Abstract
This mini-review aims at proving that waste-to-energy (WtE) is an essential cornerstone for circular economy (CE). Based on literature, the history of thermal waste treatment over the last 150 years is investigated, from open burning to WtE with resource recovery and final sink function. The results show that in the past incineration solved the issues it was designed for but often created new and sometimes even worse problems: The introduction of incineration in the 19th century improved urban sanitation, decreased waste volume and prolonged operational life of landfills. But it also polluted the environment, triggering an unprecedented scientific and engineering effort of all stakeholders. Today, WtE is one of the best investigated and optimized technologies in waste management. It enables the recovery of energy as heat and electric power and facilitates the ‘cleaning’ of cycles by the destruction of hazardous organic substances. Recent developments in resource recovery from WtE residues allow to recycle metals and, in the case of sewage sludge, even phosphorus by thermal recycling. Combined with carbon capture and storage technology, WtE stands for a quantifiable contribution to greenhouse gas reduction. Today, WtE is indispensable to reach the goals of CE, namely recycling of energy and materials, supplying safe final sinks for persistent organic substances and minimizing the need for sinks for hazardous inorganic substances.
Keywords
Introduction
This mini-review is a contribution to the 40th anniversary edition of ‘Waste Management and Research’ (WM&R). Thermal waste treatment has been a prominent topic during the past four decades, hence it makes sense to include this subject in the jubilee edition. Despite the long history of waste incineration dating back to 1874, there are still controversial opinions about this technology (e.g. Tangri, 2003; Zero Waste, 2017). Scepticism can be explained by local experiences with emissions from older incineration plants (Greenberg et al., 1977, 1978a), economic boundary conditions (Brunner and Fellner, 2007), not in my backyard NIMBY-syndrome (Brion, 1991) or general ecological and resource concerns (Zero Waste, 2017). In the past, a large potential for harmful emissions (particulate matter, heavy metals, dioxins/furans and others) has been documented, with adverse effects on air, soil and water, sometimes leading to health issues in the vicinity of incineration facilities (Brunner and Zobrist, 1983; Pudill and Weinand, 1981). Other opponents argue that the focus of waste management should be on sustainable consumption patterns, prevention, reduction at source and recycling. If waste is seen as a valuable energy source and treated in waste-to-energy (WtE) plants with efficient energy recovery, there might be less incentive to reduce waste and promote recycling. WtE might discourage recycling efforts, and thus it should be considered as a last resort (Zero Waste, 2017). In addition, incineration is seen as a source of carbon dioxide (CO2) contributing to climate change. And residues from waste incineration such as bottom ash and air pollution control (APC) residues have been recognized as significant carriers of toxic constituents requiring after-treatment and final storage (Brunner and Baccini, 1987; Lindquist, 1986).
A positive outlook on thermal waste treatment is often displayed by supporters who appreciate the reduction of waste going to landfills: precious space is saved, the release of methane and other greenhouse gases is minimized, and high flows of organic and inorganic substances in landfill leachates are prevented. In addition, WtE plants produce electricity and/or heat, which is seen as a valuable benefit if the process is cost-effective, and the emissions are strictly controlled (Khan et al., 2022). More recently, proponents point out that, if amended by thermal recycling (see below) WtE could play a significant role in supporting circular economy (CE) by extracting not only energy but also valuable metal resources such as aluminium, copper, gold, silver and others.
In view of the ambiguous situation presented above, the goal of this article is to provide an impartial base about the power and limits of advanced WtE for policy decisions in waste and resource management. It is not a technical article about incineration, but it refers to more than 50 publications that constitute the technical background (e.g. Brunner, 1996; Buekens, 2013; ISWA, 2022; Vehlow, 2013a; Wang et al., 2016). Based on the main authors in this field, the literature on thermal waste treatment is reviewed. Firstly, goals and actual performance of thermal waste treatment over time are assessed, and secondly the contribution of WtE to future CE is evaluated. For information given in Quicker et al. (2017), we focus on WtE by grate furnaces because for municipal solid waste (MSW) this technology is the most developed, is highly dependable, and has been by far most widely applied (Rogoff and Screve, 2011). Alternative thermal processes are not included. Nevertheless, most conclusions apply to other state-of-the-art thermal technologies such as fluidized bed, rotary kilns, etc. and for other waste fractions such as sewage sludge and special industrial waste, too. But for reasons of limited space, and not as a value judgement, other technologies capable of using refuse derived fuels, such as cement kilns, steel furnaces and the like are not considered here.
The value of this article lies in a comprehensive evaluation of the potential and limits of thermal waste treatment for CE. It confirms the articles showing how and why CE requires thermal waste treatment (Boloy et al., 2021; Lu, 2018; Van Caneghem et al., 2019), and it is the only one that puts WtE in a historical context of the search for the ‘final sink’, a place or process where hazardous materials can be disposed of.
Materials and methods
Three research questions are addressed: (1) How did objectives and practice of WtE evolve over the last 150 years? (2) What are the main goals of CE, particularly with respect to materials that cannot be recycled? (3) Why is WtE imperative for CE?
The following approaches are taken: on one hand, the 80 years of practical, regulatory and scientific experience of the authors in the field of thermal waste treatment are used for investigating into goals and practice of WtE. On the other hand, based on this expertise, specific literature searches are performed, such as screening electronic databases holding mostly English but also foreign language articles. Articles published on WtE on the internet and particularly in WM&R are extracted by Google Search and Scopus. In addition, queries by the multimodal large language model named Generative Pre-trained Transformer 4 (GPT-4) within specific areas of the research questions are applied. Whenever available, review articles are used to extract specific information about goals, functions, health effects, environmental performance and resource recovery by WtE. Hence, this mini-review comes close to a meta study of existing reviews.
