively suppress noise while preserving key signal features such as peak magnitude and temporal location. The mass flow rate was calculated using the equation provided below (Equation 1). 9

The theoretical value of C is calculated using the equation C≈ 22A.
The oxygen depletion factor for heat release rate (HRR) is determined using the following equation. 11
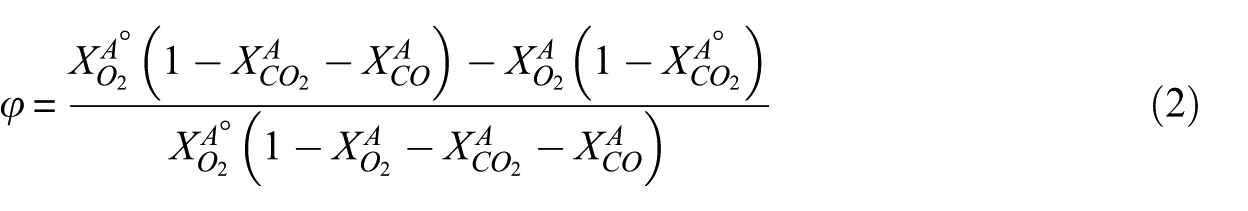
Subsequently, the heat release rate (HRR) was calculated using the following equation. 11
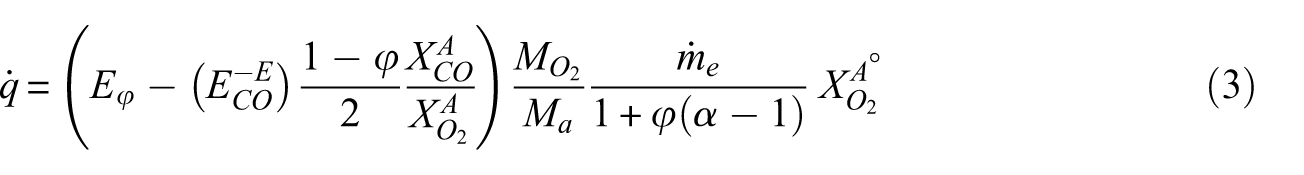
In this equation, the net heat release rate per unit mass of consumed O2 is taken as E = 13.1 MJ/kg, for carbon monoxide production ECO = 17.6 MJ/kgO2, with molecular weights Ma = 29 kg/kmol, MCO = 28 kg/kmol, MO2 = 32 kg/kmol, and the combustion expansion factor is assumed as α = 1.084.
Experimental procedure
In this study, fire experiments were conducted in a room fire test setup. Due to the high temperatures within the chimney, it was not feasible for the CO2, CO, and O2 sensors to be directly inserted into the chimney for data collection, as the sensors would be damaged by the high temperatures and dense smoke, as determined in preliminary tests. To prevent this, a system was designed that could vacuum the smoke sample from the chimney, cool it down, filter out the soot particles, and then transmit it to sensors located inside a sealed box.
The smoke extracted and conditioned from the chimney was then transferred to the sealed box containing the CO2, CO, and O2 sensors, as shown in Figure 1. This method enabled accurate measurement of the CO2, CO, and O2 volume fraction in the cooled and soot-free chimney smoke. Furthermore, an additional thermocouple was placed inside the sealed box to monitor the temperature of the gas reaching the sensors, providing protection against sudden temperature increases. Chromel (Ni–Cr) positive wire and an Alumel (Ni–Al) negative wire K-type thermocouples were used for temperature measurements. The K-type thermocouples employed in the experiments had a wire gauge of 1 mm, providing a suitable operational temperature range up to 1000°C. Figure 1 shows the long thermocouples placed for the ceiling of the room, and Figure 1 illustrates their placement points on the ceiling. Except for the point above the door, thermocouples were placed at the points indicated on the diagram, and measurements were taken.

Arrangement of measurement devices in the chimney.
For the measurement of carbon dioxide (CO2) volume fraction, the Winsen MH-Z16 infrared sensor was employed. Oxygen (O2) volume fraction was measured using the Winsen ME2-O2-Ф20 electrochemical sensor. Carbon monoxide (CO) volume fraction was determined using the MQ-7 gas sensor. Electrochemical sensors are commonly employed in full-scale fire experiments and point-based gas sampling systems due to their high sensitivity, good selectivity, and relatively fast response for O2 and CO measurements. 12 Nevertheless, it is well recognized that electrochemical sensors may be affected by factors such as sensor aging, cross-sensitivity to other combustion gases, and transient effects under rapidly changing fire conditions. As discussed by Ho, 13 when electrochemical sensors are used within a properly designed sampling system, the overall uncertainty in gas volume fraction measurements is often dominated by gas transport delay, sampling line losses, and flow non-uniformity rather than by the sensing principle itself. To address these potential limitations, a vacuum-driven gas extraction system with cooling and particulate filtration was employed in the present experiments, ensuring that all sensors operated within their specified temperature and humidity limits. This approach reduces thermal drift, minimizes cross-interference effects, and enhances the reliability of electrochemical O2 and CO measurements. The CO2 volume fraction was measured using an NDIR sensor, which is less affected by oxygen depletion and cross-sensitivity and is, therefore, considered more stable for long-duration fire experiments.
The used clothing items were burned in a dry condition, and the experiments were conducted during the summer period under low ambient humidity conditions, resulting in a comparatively limited contribution of water vapor to the sampled gas stream. Accordingly, the gas sampling system employed forced cooling and particulate filtration, without the use of dedicated water vapor filtration. In fire and combustion studies involving surface fires, such as oil burning on water and marine fire scenarios, water vapor becomes a dominant and external source of measurement uncertainty. Therefore, active moisture removal is necessary before gas sensors to ensure accurate readings.14,15 While residual moisture may introduce a secondary source of uncertainty in electrochemical O2 measurements, under the experimental conditions of this study this effect is not expected to significantly influence the interpretation of oxygen depletion trends and tenability-related assessments.
The technical specifications of the sensor are provided in Table 2. To measure the gas flow rate in the duct, a bidirectional probe and Testo 510i brand model device were employed. The technical specifications of the Testo 510i device are provided in Table 3.
Technical specifications of the sensors.

Technical specifications of the Testo 510i device.

Fire tests were conducted in different intervals to examine the temperature, fire spread, CO2, CO, and O2 emissions and HRR caused by flammable materials which contain 65% cotton and 35% polyester. in large retail textile stores. Two different fire scenarios, labeled A1 and B1, were evaluated. The tests utilized wall-mounted hangers and independent arm iron clothing hangers used in stores. Flammable materials, including t-shirts, plastic hangers, bags, dresses, pants, jeans, and scarves made from different contents, were used in the experiments. To measure HRR under free burning conditions, ignition was manually initiated from the lower part using an ignition source (torch).
The combustion process was monitored until the fuel was exhausted and the fire extinguished. No automatic extinguishing system was set up, and no manual intervention was made during the process. To analyze the effect of arrangement and fire load density in clothing stores and to determine the spread times of the fire based on distances, two different arrangements were decided. The experimental definitions are provided in Table 4.
Experimental definitions.
A1 single-row hanger experiment
In the A1 experiment, a shelf arrangement was made to simulate a wall unit 1 m wide and 1.5 m high. The shelf was placed at the far end of the room. There was a single metal hanger on the rack, and 20 kg of products were hung on the shelf. The arrangement of the clothes is shown in Figure 2. The ignition was manually initiated, and no extinguishing system was installed. The fire was allowed to self-extinguish, and measurements were taken throughout the process.

Product hanger arrangement.
B1 straight row and circular array fire spread experiment
In the B1 experiment, a wall unit with a height of 1.2 m and a width of 1 m, along with four metal arm hangers with a maximum height of 1 m, was used. A total of 40 kg of products were loaded, with 20 kg on the wall unit and 20 kg on the movable hanger. The units were arranged with a 50 cm distance between them. The arrangement of the clothes is shown in Figure 3. Ignition was manually initiated.

