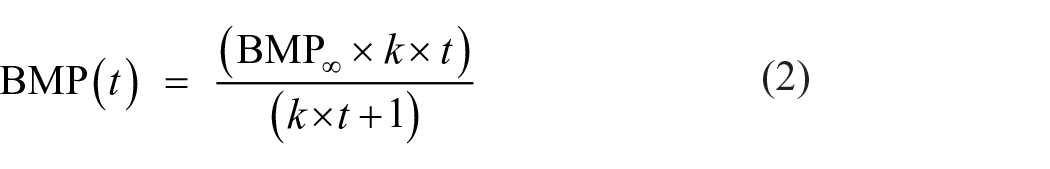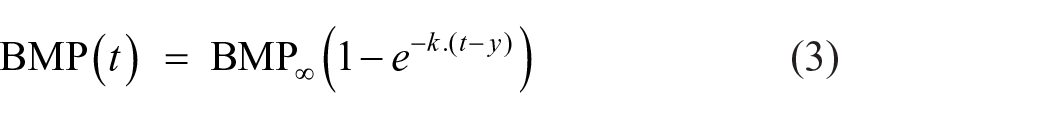Abstract
Anaerobic digestion (AD) is a prominent treatment method for the sludge produced from sewage treatment plants. Poor solid reduction and longer retention time are the main drawbacks of AD. Thermal hydrolysis (TH) is a potential pretreatment method for solubilization of sewage sludge (SS) solids thereby improving biogas production during AD post-treatment. In this study, the SS sample (total solids = 1.75 wt% and total chemical oxygen demand (COD) = 15,450 mg L−1) was subjected to TH pretreatment (temperature = 140–180°C and reaction time = 60 minutes) in a 0.7-L capacity stainless-steel high-pressure reactor. At a reaction temperature of 180°C, the maximum solid solubilization (total dissolved solids = 4652 mg L−1) and improved dewaterability (time to filter = 4.7 s.L g−1) were observed. The biochemical methane potential test results showed almost doubling of methane generation from 145 to 284 mL gCOD−1 after TH pretreatment at 180°C. The life cycle assessment approach was used to compare various SS treatment and disposal scenarios, two of which included hydrothermal pretreatment. The scenarios involving hydrothermal pretreatments showed the least global warming potential.
Keywords
Introduction
Sewage sludge (SS) is a semi-solid waste by-product which is generated from a sewage treatment plant (STP). In view of the rapid growth in sewage treatment capacity in developing nations like India, the sludge production is also expected to increase tremendously. The total solid (TS) content in the sludge from the primary clarifier and secondary clarifier (i.e. after biological treatment) can vary from 2 to 7% and 0.4 to 1.5%, respectively (Metcalf and Eddy, 2003). Since the sludge consists of active biomass, it deems unfit for direct land application (Hii et al., 2014), and the sludge disposal by means of land application is not sustainable. However, SS processing may cost up to approximately 50% of the total operational expenses in an STP (Suárez-Iglesias et al., 2017).
From the literature, it was found that anaerobic digestion (AD) is one of the efficient sludge processing technology (Pilli et al., 2015). Several benefits associated with AD technology include volume reduction, odour elimination, pathogen reduction, low energy requirement and energy recovery in the form of biogas (Metcalf and Eddy, 2003). However, the conventional AD is a slow process for SS due to the presence of high concentration of suspended solids, and the biogas yield is often lower than the theoretical estimation. Therefore, there is a need to develop suitable SS pretreatment method, which not only protect the surrounding environmental but also enable recovery of value-added products.
Hydrothermal pretreatment is considered an attractive alternative for wet biomass (e.g. SS) due to its capability of improving the sludge digestion rates due to solubilization of suspended solids, which lowers the retention time with improved biogas production (Bougrier et al., 2006; Jeong et al., 2019; Khanal et al., 2007; Li and Noike, 1992; Pilli et al., 2015; Pinnekamp, 1998; Tanaka et al., 1997; Valo et al., 2004; Zhang et al. 2014). Moreover, the SS pretreatment by the two hydrothermal pretreatment methods, that is, thermal hydrolysis (TH) and hydrothermal carbonization (HTC), is likely to offer several environmental benefits such as reduction in greenhouse gas (GHG) emissions, recovery of value-added chemicals (such as humic substances, phosphorous, etc.), improved biogas production and less or no sludge for disposal (Gupta et al., 2020; Malhotra and Garg, 2020, 2021). The type and nature of products/by-products depends upon reaction conditions; thus, the products from both the processes generally vary widely. However, high energy requirement is considered major disadvantage of HT pretreatment. Out of the above hydrothermal pretreatments, the TH process typically works for SS under mild temperatures (140–180°C) in the absence of air (Suárez-Iglesias et al., 2017). The main purpose of the process is to improve biogas generation from the waste.
To make appropriate choice, the life cycle assessment (LCA) approach helps by evaluating advantages and disadvantages from various options. Many researchers have reported comparison among different SS treatment options using LCA primarily reporting GHG emission and energy needs (Hong et al., 2009; Hospido et al., 2005; Liu et al., 2013; Lombardi et al., 2017; Mills et al., 2014; Xu et al., 2014). The studies carried out by Mills et al. (2014) and Lombardi et al. (2017) showed that the incorporation of hydrothermal pretreatment reduced environmental impacts significantly.
Up to the best of our knowledge, no study is available on SS pretreatment by HT in context of India, which reported change in biogas generation and GHG emissions after the pretreatment. Therefore, the experiments were planned to investigate the efficacy of TH process (performed at 140–180°C temperature) as pretreatment for determining the extent of sludge solubilization and improvement in the biogas yield during post-treatment. The detailed sludge characterization and biochemical methane potential (BMP) tests were performed to achieve the above objective. Subsequently, the SS disposal scenarios were formulated to compare their energy and environmental performance using the LCA approach. It is believed that the results from the experimental as well as LCA evaluation will be helpful in selecting the most sustainable option for sludge disposal.