Regarding CE, official definitions from the European Union (EU) as well as academic and applied articles highlighting CE strategies are reviewed. To examine the relationship between CE and thermal waste treatment, GPT-4 is used again to support the literature search. Special attention is given to the so-called ‘sink’ concept (Baccini, 1989; Kral et al., 2019). The concept is based on the fact that non-recyclables such as hazardous legacy substances require safe final disposal such as mineralization or safe underground storage. Finally, conclusions regarding the significance of thermal processes for CE are drawn from the information collected.
Results and discussion
Evolution of waste incineration
The first research question is answered along the timeline presented in Figure 1: goals, means and practical outcomes of thermal waste treatment are discussed in view of the goals of waste management (cf. Brunner et al., 2023).

Development of municipal waste incineration during the last 150 years, from open burning to WtE including WtER and WtER-CCS. Note that albeit this presentation insinuates a linear evolution, on a global scale the progress is highly uneven: although today WtE with emission control is state-of-the-art in affluent countries, open burning continues to prevail in low-income countries, and emission control is just starting in others. The latest developments such as WtER with carbon capture and storage WtER-CCS are still rare single applications that have passed the experimental state just recently.
Open burning: The term ‘open burning’ names the intended or accidental burning of wastes in the open air, usually without any technical device (Ramadan et al., 2022). Toxic off-streams such as smoke, fumes and leachates are directly released to the environment, and potentially hazardous residues remain. Such open burning is quite common: As long as humankind remained a marginal species on the planet, burning of wastes was not necessary. Natural processes such as aerobic and anaerobic degradation and mineralization (transformation of organic substances to inorganic carbon and CO2) converted the small flow of biogenic materials from human habitat into simple compounds that were of little harm to early humans and the environment. With the transition from nomadic to agricultural lifestyle and the development of first human settlements, population density and per capita turnover increased. As a result, the mass flow of wastes increased too. Open dumping in specially designed areas became the favourite means of waste disposal. These open dumps sometimes caught fire, accidentally or intentionally. Open burning reduces waste volume, allows to partially control pests and reduces water pollution, three benefits that became increasingly important with growing settlements. According to Vehlow (2013b), first open burning of wastes using ashes for fertilizing purposes was mentioned about 1000 BC in Jerusalem.
Open burning of waste became a widespread practice in many parts of the world (Das, 2022). In countries with low gross domestic product, it is still practiced as an unofficial and inexpensive means to dispose of waste. Short- and long-term environmental pollution from such practice is immense. The chemical content of modern consumer goods and the lack of any technical process to control oxidation and air pollution are the reasons why this practice is a major source of atmospheric pollution in emerging economies. If 1 tonne of waste is burned or smouldered in an open dump, the emissions are at least three orders of magnitude (99.9%) higher than if incinerated in a WtE plant with modern APC technologies. Hence, although open burning fulfilled the three original aims of (1) reduction of space for dumping, (2) pest control by mineralization of pathogens and organic material and (3) reduction of water pollution by decreasing organic leachates, it caused other severe problems: air and subsequent soil and water pollution.
In countries with modern waste management infrastructure, intentional open burning is very rare, but unintentional open burning of waste can still be observed: uncontrolled (Ghosal, 2022) and even controlled landfills (Morales et al., 2018), storages of single waste materials such as tires (Deutsche Welle, 2016; Ettala et al., 1996), recycling centres and packaging materials (Jimenez et al., 2023; O’Connor and Wise, 2021), they all are prone to catch fire if not special precaution is taken to prevent fire hazards.
MSW incineration
Literature is not conclusive about the first MSW incinerator: Some sources claim that the first MSW has been incinerated in 1870 in Paddington, London, in a retrofitted coal incinerator (Zakaria et al., 2021). The performance was poor, with bad odours due to smouldering waste, and the plant was not successful. Other sources name the so called ‘Destructor’ of Nottingham, England as the first MSW incinerator. This furnace has been designed and patented by Alfred Fryer and built by Manlove, Alliott & Co. Ltd in 1874 (Grace’s Guide, 2017). The incineration plant must have been quite successful because the same company built similar plants in Manchester (1876), Shoreditch (first MSW incinerator with heat recovery), Cambridge, Liverpool and other English towns. In the late 19th century, trials started to replace coal by MSW to power the coming Industrial Revolution.
Although densely populated England was the pioneer in the early days of incineration, other countries and towns followed shortly, namely the United States (1885 Governor’s Island, New York, 1885 Allegheny, Pennsylvania) and Germany that hosted the first incinerator on the European continent (1896 in Bullerdeich, Hamburg). Here, again, the triggering factor was the outbreak of cholera in 1892 that killed 8000 inhabitants within a single week. Because of fear of contagion and foreign matter, farmers refused to accept waste from Hamburg and its harbour for disposal. Landfilling as an alternative required land which became scarce in the vicinity of growing towns, landfills polluted surface and ground waters and uncontrolled landfills caught fire and had problems with odour and geotechnical stability. Hence, many countries such as Japan (1897), Denmark (1903 in Frederiksberg), Switzerland (1904 in Josefstrasse, Zürich) and Czech Republic (1905 in Brno) followed the example of England. At the turn of the century, more than 200 MSW incinerators were operating globally.
The main reasons for the introduction of incinerators were: (1) lack of sanitation with severe outbreaks of cholera and typhus caused by dumping waste on agricultural land, (2) growth of population, density, poverty and waste in the 19th century and (3) change in waste composition that threatened farming (glass, metal, others). Urban sanitation was a huge issue, with ten International Sanitary Conferences between 1851 and 1897 in major cities of the world, and with an ‘International Sanitary Convention’ aiming at global protocols to protect people from pandemic diseases. Effective urban sanitation became a distinguished responsibility for every modern city, waste collection was improved and incineration was introduced.