Product hanger dimensions B1 Straight row and circular arrangement product hanger arrangement for fire spread test.
Results
A1 single-row hanger experiment results
From the A1 experiment, temperatures were measured on the ceiling of the room and inside the chimney given in Figure 4. The maximum temperatures on the ceiling measured at the 640th second were as follows: T2: 360.5°C, T3: 352.75°C, T4: 520.75°C, T5: 303.75°C, and T6: 289.5°C. The highest temperature in the chimney was measured obtained from T1 point as 83.75°C at the 584th second.
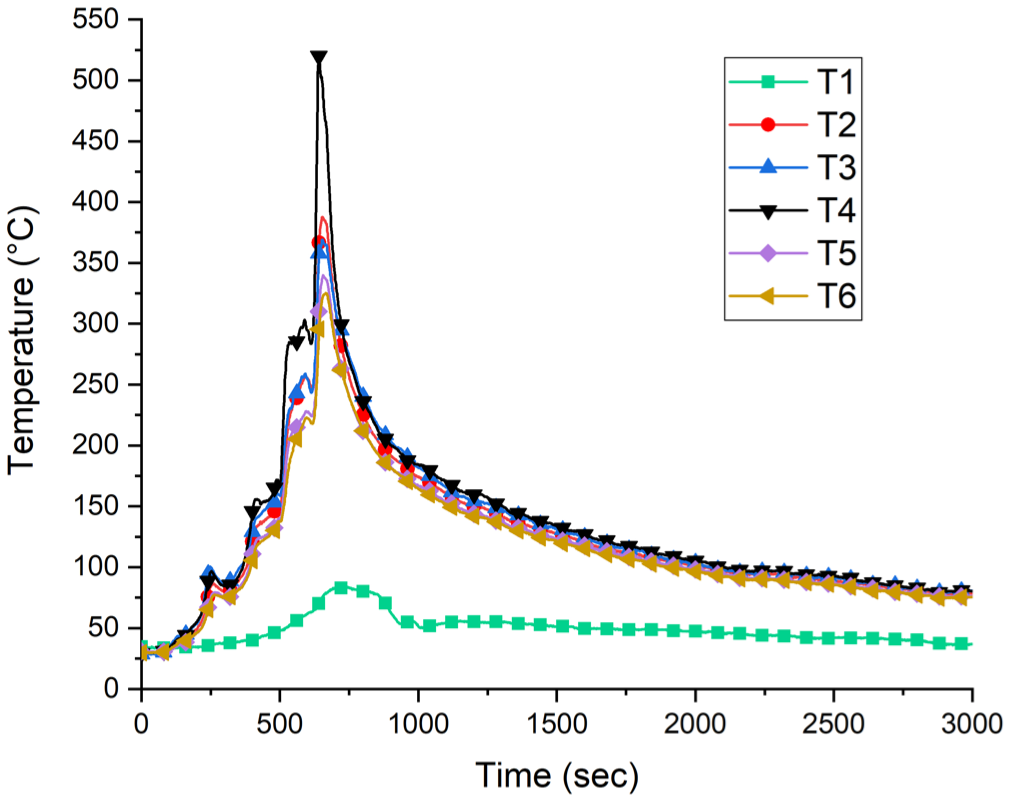
Experiment A1 temperature values.
Time-dependent HRR and CO consumption data are presented in Figure 5, respectively. In the A1 experiment, the maximum heat release rate was measured at 1991 kW at the 673rd s (11.2 min). After 2.7 min, at the 840th second (14th min), it decreased to 613.1 kW.
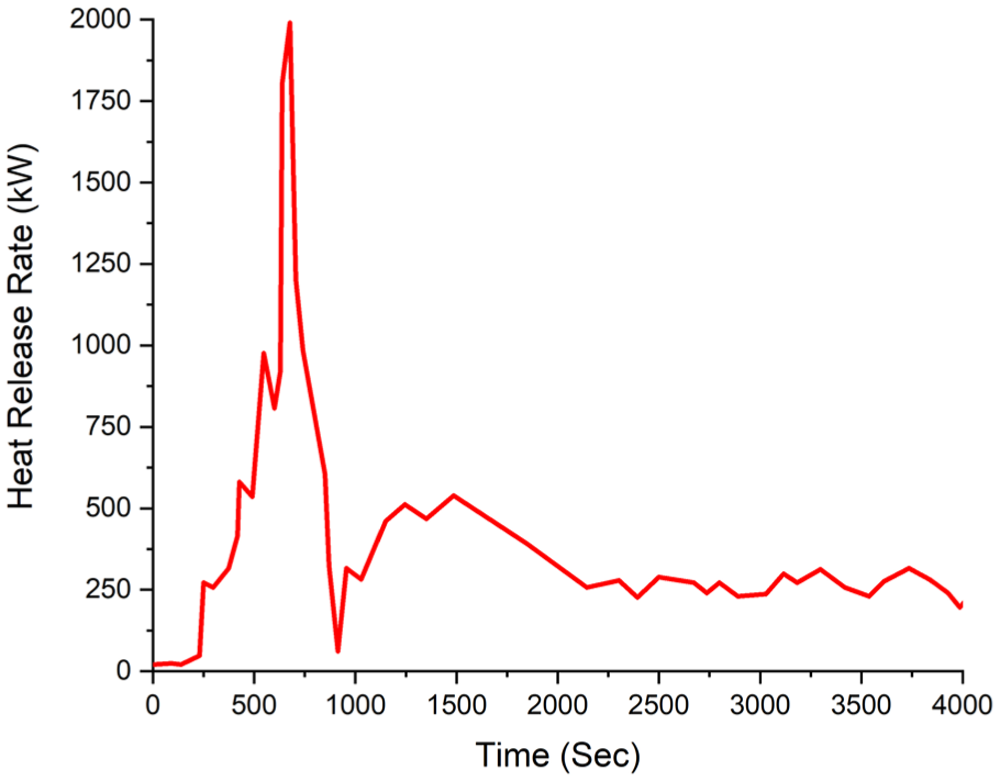
Experiment A1 HRR Heat release rate.
The maximum time-dependent CO2 volume fraction of 3.27% is shown in Figure 6. At the beginning of the experiment A1, the CO2 volume fraction remains at low levels but exhibits a sharp increase, reaching a peak of approximately 3.2% around the 700th second. Following this peak, volume fraction rapidly decreases and gradually stabilizes over time. This behavior indicates a short-term but intense gas release within the system, followed by a progressive decline in emission levels.
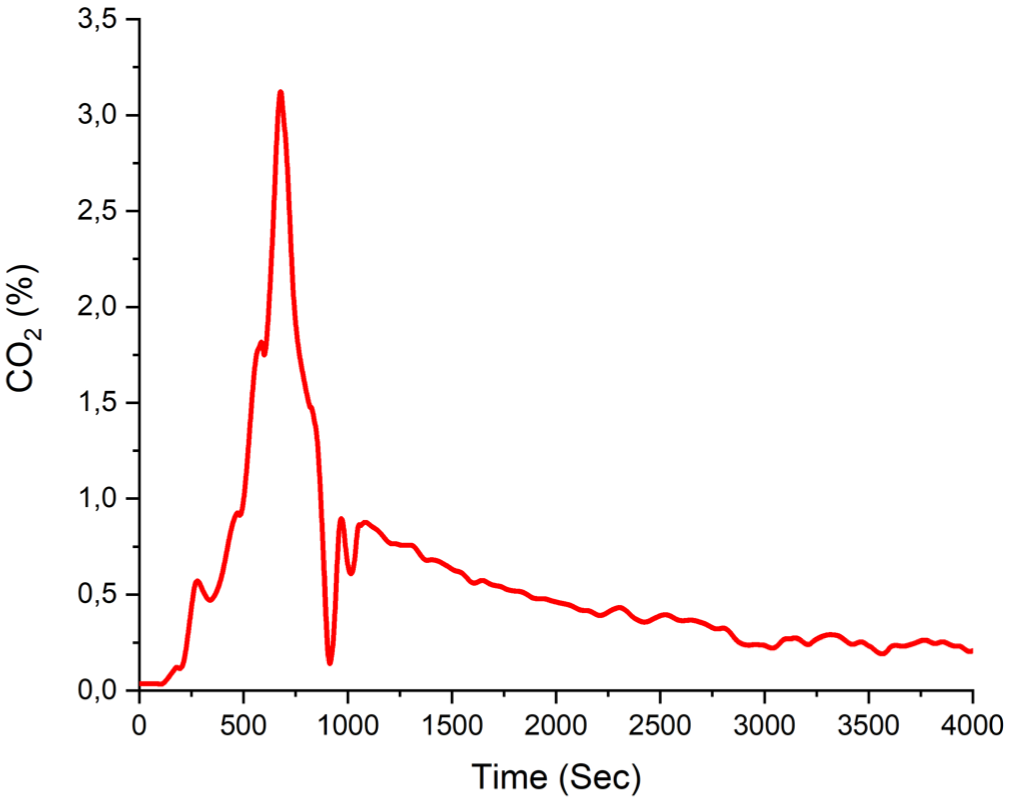
Experiment A1 CO2 volume fraction.
