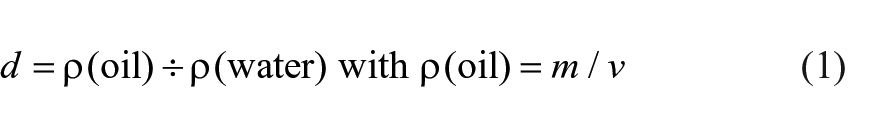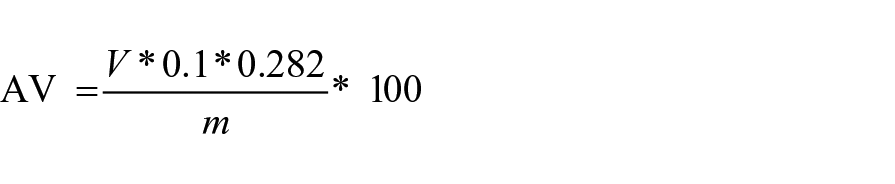Abstract
The treatment of used vegetable oil (UVO) with seven different adsorbents and through two different procedures (stirring and gravity filtration) was explored. Important differences in terms of density, turbidity, electrical resistance, free fatty acids (FFAs) content and relative fatty acid distribution were observed. Different outcomes were shown depending both on the adsorbent and on the procedure. Lower values of density and FFAs were registered for oils treated by gravity filtration with portland (respectively 0.6% and 0.81 g/ml) and celite (respectively 0.7% and 0.72 g/ml). Considering the undesired leaching from the powder to the oil, related to the turbidity, the celite resulted more suitable for the oil recycling (241 Nephelometric Turbidity Unit (NTU) for portland vs 184 NTU for celite). In addition, Fourier-transform infrared spectroscopy combined with multivariate analysis allowed to determine a chemical fingerprint relative characteristic of vegetable oils recycled by gravity or by adsorption by stirring.
Introduction
Since many years, vegetable oils have represented as a valuable source of chemicals for multiple uses, finding applications in various fields such as, for example, animal feeding (Magrinyà et al., 2012) fuel (Chrysikou et al., 2019; Hazrat et al., 2019; Ray and Prakash, 2019; Singh et al., 2022) and lubricant (Karmakar et al., 2017) production, as ingredients for cosmetics (Biermann et al., 2011) and in the construction (Asli et al., 2012) sector. The impact of this raw material on the specific market segments changes considerably depending on the geographical area. The balance between availability, technology readiness and local normative influences the choice of the raw material: as matter of example, soybean oil is mainly used in United States, Brazil and Argentina, palm and coconut oils are used in Malaysia (Ogunkunle and Ahmed, 2019). In European Union (EU), rapeseed oil is mostly used with the limitation of employing edible vegetable oils exclusively in the food chain. Also, in territories as EU, the use of used vegetable oils (UVOs) in non-food markets is widely endorsed and regulated (Ibanez et al., 2020).
UVOs are classified as wastes from the EU waste catalogue under the category edible oils and fats and thus, after collection, they can used as waste raw material of added value (European Commission, 2018). In this context, re-using of UVOs represents an easy route to cope with sustainable targets. In fact, waste cooking oils (WCOs), which constitute more than 90% of the above-mentioned UVOs family, are discarded before the formation of any unhealthy chemical. In the main decomposition process, the oxidation of C–C bonds and subsequent free radical formation must be carefully monitored. In fact, this process can end with acrolein production, and in combination with food leaching can also generate acrylamide (Fox and Stachowiak, 2007). Thus said, currently, the WCO goes out from the food chain with a low degree of degradation. In the end, it represents a valuable raw material easy to recycle and it is cheaper than the corresponding edible vegetable oil.
Within the current available technologies for UVO recycling, which are related to the specific application (Mannu et al., 2020), treatment with adsorbents or clays still represents a pivotal step. In fact, independent of the recycling process, filtration with bentonite, cellulose or other material is the best option to improve density, colour and thus viscosity of oils. Many bleaching procedures have been developed during the years (Hymore, 1996; Komadel, 2016) to remove greases, gums and other impurities from vegetable edible oil (Murray, 1991). Regarding their application to the recycling of UVOs, it is known that depending on the specific powder and on the specific activation procedure (Luna et al., 2018; Richardson, 1978), bentonites are able to retain different functional groups (Mannu et al., 2019; Serouri et al., 2021). Nevertheless, many protocols for the treatment of waste oils are referred to non-commercially available clays, and sometimes their corresponding activation procedure is not validated. Thus, from practical industrial application, it would be necessary to shed some light on commercially available and cheap raw materials which can be used for purifying UVOs. Such kind of screening represents the main target of this research.
Thus, in the present research work, WCOs arising from catering industries were subjected to treatment with different adsorbent materials. Commercial hydrophobic and hydrophilic silica-derived clays, celite, portland powder, diatomaceous earth and non-commercially but commonly available spent coffee and cork powders were used for screening purposes. For treated samples, some chemical and physical parameters were also assessed, in particular the free acidity, pH, density, turbidity and electrical resistance. Also, the fatty acid (FA) distribution was determined by 1H Nuclear Magnetic Resonance (NMR). Fourier-transform infrared (FT-IR) spectroscopy combined with multivariate analysis allowed to determine some relevant effects of specific materials.
Materials and methods
Materials
Silica HP60 and H18 were purchased by Wacker Chemie Italia S.r.l. (Italy), while celite and portland cement from Merch (Germany). Coffee powder was obtained by the dehydration of spent coffee powder originated from domestic houses, by heating in a laboratory oven at 50°C during 3 hours. Diatomaceous earths were purchased by Vozydeo–SAS (France). Cork powder was prepared by grating cork wine stoppers with a small size holes grater. UVO was collected from householders and local catering industries (Milan, Italy).