The first generation of incinerators had two goals: to destroy putrescible organic material and microorganisms contained in wastes and to decrease waste volume to be landfilled. As a result, the unsanitary wastes holding a myriad of malignant germs were transformed into a more or less sterile bottom ash with little odour and greatly reduced water pollution potential. Incineration reduced waste volume by a factor of 10, thus enabling much more waste to be disposed of in a given landfill space. In some cases, bottom ash was applied to fields, thus nutrients such as phosphorous and potassium were recycled to support food production. In other cases, bottom ash was upgraded to building materials (De Fodor, 1911). Such past use in constructions of roads and buildings may pose a future hazard (Joseph et al., 2020; Lichtensteiger and Zeltner, 1992). The fact that the early 20th century practice of using bottom ash as building material has not been continued, suggests that this building material was not successful in the long term. Unfortunately, few records exist that give a solid account of the early practice of ash application. Today, upgraded bottom ash is safe for recycling (Confederation of European Waste-to-Energy Plants (CEWEP), 2017; Chandler et al., 1997).
MSW incineration with emission control
The incineration plants built more than 120 years ago reached their goals, but they caused new and severe problems: Pollution of air, water and soil. For a long time, emissions from MSW incinerators were neglected. It was believed that high chimneys would lead to sufficient dilution, resulting in harmless concentrations on the ground level. Leachates from bottom ashes were little investigated, and filter ashes did not exist because of the lack of APC systems.
It should be noted that in the early years of incineration, waste management was a segmented field, and not treated as an entity. The consequences of the introduction of a new process, here controlled thermal waste treatment, on the entire management of wastes were not investigated. The holistic look at waste management was introduced towards the 1970s, when today’s goals for waste management were introduced: (1) protection of human health and the environment, (2) conservation of resources and (3) sustainability, defined as after-care-free waste management not depending on export in time and space. No longer was landfilling, composting, incineration or recycling a goal of waste management. These activities are just means to reach the overarching goals of environmental protection and resource conservation considering long time periods (Brunner et al., 2023). These aims make it impossible to shift problems from one sphere to another, for example, from soil to air. They demand that materials are followed through the whole chain from resources to many cycles of use to final disposal of inevitable wastes and emissions. Since they are based on scientific grounds, these objectives obey both the law of conservation of mass and the law of entropy. The latter means that material cycles cannot be closed, material will inevitably be lost from cycles, and that processes such as incineration or recycling are not able to make matter ‘disappear’.
Starting in the 1950s, incineration became increasingly popular because of the beginning age of environmental protection: It became clear that the enormous amounts of uncontrolled landfills were the source of severe water pollution. Incineration was seen as a solution to protect surface and ground water from waste. It has not been recognized yet that while the hygienic problems were solved, new challenges of inorganic (heavy metals, nitrogen oxides, sulphur oxides and hydrohalogen acids such as hydrochloric acid and hydrofluoric acid) and organic (dioxins, furans, total organic carbon (TOC)) compounds arouse. It should also be noted that waste management was the domain of civil engineers with little experience in chemical engineering. And suddenly, waste treatment processes were introduced that needed in-depth chemical knowledge of combustion and pollution control science. The new challenges such as the limit for mercury of 0.05 mgHg Nm−3 or dioxins (0.1 ng TEQ−1 Nm−3) from off-gas had to be solved with little experience from other fields. This required a new generation of chemical engineers for analysing, understanding and design of APC devices. This generational shift was also the reason for a certain reluctance of the people engaged in waste incineration to enter a new APC age. Consequently, new incinerators had to be amended by a chemical plant of about the same size as the oven in space, equipment, knowledge and investments.
The main emission problems of waste incineration without APC systems were: (1) damage to human, animals and environment due to stack emissions of heavy metals, acid gases, TOC and persistent hazardous organic compounds such as dioxins and furans, polycyclic aromatic hydrocarbons (PAHs) and polychlorinated biphenyls. (2) Flows of heavy metals and salts from the disposal of solid incineration residues on land and from the use of such materials as building materials. (3) Emissions of CO2 and traces of sulphur hexafluoride SF6 and other gases contributing to climate change. In general, high-quality studies of real damage in the vicinity of incinerators without APC exist, but they are scarce, and often hard to find and retrieve (Suess, 1987). The history of incineration, its emissions and failures has not been written yet. A recent review about the health impacts of waste incineration summarizes some of the environmental and human health issues of earlier incinerators (Tait et al., 2020). The authors state that more research is required to produce compelling evaluation results about WtE.
The few individual reports about severe incidents in the vicinity of old incinerators give a clear picture about the hazards of burning waste without or with inappropriate emission control. They document high loadings of dust and hazardous substances in workers engaged in operation and regular revisions of waste incinerators (Hebisch et al., 2008; Kuhlmann et al., 2011). Concentrations of heavy metals such as cadmium (Cd) can be significantly elevated in the blood of workers employed in incinerator plants (Pudill and Weinand, 1981). Animal fodder produced in the vicinity of an incinerator was highly contaminated by heavy metals (Zuber et al., 1975). Similar pollution of grassland and spinach has been found in the surroundings of incinerators by Rollier (1980). Other reports show the heavy metal contamination of forest roads constructed with bottom ash from incinerators. And they document the high loads of hazardous and persistent organic chemicals such as chlorinated dioxins and furans in emissions from incinerators (Buser et al., 1978).