Material and methods
Sources of SS and seed sludge
The SS sample was collected from an STP (capacity = 100 MLD) located in Nerul, Navi Mumbai, India, where sequencing batch reactor technology is used for the treatment of municipal wastewater. The sludge sample was stored at 4°C temperature to avoid any biological degradation.
The seed sludge sample was obtained from a biogas plant located at Indian Institute of Technology Bombay campus. This plant is used for anaerobic treatment of food waste. The seed sludge was incubated for 10 days at 37°C to decrease background biogas production and also to eliminate the residual substrate.
Procedure for TH
The thermal pretreatment of SS was performed in a high-pressure stainless-steel reactor (316) with a volume of 0.7 L in batch mode (Figure S1, Supplemental Information). Similar types of reactors have been used for the laboratory scale studies (Jeong et al., 2019; Li and Noike, 1992; Malhotra and Garg, 2020, 2021; Valo et al., 2004). The instrument is equipped with temperature and mixing controller using proportional integral device to maintain the temperature within ±1°C of the set value, mixing speed adjustment, liquid sampling ports and gas injection port. A digital display unit was also attached to record temperature, pressure and stirrer speed during the reaction. For a typical run, 0.5 L of SS sample was added into the reactor. After sealing, the reactor vessel was subjected to heating. Around 40 minutes were required to achieve the desired reaction temperature. During heating as well as reaction period, the SS was continuously mixed with a four-blade stirrer at a speed of 100 rpm. The TH runs were conducted at three temperatures, 140, 160 and 180°C for 60-minute duration. The corresponding vapour pressure in the reactor was recorded as 2.6, 5.1 and 8.7 kg cm−2, respectively, during the heating period. All the batch TH runs were conducted in duplicate, and no periodic sampling was performed. Furthermore, the analysis of each treated sample was performed in triplicates to determine variability in the results.
Analytical methods
The chemical oxygen demand (COD) of a sample was determined by standard closed reflux method as per the procedure described in American Public Health Association (APHA) handbook (APHA, 2012). For determining the soluble COD (sCOD), the samples filtered through 1.2-μm pore size filter were digested in a HACH COD digester 74 (DRB200, Hach company, Loveland, Colorado, USA). Total organic carbon (TOC) in the raw and treated SS samples was measured using TOC Analyzer (Shimadzu, TOC-VCSH, Kyoto, Japan). TS, total suspended solids (TSS) and total dissolved solids (TDS) in sludge samples were determined in accordance to the procedure specified in APHA (2012). For TSS calculation, 10 mL of raw and treated sludge samples were filtered through glass fibre filter (GF/B, 47 mm Whatman) in a vacuum filtration assembly. The ammonium nitrogen (NH4+–N) concentration was measured in the aqueous sludge samples using salicylate method (Willis et al., 1996).
The dewaterability properties of the raw and treated SS were measured using time to filter (TTF) apparatus. The parameter was measured in terms of the specific resistance. Buchner funnel with Whatman No. 1 filter paper (ID = 90 mm) was used for the test (APHA, 2012).
All the batch runs for TH were conducted in duplicates. Furthermore, the analysis of each sample was performed in triplicates, and the results are presented with standard error.
BMP test
The improvement in biogas production due to hydrothermal pretreatment was predicted by performing BMP test (Holliger et al., 2016; Jeong et al., 2019; Valo et al. 2004).
The BMP test was performed in 130-mL serum bottles with coiled butyl rubber stoppers. A substrate-to-inoculum ratio (SIR) of 0.5 (on volatile matter basis) was adopted (Filer et al., 2019). Based on the SIR value, the required amounts of inoculum and substrate were mixed in a glass bottle, and total volume was made up to 80 mL by adding growth medium. The composition of growth medium was adopted from Angelidaki et al. (2009). Subsequently, the bottle contents were flushed with pure N2 before tightly closing with a cap. Then, the bottles were kept in a water bath for incubation at 37°C for the entire test period. The mixing of bottle contents was performed manually once in a day. For biogas measurement, a gaseous sample was withdrawn using a glass syringe (volume = 20 mL), and the methane content was calculated in the sample using gas density method (Justesen et al., 2019). Moreover, flame test was carried out to confirm the presence of methane in biogas.
Kinetics of biogas production during BMP test
The biogas generation rate and extent of AD were determined based on the experimentally obtained methane production data during the batch BMP tests. Three different rate models were used to fit the BMP kinetic data (Filer et al., 2019; Strömberg et al., 2015; Zhang et al., 2021). The model equations are as follows:
(i) First-order rate model:
(ii) Monod type model:
(iii) Pseudo first-order model:
where BMP∞ = ultimate BMP (mL gCOD−1), BMP(t) = BMP value at time t (days), k = BMP kinetic rate constant (day−1), y = lag phase duration (days) and t = incubation time (days).
Comparison of SS management scenarios through LCA approach
Goal and scope definition
Objective
The goal of the study is to compare various sludge management options. For this purpose, the environmental impact was estimated in terms of global warming potential (GWP) and energy requirements for different sludge disposal scenarios.
Functional unit
The functional unit assumed for the study was 1 tonne of dry SS, and the life cycle inventory method was used to estimate the energy and GHG emissions.
System boundaries
The system boundary starts with production of SS in an STP. The system boundary of various scenarios were covered with centrifugation, AD, TH, HTC, transportation and final disposal of residues to landfill. Moreover, the fuel consumption in transportation, electricity used in centrifugation, heating requirements of AD, TH and HTC and losses are included. Direct emissions released within the operation phase of each unit process, such as landfilling, AD, transportation, etc., are also accounted.