Physicochemical analyses
Conductivitymeter Inolab type was used for conductivity measurements and a digital pHmeter Inolab equipped with a combined glass electrode was employed to determine pH values (Xylem Analytics Germany Sales GmbH & Co. KG.). Turbidimeter HACH DR/800 Colorimeter brand was used for the turbidity measurements.
Density was determined by the classical volumetric method (equation (1)):
FT-IR spectrometer Perkin Elmer type (Model Frontier/with a spectral range of 4000–400 cm−1 and 1 cm−1 resolution) was employed for the characterization of oil samples spectroscopically.
Free acidity determination
The free acidity was determined by the method reported by Garcia Martin and coworkers (García et al., 2019). In an Erlenmeyer flask, 20 g of UVO, 50 ml of a solution ethanol/ethyl ether (1/1) and few drops of phenolphthalein were mixed. The resulting solution was neutralized by adding dropwise a basic KOH 0.1 N solution. The following equation was used to determine the acid value (AV):
where V corresponds to the volume of KOH in mL used for neutralization, 0.1 is referred to the normality of KOH, 0.282 is the milli-equivalent of oleic acid and m is the mass of the oil subjected to analysis.
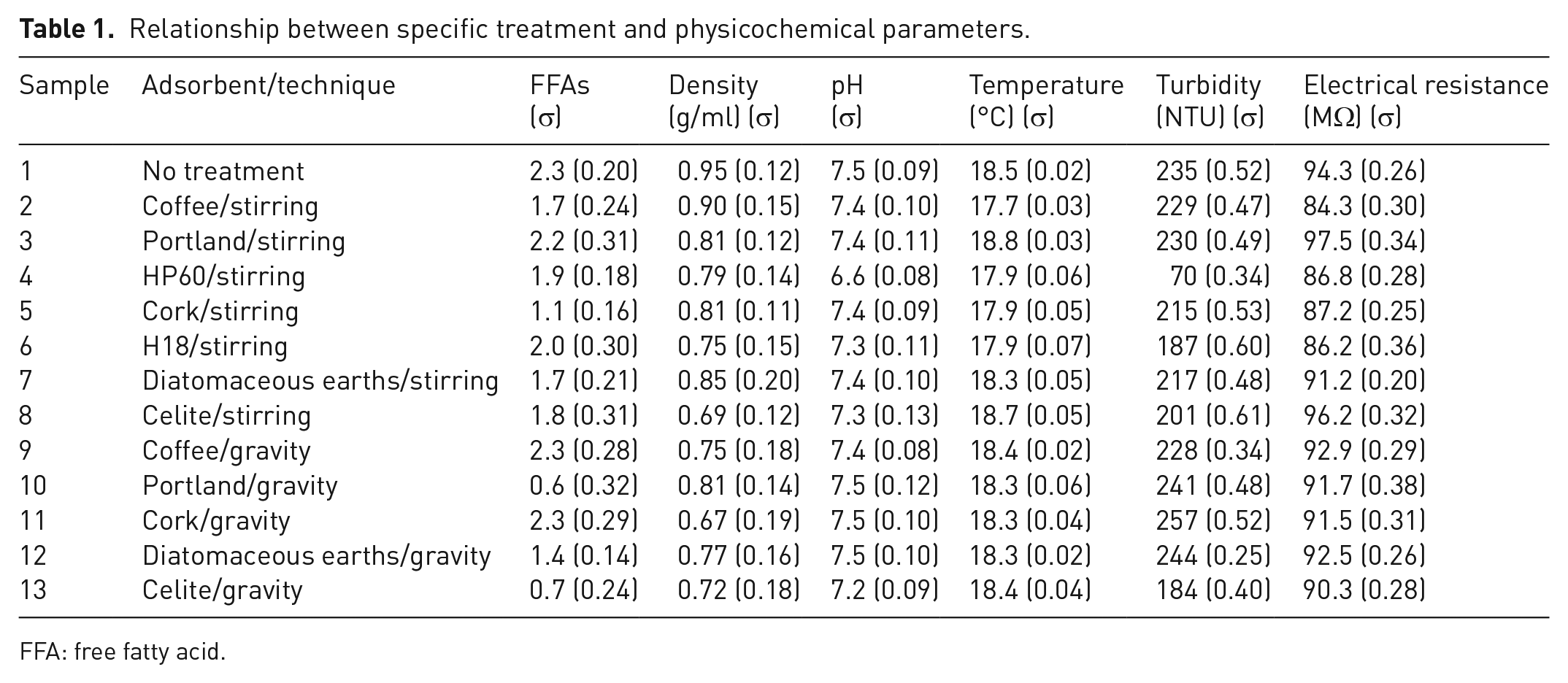
The value of each parameter was reported as the arithmetic media between three measurements. The corresponding standard deviation (σ) is then reported in Table 1.
Relationship between specific treatment and physicochemical parameters.
FFA: free fatty acid.
NMR measurements
Neat WCO samples were transferred to 5 mm NMR tubes, equipped with a coaxial tube containing deuterated dimethylsulfoxide (DMSO-d6) and tetramethylsilane as lock and chemical shift reference, respectively. NMR measurements were performed without sample spinning with a Bruker NEO 500 console (11.74 T) equipped with a direct observe BBFO (broadband including fluorine) iProbe and a variable temperature unit (1H resonance frequency of 500.13 MHz). 1D 1H spectra were recorded at 298 K with 64 scans using 65,536 points and a relaxation delay of 10 seconds, over a spectral width of 14 ppm. Data processing were performed in triplicates and standard deviations used as an estimate for the error.