These accounts show individual impacts of emissions from incinerators on human health and environment. Other articles compare flows of hazardous substances from all relevant sources of pollution in order to prioritize measures to reduce emissions on different spatial scales. Nriagu stated for the period before 1980 annual waste incineration emissions of 1.35 kilotonnes of Cd (Nriagu, 1980). Based on the total global emissions of Cd (7.2 kilotonnes year−1) by ore mining and production of zinc (2.8 kilotonnes year−1) and copper (1.53 kilotonnes year−1), he claimed that waste incineration alone causes nearly 20% of the global Cd emissions! Based on available global data about mining, consumption and disposal, Brunner and Baccini (1981) calculated global geogenic and anthropogenic Cd budgets for the 1970s. Anthropogenic flows of 17 kilotonnes year−1 are about three times larger than natural flows of 5.4 kilotonnes year−1. Because of the widespread use of Cd as stabilizer for polyvinylchloride (PVC), substantial amounts of Cd end up via waste incineration and atmosphere in the environment, namely soil and water. Thus, on a global level, the anthropogenic flow of Cd from waste incineration without APC device was about an order of magnitude larger than from natural flows!
On national and regional scales, comparable results were observed: Incinerators of the 1970s were a comparatively large source of pollution (Brunner and Zobrist, 1983). These surprise findings became more understandable when the total flow in combustible wastes was compared to the total national flow of elements such as Cd, chlorine, mercury and so on (Figure 2). Due to new products and changes in consumption, more goods made from PVC were used and discarded, and Cd as an additive in PVC increased in MSW too. Similarly, the flow of mercury in batteries and thermometers grew, and thus waste incineration became a dominant source of mercury on a national scale.
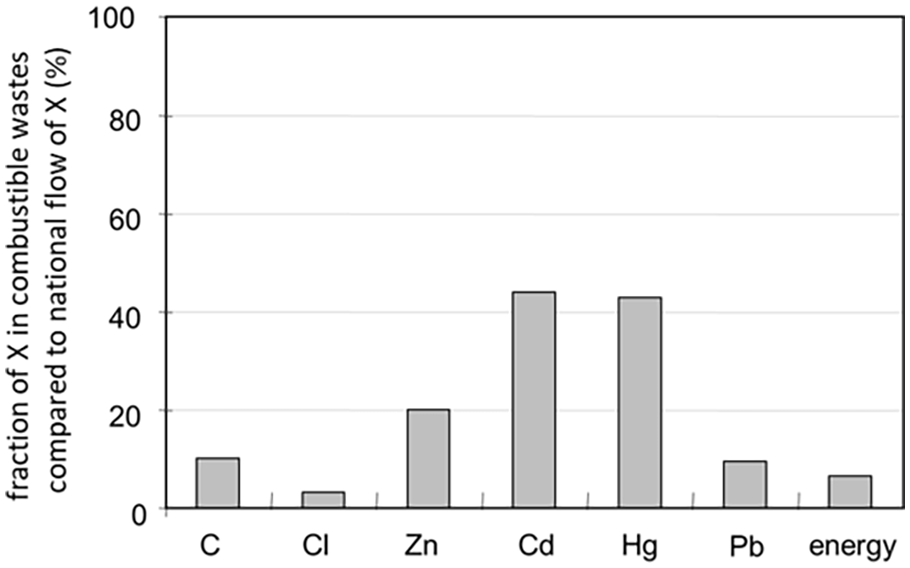
Fraction of selected elements and energy in combustible wastes compared to total national element and energy flows. Example of Switzerland in the 1980s.
Due to the large consumption of materials by modern societies, it became urgent to better understand and prevent emissions from waste incinerators. Some researchers focused on small particles in emissions from incinerators (Greenberg et al., 1978a, 1978b; Vogg, 1987). They discovered the enrichment of volatile (‘atmophile’) heavy metals such as Hg, Cd, Zn and Pb in fine dust of emissions from incinerators. Olie et al. (1977) and others found that highly toxic polychlorinated dibenzo-p-dioxins and dibenzofurans are formed in fly ash from waste incinerators, and that these chemicals concentrate on fine dust. These discoveries were the starting signals for in-depth research in the analysis, design, and optimization of advanced APC systems. It became obvious that the mere mass of dust emitted was not a sufficient criterion for waste incinerators because the most hazardous substances are concentrated in small aerosol particles. These are difficult to eliminate by the APC technology of the 1970s, consisting mostly of cyclones, electrostatic precipitators or simple scrubbers.
The necessary big step forward was also promoted by citizens groups and environmental non-governmental organizations (green NGOs) who actively opposed the construction and operation of incinerators. The joint power of green movements pointing out the problems, and of technology suppliers working out solutions finally turned out to be successful: Incineration with advanced APC systems became the best investigated field of waste management. To comply with modern APC standards, multiple stages are required for removing coarse and ultra-fine fly ash, heavy metals, inorganic acids and organic contaminants such as dioxins from flue gas. An excellent account of APC systems for waste incinerators is given in Vehlow (2015). The technological and environmental results of this progress are remarkable: Thanks to advanced APC technology, emissions to air can be reduced by two to three orders of magnitude (99–99.9%). Technologies such as selective non-catalytic reduction or catalytic reduction for the control of nitrogen oxides (DeNOx), electrostatic precipitators or bag filters for particulate removal with or without preceding injection of neutralization agents for acid gases abatement, venturi scrubbing with alkaline solutions, flue gas condensation for removal of fine particulates and gases, activated carbon treatments for mercury and dioxins and the like allow to clean off-gas to an extremely high degree. The earlier observation of high emissions in the vicinity of incinerators (Hu and Shy, 2001; Tait et al., 2020; Zuber et al., 1975) are superseded today by research results of modern, advanced WtE showing no negative impact on the environment: Damgaard et al. (2010) showed the drastic reduction of air emissions from WtE with the improvement of APC during the last 35 years. Remaining impacts can be offset by recovering energy, which otherwise is produced from fossil fuels. Van Dijk, van Doorn and van Alfen stated in their long-term (2004–2013) biomonitoring study on plants in the vicinity of Dutch waste incinerators that the emissions did not affect the quality of crops and cow milk with regard to selected heavy metals and PAHs, dioxins and furans (van Dijk et al., 2015). Equivalent results are reported by CEWEP (2019), de Titto and Savino (2019) and BMU (2005). Thus, the main and most often criticized issue of waste incineration, air pollution, has been resolved, albeit at high costs. At the same time, challenges such as contaminated bottom ash, hazardous residues from APC and waste waters from scrubbers and bottom ash come into focus and superseded the air pollution topic (cf. below). The drawback of the advancement of APC technology is the increase in total costs of waste incineration for both, investment, and operation. It can be estimated that APC with residue treatment accounts to about half of the cost of incineration. It is important to say that if for budgetary reasons APC devices are reduced, incineration cannot comply with the required advanced emission standards (Gebhardt, 2000)! This implies that economically weak regions may encounter difficulties in implementing waste incineration. Sometimes, revenue systems such as advance disposal fees or producer responsibility concepts may help to overcome such shortcomings.
