Abstract
In this study, polypropylene (PP) was recycled in a non-stirred batch reactor by slow pyrolysis at low temperature. Virgin PP and waste PP as well as mixed material of equal amounts of virgin PP plus virgin PP pyrolysis oil (ratio 1:1 w/w) were used as raw material. The highest yields of liquid product were obtained at 350°C and 400°C (82.0 and 82.3 w/w%, respectively). The density, viscosity and calorific value of the gasoline and diesel fractions of the obtained pyrolysis oils comply with EN228 and EN590 standards, respectively. The flash point corresponded to the standard only for some of the oils, but the cold filter clogging point, the pour point and especially the oxidation stability were far above the stated reference values of the standards. The pyrolysis oils as products of thermal decomposition were determined by the methods of 1H and 13C and two-dimensional-heteronuclear single quantum coherence nuclear magnetic resonance (2D-HSQC NMR) spectra. Spectral analysis showed that only very little aromatic compounds were present in the oils, but they contained many unsaturated compounds, which is presumably consistent with the measured oxidation stability and limits their use in the production of alternative fuels. The research octane number (RON) calculated from the NMR analyses corresponds to the lower limit of gasoline.
Introduction
Rapid urbanisation and economic development has led to the production of around 59 million tonnes of plastic per year in Europe in 2015 (Kassargy et al., 2017). Due to their unique properties such as lightweight, mechanical strength, corrosion resistance and, depending on the application and other specially developed properties, plastics are widely used in daily practice. The excessive use of these plastics results in large amounts of waste. Various methods are used for their disposal, such as incineration, landfilling, reuse and conversion into value-added products (Ahmad et al., 2015). Pyrolysis (thermal decomposition) has emerged as a promising option to make plastic waste useful for both the waste and energy industries (Singh et al., 2019). The main advantage of pyrolysis is the conversion of substrates into gaseous, liquid and solid fractions. Pyrolysis has been used for thousands of years to produce charcoal and carbon, and more recently pyrolysis has been used to produce oil, activated carbon, coke, carbon fibres and methanol (Butler et al., 2011; Sharma et al., 2014). The main factors affecting the plastic pyrolysis process and the molecular distribution of the pyrolysis products include the chemical composition of the feedstock, the pyrolysis temperature and heating rate, and the type of reactor. For example, the conversion rate increases with the degree of branching of the polymers: HDPE < LDPE < PP < PS (HDPE = high-density polyethylene, LDPE = low density polyethylene, PP = polypropylene, PS = polystyrene). The reaction rate also increases with increasing stability of the radicals formed during the pyrolysis process (Westerhout et al., 1997).
Influences of other factors on the pyrolysis of polymers are described in Sharuddin et al. (2016), Abbas-Abadi et al. (2014) and Buekens (2006). Polyolefins such as HDPE, LDPE and PP are the ideal waste plastics for the production of diesel and gasoline fuel (Panda et al., 2019). Pyrolysis of PP by changing the parameters to optimise the yield of liquid oil has been studied several times (Abbas-Abadi et al., 2014; Ahmad et al., 2015; Miskolczi et al., 2009). The yield of liquid products was generally highest (70–80%) in the low temperature range up to about 500°C. Above 500°C, the yield reduces with increasing gas content to a maximum of 50% (Vijayakumar and Jilse, 2018).
However, to the authors’ knowledge, there is no single successful and widely licenced technology in the literature for producing fuels by pyrolysis. Some configurations produce good-quality fuel, but usually more intensive processing is required (Butler et al., 2011; Kassargy et al., 2018).
Waste PP is distinguished from virgin PP by its age, its use and the specific material changes it undergoes. The PP ages and degrades through proliferation, cracking, and so on (White, 2006). Analyses showed that oxygen is the main element associated with the changes in the aged plastic surface (Canopoli et al., 2020). Even in buried PP, the carbonyl index was 1.5–2 times higher than that of fresh material after 10 years (Canopoli et al., 2020). Heteroatoms promote the cleavage of the alpha bonds next to them (Zhang et al., 2018).
Although recycling of waste PP is cheaper than that of virgin PP, the inferior properties and impurities of waste PP raise concerns about its effectiveness (Azeez, 2020). However, most studies have focused mainly on virgin PP (Anene et al., 2018; Yan et al., 2015). Other studies have investigated virgin PP and PP waste, but attributed the differences to the impurities and food residues of PP waste (Ciliz et al., 2004).
Almost all of the current literature and applications of pyrolysis focus on high temperatures, high heating rates and ground feedstock. This allows simplification of the highly complex pyrolysis process (Ranzi et al., 1997). However, pyrolysis at high temperatures makes most pyrolytic processes uneconomical, dangerous and they can also produce highly toxic gases that require treatment before they can be discharged to the atmosphere. Most heat sources are furnaces that cause some of the feedstock to thermally degrade under slightly different conditions, which affects the results (Jouhara et al., 2018). To increase the production of high-quality liquid products (fuel), the concept of slow pyrolysis can be applied. The advantage of slow pyrolysis over fast pyrolysis is manifold, especially the long duration in slow pyrolysis leads to better heat transfer and high liquid yield. A detailed understanding of slow pyrolysis, including the effect of the long duration, is still lacking in terms of product distribution to the target value added, and only a few reports are available (Das and Tiwari, 2018a).
In this study, virgin and waste PP and a mixture of solid virgin PP are pyrolysed with the pyrolysis oil of virgin PP (ratio 1:1 w/w) at low temperatures and low heating rate with the aim of reducing the amount of waste plastic and combining this reduction with a useful application such as the production of transport fuel. First, the optimal temperature was determined to obtain the highest possible liquid fraction at atmospheric pressure. The liquid oil obtained was analysed by nuclear magnetic resonance (NMR) spectroscopy. Subsequently, the liquid oil was separated into a diesel and a gasoline hydrocarbon fraction, which were characterised in terms of their fuel parameters, and an analysis of the fuel properties as a function of the type of PP feedstock and the process parameters was carried out.
Materials and methods
Materials
Two types of PP plastic raw materials were used: PP virgin material (JSC Retal Baltic) and PP waste from a secondary sorting plant (JSC Waste Management Center of Klaipeda Region). PP waste without composites or without visually obvious filler or additive content was used. The PP waste from the secondary sorting plant was washed with water and dried. The PP raw material was shredded into particles of ~25 mm2.
Procedure of the pyrolysis
Pyrolysis was carried out at atmospheric pressure in a non-stirred 4.5-L steel batch reactor (internal height 295 mm, internal diameter 190 mm) with a single outlet. The thermocouple in a sleeve protrudes 140 mm from the top into the reactor chamber. The reactor stands upright in the muffle furnace with the reactor head (60 mm internal height) protruding from the furnace. The outlet from the top plate on the reactor head opens via an air-cooled connection into a double-neck attachment, which sits on a flask for the pyrolysis oil and is connected to a water-cooled reflux condenser (approx. 15°C temperature) on the second neck. The reflux condenser is only used for complete condensation of the pyrolysis oil. The furnace heats the bottom and the side wall of the reactor to the desired temperature.
The reactor is filled to approximately 75% with the PP raw material (500 g) by pressing in. For the copyrolysis 500 g of a mixture of virgin PP with the pyrolysis oil of virgin PP in the ratio 1:1 w/w (called ‘mixture PP’ in short) were used. After closing and connecting the reactor, it is placed in the oven and connected to the flask and reflux condenser with the connecting piece. After the oven is switched on and the final temperature is reached, it is maintained by manual control until no more drops of condensate flow into the flasks within a time interval of 10 minutes. The rate of temperature increase ranged from 2.9°C to 3.4°C minute−1, so the process time was about 2 hours. The condensed liquid product (pyrolysis oil) was collected in a glass collecting flask; the gas was discharged through the tube built into the reactor; the solid residue remained in the reactor and was removed from it at the end of the process. The liquid phase was filtered through Whatman No. 4 filter paper to remove solids. Each pyrolysis was repeated thrice.
