Abstract
The growing consumption of electrical and electronic equipment leads to high amounts of electronic waste (e-waste), which is now considered the fastest-growing waste stream at the national and international levels. As well as being a potential secondary resource due to its precious metals content, e-waste also contains strategic metals and plastics. For instance, mobile phones have about 25–55% plastic substances. A few studies have been performed to investigate the potential of indigenous bacteria in metals’ bioleaching from the polluted environment. Heterotrophic bioleaching potential in acidic conditions had been preliminarily investigated. Two soil types of iron ore were considered the source of indigenous bacteria. Despite the acidophilic nature of the bacterial consortium, they continued their leaching activity regardless of alkaline conditions. Maximum biorecovery rate related to copper (4%) responding to the main soil, owing to the higher copper content of mobile phone waste. Chromium had the least recovery rate (⩽0.002%). Overall, the maximum metal recovery rate was 4.7%, achieved by tailing heterotrophs at an e-waste loading of 10 g l−1. Statistical analysis had shown that there was no significant difference between the metal recovery rates and soil type or even the solid-liquid ratio (p > 0.05). Although acidophilic indigenous heterotrophs could not be an appropriate alternative for a large amount of metal recovery process, they might have considerable potential in the bioremediation of e-waste plastic fractions and metals in low concentrations simultaneously.
Introduction
Environmental protection as a constitute of ecological health is preventing undesired changes to ecosystems and their components (Hamilton et al., 2018). Therefore, it focuses on identifying, evaluating and eliminating environmental hazards (Nriagu, 2019). Tracking these environmental challenges is a beneficial step to identifying harmful exposures and implementing strategies towards reducing these risks, and creating health-supportive environments (WHO, 2018, 2020).
The era of accelerated technological progress has brought comfort to lives but also introduced harmful contaminants and pollution to the environment (Basu et al., 2022). The growing consumption of electrical and electronic equipment leads to high amounts of electronic waste (e-waste), which is now considered the fastest-growing waste stream both at the national and international levels (Abalansa et al., 2021; Forti et al., 2020; UN, 2017). E-waste is a heterogeneous combination of various materials (Purchase et al., 2020). As well as being a potential secondary resource due to its precious metals content, e-waste also contains strategic metals and plastics (Ankit et al., 2021).
Critical metals include aluminium, arsenic, cadmium, chromium, cobalt, gallium, lead, mercury, nickel, platinum, tin, titanium, zinc and other rare earth elements (FederalRegister, 2021; Rai et al., 2021). These metals exist on earth naturally and are also produced by humankind’s activities. Some of these metals are essential for the growth of organisms. Still, their higher concentrations significantly damage humans’ health since they are non-degradable and persist long-term in the ecosystem (Ayangbenro and Babalola, 2018). The adverse health effects on humans include severe toxic effects on the brain, heart, liver, kidney and skeletal system damage (Ali et al., 2021).
On the other hand, plastics represent more than about 20% of the mass content of waste electrical and electronic equipment (Basu et al., 2022). For instance, mobile phones have about 25–55% plastic substances. It was estimated that the consumption of plastics in electrical and electronic equipment would reach more than 6 billion pounds in 2022. By 2040, about 400 million tons of CO2 emissions will be expected due to global total e-waste plastic production and incineration (Singh et al., 2020). E-wastes contain various kinds of polymers. High-impact polystyrene (HIPS) and acrylonitrile butadiene styrene (ABS), polystyrene (PS), polyethylene, polypropylene (PP), styrene-acrylonitrile, acrylonitrile butadiene styrene/polycarbonate (ABS/PC), polyamide, polyurethane, polyphenylene oxide (HIPS/PPO), polybutadiene terephthalate, polyamide 66 (Nylon 66) and polyphenylene sulphide are known as the main polymers in printed circuit boards (PCBs). Also, PS, ABS and PP are the main polymers in e-waste constituting about 31%, 16% and 13%, respectively (Arshadi et al., 2019). That is why minimization of the harmful effects of the contained contaminants should come under serious consideration (Basu et al., 2022).
Although advanced incineration and modern landfills can effectively manage the e-waste issue, these technologies are not sustainable and appropriate for long-term application (Awasthi and Li, 2019). Therefore, it falls to the recycling industry to help sustainable waste management, environmental protection and resource conservation through removing hazardous materials and economic recovery of metal resources from e-waste (Awasthi and Li, 2019; Işıldar et al., 2018). However, recycling e-waste is very difficult due to its complexity and toxicity (Singh et al., 2020).
Yet, for the widespread application of e-waste recycling, it is essential to consider processes with a minor release of toxic substances and affordability (Rai et al., 2021). To this aim, several approaches have been recommended for effective and eco-friendly recycling of e-waste from which biological methods and bioremediation are widely accepted (Basu et al., 2022).
Microorganisms are appropriate candidates for solving complex problems in different environmental aspects of life due to their exclusivity and unpredictable nature, and biosynthetic capacity (Kumar and Gopal, 2015). A variety of microorganisms pose excellent capability for sustainable biotechnology, especially the heterotrophic communities with promising biosynthetic activity (Kumar and Gopal, 2015; Zuñiga et al., 2020).
Bioleaching is a promising technology for bioremediation that uses capable microorganisms to recover various metals from different minerals and waste materials, including e-waste, through microbial solubilization of metals (Gopikrishnan et al., 2020). Although bioleaching has been performed since prehistoric times, its application in metal recovery from e-waste is still emerging (Valix, 2017).
Many microorganisms are applied for metal recovery, especially those isolated from mining sites, including autotrophic and heterotrophic organisms (Gopikrishnan et al., 2020). Among autotrophs, iron- and sulphur-oxidizing bacteria, particularly bacteria from the genera Acidithiobacillus and Leptospirillum, are widely studied and reported to play a significant role in bioleaching, especially in acidic conditions (Gopikrishnan et al., 2020; Valix, 2017).