Multivariate analysis
Multivariate analysis was conducted with the online tool metaboanalyst 4.0, while for the PLS-DA regression, the plsr function provided by R pls package was employed (Wehrens and Mevik, 2007).
Results and discussion
An exploratory screening was carried out on WCO samples treated by two different procedures. In the first one, the adsorbent was added to 100 g of WCO and the resulting mixture was stirred over 1 hour, before filtering on a paper. The second procedure was a classical gravity filtration: the waste oil was filtered over the adsorbent. The results relative to the acid value, pH, turbidity, electrical resistance and density are reported in Table 1.
Looking at the data reported in Table 1, it is possible to notice some important features. The starting waste oil shows a free fatty acid (FFA) content of 2.3%, which is in line with a WCO subjected to few deep-frying cycles (Azzena et al., 2020). The density value (0.95 g/ml) is slighter than the ones typical of edible vegetable oils (Noureddini et al., 1992), indicating a change in composition produced by frying, including the up taking of water from food and air humidity, showing, at the end, the same value reported for similar waste vegetable oils (Mannu et al., 2018). Turbidity and electrical resistance can vary consistently depending on the life cycle of the oil (Serouri et al., 2021) and here have been taken as a benchmark to analyse the effects of the different treatments on the original UVO.
Regarding the treated samples, the FFA content, which is strictly related to the pH, ranges from 0.7 (sample 13) to 2.3, typical of non-treated oil and samples 9 and 11. Focusing on FFA content, the best result was observed in experiments 10 (portland, gravity) and 13 (celite, gravity), for which the treatment is able to reduce the FFA content, respectively, of 75% (from 2.3 to 0.6) and 70% (from 2.3 to 0.7). It is interesting to notice consistent differences for the same process depending on the specific procedure employed. For all the tested adsorbents, a comparison between the two procedures shows that the gravity filtration resulted more effective than the suspended powder, with portland (sample 13) showing the bigger difference (from 2.2 for sample 3 to 0.6 for sample 10).
The pH values result in less variable, assessing at neutral values, with a very slight tendency to basicity. The only outliner is represented by sample 4 (treated with HP60), which showed a pH value of 6.6. Turbidity, which is related to partial solubility of the adsorbent into the oil, resulted in low for sample 4 (HP60), and, in minor extent, for samples 6 (H18, stirring) and 13 (celite, gravity). Density values range from 7.2 to 7.5 g/ml, indicating that it does not change considerably after recycling and it is not affected by the adsorbent. The only exception is represented by the sample treated by cork with the stirring process, which revealed a low density, around 6.6 g/ml. A relevant contribution in the slightly differences observed in density is provided by the presence of small amounts of water in the UVO. This is usually removed by treatment with powders while recycled cork does not show any water adsorption capacity. Generally, minor changes in the density of recycled WCOs were also observed after water washing at different pH, temperature, time and for different oil/water ratio (Vlahopoulou et al., 2018).
Finally, the electrical resistance was monitored. It is a parameter linked to the capacity of the considered matrix to allow the free movement of electrical charges and therefore to allow the passage of an electrical current. The electrical resistance of our samples ranges from 86.2 (H18, stirring) to 94.3 MΩ (untreated UVO) and, as the FFAs and pH, it confirms a different outcome in the oil composition which is related to the specific procedure (stirring or gravity). The differences observed for several parameters related to the procedure may be explained with the different contact time which, for some adsorbents, can lead to leaching phenomena. It is at the moment very difficult to correlate the composition of the adsorbent with the observed effects on the variation of the oil composition. In general, the two silica-based adsorbents HP60 and H18 differ in their interaction with water being, respectively, hydrophilic and hydrophobic. Unfortunately, being both the materials partially soluble in the vegetable oil, they can’t be used avoiding contamination. Nevertheless, as expected, hydrophobic H18 shows the higher solubility increasing the turbidity much more than HP60 (187 vs 70 NTU) (Wypych, 2016). Regarding the silica-alumina-based adsorbents (celite and diatomaceous earths), they operate in similar way and the only outlier is represented by the celite used under gravity filtration conditions, which probably interact better with specific chemical groups under long contact times which are typical of the gravity filtration. A specific physical characterization of the adsorbents would be necessary to clarify which minor components are responsible for the different behaviour. Regarding the portland cement, it has been reported as effective in removing metal traces from water (Hossain et al., 2020; Lim et al., 2019; Xu et al., 2018), but no indications have been reported for vegetable oils.
The variation in the physical parameters is a consequence of a different chemical composition upon the different treatments. Some preliminary information was extracted from the FFA profiles (Table 1), and other indications can be obtained by NMR analysis.
The FA profile of oils is the major parameter affecting their chemical and physical properties and hence their applications. It is related to the oxidative resistance and thus it determines the specific application when UVO is recycled (Borugadda and Goud, 2016; de Souza et al., 2020) 1H NMR can be fruitfully used to quantify the common unsaturated FAs (oleic, linoleic, linolenic) in a vegetable oil (Knothe and Kenar, 2004), with several advantages with respect to gas chromatography, which is usually the method of choice for determining the FA profile. Proton NMR needs indeed small sample volumes that are analysed in a fast and non-destructive way, giving the simultaneous identification of multiple components in a single experiment (Popescu et al., 2015). In addition, 1H NMR method can be even applied directly to neat oils, with no treatment of the sample. Figure 1 shows the spectra of the neat WCO samples investigated in this work, including the assignment of the characteristic peaks (García et al., 2019; Miyake et al., 1998).
