Abstract
This study addresses the urgent issue of water pollution caused by iron (Fe) and manganese (Mn) ions. It introduces an innovative approach using graphene oxide (GO) and GO-decorated polyethersulphone (PES) membranes to efficiently remove these ions from contaminated water. The process involves integrating GO into PES membranes to enhance their adsorption capacity. Characterization techniques, including scanning electron microscopy, Fourier-transform infrared, and contact angle measurements, were used to assess structural and surface properties. The modified membranes demonstrated significantly improved adsorption compared to pristine PES. Notably, they achieved over 94% removal of Mn2+ and 93.6% of Fe2+ in the first filtration cycle for water with an initial concentration of 100 ppm. Continuous filtration for up to five cycles maintained removal rates above 60%. This research advances water purification materials, offering a promising solution for heavy metal ion removal. GO-decorated PES membranes may find application in large-scale water treatment, addressing environmental and public health concerns.
Keywords
Introduction
The contamination of water sources by heavy metal ions, particularly iron (Fe) and manganese (Mn), has emerged as a global concern due to its profound impact on environmental ecosystems and public health (Fernandez-Luqueno et al., 2013; Mukherjee et al., 2021; Shen et al., 2023; Yang et al., 2022). The escalation of industrial activities, mining operations and urbanization has unleashed a torrent of heavy metals into aquatic systems, resulting in severe pollution that jeopardizes ecosystems and human well-being (Danielopol et al., 2003; Jin et al., 2022; Meng et al., 2022). Traditional methods for heavy metal removal from polluted water, such as precipitation, coagulation and membrane filtration, often grapple with limitations in terms of efficiency and cost-effectiveness (Punia et al., 2022). Consequently, there is an urgent call for innovative and effective technologies to combat this challenge.
In recent years, membrane-based separation processes have risen to prominence in the realm of water purification, owing to their exceptional selectivity, cost-efficiency and operational simplicity (Jin et al., 2022; Lee et al., 2016). Membrane filtration, in particular, offers the advantage of continuous operation, which not only minimizes energy consumption but also guarantees a consistent and efficient purification process (Lee et al., 2016). This technology has witnessed remarkable advancements in materials and techniques, establishing itself as an indispensable component in diverse industrial processes, notably in wastewater treatment (Zhang and Chung, 2017; Ren et al., 2023).
Within the spectrum of materials employed for membrane construction, polymeric polymers, including cellulose, polyamide and polyethersulphone (PES), have gained popularity due to their porous structure, mechanical resilience and cost-effectiveness (Huang et al., 2015; Yadav et al., 2020). Among these, PES membranes have emerged as promising candidates for water treatment applications, primarily because of their high durability and permeability (Huang et al., 2015; Ganesh et al., 2013; Jin et al., 2013; Wang et al., 2022). However, a persistent challenge remains in augmenting the adsorption capacity and selectivity of PES membranes for heavy metal ions.
One promising avenue to enhance membrane adsorption properties is surface modification using nanomaterials. The integration of nanomaterials into polymeric membranes has shown great potential in addressing the limitations associated with conventional polymeric membranes, making them more adept at removing heavy metal ions and other contaminants. These nanomaterials, such as GO, carbon nanotubes and various nanoparticles, endow membranes with improved properties, including enhanced adsorption capacity, mechanical strength and hydrophilicity. Through these innovations, membrane technology has become a frontrunner in the quest for efficient heavy metal ion removal from water sources.
Nevertheless, polymeric membranes do have their inherent challenges, including suboptimal separation performance, limited water permeability, susceptibility to fouling and reduced lifespan (Ganesh et al., 2013; Hu and Mi, 2013). To surmount these obstacles, various strategies have been explored. One approach involves the incorporation of additives that enhance membrane hydrophilicity and water permeability. These modifications aim to mitigate the limitations associated with conventional polymeric membranes, paving the way for more effective heavy metal ion removal technologies.
GO, a derivative of graphene, is a versatile nanomaterial celebrated for its diverse properties and applications (Ganesh et al., 2013; Hu and Mi, 2013; Wang et al., 2022). With the two-dimensional structure, consisting of a single layer of carbon atoms arranged in a hexagonal lattice, GO boasts a substantial surface area. This extensive surface area is a boon for applications such as adsorption, catalysis and sensing, as it provides ample active sites for interactions (Wang et al., 2022). What sets GO apart is its exceptional flexibility when assembled into thin films or membranes (Hu and Mi, 2013). This attribute proves invaluable in applications like flexible electronics, sensors and barrier materials, as these films readily conform to diverse surfaces and structures (Marcano et al., 2010). Although thermal conductivity of GO is not as remarkable as pristine graphene, it still outperforms many materials, making it beneficial for heat management in electronics (Huang et al., 2011). GO’s impermeable structure equips it to serve as an effective barrier material, proficiently obstructing the passage of gases and liquids. This feature is indispensable in gas and liquid separation, packaging materials and protective coatings, where impermeability is paramount. GO is equally impressive in terms of mechanical properties. Its high tensile strength and stiffness, attributable to the covalent carbon-carbon bonds in the graphene lattice, render it a valuable candidate for reinforcing composite materials. Although GO’s electron mobility is generally lower than that of pristine graphene due to the presence of oxygen-containing functional groups, it still exhibits relatively high electron mobility compared to many other materials. This quality is pivotal in electronic applications like field-effect transistors and sensors. It is noteworthy that properties of GO can be tailored for specific applications through chemical functionalization and material engineering. This adaptability makes it a prized material in fields such as materials science, electronics, energy storage and environmental engineering. Figure 1 illustrates various properties of GO (Ganesh et al., 2013; Hu and Mi, 2013; Huang et al., 2011; Marcano et al., 2010; Wang et al., 2022).