The four scenarios identified for SS treatment and disposal were as follows:
Sludge centrifugation and landfilling with gas recovery
AD of SS followed by centrifugation and landfilling of digestate
SS pretreatment by TH followed by AD and landfilling of the solid fraction with gas recovery
Sludge dewatering followed by HTC and utilization of liquid fraction and solid hydrochar obtained after the pretreatment for resource recovery
Various SS treatment scenarios are illustrated in Figure 1, whereas the detailed description of each scenario along with the detailed calculation is provided in Section S2 (Supplemental Information). The GWP and energy input for each step of all above scenarios are presented in Table 1.
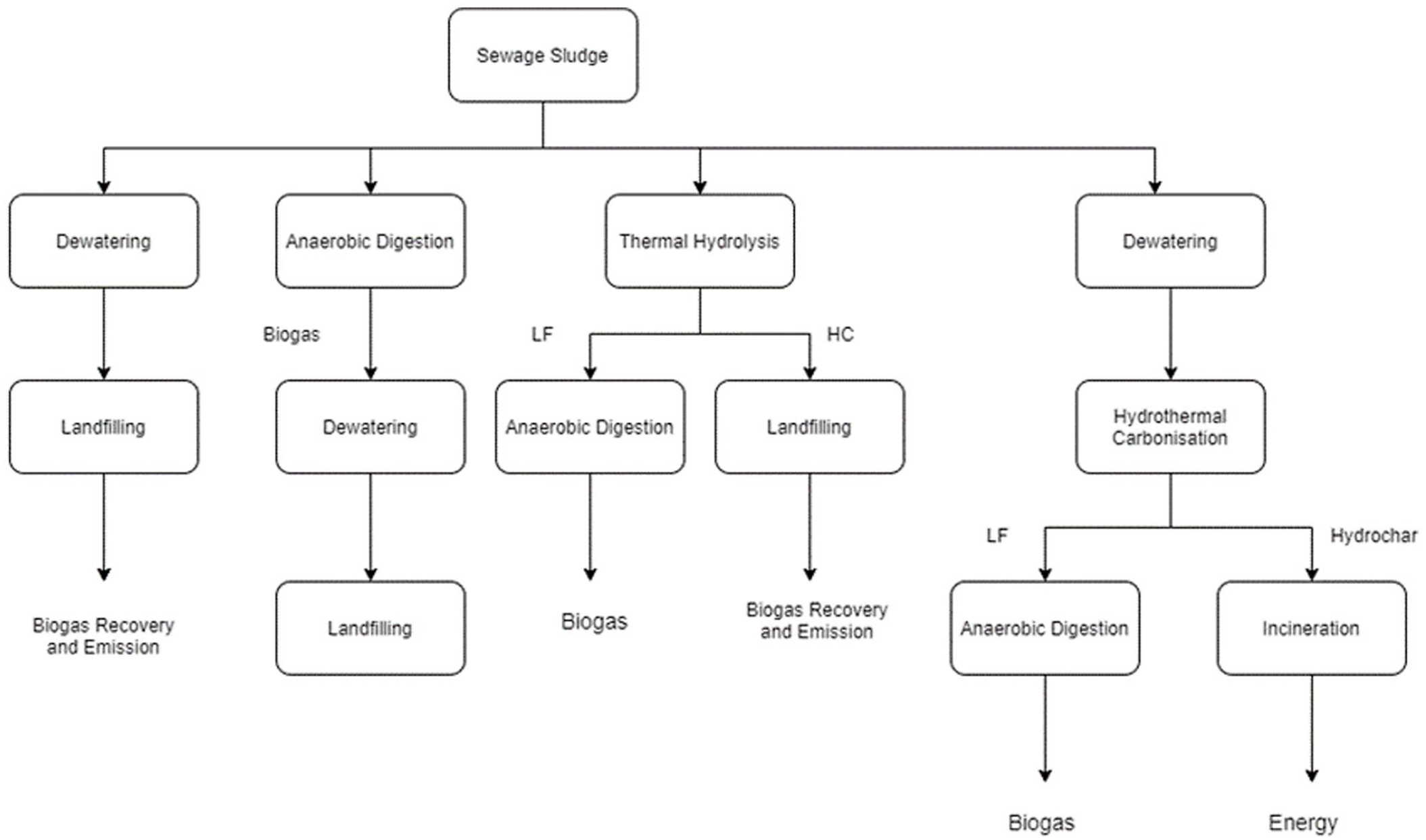
Various sewage sludge treatment scenarios and system boundaries.
Global warming potential and energy input of considered scenarios.