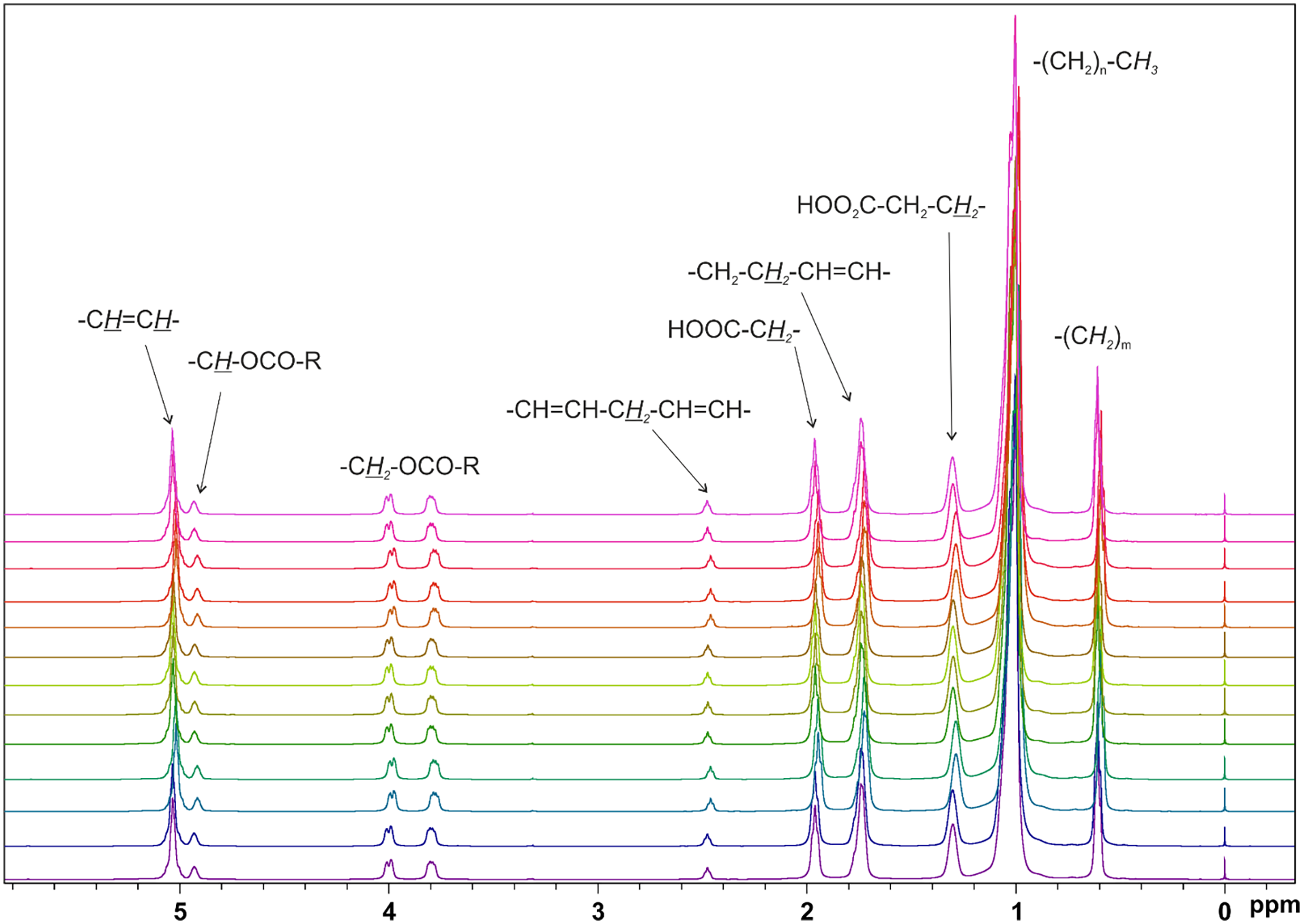
Overlap of 1H NMR spectra of WCO samples.
All WCO spectra show qualitatively the same pattern. Quantitative information, that is, the main FA concentrations, can be obtained by combining the various signal integrals, which are strictly proportional to the number of protons, using standard equations already reported (García et al., 2019). Table 2 lists the main FA profile of samples 1–13 calculated using 1H signals corresponding to the terminal methyl group at 0.6 ppm, the allyl methylene group at 1.7 ppm and the divinyl methylene group at 2.5 ppm. The iodine value (IV) can be also derived including the integral of the olefinic protons at 5.0 ppm. Despite not being a quantitative measure, IV is an empirical number useful to define the total amount of unsaturation, with 86 the reference value for oleic acid (García et al., 2019).
FA profile.