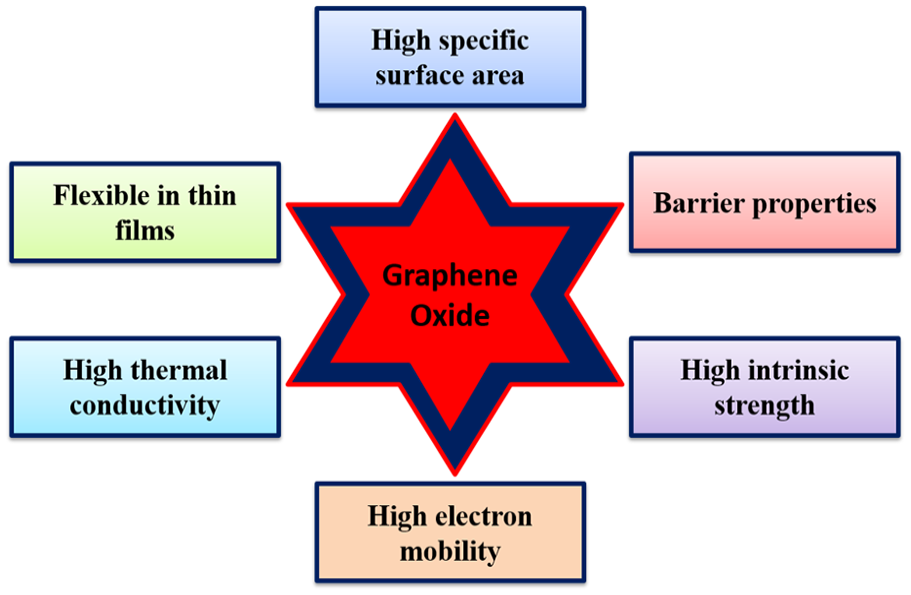
Some common properties of GO.
Several studies have illuminated the potential of GO-based composite membranes in addressing water contamination challenges (Cui et al., 2022; Hussain et al., 2023; Kamalam et al., 2023; Liu et al., 2020). For instance, the integration of GO into PES membranes has demonstrated improved salt and dye rejection percentages (Anand et al., 2018; Goyat et al., 2022a, 2022b; Koulivand et al., 2020). In line with these developments, the present study endeavours to harness the advantages of GO to engineer enhanced PES-based microfiltration membranes using the phase inversion technique (Gao et al., 2013; Kim et al., 2016; Wei et al., 2018).
This study aims to synthesize and characterize GO-decorated PES membranes and evaluate their efficiency in removing heavy metal ions, specifically Fe and Mn, from contaminated water. The surface modification of PES membranes with GO is anticipated to enhance their adsorption capacity, improving the overall efficiency of heavy metal ion removal compared to pristine PES membranes.
It is important to note that our research contributes to the United Nations Sustainable Development Goals (SDGs), specifically SDG 6, which focuses on ensuring the availability and sustainable management of water and sanitation for all. Thus, this research is achieving SDG 6 by providing an innovative solution for the removal of heavy metal ions, particularly iron (Fe) and manganese (Mn), from contaminated water sources. The use of GO-decorated PES membranes enhances the efficiency of water purification processes, supporting the sustainable management of water resources, which is a key component of SDG 6. By developing advanced materials like GO-decorated PES membranes, we are contributing to a more sustainable and circular approach to water treatment. These membranes can be used repeatedly, reducing the need for disposable filters and promoting the reuse and recycling of materials in the water purification process.
Therefore, through detailed characterization and adsorption experiments, this research seeks to provide valuable insights into the development of advanced membrane materials for efficient removal of heavy metal ions from contaminated water. The outcomes of this study could significantly contribute to addressing environmental pollution and ensuring access to clean and safe water resources, thus emphasizing the importance of this research in addressing a critical global challenge.
Experimental details
Materials
PES obtained from BASF Germany constituted the polymer matrix for the membranes. All chemicals employed were of analytical reagent (AR) grade and procured from Sigma-Aldrich, including natural graphite powder, concentrated sulphuric acid (H2SO4), orthophosphoric acid (H3PO4), hydrochloric acid (HCl), potassium permanganate (KMnO4), hydrogen peroxide (H2O2), ethanol (C2H5OH) and N,N-dimethylformamide (DMF). These reagents and solvents were used without further purification.
Synthesis of GO
The synthesis of GO was achieved by converting graphite powder into GO through a modified Hummers process as shown in Figure 2 (Goyat et al., 2022b; Marcano et al., 2010). Initially, 1 g of graphite powder was introduced into a mixture of concentrated acids (H2SO4, 45 mL/H3PO4, 5 mL). Subsequently, 6 g of KMnO4 was gradually added to the solution, which was then stirred for 10 hours on a hot plate at 60°C. After cooling to room temperature, 250 mL of ice-cold water was carefully added to the solution. To prevent further oxidation, 10 mL of H2O2 was added (Marcano et al., 2010). The resulting product was filtered, washed with distilled water, air-dried and stored for further use.