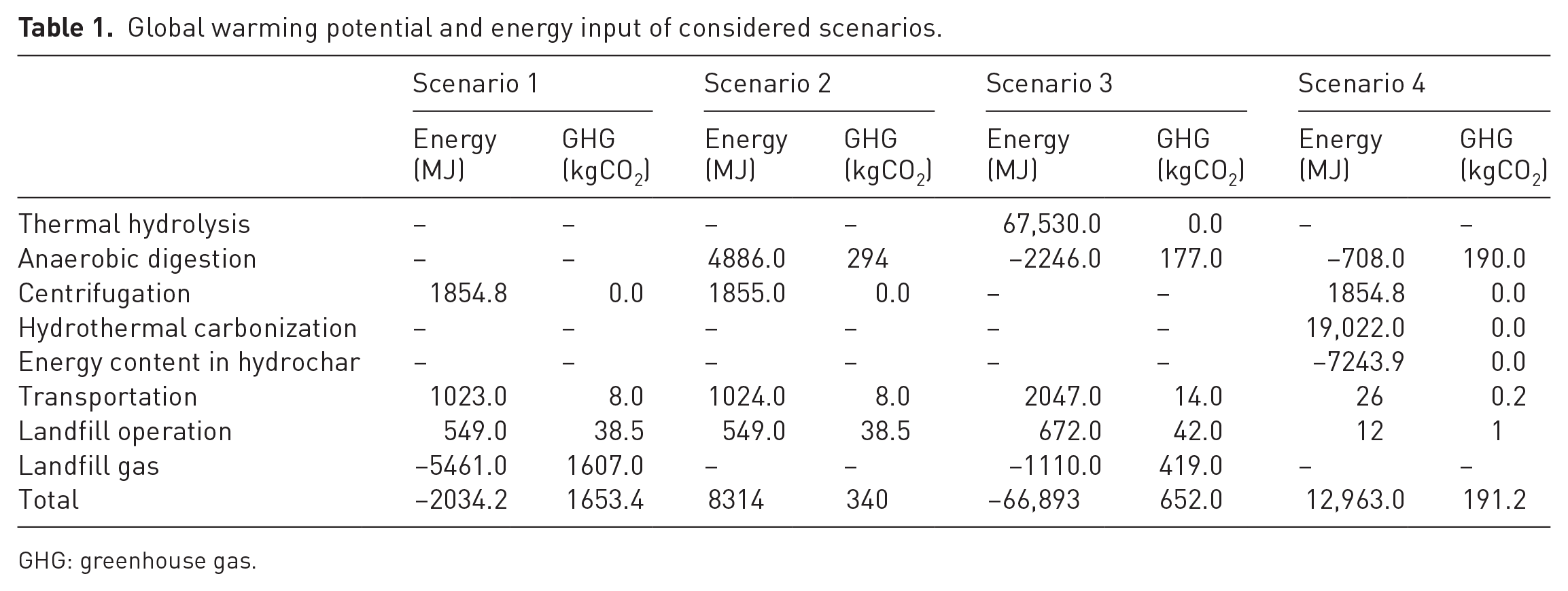
GHG: greenhouse gas.
Following assumptions were made during evaluation of various scenarios:
The impacts caused by infrastructure construction were not included in the analysis.
Indirect emissions were excluded from the calculation.
Direct CO2 emissions from biological degradation of solids and the combustion of biomass, which is defined as ‘carbon neutral’, were also excluded from the calculation of GWP.
The electrical efficiency was 33%, whereas heating efficiency was assumed to be 50%.
The time horizon chosen for the study is 100 years for both emission inventory and impact assessment.
A transportation distance of 10 km is taken from STP to landfill.
Results and discussion
Characteristics of SS and seed sludge
Before the commencement of any experimental run, the sludge was mixed vigorously to obtain a homogenous mixture, whereas the seed sludge was sieved through screens to remove large suspended particles and fibrous matter. The physicochemical properties of SS and seed sludge are shown in Table 2.
Various physicochemical parameters of raw sewage sludge and seed sludge.
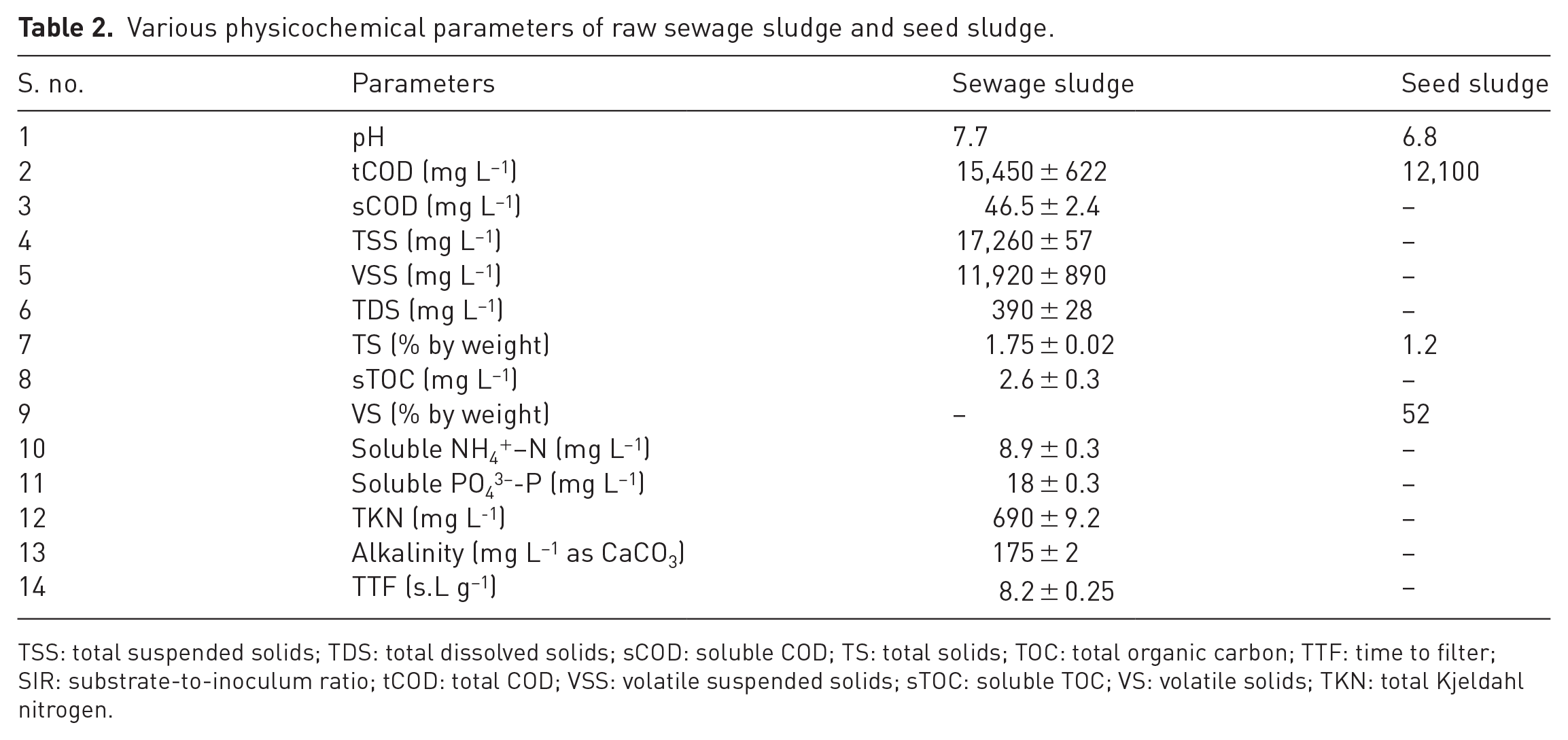
TSS: total suspended solids; TDS: total dissolved solids; sCOD: soluble COD; TS: total solids; TOC: total organic carbon; TTF: time to filter; SIR: substrate-to-inoculum ratio; tCOD: total COD; VSS: volatile suspended solids; sTOC: soluble TOC; VS: volatile solids; TKN: total Kjeldahl nitrogen.