Regarding heterotrophic bacteria, there are few available reports; of those, the genera Bacillus and Pseudomonas are introduced as the most heterotrophic strains used in bioleaching of various metals. These bacteria consume organic carbon and secrete organic acids that help metal solubilization (Gopikrishnan et al., 2020; Valix, 2017). Since these are neutrophilic bacteria, their optimal growth pH is between 5 and 9 (Jin and Kirk, 2018). Alongside the metal solubilization, some reports regarding the biodegradation of high molecular weight organic polymers using potent microorganisms such as Aspergillus glaucus, A. niger, Pseudomonas sp., Bacillus siamensis, Klebsiella pneumonia, Micrococcus sp., Staphylococcus sp. and Moraxella sp. However, the degradation mechanism is not well-defined. Researchers have proposed different paths, including aerobic biodegradation through aerobic respiration resulting in H2O and CO2 release and molecular structure break of plastics due to the secreted enzymes and other extracellular compounds (Kathiresan, 2004; Zeenat et al., 2021).
There have been several studies on the bioleaching process using microorganisms isolated from different environments; meanwhile, a few studies have been performed to investigate the potential of indigenous microorganisms in the environment. Due to the abundance of heterotrophic bacteria, in this study, bioleaching potential in acidic conditions has preliminarily been investigated. They consider that the best bioleaching efficiencies typically occur at low pH values (Díaz et al., 2018). The plastic degradation capability of the isolated bacteria was also studied to develop efficient microorganisms and their products.
Materials and methods
Preparation of samples
In the present study, mobile phones were used to source heavy metals pollution. Waste-printed circuit boards (WPCBs) used in mobile phones usually contain greater concentrations of metals than those used in PCs (Bryan et al., 2020). These materials were shredded and sterilized through tyndallization (Baird et al., 2018). Inductively coupled plasma-mass (ICP -MS) spectrometry was used to characterize the primary concentration of heavy metals.
Acidophilic heterotrophic bacteria adaptation
The soil of Iran’s central iron ore located in the Bafq mining district was used as the source of heterotrophic consortium (Moore and Modabberi, 2003). In this matter, two soil samples, including the main soil and tailing soil of iron ore, namely M and T samples, were taken from the soil surface (0–30 cm) and mixed well to achieve homogenous samples.
Bacterial isolation was conducted by adding 10 g of each soil type to 100-ml flasks containing 50 ml nutrient solution and incubating in a shaker incubator (temperature of 30°C and agitation rate of 150 rpm) for 7 days (Hassanshahian and Ghoebani, 2018). Nutrient solution was composed of the following mineral salts: (NH4)2SO4 3.0 g l−1, KCl 0.1 g l−1, K2HPO4 0.5 g l−1, MgSO4.7H2O 0.5 g l−1, Ca(NO3)2.4H2O 0.01 g l−1 and 1% w/v glucose. Glucose was filter-sterilized by passage through 0.2-μm membrane filters. To establish acidic conditions, the pH of the solutions was adjusted to 3 (Keenleyside, 2019). All culture media and chemicals were of analytical grade, purchased from Merck. H2SO4 solution (1N) was used for pH adjustment.