The maximum time-dependent CO volume fraction of 229 ppm is shown in Figure 7. During the initial 800 s, a fluctuating degree of approximately 90 ppm was observed. It is understood that the CO volume fraction stabilized at a certain level during the flaming combustion phase up to the 800th second. Starting from the 1000th second, due to the transition into the smoldering phase and the consequent reduction in complete combustion reactions, the CO volume fraction exhibited a continuous increase until approximately the 2600th second, after which a decreasing trend was observed.
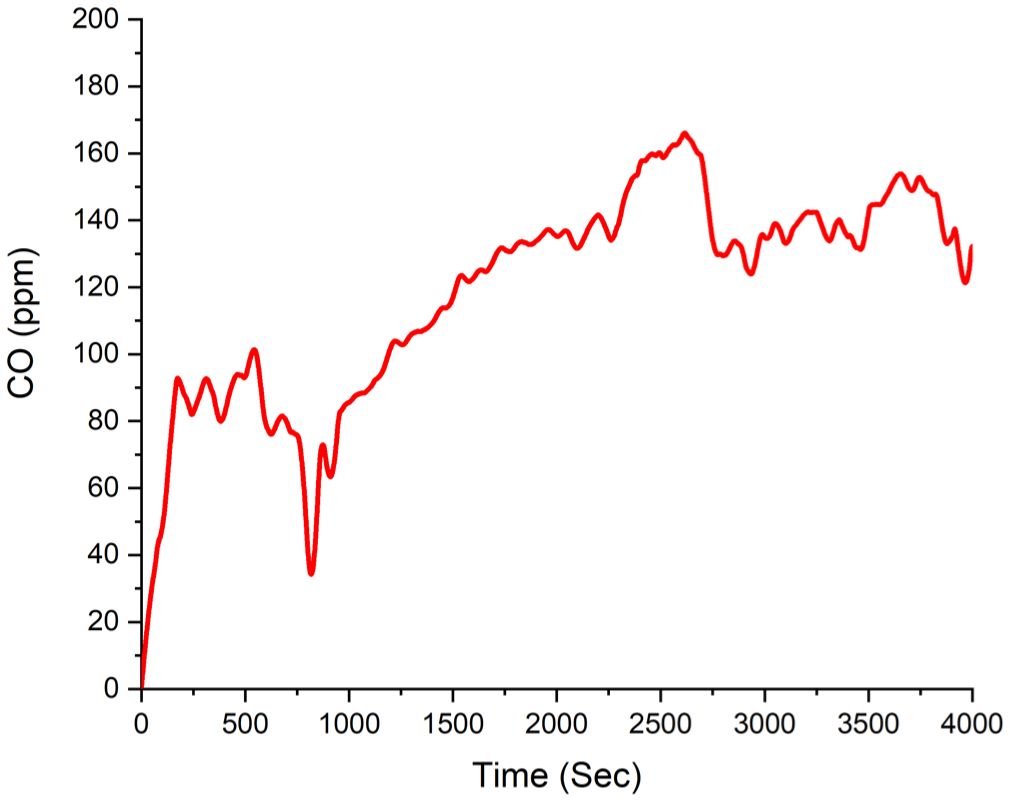
Experiment A1 CO volume fraction.
The minimum time-dependent O2 volume fraction of 17.66% is calculated and shown in Figure 8. Initially, the O2 volume fraction is around 20.9%, but it rapidly decreases to minimum within a short period. Following this sharp decline, volume fraction begins to increase and gradually stabilizes over time. This fluctuation indicates that combustion or reaction processes within the system led to oxygen consumption, followed by a gradual reintroduction of oxygen into the environment to restore balance.
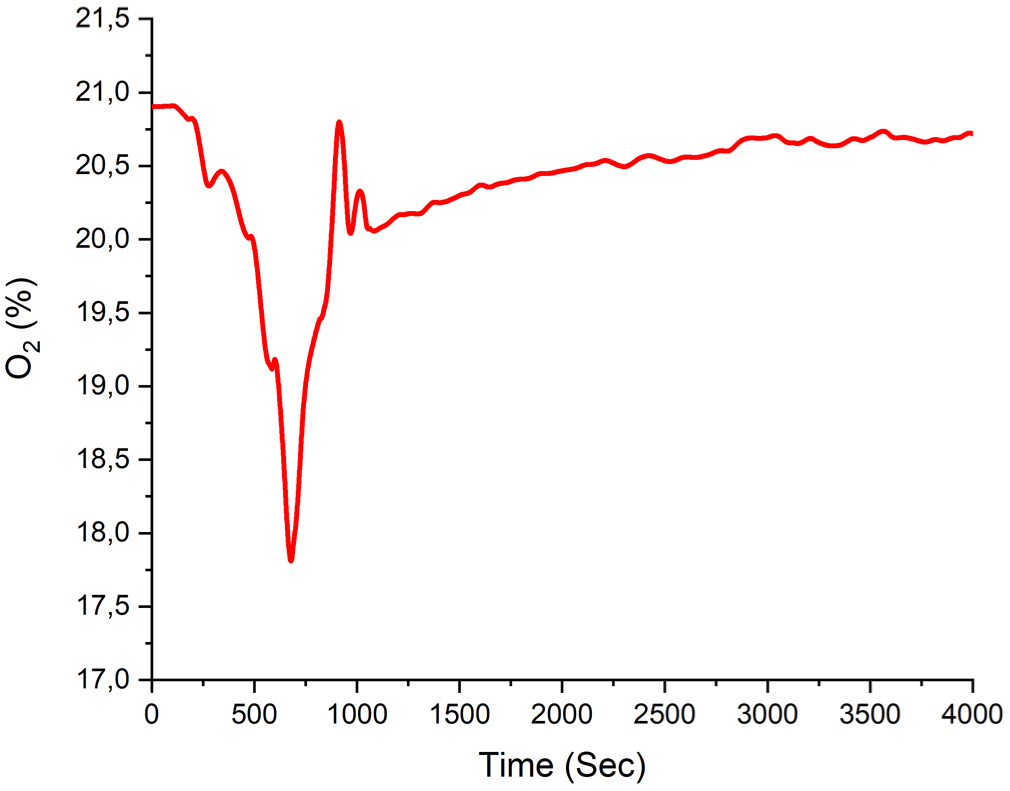
Measured O2 volume fraction in Experiment A1.
Experiment A1 Process: At 0 s, ignition was initiated from the right side of the hanger. At 150 s, the flame had reached the metal hanger. At 180 s, due to the effect of heat, the plastic hanger melted, causing the burning product to fall to the ground. At 300 s, the remaining products on the hanger reignited. At 400 s, the flame tongue reached the ceiling from the central part of the hanger. At 490 s, all remaining products on the hanger were engulfed in flames, and some unburned gases were observed to ignite. Due to the heat, plastic hangers completely melted, leading to the fall of all remaining products to the ground. At 510 s, the temperature reached 520°C. At 640 s, it peaked at 520.75°C. By 740 s, the temperature had dropped to 198°C, marking the transition to the fire decay phase.