Visual observations of graphite powder before oxidation and GO after oxidation.
Preparation of PES/GO nanocomposite membranes
The fabrication of PES/GO nanocomposite membranes utilized the phase inversion technique (Junaidi et al., 2018, 2019). To initiate the creation of PES/GO nanocomposite membranes, a PES solution was prepared by dissolving 1000 mg of PES pellets in DMF at 60°C. Separately, 10 mg of GO was dispersed in 10 mL of DMF and subjected to ultrasonication using a Ultasonic Probe sonicator for 30 minutes to exfoliate the GO sheets. The GO suspension was then added to the PES solution at 60°C and stirred for 4 hours at room temperature. The resultant nanocomposite slurry was casted onto a glass plate using a doctor blade. The nanocomposite membrane was formed by swiftly immersing the glass plate in deionized water. Afterward, the membranes were washed and air-dried in an oven.
Characterization of GO and embedded membrane
The synthesized material’s characteristics were assessed using a ultraviolet–visible (UV–-Vis) spectrophotometer. Chemical structural features, morphology and functional groups present in the material were examined using FTIR spectroscopy and field-emission scanning electron microscopy (FESEM). Hydrophilicity was assessed through contact angle measurements, whereas pore size analysis and morphological examination were performed to understand membrane properties.
Heavy metal ion uptake via filtration setup
The efficiency of the GO-embedded PES membranes in capturing heavy metal ions (HMIs) was evaluated through filtration experiments. The filtration process was carried out over four to five cycles to gage the membrane’s performance. In each cycle, 2.0 L of water was passed through the membrane. Following each cycle, the membranes were cleansed with deionized water to eliminate any residual solution, rendering them ready for subsequent cycles.
Heavy metal uptake study
Stock solutions of Fe2+ and Mn2+ HMIs at 1000 ppm were prepared using ferric chloride and manganese acetate, respectively, in 1000 mL of deionized water. These stock solutions were further diluted to obtain 100 ppm solutions. The pH of the solutions was adjusted using H2SO4 and NaOH solutions. At ambient temperature, the diluted solutions were subjected to filtration through the prepared membrane using a filtration assembly. The removal of Fe2+ and Mn2+ HMIs in the permeate was quantified using an atomic absorption spectrophotometer. The percentage removal of metal ions was calculated using equation (1):

where,
Results and discussion
Characterization of GO and its embedded membrane
Spectroscopic and morphological characterizations of GO
The successful synthesis of GO was confirmed through various spectroscopic techniques, shedding light on its structural attributes. UV–Vis spectroscopy unveiled the characteristic absorbance peaks associated with π→π* transitions in conjugated systems (Liu et al., 2022; Yadav and Lochab, 2019). The absorption peak observed at
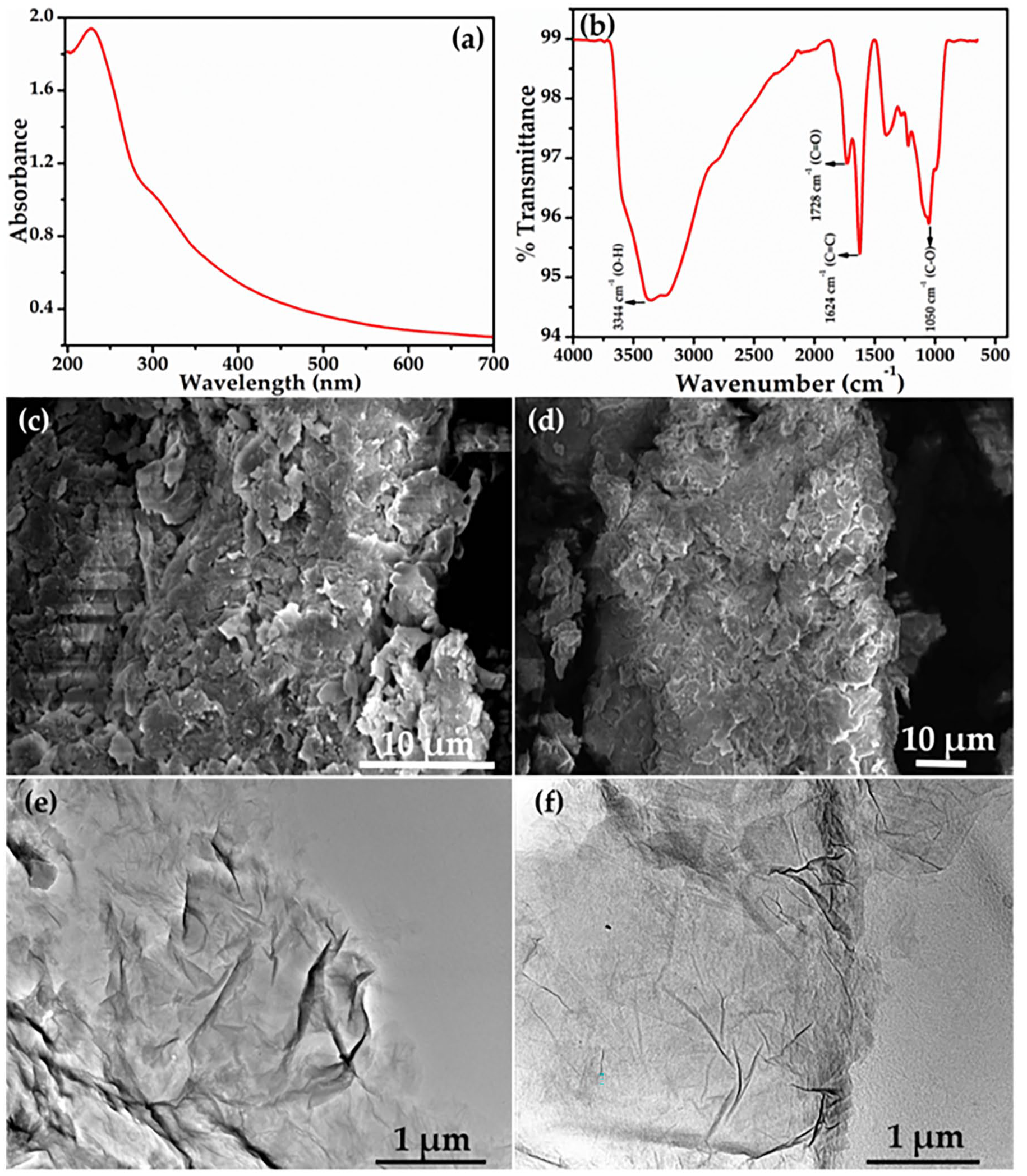
Typical (a) UV–Vis spectrum, (b) FTIR spectrum, (c, d) FESEM, and (e, f) transmission electron microscopy (TEM) images of as-prepared GO nanosheets.