To enumerate acidophilic heterotrophic populations, sterilized phosphate buffer with pH = 3 was used. 0.1 ml inoculum of each dilution was transferred to Petri dishes containing sodium thiosulphate agar and glucose and then were incubated at 30°C for 24–48 hours.
Adaptation was performed inside a shaker-incubator; by adding 30 ml bacterial suspension (previous section) into 250-ml flasks containing 30 ml of appropriate minerals with pH = 3, and 30 mg crushed e-waste. The flasks were incubated for 7 days (30°C and 150 rpm), and their e-waste components gradually increased to 100 mg during 21 days of the adaptation period. A fresh medium was slowly added to the flasks, and pH was controlled periodically to maintain the appropriate conditions for the growth of the acidophilic heterotrophs.
Heavy metals solubilization
The bacterial suspension was transferred to perform heavy metals leaching for 15 days as a bioleaching experiment. Three different solid-liquid ratios (S/L) were considered, including 5, 10 and 15 g l−1. All experiments were carried out in 100-ml flasks containing 60 ml appropriate medium and 10% v/v bacterial suspension (Figure 1).
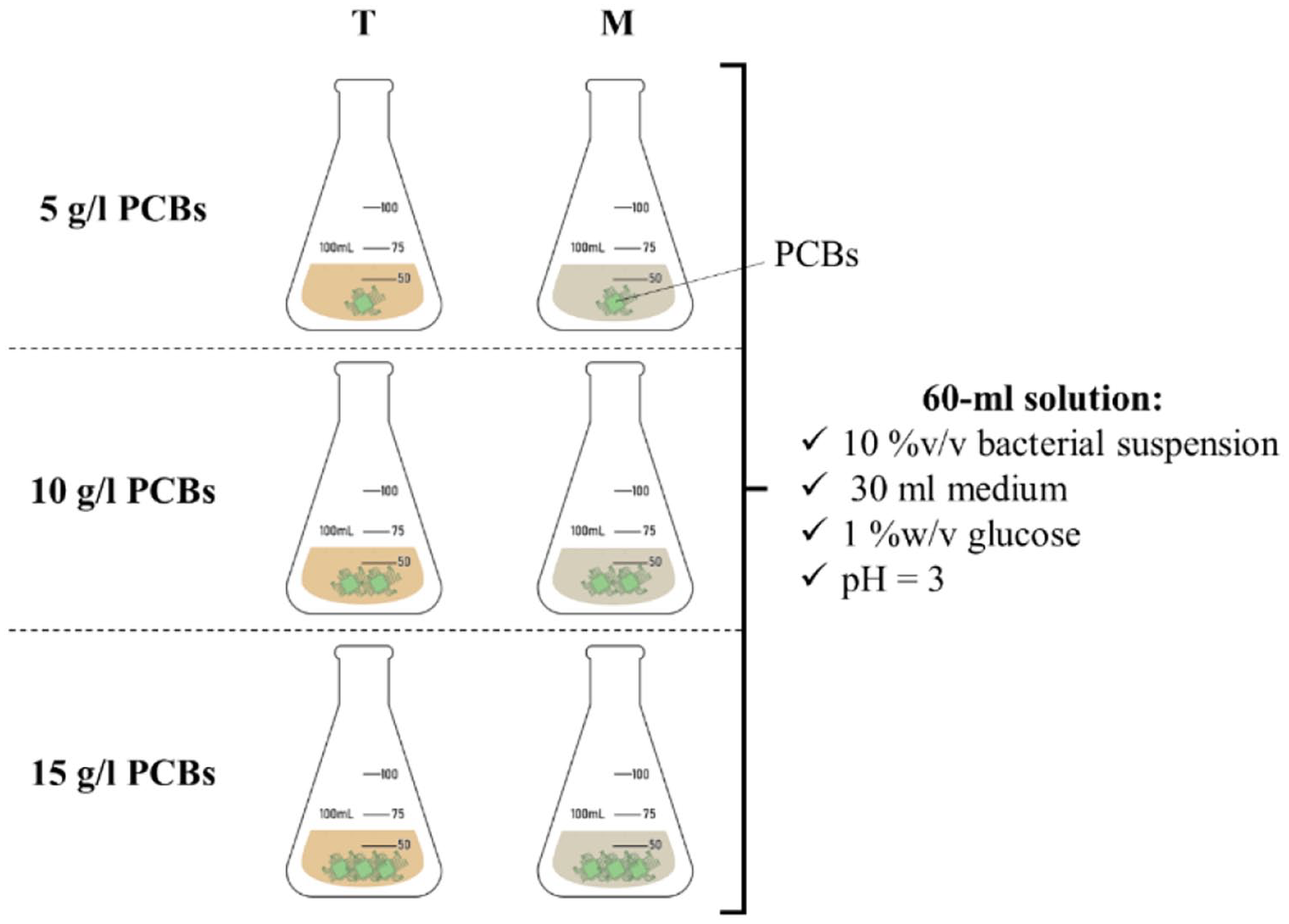
Schematic of bioleaching setups (pH = 3, bacterial suspension = 10%, glucose = 1% w/v).
Additionally, pH, optical density (OD595) and dissolved oxygen (DO) were measured periodically. Also, leached metal concentration was determined by ICP-MS weekly. At last, e-waste weight change was investigated.
Statistical analysis
SPSS software version 26 (Two-way ANOVA analysis) was used to compare the mean differences between metals’ recovery rates.
Results and discussion
Mobile phone contents and soil bacteria enumeration
So far, several pieces of research have been performed using autotrophs, and less is available about heterotrophs in metal recovery. In contrast, heterotrophic bacteria have great potential for remediation of organic pollutants. The main purpose of this study was to investigate the ability of the heterotrophic consortium of mineral areas to recover heavy metals from polluted environments. Figure 2 shows the concentration of total metals in the mobile phone waste in this survey.
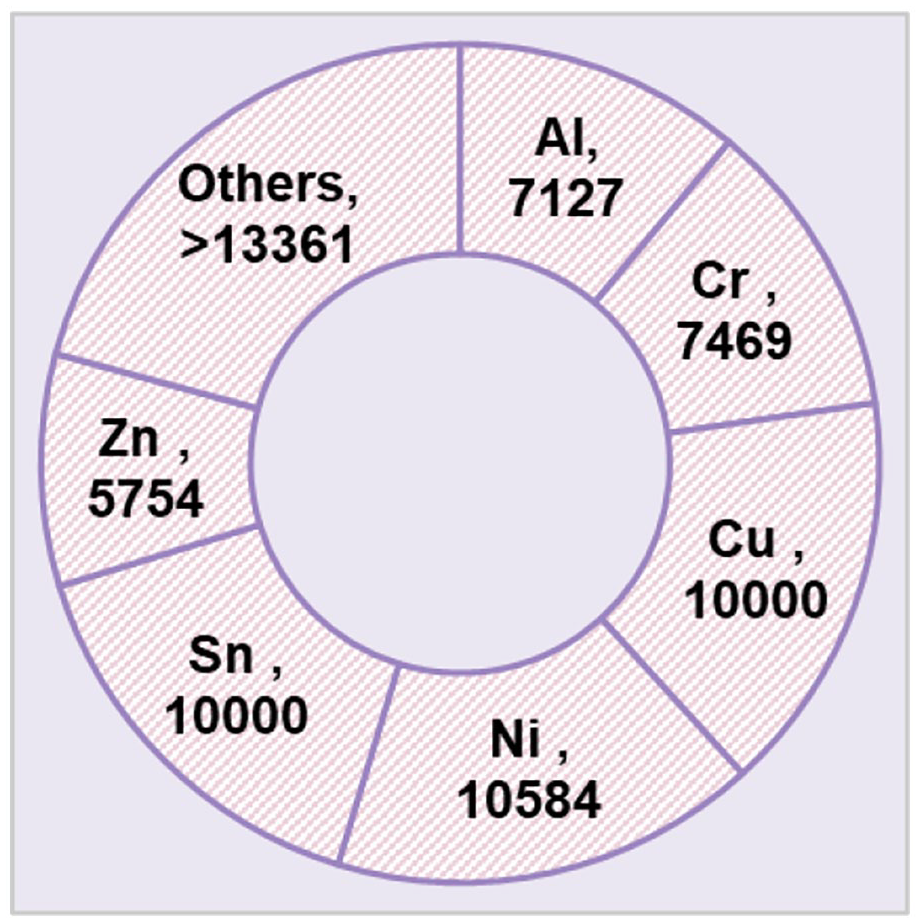
The concentrations of metals in WPCBs as ppm (mobile phone).
According to Figure 2, nickel, copper, tin, chromium, aluminium and zinc were measured as heavy metals with the highest concentrations in the mobile phone waste. It was reported that, on average, PCBs typically are made up of about 7.0% Fe, 27.0% Cu, 2.0% Al, 1.5% Zn, 0.5% Ni, 2000 ppm (Ajiboye et al., 2019). Ajiboye et al. (2019) analysed the WPCBs metal composition and observed that most metals included base metals such as copper (41.64%), iron (11.11%), aluminium (5.05%), nickel (2.28%), zinc (1.79%), tin (1.63%) and lead (1.09%). Yamane et al. (2011) also declared that their studied PCBs from discarded computers mainly consist of copper, lead, aluminium, iron, tin, cadmium and nickel.