At 60 s, the smoke was observed to descend to a height of 150 to 170 cm, yet visibility remained clear up to these levels. In the relatively cooler and less illuminated left section, the difference in smoke height was recorded as 24 cm. By 120 s, the visibility became hazy, making it difficult to distinguish the colors of the products. At 270 s, dense black smoke was seen descending to 80 cm on the left side and 110 cm near the doorway. By 310 s, the smoke level had further dropped to 76 cm on the left side and 95 cm at the doorway. It was also observed that the smoke had completely covered the upper portions of the hanging garments.
A series of time-dependent photographs taken by a camera positioned at the doorway facing the fire are presented in Figure 9.

Photographs showing the progress of Experiment A1.
B1 straight row and circular array fire spread experiment results
Temperatures were measured on the ceiling of the room and in the chimney. The maximum temperature measured for Experiment B1 was T1: 74.25°C, T2: 360.25°C, T3: 365.75°C, T4: 542.25°C, T5: 284.5°C, and T6: 295.5°C at 394 s. The highest temperature in the chimney was 143.5°C at 876th second (T1).
The time-dependent ambient temperature data are presented in Figure 10. The heat release rate (HRR) and oxygen consumption data over time are illustrated in Figures 11 and 14, respectively. For Experiment B1, the maximum HRR was recorded at 422 s (7 min) as 2308 kW. After 3.28 min, at 619 s (10.3 min), it decreased to 781 kW. Subsequently, at 661 s, it peaked again, reaching 2156 kW.
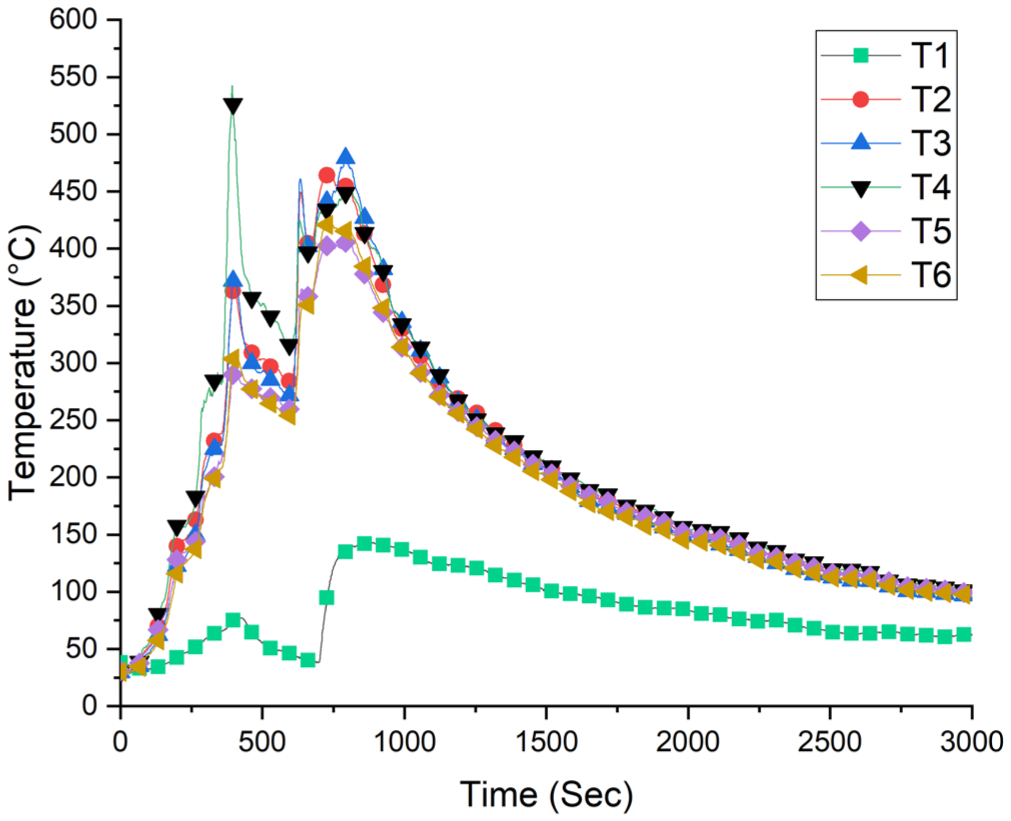
Experiment B1 temperature values.
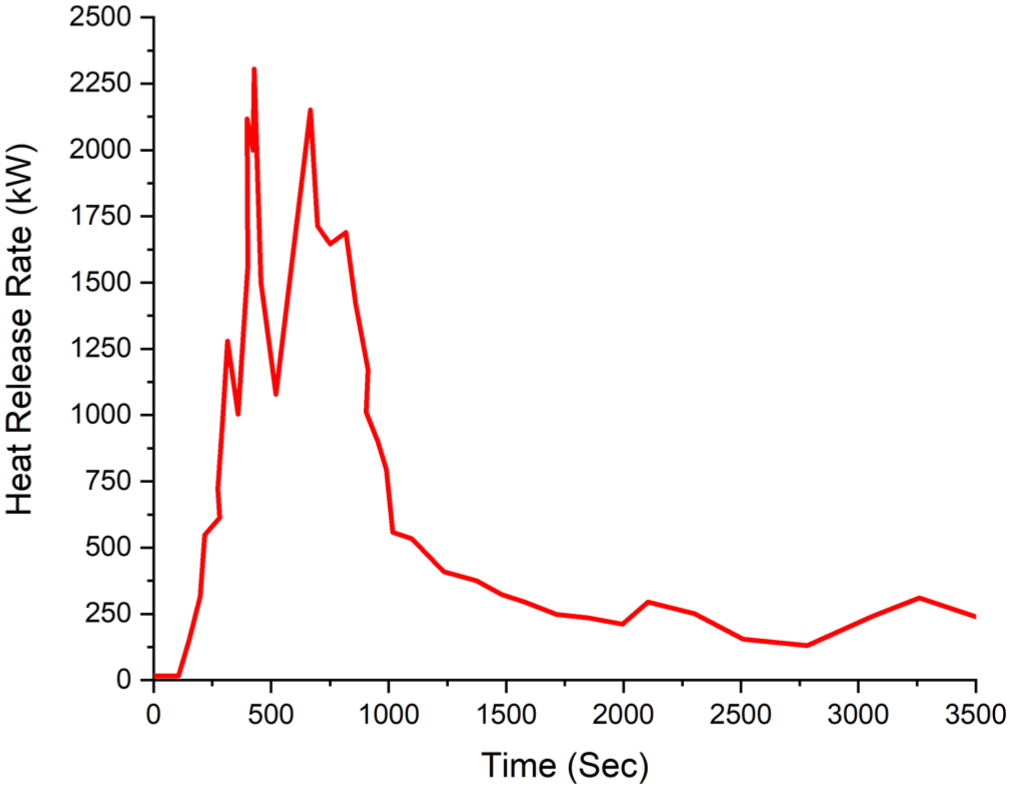
Experiment B1 HRR Heat release rate
Figure 11 presents the variation of heat release rate (HRR) over time for Experiment B1. At the onset of the experiment, the HRR increases rapidly, forming two distinct peaks around the 400th and 600th seconds. At 494th second, as can be seen in the figure, there is a sudden drop and rise after the peak, during which the fire is seen to spread.
The maximum time-dependent carbon dioxide volume fraction of 3% is shown in Figure 12.