Waste-to-energy
WtE stands for the conversion of waste into usable forms of energy such as heat, fuel and electricity. It includes processes such as incineration, co-incineration, gasification, pyrolysis, anaerobic digestion and landfill gas recovery. In the following, grate incineration as the most widely applied conversion technology of MSW into heat and electricity is discussed only. Waste has been used as an energy carrier since the beginning of waste incineration. At first, it was a means to supplement coal fired furnaces. Later, energy was extracted from incineration off-gas to cool the off-gas, thus enabling APC at lower temperatures. Today, energy recovery for heating and/or electricity generation is an essential, standard feature of state-of-the-art WtE. Substituting coal, oil and gas by MSW improves the ecological as well as the economic performance of incineration. In 2021, more than 2200 WtE plants were running worldwide, converting about 260 million tonnes year−1 of waste into useful energy (Gleis, 2018). For 2031, it is estimated that about 3000 WtE plants with a total capacity of 630 million tonnes year−1 will be operational globally (Havel, 2022). On a country scale, energy contained in non-recyclable burnable wastes amounts to about 5% of total primary energy consumed (cf. Figure 2). But for regional supply of heat, it can be significant and can contribute valuable band energy for the grid.
A reference document of the EU defines best available technology for WtE (Neuwahl et al., 2019). It covers the energy recovery from waste in incineration and co-incineration plants, and the treatment of resulting bottom ash, slags, filter residues and any liquid effluents. Modern WtE is considered a sophisticated, dependable and environmentally safe technology to treat MSW (Ardolino et al., 2020). The main advantage of WtE is the effective mineralization of even complex and heterogeneous waste mixtures, converting organic carbon reliably into useful energy, CO2, water and inorganic residues (Brunner, 2012). Because of the large size of bunkers, feed hoppers and grate furnaces of modern WtE plants, pretreatment of MSW is usually not required. This unique selling point contrasts with other waste processing such as mechanical-biological treatment or composting which are more susceptible to variations of input composition. Only exceptionally large objects may require shredding to avoid mechanical blockages.
WtER
Driven by increasing cost and scarcity of landfill space, the disposal of solid residues from WtE became a growing challenge. The problem was enhanced when it was recognized that both bottom ash and APC residues hold hazardous constituents and had to be treated in part as special waste (Brunner and Baccini, 1987). Hence, pathways were investigated how to reuse or treat WtE residues (Sakai and Hiraoka, 2000). Today, several processes are available for upgrading, reusing, stabilizing and disposing of bottom ash and filter residues (Quicker, 2018; Sakai and Hiraoka, 2000; Vehlow, 2011). The objective is to contribute to resource recovery on one hand, and to ‘clean’ material cycles of the CE on the other hand by destroying and/or immobilizing hazardous substances. Figure 2 shows the high fraction of national metal flows reaching MSW. If these metals are recovered from bottom ash and APC residues, incineration becomes a means to actively contribute to CE not only by energy recovery but also by materials recycling, and by ‘cleaning’ material cycles from toxic metals like Hg, Cd, Sb and Pb, too. The quantitative contribution may seem small, but the recovery is attractive and important because of the high ecologic and economic value of elements such as gold, silver, copper and aluminium. A life cycle assessment (LCA) study performed by Mehr et al. (2021) confirmed the large environmental benefits of the recovery of non-ferrous metals from bottom ash, especially Cu and Au. Compared to the savings by energy recovery (215 kg CO2-eq per tonne of MSW), enhanced metal recovery may save up to 140 kg CO2-eq per tonne of MSW. Further progress will increase these numbers by around 30% (Böni, 2020; Zenger, 2022). Altogether, Bunge estimates that ‘extended processing’ of MSW bottom ash equals about 15% of the cumulative environmental benefit of recycling measures for municipal waste in Switzerland (Bunge, 2017).
The full potential of WtE to recycle metals was figured out in a comprehensive substance flow analysis of precious metals and rare earth elements in Switzerland (Morf et al., 2013). Although metals, electric and electronic waste, glass, batteries and others are separately collected at high rates, this study proves substantial amounts of metals in the MSW input of the WtE plant. Many valuable substances, such as Au and Ag are enriched in specific outputs of bottom ash treatment (e.g. non-ferrous metal fractions) and can be recovered. The results of this pioneering analysis allow crucial improvements in metal reclamation from WtE plants. They have been successfully implemented in the Hinwil WtE plant recovering Au, Ag, Al, Cu, Fe, stainless steel, Pb, Pd, Zn and Fe (ZAV, 2023). The concept as described in Böni and Morf (2018) is applied in other WtE plants too. More work has been published by Muchova et al. (2009), Born (2018), Pérez-Martínez et al. (2019), Allegrini et al. (2015), Biganzoli and Grosso (2013), Biganzoli et al. (2014) and Gökelma et al. (2021). A review of the state-of-the-art of metal recovery from bottom ash is given in Šyc et al. (2020).