The present study did not investigate other metals, including sodium, potassium, magnesium, iron, etc. Due to the abundance of tailing in the Bafq iron mine area, two soil samples were investigated, including the main soil and tailing. Also, the community and morphology of bacterial isolates are indicated in Figure 3.
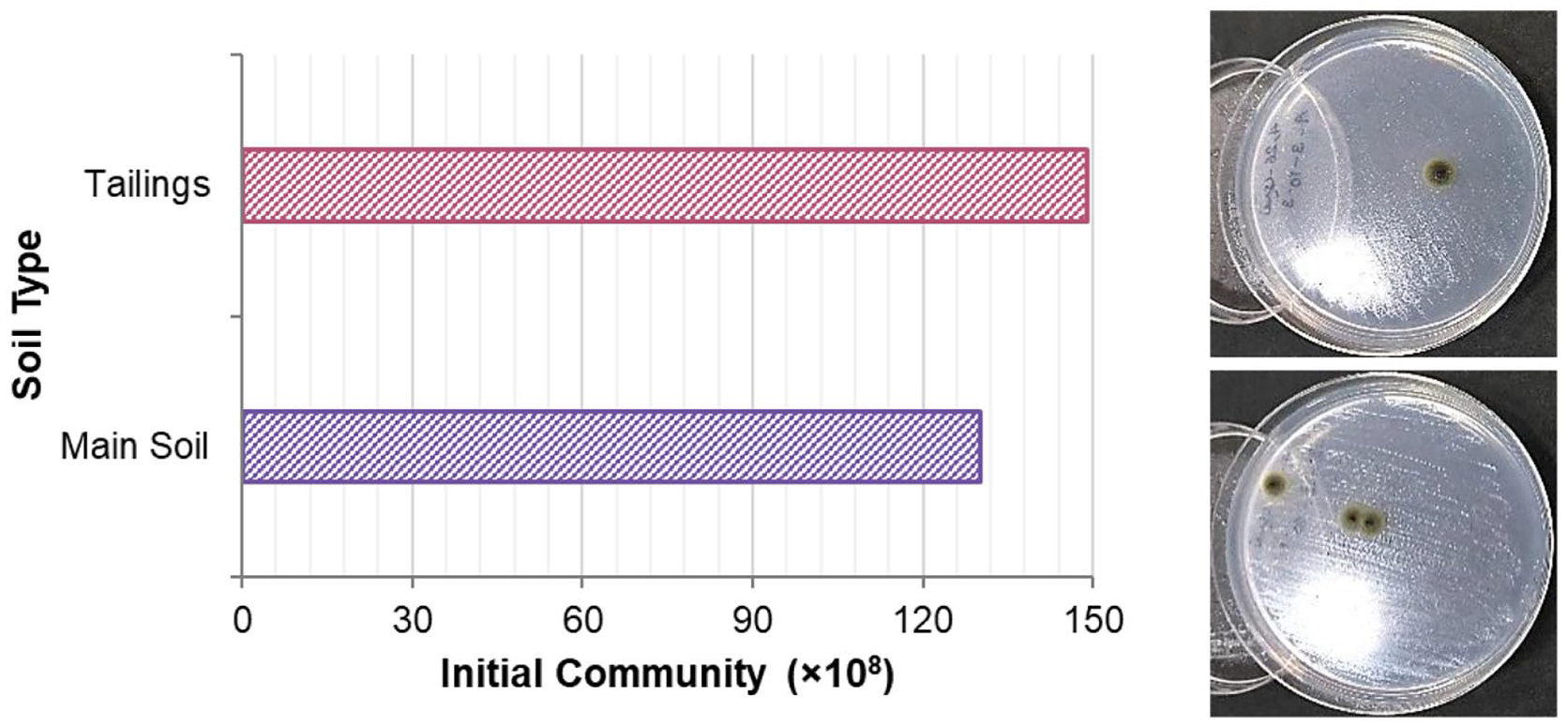
Initial bacterial community and morphological characteristics as the soil type.
As shown in Figure 3, tailing had a bit more bacterial community than the main soil. However, both soil samples had enough bacterial content to perform bioleaching experiments. Also, the formed bacterial colonies of both samples were green with a dry surface, a dark core and lighter edges.
Bacterial adaptation
Bacterial adaptation was carried out through the gradual addition of the WPCBs. Alongside, main environmental parameters, including OD595 and DO, were monitored, and the results are presented in Figure 4.