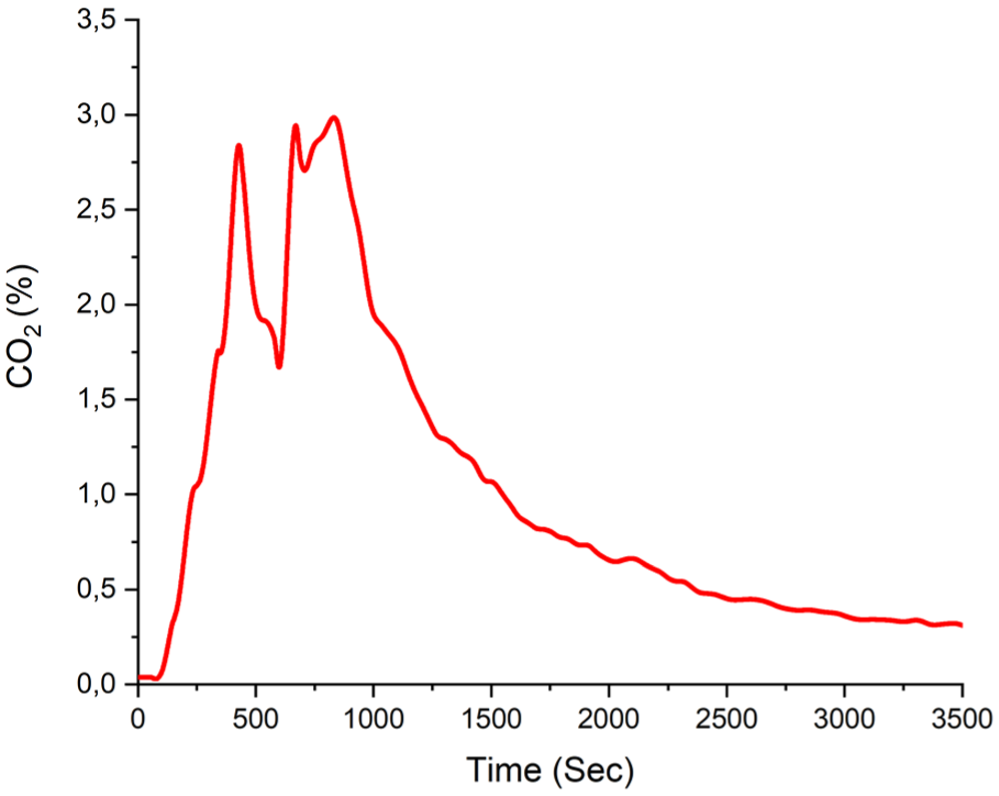
Experiment B1 carbon dioxide volume fraction.
The maximum time-dependent carbon monoxide volume fraction of 250 ppm is shown in Figure 13.
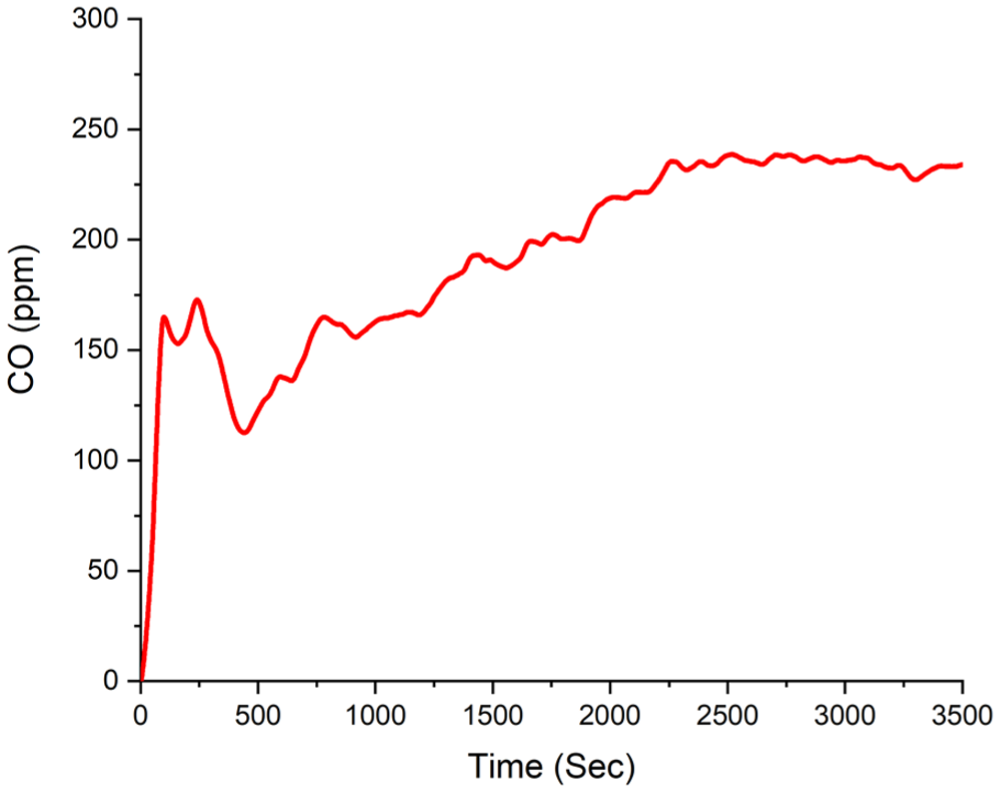
Experiment B1 carbon monoxide volume fraction.
During the experiment, the time-dependent oxygen volume fraction was calculated as a minimum of 17.92%, as shown in Figure 14.
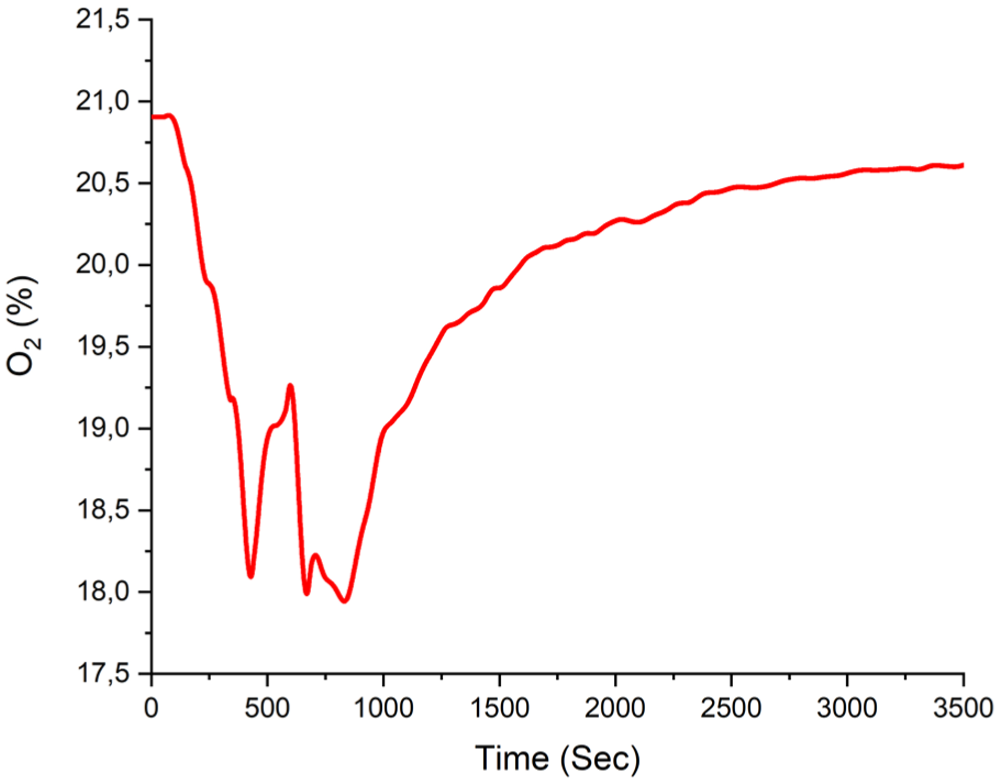
Experiment B1 calculated oxygen volume fraction.