MSW and bottom ash are highly heterogeneous materials with inherent challenges in sampling, particularly (1) sampling correctness, (2) sampling bias and (3) sampling variance as defined in the theory of sampling (TOS) by Esbensen and Wagner (2014). The recent advances in bottom ash treatment have been made possible by new methodologies for representative sampling and analysis. The necessary solutions that fulfil TOS concept requirements are sophisticated and expensive and are presented in Skutan and Gloor (2014) and Skutan (2023).
Although the ecological and economical potential of metal recovery from APC residues is far less attractive than from bottom ash, it is important to close these cycles too: (1) for certain metals like Zn, the recoverable mass from waste and sewage sludge incineration plants represents substantial amounts of the national consumption (>20%) as calculated from references in (Meylan, 2017). (2) The recovery of hazardous heavy metals, like Cd, Pb, Hg and so on is important to avoid potential future emissions in landfills. Today, metal recovery from APC residues is state-of-the-art (e.g. Bunge et al., 2023; Bühler and Schlumberger, 2010).
WtER and CCS
Depending on the domestic energy production mix and the role of WtE in MSW management, modern WtE-plants emit 1–10% of total national CO2-emissions. From an environmental point of view, the last unresolved topic of WtE is: (1) the reduction of CO2 emissions of fossil origins and (2) the use of negative emission potentials by eliminating biogenic CO2 in the flue-gas applying carbon capture and storage (CCS) techniques. In Europe, first CO2 capture plants are both in operation and planned in Norway and in the Netherlands. The Norwegian Longship CCS Project (see https://ccsnorway.com/capture-studies/), initiated in 2014 and promoted by Gassnova SF, a state-owned enterprise under the Norwegian Ministry of Petroleum and Energy, was one of the global early movers to establish full-scale CCS for WtE in Hafslund Oslo Celsio (earlier Fortum Oslo Varme). In Duiven (NL), a large-scale WtE plant is capturing CO2 since 2019, and in Twente (NL) since 2021; 400,000 tonnes of CO2 will be captured from the Oslo WtE plant. Another WtE combined with CO2-capturing exists in Saga-City, Japan, operating since 2016, and applying the same technology as used for large US power plants. Switzerland plans CCS for all WtE works as a key element in achieving net zero greenhouse gas emissions by 2050 (Bundesrat, 2022). A new CO2 competence centre aims at gaining sufficient technical and economic expertise by building and operating a first 100,000 tonnes year−1 WtE plant before 2030 (ZAR, 2023a). The result will supply a decision base for CCS and WtE from both a technical, economic, ecologic and social perspective. A holistic evaluation such as in Bisinella et al. (2021) is essential. Their LCA modelling of WtE and CCS with monoethanolamine illustrates that despite energy penalties, CCS may lower the output of CO2-eq tonne−1 by 700 kg compared to WtE without CCS, quite a substantial reduction when compared to the medium emission of 1000 kg CO2 tonne−1 of MSW. Muslemani et al. (2023) conclude in their study about negative emissions from WtE and CCS that, if the existing European 100 million tonnes installed capacity of WtE plants were retrofitted with CCS technology, negative emissions in the range of 50–70 million tonnes CO2 year−1 could be generated. To realize this potential benefit of CCS, more and evenly located CO2 storage sites are needed in Europe, including a CO2 transportation network (Rosa et al., 2021). The CEWEP shows in a recent study how WtE, supported by EU policies could become a key process to reach the ambitious climate targets of the European Green Deal (Poretti and Engler, 2022).
Evolution and objectives of CE
Historically, recycling of wastes has been a common and widely applied practice, particularly in agriculture (manure, compost), primary industry (metals and metal ores from tailings), the construction sector (masonry and stones) and for consumer goods too. Especially during wartimes, periods of extreme poverty and scarcity of certain raw materials, it became a necessity to recover materials instead of discarding them in dumps (Klinglmair and Fellner, 2010). Environmental considerations for reuse and recycling are comparatively new and became relevant only in the aftermath of the study by the Club of Rome (Meadows et al., 1972): The report ‘Limits to growth’ presented models for future scenarios of resource consumption that – according to the authors – are required to prevent a ‘sudden and uncontrollable decline in both population and industrial capacity’. The report was quite challenged after its publication (Bardi, 2011), particularly from economists such as Julian Simon (Aligica, 2009; Simon, 1996). Nevertheless, recycling became a key issue on the agenda of modern waste management, with paper, biomass, metal, glass, plastic and other constituents of MSW as separately collected items. And the fast development of powerful economies such as the BRICS states (Brazil, Russia, India, China, South Africa) created a large demand for resources for both, infrastructure development and consumer goods. At the same time, new materials such as lithium and rare elements (e.g. neodymium) entered the market, opening the race for resource demand once again. The results are rising prices and partial shortage of certain materials, which are seen as a threat to future development. The stage is set to tackle this situation by the ‘new’ strategy of CE for both environmental and resource reasons.