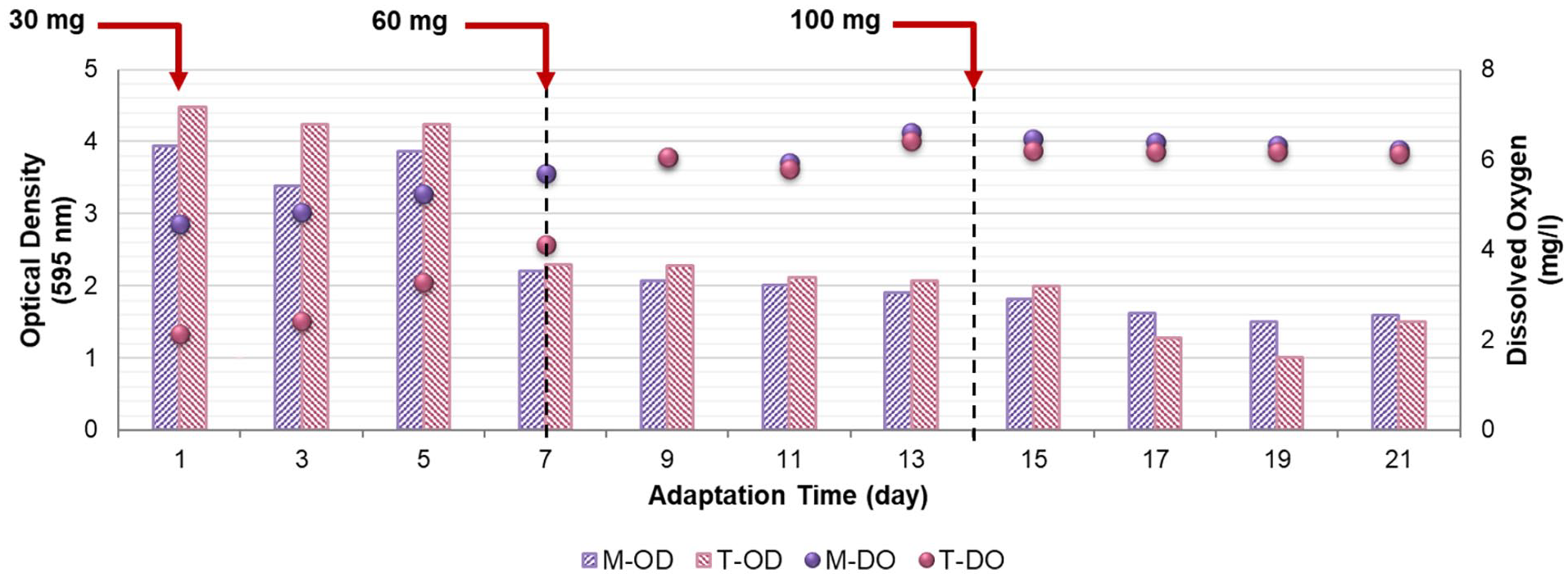
OD595 and DO variations during bacterial adaptation (e-waste was gradually added in three stages with concentrations of 30, 60 and 100 mg).
On the first days of adaptation, high optical densities were observed. However, weekly WPCBs level-up decreased the OD trend (Figure 3). As opposed to OD595, DO charts had an increasing trend during the adaptation process, which could probably result from continuous agitation inside the incubator shaker providing the required oxygen for the aerobic bacteria (Bates et al., 2016). The decreasing trend of OD can be attributed to two reasons; Adding higher loadings of WPCBs increases the toxicity of the medium containing the bacteria, which can affect bacterial growth (Wu et al., 2018).
Also, according to some papers, high concentrations of DO lead to an inhibitory effect on bacterial growth, despite oxygen being an electron acceptor through aerobic bacterial metabolism (Baez and Shiloach, 2014; Guezennec et al., 2016, 2017; Li et al., 2015). This deleterious effect could be due to the oxidative stress induced by the activation of reactive oxygen species due to the change in oxidation states of metal ions in the medium (Baez and Shiloach, 2014; Guezennec et al., 2017).
Bioleaching experiments
Figure 5 shows the environmental parameters measured during bioleaching experiments.
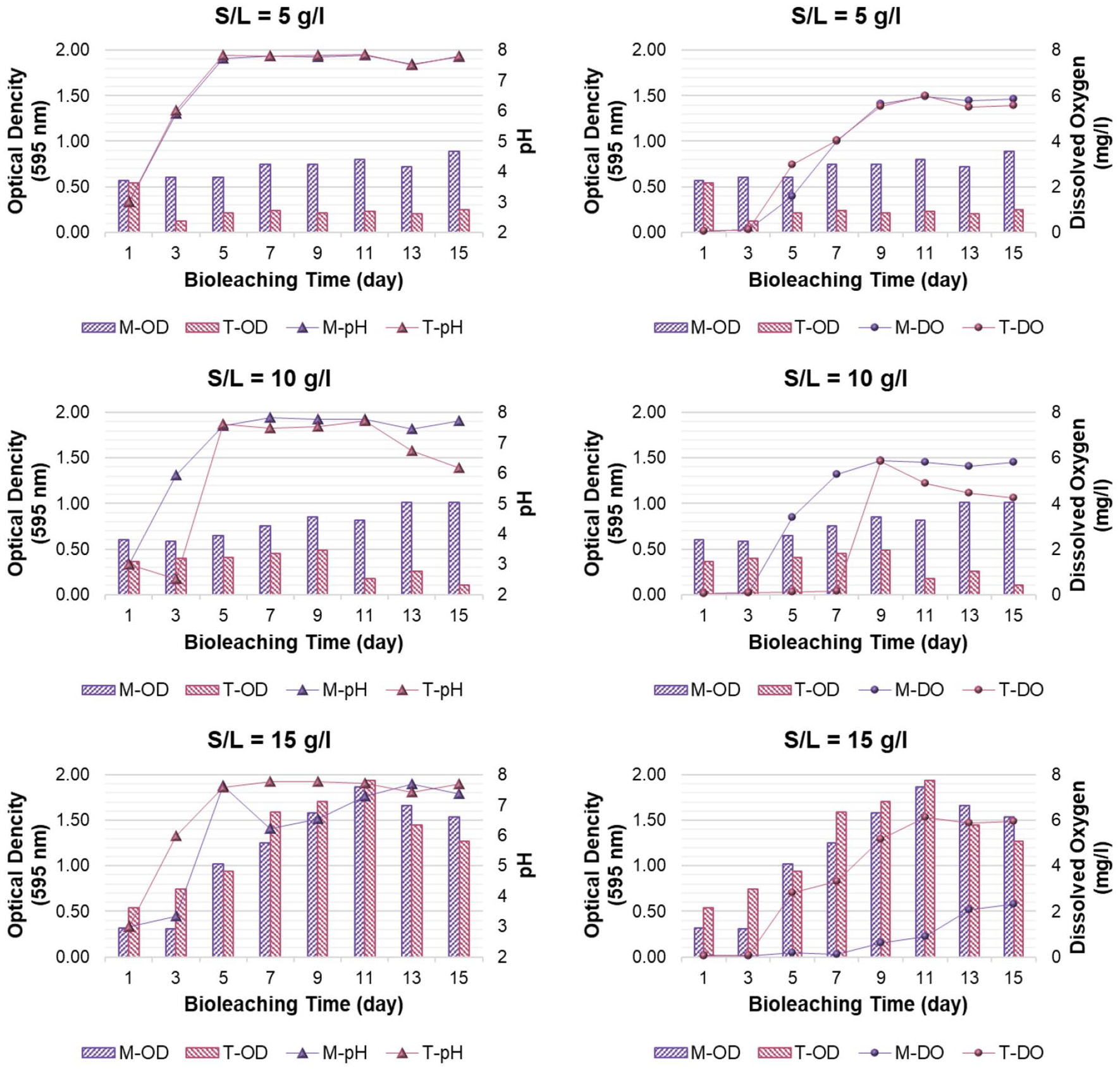
pH, OD595 and DO variations during bioleaching.