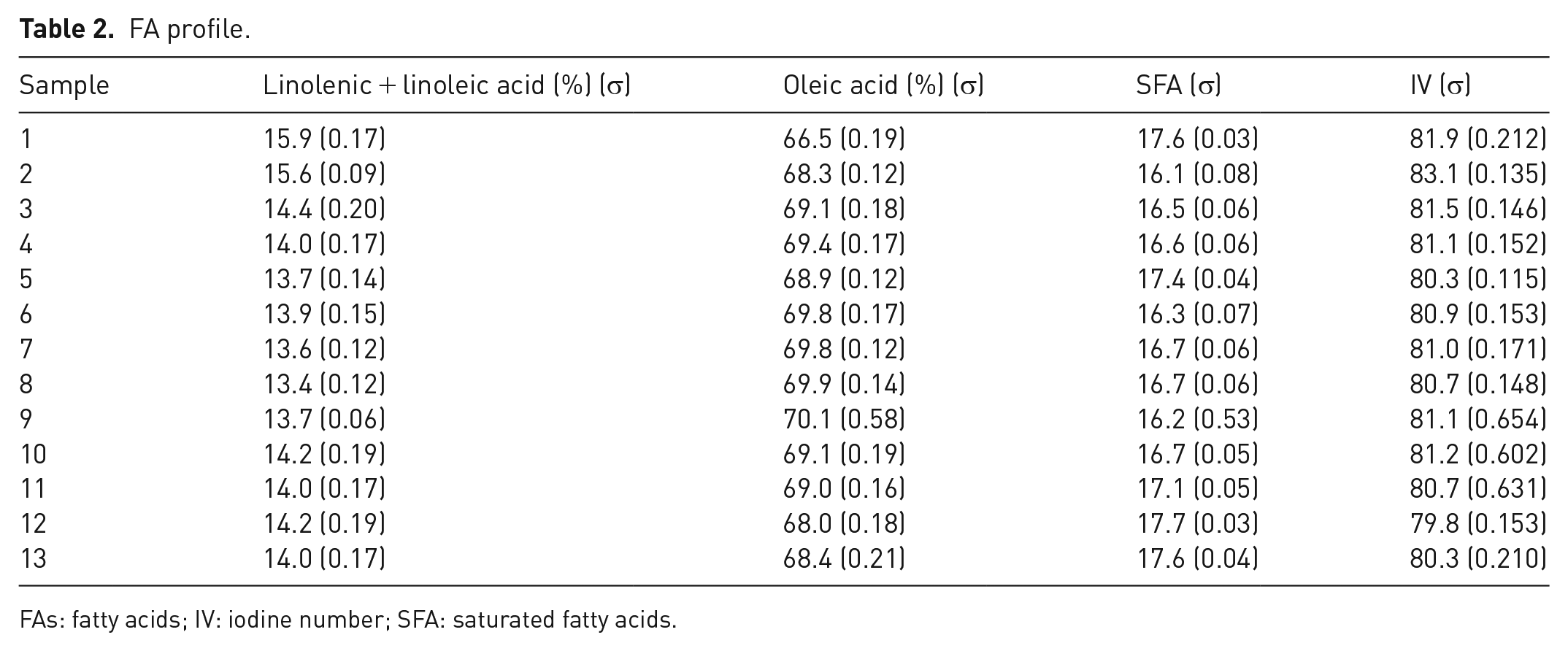
FAs: fatty acids; IV: iodine number; SFA: saturated fatty acids.
As expected, FA profile for samples 1–13 indicates low variability within different treatments. Considering the linoleic + linolenic as well as oleic percentages, it is possible to notice that from starting raw material (sample 1), the relative distribution of the two groups shows a variability below 2.4% and 3.5%, respectively. In particular, sample 8 (celite, stirring) and sample 9 (coffee, gravity) show the higher variability with respect to the starting material in terms of linoleic + linolenic component and oleic component, respectively. Overall, a decrease in the linoleic + linolenic component and an increase in the oleic portion is observed in all treatments. Comparing same adsorbents but different protocol, the FA profile confirms the different outcomes obtained by stirring or by gravity filtering. An increment in oleic acid is observed in the case of samples 2–9 (coffee, 68.3% vs 70.1%) while a decrement has been registered for samples 7–12 (diatomaceous earths, 69.8% vs 68.0%) and 8–13 (celite, 69.9% vs 68.4%). The reason behind the different FA profile observed when different procedures are used is related to the increased surface contact obtained by stirring the adsorbent with the oil. In this way, the adsorption ability of the material is enhanced and the capacity to retain specific molecules or chemical groups is highlighted. Regarding the oleic acid content, it is substantially unchanged in samples 3–10 (portland, 69.1% in both samples) and 5–11 (cork, 68.9% vs 69.0%). The level of saturated FAs seems not to be greatly affected by the nature of the adsorbent or the specific protocol and it ranges from 16.1% (sample 2) to 17.7% (sample 12). Finally, iodine number, which results basically unchanged upon the different treatments, shows lower values (around 80–83) than other WCOs (Mannu et al., 2019), indicating a relative low degradation of the specific UVO considered.
FT-IR analyses
The first qualitative assessment of the composition of samples 1–13 through FT-IR spectroscopy was conducted. As expected, no evident changes in the main chemical groups of untreated and treated samples were observed and the spectra obtained are superposable. In Figure 2, the spectrum corresponding to the starting material (sample 1) is compared with the samples treated with diatomaceous earths and celite (7 and 8 obtained by stirring and 12 and 13 by gravity filtration).
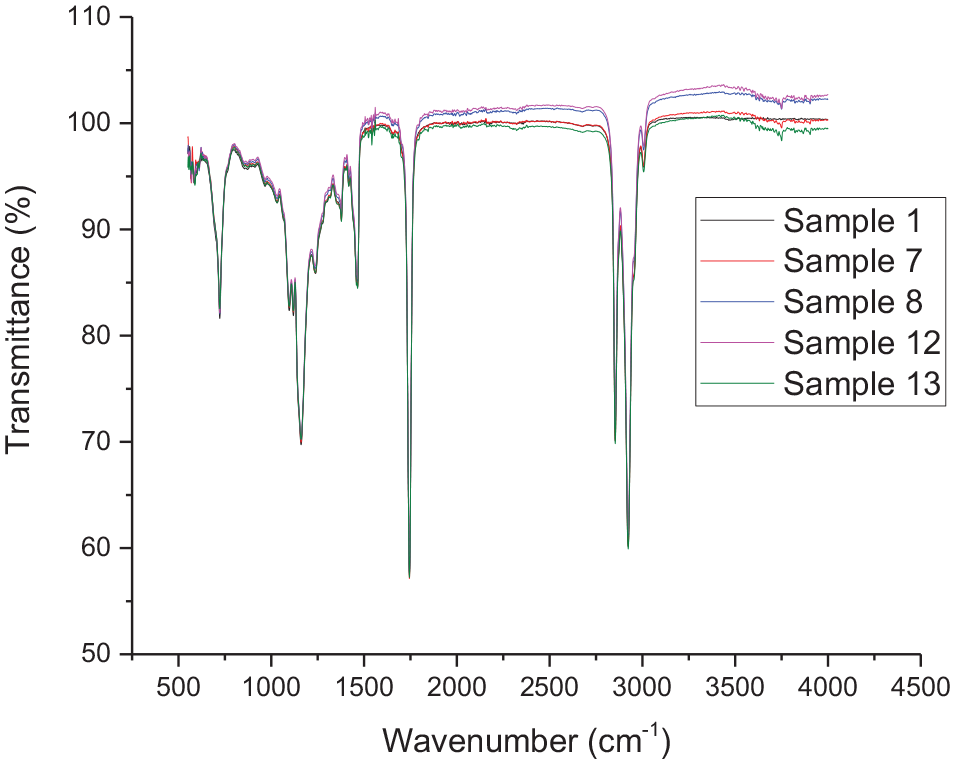
FT-IR spectra (transmittance mode) of samples 1, 7, 8, 12 and 13.