Distillation of the PP pyrolysis oil to recover the hydrocarbon fraction for gasoline and diesel
The pyrolysis oil was distilled by separating the fraction boiling at 190°C (hereafter referred to as the gasoline range fraction) and the fraction boiling at 190–320°C (diesel range fraction). The substance remaining in the container after atmospheric distillation is a residue fraction (>320°C).
NMR analysis
NMR spectral analysis of the pyrolysis oil 1H and 13C NMR spectra were recorded using a VarianUnity Inova 300 spectrometer at 302 K (29°C). The chemical shifts (δ) of the hydrogen and carbon atoms of the pyrolysis oil were recorded by comparison with the reference tetramethylsilane (TMS, δ = 0 ppm) in deuterated chloroform (CDCl3) with a chemical shift δ = 7.26 ppm for 1H NMR spectra and δ = 77.0 ppm for 13C NMR spectra. Interactions between aliphatic carbon and the bonded hydrogen atoms were detected with two-dimensional heteronuclear single quantum correlation (2D-HSQC) NMR spectra.
Physical properties of the pyrolysis liquids
The density was determined according to EN ISO 12185:2001 (2001) with a DMA 4500 densimeter. The viscosity was determined according to EN ISO 3104:2020 (2020) with a capillary viscometer at 40°C. The calorific value was determined according to EN ISO 8217:2017 (2017) with an IKA calorimeter C500300 C. The oxidation stability was determined according to EN 16091:2011 (2011) with an automatic oxidation stability analyser PetroOXY. Flash point was determined according to EN 22719:1993 (1993) using an FP93 5G2 automatic analyser, which allows the determination of the flash point of the closed Pensky–Martens crucible of petroleum products. The pour point was determined according to ISO 3016:2019 (2019) using a fully automatic CPP 5Gs instrument to measure the pour point of the fuel. The analyses were performed in three replicates.
Statistical analysis
One-way analysis of variance (ANOVA) was used to compare the yield of pyrolysis products and the physical properties of the liquid fraction. The mean values were compared using Fisher’s least significant difference test. The statistical significance of the results was determined at p < 0.05. The statistical software R 3.4.4 was used (R Core Team, 2019).
Results and discussion
Effect of temperature on product yield (mass balance)
In order to determine the optimum pyrolysis temperature, a slow low temperature pyrolysis was carried out with virgin PP. The yields of liquid, gaseous and solid products from virgin PP obtained from non-catalytic degradation at 300°C, 350°C and 400°C are shown in Figure 1.
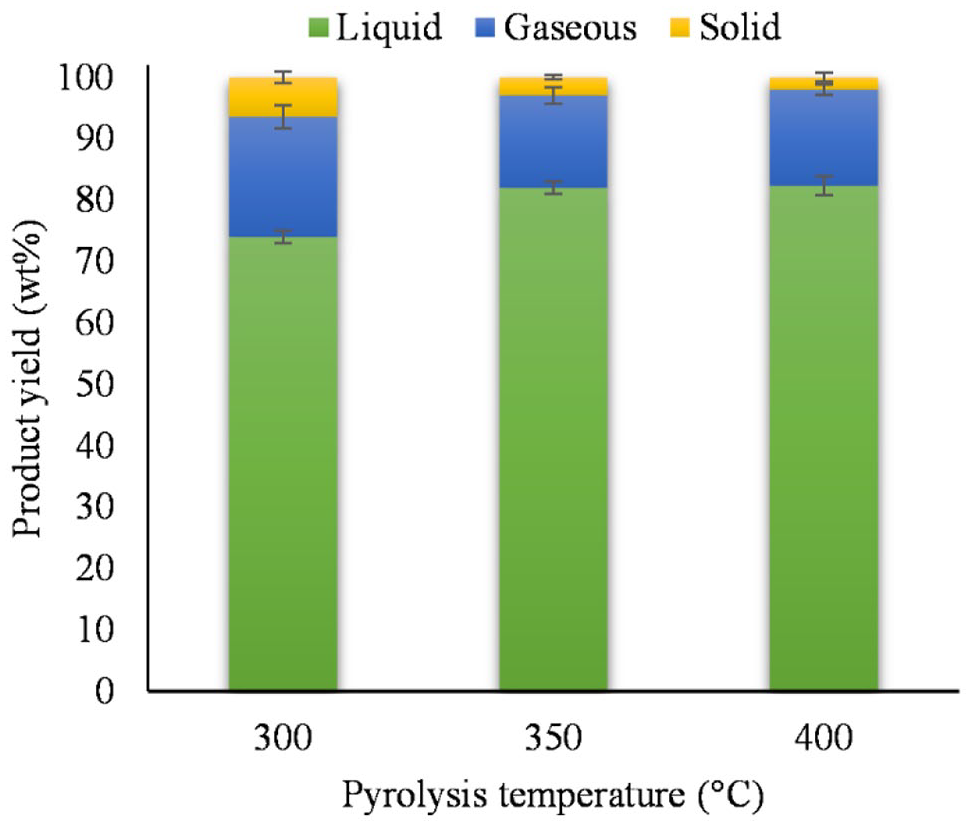
PP yields of pyrolysis products at different temperatures (mean ± standard deviation).
The results of the study show (Figure 1) that the pyrolysis temperature affects the yield of virgin PP products. Pyrolysis of virgin PP at 300°C (74.0 wt%) yields less (p < 0.001) liquid product than pyrolysis at 350°C or 400°C (82.0 and 82.3 wt%). Some authors suggest that this could be due to the branched structure of the easily degradable polyolefin (Ahmad et al., 2015). In principle, however, the high liquid oil yields confirm the conclusion of Vijayakumar and Jilse (2018) and Wirawan and Farizal (2019), who recommend pyrolysis temperatures below 500°C.
The largest amount of gas is obtained mainly from pyrolysis at 300°C with 19.6 wt%, which is more than from pyrolysis at 350°C or 400°C (p = 0.016). Pyrolysis at 350°C or 400°C gives almost the same gas yields (15.0 and 15.6 wt%). When evaluating the formation of solid residues, pyrolysis with virgin PP feedstock at 300°C (6.4 wt%) was found to produce more solid residues (p < 0.001) than pyrolysis at 350°C (3.0 wt%) and 400°C (2.0 wt%).
A similar trend in terms of solid yield was also observed in López et al. (2011), according to which the solid content decreased with increasing temperature. However, the trend was much weaker than in our case. In contrast the liquid yield in this study increased slightly with increasing pyrolysis temperature. The gas yield decreased to the same extent. Although the reactor design of López et al. (2011) is very similar to that of this study, the results do not seem to be easily extrapolated to the results of this study. However, the heating rate of the furnace in this study is significantly lower, no nitrogen gas is passed through the reactor and the volume of the reactor is larger in this study. Due to the lower heating rate and the lack of nitrogen gas flow, the pyrolysis material in this study remains longer in the reactor and can form more liquid pyrolysate, as described in López et al. (2011); Seth and Sarkar (2004); Hujuri et al. (2011); Shah et al. (2010). The extended residence time of the feedstock in the reactor enables secondary reactions and significantly alters the slow pyrolysis with slow heating of the products (Jouhara et al., 2018). For the further investigations, PP pyrolysis at 350°C was chosen.