As shown in Figure 5, in all bioleaching set-ups, the bacterial consortia of the main soil showed increasing OD. It seems that the preadapted heterotrophs of tailing have maintained their growth trait, being influenced negatively by increasing DO during the bioleaching period. Although microorganisms need more time to adapt to higher levels of e-waste (Shaikh et al., 2018), it can be observed that the main soil heterotrophic bacteria have adapted well to the toxic e-waste. However, at S/L = 15 g l−1, the isolates of the main soil and tailing had a similar OD trend, showing decreasing growth on the ending days, which can be due to the high toxicity of e-waste loading (Wu et al., 2018).
Although the isolates were meant to be acidophiles acidifying the medium, an opposite phenomenon was observed. In all bioleaching set-ups, pH tended to have neutral/slightly alkaline values.
The fact is that WPCBs have an alkaline nature resulting in the release of alkaline compounds (Rasoulnia et al., 2021). Since heterotrophic bacteria produce organic acid due to carbon consumption, this leads to a decrease in the medium pH (Feliatra et al., 2019; Mei et al., 2021). Yet, the amount produced organic acids in this study was not enough to neutralize the alkalinity of e-waste.
Also, the bacterial communities were monitored periodically. Heterotrophic enumeration changes during the bioleaching process are displayed in Figure 6.
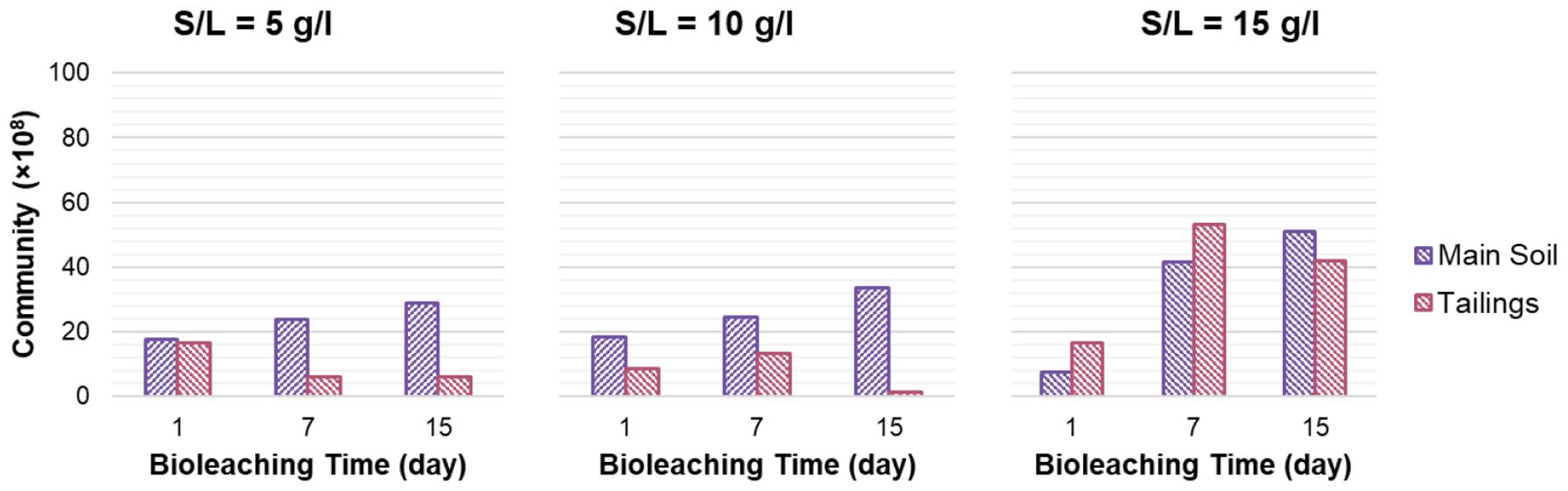
Variation of heterotrophic bacterial communities during metals solubilizations.
Although it is believed that higher concentrations of e-waste lead to inhibition of bacterial activity (Wu et al., 2018), the main soil bacterial community showed an increasing growth trend during bioleaching of all three WPCBs loadings in this study (Figure 6), suggesting that the studied conditions are providing the favoured substances, especially carbon source for these bacteria. Debbarma et al. isolated some indigenous microbial strains possessing the potential to degrade e-waste plastics. As they observed, the microbial consortium showed accelerated growth in the presence of e-waste (Debbarma et al., 2018). Meanwhile, the bacterial community of tailing soil decreased during the process, except for the density of 15 g l−1, in which the bacterial population of tailing soil showed an increasing trend within the first week of bioleaching and then started to decrease (Figure 6).
The calculated rate of metal recovery is presented in Table 1.
Metal recovery rate achieved by acidophilic heterotrophs*.