SS contained very high concentration of suspended impurities in comparison to dissolved solids. Volatile suspended solids (VSS) to TSS ratio was approximately 0.7, and total Kjeldahl nitrogen (TKN) was 690 mg L−1. Among other parameters, PO43−-P and NH4+–N concentrations in the sludge sample were 18 and approximately 9 mg L−1, respectively.
The sieved seed residue had total COD (tCOD) (approximately 12,000 mg L−1), whereas TS concentration was 1.2%. Volatile solids (VS) concentration in the seed sludge was lower than that in SS.
Effect of hydrothermal pretreatment on SS properties
SS solubilization
In the study, the extent of SS solubilization after TH process was examined by measuring TSS, TDS, soluble TOC (sTOC) and sCOD (Figure 2(a)). With an increase in reaction temperature, the solubilization was also enhanced. The sCOD of the untreated and treated SS sample (at 180°C) were 46.5 and approximately 4000 mg L−1, respectively. The TDS and sTOC concentrations in the treated sludge were increased to approximately 2450–4650 and approximately 735–1700 mg L−1, respectively (initial TDS = approximately 390 mg L−1 and sTOC = 2.6 mg L−1). The TSS concentration was reduced with increase in temperature. Sludge solubilization at elevated temperatures has been reported in earlier studies (Jeong et al., 2019; Li and Noike, 1992; Valo et al., 2004).
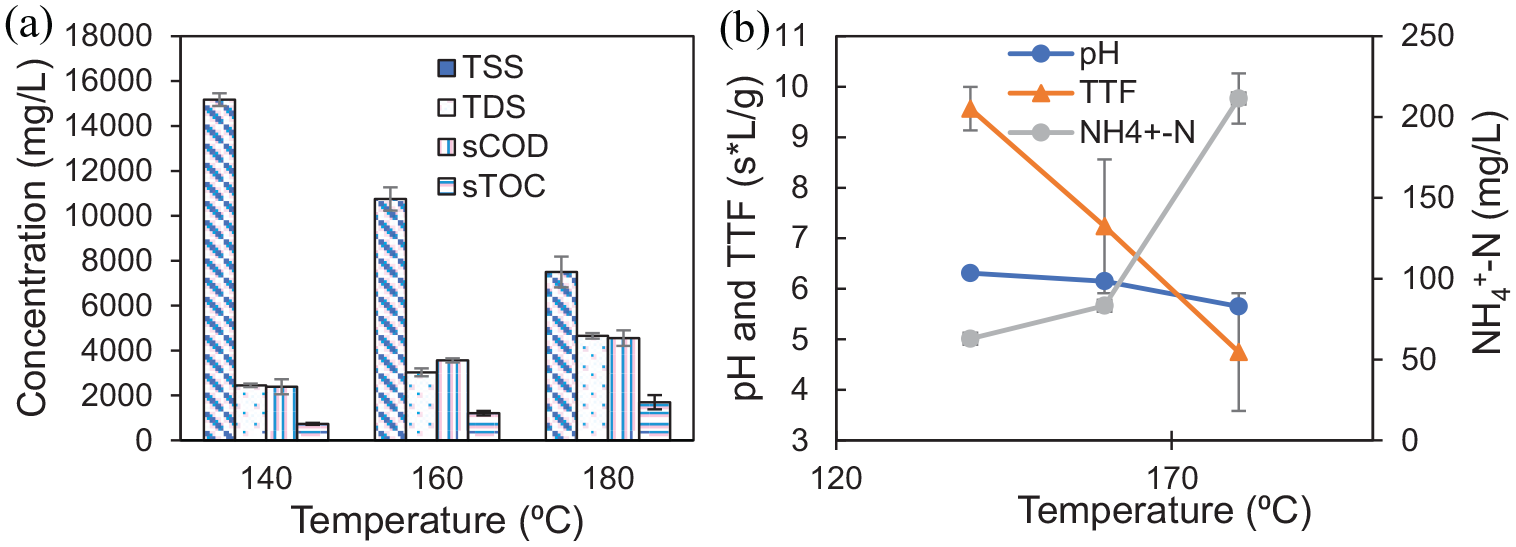
Effect of TH temperature: (a) TSS, TDS, sTOC and sCOD concentrations and (b) pH, TTF and NH4+–N of SS (reaction time = 60 minutes).
Change in pH, TTF and ammonium nitrogen concentration
It was observed that the pH of SS reduced to 6.3 from an initial value of 7.7 (raw sludge) at a reaction temperature of 140°C. Marginal reduction in the pH of treated sludge was observed after TH at higher temperatures (Figure 2(b)). The reduction in pH is an indication of the formation of solubilized organic species having acidic nature.