At 0 s, ignition was initiated on the right side of the wall unit located on the left wall. At 15 s, the ignited product detached from the hanger and fell to the ground. At 120 s, the fire reached the remaining hanging products. At 210 s, the flames extended beyond the hanger rod and reached the ceiling. At 330 s, unburned pyrolysis gases became visibly distinct. At 390 s, all the products on the wall unit were engulfed in flames. At 420 s, some products on the free-standing rack fell to the ground due to the heat. At 450 s, the formation of pyrolysis gases became highly pronounced on both the fallen and hanging products. At 493 s, fire spread occurred. At 510 s, flames advanced toward the hanging products. At 540 s, flames completely engulfed the products on the fire-facing side of the free-standing rack. At 570 s, all products on the free-standing rack ignited and fell to the ground. A total of 77 s after the maximum HRR point, the entire pile was observed to be fully involved in flames.
Smoke Progression and Density: At 15 s, the smoke level dropped to 170 cm, with a grayish color, and visibility remained clear. At 60 s, a slight blurring of visibility was observed. At 120 s, as the flames intensified, visibility improved again. At 300 s, the smoke level dropped to 110 cm. At 490 s, prior to maximum HRR point, pyrolysis gases became highly distinct, and the smoke level dropped to 75 cm. At 510 s, following maximum HRR point, the gray smoke beneath the dense black smoke disappeared. At 540 s, the smoke level further dropped to 60 cm. At 600 s, the density of black smoke significantly increased.
The stages of the fire process were recorded using a camera positioned at the door opening, and a time-sequenced series of photographs is presented in Figure 15.

(Continued)
Verification and comparison
In Figure 16, the HRR data obtained from two separate experiments are presented in a comparative manner alongside previous experimental studies in the literature. In the graph, the study conducted by Zalok and Hadjisophocleous 3 represents retail environments with total 35.44 kg of cloth as the major combustible content, while the study by Chow et al. 1 represents a fire and flashover experiment conducted with 96 t-shirts. The highest heat release rates measured in the literature were 1528 kW (Chow et al.), 1500 kW (Candy et al.), and 2400 kW (Chow et al.). When the HRR values obtained from this study are compared with those reported in the literature, it is observed that the maximum heat release rates are quite similar and the trends are consistent. However, some discrepancies are observed in the time taken to reach peak energy at the initial stage. These differences are thought to be due to factors such as the density of clothing arrangement, the energy of the initial ignition source (torch), and the diversity of the clothing materials used in the experiments.
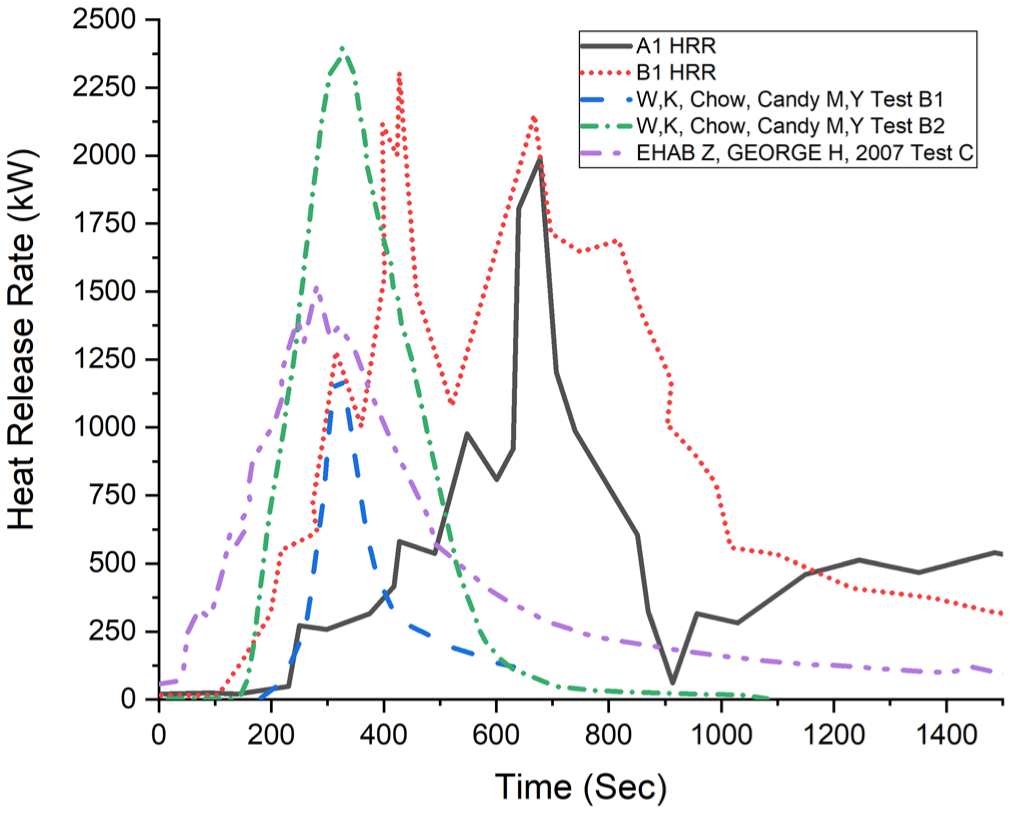
Heat release rate comparison.
Discussion and conclusion
This study focuses on the characterization of potential fires in retail textile stores, improving evacuation processes, and evaluating product placement in terms of fire safety. The research involves small-scale preliminary studies of experimental setups representing the product groups and fire loads of textile stores operating in 500 different locations across 30 countries worldwide. The products selected for the experiments were chosen using a random sampling method from various product groups, with the aim of obtaining homogeneous fuel packages.
The “A1” experimental setup represents the second experiment with a single-row hanging product wall unit containing a total of 20 kg of flammable products. The “B1” experimental setup represents a configuration with a linear wall unit containing a total of 40 kg of flammable products and a circularly arranged free-standing product unit, with a 50 cm distance between them.
In the experiments, a total of 94 hangers, each with a 1-m-long hanger rod, were used. These hangers vary according to the product groups, and as a result, their weights differ. The hangers used in the adult group weighed 95, 60, and 35 g, respectively. In the children and baby groups, the hangers weighed 20, 30, and 35 g. In the tests, the total weight of the hangers accounted for 29% of the weight of the products, which means that 5.8 kg of hangers were used for 20 kg of products. The combustion products of plastic include highly toxic substances such as carbon monoxide, hydrochloric acid, hydrogen cyanide, and nitrogen oxides. In the characterization of design fires in retail textile stores, the characterization of additional materials used, apart from the products, should also be examined. It should be discussed that these auxiliary products should be replaced with products that do not contribute to the fire and do not produce smoke.