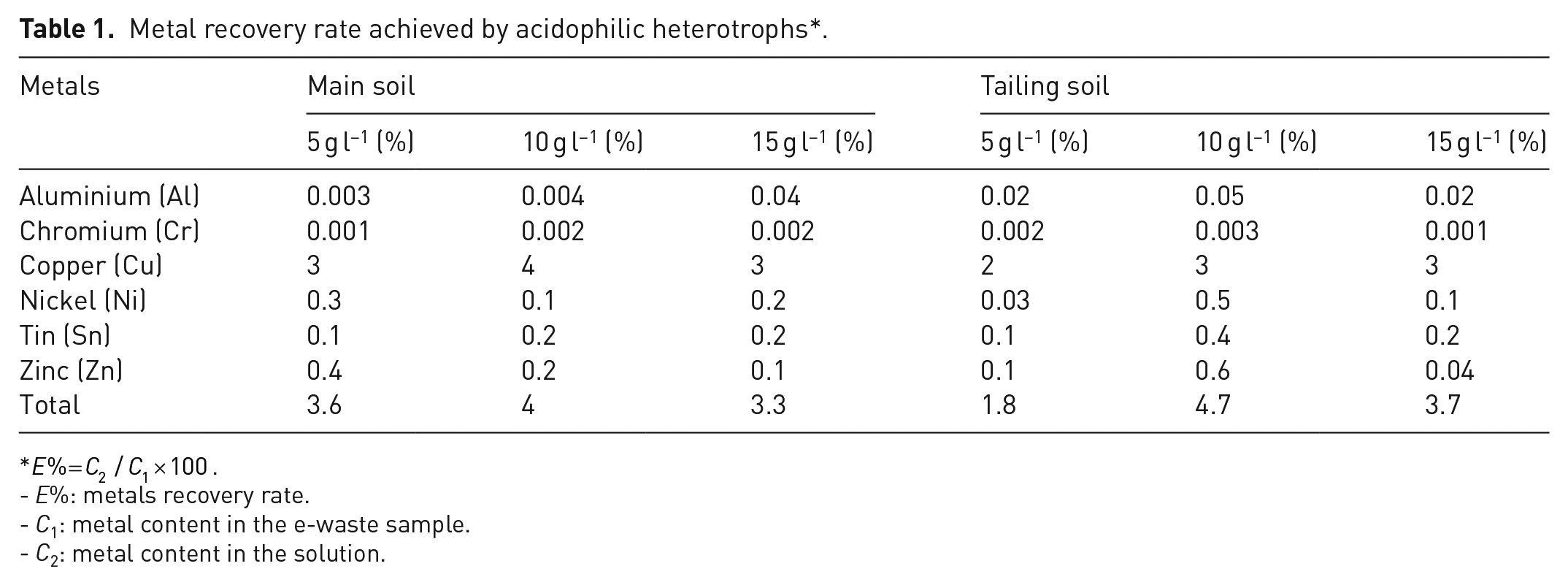
- E%: metals recovery rate.
- C1: metal content in the e-waste sample.
- C2: metal content in the solution.
As presented in Table 1, the hiighest biorecovery rate corresponded to copper (4%) by heterotrophs of the main soil at S/L = 10 g l−1. The higher leaching value of copper could be owing to the large proportion of this metal in WPCBs. The biorecovery rates for other metals were negligible; among them, chromium was the metal with the least recovery rate (⩽0.002%). It might mean that the isolates were more susceptible to chromium than other metals. Rodríguez et al. investigated the tolerance behaviour of some heterotrophic bacteria isolated from the natural waters of a mining area. They reported an overall toxicity pattern for all tested bacteria (Hg > Co > Cr > Zn > Ag = Cu), showing that chromium was more toxic than copper (Escamilla-Rodríguez et al., 2021).
Figure 7 compares the weight changes of different e-waste loadings (5, 10 and 15 g l−1) during bioleaching experiments.
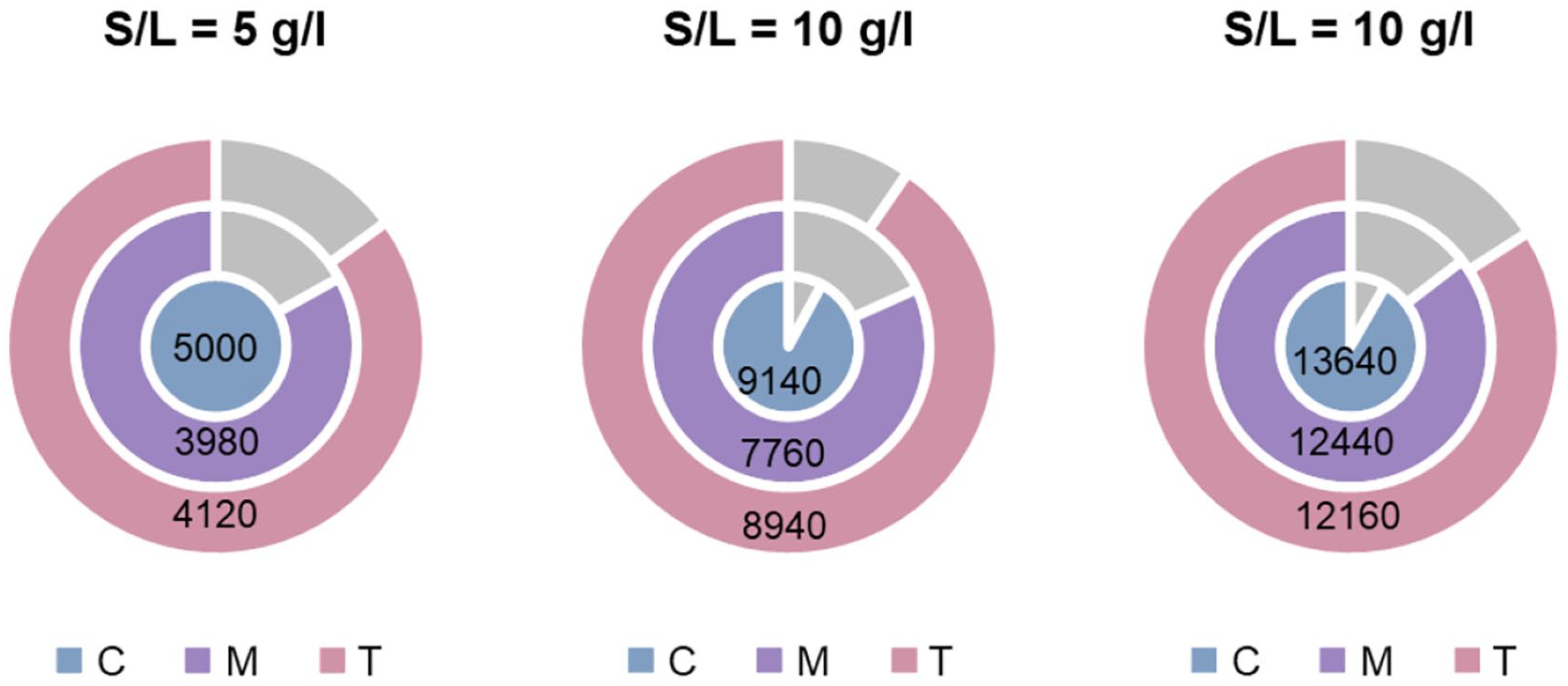
Changes in WPCBs weight (mg) after bioleaching process based on S/L ratio.