From a qualitative visual inspection of the spectra reported in Figure 2, it is not possible to ketch evident differences between the samples. Nevertheless, it was recently reported that through multivariate analysis it is possible to bring to light hidden information in raw FT-IR spectra of edible and waste vegetable oils (Mannu et al., 2022). Thus, with the aim to highlight the changes in composition produced by different adsorbents and by different procedures, the acquired data were subjected to multivariate analysis.
Multivariate analysis of FT-IR data
The raw relative integrals of all the signals detected by FT-IR spectroscopy and referred to samples of recycled UVO 2, 3, 5 and 7–13 were subjected to multivariate analysis. The spectra of oil samples treated with silica H18 and HP60 were discarded for the intensive leaching observed during the experimental procedure, which made these powders unsuitable for practical purposes. Also, for comparing reasons, sample 1 was not included in the statistical treatment. Thus, unsupervised principal component analysis was performed to check if the two different processes (stirring and gravity) were able to produce different clusters (Figure 3).
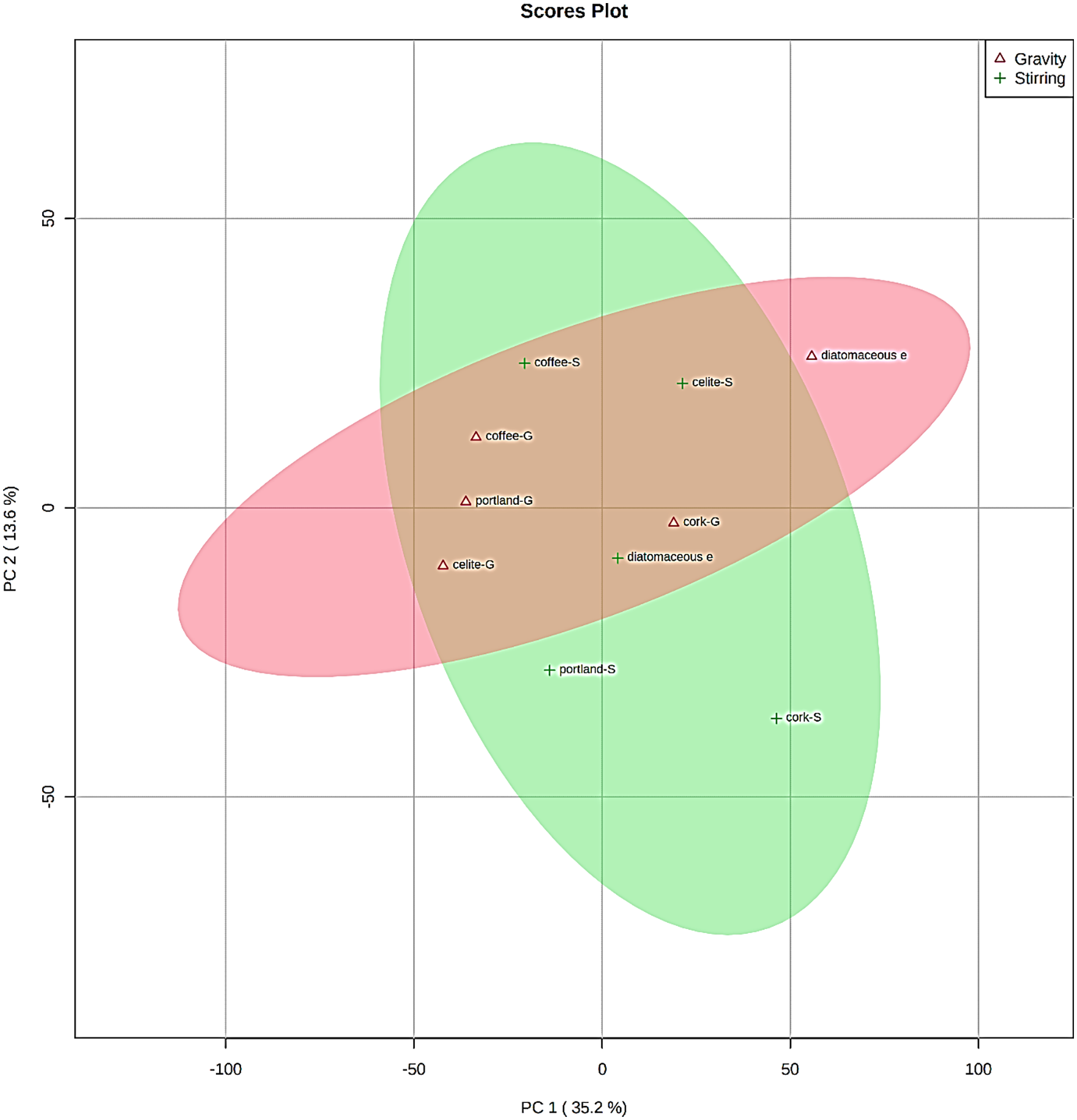
2D PCA scores plot for WCO samples treated with different procedures.