Effect of the raw material PP on the product yields (mass balance)
Two pyrolysis processes were carried out: feed with virgin PP and feed with waste PP. The yield of the liquid fraction at 350°C depended on the type of PP feedstock (Figure 2). The results of the study showed that the lowest yield of liquid product at 350°C was obtained when pyrolysis was carried out with waste PP (69.7 wt%), but when virgin PP was used, the yield obtained was higher (82.0 wt%) (p < 0.001). A similar yield of the liquid fraction of 65.44 wt% from the pyrolysis of PP waste (temperature 460°C) was obtained by Yan et al. (2015). In Sogancioglu et al. (2017), higher liquid fraction yields – 81.47 and 79.62 wt% – were obtained with washed and unwashed waste PP at 300°C, while at 400°C the yields were 80.55 and 78.70 wt%. However, this also shows that the reflux in the reactor at the low temperatures at the beginning of the pyrolysis does not lead to an increased yield of liquid product, as described, for example, in Seth and Sarkar (2004).
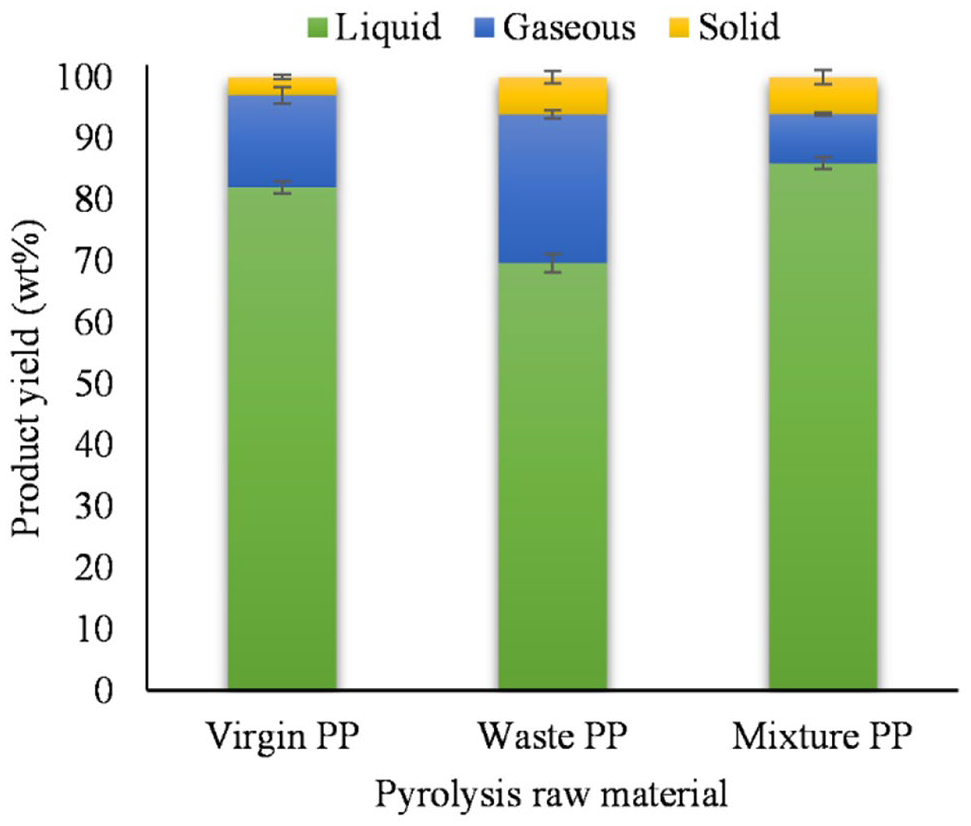
Yields of pyrolysis products at 350°C with different PP feedstocks (mean ± standard deviation).
Pyrolysis with virgin PP produces 15.0 wt% gaseous phases, which is less than pyrolysis with waste PP at 24.3 wt% (p < 0.001). A similar amount of a gaseous fraction with virgin PP (450°C) was obtained by Kassargy et al. (2017), and when the raw material was waste PP, it was 19 wt% (Kassargy et al., 2018). In Singh et al. (2019), a gas fraction of 9.5 wt% was obtained when the raw material was waste PP and the pyrolysis temperature was 450°C.
It was found that the formation of solid residue was lower in the pyrolysis of virgin PP (3.0 wt%) than in the pyrolysis with waste PP (6.1 wt%) (p = 0.009). In Singh et al. (2019), a higher amount of solid residue of 8.0 wt% was obtained when the raw material was waste PP and the pyrolysis temperature was 450°C.
Physical properties of PP pyrolysis oil
Pyrolysis products are sources of hydrocarbons that can have a wide range of applications. Therefore, to assess the feasibility of using liquid pyrolysis products from different PP feedstocks (virgin PP, waste PP) in fuel production, their physical properties were determined (Table 1). The quality of the pyrolysis oil obtained in the trials was assessed against a number of parameters that are important for the fuel and can determine the possibilities for further refining or direct use.
Physical properties of the liquid fraction of PP pyrolysis.
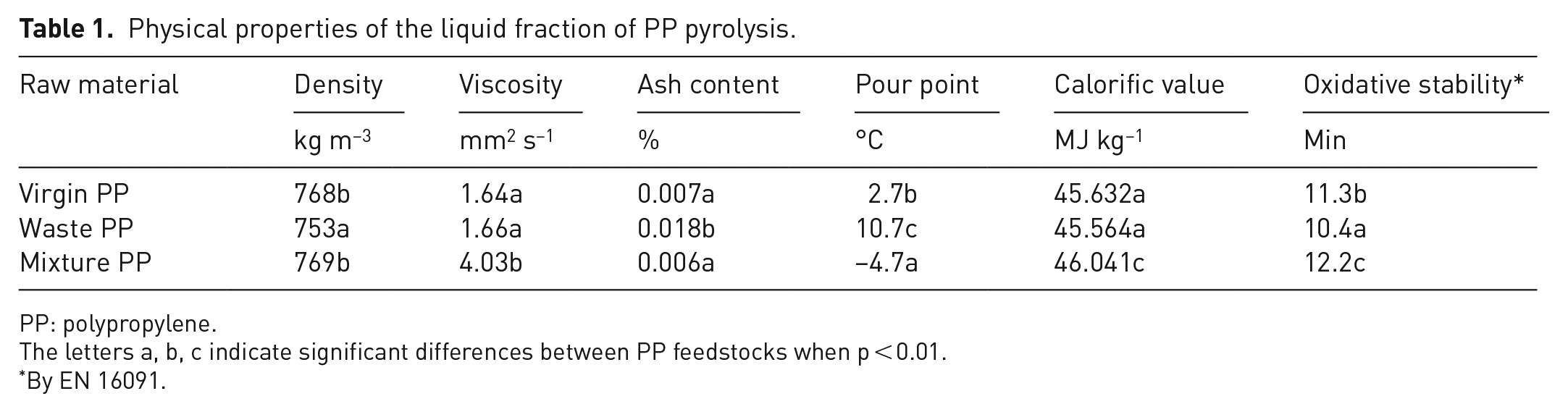
PP: polypropylene.
The letters a, b, c indicate significant differences between PP feedstocks when p < 0.01.
By EN 16091.