Today, CE is a popular object of great topicality for society, public policy, for economic and resource research and for implementation in many economic sectors. Literature searches of the term ‘circular economy’ in Google Scholar yield close to 0.4 million articles. The diversity of the origin and content of these articles is large. There is no uniform and generally accepted answer yet to the second research question stated above ‘What are the main goals of CE, particularly with respect to materials that cannot be recycled?’. Kirchherr et al. (2017) concluded in their systematic analysis of 114 definitions of CE that CE ‘means many different things to different people’ and provided evidence for their statement. According to them, CE is most often depicted as a combination of reduce, reuse and recycle activities. A systemic shift towards CE is sometimes postulated as necessary. In addition, there are not many links found between CE concepts and sustainable development including future generations and social equity.
Based on relevant literature, the following two approaches to CE can be discerned:
Objectives of CE in a narrower sense: – Conservation of primary resources by multiple cycling of materials: Reuse and recycling of materials, thus reducing the need for primary materials, of energy and land by replacing mining and primary ore processing by secondary mining (e.g. ‘urban mining’) – Reduction of emissions by replacing mining and primary ore processing by less polluting secondary material cycling – Increase of resource autonomy and decrease of dependency on foreign resources
An example of such an approach to CE by a recycling strategy is given in US EPA (2021).
2. Objectives of CE in a wider sense:
In addition to the above waste-oriented CE, the aims are expanded and include a systemic view, with social issues, too. Often, three key elements are in focus (Webster, 2021): (1) design for circularity, (2) close links between material cycles and finance systems and (3) a holistic interpretation of the CE concept, replacing the ‘machine economy’ by a ‘nested living system’.
De facto, a mixture of objectives 1 and 2 is most often stated in national and regional policies and legislation. The EU understands CE as a model of production and consumption, with maximum extension of the life cycle of products. It involves sharing, leasing, reusing, repairing, refurbishing and recycling existing materials and products as long as possible (European Parliament, 2023). Based on the United Nations Sustainable Development Goals and the Paris Agreement, the Japanese government refers to the ‘circular and ecological economy’ CE as a closed circuit of consumption without the production of wastes, and thus no need for more resources (Japan Ministry of the Environment, 2019). Public and private sectors are committed to transform into a CE by 2050.
In China, the government has included CE in their 14th five-year plan 2021–2025. The goals are to increase resource efficiency by resource conservation and recycling, boost high-quality development and foster national resource security. The plan includes green design and production and makes CE a national priority. China aims at setting up recycling industries and systems covering the entire society until 2025. Recycling targets are set for key materials such as scrap steel, non-ferrous metals, paper, agricultural wastes and more.
Limits to CE
While the first half of the second research question ‘What are the main goals of CE . . .’, has been answered above, the second part ‘. . . particularly with respect to materials that cannot be recycled?’ is still open: Most actors agree that CE must be amended by mechanisms to safely destroy, transform or dispose of wastes and materials that contain hazardous substances. It is important to acknowledge that besides practical considerations, there are also theoretical boundaries that prevent total recycling of materials. According to Stumm and Davis (1991), cycling of resources is urgently needed, but the law of entropy limits the extent of recycling. Complete cycles are not possible. With their statement, they follow Georgescu-Roegen and his ideas presented in the book ‘The Entropy Law and the Economic Process’ (Georgescu-Roegen, 1971), arguing that natural resources are irreversibly degraded when used in economic processes. Even if this concept has been criticized by economists, it cannot be denied that the entropy law (the second law of thermodynamics) is a fundamental principle of physics. Dissipation of matter increases entropy, or the other way round, to concentrate matter requires energy (Rechberger and Brunner, 2002). The larger the dissipation the more energy is needed to collect and concentrate a given amount of matter. A cycling strategy aiming at complete cycles would require an infinite amount of energy and economic resources. While almost complete cycles may be attempted for rare and expensive materials with specific applications, it is not possible to apply this to the complete set of materials used by society: Much of the gold exploited from the Earth’s crust can be recovered with a high recycling rate, but most of the zinc used is dissipated and lost. The reason are differences in application, chemical speciation and biogeochemical behaviour of the tens of thousands of substances that are in use today (Brunner, 2013).
In addition to (1) the entropic and economic limits to the CE, further barriers exist. (2) The largest mass of materials is embedded in the long living anthropogenic stock and is only available for reuse after long time periods (Baccini and Brunner, 2012). (3) Hazardous chemicals are important and ubiquitously present in today’s world. They serve valuable purposes to increase life span of infrastructure, for transportation, to ensure human health, for stabilizing plastic materials in long living goods, for space and military purposes, for agricultural practices and more. Many of the highly dangerous substances have been banned but still exist as legacy chemicals that cannot be recycled but must be extracted from cycles. (4) In addition, some substances endanger technical recycling processes and must be kept away from cycles for technology and material quality reasons. (5) In accidents, hazardous chemicals and isotopes can be formed or released contaminating large amounts of materials that cannot be recycled. Examples comprise the European wide contamination of sewage sludge by radionuclides of 131I, 137Cs and 90Sr after the Chernobyl nuclear disaster (IAEA, 2006). (6) And finally, certain goods contaminated by, for example, pandemic microorganisms cannot be recycled either.
It is interesting to note that in most strategy articles about CE, these limits are rarely acknowledged, and thus there are only few recipes offered how to deal with such unrecyclable materials (Bodar et al., 2018). The study on copper-recycling during war periods by Klinglmair and Fellner (2010) shows that, depending on the material, the fraction available for recycling can be rather small (10%). Studies on flame retardants show high concentrations of chlorinated organophosphate esters and polybrominated diphenyl ethers in waste childcare articles made from recyclates (Harrad et al., 2022). Thus, it is a relevant question what to do with the non-recyclables (e.g. copper dissipated as copper sulphate, metallic copper from brake linings or catenary wires, flame retardants from consumer products, etc.). The EU (European Parliament, 2022) stated rightly about legacy substances (‘forever chemicals’): There are risks that legacy substances re-enter the economy after their disposal. These risks must be considered in the EU CE plan and in waste management regulation. New rules are introduced for several persistent organic pollutants, with lower permitted levels in products. Waste materials holding levels of pollutants that are too high cannot be recycled and must be destroyed or incinerated. ‘Products containing hazardous chemicals need to be treated in such a way that the pollutants are: (1) destroyed (for example, through incineration), (2) irreversibly transformed or (3) permanently stored (for example, in deep, underground, hard rock formations, salt mines, or a landfill site for hazardous waste)’. For harmful chemicals below a certain threshold concentration, recycling could be allowed. How to find the acceptable amount of materials lost ‘forever’ due to dissipation is not addressed yet.