As expected, even if some differences between the two treatments were observed in terms of chemical–physical properties of the oil, a clear trend was not detected. In fact, the two clusters partially overlap evidencing differentiation just for portland and cork (stirring), and for diatomaceous earths (gravity).
Nevertheless, as demonstrated for similar studies on wine (Mannu et al., 2020) classification and for the distinction of edible and UVO (Mannu et al., 2018), it is possible to determine a chemical fingerprint characteristic of each process by partial least square discriminant analysis (PLS-DA). PLS regression can be useful for analysing systems characterized by a number of variables higher than the number of observations(Brereton and Lloyd, 2014; Ramadan et al., 2006; Westerhuis et al., 2008). In Figure 4, the 2D PLS-DA plots are reported.
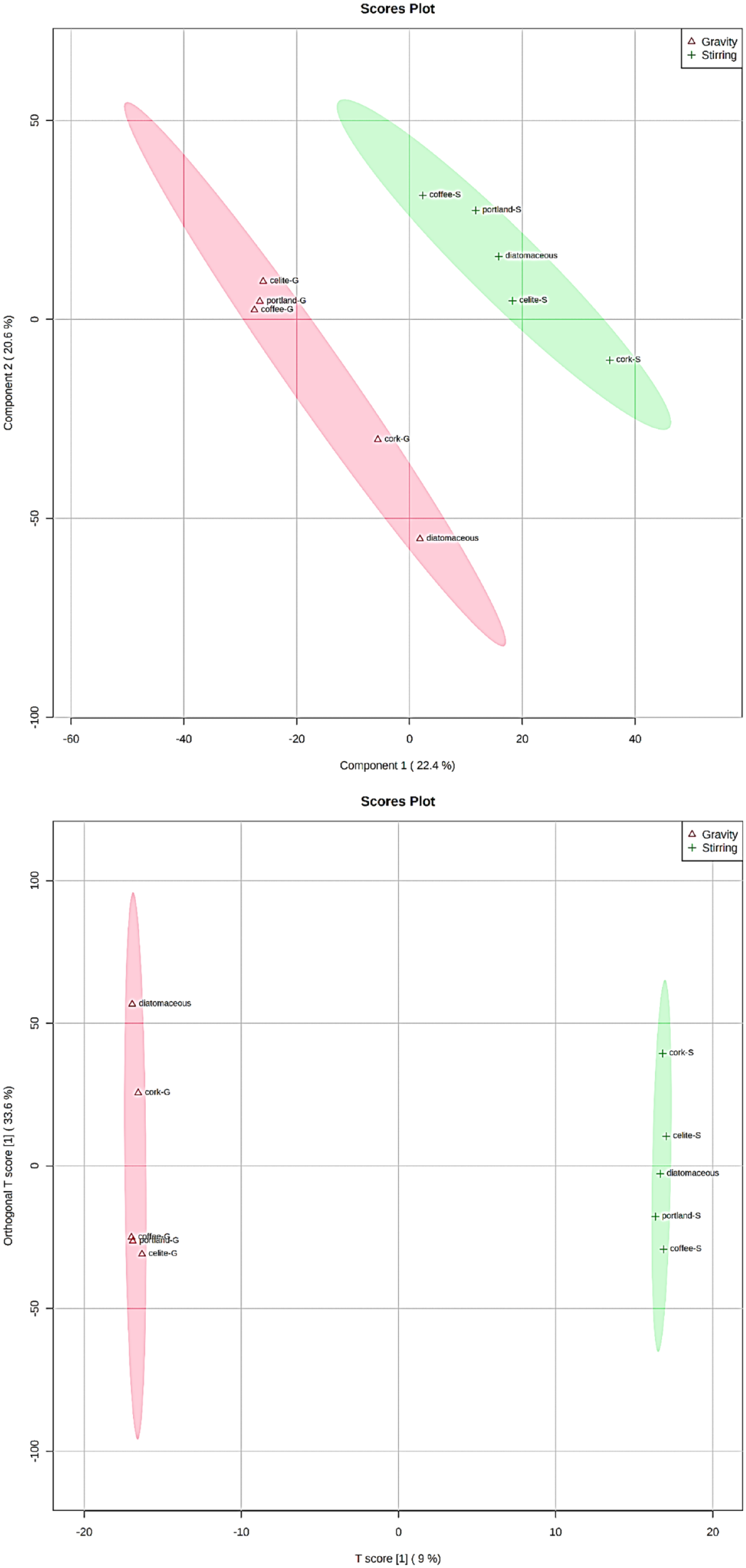
2D PLS-DA and o-PLS-DA score plots for WCO samples treated with different procedures.
Supervised PLS-DA methodology allows to generate two different clusters relative to samples of UVO treated with different adsorbents by stirring or by gravity filtering. Thus, the integrals more statistical relevant for each procedure which show a different value within all the adsorbents considered, can be isolated and used to determine a chemical fingerprint through the so-called variable importance in projection (VIP) plot (Figure 5).