The results of the study showed that when using PP waste for pyrolysis, the density of the oils is lower (753 kg m–3) than when using virgin PP (768 kg m–3) as raw material (p < 0.001). The density values we obtained are very similar to those found by Singh et al. (2019) at a temperature of 300–600°C. Virgin PP (1.64 mm2 s−1) and waste PP (1.66 mm2 s−1) resulted in lower viscosity of the liquid (p < 0.001). The slow pyrolysis of different PP feedstocks at low temperature has a higher viscosity value than mentioned in Kassargy et al. (2017). When the feedstock was waste PP, the pour point was 10.7°C, but when the feedstock was virgin PP, it was 2.7°C (p < 0.001). When virgin PP was used as feedstock for pyrolysis, the calorific value and oxidation stability values were higher than when the feedstock was waste PP (p = 0.001 and p = 0.003, respectively). The low oxidation stability may be due to the high proportion of compounds with double bonds (Pullen and Saeed, 2012).
The liquid fraction obtained from different PP feedstocks during the pyrolysis process was characterised by low oxidation stability and a high pour point. The low oxidation stability of PP oils leads to additional problems in use, transport, storage and so on. As can be seen from Table 1, the liquid fractions obtained from different PP feedstocks do not meet the fuel requirements (neither for gasoline nor for diesel).
Pyrolysis of a mixture of virgin PP with the pyrolysis oil of virgin PP
A second pyrolysis was now carried out in a 1:1 (w/w) mixture of virgin PP and the pyrolysis oil from virgin PP. It was expected that similar to Seth and Sarkar (2004) or Abnisa et al. (2014), a prolonged distillation under reflux would lead to improved products. As already described above in section ‘Influence of temperature on product yields’, the reactor dimensioning should lead to a prolonged residence time of the mixture in the reactor and, assuming as described in Jouhara et al. (2018), to secondary reactions and significantly changed pyrolysis products. The second pyrolysis was again carried out at 350°C.
The liquid yield was 86.0 wt% and is higher than in the pyrolysis of solid, virgin PP without oil addition (p < 0.001). However, this is to be expected in such a case. In the pyrolysis of the mixture (Abnisa et al., 2014), which presumably took place at almost the same reaction temperature and other reaction conditions, the fresh virgin PP may have reacted with the double bonds of the pyrolysis oil. Since the liquid fraction has increased somewhat while the gas fraction of about 8.0 wt% has decreased compared with the other two pyrolysis processes, this seems to have taken place. But presumably for the hydrocarbons with an already high proportion of double bonds or preformed structure to the polyaromatic, this reaction is in competition with the dehydrogenation to solid, carbon-rich solids, the proportion of 6.0 wt% has also increased somewhat.
As shown in Table 1, no changes in physical properties were observed. The expectations based on Seth and Sarkar (2004) were therefore not fulfilled.
Spectral analysis of PP pyrolysis oils
The NMR analysis of PP pyrolysis oil showed no difference between the pyrolysis oil obtained from virgin PP feedstock and that obtained from waste PP, therefore the spectra of waste PP pyrolysis oil are not shown. Analysis of the 1H NMR spectra of pyrolysis oil from virgin PP and PP blend, shown in Figure 3, indicates that their chemical composition is quite similar. Pyrolysis oil from virgin PP (3a) has chemical shifts ranging from 0.89 to 4.77 ppm for all protons and blend PP (3b) has chemical shifts ranging from 0.89 to 4.79 ppm for all protons. No signals were observed in the spectrum that could be associated with aromatic protons.
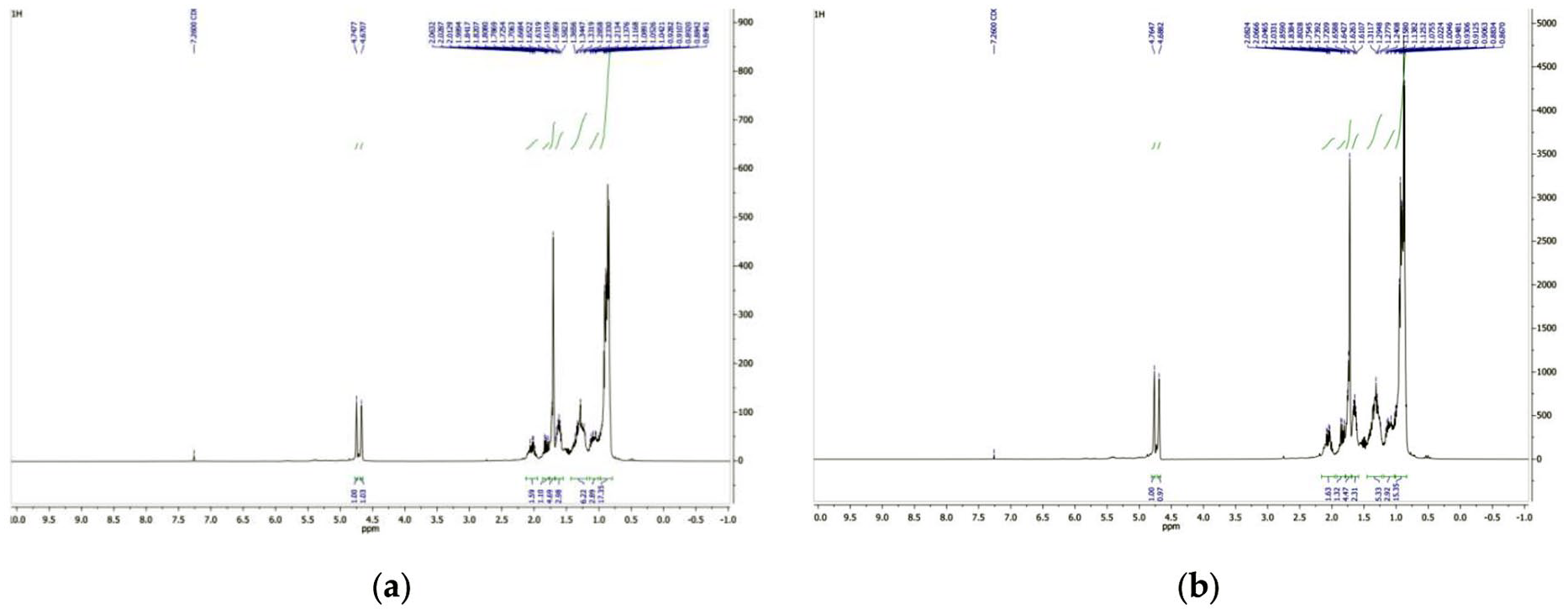
1H NMR spectra of virgin PP (a) and mixture PP (b) pyrolysis oil.
The spectra of the 1H NMR products show two equivalent signals with chemical shifts in the spectrum of the virgin PP pyrolysis oil of 4.70–4.77 ppm and 4.71–4.79 ppm of the mixture PP, respectively. The obtained ratio of these protons is 1:1. It is known from the literature that such an image belongs to the terminal double-bonded protons of the methylidene group (Pullen and Saeed, 2012). Signals with chemical shifts of 0.89–2.10 ppm for virgin PP pyrolysis oil and 0.89–2.13 ppm for the PP mixture in the 1H NMR spectra are assigned to the alkyl groups of the aliphatic and cyclic fragments, respectively.
The 13C NMR spectra of virgin PP and blend PP pyrolysis oil are shown in Figure 4.