In the experiments, the volume fraction of carbon dioxide (CO2), carbon monoxide (CO), and oxygen (O2) is shown over time in Figures 17–19. Carbon dioxide can cause simple asphyxia while a slight increase in the volume fraction of carbon monoxide in the air leads to respiratory disturbances. When the volume fraction reaches 1.5%, breathing difficulties and an increase in heart rate are observed. At a volume fraction of 3%, muscle pain, fainting, and the risk of death occur. The carbon dioxide volume fraction obtained in the experiments was found to range between 3% and 4%. From the eighth minute on average, the levels reached are sufficient to pose a risk of fainting and death. 16
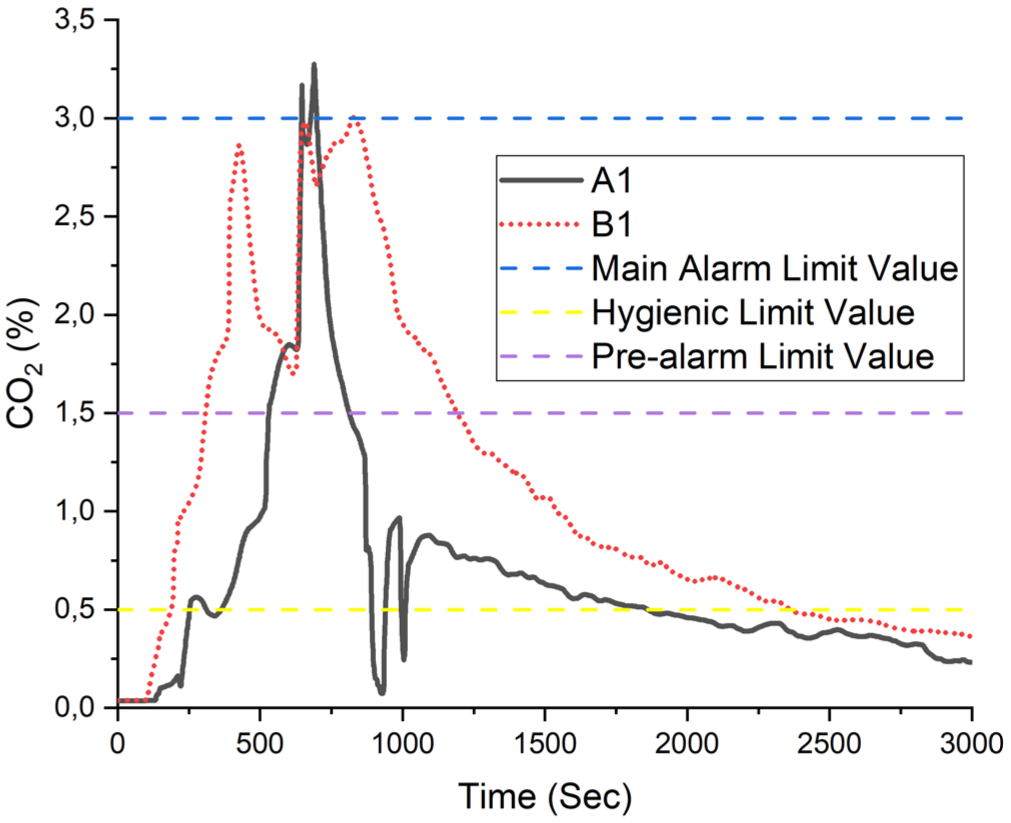
Carbon dioxide volume fraction.
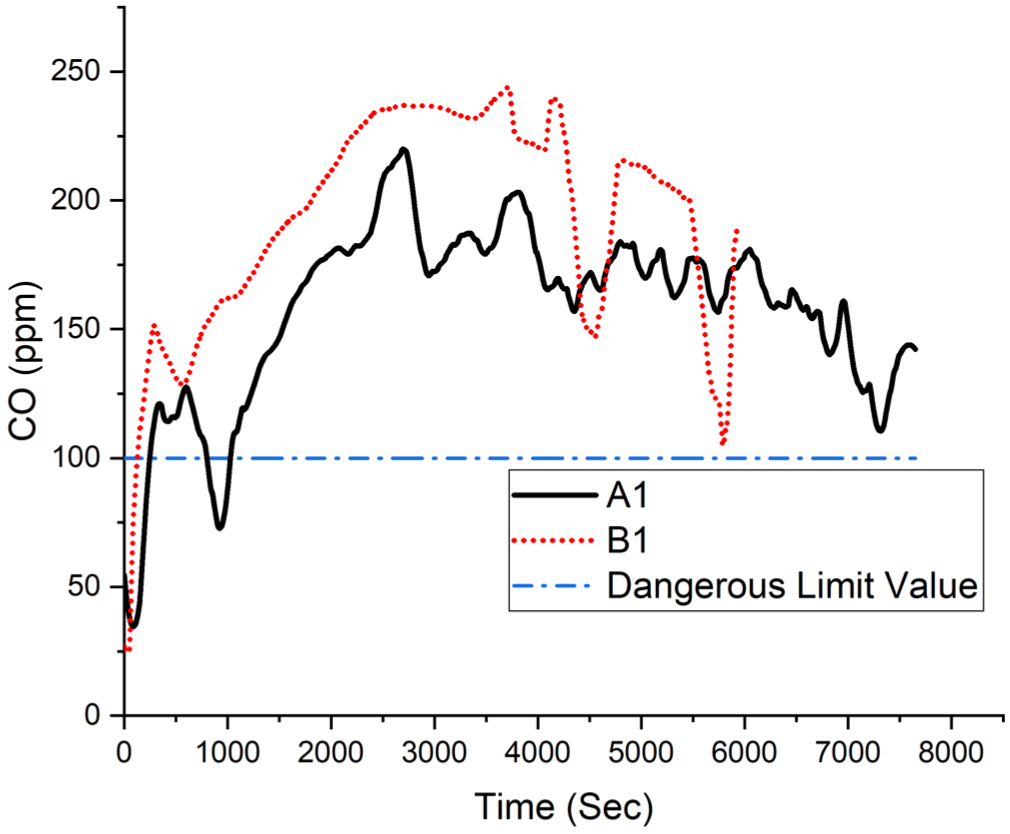
Carbon monoxide volume fraction.
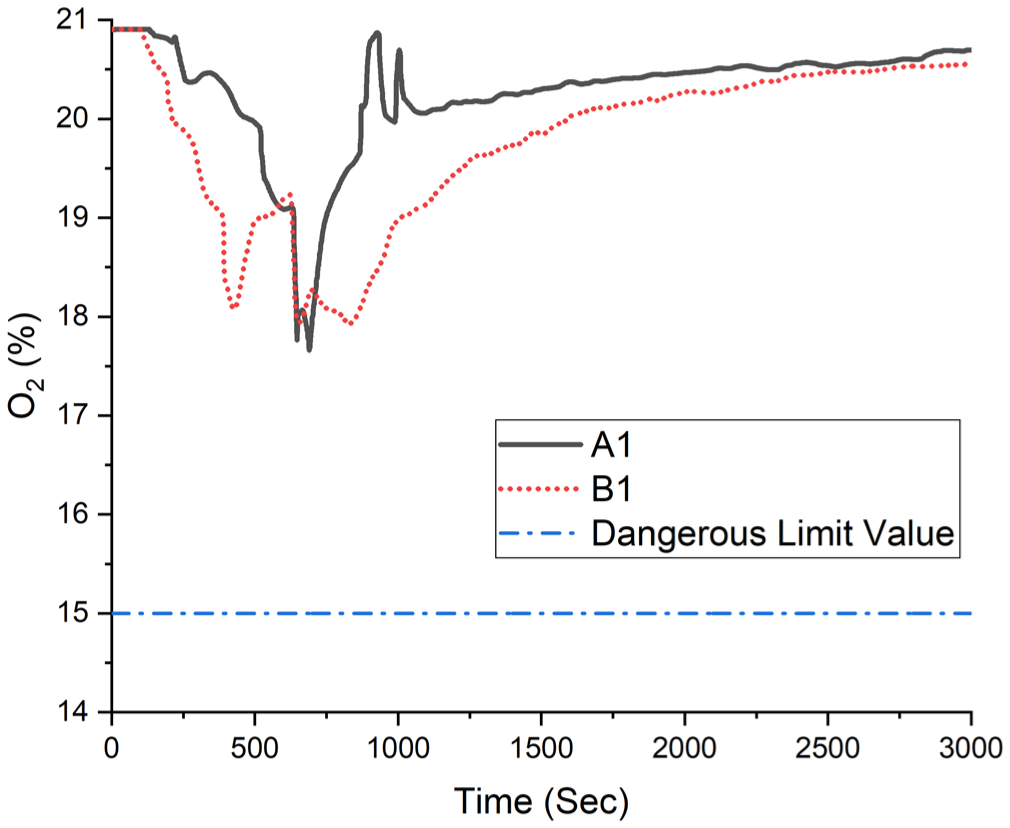
Oxygen volume fraction.