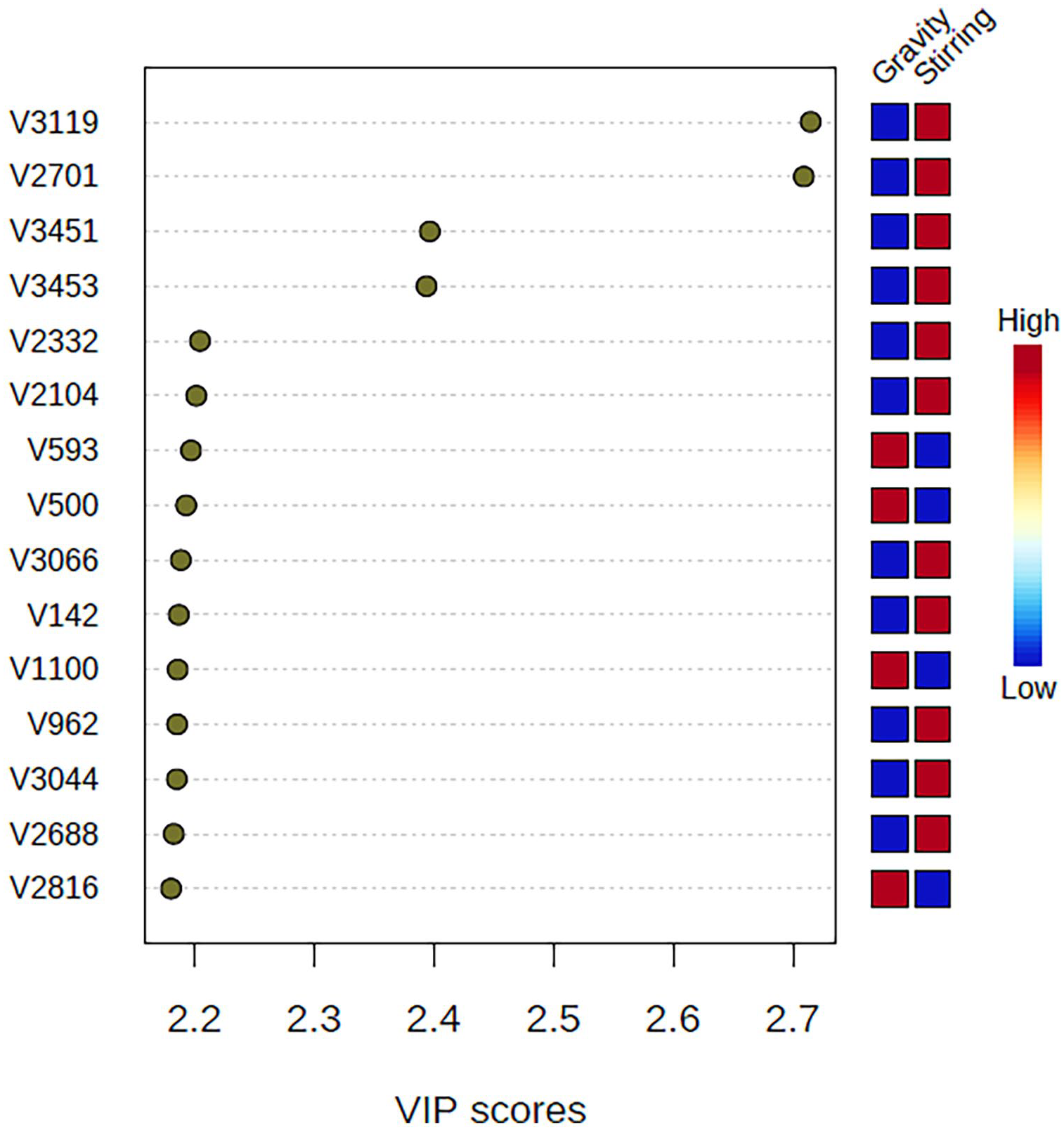
Important features identified by PLS-DA. The coloured boxes on the right indicate the relative concentrations of the corresponding FT-IR signals in each category considered.
As shown in Figure 5, each process can be characterized by a specific combination of 15 relative integrals which characterize the procedure independently of the adsorbent used. This sequence represents a chemical fingerprint typical to the recycling methodology and to the matrix considered.
Conclusions
Seven different adsorbent materials were screened in two different procedures aimed to recycle UVOs. The effectiveness of the adsorbents and of the specific process were assessed through chemical–physical analyses of oil samples prior and after treatment.
The current study concluded that treatment with porous materials can improve the chemical–physical properties of UVOs, especially in terms of density and FFA content. Considering the contamination and decomposition occurring during frying, the removal of FFAs as well as the lowering of density can be considered as key parameters in assessing the degree of regeneration of the waste. More in detail, the overall distribution of the FAs between oleic, linoleic and linolenic, is determined by 1H NMR, and the results are also affected by the treatment, although in minor extent. The specific characteristics of each powder influence the variation of the oil’s characteristics, which are related to the specific chemical profile, with celite, cork and diatomaceous earths resulting more active. In particular, celite seems to use for removing FFAs, while cork and diatomaceous earths can be suggested for lowering the density of the recycled oil. The celite seems to be efficient for each adsorbent should also be considered in combination with the specific process (stirring or gravity filtration), which can lead to different outcomes, as highlighted by FT-IR-based multivariate analysis. At the end, it is evident that the correct choice of the best adsorbent should be related to the specific application. In this context, the availability of the data provided in the current study can represent a useful toolbox for further industrial applications. In the end, starting from the above reported results would be possible to formulate specific combination of adsorbents. For example, in lubricant production (low density and absence of FFAs required), the values herein obtained (about 0.7 g/ml for density and 0.7% of FFAs) are already suitable and can be achieved with cheap sustainable powders which, in combination with the waste nature of the raw material, represent ideal candidates for designing a circular production.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The present work was funded by the WORLD Project-RISE, a project that has received funding from the European Union’s Horizon 2020 research and innovation programme, under the Marie Skłodowska-Curie, Grant Agreement No. 873005.