The dewaterability of untreated and treated SS was measured as TTF, which was normalized with TSS of the sample. It was observed that TTF of sludge was first increased to 9.5 s.L g−1 after TH at 140°C from an initial value for raw SS of 8.2 s.L g−1 possibly due to disintegration of larger solid particles into colloids, which inhibit the filtration process. However, the TTF was found to decrease significantly with further increase in reaction temperature (Figure 2(b)). For instance, TTF values were 7.2 and 4.7 s.L g−1 at temperatures 160 and 180°C, respectively. The significant improvement in dewaterability of the SS after hydrothermal pretreatment at higher temperatures was attributed to the release of bound and intracellular water present in the SS structure. The water is released due to several physical and chemical reactions that take place during TH (Pilli et al., 2015).
The NH4+–N concentration was also determined in the solubilized sludge after TH pretreatment. The production of NH4+–N is attributed to degradation of protein molecules present in SS. Its concentration should be known if the pretreated sludge is subjected to AD process since NH4+–N concentration greater than 1500 mg L−1 cause ammonia toxicity during the biological treatment (Ngo et al., 2021). In the present study, the NH4+–N concentration increased from approximately 9 mg L−1 (in untreated sludge) to a maximum of approximately 210 mg L−1 after 60 minutes hydrothermal treatment at 180°C temperature (Figure 2(b)).
Improvement in sludge settling properties
After TH pretreatment, most of the particulates present in SS sample were in solubilized form. The suspended solids remaining after the pretreatment were much lower as compared to those in the raw sludge. The settling properties of SS were remarkably improved after TH at 160 and 180°C, and the residual solids settled rapidly within 2–3 minutes after pouring the treated sludge in measuring cylinder (capacity = 1000 mL) (Figure S2; Supplemental Information). However, no improvement in sludge settling was observed after TH at 140°C.
Effect of hydrothermal pretreatment on BMP
Biogas and methane generation
After hydrothermal pretreatment, the BMP of SS was increased significantly due to cell lysis and breakdown of extracellular polymeric substances. The improvement in biogas generation was observed with increase in pretreatment temperature. The total biogas produced from the raw SS and treated sludge (at 140, 160 and 180°C) was 294, 424, 532 and 516 mL per g of COD added, respectively (Figure 3(a)), whereas the corresponding methane production was 145, 250, 286 and 284 mL gCOD−1 added (Figure 3(b)). However, no improvement in gas generation was observed when the sludge pretreatment was performed above 160°C despite higher solubilization. This result suggests that recalcitrant melanoidins may have formed due to reaction between proteins and carbohydrates at and above 180°C temperature (Zhang et al., 2021). Moreover, the nature of volatile fatty acids may affect the gas production. The lag phase of 2.4 days could be observed during anaerobic treatment of raw SS, whereas the hydrothermal pretreatment eliminated the lag phase during anaerobic treatment. The BMP of the pretreated SS at 140–180°C was 1.7–2 times as compared to the untreated SS. Similar results have been reported in earlier studies on TH of SS (Li and Noike, 1992; Tanaka et al., 1997).
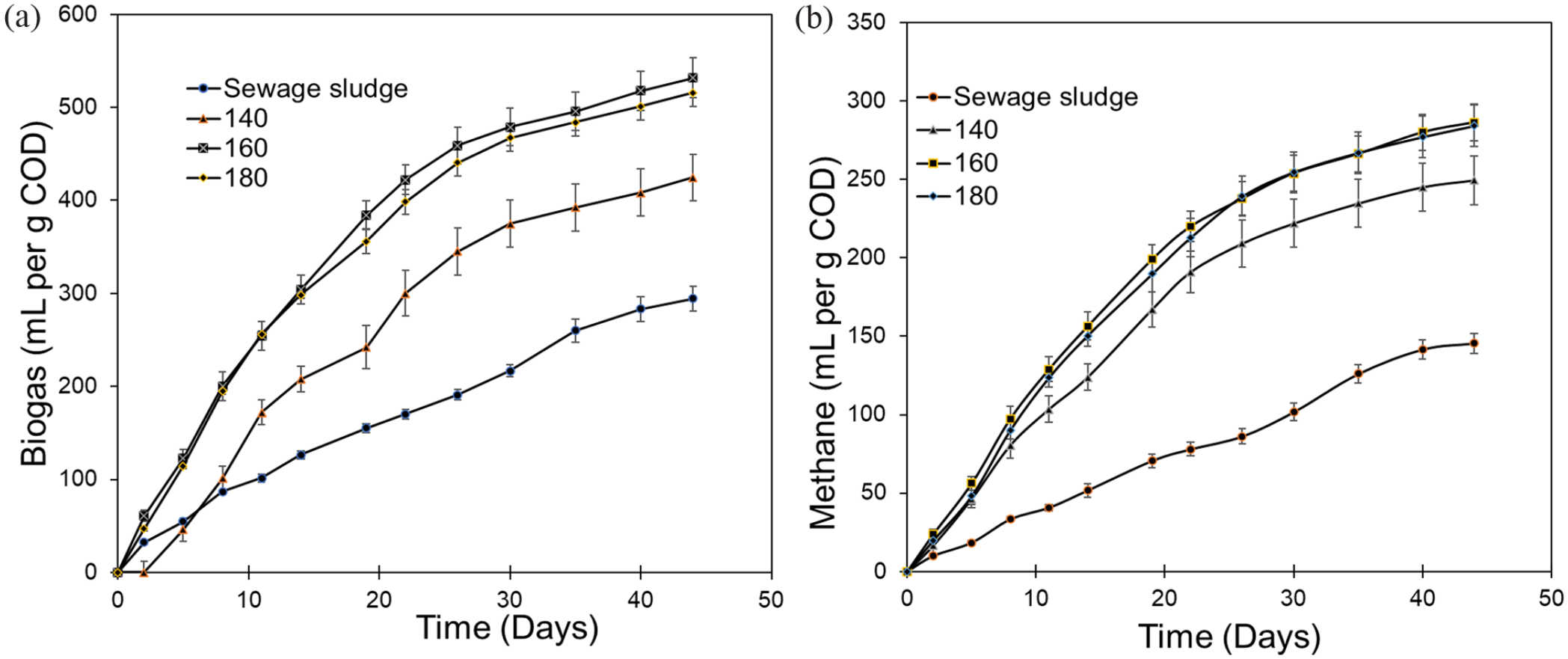
BMP test results: (a) biogas production and (b) methane generation with time from untreated and pretreated SS samples.