In the assessment of hazards occurring during a fire, threshold values related to oxygen deficiency and exposure to toxic gases must be considered. In this context, Table 5 summarizes the limit values for different hazard types based on varying exposure durations. 16
Threshold values for hazard types. 16

Figure 17 illustrates the temporal variation of carbon dioxide (CO2) volume fraction measured at points A1 and B1 during the experimental period. CO2 is a common combustion by-product and poses a suffocation hazard by displacing oxygen in enclosed environments. The graph reveals that CO2 levels at point A1 peaked above 3%, exceeding the main alarm threshold, while point B1 recorded volume fraction approaching 2.5%. These values significantly surpass the hygienic exposure limit of 0.5% established for occupational settings. 17 Prolonged exposure to elevated CO2 levels may lead to symptoms such as headaches, dizziness, cognitive impairment, and in extreme cases, loss of consciousness. 18 The fact that both the pre-alarm and main alarm thresholds were exceeded indicates that the environment reached a critical level of hazard that would necessitate immediate evacuation. 19
Carbon monoxide (CO) is a chemically induced asphyxiant that impairs oxygen transport by forming carboxyhemoglobin (COHb), thereby inhibiting oxygen binding to hemoglobin. The affinity of CO for hemoglobin is approximately 200–250 times greater than that of oxygen, 20 leading to significant reductions in oxygen delivery to tissues. 21 Exposure to CO volume fraction of 100 ppm can result in carboxyhemoglobin levels of 16%, sufficient to produce clinical symptoms. 22 The National Institute for Occupational Safety and Health (NIOSH) recommends a short-term exposure limit (STEL) of 200 ppm for 15 min. 23 In fire scenarios, the combination of CO and hydrogen cyanide (HCN) poses a greater risk than exposure to either gas alone, as CO impairs oxygen transport while HCN inhibits cellular respiration, together leading to a more severe and potentially fatal asphyxiation. 24
Clinical symptoms of hypoxia begin to appear when oxygen volume fraction drops below 15% for periods longer than 15 min, while levels between 6% and 10% can result in fatal outcomes due to severe oxygen deprivation. In the experimental setup, oxygen volume fraction values were recorded and are presented in Figure 19. The results indicate that the oxygen level decreased to approximately 17.66%, which, although not immediately lethal, signals the onset of physiological stress and impaired cognitive and motor functions.16,19
Figure 20 presents the temporal evolution of the smoke layer height for both experimental configurations (A1 and B1). The smoke layer height data were obtained based on visual observations from the video recordings captured during the experiments. In both cases, a rapid descent of the smoke layer was observed during the early stages of fire growth, followed by a quasi-steady phase once the fire reached a fully developed state. For the A1 configuration, the smoke layer height decreased rapidly within the first 300 s and stabilized at approximately 75–80 cm. In contrast, the B1 configuration exhibited a more pronounced smoke accumulation, with the smoke layer descending to approximately 45–50 cm within 600 s. This difference can be attributed to the higher fuel load and enhanced fire growth rate in the B1 configuration, which resulted in increased smoke production and reduced tenability conditions. From a tenability perspective, a smoke layer height below 2.0 m significantly impairs visibility and occupant evacuation, while values below 1.0 m are generally considered critical for safe egress. In the present experiments, both configurations reached critical smoke layer heights within a few minutes after ignition. In particular, the B1 configuration reached a smoke layer height below 0.5 m, indicating severe visibility loss and untenable conditions.
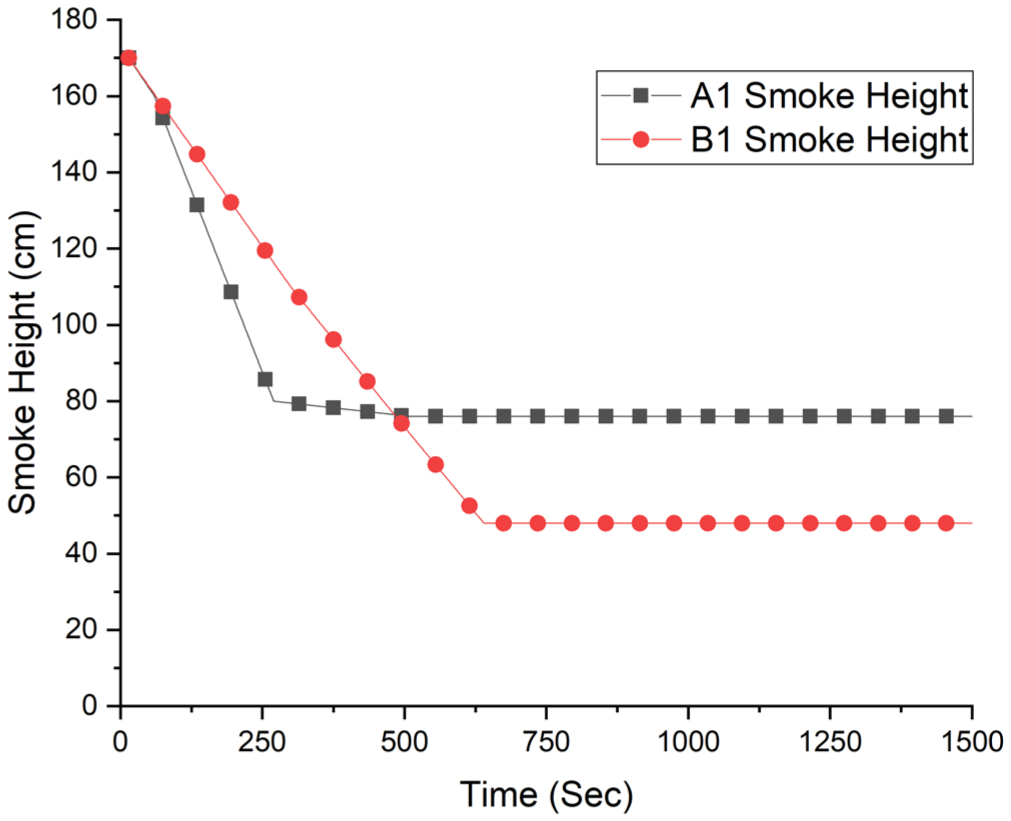
Temporal variation of smoke layer height for the A1 and B1 experimental configurations.
Within the scope of this study, a full-scale fire hazard preliminary assessment was carried out in the retail textile sector, and the following conclusions were drawn from the data obtained
In the A1 experiment, the oxygen volume fraction dropped below the dangerous threshold of 19% at around 700 s, while in the B1 experiment, it fell below this level at approximately 350 s and again at 700 s. These findings indicate that in both scenarios, the ambient oxygen level reaches a critical point roughly 11 min after the fire ignition.
It was observed that the CO volume fraction exceeded the dangerous threshold of 0.01% at approximately 100 s in the A1 experiment and at 110 s in the B1 experiment. This suggests that carbon monoxide levels rise above critical limits roughly 1.5 min after the fire begins in both cases.
In the A1 experiment, CO2 volume fraction exceeded the hygienic threshold of 0.5% at around 250 s, while in the B1 experiment this occurred at approximately 235 s. This implies that the CO2 exposure level crosses the hygienic limit roughly 4 min after ignition in both cases.
In the experimental studies, the maximum HRR was found to be 1991 kW in the A1 experiment with a single hanging group, and 2308 kW in the B1 experiment with double hanging groups.
In experiment B1, it was observed that fire spread to circular hanger group occurred at 493 s.
For effective fire safety management, factors such as store structures, building performance, store sizes, store concepts, and climate conditions must be considered. These factors should be evaluated in terms of design criteria such as exit widths, evacuation times, and floor heights. Various fire dynamics simulators can be used for modeling purposes. In future studies, a validation study of fire simulations is planned using the data obtained. Also, further experimental investigations are planned to examine the effects of different spacing distances between hangers on flame spread, as well as the influence of textile products quantity and variations in material composition and ratios of the burning products on the HRR and emission characteristics. Furthermore, simulations will be conducted in a full-scale model of a sample store to collect data, which will then be used to analyze the effects of fire-induced smoke and heat on evacuation scenarios by employing evacuation models.
Footnotes
Appendix
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