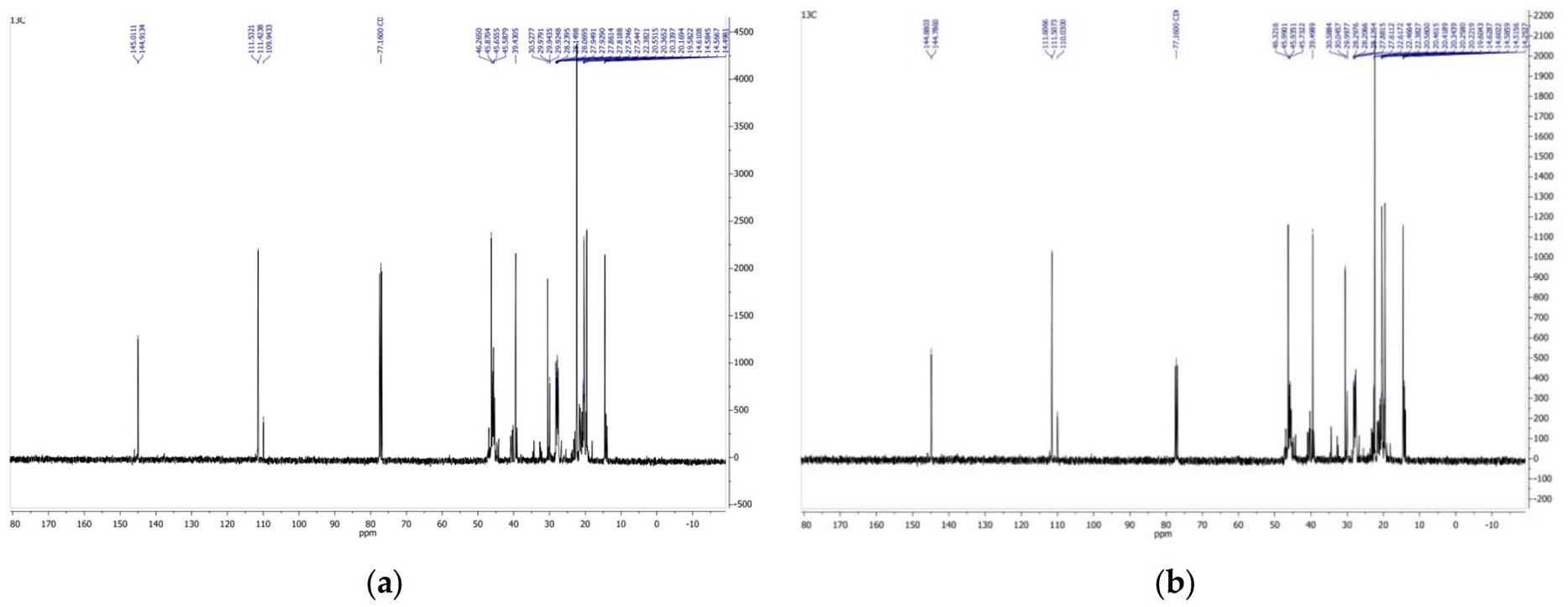
13C NMR spectra of virgin PP (a) and mixed PP (b) pyrolysis oil.
Examination of the 13C NMR spectra (Figure 4) shows that the carbon atoms in the alkene appear sp2 hybridised in the spectrum between 100 and 150 ppm. The 13C NMR spectrum shows that the carbon atoms of the pyrolysis oils with chemical shifts from 14.4 to 46.0 ppm belong to paraffinic carbon atoms, and the signals with chemical shifts of 109.7, 111.2, 111.3, 144.7 and 144.8 ppm can be assigned to the sp2 hybridisation carbon atoms. The spectral data obtained show that the pyrolysis products have unsaturated compounds. According to the literature (Hesse et al., 2005; Ure et al., 2019), the signals obtained can be assigned to α-alkenes with a chemical shift of 100–150 ppm.
Other authors (Myers et al., 1975) studied gasoline and showed that 1H NMR spectra can be used to identify groups of compounds in fuels with high accuracy and estimate their molar ratios. The signal peaks do not overlap and the compounds can be determined quite accurately. The authors propose to use NMR analysis not only for qualitative but also for quantitative determination of the amounts of compounds. Using NMR spectroscopy, Myers et al. (1975) and Ure et al. (2019) identified the pyrolysis compounds by class and calculated the content of paraffin, olefins and aromatic compounds in the pyrolysis oil obtained by slow PP pyrolysis. In their study, unlike ours, small amounts of aromatic compounds were detected. The chemical shifts in the signals of the 1H and 13C NMR spectra of the pyrolysis oil compounds determined by the authors are in agreement with the chemical shifts found in our study.
To confirm and further analyse the chemical composition of the obtained pyrolysis products, their 2D-HSQC spectra were recorded. The obtained HSQC spectra of all three PP liquid fractions (PP virgin, PP waste and PP mixture) are almost identical, therefore only the spectrum of the PP mixture is presented, discussed and shown in Figure 5.
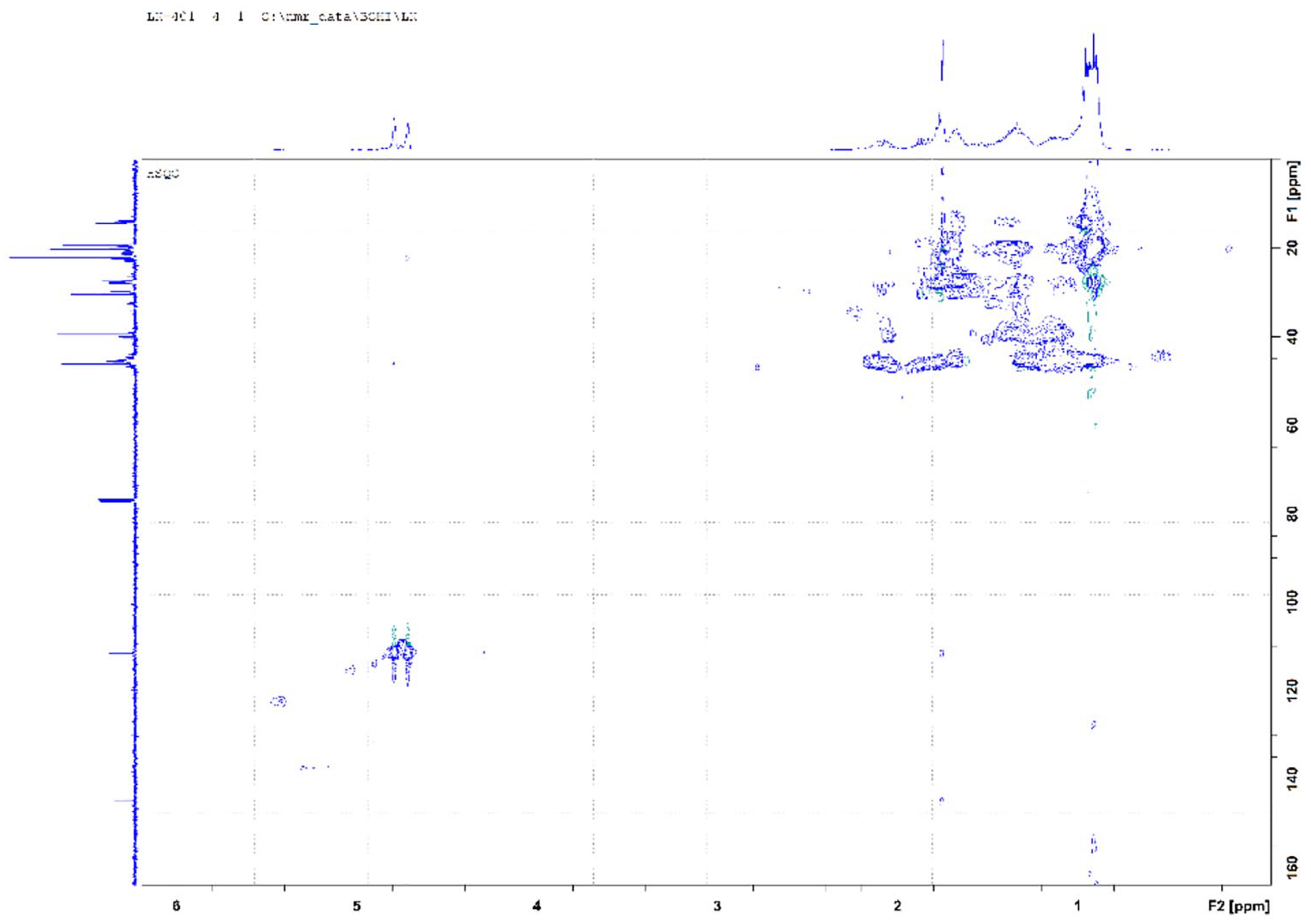
HSQC spectrum of the PP-pyrolysis oil mixture.