Determination of kinetic rate constant
Out of the three models (discussed in ‘Kinetics of biogas production during BMP test’ section), which were used to determine the kinetic rate constant, the first-order model showed the best fit to the BMP data (Table 3). With this model, the kinetic rate constant for raw SS was found to be 0.028 day−1. The kinetic rate constants for methane generation from the hydrothermally pretreated SS (at 140, 160 and 180°C temperatures) were obtained as 0.035, 0.036 and 0.035 day−1, respectively. The highest rate constant was observed with the SS treated at 160°C, which was approximately 1.3 times than that for the untreated sludge. The lower rate constant value at 180°C is possibly due to the formation of inhibitory and/or refractory organic substances such as melanoidins as discussed earlier. It is clear from the model results that TH temperature above 160°C eliminated lag phase of AD. Filer et al. (2019) have reported first-order kinetic rate constant in range of 0.151–15.2 day−1 for waste-activated SS. However, it is difficult to compare kinetic constant among various studies due to the variation in sludge characteristics, origin of inoculum and operating conditions.
Kinetic parameters estimated using four models.
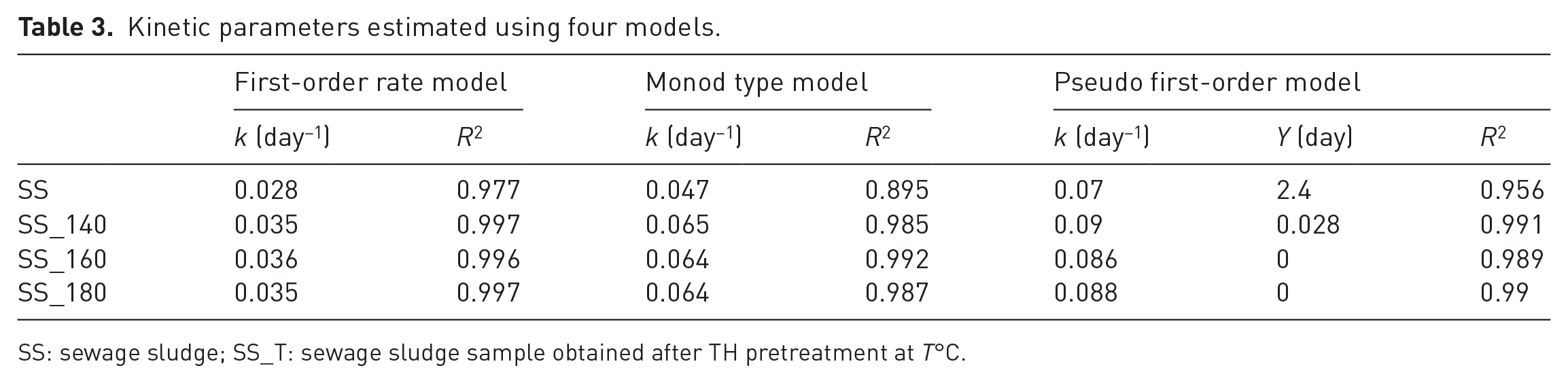
SS: sewage sludge; SS_T: sewage sludge sample obtained after TH pretreatment at T°C.
Comparison of various sludge treatment options based on LCA approach
Estimation of GWP
A comparison of GWP for the four scenarios is shown in Figure 4(a). The GWP for scenarios 1, 2, 3 and 4 were 1653, 340, 652 and 191 kgCO2 eq., respectively. Scenario 4 showed the least GWP (i.e. 191 kgCO2 eq.), whereas scenario 1 indicates the highest GWP with 1653 kgCO2 eq. The contribution of non-recovered landfill gas was highest to GWP for scenarios 1 and 3. Since it was assumed that approximately 10% of biogas is lost to environment during AD process, higher GWP was observed for scenario 2 though it was much less than scenarios 1 and 3. The GWP of other processes such as centrifugation, TH and HTC was not considered because these processes are not expected to result any direct emissions.