According to Zeng et al. (2022), circular flows of hazardous substances may impose risks for public health and environment. Therefore, CE must be organized with caution, there is no benefit for an overemphasis of anthropogenic circularity. In addition, the management of waste flows in a CE might result in an uneven distribution of risks and costs among the global economy. Thus, for an effective CE, Zeng et al. (2022) asked for global restrictions on recycling of toxic materials and suggest appropriate regulations, for example, in the United Nations Basel Convention.
One of the crucial resources is phosphorus (P). Thus, P recovery from sewage sludge is important. Due to contaminants in sludge such as heavy metals and persistent organic pollutants, recycling of sludge in agriculture is prohibited in many countries. Sewage sludge incineration combined with P recovery from ashes acts as a cleaning process for P (Morf et al., 2019). Such an ‘urban phosphorus-mining’ strategy is proposed in Switzerland (AWEL, 2012; ZAR, 2023b) and discussed in other countries.
The need for sinks is defined in Brunner and Kral (2010), namely processes (transformations) or places for final disposal of hazardous materials. In his historical perspective, Tarr (1996) deplored that pollution control often moves waste from one medium to another. He asked to address the issue of the ‘ultimate sinks’ for urban wastewater, off gas and wastes in a holistic way. Other authors pointed out that sources and sinks are necessary elements of natural as well as anthropogenic systems, and that a CE cannot solve the waste problem without providing sinks for non-recyclables (Brunner, 2010; Stanisavljevic and Brunner, 2021). Kral et al. (2019) used case studies to show the link between quantity and quality of recycled material, and the need for final sinks. They discussed the optimization challenge of keeping cycles clean by costly measures for safe disposal of hazardous materials. They concluded that sinks such as WtE plants and landfills are key for designing a CE compatible with environmental and resource-oriented goals.
In summary, it can be said that CE requires sinks. CE cannot be successful without a concept for the disposal of non-recyclables. Natural sinks such as the atmosphere, hydrosphere and lithosphere with biogeochemical processes and anthropogenic sinks with corresponding transformation processes (mineralization by WtE, physical-chemical processes, microbial processes) must be provided by waste management to host, dilute and/or destroy hazardous materials. Some natural sinks are non-renewable resources and must be used with caution. If overloaded, their carrying capacity is decreasing, and ecosystems may deteriorate. Material flows into sinks must either be lower than metabolic capacity of the sink or must be so low that they do not significantly change sink concentrations even for long time periods (Baccini, 1989). Appropriate sinks for non-recyclable organic materials are WtE plants (Brunner, 2013). The conditions in a modern incinerator ensure that most any type of organic substance is mineralized and transformed into products such as CO2, water and inorganic salts. As shown before, advanced APC technologies eliminate the small fraction remaining in the off gas to insignificant traces. For inorganic residues that are highly immobilized, sites that exclude materials from the hydrological and atmospheric systems represent suitable final sinks.
Conclusions
The benefit of WtE for CE becomes evident when comparing the answers to the research questions about goals and means of WtE and goals of CE for materials that cannot be directly recycled: For theoretical (entropy) as well as practical (collection efficiency) and economic (costs of high recycling rates) reasons, full material cycles are not feasible. CE depends upon the controlled discharge of non-recyclables form cycles. They must either be destroyed or disposed of in safe long-term sinks. Over the last 100 years, WtE has been developed into a highly advanced and environmentally sound technology. Today, WtE serves as a final sink for non-recyclables such as hazardous organic substances and pathogenic micro-organisms. For successful implementation of CE, such sinks are imperative. WtE supports the main purpose of CE, namely concentrating and recycling of secondary resources that direct recycling systems are not able to recover, such as metals from MSW (Fe, Al, Cu, Zn, Au, Ag) and non-metals from sewage sludge (P). The highest overall recovery rates are achieved by a combination of recycling separately collected scrap metals and recovery of metals from WtE residues. In addition, WtE transforms wastes of high volume and solubility into residues of smaller volumes and solubility, hence decreasing the need for landfills. In addition, WtE supplies power, heat and even cooling to the CE. Combined with CCS technology this reduces greenhouse gas emissions and can generate negative CO2 emissions due to capturing of biogenic C.
The combined benefits summarized here cannot be provided to CE by any other single technology. Thus, for best performance, CE and WtE with state-of-the-art emission control, resource recovery and CCS belong together, resulting in more recycling, less loadings of the environment and less future risks. The relevance of this statement for resource and environmental policy is that modern WtE is a mandatory means to complement any CE policy. Policies about CE must include a strategy what to do with non-recyclables. This requires knowledge about total material flows and stocks. Theoretical and practical limits of a CE must be explored, and kind and amount of sinks required to accommodate non-recyclable materials, wastes and losses must be identified and quantified. Quantification of losses from cycles is paramount and determines prospective challenges regarding sink capacities that must be provided in the future. Hence, WtE will not only become more important as a source of secondary resources. Upgraded with CCS, it will be instrumental to reach the goals of a CE, namely recycling of energy and materials, capturing carbon to prevent climate change and minimizing the need for sinks.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