The 2D-HSQC spectrum shows that 1H NMR signals with chemical shifts of 4.69 and 4.77 ppm belong to two hydrogen atoms attached to the first methylidene carbon atom with chemical shifts of 109.8, 111.3 and 111.4 ppm in the 13C NMR part of the α-alkenes.
Only a small fraction of CH3 groups with a chemical shift of 1.73 ppm are attached to the second unsaturated carbon atom, and a large fraction of such methyl groups are attached to aliphatic and cyclic structures, and as confirmed by the analysis of the 2D-HSQC spectra, the pyrolysis oils contain no or very few aromatic compounds, which was already mentioned when discussing the 1H NMR spectra of virgin PP and mixed PP pyrolysis oil.
Onwudili et al. (2009) analysed the NMR spectra of pyrolysis oils and found that for pyrolysis carried out at higher temperatures, the paraffin content in the pyrolysis oil decreased due to secondary cyclisation and aromatisation reactions when the pyrolysis was carried out above 500°C and under high pressure (4.31 MPa). In the pyrolysis carried out in this study at atmospheric pressure and 350°C, only secondary cyclisation reactions took place, but no aromatic compounds were formed. Other authors found that at the beginning of PP pyrolysis (up to 390 minutes) the yield of olefins is maximum (Das and Tiwari, 2018a). With increasing pyrolysis time, the concentration of paraffin increases and the concentration of olefins decreases. The temperature of the pyrolysis had the same effect. Das and Tiwari (2018b) and Almeida and de Fatima Marques (2016) have shown that the pyrolysis of LD or HDPE as well as PP produces a high proportion of paraffins and olefins. This was also proven by Das and Tiwari (2018b) by 1H-NMR spectroscopy.
In the pyrolysis process, α-alkenes are formed during the β-cleavage reactions, while saturated compounds are obtained in the initiation and termination reactions (Almeida and de Fatima Marques, 2016). The amount of saturated compounds in the product indicates the extent of initiation and termination reactions, and the amount of α-alkenes indicates the potential for β-cleavage reactions to occur. The obtained results show that during PP pyrolysis not only β-decomposition occurs intensively, but also other reactions in which many compounds with different chemical structures are formed, confirming that the process proceeds according to the mechanism of thermal fission. The C,C-single bond is the weakest compound of polyalkenes (Marcilla et al., 2009), and beta cleavage stabilises a radical, creating a double bond. According to Almeida and de Fatima Marques (2016) or Miskolczi and Nagy (2012), the pyrolysis mechanism is still being studied and discussed. Estimation of the amount of unsaturated compounds in the product is very important to decide on further use.
The fact that the different pyrolysis oils appear to be identical in terms of NMR and other properties could confirm that there are no differences in the chemical processes involved in the pyrolysis of virgin and recycled PP. However, a recycled and then presumably aged PP may be oxidised, which have activating effects on the neighbouring bonds. However, since the further mechanism is probably similar, a similar product is formed. This was not to be expected from the outset, however, as different polymers can interact with each other even if their chemistry is different (see, for example, Wu et al., 2020).
NMR spectroscopy can be used to calculate the chemical and physical properties of petroleum products (Edwards, 2011; Petrakis and Fraissard, 1984). For this purpose, the spectra are divided into six ranges and the signals of these ranges are integrated using the scheme of Das and Tiwari (2018b) (Table 2 lists these values for the pyrolysis oils of virgin and mixed PP).
NMR spectral region and integral area of the pyrolysis oils of virgin and mixed PP.

NMR: nuclear magnetic resonance; PP: polypropylene.
Table 3 shows the percentages of paraffinic, olefinic and aromatic hydrocarbons by volume calculated from the values in Table 2, as well as the H/C ratio, the isoparaffin index and the RON value according to the equations from Das and Tiwari (2018b).
Properties of the pyrolysis oils of different PP types determined by 1H NMR spectra.
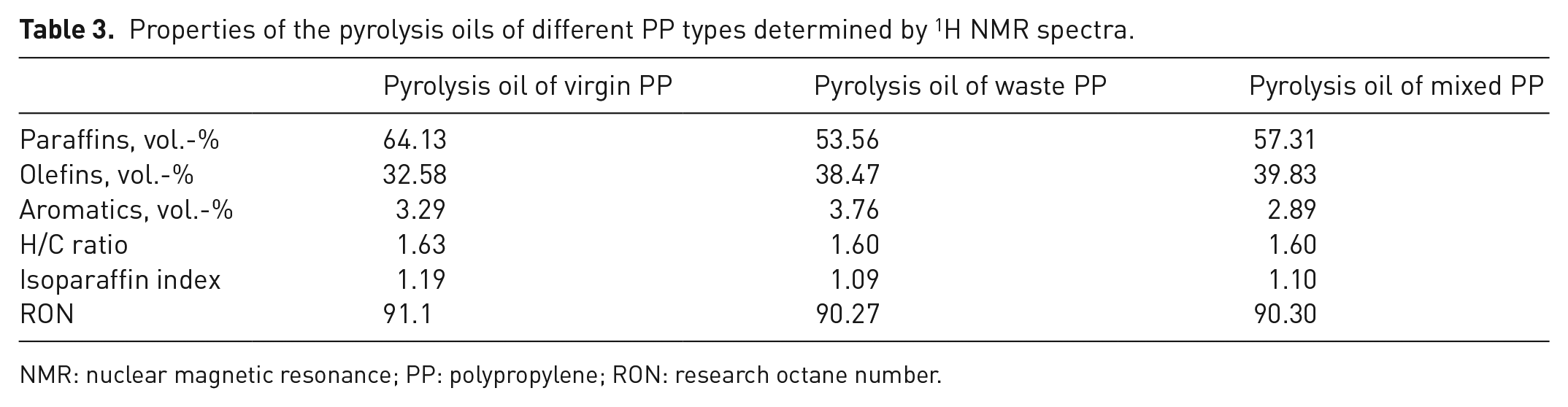
NMR: nuclear magnetic resonance; PP: polypropylene; RON: research octane number.
The pyrolysis oils mainly have a paraffin content, as was also found in the analysis of the spectrum based on the functionalities (Table 3). The aromatics content is about 3%, which is the lower limit for commercially available fuels (Hancsok et al., 2007).
However, the somewhat lower H/C ratios of 1.6 compared with gasoline and diesel fuels should lead to low greenhouse gas emissions according to Annamalai et al. (2018). The isoparaffin index is a measure of the degree of branching of the paraffins in the blends. A high isoparaffin index results in a high octane number of a fuel (Demirbas et al., 2015) and high oxidation stability (Hancsok et al., 2007). The isoparaffin indexes determined in this work are low compared with commercially available fuels (Hancsok et al., 2007). The RON of every pyrolysis oils is at the lower limit of gasoline fuels, but the oil should be further developed for use as a fuel.