Considering total studied metals, the highest recovery rate was 4.7%, achieved by heterotrophs of tailing at S/L = 10 g l−1. Regarding the WPCBs weight decrease due to bioleaching, the most weight change was done by the heterotrophs of tailing at S/L = 15 g l−1, leading to a 2840 mg decrease (Figure 7). Although the mechanism of the considerable WPCBs weight decrease is not surgically known, the capability of these acidophilic heterotrophs in this matter suggests their probable ability to consume plastics as a source of carbon or to penetrate easily into the polymers and biodegrade them into monomers (Kathiresan, 2004; Zeenat et al., 2021). Although these heterotrophic isolates did not show significant metal leaching performance, their application in the remediation of plastic pollution could be beneficial.
Analysed surface morphology by SEM microscopy is illustrated in Figure 8.

WPCBs appearance (a) before and (b) after bioleaching process.
The results from SEM analysis (Figure 8) indicated that the bioleached sample had a more crushed surface than the raw one, which confirms the bacterial activity.
Statistical analysis
Correlations of metals’ biorecovery rate with solid-liquid ratio and soil type were evaluated using Two-way ANOVA tests (Table 2).
Correlation of biorecovered metals with soil type and solid-liquid ratio.
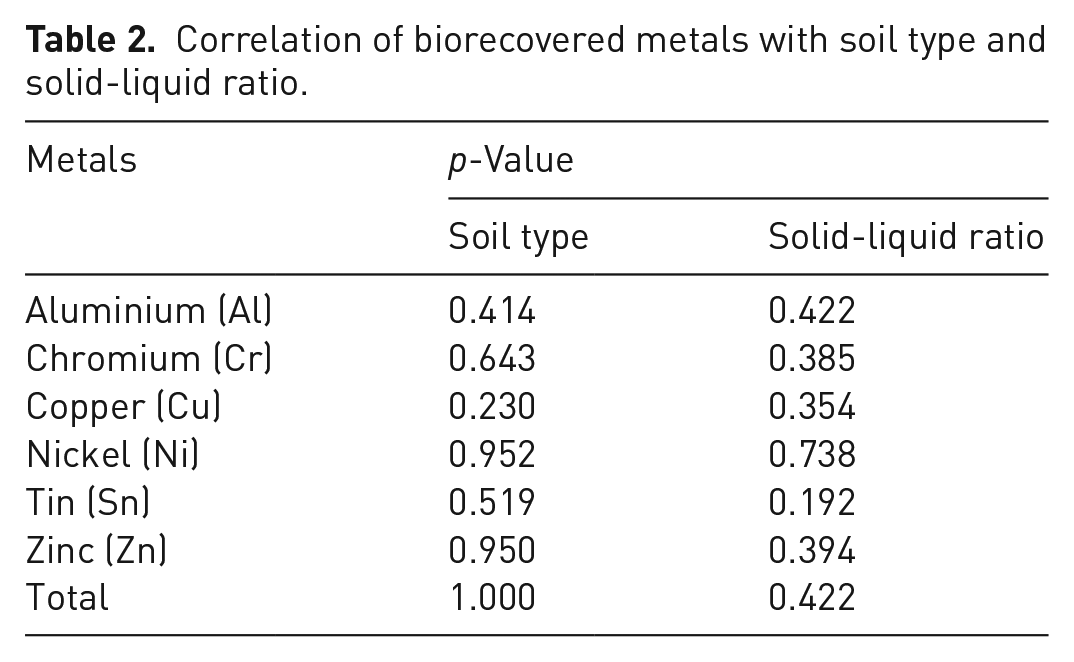
As shown, there was no significant difference between the metal recovery rates and soil type or even the solid-liquid ratio (p > 0.05).
Conclusions
The main objective of this study was to obtain preliminary data on the feasibility of the heterotrophic consortia in solubilizing hazardous metals and degrading plastics contained in WPCBs in acidic conditions. The iron mine soil had shown rich total heterotrophic bacterial counts of up to 130 × 104 and 149 × 104 in the main soil and the tailing soil, respectively.
In bioleaching set-ups, pH tended to neutral/slightly alkaline values, probably due to the alkaline nature of the WPCBs. Despite the acidophilic nature of the bacterial consortium, they continued their leaching activity regardless of alkaline conditions. The highest biorecovery rate was achieved by heterotrophs of the main soil corresponding to copper (4%) at S/L = 10 g l−1, owing to the higher amount of this metal in WPCBs. Other metals had a nominal bioleaching rate; among them, chromium showed the least recovery rate (⩽0.002%). It might be concluded that chromium has a higher degree of toxicity to these bacteria. Hence, it is suggested to conduct quantification tests to measure the toxicity level of the heavy metals and determine the bacterial tolerance towards these toxic metals. Considering the entire metals involved, the highest recovery rate was 4.7%, achieved by heterotrophs of tailing at S/L = 10 g l−1. Although acidophilic indigenous heterotrophs could not be an appropriate alternative for the extensive metal recovery process, they could have considerable potential in the bioremediation of WPCBs plastic fractions and metals in low concentrations simultaneously. This reveals that central iron ore soil of Iran country can be an appropriate source of microorganisms with potential for plastic degradation.
Footnotes
Acknowledgements
Authors would like to thank Iran central iron ore company for supplying the soil samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical consideration
This study was approved at Ethics committee of Shahid Sadoughi University of Medical Sciences. (ID: IR.SSU.SPH.REC.1399.053).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project funded by Shahid Sadoughi University of Medical Sciences. (ID: 7980).