FESEM was harnessed to delve into the intricate morphology of the synthesized GO, unravelling its structural intricacies. The FESEM images depicted a sheet-like structure marked by a substantial surface area (Figure 3(c and d)). The creases and folds scattered across the GO surfaces imparted a distinct crumpled appearance, reminiscent of layered structures. This unique morphology alluded to the stacked arrangement of GO layers, contributing to its multi-layer configuration (Huang et al., 2011). With a well-characterized GO structure in hand, the material was seamlessly integrated into the membrane matrix, laying the groundwork for subsequent morphological, hydrophilicity, and porosity investigations. The transmission electron microscopy (TEM) observations provide valuable insights into the structural properties of the GO. The TEM images presented in Figure 3(e and f) offer a comprehensive view of the prepared GO. Through these TEM images, we gain valuable insights into the intricate morphological attributes of the GO (Dave et al., 2016). Notably, the images distinctly showcase the characteristic appearance of GO, which adopt a flake-like configuration with a transparent nature. Upon closer examination, it becomes evident that the surfaces of the GO display a textured and nuanced structure. This is particularly evident through the presence of visible wrinkles and folds that are distributed randomly across the GO framework (Goyat et al., 2022b). Furthermore, the observed size distribution of the GONs falls within the micrometer scale, aligning with the intended nanosheet morphology. This consistency in size further supports the successful synthesis and formation of the desired GON structure (Aliyev et al., 2019; Dave et al., 2016; Goyat et al., 2022b; Marcano et al., 2010).
Morphological and compositional characterizations of GO-PES membrane
Leveraging FESEM, a deeper dive into the morphological traits of the GO-embedded PES membrane was undertaken (Figure 4(a and b)) (Junaidi et al., 2018, 2019; Kim et al., 2016). The membrane exhibited a remarkable porous architecture adorned with numerous micro-scale pores, a structural design that magnified its hydrophilicity and facilitated enhanced water flux (Junaidi et al., 2018). The incorporation of GO was notably correlated with an elevation in pore count and size within the membrane structure, effectively contributing to the amplification of water permeability and filtration efficiency (Junaidi et al., 2018, 2019; Kim et al., 2016). Further, the UV–Vis spectrum of GO-decorated PES would likely show characteristics about peaks or transitions among the conjugated systems. Herein, GO exhibits a distinct UV–Vis adsorption peak around 258 nm. This peak arises from the π→π* transitions within the GO structure. Concurrently, in the context of the PES membrane, the presence of a UV–Vis adsorption peak around 290 nm is anticipated. This peak is attributed to the aromatic rings within the PES structure (Figure 2(c)) (Ly et al., 2020; Khan et al., 2016). The positions of the peaks may vary due to some factors such as degree of decoration and experimental compositions.