Distillation fraction of pyrolysis oils
Fractional distillation has been used to produce fuel from oils. At least one of the fractions should have a higher oxidative stability. It was expected that the individual fractions would have better properties relevant to fuel specifications. The yields of the fractions for the gasoline range and the diesel range are shown in Figure 6.
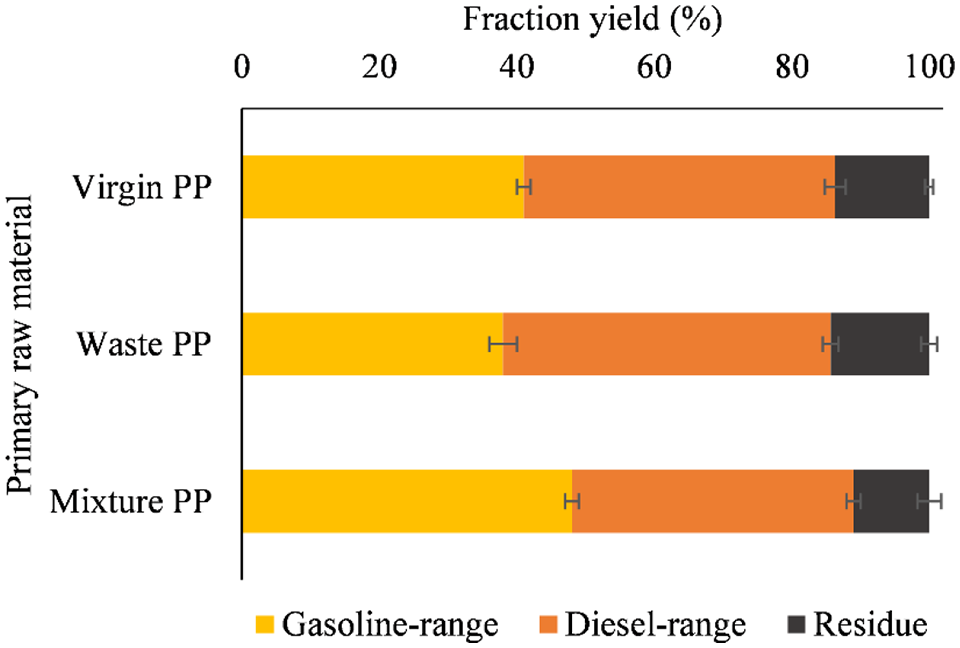
Yield of distillate fractions (%) from different PP pyrolysis oil feedstock.
It can be observed that different amounts of gasoline fractions are obtained with different PP feedstocks (p < 0.001). The lowest gasoline range fraction (38.0%) was obtained from waste PP and the highest (48.0%) from blended PP. The residue was 11.0% from mixed PP, 13.7% from virgin PP and 14.3% from waste PP, while the yields of light products were 89.0%, 86.3% and 85.7%, respectively, which is certainly a good result. The yields of the distillation fractions obtained showed that different amounts of fractions in the gasoline range (38.0–48.0%) and diesel range (41.0–47.7%) can be obtained from the different PP feedstocks in all cases and PP can be suitable for fuel production.
Properties of the gasoline fraction
The determined properties of the gasoline range fraction are shown in Table 4. The results of the investigation show that the density of the gasoline range fraction is not influenced by the PP raw material (p = 0.665). All densities meet the requirements for gasoline according to EN 228. In Kassargy et al. (2017), the density of the gasoline range fraction of PP was determined to be 710 g cm−1, but pyrolysis was then carried out at a higher temperature. The viscosity value determined was 0.631 mm2 s−1 when the raw material was blended PP, which was lower (p < 0.001) than when the raw material was virgin PP (1.113 mm2 s−1) and waste PP (1.112 mm2 s−1). This could be due to the easier thermal decomposition of the added liquid PP fraction. The calorific value of the gasoline fraction was 45.365 MJ kg−1 for the virgin PP feedstock, 45.610 MJ kg−1 for the waste PP feedstock and 45.325 MJ kg−1 for the blend PP feedstock. The calorific values met the requirements of EN 228 and the same trend as for the calorific value study was observed for oxidation stability, another property of the gasoline fraction: oxidation stability was the lowest for the blended PP (p < 0.001) and highest for the waste PP (9.6 minutes).
Physical properties of the gasoline range fraction (different letters indicate significant differences between PP feedstocks when p < 0.01) compared with EN 228.
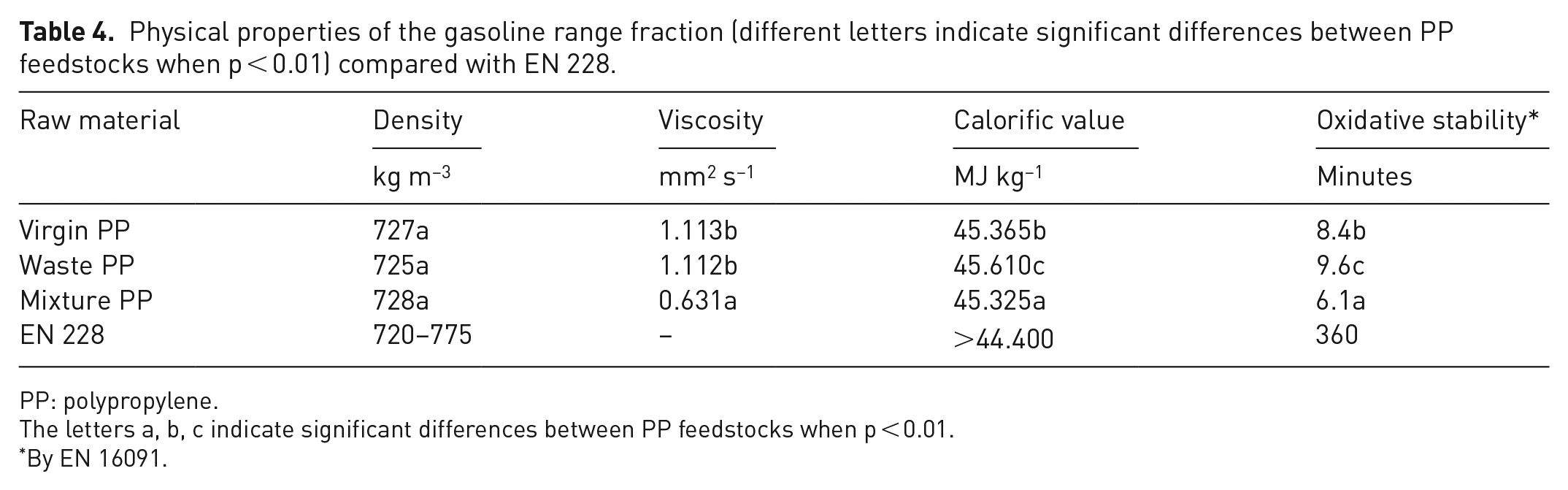
PP: polypropylene.
The letters a, b, c indicate significant differences between PP feedstocks when p < 0.01.
By EN 16091.
The oxidation stability of the gasoline fraction is even lower compared with the oxidation stability of the pyrolysis oils obtained from different PP feedstocks, but in terms of other physical parameters, the gasoline fraction from blended PP is the most consistent with the EN 228 standard. Therefore, it may be difficult to use the gasoline fraction for further processing as well.