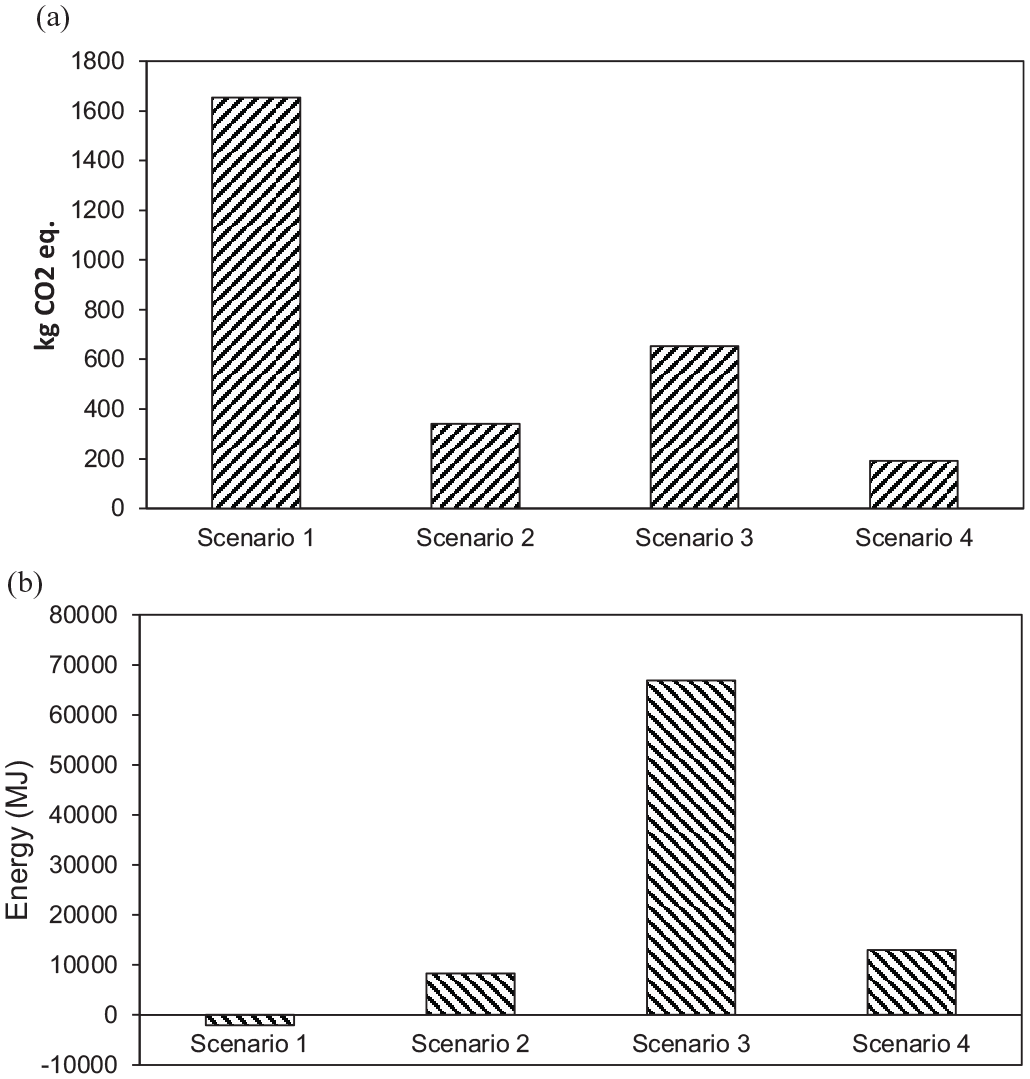
(a) Prediction of GWP and (b) estimation of net energy gain or loss from different SS treatment and disposal scenarios.
For the scenario mechanical dewatering and landfilling of SS with biogas recovery, the GWP was predicted as 1992 and 1180 kgCO2 eq., respectively (Liu et al., 2013; Lombardi et al., 2017). In another study, Hospido et al. (2005) reported GWP of 250 kgCO2 eq. for the scenario AD of SS followed by mechanical dewatering and land application. This value was comparatively lower as compared to that obtained in the present study since biogas loss was ignored during AD process in the previous study. Mills et al. (2014) considered TH of SS with AD and land recycling of the digestate and reported GWP of 600 kgCO2 eq., which is comparable with our study.
Estimation of energy requirements
A comparison of energy input requirements for the four scenarios is presented in Figure 4(b). The energy input for scenarios 1, 2, 3 and 4 was −2034, 8314, 66,893, and 12,963 MJ, respectively. Only scenario 1 is likely to produce energy, whereas scenario 3 involving TH was adjudged as the most energy-intensive. The scenario 3 based on AD process showed higher energy consumption compared to its production (as methane) because more energy was required for heating and operation. Scenario 4 involving HTC as pretreatment process showed more energy consumption; however, it was still lower than the scenario used TH as pretreatment (i.e. scenario 3).
Conclusions
TH is a potential pretreatment method for SS by which high degree of solubilization of the sludge solids can be achieved. The pretreatment improved significant biogas yield (almost twice than that observed during AD of untreated SS) with high methane content. Although the sludge solubilization was highest after TH treatment at 180°C, but no improvement in biogas yield is observed probably due to the formation of recalcitrant pollutants. Furthermore, dewaterability and settling properties of the treated SS samples at 160 and 180°C were substantially improved. At pretreatment temperature of 160°C, higher rate of anaerobic biodegradation was observed with kinetic rate constant of 0.036 day−1, which was 1.3 times the rate constant for raw SS.
The LCA approach was used to compare GWP and energy requirements of four sludge disposal scenarios. The results showed that landfilling of centrifuged sludge with gas recovery is an only energy producing treatment option, whereas the fourth scenario involving HTC pretreatment showed the least GWP. It is clear from the analysis that hydrothermal treatment may reduce GWP significantly, but energy requirements are found to be much higher in comparison to other treatments. Hence, there is a need to develop heat recycling and resource recovery options after hydrothermal treatment. Moreover, a detailed cost–benefit analysis should be performed. However, the choice of SS treatment and disposal technology will also depend upon the location and scale of operation which will also have great impact on the economics of process.
Supplemental Material
sj-docx-1-wmr-10.1177_0734242X231171044 – Supplemental material for Thermal hydrolysis of sewage sludge: Improvement in biogas generation and prediction of global warming potential
Supplemental material, sj-docx-1-wmr-10.1177_0734242X231171044 for Thermal hydrolysis of sewage sludge: Improvement in biogas generation and prediction of global warming potential by Diwakar Kumar Singh and Anurag Garg in Waste Management & Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The first author acknowledges the fellowship and other financial assistance by Ministry of Human Resource Development, New Delhi, India, under Prime Minister’s Research Fellow scheme. Furthermore, the authors also acknowledge the Department of Science and Technology, New Delhi, India, for financial support to carry out the research in this field (grant reference no.: DST/SSTP/2018/204).
Data availability statement
All data, models and code generated or used during the study appear in the published article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.