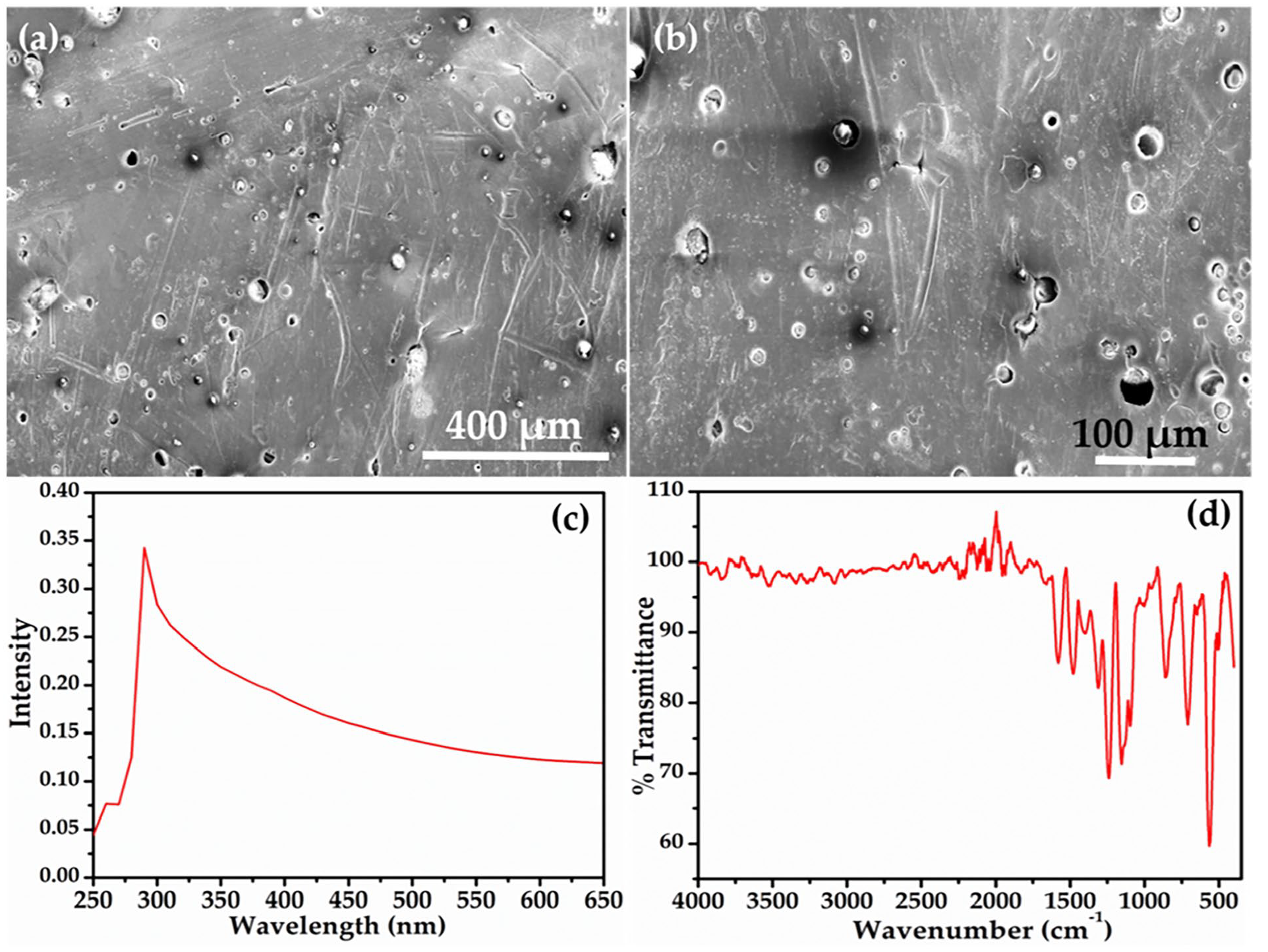
Typical (a, b) FESEM Images, (c) UV–Vis spectrum, and (d) FTIR spectrum of as-prepared PES-GO membrane.
The FTIR analysis of the prepared membrane was imperative in deciphering its chemical composition and the intricate web of functional groups adorning its surface (Figure 4(d)). While the major peaks were attributed to PES, which is the base material used for the membrane fabrication, the spectrum revealed additional signatures. Prominent absorption bands at 1158 and 1100 cm−1 corresponded to the symmetric and asymmetric absorption of -SO2 groups, characteristic of the membrane’s composition (Belfer et al., 2000; Klaysom et al., 2011). Furthermore, peaks within the spectral range of 3300–3400 cm−1 were indicative of hydroxyl moieties within the GO functional groups. The FTIR analysis effectively underscored the successful integration of GO into the membrane’s structure (Belfer et al., 2000; Klaysom et al., 2011).
Hydrophilicity and porosity analysis
Hydrophilicity assessment is crucial for membrane functionality, primarily governed by contact angle measurements. The prepared membrane’s contact angle was determined after drying at 25°C for 2 hours. The contact angle of water droplets on the GO-embedded PES membrane’s surface was measured using a goniometer. Traditionally, pure PES membranes have been recognized as hydrophobic, restricting water passage (Ahmad et al., 2013; Fu and Zhang, 2019). In contrast, the contact angle of the GO-PES membrane was 64°, indicative of its hydrophilic nature. This alteration resulted from the addition of GO, which effectively reduced the contact angle, thus conferring hydrophilicity to the membrane. Consequently, incorporation of GO particles augmented water flux, pore size, permeability, and hydrophilicity, enhancing the membrane’s filtration performance (Ahmad et al., 2013; Fu and Zhang, 2019). This behaviour could be attributed to the presence of oxygen-containing polar moieties, particularly carbonyl groups, on the GO surface, as validated by FTIR spectra (Goyat et al., 2022b; Marcano et al., 2010). Ultimately, the membrane met the requisite criteria for efficient water filtration, primarily attributed to its newfound hydrophilic behaviour.