Properties of the diesel range fraction
The results of the study showed (Table 5) that the PP raw material had an effect on the density value of the diesel range fraction; the mixed PP had the lowest density and the waste and virgin PP had the highest density (p < 0.001). The density values of the different PP primary raw materials were below EN 590. The viscosity value of the fraction from the diesel range of the blended PP (3.921 mm2 s−1) was higher (p < 0.001) than that of the virgin PP (2.311 mm2 s−1) or the waste PP (2.468 mm2 s−1). The viscosity of the diesel fraction from different feedstocks was in accordance with EN 590. The calorific value of the diesel fraction increased from virgin PP (46.368 MJ kg−1) to waste PP (46.481 MJ kg−1) to liquid phase blend PP (46.726 MJ kg−1). The values were above the minimum heating value specified in EN 590. The lowest cold filter clogging point was determined for the virgin PP raw material at −6.5°C, while that of the waste PP was −5.8°C. The highest flash point of the diesel fraction was 57.4°C for waste PP and the lowest 22.7°C for blend PP. The decrease in the flash point of mixture PP could be due to the formation of lighter components. However, the flash point of the diesel fraction of virgin PP and waste PP only corresponded to the value given in EN 590. The pour point of the diesel range fraction from virgin PP was −23.2°C and lower (p < 0.001) than that of the diesel range fraction from blend PP (−28.7°C), but higher than that of waste PP (−16.5°C).
Physical properties of the diesel fraction compared with EN 590.
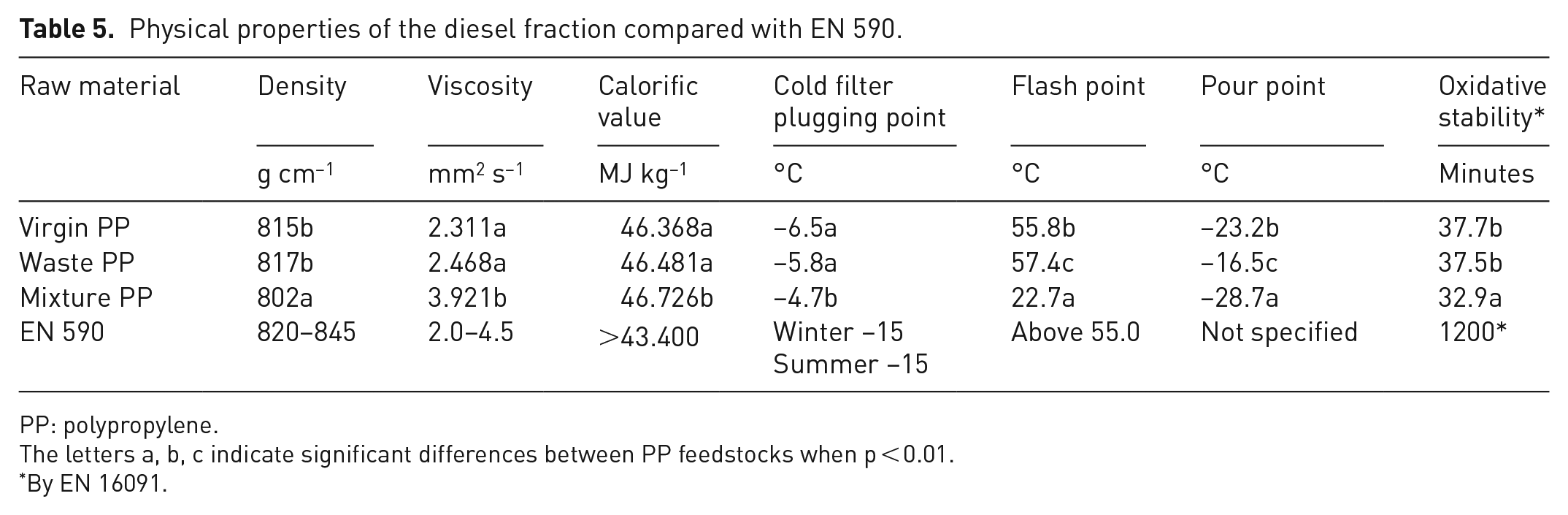
PP: polypropylene.
The letters a, b, c indicate significant differences between PP feedstocks when p < 0.01.
By EN 16091.
The oxidation stability of the diesel range fraction of blend PP (32.9 minutes) was lower (p < 0.001) than that of feedstock virgin (37.7 minutes) or waste PP (37.5 minutes). After atmospheric distillation, the oxidative stability of the diesel fraction increased about threefold compared with the corresponding pyrolysis oils.
In the end, neither virgin nor waste PP nor a product from the second pyrolysis of the pyrolysis oils from virgin and waste PP could be successfully converted into a directly usable fuel, which is in contrast with the literature (Ahmad et al., 2015; Seth and Sarkar, 2004; Wirawan and Farizal, 2019). One possible reason for the low oxidative stability of the resulting pyrolysis oils is mentioned in Hujuri et al. (2011), namely that abstraction is the preferred route for radical stabilisation. For this reason, the pyrolysis oils could be a good source of organic compounds or specific chemicals that can be used in the chemical industry or require additional processing. This is also supported in a similar way by Palmay-Paredes et al. (2021) and Le Gresley et al. (2019).
Conclusion
In this study, the performed slow non-catalytic low temperature pyrolysis of virgin polypropylene showed that a temperature of 350°C in the reactor is sufficient to degrade polypropylene to a liquid and this temperature allows to obtain 82 wt% of a liquid product.
The results of the study of oil properties showed that the use of oils in the production of alternative fuels can be limited mainly by low oxidative stability: 10.4–12.2 minutes. The 1H and 13C NMR spectral analysis showed that the stability of oils was determined by the presence of a high content of unsaturated double-bonded compounds. The obtained signal in the 13C NMR spectrum with a chemical shift of 144.6 ppm indicates the formation of α-alkenes with a quaternary second carbon atom in their structure, during thermal β-cleavage. The values for the H/C ratio and the isoparaffin index determined from the NMR spectra are too low compared with commercial fuels, which is probably due to the high-volume fraction of olefins compared with the share of paraffins. The RON of any pyrolysis oil is already at the lower limit of gasoline, but needs to be increased.
At temperatures of up to 190°C, the density and calorific values of the gasoline fractions from different PP feedstocks were consistent with the gasoline standard. However, the oxidation stability was further reduced to 6.1–9.6 minutes compared with the oxidation stability of the corresponding oils.
Viscosity, heating values and flash point of the diesel range fractions obtained from different PP feedstocks at 190–320°C (but not the PP blends) correspond to the diesel standard. However, the oxidation stability of the diesel range fraction of virgin, waste PP and blend PP and virgin PP pyrolysis oil increased to 32.9–37.37 minutes (2.7–3.6 times increased compared with the corresponding oil).
The fact that the distilled fractions appear to be identical in terms of NMR and other properties could confirm that there are no differences in the chemical processes involved in the pyrolysis of new and recycled PP. However, a recycled and then presumably aged PP may be oxidised and contain carbonyl groups. These carbonyl groups activate the alpha bonds. However, since the further mechanism is probably similar, a similar product is formed. However, this was not to be expected in advance, because different polymers can interact with each other even though their chemistry is different (see, for example, Wu et al., 2020).
In order to use the pyrolysis oil or the diesel and gasoline fractions from PP virgin, PP waste and PP blends (virgin PP and virgin PP pyrolysis oil) from this study for the production of alternative fuels, they should be treated to increase the oxidation stability and other physical properties.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