Uptake of HMIs via filtration assembly using GO-PES membrane
The synthesized GO-embedded PES membranes were subjected to a rigorous investigation, assessing their efficacy in the uptake of HMIs, particularly Fe2+ and Mn2+metal ions. Employing a tailored filtration assembly, a series of experiments were conducted to ascertain the membrane’s prowess in removing these potentially hazardous metal ions from solution. The experiments involved the filtration of 100 ppm concentrations of Fe2+ and Mn2+ metal ion solutions consecutively through the GO-PES membrane using filtration assembly. The collected permeate samples, segregated into 100.0 mL portions, were meticulously analysed for residual metal ion concentrations.
The assessment of the percentage removal efficiency was facilitated through the utilization of Atomic Absorption Spectroscopy (AAS), a robust analytical technique known for its precision and accuracy in quantifying trace elements within a sample (Anwar et al., 2019; Das et al., 2009). The AAS analysis allowed for the determination of the percentage removal of Fe2+ and Mn2+ ions in the permeate after each filtration cycle. This sequential evaluation enabled insights into the membrane’s capacity to effectively capture and retain HMIs over multiple filtration iterations.
The initial filtration cycle yielded striking results, with the GO-PES membrane exhibiting remarkable efficiency in the removal of Fe2+ and Mn2+ ions. The percentage removal efficiency after the first cycle was noted to be 94% for Mn2+ ions and an impressive 93.6% for Fe2+ ions. These findings are indeed noteworthy, signifying the membrane’s efficacy in conserving heavy metals from the filtration process. The ability of the GO-PES membrane to achieve such substantial removal rates underscores its potential as an effective medium for the treatment of metal-laden waters.
Figure 5 visually encapsulates the dynamic behaviour of the membrane’s performance over subsequent filtration cycles. The presented graph highlights the percentage removal of HMIs as a function of filtration cycles. As the cycles progress, a gradual reduction in the efficiency of heavy metal absorption is evident (Chai et al., 2021). This reduction can be attributed to factors such as the clogging of membrane pores or the saturation of active surface sites on the membrane’s structure (Al-Amoudi and Lovitt, 2007; Lalia et al., 2013; Purkait et al., 2018). The diminishing removal efficiency highlights the importance of periodic membrane maintenance and regeneration to ensure consistent and sustained performance throughout extended filtration processes.
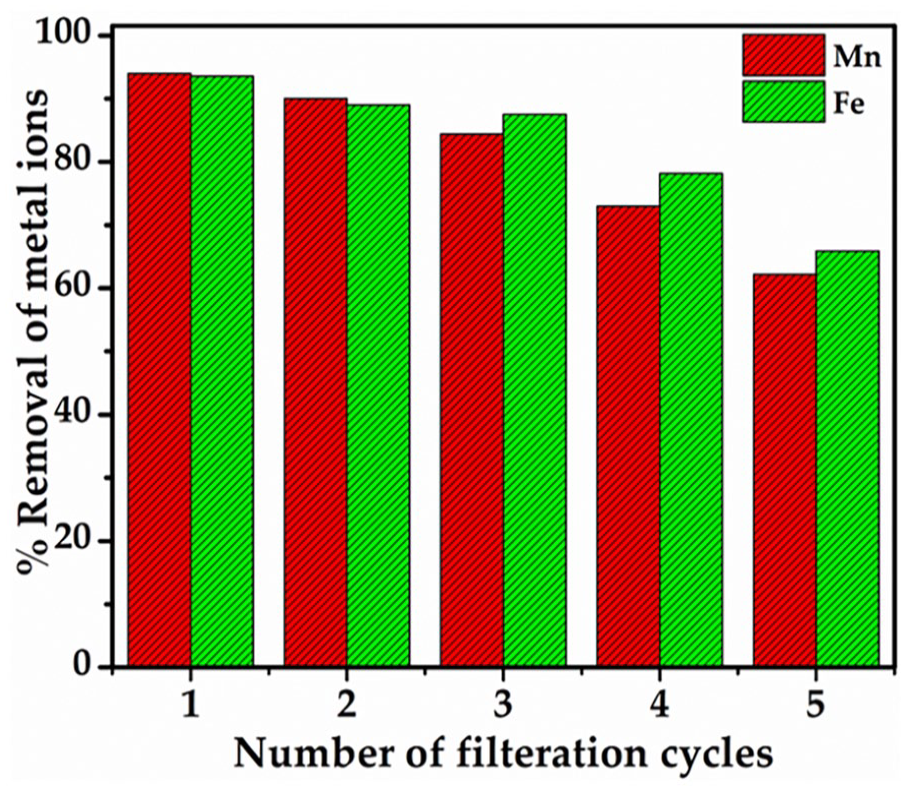
Graph showing the percentage removal of the HMIs.
The outcomes of these filtration experiments substantiate the GO-embedded PES membrane’s efficacy in heavy metal ion removal, providing a solid foundation for its potential application in water purification and treatment scenarios. The demonstrated conservation rates for Fe2+ and Mn2+ ions underscore the membrane’s relevance in addressing environmental contamination arising from heavy metal pollution. However, further investigations could delve into exploring the membrane’s performance against a broader spectrum of HMIs and evaluating its robustness in real-world water treatment applications. Moreover, optimizing membrane regeneration techniques could potentially mitigate the decline in removal efficiency observed across filtration cycles, enhancing the membrane’s overall sustainability and longevity in practical scenarios.
Table 1 presents a comparative assessment of composite membranes in terms of their effectiveness in removing various metal ions from water. It is evident that the amalgamation of distinct materials in these composite membranes results in varied metal ion removal efficiencies. Prior research endeavours involving diverse polymer types combined with different composites have yielded membranes with removal capacities ranging from 60% to 86% (Alotaibi et al., 2021; Ibrahim et al., 2022; Karki and Ingole, 2022; Marjani et al., 2020). However, the present study introduces a novel approach by incorporating GO into a PES matrix, demonstrating exceptional performance in the removal of metal ions.
Comparative heavy metal ion removal efficiency of the composite membranes.
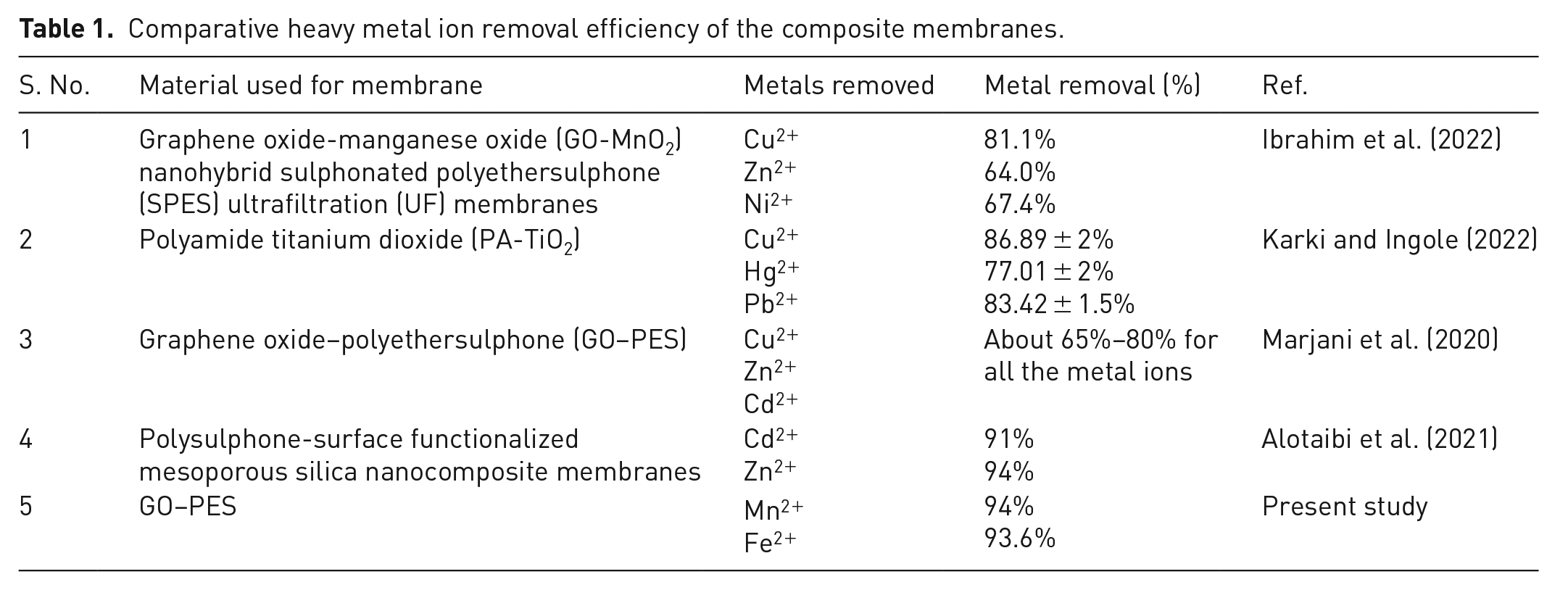
In this study, we synthesized and incorporated GO into the PES membrane, showcasing remarkable results. The developed composite membrane exhibited exceptional heavy metal ion removal efficiency, with an astounding 94% removal rate for Mn ions and an impressive 93.6% for Fe ions in the initial filtration cycle. Notably, the membrane’s performance remained consistently high even after five consecutive filtration cycles, retaining a metal removal capacity of 50%. These results underscore the superior capability of the GO-decorated PES membrane to effectively remove HMIs from contaminated water sources, outperforming many other composite membranes previously reported in the literature. The outstanding performance of this novel composite membrane holds promise for its practical application in water treatment processes and environmental remediation, addressing critical challenges related to heavy metal ion pollution in water bodies. This significant advancement in membrane technology highlights the potential of the GO-PES composite membrane to contribute to the development of efficient and sustainable solutions for water purification and environmental protection. The study represents a significant step forward in the quest for innovative materials that can address the pressing global issue of heavy metal contamination in water sources.
Mechanism underlying HMI adsorption
The adsorption process of HMIs onto the GO decorated PES membrane is intricately governed by a complex interplay of diverse interactions, ultimately leading to highly efficient metal ion removal (Ayub and Othman, 2023). GO, renowned for its extensive specific surface area and the presence of diverse functional groups, such as epoxy, carboxy, and keto moieties, exhibits remarkable versatility in its interactions with metal ions (Goyat et al., 2022b; Marcano et al., 2010). These interactions encompass electrostatic attraction, complexation, and chemical bonding, collectively orchestrating the multifaceted adsorption phenomenon. The unique surface properties of the PES membrane further contribute to the stability and dispersion of GO particles, thereby synergistically optimizing the uptake of HMIs from water sources (Ayub and Othman, 2023; Giwa and Hasan, 2020).
The remarkable properties of GO play a pivotal role in the adsorption mechanism. The vast surface area inherent to GO nanosheets provides an abundance of binding sites for metal ions. The functional groups present on GO, particularly the epoxy, carboxy, and keto moieties, actively participate in intricate interactions with metal ions (Ayub and Othman, 2023; Giwa and Hasan, 2020; Khan et al., 2021; Moradi et al., 2020). Electrostatic forces come into play as metal ions are drawn toward the charged functional groups residing on the surface of GO (Moradi et al., 2020). Furthermore, the capacity of these functional groups to form coordination complexes with metal ions results in the phenomenon of complexation (Blesa et al., 2000). This complexation interaction involves the reversible binding of metal ions to the functional groups, significantly contributing to the efficient removal of HMIs (Blesa et al., 2000).
The complex interplay of these interactions creates a dynamic equilibrium between metal ions in the solution and those adsorbed on the GO-decorated PES membrane. This equilibrium ensures the continual adsorption of HMIs from the surrounding water, ultimately leading to the high efficiency observed in our study. Moreover, the intrinsic properties of the PES membrane, including its porous structure and enhanced hydrophilicity, further enhance the overall adsorption process. The collaborative effect of GO and PES in optimizing the adsorption mechanism sheds light on the immense potential of these composite membranes as efficient adsorbents for heavy metal removal from water sources. This understanding not only advances the field of water purification but also offers promising solutions for environmental remediation and the protection of ecosystems.
Furthermore, functional groups on GO can form covalent bonds with metal ions, leading to their immobilization on the membrane surface (Ahmad et al., 2020; Duru et al., 2016). The strong binding resulting from these chemical interactions ensures the stability of the adsorbed metal ions. Here, the intricate chemical interactions that transpire between the GO surface and the HMIs in the water encompass various chemical forces, including but not limited to electrostatic attractions, coordination bonding, and complexation phenomena. These mechanisms contribute to the overall adsorption process, where HMIs are effectively captured by the GO-modified PES membrane (Moradi et al., 2020). The significance of the strong binding resulting from these chemical interactions lies in the stability it confers to the adsorbed metal ions on the surface of the membrane (Blesa et al., 2000). This stability is pivotal for preventing the desorption or release of these metal ions back into the water phase. Therefore, this is crucial for preventing their desorption back into the water phase (Ahmad et al., 2020; Blesa et al., 2000; Moradi et al., 2020).
The introduction of GO into the PES matrix establishes a mutually beneficial relationship, with each material enhancing the performance of the other. The PES membrane’s intrinsic characteristics, such as its surface morphology and hydrophilicity, create an ideal environment for the dispersion and attachment of GO particles. The porous structure of the membrane acts as a scaffold, effectively preventing the aggregation of GO and bolstering its stability during the adsorption process. This synergistic collaboration between GO and PES culminates in a substantial enhancement of the composite membrane’s adsorption efficiency (Ahmad et al., 2020; Blesa et al., 2000; Duru et al., 2016; Khan et al., 2021; Moradi et al., 2020).
The adsorption of HMIs onto the GO-decorated PES membrane is a multifaceted process characterized by intricate interactions. The exceptional specific surface area of GO, its functional groups, and its electrostatic properties work in harmony to facilitate the efficient capture of metal ions through a combination of electrostatic attraction, complexation, and chemical bonding (Ahmad et al., 2020; Blesa et al., 2000; Duru et al., 2016; Khan et al., 2021; Lim et al., 2017; Moradi et al., 2020). The integration of GO with the PES membrane not only optimizes the stability and uniform dispersion of GO but also significantly augments the overall adsorption capacity.
This comprehensive insight into the adsorption mechanism provides valuable clarity on the potential of GO-PES composite membranes as highly effective adsorbents for the removal of heavy metals from water sources. It offers promising solutions for water purification and environmental remediation, addressing the critical need to combat water pollution and protect ecosystems (Banti et al., 2020; Lim et al., 2017; Naddeo et al., 2023; Tsangas et al., 2023; Zorpas et al., 2021).
By elucidating the synergistic relationship between GO and PES and the intricate mechanisms governing heavy metal ion adsorption, our research contributes to the advancement of efficient and sustainable water treatment technologies, aligning with the global imperative for clean and safe water resources.
Conclusions
This study successfully synthesized GO through an improved Hummer’s method and integrated it into a PES membrane matrix. The investigation into GO-decorated PES membranes for the removal of HMIs, specifically iron (Fe) and manganese (Mn), from contaminated water has yielded promising results and significant insights. Characterization techniques including scanning electron microscopy (SEM), FTIR spectroscopy and ultraviolet–visible (UV–Vis) spectroscopy confirmed the successful GO synthesis. The distinct
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Deanship of Scientific Research at Najran University, Najran, Kingdom of Saudi Arabia for funding under the Research Group funding programme Grant no. NU/RG/SERC/12/36.
